Qualitative and Quantitative Metabolite Comparison of Grain, Persimmon, and Apple Vinegars with Antioxidant Activities
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Vinegar Samples
2.3. Non-Targeted Metabolite Analysis
2.3.1. GC-TOF-MS Metabolite Analysis
2.3.2. UHPLC-Orbitrap-MS/MS Metabolite Analysis
2.3.3. HS-SPME-GC-TOF-MS Metabolite Analysis
2.4. Determination of Antioxidant Activities and Total Flavonoid and Phenolic Content
2.5. Metabolite Quantification Analysis
2.5.1. Quantification of Organic Acids
2.5.2. Quantification of Amino Acids
2.5.3. Quantification of Fatty Acids
2.6. Data Processing and Multivariate Statistical Analysis
3. Results and Discussion
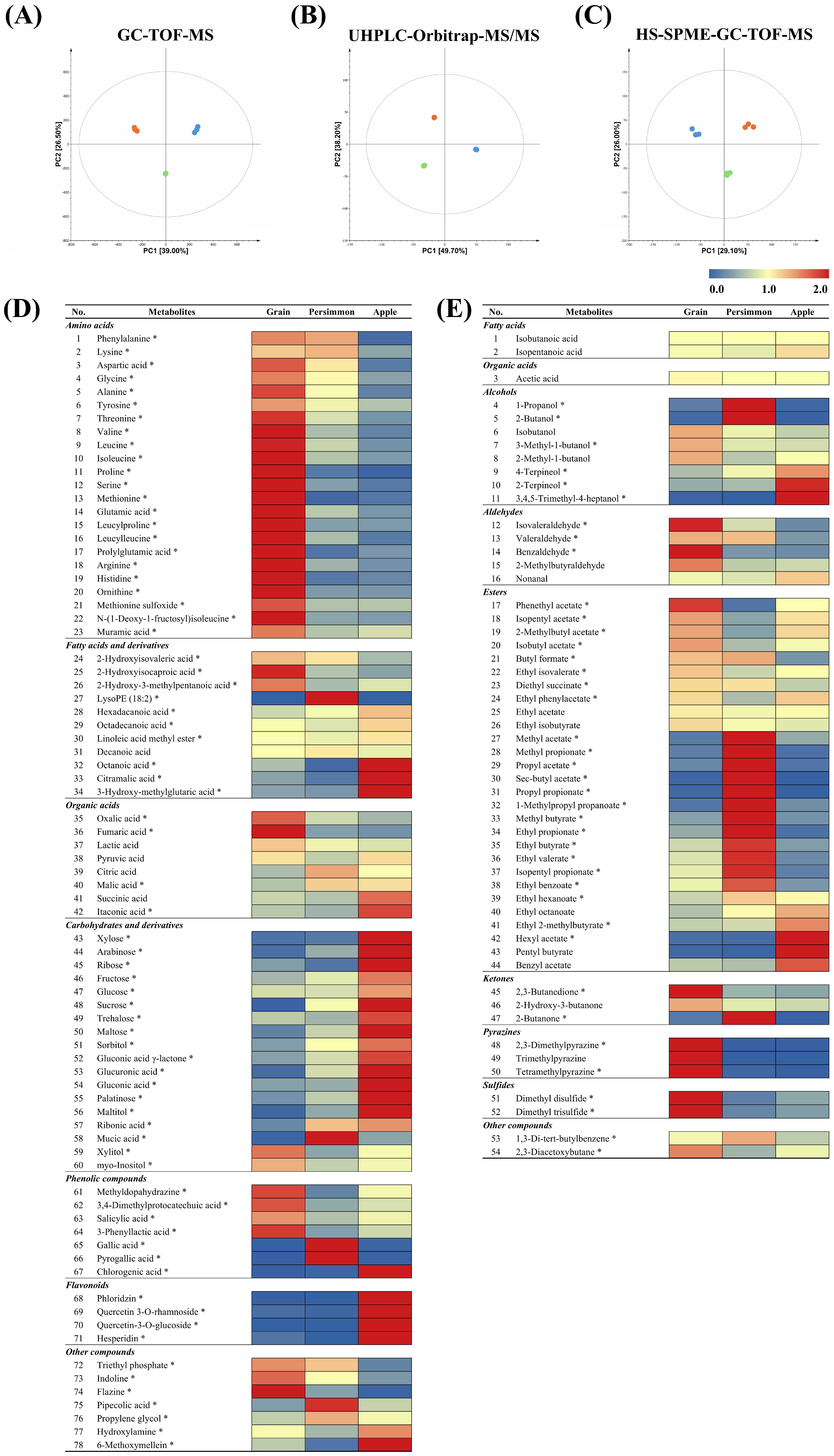
3.1. Non-Targeted Metabolite Profiling of Grain, Persimmon, and Apple Vinegars
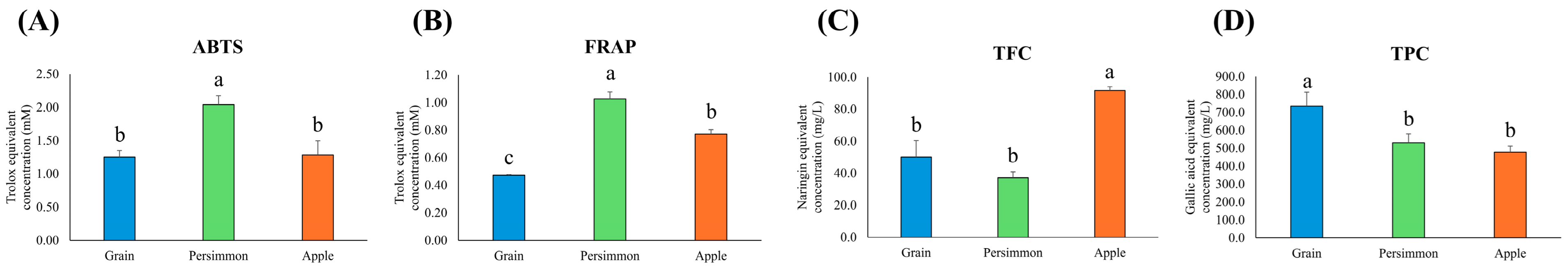
3.2. Antioxidant Activity of Grain, Persimmon, and Apple Vinegars
3.3. Metabolite Comparison in Individual Vinegar Samples
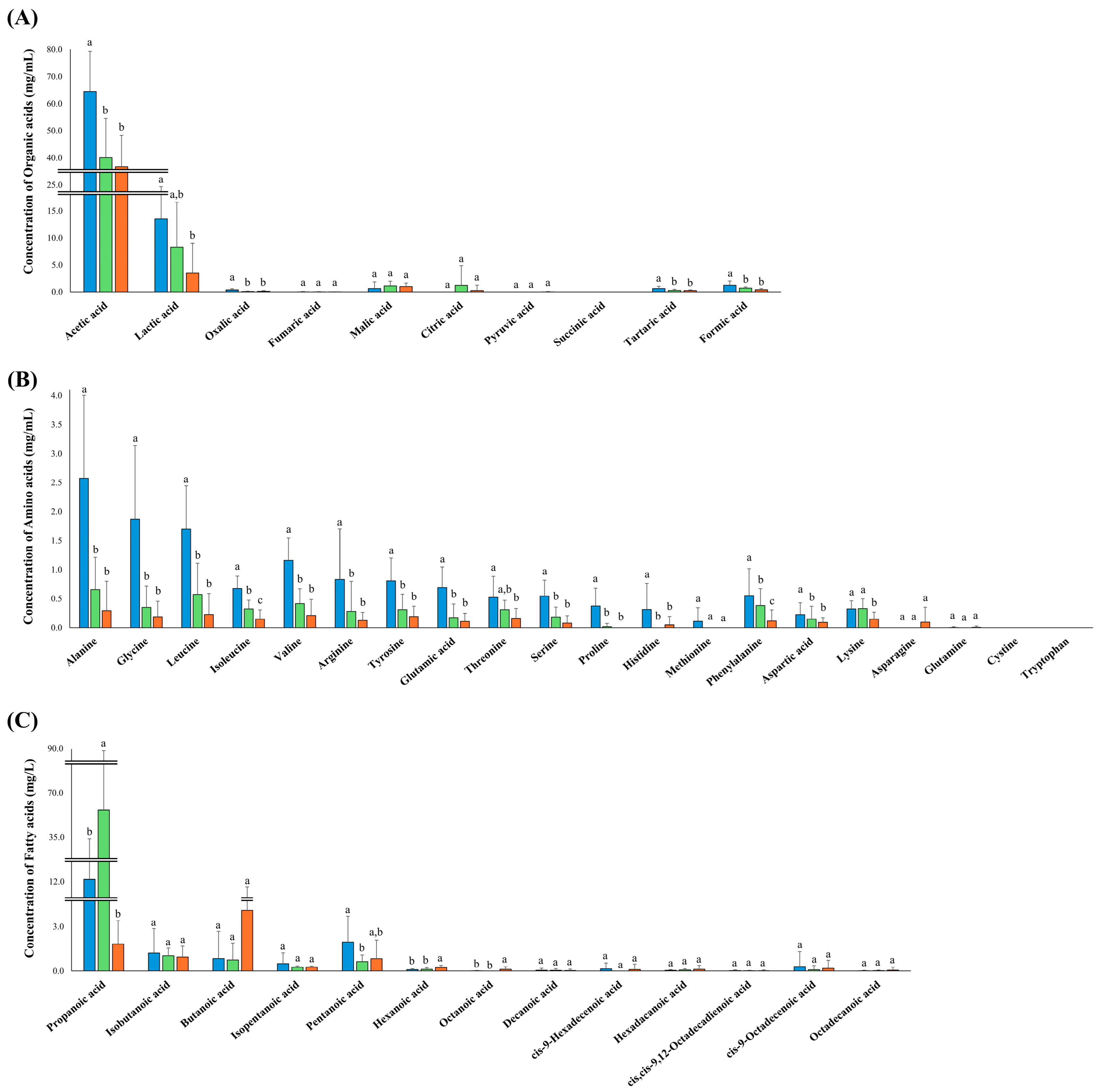
3.4. Quantitative Analysis of Organic Acids, Amino Acids, and Fatty Acids
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Liang, J.; Xie, J.; Hou, L.; Zhao, M.; Zhao, J.; Cheng, J.; Wang, S.; Sun, B.-G. Aroma Constituents in Shanxi Aged Vinegar before and after Aging. J. Agric. Food Chem. 2016, 64, 7597–7605. [Google Scholar] [CrossRef]
- Xia, T.; Zhang, B.; Duan, W.; Zhang, J.; Wang, M. Nutrients and Bioactive Components from Vinegar: A Fermented and Functional Food. J. Funct. Foods 2020, 64, 103681. [Google Scholar] [CrossRef]
- Kim, S.H.; Cho, H.K.; Shin, H.S. Physicochemical Properties and Antioxidant Activities of Commercial Vinegar Drinks in Korea. Food Sci. Biotechnol. 2012, 21, 1729–1734. [Google Scholar] [CrossRef]
- Fan, J.; Zhang, Y.; Chang, X.; Zhang, B.; Jiang, D.; Saito, M.; Li, Z. Antithrombotic and Fibrinolytic Activities of Methanolic Extract of Aged Sorghum Vinegar. J. Agric. Food Chem. 2009, 57, 8683–8687. [Google Scholar] [CrossRef]
- Kong, Y.; Zhang, L.-L.; Sun, Y.; Zhang, Y.-Y.; Sun, B.-G.; Chen, H.-T. Determination of the Free Amino Acid, Organic Acid, and Nucleotide in Commercial Vinegars. J. Food Sci. 2017, 82, 1116–1123. [Google Scholar] [CrossRef] [PubMed]
- Pazuch, C.M.; Siepmann, F.B.; Canan, C.; Colla, E. Vinegar: Functional Aspects. Científica 2015, 43, 302–308. [Google Scholar] [CrossRef]
- Verzelloni, E.; Tagliazucchi, D.; Conte, A. Relationship between the Antioxidant Properties and the Phenolic and Flavonoid Content in Traditional Balsamic Vinegar. Food Chem. 2007, 105, 564–571. [Google Scholar] [CrossRef]
- Petsiou, E.I.; Mitrou, P.I.; Raptis, S.A.; Dimitriadis, G.D. Effect and Mechanisms of Action of Vinegar on Glucose Metabolism, Lipid Profile, and Body Weight. Nutr. Rev. 2014, 72, 651–661. [Google Scholar] [CrossRef] [PubMed]
- Budak, N.H.; Kumbul Doguc, D.; Savas, C.M.; Seydim, A.C.; Kok Tas, T.; Ciris, M.I.; Guzel-Seydim, Z.B. Effects of Apple Cider Vinegars Produced with Different Techniques on Blood Lipids in High-Cholesterol-Fed Rats. J. Agric. Food Chem. 2011, 59, 6638–6644. [Google Scholar] [CrossRef]
- Kandylis, P.; Bekatorou, A.; Dimitrellou, D.; Plioni, I.; Giannopoulou, K. Health Promoting Properties of Cereal Vinegars. Foods 2021, 10, 344. [Google Scholar] [CrossRef]
- Song, N.E.; Jeong, D.Y.; Baik, S.H. Application of Indigenous Saccharomyces Cerevisiae to Improve the Black Raspberry (Rubus Coreanus Miquel) Vinegar Fermentation Process and Its Microbiological and Physicochemical Analysis. Food Sci. Biotechnol. 2018, 28, 481–489. [Google Scholar] [CrossRef] [PubMed]
- Gomes, R.J.; Borges, M.d.F.; Rosa, M.d.F.; Castro-Gómez, R.J.H.; Spinosa, W.A. Acetic Acid Bacteria in the Food Industry: Systematics, Characteristics and Applications. Food Technol. Biotechnol. 2018, 56, 139. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.H.; Lu, Z.M.; Zhang, X.J.; Wang, Z.M.; Yu, Y.J.; Shi, J.S.; Xu, Z.H. Metagenomics Reveals Flavour Metabolic Network of Cereal Vinegar Microbiota. Food Microbiol. 2017, 62, 23–31. [Google Scholar] [CrossRef]
- Xu, W.; Huang, Z.; Zhang, X.; Li, Q.; Lu, Z.; Shi, J.; Xu, Z.; Ma, Y. Monitoring the Microbial Community during Solid-State Acetic Acid Fermentation of Zhenjiang Aromatic Vinegar. Food Microbiol. 2011, 28, 1175–1181. [Google Scholar] [CrossRef] [PubMed]
- Budak, N.H.; Aykin, E.; Seydim, A.C.; Greene, A.K.; Guzel-Seydim, Z.B. Functional Properties of Vinegar. J. Food Sci. 2014, 79, R757–R764. [Google Scholar] [CrossRef]
- Liu, R.C.; Li, R.; Wang, Y.; Jiang, Z.T. Analysis of Volatile Odor Compounds and Aroma Properties of European Vinegar by the Ultra-Fast Gas Chromatographic Electronic Nose. J. Food Compos. Anal. 2022, 112, 104673. [Google Scholar] [CrossRef]
- Xia, T.; Yao, J.; Zhang, J.; Duan, W.; Zhang, B.; Xie, X.; Xia, M.; Song, J.; Zheng, Y.; Wang, M. Evaluation of Nutritional Compositions, Bioactive Compounds, and Antioxidant Activities of Shanxi Aged Vinegars During the Aging Process. J. Food Sci. 2018, 83, 2638–2644. [Google Scholar] [CrossRef]
- Seok, H.; Lee, J.Y.; Park, E.M.; Park, S.E.; Lee, J.H.; Lim, S.; Lee, B.W.; Kang, E.S.; Lee, H.C.; Cha, B.S. Balsamic Vinegar Improves High Fat-Induced Beta Cell Dysfunction via Beta Cell ABCA1. Diabetes Metab. J. 2012, 36, 275–279. [Google Scholar] [CrossRef]
- Launholt, T.L.; Kristiansen, C.B.; Hjorth, P. Safety and Side Effects of Apple Vinegar Intake and Its Effect on Metabolic Parameters and Body Weight: A Systematic Review. Eur. J. Nutr. 2020, 59, 2273–2289. [Google Scholar] [CrossRef]
- Valero, E.; Berlanga, T.M.; Roldán, P.M.; Jiménez, C.; García, I.; Mauricio, J.C. Free Amino Acids and Volatile Compounds in Vinegars Obtained from Different Types of Substrate. J. Sci. Food Agric. 2005, 85, 603–608. [Google Scholar] [CrossRef]
- Gonzalez-Covarrubias, V.; Martínez-Martínez, E.; Bosque-Plata, L. Del The Potential of Metabolomics in Biomedical Applications. Metabolites 2022, 12, 194. [Google Scholar] [CrossRef]
- Tang, J. Microbial Metabolomics. Curr. Genom. 2011, 12, 391. [Google Scholar] [CrossRef]
- Manickam, S.; Rajagopalan, V.R.; Kambale, R.; Rajasekaran, R.; Kanagarajan, S.; Muthurajan, R. Plant Metabolomics: Current Initiatives and Future Prospects. Curr. Issues Mol. Biol. 2023, 45, 8894–8906. [Google Scholar] [CrossRef]
- Mun, H.I.; Kwon, M.C.; Lee, N.R.; Son, S.Y.; Song, D.H.; Lee, C.H. Comparing Metabolites and Functional Properties of Various Tomatoes Using Mass Spectrometry-Based Metabolomics Approach. Front. Nutr. 2021, 8, 659646. [Google Scholar] [CrossRef]
- Salazar-Orbea, G.; García-Villalba, R.; Sánchez-Siles, L.M.; Tomás-Barberán, F.A.; García, C.J. Untargeted Metabolomics Reveals New Markers of Food Processing for Strawberry and Apple Purees. Molecules 2022, 27, 7275. [Google Scholar] [CrossRef] [PubMed]
- Yokota, Y.; Akihiro, T.; Boerzhijin, S.; Yamada, T.; Makino, Y. Effect of the Storage Atmosphere on Metabolomics of Harvested Tomatoes (Solanum Lycopersicum L.). Food Sci. Nutr. 2019, 7, 773. [Google Scholar] [CrossRef] [PubMed]
- Lal Bajya, S.; Shankar Bunkar, D.; Kumar Goyal, S.; Kumar Singh, M.; Kumar Paswan, V.; Lal, S.; Dhyani, P. Foodomics-Based Metabolites Profiling of the Greek Yogurt Incorporated with Unripened Papaya Peel Powder. Food Chem. Mol. Sci. 2024, 8, 100199. [Google Scholar] [CrossRef] [PubMed]
- Afshari, R.; Pillidge, C.J.; Read, E.; Rochfort, S.; Dias, D.A.; Osborn, A.M.; Gill, H. New Insights into Cheddar Cheese Microbiota-Metabolome Relationships Revealed by Integrative Analysis of Multi-Omics Data. Sci. Rep. 2020, 10, 3164. [Google Scholar] [CrossRef]
- Tak, H.J.; Lee, N.R.; Yang, S.; Kim, S.Y.; Lee, C.H. Qualitative and Quantitative Metabolite Comparison of Korean Traditional Alcoholic Beverages: Takju, Yakju, and Traditional-Soju. Foods 2024, 13, 956. [Google Scholar] [CrossRef]
- Pinu, F.R.; De Carvalho-Silva, S.; Uetanabaro, A.P.T.; Villas-Boas, S.G. Vinegar Metabolomics: An Explorative Study of Commercial Balsamic Vinegars Using Gas Chromatography-Mass Spectrometry. Metabolites 2016, 6, 22. [Google Scholar] [CrossRef]
- Ren, M.; Wang, X.; Tian, C.; Li, X.; Zhang, B.; Song, X.; Zhang, J. Characterization of Organic Acids and Phenolic Compounds of Cereal Vinegars and Fruit Vinegars in China. J. Food Process. Preserv. 2017, 41. [Google Scholar] [CrossRef]
- Piergiovanni, M.; Giliberti, C.; Maffezzoni, C.; Errico, D.; Blandino, M.; Dall’Asta, C.; Mattarozzi, M.; Bianchi, F.; Giannetto, M.; Careri, M. Electronic Nose Technology for the Detection of Ergot Alkaloid in Soft Wheat and Identification of the Relevant Volatile Compounds by Solid Phase Microextraction/Gas Chromatography-High Resolution Orbitrap-Mass Spectrometry Coupled to Chemometrics. Food Chem. 2025, 484, 144455. [Google Scholar] [CrossRef]
- Al-Dalali, S.; Zheng, F.; Sun, B.; Zhou, C.; Li, M.; Chen, F. Effects of Different Brewing Processes on the Volatile Flavor Profiles of Chinese Vinegar Determined by HS-SPME-AEDA with GC-MS and GC-O. LWT 2020, 133, 109969. [Google Scholar] [CrossRef]
- Hijazi, A.; Pisano, I.; Illek, P.; Leahy, J.J. A Rapid HPLC Method for the Simultaneous Determination of Organic Acids and Furans: Food Applications. Beverages 2022, 8, 6. [Google Scholar] [CrossRef]
- Herbert, P.; Santos, L.; Alves, A. Simultaneous Quantification of Primary, Secondary Amino Acids, and Biogenic Amines in Musts and Wines Using OPA/3-MPA/FMOC-CI Fluorescent Derivatives. J. Food Sci. 2001, 66, 1319–1325. [Google Scholar] [CrossRef]
- Jajic, I.; Krstovic, S.; Glamocic, D.; Jakšic, S.; Abramovic, B. Validation of an HPLC Method for the Determination of Amino Acids in Feed. J. Serbian Chem. Soc. 2013, 78, 839–850. [Google Scholar] [CrossRef]
- Chen, G.; Zhang, Q. Simultaneous Quantification of Free Fatty Acids and Acylcarnitines in Plasma Samples Using Dansylhydrazine Labeling and Liquid Chromatography–Triple Quadrupole Mass Spectrometry. Anal. Bioanal. Chem. 2020, 412, 2841–2849. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Ruan, H.; Zong, Z.; Mao, F.; Wang, Y.; Jiao, Y.; Xu, L.; Yang, T.; Li, W.; Liu, X. A Simulated Strategy for Analysis of Short- to Long- Chain Fatty Acids in Mouse Serum beyond Chemical Standards. J. Chromatogr. B 2021, 1180, 122895. [Google Scholar] [CrossRef]
- Jayaprakash, G.; Bains, A.; Chawla, P.; Fogarasi, M.; Fogarasi, S. A Narrative Review on Rice Proteins: Current Scenario and Food Industrial Application. Polymers 2022, 14, 3003. [Google Scholar] [CrossRef]
- Tyagi, A.; Lim, M.J.; Kim, N.H.; Barathikannan, K.; Vijayalakshmi, S.; Elahi, F.; Ham, H.J.; Oh, D.H. Quantification of Amino Acids, Phenolic Compounds Profiling from Nine Rice Varieties and Their Antioxidant Potential. Antioxidants 2022, 11, 839. [Google Scholar] [CrossRef] [PubMed]
- Kalpanadevi, C.; Muthukumar, S.P.; Govindaraju, K.; Subramanian, R. Rice Bran Protein: An Alternative Plant-Based Protein to Ameliorate Protein Malnourishment. J. Cereal. Sci. 2021, 97, 103154. [Google Scholar] [CrossRef]
- Ma, R.; Zhan, J.; Lu, H.; Chang, R.; Tian, Y. Interactions between Recrystallized Rice Starch and Flavor Molecules. Food Hydrocoll. 2022, 124, 107271. [Google Scholar] [CrossRef]
- Ho, C.W.; Lazim, A.M.; Fazry, S.; Zaki, U.K.H.H.; Lim, S.J. Varieties, Production, Composition and Health Benefits of Vinegars: A Review. Food Chem. 2017, 221, 1621–1630. [Google Scholar] [CrossRef]
- Zhao, J.; Boatright, W.L. Static Headspace Analysis of Odorants in Commercial Rice Proteins. Food Chem. 2017, 221, 345–350. [Google Scholar] [CrossRef]
- Yue, Z.; Gong, B.; Cheng, W.; Wu, K.; Yang, X.; Wang, Y.; Liu, C.; Dong, Y.; Xu, Y. Identification of Main Metabolites Correlated with the Sensory Attributes of Diospyros Kaki. Cv. “Taishuu” through a Large-Scale Comprehensive Analysis by Sensory Evaluation, Electronic Tongue and Metabolomics. LWT 2024, 195, 115834. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, S.; Meng, Q.; Song, H.; Wang, X. Characterization of Key Odor-Active Compounds in Draft Beers for the Chinese Market Using Molecular Sensory Science Approaches. Molecules 2024, 29, 2537. [Google Scholar] [CrossRef]
- Lee, J.H.; Lee, Y.B.; Seo, W.D.; Kang, S.T.; Lim, J.W.; Cho, K.M. Comparative Studies of Antioxidant Activities and Nutritional Constituents of Persimmon Juice (Diospyros Kaki L. Cv. Gapjubaekmok). Prev. Nutr. Food Sci. 2012, 17, 141. [Google Scholar] [CrossRef]
- Alavi Rafiee, S.; Farhoosh, R.; Sharif, A. Antioxidant Activity of Gallic Acid as Affected by an Extra Carboxyl Group than Pyrogallol in Various Oxidative Environments. Eur. J. Lipid Sci. Technol. 2018, 120, 1800319. [Google Scholar] [CrossRef]
- Colamartino, M.; Santoro, M.; Duranti, G.; Sabatini, S.; Ceci, R.; Testa, A.; Padua, L.; Cozzi, R. Evaluation of Levodopa and Carbidopa Antioxidant Activity in Normal Human Lymphocytes In Vitro: Implication for Oxidative Stress in Parkinson’s Disease. Neurotox. Res. 2015, 27, 106–117. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.W.; Jung, E.; Kim, S.; Lee, K.E.; Youm, J.K.; Park, D. Antagonist Effects of Veratric Acid against UVB-Induced Cell Damages. Molecules 2013, 18, 5405–5419. [Google Scholar] [CrossRef]
- Moustafa-Farag, M.; Mohamed, H.I.; Mahmoud, A.; Elkelish, A.; Misra, A.N.; Guy, K.M.; Kamran, M.; Ai, S.; Zhang, M. Salicylic Acid Stimulates Antioxidant Defense and Osmolyte Metabolism to Alleviate Oxidative Stress in Watermelons under Excess Boron. Plants 2020, 9, 724. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Wang, Y.; Wang, M.; Kong, Y.; Li, X.; Song, D.; Zeng, X.; Yang, Y.; Fan, X.; Gong, H. Improvement of Antioxidant Capacity, Aroma Quality, and Antifungal Ability of Cherry by Phenyllactic Acid Treatment during Low Temperature Storage. Front. Plant Sci. 2024, 15, 1529127. [Google Scholar] [CrossRef]
- Park, S.; Park, S.K. Anti-Oxidant and Anti-Aging Effects of Phlorizin Are Mediated by DAF-16-Induced Stress Response and Autophagy in Caenorhabditis Elegans. Antioxidants 2022, 11, 1996. [Google Scholar] [CrossRef]
- Huang, J.; Xie, M.; He, L.; Song, X.; Cao, T. Chlorogenic Acid: A Review on Its Mechanisms of Anti-Inflammation, Disease Treatment, and Related Delivery Systems. Front. Pharmacol. 2023, 14, 1218015. [Google Scholar] [CrossRef] [PubMed]
- Septembre-Malaterre, A.; Boumendjel, A.; Seteyen, A.L.S.; Boina, C.; Gasque, P.; Guiraud, P.; Sélambarom, J. Focus on the High Therapeutic Potentials of Quercetin and Its Derivatives. Phytomed. Plus 2022, 2, 100220. [Google Scholar] [CrossRef]
- Tsatsop Tsague, R.K.; Kenmogne, S.B.; Djiobie Tchienou, G.E.; Parra, K.; Ngassoum, M.B. Sequential Extraction of Quercetin-3-O-Rhamnoside from Piliostigma Thonningii Schum. Leaves Using Microwave Technology. SN Appl. Sci. 2020, 2, 1230. [Google Scholar] [CrossRef]
- Parhiz, H.; Roohbakhsh, A.; Soltani, F.; Rezaee, R.; Iranshahi, M. Antioxidant and Anti-Inflammatory Properties of the Citrus Flavonoids Hesperidin and Hesperetin: An Updated Review of Their Molecular Mechanisms and Experimental Models. Phytother. Res. 2015, 29, 323–331. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tak, H.-J.; Yang, S.; Kim, S.-Y.; Lee, N.-R.; Lee, C.H. Qualitative and Quantitative Metabolite Comparison of Grain, Persimmon, and Apple Vinegars with Antioxidant Activities. Antioxidants 2025, 14, 1029. https://doi.org/10.3390/antiox14081029
Tak H-J, Yang S, Kim S-Y, Lee N-R, Lee CH. Qualitative and Quantitative Metabolite Comparison of Grain, Persimmon, and Apple Vinegars with Antioxidant Activities. Antioxidants. 2025; 14(8):1029. https://doi.org/10.3390/antiox14081029
Chicago/Turabian StyleTak, Hyun-Ji, Sowon Yang, So-Young Kim, Na-Rae Lee, and Choong Hwan Lee. 2025. "Qualitative and Quantitative Metabolite Comparison of Grain, Persimmon, and Apple Vinegars with Antioxidant Activities" Antioxidants 14, no. 8: 1029. https://doi.org/10.3390/antiox14081029
APA StyleTak, H.-J., Yang, S., Kim, S.-Y., Lee, N.-R., & Lee, C. H. (2025). Qualitative and Quantitative Metabolite Comparison of Grain, Persimmon, and Apple Vinegars with Antioxidant Activities. Antioxidants, 14(8), 1029. https://doi.org/10.3390/antiox14081029






