Therapeutic Potential of Apocynin: A Promising Antioxidant Strategy for Acute Kidney Injury
Abstract
1. Introduction
1.1. Oxidative Stress in Acute Kidney Injury
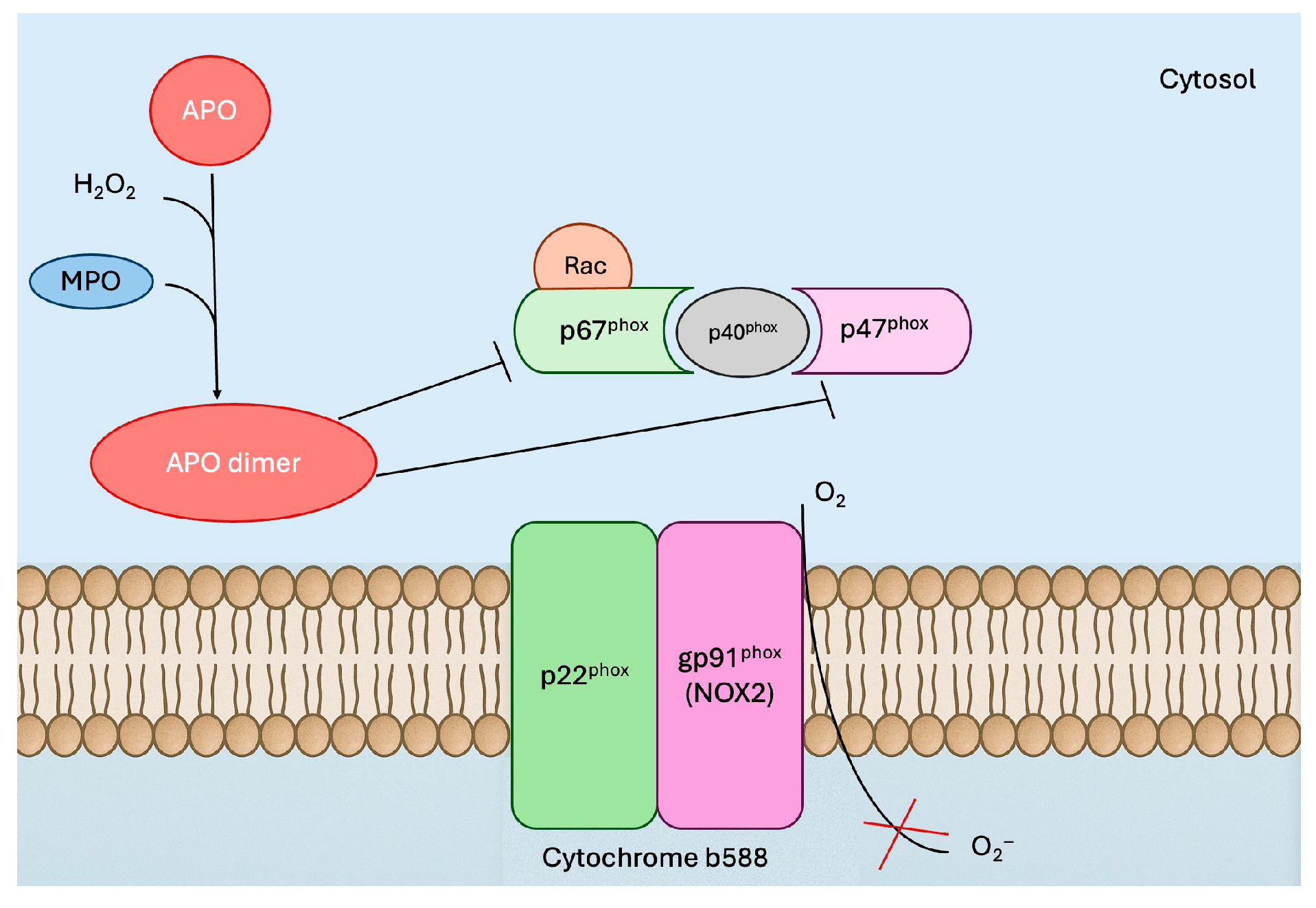
1.2. Apocynin as an Antioxidant
2. Materials and Methods
3. Results
4. Discussion
4.1. The Effects of Apocynin in Ischemia–Reperfusion Acute Kidney Injury
4.2. The Effects of Apocynin in Nephrotoxic Acute Kidney Injury
4.3. The Effects of Apocynin in Other Types of Acute Kidney Injury
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ronco, C.; Bellomo, R.; Kellum, J.A. Acute kidney injury. Lancet 2019, 394, 1949–1964. [Google Scholar] [CrossRef] [PubMed]
- Pickkers, P.; Darmon, M.; Hoste, E.; Joannidis, M.; Legrand, M.; Ostermann, M.; Prowle, J.R.; Schneider, A.; Schetz, M. Acute kidney injury in the critically ill: An updated review on pathophysiology and management. Intensive Care Med. 2021, 47, 835–850. [Google Scholar] [CrossRef] [PubMed]
- Hoste, E.A.; Bagshaw, S.M.; Bellomo, R.; Cely, C.M.; Colman, R.; Cruz, D.N.; Edipidis, K.; Forni, L.G.; Gomersall, C.D.; Govil, D.; et al. Epidemiology of acute kidney injury in critically ill patients: The multinational AKI-EPI study. J. Am. Soc. Nephrol. 2015, 26, 1411–1423. [Google Scholar] [CrossRef] [PubMed]
- Makris, K.; Spanou, L. Acute kidney injury: Definition, pathophysiology and clinical phenotypes. Clin. Biochem. Rev. 2016, 37, 85–98. [Google Scholar] [PubMed]
- Gyurászová, M.; Gurecká, R.; Bábíčková, J.; Tóthová, Ľ. Oxidative stress in acute kidney injury: Implications for Noninvasive Monitoring and Identification of Biomarkers. Oxid. Med. Cell Longev. 2020, 2020, 5478708. [Google Scholar] [CrossRef]
- Gaut, J.P.; Liapis, H. Acute kidney injury pathology and pathophysiology: A retrospective review. Clin. Kidney J. 2021, 14, 526–536. [Google Scholar] [CrossRef]
- Kaur, A.; Tiwari, R.; Tiwari, G.; Ramachandran, V. Resveratrol: A vital therapeutic agent with multiple health benefits. Drug Res. 2021, 72, 5–17. [Google Scholar] [CrossRef]
- Piko, N.; Bevc, S.; Hojs, R.; Ekart, R. The role of oxidative stress in kidney injury. Antioxidants 2023, 12, 1772. [Google Scholar] [CrossRef]
- Tomsa, A.M.; Alexa, A.L.; Junie, M.L.; Rachisan, A.L.; Ciumarnean, L. Oxidative stress as a potential target in acute kidney injury. PeerJ 2019, 7, e8046. [Google Scholar] [CrossRef]
- Su, H.; Wan, C.; Song, A.; Qiu, Y.; Xiong, W.; Zhang, C. Oxidative stress and renal fibrosis: Mechanisms and therapies. Adv. Exp. Med. Biol. 2019, 1165, 585–604. [Google Scholar] [CrossRef]
- Naito, H.; Nojima, T.; Fujisaki, N.; Tsukahara, K.; Yamamoto, H.; Yamada, T.; Aokage, T.; Yumoto, T.; Osako, T.; Nakao, A. Therapeutic strategies for ischemia reperfusion injury in emergency medicine. Acute Med. Surg. 2020, 7, e501. [Google Scholar] [CrossRef]
- Zweier, J.L.; Flaherty, J.T.; Weisfeldt, M.L. Direct measurement of free radical generation following reperfusion of ischemic myocardium. Proc. Natl. Acad. Sci. USA 1987, 84, 1404–1407. [Google Scholar] [CrossRef] [PubMed]
- Chouchani, E.T.; Pell, V.R.; James, A.M.; Work, L.M.; Saeb-Parsy, K.; Frezza, C.; Krieg, T.; Murphy, M.P. A unifying mechanism for mitochondrial superoxide production during ischemia-reperfusion injury. Cell Metab. 2016, 23, 254–263. [Google Scholar] [CrossRef] [PubMed]
- Friedewald, J.J.; Rabb, H. Inflammatory cells in ischemic acute renal failure. Kidney Int. 2004, 66, 486–491. [Google Scholar] [CrossRef] [PubMed]
- Basile, D.P.; Anderson, M.D.; Sutton, T.A. Pathophysiology of acute kidney injury. Compr. Physiol. 2012, 2, 1303–1353. [Google Scholar] [CrossRef]
- McCord, J.M. Oxygen-derived free radicals in postischemic tissue injury. N. Engl. J. Med. 1985, 312, 159–163. [Google Scholar] [CrossRef]
- Himmelfarb, J.; McMonagle, E.; Freedman, S.; Klenzak, J.; McMenamin, E.; Le, P.; Pupim, L.B.; Ikizler, T.A. Oxidative stress is increased in critically ill patients with acute renal failure. J. Am. Soc. Nephrol. 2004, 15, 2449–2456. [Google Scholar] [CrossRef]
- Parazella, M.A.; Rosner, M.H. Drug-induced acute kidney injury. Clin. J. Am. Soc. Nephrol. 2022, 17, 1220–1233. [Google Scholar] [CrossRef]
- Hosohata, K. Role of oxidative stress in drug-induced kidney injury. Int. J. Mol. Sci. 2016, 17, 1826. [Google Scholar] [CrossRef]
- Dennis, M.; Witting, P.K. Protective role for antioxidants in acute kidney disease. Nutrients 2017, 9, 718. [Google Scholar] [CrossRef]
- Kusirisin, P.; Chattipakorn, S.C.; Chattipakorn, N. Contrast-induced nephropathy and oxidative stress: Mechanistic insights for better interventional approaches. J. Transl. Med. 2020, 18, 400. [Google Scholar] [CrossRef] [PubMed]
- Rashid, H.; Jali, A.; Akhter, M.S.; Abdi, S.A.H. Molecular mechanisms of oxidative stress in acute kidney injury: Targeting the loci by resveratrol. Int. J. Mol. Sci. 2024, 25, 3. [Google Scholar] [CrossRef] [PubMed]
- Boshtam, M.; Kouhpayeh, S.; Amini, F.; Azizi, Y.; Najaflu, M.; Shariati, L.; Khanahmad, H. Antiinflammatory effects of apocynin: A narrative review of the evidence. All Life 2021, 14, 997–1010. [Google Scholar] [CrossRef]
- Brandes, R.P.; Weissmann, N.; Schröder, K. Redox-mediated signal transduction by cardiovascular NOX NADPH oxidases. Free Radic. Biol. Med. 2014, 76, 208–226. [Google Scholar] [CrossRef]
- Heumüller, S.; Wind, S.; Barbosa Sicard, E.; Schmidt, H.H.H.W.; Busse, R.; Schröder, K.; Brandes, R.P. Apocynin is not an inhibitor of vascular NADPH oxidases but an antioxidant. Hypertension 2008, 51, 211–217. [Google Scholar] [CrossRef]
- Ximenes, V.F.; Kanegae, M.P.; Rissato, S.R.; Galhiane, M.S. The oxidation of apocynin catalyzed by myeloperoxidase: Proposal for NADPH oxidase inhibition. Arch. Biochem. Biophys. 2007, 457, 134–141. [Google Scholar] [CrossRef]
- Barbieri, S.S.; Cavalca, V.; Eligini, S.; Brambilla, M.; Caiani, A.; Tremoli, E.; Colli, S. Apocynin prevents cyclooxygenase 2 expression in human monocytes through NADPH oxidase and glutathione redox-dependent mechanisms. Free Radic. Biol. Med. 2004, 37, 156–165. [Google Scholar] [CrossRef]
- Kanegae, M.P.; da Fonseca, L.M.; Brunetti, I.L.; Silva, S.O.; Ximenes, V.F. The reactivity of ortho-methoxy-substituted catechol radicals with sulfhydryl groups: Contribution for the comprehension of the mechanism of inhibition of NADPH oxidase by apocynin. Biochem. Pharmacol. 2007, 74, 457–464. [Google Scholar] [CrossRef]
- Mora-Pale, M.; Kwon, S.J.; Linhardt, R.J.; Dordick, J.S. Trimer hydroxylated quinone derived from apocynin targets cysteine residues of p47phox preventing the activation of human vascular NADPH oxidase. Free Radic. Biol. Med. 2012, 52, 962–969. [Google Scholar] [CrossRef]
- Mohammad, A.; Babiker, F.; Al-Bader, M. Effects of apocynin, a NADPH oxidase inhibitor, in the protection of the heart from ischemia/reperfusion injury. Pharmaceutics 2023, 16, 492. [Google Scholar] [CrossRef]
- Riganti, C.; Costamagna, C.; Bosia, A.; Ghigo, D. The NADPH oxidase inhibitor apocynin (acetovanillone) induces oxidative stress. Toxicol. Appl. Pharmacol. 2006, 212, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Vejrazka, M.; Micek, R.; Stipek, S. Apocynin inhibits NADPH oxidase in phagocytes but stimulates ROS production in non-phagocytic cells. Biochim. Biophys. Acta 2005, 1722, 143–147. [Google Scholar] [CrossRef] [PubMed]
- Dharmarajah, J.; Arthur, J.F.; Sobey, C.G.; Drummond, G.R. The anti-platelet effects of apocynin in mice are not mediated by inhibition of NADPH oxidase activity. Naunyn Schmiedebergs Arch. Pharmacol. 2010, 382, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Schlüter, T.; Steinbach, A.C.; Steffen, A.; Rettig, R.; Grisk, O. Apocynin-induced vasodilation involves Rho kinase inhibition but not NADPH oxidase inhibition. Cardiovasc. Res. 2008, 80, 271–279. [Google Scholar] [CrossRef]
- Stefanska, J.; Pawliczak, R. Apocynin: Molecular aptitudes. Mediat. Inflamm. 2008, 2008, 106507. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Altintas, R.; Polat, A.; Vardi, N.; Oguz, F.; Beytur, A.; Sagir, M.; Yildiz, A.; Parlakpinar, H. The protective effects of apocynin on kidney damage caused by renal ischemia/reperfusion. J. Endourol. 2013, 27, 617–624. [Google Scholar] [CrossRef]
- Li, Z.; Wang, Y. Effect of NADPH oxidase inhibitor-apocynin on the expression of Src homology-2 domain-containing phosphatase-1 (SHP-1) exposed renal ischemia/reperfusion injury in rats. Toxicol. Rep. 2015, 4, 1111–1116. [Google Scholar] [CrossRef]
- Choi, E.K.; Jung, H.; Kwak, K.H.; Yeo, J.; Yi, S.J.; Park, C.Y.; Ryu, T.H.; Jeon, Y.H.; Park, K.M.; Lim, D.G. Effects of allopurinol and apocynin on renal ischemia-reperfusion injury in rats. Transplant. Proc. 2015, 47, 1633–1638. [Google Scholar] [CrossRef]
- Hu, B.; Wu, Y.; Tong, F.; Liu, J.; Shen, X.; Shen, R.; Xu, G. Apocynin alleviates renal ischemia/reperfusion injury through regulating the level of zinc and metallothionein. Biol. Trace Elem. Res. 2017, 178, 301–308. [Google Scholar] [CrossRef]
- Kovacevic, S.; Ivanov, M.; Miloradovic, Z.; Brkic, P.; Vajic, U.J.; Zivotic, M.; Mihailovic-Stanojevic, N.; Jovovic, D.; Karanovic, D.; Jeremic, R.; et al. Hyperbaric oxygen preconditioning and the role of NADPH oxidase inhibition in postischemic acute kidney injury induced in spontaneously hypertensive rats. PLoS ONE 2020, 15, e0226974. [Google Scholar] [CrossRef]
- Lima, N.K.S.; Farias, W.R.A.; Cirilo, M.A.S.; Oliveira, A.G.; Farias, J.S.; Aires, R.S.; Muzi-Filho, H.; Paixão, A.D.O.; Vieira, L.D. Renal ischemia-reperfusion leads to hypertension and changes in proximal tubule Na⁺ transport and renin-angiotensin-aldosterone system: Role of NADPH oxidase. Life Sci. 2021, 266, 118879. [Google Scholar] [CrossRef]
- Kovacevic, S.; Ivanov, M.; Zivotic, M.; Brkic, P.; Miloradovic, Z.; Jeremic, R.; Mihailovic-Stanojevic, N.; Vajic, U.J.; Karanovic, D.; Jovovic, D.; et al. Immunohistochemical analysis of 4-HNE, NGAL, and HO-1 tissue expression after apocynin treatment and HBO preconditioning in postischemic acute kidney injury induced in spontaneously hypertensive rats. Antioxidants 2021, 10, 1163. [Google Scholar] [CrossRef] [PubMed]
- Révész, C.; Kaucsár, T.; Godó, M.; Bocskai, K.; Krenács, T.; Mócsai, A.; Szénási, G.; Hamar, P. Neutrophils and NADPH oxidases are major contributors to mild but not severe ischemic acute kidney injury in mice. Int. J. Mo. Sci. 2024, 25, 2948. [Google Scholar] [CrossRef] [PubMed]
- Chirino, Y.I.; Sánchez-González, D.J.; Martínez-Martínez, C.M.; Cruz, C.; Pedraza-Chaverri, J. Protective effects of apocynin against cisplatin-induced oxidative stress and nephrotoxicity. Toxicology 2008, 245, 18–23. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Luo, X.; Pan, H.; Huang, W.; Wang, X.; Wen, H.; Shen, K.; Jin, B. Pharmacological inhibition of NADPH oxidase protects against cisplatin induced nephrotoxicity in mice by two step mechanism. Food Chem. Toxicol. 2015, 83, 251–260. [Google Scholar] [CrossRef]
- Meng, X.M.; Ren, G.L.; Gao, L.; Yang, Q.; Li, H.D.; Wu, W.F.; Huang, C.; Zhang, L.; Lv, X.W.; Li, J. NADPH oxidase 4 promotes cisplatin-induced acute kidney injury via ROS-mediated programmed cell death and inflammation. Lab. Investig. 2018, 98, 63–78. [Google Scholar] [CrossRef]
- Abdelrahman, R.S. Protective effect of apocynin against gentamicin-induced nephrotoxicity in rats. Hum. Exp. Toxicol. 2018, 37, 27–37. [Google Scholar] [CrossRef]
- Tan, Y.C.; Sattar, M.A.; Ahmed, A.F.; Khan, N.A.; Murugaiyah, V.; Ahmad, A.; Hassan, Z.; Kaur, G.; Abdulla, M.H.; Johns, E.J. Apocynin and catalase prevent hypertension and kidney injury in Cyclosporine A-induced nephrotoxicity in rats. PLoS ONE 2020, 15, e0231472. [Google Scholar] [CrossRef]
- Ahmad, A.; Mondello, S.; Di Paola, R.; Mazzon, E.; Esposito, E.; Catania, M.A.; Italiano, D.; Mondello, P.; Aloisi, C.; Cuzzocrea, S. Protective effect of apocynin, a NADPH-oxidase inhibitor, against contrast-induced nephropathy in the diabetic rats: A comparison with n-acetylcysteine. Eur. J. Pharmacol. 2012, 674, 397–406. [Google Scholar] [CrossRef]
- Ageena, S.A.; Bakr, A.G.; Mokhlis, H.A.; Abd-Ellah, M.F. Renoprotective effects of apocynin and/or umbelliferone against acrylamide-induced acute kidney injury in rats: Role of the NLRP3 inflammasome and Nrf-2/HO-1 signaling pathways. Naunyn Schmiedebergs Arch. Pharmacol. 2025, 398, 569–580. [Google Scholar] [CrossRef]
- Mittwede, P.N.; Xiang, L.; Lu, S.; Clemmer, J.S.; Hester, R.L. Oxidative stress contributes to orthopedic trauma-induced acute kidney injury in obese rats. Am. J. Physiol. Ren. Physiol. 2015, 308, F157–F163. [Google Scholar] [CrossRef]
- Yang, X.; Zhao, K.; Deng, W.; Zhao, L.; Jin, H.; Mei, F.; Zhou, Y.; Li, M.; Wang, W. Apocynin attenuates acute kidney injury and inflammation in rats with acute hypertriglyceridemic pancreatitis. Dig. Dis. Sci. 2020, 65, 1735–1747. [Google Scholar] [CrossRef] [PubMed]
- Awad, A.S.; Rouse, M.; Huang, L.; Vergis, A.L.; Reutershan, J.; Cathro, H.P.; Linden, J.; Okusa, M.D. Compartmentalization of neutrophils in the kidney and lung following acute ischemic kidney injury. Kidney Int. 2009, 75, 689–698. [Google Scholar] [CrossRef] [PubMed]
- Connell, B.J.; Saleh, M.C.; Khan, B.V.; Saleh, T.M. Apocynin may limit total cell death following cerebral ischemia and reperfusion by enhancing apoptosis. Food Chem. Toxicol. 2011, 49, 3063–3069. [Google Scholar] [CrossRef] [PubMed]
- Kouki, A.; Ferjani, W.; Ghanem-Boughanmi, N.; Ben-Attia, M.; Dang, P.M.; Souli, A.; El-Benna, J. The NADPH oxidase inhibitors apocynin and diphenyleneiodonium protect rats from LPS-induced pulmonary inflammation. Antioxidants 2023, 12, 770. [Google Scholar] [CrossRef]
- Simonyi, A.; Serfozo, P.; Lehmidi, T.M.; Cui, J.; Gu, Z.; Lubahn, D.B.; Sun, A.Y.; Sun, G.Y. The neuroprotective effects of apocynin. Front. Biosci. Elite Ed. 2012, 4, 2183–2193. [Google Scholar] [CrossRef]
- Yu, J.; Weiwer, M.; Linhardt, R.J.; Dordick, J.S. The role of the methoxyphenol apocynin, a vascular NADPH oxidase inhibitor, as a chemopreventative agent in the potential treatment of cardiovascular diseases. Curr. Vasc. Pharmacol. 2008, 6, 204–217. [Google Scholar] [CrossRef]
- Ivanov, M.; Mihailovic-Stanojevic, N.; Grujic Milanovic, J.; Jovovic, D.; Markovic-Lipkovski, J.; Cirovic, S.; Miloradovic, Z. Losartan improved antioxidant defense, renal function and structure of postischemic hypertensive kidney. PLoS ONE 2014, 9, e96353. [Google Scholar] [CrossRef] [PubMed]
- Sachse, A.; Wolf, G. Angiotensin II-induced reactive oxygen species and the kidney. J. Am. Soc. Nephrol. 2007, 18, 2439–2446. [Google Scholar] [CrossRef]
- Liu, Y. Renal fibrosis: New insights into the pathogenesis and therapeutics. Kidney Int. 2006, 69, 213–217. [Google Scholar] [CrossRef] [PubMed]
- Vieira, L.D.; Farias, J.S.; De Queiroz, D.B.; Cabral, E.V.; Lima-Filho, M.M.; Sant’Helena, B.R.M.; Aires, R.S.; Ribeiro, V.S.; Santos-Rocha, J.; Xavier, F.E.; et al. Oxidative stress induced by prenatal LPS leads to endothelial dysfunction and renal haemodynamic changes through angiotensin II/NADPH oxidase pathway: Prevention by early treatment with α-tocopherol, Biochim. Biophys. Acta Mol. Basis Dis. 2018, 1864, 3577–3587. [Google Scholar] [CrossRef]
- Perassa, L.A.; Graton, M.E.; Potje, S.R.; Troiano, J.A.; Lima, M.S.; Vale, G.T.; Pereira, A.A.; Nakamune, A.C.; Sumida, D.H.; Tirapelli, C.R.; et al. Apocynin reduces blood pressure and restores the proper function of vascular endothelium in SHR. Vasc. Pharmacol. 2016, 87, 38–48. [Google Scholar] [CrossRef]
- Feng, W.; Remedies, C.E.; Obi, I.E.; Aldous, S.R.; Meera, S.I.; Sanders, P.W.; Inscho, E.W.; Guan, Z. Restoration of afferent arteriolar autoregulatory behavior in ischemia-reperfusion injury in rat kidneys. Am. J. Physiol. Ren. Physiol. 2021, 320, F429–F441. [Google Scholar] [CrossRef]
- Daugaard, G.; Abildgaard, U. Cisplatin nephrotoxicity. A review. Cancer Chemother. Pharmacol. 1989, 25, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Arany, I.; Safirstein, R.L. Cisplatin nephrotoxicity. Semin. Nephrol. 2003, 23, 460–464. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Novoa, J.M.; Quiros, Y.; Vicente, L.; Morales, A.I.; Lopez-Hernandez, F.J. New insights into the mechanism of aminoglycoside nephrotoxicity: An integrative point of view. Kidney Int. 2011, 79, 33–45. [Google Scholar] [CrossRef] [PubMed]
- Laaroussi, H.; Bakour, M.; Ousaaid, D.; Ferreira-Santos, P.; Genisheva, Z.; El Ghouizi, A.; Aboulghazi, A.; Teixeira, J.A.; Lyoussi, B. Protective effect of honey and propolis against gentamicin-induced oxidative stress and hepatorenal damages. Oxid. Med. Cell Longev. 2021, 2021, 9719906. [Google Scholar] [CrossRef]
- Albukhari, T.A.; Bagadood, R.M.; Bokhari, B.T.; Filimban, W.A.; Sembawa, H.; Nasreldin, N.; Gadalla, H.E.; El-Boshy, M.E. Chrysin attenuates gentamicin-induced renal injury in rats through modulation of oxidative damage and inflammation via regulation of Nrf2/AKT and NF-kB/KIM-1 pathways. Biomedicines 2025, 13, 271. [Google Scholar] [CrossRef]
- Hegazy, A.M.; Mosaed, M.M.; Elshafey, S.H.; Bayomy, N.A. 6-gingerol ameliorates gentamicin induced renal cortex oxidative stress and apoptosis in adult male albino rats. Tissue Cell 2016, 48, 208–216. [Google Scholar] [CrossRef]
- Laorodphun, P.; Cherngwelling, R.; Panya, A.; Arjinajarn, P. Curcumin protects rats against gentamicin-induced nephrotoxicity by amelioration of oxidative stress, endoplasmic reticulum stress and apoptosis. Pharm. Biol. 2022, 60, 491–500. [Google Scholar] [CrossRef]
- Ahmed, H.I.; Mohamed, E.A. Candesartan and epigallocatechin-3-gallate ameliorate gentamicin-induced renal damage in rats through p38-MAPK and NF-κB pathways. J. Biochem. Mol. Toxicol. 2019, 33, e22254. [Google Scholar] [CrossRef]
- Akcakavak, G.; Kazak, F.; Karatas, O.; Alakus, H.; Alakus, I.; Kirgiz, O.; Celik, Z.; Yilmaz Deveci, M.Z.; Ozdemir, O.; Tuzcu, M. Eucalyptol regulates Nrf2 and NF-kB signaling and alleviates gentamicin-induced kidney injury in rats by downregulating oxidative stress, oxidative DNA damage, inflammation, and apoptosis. Toxicol. Mech. Methods 2024, 34, 413–422. [Google Scholar] [CrossRef] [PubMed]
- Jado, J.C.; Humanes, B.; González-Nicolás, M.; Camaño, S.; Lara, J.M.; López, B.; Cercenado, E.; García-Bordas, J.; Tejedor, A.; Lázaro, A. Nephroprotective effect of cilastatin against gentamicin-induced renal injury in vitro and in vivo without altering its bactericidal efficiency. Antioxidants 2020, 9, 821. [Google Scholar] [CrossRef] [PubMed]
- Ali, F.E.M.; Sayed, A.M.; El-Bahrawy, A.H.; Omar, Z.M.M.; Hassanein, E.H.M. Targeting KEAP1/Nrf2, AKT, and PPAR-γ signals as a potential protective mechanism of diosmin against gentamicin-induced nephrotoxicity. Life Sci. 2021, 275, 119349. [Google Scholar] [CrossRef]
- Tomşa, A.M.; Răchişan, A.L.; Pandrea, S.L.; Benea, A.; Uifălean, A.; Toma, C.; Popa, R.; Pârvu, A.E.; Junie, L.M. Curcumin and vitamin C attenuate gentamicin-induced nephrotoxicity by modulating distinctive reactive species. Metabolites 2022, 13, 49. [Google Scholar] [CrossRef]
- Bledsoe, G.; Crickman, S.; Mao, J.; Xia, C.F.; Murakami, H.; Chao, L.; Chao, J. Kallikrein/kinin protects against gentamicin-induced nephrotoxicity by inhibition of inflammation and apoptosis. Nephrol. Dial. Transplant. 2006, 21, 624–633. [Google Scholar] [CrossRef]
- Kalayarasan, S.; Prabhu, P.N.; Sriram, N.; Manikandan, R.; Arumugam, M.; Sudhandiran, G. Diallyl sulfide enhances antioxidants and inhibits inflammation through the activation of Nrf2 against gentamicin-induced nephrotoxicity in Wistar rats. Eur. J. Pharmacol. 2009, 606, 162–171. [Google Scholar] [CrossRef]
- Cachofeiro, V.; Goicochea, M.; de Vinuesa, S.G.; Oubiña, P.; Lahera, V.; Luño, J. Oxidative stress and inflammation, a link between chronic kidney disease and cardiovascular disease. Kidney Int. Suppl. 2008, 74, S4–S9. [Google Scholar] [CrossRef]
- Maldonado, P.D.; Barrera, D.; Rivero, I.; Mata, R.; Medina-Campos, O.N.; Hernández-Pando, R.; Pedraza-Chaverrí, J. Antioxidant S-allylcysteine prevents gentamicin-induced oxidative stress and renal damage. Free Radic. Biol. Med. 2003, 35, 317–324. [Google Scholar] [CrossRef]
- Kadkhodaee, M.; Khastar, H.; Faghihi, M.; Ghaznavi, R.; Zahmatkesh, M. Effects of co-supplementation of vitamins E and C on gentamicin-induced nephrotoxicity in rat. Exp. Physiol. 2005, 90, 571–576. [Google Scholar] [CrossRef] [PubMed]
- Geenen, R.W.F.; Kingma, H.J.; van der Molen, A.J. Contrast-induced nephropathy: Pharmacology, pathophysiology and prevention. Insights Imaging 2013, 4, 811–820. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, J. Contrast-induced acute kidney injury: A review of definition, pathogenesis, risk factors, prevention and treatment. BMC Nephrol. 2024, 25, 140. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, S.; Koyasu, S. Mechanisms of action of cyclosporine. Immunopharmacology 2000, 47, 119–125. [Google Scholar] [CrossRef]
- Abd-Eldayem, A.M.; Makram, S.M.; Messiha, B.A.S.; Abd-Elhafeez, H.H.; Abdel-Reheim, M.A. Cyclosporine-induced kidney damage was halted by sitagliptin and hesperidin via increasing Nrf2 and suppressing TNF-α, NF-κB, and Bax. Sci. Rep. 2024, 14, 7434. [Google Scholar] [CrossRef]
- Yoon, H.E.; Yang, C.W. Established and newly proposed mechanisms of chronic cyclosporine nephropathy. Korean J. Intern. Med. 2009, 24, 81. [Google Scholar] [CrossRef] [PubMed]
- Modlinger, P.; Chabrashvili, T.; Gill, P.S.; Mendonca, M.; Harrison, D.G.; Griendling, K.K.; Li, M.; Raggio, J.; Wellstein, A.; Chen, Y.; et al. RNA silencing in vivo reveals role of p22phox in rat angiotensin slow pressor response. Hypertension 2006, 47, 238–244. [Google Scholar] [CrossRef]
- De Keulenaer, W.G.; Alexander, R.W.; Ushio-Fukai, M.; Ishizaka, N.; Griendling, K.K. Tumour necrosis factor alpha activates a p22phox-based NADH oxidase in vascular smooth muscle. Biochem. J. 1998, 329, 653–657. [Google Scholar] [CrossRef]
- Brandes, R.P.; Viedt, C.; Nguyen, K.; Beer, S.; Kreuzer, J.; Busse, R.; Görlach, A. Thrombin-induced MCP-1 expression involves activation of the p22phox-containing NADPH oxidase in human vascular smooth muscle cells. Thromb. Haemost. 2001, 85, 1104–1110. [Google Scholar] [PubMed]
- Griendling, K.K. NADPH oxidases: New regulators of old functions. Antioxid. Redox Signal. 2006, 8, 1443–1445. [Google Scholar] [CrossRef]
- Nankivell, B.J.; Borrows, R.J.; Fung, C.L.; O’Connell, P.J.; Allen, R.D.; Chapman, J.R. The natural history of chronic allograft nephropathy. N. Engl. J. Med. 2003, 349, 2326–2333. [Google Scholar] [CrossRef]
- Djamali, A.; Wilson, N.A.; Sadowski, E.A.; Zha, W.; Niles, D.; Hafez, O.; Dorn, J.R.; Mehner, T.R.; Grimm, P.C.; Hoffmann, F.M.; et al. Nox2 and cyclosporine-induced renal hypoxia. Transplantation 2016, 100, 1198–1210. [Google Scholar] [CrossRef]
- Vetter, M.; Chen, Z.J.; Chang, G.D.; Che, D.; Liu, S.; Chang, C.H. Cyclosporin A disrupts bradykinin signaling through superoxide. Hypertension 2003, 41, 1135–1141. [Google Scholar] [CrossRef]
- Ciarcia, R.; Damiano, S.; Florio, A.; Spagnuolo, M.; Zacchia, E.; Squillacioti, C.; Mirabella, N.; Florio, S.; Pagnini, U.; Garofano, T.; et al. The protective effect of apocynin on cyclosporine A-induced hypertension and nephrotoxicity in rats. J. Cell Biochem. 2015, 116, 1848–1856. [Google Scholar] [CrossRef] [PubMed]
- Elhelaly, A.E.; AlBasher, G.; Alfarraj, S.; Almeer, R.; Bahbah, E.I.; Fouda, M.M.A.; Bungău, S.G.; Aleya, L.; Abdel-Daim, M.M. Protective effects of hesperidin and diosmin against acrylamide-induced liver, kidney, and brain oxidative damage in rats. Environ. Sci. Pollut. Res. Int. 2019, 26, 35151–35162. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Cao, X.; Hu, X.; Li, S.; Wang, J. The anti-apoptotic, antioxidant and anti-inflammatory effects of curcumin on acrylamide-induced neurotoxicity in rats. BMC Pharmacol. Toxicol. 2020, 21, 62. [Google Scholar] [CrossRef] [PubMed]
- Song, D.; Xu, C.; Holck, A.L.; Liu, R. Acrylamide inhibits autophagy, induces apoptosis and alters cellular metabolic profiles. Ecotoxicol. Environ. Saf. 2021, 208, 111543. [Google Scholar] [CrossRef]
- Tanase, D.M.; Gosav, E.M.; Radu, S.; Costea, C.F.; Ciocoiu, M.; Carauleanu, A.; Lacatusu, C.M.; Maranduca, M.A.; Floria, M.; Rezus, C. The predictive role of the biomarker kidney molecule-1 (KIM-1) in acute kidney injury (AKI) cisplatin-induced nephrotoxicity. Int. J. Mol. Sci. 2019, 20, 5238. [Google Scholar] [CrossRef]
- Jin, H.Z.; Yang, X.J.; Zhao, K.L.; Mei, F.C.; Zhou, Y.; You, Y.D.; Wang, W.X. Apocynin alleviates lung injury by suppressing NLRP3 inflam masome activation and NF-κB signaling in acute pancreatitis. Int. Immunopharmacol. 2019, 75, 105821. [Google Scholar] [CrossRef]
- Xiang, L.; Lu, S.; Mittwede, P.N.; Clemmer, J.S.; Hester, R.L. Inhibition of NADPH oxidase prevents acute lung injury in obese rats following severe trauma. Am. J. Physiol. Heart Circ. Physiol. 2014, 306, H684–H689. [Google Scholar] [CrossRef]
- Schonfeld, P.; Wieckowski, M.R.; Lebiedzinska, M.; Wojtczak, L. Mitochondrial fatty acid oxidation and oxidative stress: Lack of reverse electron transfer-associated production of reactive oxygen species. Biochim. Biophys. Acta 2010, 1797, 929–938. [Google Scholar] [CrossRef]
- Ge, N.; Xia, Q.; Yang, Z.H.; Ding, Q.F.; Zeng, Z. Vascular endothelial injury and apoptosis in rats with severe acute pancreatitis. Gastroenterol. Res. Pract. 2015, 2015, 235017. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Wang, W.; Shi, Q.; Zhao, L.; Mei, F.; Li, C.; Zuo, T.; He, X. Paeoniflorin ameliorates acute necrotizing pancreatitis and pancreatitis-induced acute renal injury. Mol. Med. Rep. 2016, 14, 1123–1131. [Google Scholar] [CrossRef] [PubMed]
- Wan, C.; Su, H.; Zhang, C. Role of NADPH oxidase in metabolic disease-related renal injury: An update. Oxid. Med. Cell Longev. 2016, 2016, 7813072. [Google Scholar] [CrossRef] [PubMed]
- Kounatidis, D.; Tzivaki, I.; Daskalopoulou, S.; Daskou, A.; Adamou, A.; Rigatou, A.; Sdogkos, E.; Karampela, I.; Dalamaga, M.; Vallianou, N.G. Sepsis-Associated Acute Kidney Injury: What’s New Regarding Its Diagnostics and Therapeutics? Diagnostics 2024, 14, 2845. [Google Scholar] [CrossRef]
- Ow, C.P.C.; Trask-Marino, A.; Betrie, A.H.; Evans, R.G.; May, C.N.; Lankadeva, Y.R. Targeting oxidative stress in septic acute kidney injury: From theory to practice. J. Clin. Med. 2021, 10, 3798. [Google Scholar] [CrossRef]
- Nlandu-Khodo, S.; Dissard, R.; Hasler, U.; Schäfer, M.; Pircher, H.; Jansen-Durr, P.; Krause, K.H.; Martin, P.Y.; de Seigneux, S. NADPH oxidase 4 deficiency increases tubular cell death during acute ischemic reperfusion injury. Sci. Rep. 2016, 6, 38594. [Google Scholar] [CrossRef]
- Chen, H.C.; Hou, H.Y.; Sung, J.M.; Shieh, C.C. Deletion of NADPH oxidase 2 attenuates cisplatin-induced acute kidney injury through reducing ROS-induced proximal tubular cell injury and inflammation. Front. Med. 2023, 10, 1097671. [Google Scholar] [CrossRef]
- Karim, A.S.; Reese, S.R.; Wilson, N.A.; Jacobson, L.M.; Zhong, W.; Djamali, A. Nox2 is a mediator of ischemia reperfusion injury. Am. J. Transplant. 2015, 15, 2888–2899. [Google Scholar] [CrossRef]
- Bhatt, N.P.; Park, J.Y.; Lee, H.J.; Kim, S.S.; Kwon, Y.S.; Chun, W. Apocynin protects mesangial cells from lipopolysaccharide-induced inflammation by exerting heme oxygenase 1-mediated monocyte chemoattractant protein-1 suppression. Int. J. Mol. Med. 2017, 40, 1116–1124. [Google Scholar] [CrossRef]
- Shen, W.C.; Liang, C.J.; Huang, T.M.; Liu, C.W.; Wang, S.H.; Young, G.H.; Tsai, J.S.; Tseng, Y.C.; Peng, Y.S.; Wu, V.C.; et al. Indoxyl sulfate enhances IL-1β-induced E-selectin expression in endothelial cells in acute kidney injury by the ROS/MAPKs/NFκB/AP-1 pathway. Arch. Toxicol. 2016, 90, 2779–2792. [Google Scholar] [CrossRef]
- Lu, Q.B.; Du, Q.; Wang, H.P.; Tang, Z.H.; Wang, Y.B.; Sun, H.J. Salusin-β mediates tubular cell apoptosis in acute kidney injury: Involvement of the PKC/ROS signaling pathway. Redox Biol. 2020, 30, 101411. [Google Scholar] [CrossRef]
- Su, L.; Zhang, J.; Gomez, H.; Kellum, J.A.; Peng, Z. Mitochondria ROS and mitophagy in acute kidney injury. Autophagy 2023, 19, 401–414. [Google Scholar] [CrossRef]
- Chiang, C.H.; Chen, C.; Fang, S.Y.; Lin, S.C.; Chen, J.W.; Chang, T.T. Xanthine oxidase/NADPH oxidase inhibition by hydralazine attenuates acute kidney injury and prevents the transition of acute kidney injury to chronic kidney disease. Life Sci. 2023, 327, 121863. [Google Scholar] [CrossRef] [PubMed]
- Roe, N.D.; Ren, J. Nitric oxide synthase uncoupling: A therapeutic target in cardiovascular diseases. Vasc. Pharmacol. 2012, 57, 168–172. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.; Yuan, Y.; Yuan, F.; Li, X. Acute kidney injury: Exploring endoplasmic reticulum stress-mediated cell death. Front. Pharmacol. 2024, 15, 1308733. [Google Scholar] [CrossRef] [PubMed]
- Vermot, A.; Petit-Härtlein, I.; Smith, S.M.E.; Fieschi, F. NADPH Oxidases (NOX): An Overview from Discovery, Molecular Mechanisms to Physiology and Pathology. Antioxidants 2021, 10, 890. [Google Scholar] [CrossRef]
- Touyz, R.-M. Apocynin, NADPH oxidase, and vascular cells: A complex matter. Hypertension 2008, 51, 172–174. [Google Scholar] [CrossRef]

| Author | Experimental Protocol | Apocynin Protocol | Main Findings |
|---|---|---|---|
| Altintas et al., 2013 [37] | Ischaemia (60 min)-reperfusion Injury induced in Wistar albino rats | 30 min before ischemia, 20 mg/kg i.p. (one group) and 30 min before reperfusion, 20 mg/kg i.p. (other group) | APO applied 30 min before ischemia significantly decreased serum levels of creatinine and urea, decreased lipid peroxidation and MPO activity, increased GPx activity in kidney tissue, and improved renal morphology (evaluated by semiquantitative analysis). APO applied 30 min after ischemia also significantly decreased serum creatinine and urea, increased only GPx activity in kidney, improved renal morphology, and decreased caspase-3 positive cells in tubular immunostaining. |
| Li and Wang, 2015 [38] | Ischaemia (45 min)-reperfusion Injury induced in Sprague—Dawley rats | 30 min before reperfusion, 50 mg/kg i.p. | APO significantly decreased creatinine and urea serum levels, decreased lipid peroxidation and iNOS activity, improved SOD activity, increased NO levels, decreased TNF-α levels, decreased TLR4 and NF-κB, and increased SHP-1 expression in kidney tissue. |
| Choi et al., 2015 [39] | Ischaemia (30 min)-reperfusion Injury induced in Sprague—Dawley rats | 1 h before ischemia, 20 mg/kg i.p. | APO significantly decreased serum levels of creatinine and urea, decreased lipid peroxidation, and improved renal morphology (significant recovery in the tubular cells). |
| Hu et al., 2016 [40] | Ischaemia (45 min)-reperfusion Injury induced in Sprague—Dawley rats | 30 min before ischemia, 50 mg/kg i.p. | APO decreased serum creatinine and urea levels, increased serum and kidney tissue zinc levels, increased metallothionen expression, decreased lipid peroxidation and increased SOD activity, increased IL-4 and IL-10, and decreased IL-6 and TNF-α levels in kidney tissue. |
| Kovacevic et al., 2020 [41] | Ischemia (45 min)-reperfusion injury induced in Spontaneously hypertensive rats | 5 min before reperfusion, 40 mg/kg i.v. | APO improved renal hemodynamics (increased RBF and decreased RVR), decreased creatinine, urea, and phosphate levels in plasma, and improved renal morphology (moderately intensive tubular necrosis, reduced tubular dilatation, and a smaller number of PAS-positive casts). |
| Lima et al., 2021 [42] | Ischaemia (45 min)-reperfusion Injury induced in Wistar albino rats | 24 h before and after ischemia, 100 mg/kg in drinking water (acute treatment), 24 h after ischemia, 100 mg/kg in drinking water, 4 weeks (chronic treatment) | APO reduced creatinine and urea serum levels 24 h after reperfusion. Acute APO treatment decreased lipid peroxidation, superoxide anion production, NOX oxidase activity, ouabain-sensitive Na+-K+-ATPase activity, AT1 and AT2 receptor expression and increased furosemide-sensitive, ouabain-resistant Na+-K+-ATPase activity, ACE 1 and ACE 2 activity, and AT2 receptor expression in kidney tissue 4 weeks after AKI induction. Chronic APO treatment also decreased lipid peroxidation, superoxide anion production, NOX oxidase activity, and AT1 receptor expression, and increased furosemide-sensitive, ouabain-resistant Na+-K+-ATPase activity, AT2 receptor expression, and ACE 1 and PKCλ activity in kidney tissue 4 weeks after AKI induction. |
| Kovacevic et al., 2021 [43] | Ischaemia (45 min)-reperfusion injury induced in Spontaneously hypertensive rats | 5 min before reperfusion, 40 mg/kg i.v. 2 sessions | APO improved clearances of urea, creatinine, and phosphate, decreased expression of 4-HNE and NGAL, and provoked different HO-1 immunohistochemical expression patterns in kidney tissue. |
| Revesz et al., 2024 [44] | Ischaemia (15, 20 or 30 min)-reperfusion Injury induced in C57BL/6 mice | 15 min before (i.v.), immediately after (i.p.), and 120 min after (i.p.) ischemia, 10 mg/kg i.p. | APO decreased urea and NGAL levels in plasma, increased NRF2, HO-1, and GPx3, and decreased NGAL, TNF-α, and F4/80 mRNA expression in kidney tissue and improved renal morphology (decreased ATN score) after mild (15 min) ischemia. APO did not have beneficial effects after moderate (20 min) or severe (30 min) ischemia. |
| Author | Experimental Protocol | Apocynin Protocol | Main Findings |
|---|---|---|---|
| Chirino et al., 2008 [45] | Cisplatin-induced nephrotoxicity in Wistar albino rats | 7 days before and 3 days after single i.p. cisplatin injection (7.5 mg/kg), 2 g/L in drinking water. | APO significantly decreased serum levels of creatinine and urea, proteinuria and urinary GST and NAG excretion, decreased lipid peroxidation, immunohistochemical expression of 4-HNE and 3-NT in kidney tissue, and improved renal morphology (nearly regular morphology of epithelial tubular cells with significant reduction of cast formation of 85% in renal cortex and of 98% in renal medulla). |
| Wang et al., 2015 [46] | Cisplatin-induced nephrotoxicity in C56BL/6 mice | 2 h before and 2 days after single i.p. cisplatin injection (20 mg/kg), 10 mg/kg i.p. | APO significantly decreased creatinine and urea in serum, improved kidney morphology (necrosis, protein cast, vacuolation, and desquamation of epithelial cells in the renal tubules was significantly attenuated, up to 40%), decreased oxidative stress (3-NT, 4-HNE), NOX activity, decreased inflammation (decreased TNF-α, IL-1β expression and MPO activity), and decreased apoptosis (decreased cleaved caspase 3, DNA fragmentation, TUNEL + cells) in kidney tissue. |
| Meng et al., 2017 [47] | Cisplatin-induced nephrotoxicity in mice | 1 day before single i.p. cisplatin injection (20 mg/kg), 100 mg/kg i.p. | APO significantly decreased creatinine and urea in serum, decreased lipid peroxidation, NOX 4 protein expression, KIM-1 expression, decreased inflammation (decreased TNF-α, IL-1β and IL-6 expression), decreased RIPK1, RIPK3, P-MLKL and cleaved caspase 3 in kidney tissue, and improved renal morphology (tubular necrosis, cast formation and tubular dilatation significantly reduced). |
| Abdelrahman, 2017 [48] | Gentamicin-induced nephrotoxicity in Wistar albino rats | 7 days before and 7 days along with i.p. gentamicin injection (100 mg/kg, 7 days), 10 mg/kg i.p. | APO decreased serum creatinine and urea levels, increased creatinine clarence, decreased proteinuria, decreased lipid peroxidation and NO content, increased SOD activity, decreased CD95 in kidney tissue, and improved renal morphology (normal renal tubular epithelium lining renal tubules). |
| Tan et al., 2020 [49] | Cyclosporine A-induced nephrotoxicity in Wistar-Kyoto rats | 14 days along with cyclosporine-A administration (25 mg/kg/day, via gavage), 2.5 mmol/L/day orally | APO decreased plasma creatinine and urea, increased creatinine clarence, decreased urine output, fractional excretion of sodium, urinary sodium/potassium ratio, decreased BUN and proteinuria, improved renal cortical blood perfusion, decreased lipid peroxidation, increased SOD activity and total antioxidant capacity, decreased NOX 4 mRNA expression in kidney tissue, and improved renal morphology (no severe renal tubular ischemia, no abscesses in renal interstitial area, normal glomerular apparatus, no neutrophils). |
| Ahmad et al., 2012 [50] | Contrast-induced nephrotoxicity in diabetic Wistar albino rats | 30 min before i.v. iomeprol injection (10 ml/kg), 5 mg/kg i.v. | APO decreased plasma creatinine and urea, increased creatinine clarence, decreased plasma and urinary NGAL, decreased urinary αGST, decreased immunohistochemical 3-NT, TNF-α, IL-1β expression, PARP activation and number of apoptotic calls or fragments in kidney tissue, and improved renal morphology (significantly decreased histological score). |
| Ageena et al., 2021 [51] | Acrylamide-induced nephrotoxicity in Wistar albino rats | 10 days along with acrylamide administration (40 mg/kg/day, i.p.), 100 mg/kg orally | APO decreased serum creatinine, urea, uric acid, potassium, magnesium, and KIM-1, decreased lipid peroxidation, increased GSH and SOD levels, increased NRF-2 and HO-1 expression, decreased TNF-α, caspase 1, IL-1β, GSDMD, ASC, and NLRP3 expression in kidney tissue, with mild improvement in renal morphology (moderate damage to proximal and distal convoluted tubules, moderate hyperemia of the capillary tufts and blood vessels, and moderate interstitial hemorrhage). |
| Mittwede et al., 2015 [52] | Orthopedic trauma-induced acute kidney injury in obese Zucker rats | Immediately after trauma, 50 mg/kg, i.p., and during next 24 h in drinking water, 2 mmol. | APO decreased creatinine levels in plasma, improved GFR, decreased urinary KIM-1, albumin excretion, and urinary lipid peroxidation, decreased NOX activity and IL-6 in kidney tissue. |
| Yang et al., 2020 [53] | Acute hypertriglyceridemic pancreatitis- related acute kidney injury induced in Sprague–Dawley rats | 30 min before induction, 50 mg/kg, subcutaneously | APO significantly decreased creatinine and urea serum levels, decreased NOX2, NOX4 expression, ROS levels, TNF-α, NF-κβ, GSK-3β and increased p-AKT expression, reduced MPO and CD68 positive cells, decreased apoptotic index, and improved renal morphology (histopathological score was significantly reduced). |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nesovic Ostojic, J.; Kovacevic, S.; De Luka, S.R.; Ivanov, M.; Nenadovic, A.; Vukovic, A. Therapeutic Potential of Apocynin: A Promising Antioxidant Strategy for Acute Kidney Injury. Antioxidants 2025, 14, 1025. https://doi.org/10.3390/antiox14081025
Nesovic Ostojic J, Kovacevic S, De Luka SR, Ivanov M, Nenadovic A, Vukovic A. Therapeutic Potential of Apocynin: A Promising Antioxidant Strategy for Acute Kidney Injury. Antioxidants. 2025; 14(8):1025. https://doi.org/10.3390/antiox14081025
Chicago/Turabian StyleNesovic Ostojic, Jelena, Sanjin Kovacevic, Silvio R. De Luka, Milan Ivanov, Aleksandra Nenadovic, and Andrija Vukovic. 2025. "Therapeutic Potential of Apocynin: A Promising Antioxidant Strategy for Acute Kidney Injury" Antioxidants 14, no. 8: 1025. https://doi.org/10.3390/antiox14081025
APA StyleNesovic Ostojic, J., Kovacevic, S., De Luka, S. R., Ivanov, M., Nenadovic, A., & Vukovic, A. (2025). Therapeutic Potential of Apocynin: A Promising Antioxidant Strategy for Acute Kidney Injury. Antioxidants, 14(8), 1025. https://doi.org/10.3390/antiox14081025








