Investigation into the Thermal Response and Pharmacological Activity of Substituted Schiff Bases on α-Amylase and α-Glucosidase
Abstract
1. Introduction
2. Experimental Details
2.1. General
2.2. Synthesis
2.3. Electronic Absorption Studies
2.4. Antioxidant Assays
2.4.1. DPPH Radical Scavenging Activity
2.4.2. ABTS Radical Cation Decolourisation Assay
2.4.3. H2O2-Scavenging Activity
2.4.4. Reducing Power Assay
2.5. Antimicrobial Studies
2.5.1. Preparation of Resazurin-Based Indicator and Solution of Schiff Bases
2.5.2. Test Microorganisms
2.5.3. Preparation of Standardised Inoculum
2.5.4. Antimicrobial Assay of Schiff Bases 1–9
2.6. In Vitro Antidiabetic Activity
2.6.1. α-Amylase Inhibitory Activity
2.6.2. α-Glucosidase Inhibitory Activity
3. Results
3.1. Synthesis and Spectra of Compound (1–9)
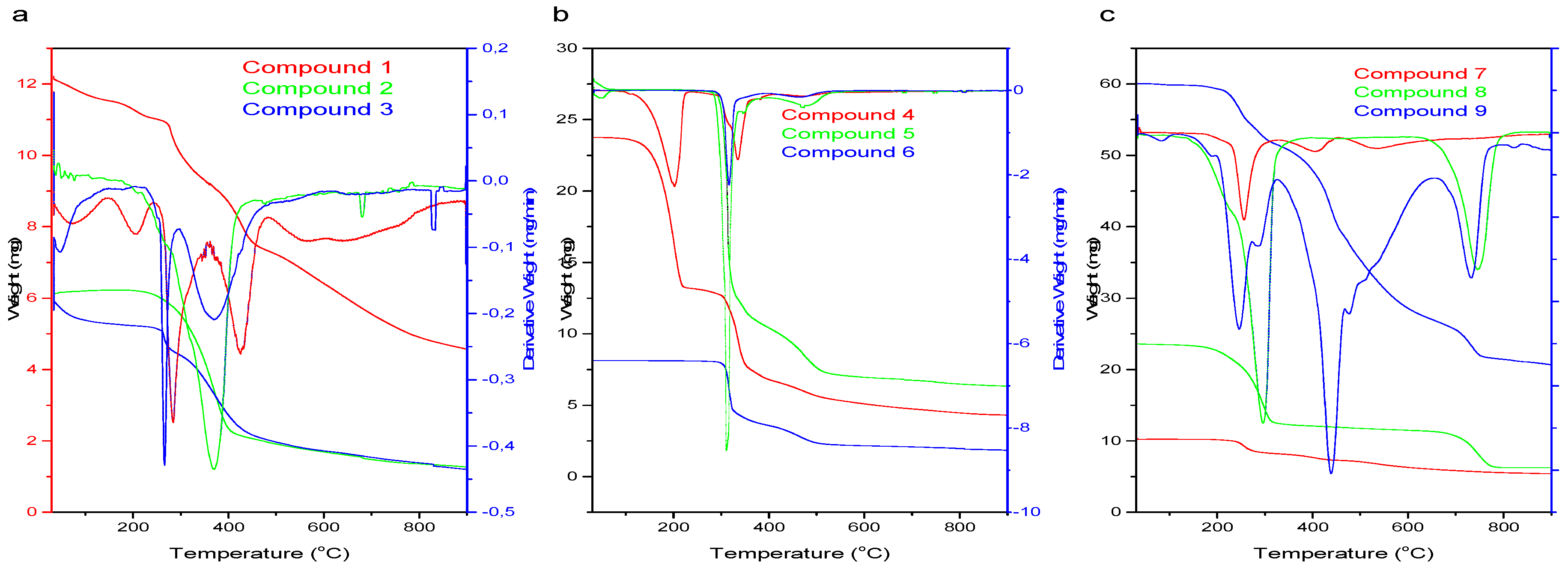
3.2. Thermal Profile of Schiff Bases (1–9)
3.3. X-ray Diffraction
3.4. 1H and 13C NMR Spectra of Schiff Bases 1–9
3.5. Antioxidant Activity of Schiff Bases 1–9
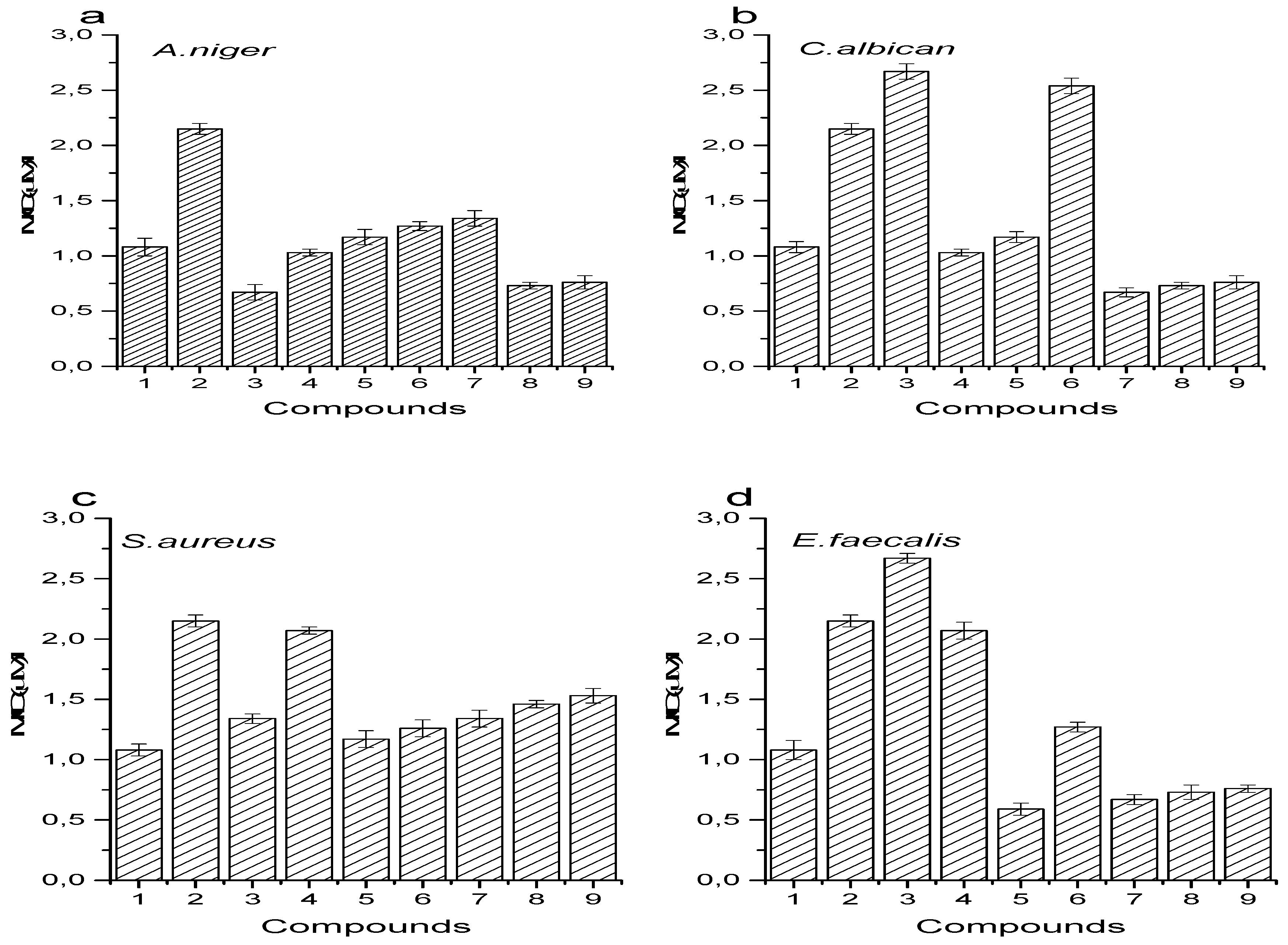
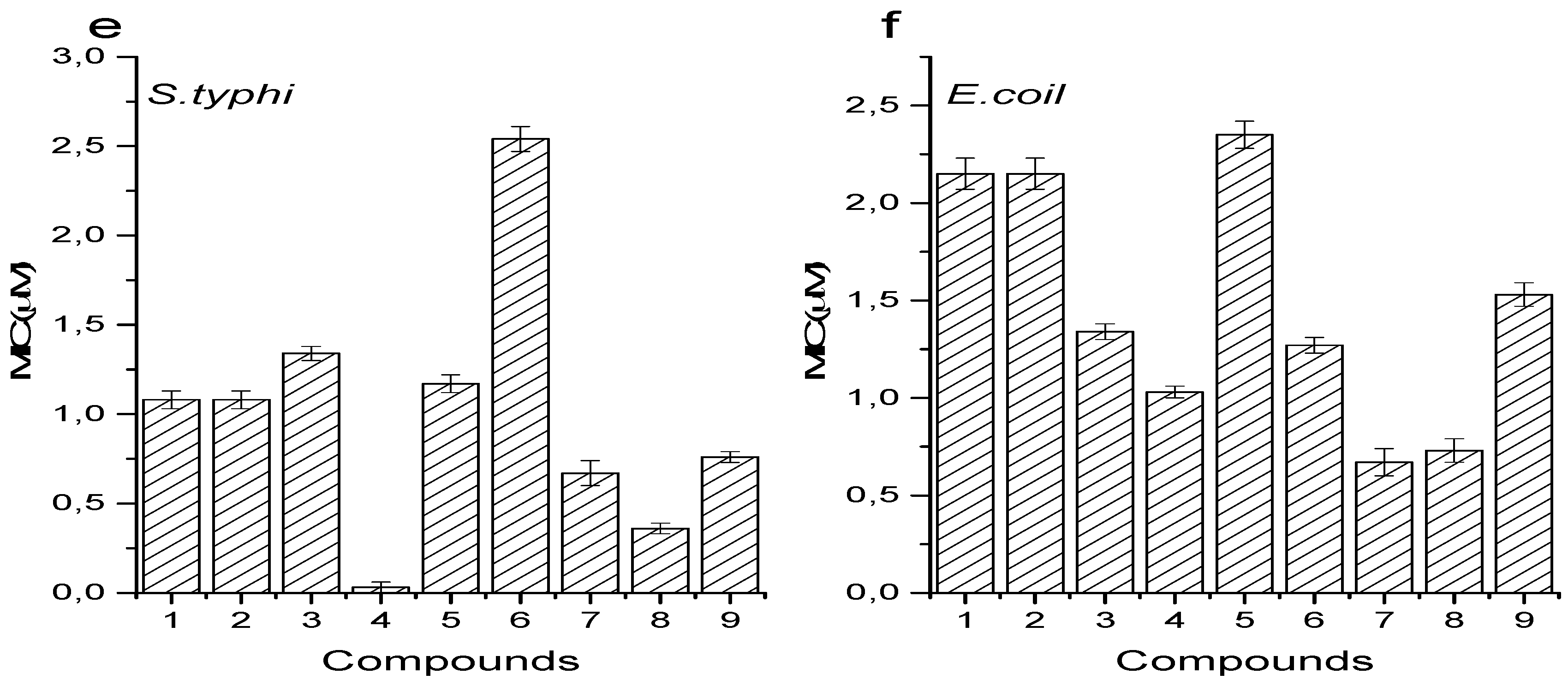
3.6. Antimicrobial Activity
3.7. In Vitro -Glucosidase and -Amylase Inhibitory Activities
4. Discussion
4.1. Synthesis and Spectra of Compound 1–9
4.2. Thermal Profile and Crystal Sizes of Schiff Bases (1–9)
4.3. 1H and 13C NMR Spectra of Schiff Bases 1–9
4.4. Antioxidant Activity of Schiff Bases 1–9
4.5. Antimicrobial Activity
4.6. In Vitro -Glucosidase and -Amylase Inhibitory Activities
5. Statistical Analysis
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Centers for Disease Control and Prevention. Diabetes—A Major Health Problem. Available online: https://www.cdc.gov/diabetes/ndep/pdfs/ppod-guide-diabetes-major-health-problem.pdf (accessed on 8 January 2018).
- Sunny, C.; Esther, N.O.; Andrew, E.U.; Anthonia, O.; Olorogun, S.K. The Clinical Practice Guidelines for Diabetes Management in Nigeria, 2nd ed.; Diabetes Association of Nigeria (DAN): Port Harcourt, Nigeria, 2013; Volume 2, ISBN 978-978-496-316-4. [Google Scholar]
- Soltani, A.; Pourian, M.; Davani, B.M. Correction to: Does this patient have Pheochromocytoma? A systematic review of clinical signs and symptoms. J. Diabetes Metab. Disord. 2017, 16, 42. [Google Scholar] [CrossRef] [PubMed]
- Krishan, P.; Singh, G.; Bedi, O. Carbohydrate restriction ameliorates nephropathy by reducing oxidative stress and upregulating HIF-1α levels in type-1 diabetic rats. J. Diabetes Metab. Disord. 2017, 16, 47. [Google Scholar] [CrossRef] [PubMed]
- Pratley, R.E. The Early Treatment of Type 2 Diabetes. Am. J. Med. 2013, 126, S2–S9. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, H. Microbiota associated with type 2 diabetes and its related complications. Food Sci. Hum. Wellness 2013, 2, 167–172. [Google Scholar] [CrossRef]
- McArdle, M.A.; Finucane, O.M.; Connaughton, R.M.; McMorrow, A.M.; Roche, H.M. Mechanisms of obesity-induced inflammation and insulin resistance: Insights into the emerging role of nutritional strategies. Front. Endocrinol. (Lausanne) 2013, 4, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Lee, J. Adipose tissue macrophages in the development of obesity-induced inflammation, insulin resistance, and type 2 Diabetes. Arch. Pharm. Res. 2013, 36, 208–222. [Google Scholar] [CrossRef] [PubMed]
- LeBleu, V.S.; MacDonald, B.; Kalluri, R. Structure and Function of Basement Membranes. Exp. Biol. Med. 2007, 232, 1121–1129. [Google Scholar] [CrossRef] [PubMed]
- Giacco, F.; Brownlee, M. Oxidative stress, and diabetic complications. Circ. Res. 2010, 107, 1058–1070. [Google Scholar] [CrossRef] [PubMed]
- Sabu, M.C.; Kuttan, R. Anti-diabetic activity of medicinal plants and its relationship with their antioxidant property. J. Ethnopharmacol. 2002, 81, 155–160. [Google Scholar] [CrossRef]
- Nalini, P.; Poonam, Y. Synthesis and Biological activities of some new Phthalides. Orient. J. Chem. 2012, 2, 57–61. [Google Scholar]
- Golcu, A.; Tumer, M.; Demirelli, H.; Wheatley, R.A. Cd(II) and Cu(II) complexes of polydentate Schiff base ligands: Synthesis, characterization, properties and biological activity. Inorg. Chim. Acta 2005, 358, 1785–1797. [Google Scholar] [CrossRef]
- El-Saied, F.A.; Salem, T.A.; Aly, S.A.; Shakdofa, M.E. Evaluation of Anti-Hyperglycemic Effect of Synthetic Schiff Base Vanadium(IV) Complexes. Pharm. Chem. J. 2017, 51, 833–842. [Google Scholar] [CrossRef]
- Yalcin, I.; Oren, I.; Sener, E.; Akin, A.U. Structure-activity, N. dines As, benzothiazoles and benzimidazoles Med., antimicrobial agents. Eur. J. Chem. 1992, 27, 401–406. [Google Scholar] [CrossRef]
- Thangadurai, T.D.; Natarajan, K. Tridentate Schiff base complexes of ruthenium(III) containing ONS/ONO donor atoms and their biocidal activities. Transit. Met. Chem. 2001, 26, 717–722. [Google Scholar] [CrossRef]
- Ali, S.S.; Kasoju, N.; Luthra, A.; Singh, A.; Sharanabasava, H.; Sahu, A.; Bora, U. Indian medicinal herbs as sources of antioxidants Indian medicinal herbs as sources of antioxidants. Food Res. Int. 2008, 41, 1–15. [Google Scholar] [CrossRef]
- Burčul, F.; Generalić Mekinić, I.; Radan, M.; Rollin, P.; Blažević, I. Isothiocyanates: Cholinesterase inhibiting, antioxidant, and anti-inflammatory activity. J. Enzyme Inhib. Med. Chem. 2018, 33, 577–582. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.X.; Liu, Y.; Cia, L.H.; Hu, Y.J.; Yin, J.; Hu, P.Z. Inhibitory study of some novel Schiff base derivatives on Staphylococcus aureus by microcalorimetry. Thermochim. Acta 2006, 440, 51–56. [Google Scholar] [CrossRef]
- Mezeiova, E.; Spilovska, K.; Nepovimova, E.; Gorecki, L.; Soukup, O.; Dolezal, R.; Malinak, D.; Janockova, J.; Jun, D.; Kuca, K.; et al. Profiling donepezil template into multipotent hybrids with antioxidant properties. J. Enzyme Inhib. Med. Chem. 2018, 33, 583–606. [Google Scholar] [CrossRef] [PubMed]
- Moffett, R.B. Organic Syntheses; Rabjohn, N., Ed.; John Wiley & Sons, Inc.: New York, NY, USA, 1975; Volume 59, ISBN 3-540-07440-6. [Google Scholar]
- Tran, H.V.; Tran, L.D.; Cham, T.V.; Hoang, D.N.; Thinh, N.P.; Dien, G.N.; Phuc, X. Synthesis, characterization, antibacterial and antiproliferative activities of monodisperse chitosan-based silver nanoparticles. Colloids Surf. A 2010, 360, 32–40. [Google Scholar] [CrossRef]
- Blois, M.S. Antioxidant determinations by the use of a stable free radical [10]. Nature 1958, 181, 1199–1200. [Google Scholar] [CrossRef]
- Wolfenden, B.S.; Willson, R.L. Radical-cations as reference chromogens in kinetic studies of one-electron transfer reactions: Pulse radiolysis studies of 2,2′-azinobis-(3-ethylbenzthiazoline-6-sulphonate). J. Chem. Soc. Perkin Trans. 2 1982, 2, 805–812. [Google Scholar] [CrossRef]
- Ruch, R.J.; Cheng, S.-J.; Klaunig, J.E. Prevention of cytotoxicity and inhibition of intercellular communication by antioxidant catechins isolated from Chinese green tea. Carcinogenesis 1989, 10, 1003–1008. [Google Scholar] [CrossRef] [PubMed]
- Oyaizu, M. Antioxidative activities of browning products of glucosamine fractionated by organic solvent and thin layer chromatography. Nippon Shokuhin Kogyo Gakkaishi 1988, 35, 771–775. [Google Scholar] [CrossRef]
- Clinical and Laboratory Standards Institute. M100: Performance Standards for Antimicrobial Susceptibility Testing, 28 Edition. Available online: https://clsi.org/standards/products/microbiology/documents/m100/ (accessed on 11 January 2018).
- De Rapper, S.; Kamatou, G.; Viljoen, A.; Van Vuuren, S. The in vitro antimicrobial activity of Lavandula angustifolia essential oil in combination with other aroma-therapeutic oils. Evid.-Based Complement. Altern. Med. 2013, 2013, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Akhalwaya, S.; van Vuuren, S.; Patel, M. An in vitro investigation of indigenous South African medicinal plants used to treat oral infections. J. Ethnopharmacol. 2018, 210, 359–371. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.; Kai, G.; Ni, X.; Yang, F.; Chen, X. Interaction of natural polyphenols with α-amylase in vitro: Molecular property–affinity relationship aspect. Mol. Biosyst. 2011, 7, 1883. [Google Scholar] [CrossRef] [PubMed]
- Ranilla, L.G.; Kwon, Y.I.; Apostolidis, E.; Shetty, K. Phenolic compounds, antioxidant activity and in vitro inhibitory potential against key enzymes relevant for hyperglycemia and hypertension of commonly used medicinal plants, herbs and spices in Latin America. Bioresour. Technol. 2010, 101, 4676–4689. [Google Scholar] [CrossRef] [PubMed]
- Wawer, V.; Koleva, T.; Dudev, I. 1H and 13C NMR study and AM1 calculations of some azobenzenes and N-benzylideneanilines: Effect of substituents on the molecular planarity. J. Mol. Struct. 1997, 412, 153–159. [Google Scholar]
- Fan, C.; Chen, J.; Chen, Y.; Ji, J.; Teng, H.H. Relationship between solubility and solubility product: The roles of crystal sizes and crystallographic directions. Geochim. Cosmochim. Acta 2006, 70, 3820–3829. [Google Scholar] [CrossRef]
- Black, S.; Muller, F. On the effect of temperature on aqueous solubility of organic solids. Org. Process. Res. Dev. 2010, 14, 661–665. [Google Scholar] [CrossRef]
- Bowman, B.T.; Sans, W.W. Effect of temperature on the water solubility of insecticides. J. Environ. Sci. Health Part B 1985, 20, 625–631. [Google Scholar] [CrossRef]
- Abramowitz, R.; Yalkowsky, S.H. Melting point, boiling point, and symmetry. Pharm. Res. 1990, 7, 942–947. [Google Scholar] [CrossRef] [PubMed]
- Ohira, T.; Yamamoto, O. Correlation between antibacterial activity and crystallite size on ceramics. Chem. Eng. Sci. 2012, 68, 355–361. [Google Scholar] [CrossRef]
- Phonsri, W.; Macedo, D.S.; Davies, C.G.; Jameson, G.N.L.; Moubaraki, B.; Murray, K.S. Heteroleptic iron(iii) Schiff base spin crossover complexes: Halogen substitution, solvent loss and crystallite size effects. Dalt. Trans. 2017, 46, 7020–7029. [Google Scholar] [CrossRef] [PubMed]
- Neelofar, N.A.; Khan, A.; Amir, S.; Khan, N.A.; Muhammad, B. Synthesis of Schiff bases derived from 2-hydroxy-1-naphth-aldehyde and their tin(II) complexes for antimicrobial and antioxidant activities. Bull. Chem. Soc. Ethiop. 2018, 31, 445–456. [Google Scholar] [CrossRef]
- Iftikhar, B.; Javed, K.; Khan, M.S.U.; Akhter, Z.; Mirza, B.; Mckee, V. Synthesis, characterization and biological assay of Salicylaldehyde Schiff base Cu(II) complexes and their precursors. J. Mol. Struct. 2018, 1155, 337–348. [Google Scholar] [CrossRef]
- Amarowicz, R.; Pegg, R.; Rahimi, P.; Barl, B.; Weil, J. Free radical scavenging capacity and antioxidant activity of selected plant species from the Canadian prairies. Food Chem. 2004, 84, 551–562. [Google Scholar] [CrossRef]
- Shimada, K.; Fujikawa, K.; Yahara, K.; Nakamura, T. Soybean, Antioxidative properties of xanthan on the autoxidation of Chemistry, oil in cyclodextrin emulsion. J. Agric. Food 1992, 40, 945–948. [Google Scholar] [CrossRef]
- Xiao, J.; Ni, X.; Kai, G.; Chen, X. A Review on Structure–Activity Relationship of Dietary Polyphenols Inhibiting α-Amylase. Crit. Rev. Food Sci. Nutr. 2013, 53, 497–506. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, K.; Milosavic, N.; Popovic, M.M.; Prodanovic, R.; Knezevic, Z.D.; Jankov, R. Preparation and studies on immobilized α-glucosidase from baker’s yeast Saccharomyces cerevisiae. J. Serb. Chem. Soc. 2007, 72, 1255–1263. [Google Scholar] [CrossRef]


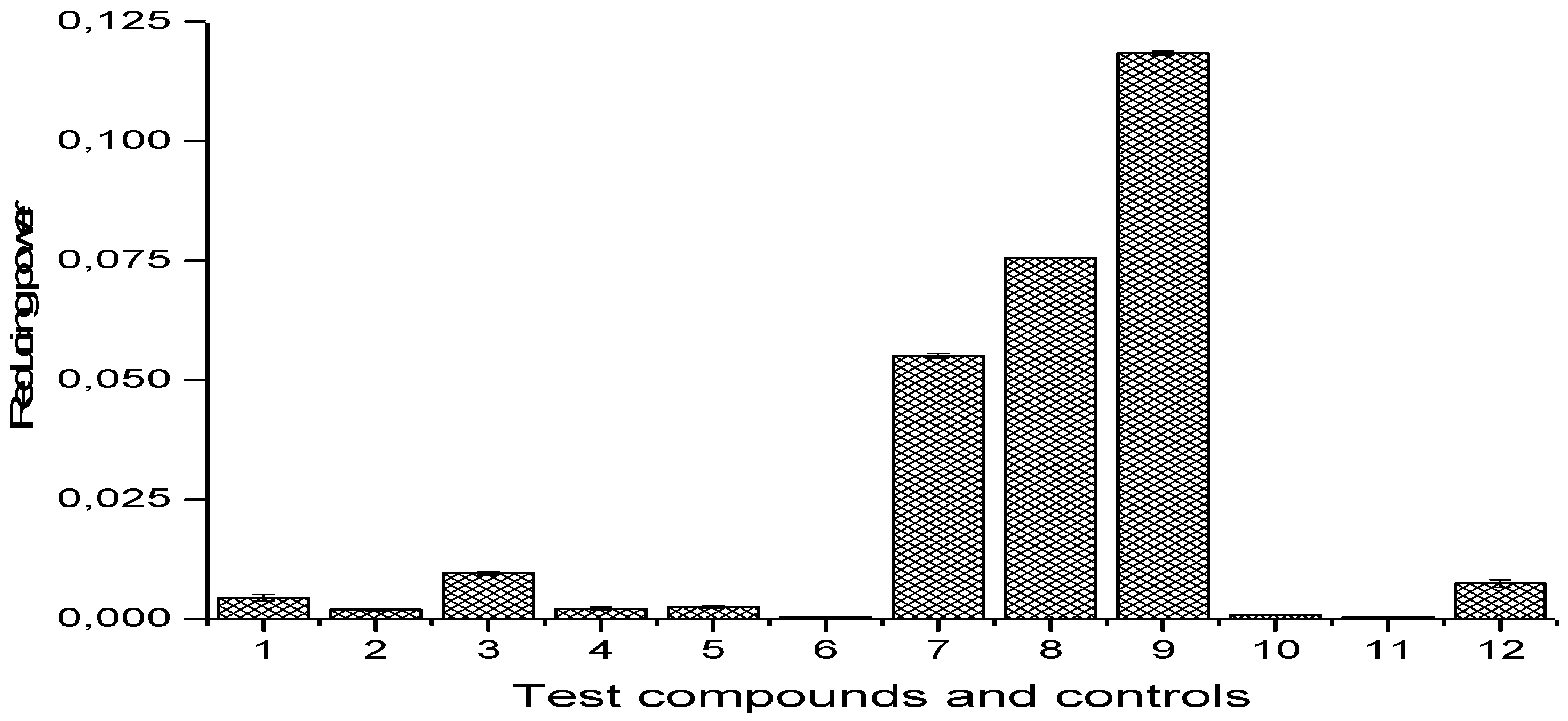
| Schiff Bases | M.F | M.pt (°C) | Yield % | Nature | ||
|---|---|---|---|---|---|---|
| 1 | 232.07 | 285 | 61.8 | Regatta | 393 | |
| 2 | 232.06 | 370 | 77.1 | Mandarin | 470 | |
| 3 | 187.07 | 266 | 63.8 | Peach Puff | 321 | 0.8236 |
| 4 | 242.07 | 202 | 27.3 | Silver | 435 | 2.5340 |
| 5 | 213.08 | 305 | 95.6 | Light gray | 498 | |
| 6 | 197.08 | 302 | 39.7 | Grey | 472 | 2.8978 |
| 7 | 372.04 | 200 | 57.3 | Orange | 455 | 5.3109 |
| 8 | 343.05 | 296 | 93.7 | Dirty green | 397 | 2.6220 |
| 9 | 327.06 | 246 | 73.1 | Chocolate | 394 | 2.3770 |
| Schiff Bases | FT-IR (cm−1) | ||||
|---|---|---|---|---|---|
| 1 | 3352 | 1698 | 1508 | 1529 | 3590 |
| 2 | 3450 | 1698 | - | 1526 | 3338 |
| 3 | 3450 | 1690 | - | 1575 | 3660 |
| 4 | 3219 | 1660 | 1347 | 1521 | - |
| 5 | 3232 | 1655 | - | 1432 | - |
| 6 | 3236 | 1653 | - | 1533 | - |
| 7 | 3437 | 1689 | 1502 | 1575 | - |
| 8 | 3337 | 1613 | - | 1514 | - |
| 9 | 3567 | 1614 | - | 1507 | - |
| Schiff Bases | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 |
|---|---|---|---|---|---|---|---|---|---|
| Crystallite size (nm) | 13.01 | 10.33 | 10.25 | 11.11 | 6.19 | 6.67 | 13.13 | 18.55 | 22.62 |
| % change in crystallite size | −26.93 | −0.78 | − | −66.57 | 8.00 | − | 41.95 | 17.99 | − |
| Antioxidant Assay | |||
|---|---|---|---|
| DPPH | ABTS | H2O2-Scavenging | |
| Compounds | IC50 (µM) | IC50 (µM) | IC50 (µM) |
| 1 | 290 ± 9 | 410 ± 6 | 350 ± 5 |
| 2 | 390 ± 5 | 510 ± 5 | 410 ± 6 |
| 3 | 360 ± 5 | 450 ± 7 | 410 ± 8 |
| 4 | 290 ± 6 | 370 ± 6 | 370 ± 7 |
| 5 | 260 ± 5 | 110 ± 5 | 320 ± 6 |
| 6 | 380 ± 2 | 260 ± 6 | 380 ± 5 |
| 7 | 200 ± 0 | 50 ± 5 | 160 ± 90 |
| 8 | 210 ± 5 | 140 ± 5 | 240 ± 17 |
| 9 | 220 ± 6 | 50 ± 4 | 190 ± 11 |
| 10 | 420 ± 3 | 580 ± 7 | 460 ± 11 |
| 11 | 350 ± 5 | 530 ± 6 | 440 ± 7 |
| 12 | 240 ± 3 | 270 ± 8 | 270 ± 10 |
| Compounds | MBC (µM) | MFC (µM) | ||||
|---|---|---|---|---|---|---|
| S. aureus | E. faecalis | E. coli | S. typhi | A. niger | C. albicans | |
| 1 | 4.31 | 4.31 | 4.31 | 2.15 | >4.31 * | >4.31 * |
| 2 | 4.31 | >4.31 * | 4.31 | >4.31 * | >4.31 * | >4.31 * |
| 3 | >5.35 * | >5.35 * | 2.67 | >5.35 * | >5.35 * | >5.35 * |
| 4 | 4.31 | >4.31 * | 2.07 | >4.31 * | >4.31 * | >4.31 * |
| 5 | >4.69 * | 4.69 | 4.69 | >4.69 * | >4.69 * | >4.69 * |
| 6 | >5.07 * | >5.07 * | 2.54 | >5.07 * | >5.07 * | >5.07 * |
| 7 | >2.69 * | >2.69 * | 1.34 | >2.69 * | 2.69 | 1.53 |
| 8 | >2.92 * | >2.92 * | 1.46 | >2.92 * | 1.46 | 1.46 |
| 9 | >3.06 * | >3.06 * | 3.06 | >3.06 * | 1.53 | 1.53 |
| Schiff Bases | -Glucosidase | -Amylase |
|---|---|---|
| 1 | 5.40± 0.44 | 4.20 ± 0.36 |
| 2 | 1.20 ± 0.51 | 0.91 ± 0.09 |
| 3 | 5.60 ± 0.82 | 2.00 ± 0.74 |
| 4 | 13.94 ± 0.91 | 13.77 ± 0.17 |
| 5 | 13.83 ± 12.5 | 13.83 ± 0.84 |
| 6 | 14.18 ± 17.3 | 14.14 ± 0.17 |
| 7 | 9.61 ± 0.84 | 7.30 ± 0.16 |
| 8 | 5.00 ± 0.82 | 4.20 ± 0.34 |
| 9 | 6.60 ± 0.97 | 5.61 ± 0.42 |
| Acarbose | 8.33 ± 7.20 | 6.80 ± 1.25 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Okoli, B.J.; Modise, J.S. Investigation into the Thermal Response and Pharmacological Activity of Substituted Schiff Bases on α-Amylase and α-Glucosidase. Antioxidants 2018, 7, 113. https://doi.org/10.3390/antiox7090113
Okoli BJ, Modise JS. Investigation into the Thermal Response and Pharmacological Activity of Substituted Schiff Bases on α-Amylase and α-Glucosidase. Antioxidants. 2018; 7(9):113. https://doi.org/10.3390/antiox7090113
Chicago/Turabian StyleOkoli, Bamidele Joseph, and Johannes Sekomeng Modise. 2018. "Investigation into the Thermal Response and Pharmacological Activity of Substituted Schiff Bases on α-Amylase and α-Glucosidase" Antioxidants 7, no. 9: 113. https://doi.org/10.3390/antiox7090113
APA StyleOkoli, B. J., & Modise, J. S. (2018). Investigation into the Thermal Response and Pharmacological Activity of Substituted Schiff Bases on α-Amylase and α-Glucosidase. Antioxidants, 7(9), 113. https://doi.org/10.3390/antiox7090113






