Extraction of Antioxidant Phenolic Compounds from Brewer’s Spent Grain: Optimization and Kinetics Modeling
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Optimization of Polyphenols Extraction in Batch System
2.2.2. Ultrasound and Microwave Assisted Extraction Methods
2.2.3. Total Phenolic Content (TPC) and Antioxidant Capacity Determination
2.2.4. Modeling of Kinetics Profile and Statistical Analysis
3. Results and Discussion
3.1. Extraction Optimization in Batch System
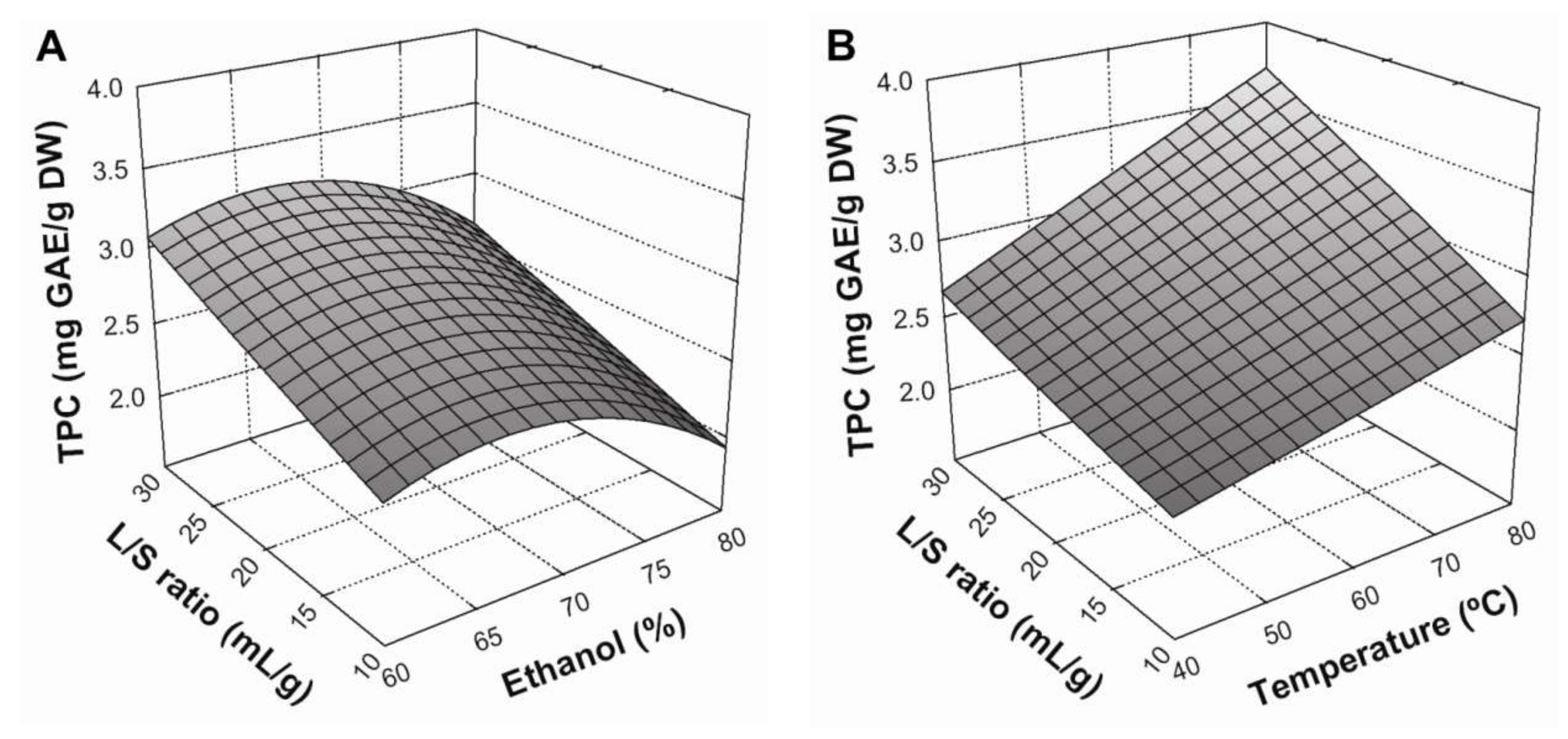
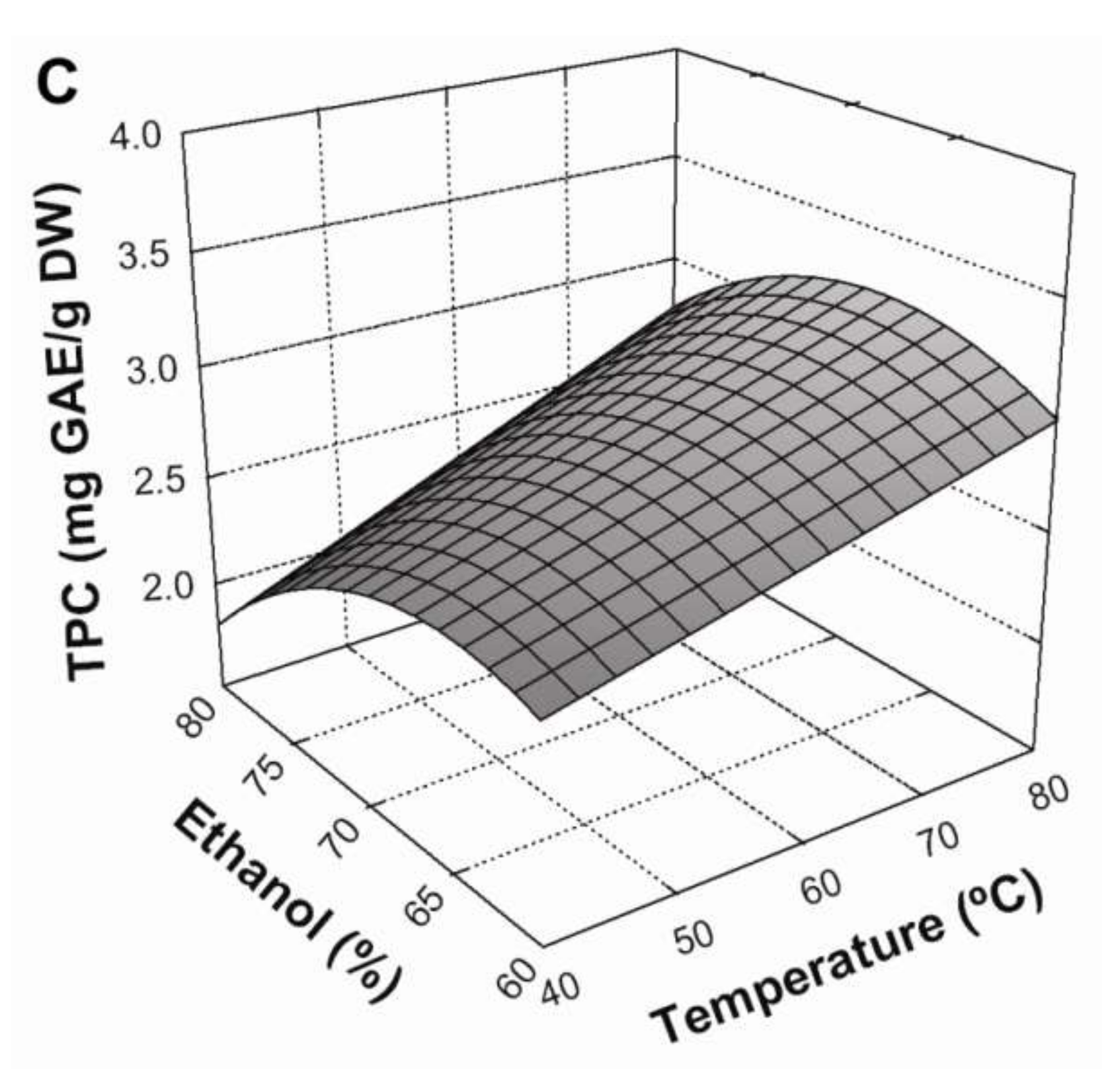
3.2. Effect of Extraction Conditions on TPC Yield
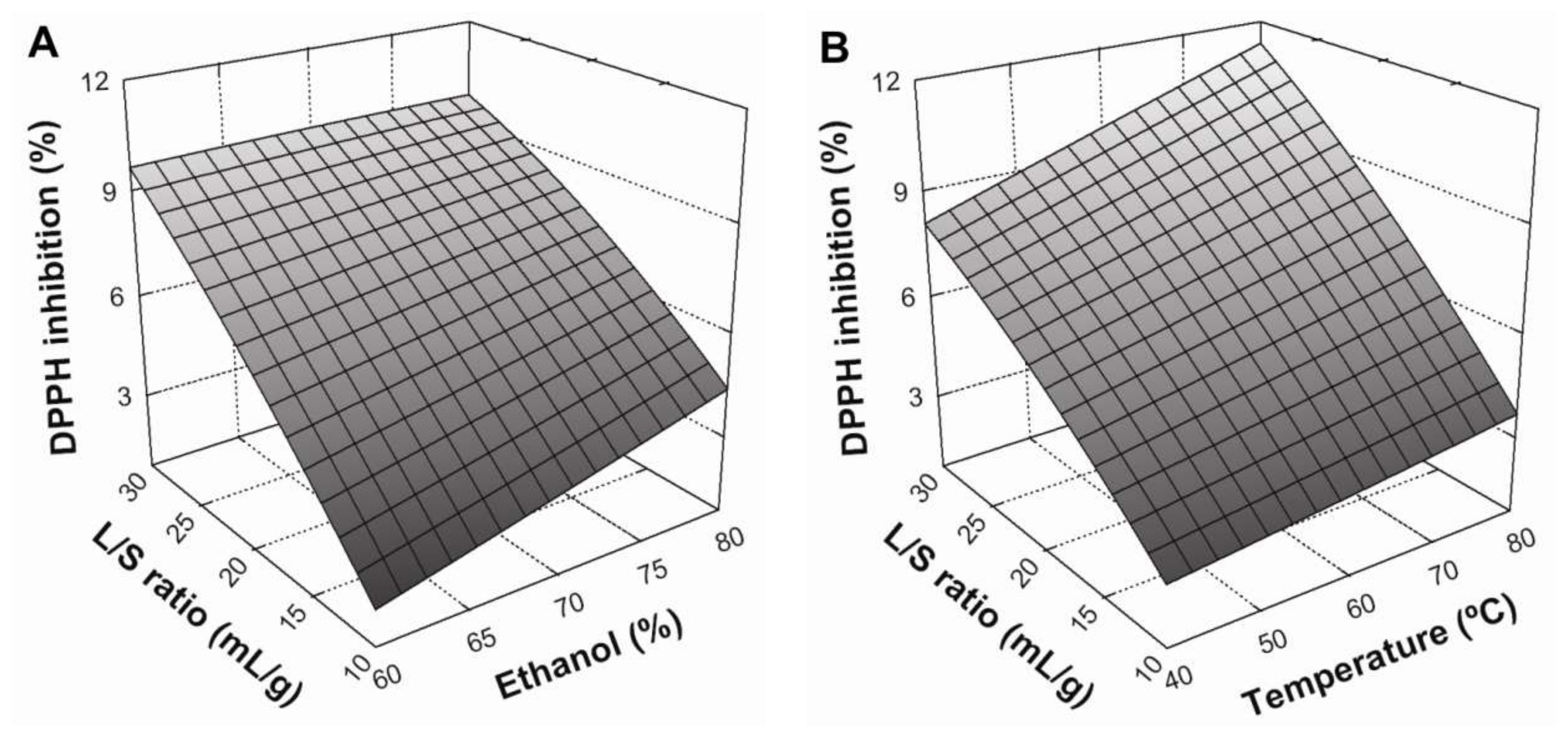
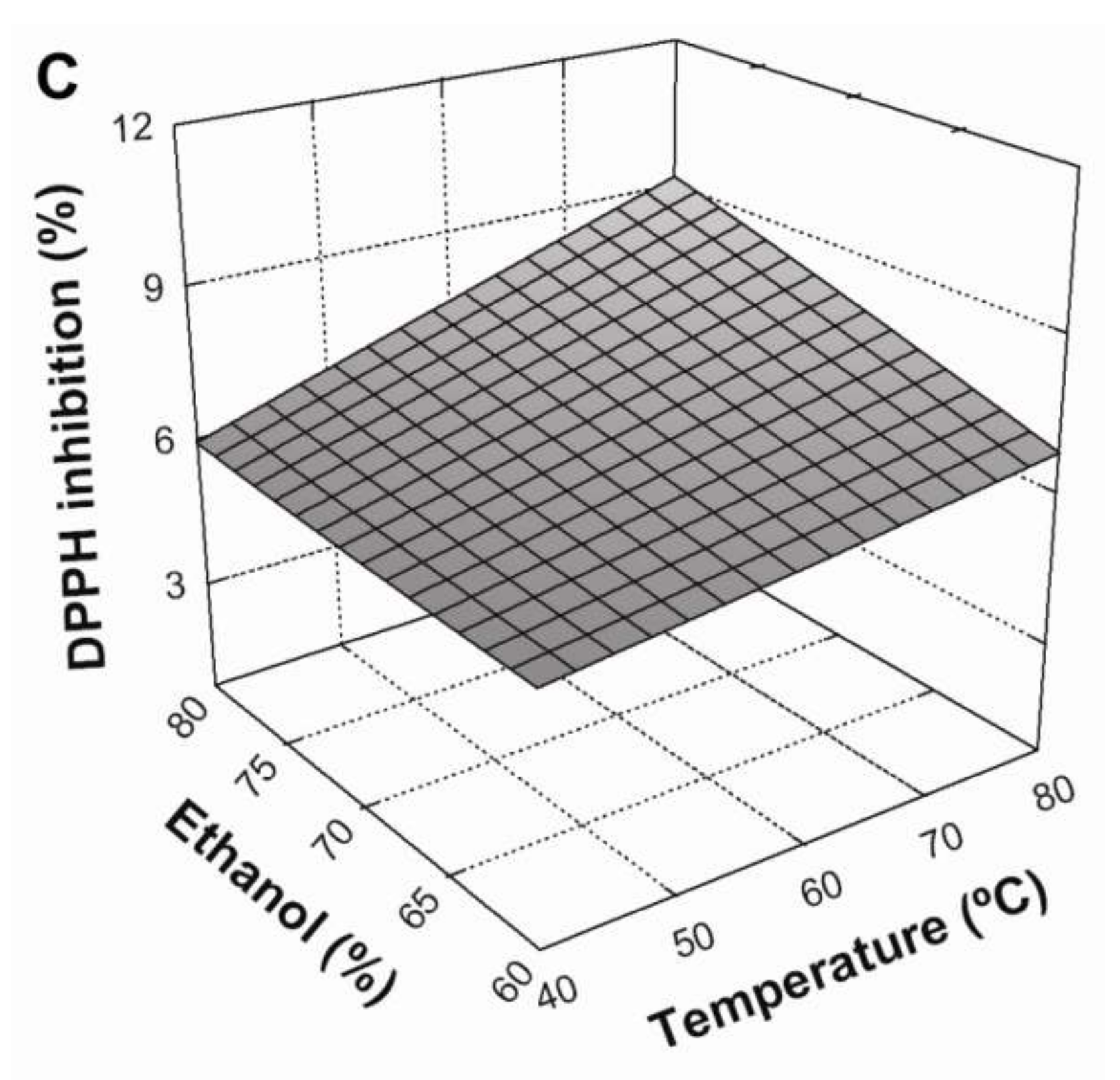
3.3. Effect of Extraction Conditions on DPPH Radical Scavenging
3.4. Optimization of the Extraction Conditions
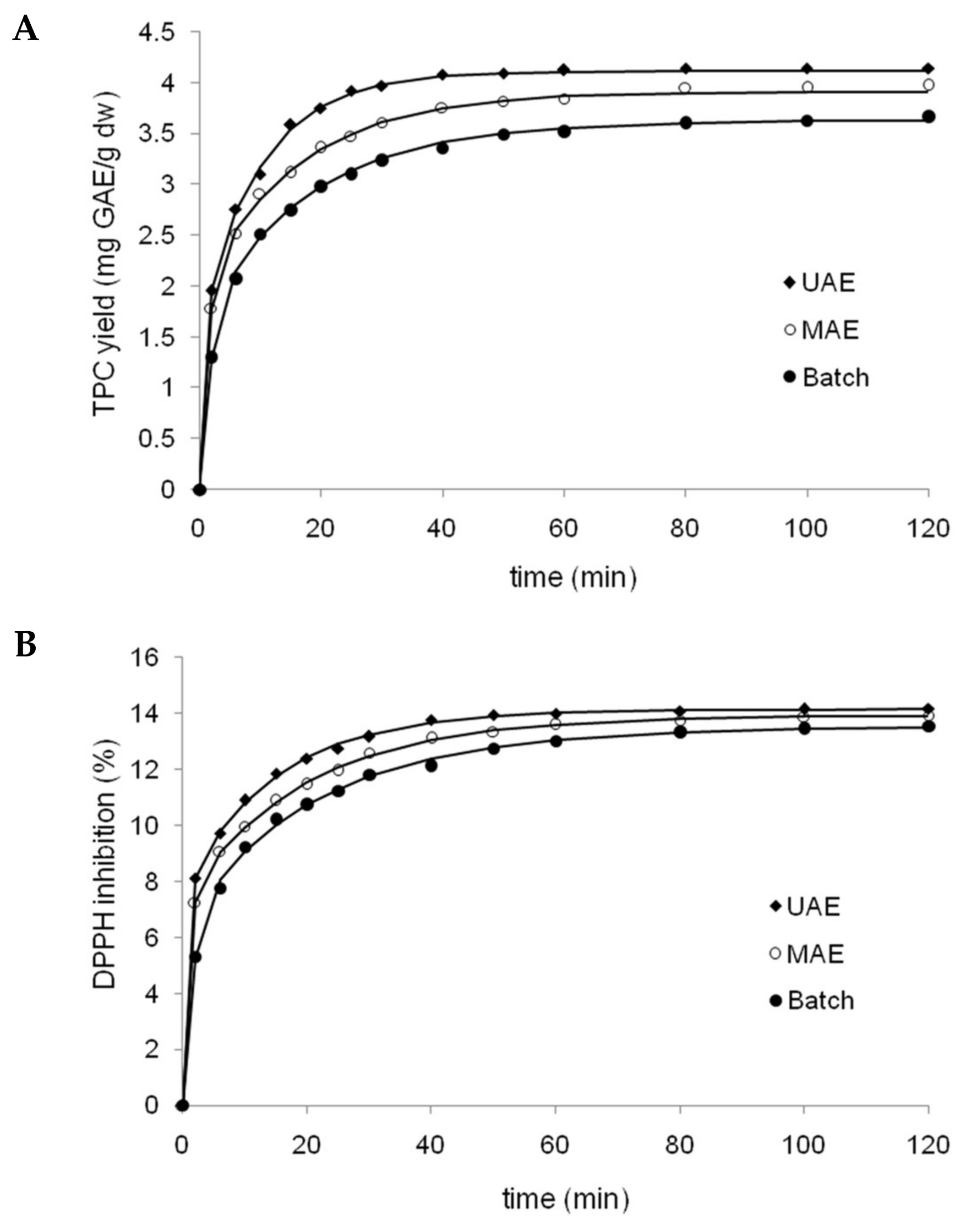
3.5. Extraction Kinetics Study
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Moreira, M.M.; Morais, S.; Carvalho, D.O.; Barros, A.A.; Delerue-Matos, C.; Guido, L.F. Brewer’s spent grain from different types of malt: Evaluation of the antioxidant activity and identification of the major phenolic compounds. Food Res. Int. 2013, 54, 382–388. [Google Scholar] [CrossRef]
- Mussato, S.I. Biotechnological potential of brewing industry by-products. In Biotechnology for Agro-Industrial Residues Utilisation, 1st ed.; Singh-Nee Nigam, P., Pandey, A., Eds.; Springer: Dordrecht, The Netherlands, 2009; pp. 313–326. ISBN 978-1-4020-9941-0. [Google Scholar]
- Guo, M.; Du, J.; Zhang, Z.; Zhang, K.; Jin, Y. Optimization of brewer’s spent grain-enriched biscuits processing formula. J. Food Process Eng. 2014, 37, 122–130. [Google Scholar] [CrossRef]
- Liyana-Pathirana, C.; Shahidi, F. Optimization of extraction of phenolic compounds from wheat using response surface methodology. Food Chem. 2005, 93, 47–56. [Google Scholar] [CrossRef]
- Chan, C.-H.; Yusoff, R.; Ngoh, G.-C. Modeling and kinetics study of conventional and assisted batch solvent extraction. Chem. Eng. Res. Des. 2014, 92, 1169–1186. [Google Scholar] [CrossRef]
- Xi, J.; He, L.; Yan, L. Kinetic modeling of pressure-assisted solvent extraction of polyphenols from green tea in comparison with the conventional extraction. Food Chem. 2015, 166, 287–291. [Google Scholar] [CrossRef] [PubMed]
- Tušek, A.J.; Benković, M.; Cvitanović, A.B.; Valinger, D.; Jurina, T.; Kljusurić, J.G. Kinetics and thermodynamics of the solid-liquid extraction process of total polyphenols, antioxidants and extraction yield from Asteraceae plants. Ind. Crops Prod. 2016, 91, 205–214. [Google Scholar] [CrossRef]
- Carciochi, R.A.; Manrique, G.D.; Dimitrov, K. Optimization of antioxidant phenolic compounds extraction from quinoa (Chenopodium quinoa) seeds. J. Food Sci. Technol. 2015, 52, 4396–4404. [Google Scholar] [CrossRef] [PubMed]
- Carciochi, R.A.; D’Alessandro, L.G.; Manrique, G.D. Effect of roasting conditions on the antioxidant compounds of quinoa seeds. Int. J. Food Sci. Technol. 2016, 51, 1018–1025. [Google Scholar] [CrossRef]
- Meneses, N.G.T.; Martins, S.; Teixeira, J.A.; Mussatto, S.I. Influence of extraction solvents on the recovery of antioxidant phenolic compounds from brewer’s spent grains. Sep. Purif. Technol. 2013, 108, 152–158. [Google Scholar] [CrossRef]
- McCarthy, A.L.; O’Callaghan, Y.C.; Neugart, S.; Piggott, C.O.; Connolly, A.; Jansen, M.A.K.; Krumbein, A.; Schreiner, M.; Fitzgerald, R.J.; O’Brien, N.M. The hydroxycinnamic acid content of barley and brewers’ spent grain (BSG) and the potential to incorporate phenolic extracts of BSG as antioxidants into fruit beverages. Food Chem. 2013, 141, 2567–2574. [Google Scholar] [CrossRef] [PubMed]
- D’Alessandro, L.G.; Kriaa, K.; Nikov, I.; Dimitrov, K. Ultrasound assisted extraction of polyphenols from black chokeberry. Sep. Purif. Technol. 2012, 93, 42–47. [Google Scholar] [CrossRef]
- Patricelli, A.; Assogna, A.; Casalaina, A.; Emmi, E.; Sodini, G. Fattori che influenzano l’estrazione dei lipidi da semi decorticati di girasole. Riv. Ital. Sostanze Grasse 1979, 56, 136–142. [Google Scholar]
- Mustapa, A.N.; Martin, A.; Gallego, J.R.; Mato, R.B.; Cocero, M.J. Microwave-assisted extraction of polyphenols from Clinacanthus nutans Lindau medicinal plant: Energy perspective and kinetics modeling. Chem. Eng. Process. 2015, 97, 66–74. [Google Scholar] [CrossRef]
- Chemat, F.; Huma, Z.; Khan, M.K. Applications of ultrasound in food technology: Processing, preservation and extraction. Ultrason. Sonochem. 2011, 18, 813–835. [Google Scholar] [CrossRef] [PubMed]
- Veggi, P.C.; Martinez, J.; Meireles, M.A.A. Fundamentals of microwave extraction. In Microwave-Assisted Extraction for Bioactive Compounds: Theory and Practice, 1st ed.; Chemat, F., Cravotto, G., Eds.; Springer: New York, NY, USA, 2013; pp. 15–52. ISBN 978-1-4899-7361-0. [Google Scholar]
| Run | Temperature (°C; X1) | Ethanol Concentration (%; v/v; X2) | Liquid/Solid Ratio (mL/g; X3) | TPC (mg GAE/g DW) | DPPH Inhibition (%) |
|---|---|---|---|---|---|
| 1 | 40 (−1) | 60 (−1) | 10:1 (−1) | 2.15 | 1.86 |
| 2 | 80 (+1) | 60 (−1) | 10:1 (−1) | 2.33 | 2.23 |
| 3 | 40 (−1) | 80 (+1) | 10:1 (−1) | 1.59 | 3.17 |
| 4 | 80 (+1) | 80 (+1) | 10:1 (−1) | 2.34 | 5.68 |
| 5 | 40 (−1) | 60 (−1) | 30:1 (+1) | 2.67 | 8.40 |
| 6 | 80 (+1) | 60 (−1) | 30:1 (+1) | 3.57 | 10.99 |
| 7 | 40 (−1) | 80 (+1) | 30:1 (+1) | 2.02 | 7.40 |
| 8 | 80 (+1) | 80 (+1) | 30:1 (+1) | 3.16 | 11.93 |
| 9 | 40 (−1) | 70 (0) | 20:1 (0) | 2.41 | 6.50 |
| 10 | 80 (+1) | 70 (0) | 20:1 (0) | 3.19 | 7.59 |
| 11 | 60 (0) | 60 (−1) | 20:1 (0) | 2.85 | 5.70 |
| 12 | 60 (0) | 80 (+1) | 20:1 (0) | 2.28 | 7.85 |
| 13 | 60 (0) | 70 (0) | 10:1 (−1) | 2.59 | 2.61 |
| 14 | 60 (0) | 70 (0) | 30:1 (+1) | 3.07 | 9.87 |
| 15 | 60 (0) | 70 (0) | 20:1 (0) | 2.74 | 7.11 |
| 16 | 60 (0) | 70 (0) | 20:1 (0) | 2.89 | 6.95 |
| 17 | 60 (0) | 70 (0) | 20:1 (0) | 2.83 | 6.56 |
| 18 | 60 (0) | 70 (0) | 20:1 (0) | 2.89 | 6.56 |
| Name | Model | Equation |
|---|---|---|
| Peleg | (1) | |
| Page | (2) | |
| Patricelli | (3) |
| Statistical Parameters | Equation |
|---|---|
| (4) | |
| (5) | |
| (6) | |
| (7) |
| Source | TPC (R2 = 0.9701) | DPPH (R2 = 0.9864) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| DF | SS | MS | F-Value | p-Value | DF | SS | MS | F-Value | p-Value | |
| Model | 6 | 3.83 | 0.64 | 59.49 | <0.0001 | 7 | 134.34 | 19.19 | 103.33 | <0.0001 |
| Lack of Fit | 8 | 0.10 | 0.01 | 2.62 | 0.2300 | 7 | 1.62 | 0.23 | 2.90 | 0.2056 |
| Pure Error | 3 | 0.015 | 0.005 | 3 | 0.239 | 0.080 | ||||
| Response | Polynomial Equation | |
|---|---|---|
| TPC (mg GAE/g DW) | y = 2.83 + 0.38X1 − 0.22X2 + 0.35X3 + 0.10X1X2 + 0.14X1X3 − 0.33X22 | (8) |
| DPPH radical scavenging (%) | y = 6.85 + 1.11X1 + 0.69X2 + 3.30X3 + 0.51X1X2 + 0.53X1X3 − 0.60X2X3 − 0.44X32 | (9) |
| Response Variable | Model | Extraction Method | Coefficient | Statistical Parameter | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| K1 | K2 | C1 | C2 | RMSE | R2 | adjR2 | %AARD | |||
| TPC | Peleg | BE | 1.28 | 0.27 | - | - | 0.168 | 0.939 | 0.936 | 2.389 |
| UAE | 0.68 | 0.23 | - | - | 0.298 | 0.825 | 0.817 | 7.520 | ||
| MAE | 0.84 | 0.25 | - | - | 0.193 | 0.919 | 0.916 | 3.147 | ||
| Page | BE | −0.62 | 0.17 | - | - | 0.330 | 0.796 | 0.788 | 9.216 | |
| UAE | −0.90 | 0.11 | - | - | 0.310 | 0.808 | 0.800 | 8.120 | ||
| MAE | −0.78 | 0.13 | - | - | 0.278 | 0.836 | 0.829 | 6.539 | ||
| Patricelli | BE | 0.53 | 0.06 | 1.64 | 1.98 | 0.115 | 0.968 | 0.967 | 0.915 | |
| UAE | 1.34 | 0.10 | 1.63 | 2.48 | 0.100 | 0.954 | 0.952 | 0.689 | ||
| MAE | 0.79 | 0.06 | 1.95 | 1.96 | 0.105 | 0.970 | 0.968 | 0.770 | ||
| DPPH | Peleg | BE | 0.33 | 0.07 | - | - | 0.210 | 0.993 | 0.993 | 3.718 |
| UAE | 0.15 | 0.07 | - | - | 0.221 | 0.988 | 0.987 | 4.131 | ||
| MAE | 0.20 | 0.07 | - | - | 0.243 | 0.988 | 0.987 | 5.017 | ||
| Page | BE | −1.85 | 0.08 | - | - | 0.275 | 0.988 | 0.987 | 6.411 | |
| UAE | −2.13 | 0.05 | - | - | 0.226 | 0.987 | 0.987 | 4.323 | ||
| MAE | −2.01 | 0.06 | - | - | 0.218 | 0.990 | 0.989 | 4.029 | ||
| Patricelli | BE | 0.61 | 0.05 | 6.56 | 6.93 | 0.125 | 0.997 | 0.996 | 1.076 | |
| UAE | 1.47 | 0.07 | 7.72 | 6.40 | 0.083 | 0.997 | 0.996 | 0.478 | ||
| MAE | 1.15 | 0.05 | 7.34 | 6.56 | 0.070 | 0.996 | 0.996 | 0.342 | ||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carciochi, R.A.; Sologubik, C.A.; Fernández, M.B.; Manrique, G.D.; D’Alessandro, L.G. Extraction of Antioxidant Phenolic Compounds from Brewer’s Spent Grain: Optimization and Kinetics Modeling. Antioxidants 2018, 7, 45. https://doi.org/10.3390/antiox7040045
Carciochi RA, Sologubik CA, Fernández MB, Manrique GD, D’Alessandro LG. Extraction of Antioxidant Phenolic Compounds from Brewer’s Spent Grain: Optimization and Kinetics Modeling. Antioxidants. 2018; 7(4):45. https://doi.org/10.3390/antiox7040045
Chicago/Turabian StyleCarciochi, Ramiro A., Carlos A. Sologubik, María B. Fernández, Guillermo D. Manrique, and Leandro Galván D’Alessandro. 2018. "Extraction of Antioxidant Phenolic Compounds from Brewer’s Spent Grain: Optimization and Kinetics Modeling" Antioxidants 7, no. 4: 45. https://doi.org/10.3390/antiox7040045
APA StyleCarciochi, R. A., Sologubik, C. A., Fernández, M. B., Manrique, G. D., & D’Alessandro, L. G. (2018). Extraction of Antioxidant Phenolic Compounds from Brewer’s Spent Grain: Optimization and Kinetics Modeling. Antioxidants, 7(4), 45. https://doi.org/10.3390/antiox7040045





