Antioxidant Nanohybrid Materials Derived via Olive Leaf Extract Incorporation in Layered Double Hydroxide: Preparation, Characterization, and Evaluation for Applications
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Enzymatic-Assisted Extraction
2.3. OLE@LDH_Zn/Al Nanohybrids’ Preparation
2.4. Phytochemical Analyses of OLE
2.5. Instrumental Structural Analysis of OLE@LDH_Zn/Al_x/1 Nanohybrids
2.6. EC50 Estimation of OLE@LDH_Zn/Al_x/1 Nanohybrids
2.6.1. In Vitro Antioxidant Activity Determination of Pure OLE and Obtained OLE@LDH_Zn/Al_x/1 Nanohybrids via the 2,2-Diphenyl-1-picrylhydrazyl (DPPH) Assay Method
2.6.2. Antioxidant Activity of O OLE@LDH_Zn/Al_x/1 Nanohybrids with the 2,2′-Azino-bis(3-ethylbenzothiazoline-6-sulfonic Acid) Diammonium Salt (ABTS) Assay
2.6.3. Antioxidant Activity of O OLE@LDH_Zn/Al_x/1 Nanohybrids with the Ferric Reducing Antioxidant Power (FRAP) Assay
2.7. Total Polyphenol Content (TPC) of OLE@LDH_Zn/Al_x/1 Nanohybrids
2.8. Antibacterial Activity
2.8.1. Antimicrobial Activity of OLE@LDH_Zn/Al_x/1 Nanohybrids
2.8.2. Disk Diffusion Susceptibility Test
2.8.3. Resazurin-Based 96-Well Plate Microdilution Method
2.9. Statistical Analysis
3. Results
3.1. HPLC-DAD Analyses of OLE
3.2. Physicochemical Characterization of OLE@LDH_Zn/Al_x/1 Nanohybrids
3.2.1. XRD Analysis of OLE@LDH_Zn/Al_x/1 Nanohybrids
3.2.2. FTIR of OLE@LDH_Zn/Al_x/1 Nanohybrids
3.2.3. HR-SEM Analysis of OLE@LDH_Zn/Al_x/1 Nanohybrids
3.3. Antioxidant Activity of OLE@LDH_Zn/Al_x/1 Nanohybrids
3.3.1. EC50 Estimation
3.3.2. Total Phenolic Content (TPC) Estimation
3.4. Antibacterial Activity of OLE@LDH_Zn/Al_x/1 Nanohybrids
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cooney, R.; de Sousa, D.B.; Fernández-Ríos, A.; Mellett, S.; Rowan, N.; Morse, A.P.; Hayes, M.; Laso, J.; Regueiro, L.; Wan, A.H.L.; et al. A Circular Economy Framework for Seafood Waste Valorisation to Meet Challenges and Opportunities for Intensive Production and Sustainability. J. Clean. Prod. 2023, 392, 136283. [Google Scholar] [CrossRef]
- Esposito, B.; Sessa, M.R.; Sica, D.; Malandrino, O. Towards Circular Economy in the Agri-Food Sector. A Systematic Literature Review. Sustainability 2020, 12, 7401. [Google Scholar] [CrossRef]
- Hamam, M.; Chinnici, G.; Di Vita, G.; Pappalardo, G.; Pecorino, B.; Maesano, G.; D’Amico, M. Circular Economy Models in Agro-Food Systems: A Review. Sustainability 2021, 13, 3453. [Google Scholar] [CrossRef]
- Panáček, D.; Zdražil, L.; Langer, M.; Šedajová, V.; Baďura, Z.; Zoppellaro, G.; Yang, Q.; Nguyen, E.P.; Álvarez-Diduk, R.; Hrubý, V.; et al. Graphene Nanobeacons with High-Affinity Pockets for Combined, Selective, and Effective Decontamination and Reagentless Detection of Heavy Metals. Small 2022, 18, 2201003. [Google Scholar] [CrossRef]
- Rontogianni, A.; Chalmpes, Ν.; Nikolaraki, E.; Botzolaki, G.; Androulakis, A.; Stratakis, A.; Zygouri, P.; Moschovas, D.; Avgeropoulos, A.; Karakassides, M.A.; et al. Efficient CO2 Hydrogenation over Mono- and Bi-Metallic RuNi/MCM-41 Catalysts: Controlling CH4 and CO Products Distribution through the Preparation Method and/or Partial Replacement of Ni by Ru. Chem. Eng. J. 2023, 474, 145644. [Google Scholar] [CrossRef]
- Cansado, I.P.d.P.; Mourão, P.A.M.; Castanheiro, J.E.; Geraldo, P.F.; Suhas; Suero, S.R.; Cano, B.L. A Review of the Biomass Valorization Hierarchy. Sustainability 2025, 17, 335. [Google Scholar] [CrossRef]
- Joshi, N.C.; Sinha, S.; Bhatnagar, P.; Nath, Y.; Negi, B.; Kumar, V.; Gururani, P. A Concise Review on Waste Biomass Valorization through Thermochemical Conversion. Curr. Res. Microb. Sci. 2024, 6, 100237. [Google Scholar] [CrossRef]
- Areti, H.A.; Muleta, M.D.; Abo, L.D.; Hamda, A.S.; Adugna, A.A.; Edae, I.T.; Daba, B.J.; Gudeta, R.L. Innovative Uses of Agricultural By-Products in the Food and Beverage Sector: A Review. Food Chem. Adv. 2024, 5, 100838. [Google Scholar] [CrossRef]
- Lai, W.T.; Khong, N.M.H.; Lim, S.S.; Hee, Y.Y.; Sim, B.I.; Lau, K.Y.; Lai, O.M. A Review: Modified Agricultural by-Products for the Development and Fortification of Food Products and Nutraceuticals. Trends Food Sci. Technol. 2017, 59, 148–160. [Google Scholar] [CrossRef]
- Ruff, A.J. Food Industry Side Streams: An Unexploited Source for Biotechnological Phosphorus Upcycling. Curr. Opin. Biotechnol. 2024, 90, 103209. [Google Scholar] [CrossRef]
- Salvatore, I.; Leue-Rüegg, R.; Beretta, C.; Müller, N. Valorisation Potential and Challenges of Food Side Product Streams for Food Applications: A Review Using the Example of Switzerland. Future Foods 2024, 9, 100325. [Google Scholar] [CrossRef]
- Chalmpes, N.; Tantis, I.; Alsmaeil, A.W.; Aldakkan, B.S.; Dimitrakou, A.; Karakassides, M.A.; Salmas, C.E.; Giannelis, E.P. Elevating Waste Biomass: Supercapacitor Electrode Materials Derived from Spent Coffee Grounds. Energy Fuels 2025, 39, 1305–1315. [Google Scholar] [CrossRef]
- Guillard, V.; Gaucel, S.; Fornaciari, C.; Angellier-Coussy, H.; Buche, P.; Gontard, N. The Next Generation of Sustainable Food Packaging to Preserve Our Environment in a Circular Economy Context. Front. Nutr. 2018, 5, 121. [Google Scholar] [CrossRef]
- Gupta, S.; Sharma, S.; Kumar Nadda, A.; Saad Bala Husain, M.; Gupta, A. Biopolymers from Waste Biomass and Its Applications in the Cosmetic Industry: A Review. Mater. Today Proc. 2022, 68, 873–879. [Google Scholar] [CrossRef]
- Al-Maqtari, Q.A.; Rehman, A.; Mahdi, A.A.; Al-Ansi, W.; Wei, M.; Yanyu, Z.; Phyo, H.M.; Galeboe, O.; Yao, W. Application of Essential Oils as Preservatives in Food Systems: Challenges and Future Prospectives—A Review. Phytochem. Rev. 2022, 21, 1209–1246. [Google Scholar] [CrossRef]
- Almeida-Souza, F.; Magalhães, I.F.B.; Guedes, A.C.; Santana, V.M.; Teles, A.M.; Mouchrek, A.N.; Calabrese, K.S.; Abreu-Silva, A.L. Safety Assessment of Essential Oil as a Food Ingredient. In Essential Oils: Applications and Trends in Food Science and Technology; Santana de Oliveira, M., Ed.; Springer International Publishing: Cham, Switzerland, 2022; pp. 123–171. ISBN 978-3-030-99476-1. [Google Scholar]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological Effects of Essential Oils—A Review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef] [PubMed]
- Salas, E.; Oliveira, J.; Perez-Gregorio, R. Editorial: Natural Extracts as Food Ingredients: From Chemistry to Health. Front. Nutr. 2023, 10, 1306307. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Sun, H.; Weng, Y. Natural Extracts and Their Applications in Polymer-Based Active Packaging: A Review. Polymers 2024, 16, 625. [Google Scholar] [CrossRef]
- Giannakas, A.E. Plant Extracts-Based Food Packaging Films. In Natural Materials for Food Packaging Application; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2023; pp. 23–49. ISBN 978-3-527-83730-4. [Google Scholar]
- Giannakas, A.E. 7-Bionanocomposites with Hybrid Nanomaterials for Food Packaging Applications. In Advances in Biocomposites and their Applications; Karak, N., Ed.; Woodhead Publishing Series in Composites Science and Engineering; Woodhead Publishing: Cambridge, UK, 2024; pp. 201–225. ISBN 978-0-443-19074-2. [Google Scholar]
- Deshmukh, R.K.; Hakim, L.; Akhila, K.; Ramakanth, D.; Gaikwad, K.K. Nano Clays and Its Composites for Food Packaging Applications. Int. Nano Lett. 2023, 13, 131–153. [Google Scholar] [CrossRef]
- Blinka, T.A.; Edwards, F.B.; Miranda, N.R.; Speer, D.V.; Thomas, J.A. Zeolite in Packaging Film. U.S. Patent 5834079A, 10 November 1998. [Google Scholar]
- Salmas, C.E.; Giannakas, A.E.; Karabagias, V.K.; Moschovas, D.; Karabagias, I.K.; Gioti, C.; Georgopoulos, S.; Leontiou, A.; Kehayias, G.; Avgeropoulos, A.; et al. Development and Evaluation of a Novel-Thymol@Natural-Zeolite/Low-Density-Polyethylene Active Packaging Film: Applications for Pork Fillets Preservation. Antioxidants 2023, 12, 523. [Google Scholar] [CrossRef]
- Kumari, S.; Soni, S.; Sharma, A.; Kumar, S.; Sharma, V.; Jaswal, V.S.; Bhatia, S.K.; Sharma, A.K. Layered Double Hydroxides Based Composite Materials and Their Applications in Food Packaging. Appl. Clay Sci. 2024, 247, 107216. [Google Scholar] [CrossRef]
- Daniel, S.; Thomas, S. 1-Layered Double Hydroxides: Fundamentals to Applications. In Layered Double Hydroxide Polymer Nanocomposites; Thomas, S., Daniel, S., Eds.; Woodhead Publishing Series in Composites Science and Engineering; Woodhead Publishing: Cambridge, UK, 2020; pp. 1–76. ISBN 978-0-08-102261-0. [Google Scholar]
- Alexandre, M.; Dubois, P. Polymer-Layered Silicate Nanocomposites: Preparation, Properties and Uses of a New Class of Materials. Mater. Sci. Eng. R Rep. 2000, 28, 1–63. [Google Scholar] [CrossRef]
- Chalmpes, N.; Kouloumpis, A.; Zygouri, P.; Karouta, N.; Spyrou, K.; Stathi, P.; Tsoufis, T.; Georgakilas, V.; Gournis, D.; Rudolf, P. Layer-by-Layer Assembly of Clay–Carbon Nanotube Hybrid Superstructures. ACS Omega 2019, 4, 18100–18107. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, L.H.; Trigueiro, P.; Souza, J.S.N.; de Carvalho, M.S.; Osajima, J.A.; da Silva-Filho, E.C.; Fonseca, M.G. Montmorillonite with Essential Oils as Antimicrobial Agents, Packaging, Repellents, and Insecticides: An Overview. Colloids Surf. B Biointerfaces 2022, 209, 112186. [Google Scholar] [CrossRef] [PubMed]
- Deshmukh, R.K.; Kumar, L.; Gaikwad, K.K. Halloysite Nanotubes for Food Packaging Application: A Review. Appl. Clay Sci. 2023, 234, 106856. [Google Scholar] [CrossRef]
- Singha Roy, A.; Kesavan Pillai, S.; Ray, S.S. Layered Double Hydroxides for Sustainable Agriculture and Environment: An Overview. ACS Omega 2022, 7, 20428–20440. [Google Scholar] [CrossRef]
- Ghotbi, M.Y.; Hussein, M.Z.B.; Yahaya, A.H.; Rahman, M.Z.A. LDH-Intercalated d-Gluconate: Generation of a New Food Additive-Inorganic Nanohybrid Compound. J. Phys. Chem. Solids 2009, 70, 948–954. [Google Scholar] [CrossRef]
- Bugatti, V.; Bernardo, P.; Clarizia, G.; Viscusi, G.; Vertuccio, L.; Gorrasi, G. Ball Milling to Produce Composites Based of Natural Clinoptilolite as a Carrier of Salicylate in Bio-Based PA11. Polymers 2019, 11, 634. [Google Scholar] [CrossRef]
- Mondal, S.; Dasgupta, S.; Maji, K. MgAl- Layered Double Hydroxide Nanoparticles for Controlled Release of Salicylate. Mater. Sci. Eng. C 2016, 68, 557–564. [Google Scholar] [CrossRef]
- Aisawa, S.; Higashiyama, N.; Takahashi, S.; Hirahara, H.; Ikematsu, D.; Kondo, H.; Nakayama, H.; Narita, E. Intercalation Behavior of L-Ascorbic Acid into Layered Double Hydroxides. Appl. Clay Sci. 2007, 35, 146–154. [Google Scholar] [CrossRef]
- Rahmanian, O.; Dinari, M.; Neamati, S. Synthesis and Characterization of Citrate Intercalated Layered Double Hydroxide as a Green Adsorbent for Ni2+ and Pb2+ Removal. Environ. Sci. Pollut. Res. 2018, 25, 36267–36277. [Google Scholar] [CrossRef] [PubMed]
- Quintieri, L.; Bugatti, V.; Caputo, L.; Vertuccio, L.; Gorrasi, G. A Food-Grade Resin with LDH–Salicylate to Extend Mozzarella Cheese Shelf Life. Processes 2021, 9, 884. [Google Scholar] [CrossRef]
- Bugatti, V.; Vertuccio, L.; Zuppardi, F.; Vittoria, V.; Gorrasi, G. PET and Active Coating Based on a LDH Nanofiller Hosting P-Hydroxybenzoate and Food-Grade Zeolites: Evaluation of Antimicrobial Activity of Packaging and Shelf Life of Red Meat. Nanomaterials 2019, 9, 1727. [Google Scholar] [CrossRef]
- Viscusi, G.; Bugatti, V.; Vittoria, V.; Gorrasi, G. Antimicrobial Sorbate Anchored to Layered Double Hydroxide (LDH) Nano-Carrier Employed as Active Coating on Polypropylene (PP) Packaging: Application to Bread Stored at Ambient Temperature. Future Foods 2021, 4, 100063. [Google Scholar] [CrossRef]
- Bugatti, V.; Vertuccio, L.; Zara, S.; Fancello, F.; Scanu, B.; Gorrasi, G. Green Pesticides Based on Cinnamate Anion Incorporated in Layered Double Hydroxides and Dispersed in Pectin Matrix. Carbohydr. Polym. 2019, 209, 356–362. [Google Scholar] [CrossRef]
- Nalawade, P.; Aware, B.; Kadam, V.; Hirlekar, R. Layered Double Hydroxides: A Review. J. Sci. Ind. Res. 2009, 68, 267–272. [Google Scholar]
- Mir-Cerdà, A.; Granados, M.; Saurina, J.; Sentellas, S. Olive Tree Leaves as a Great Source of Phenolic Compounds: Comprehensive Profiling of NaDES Extracts. Food Chem. 2024, 456, 140042. [Google Scholar] [CrossRef] [PubMed]
- Debs, E.; Abi-Khattar, A.-M.; Rajha, H.N.; Abdel-Massih, R.M.; Assaf, J.-C.; Koubaa, M.; Maroun, R.G.; Louka, N. Valorization of Olive Leaves through Polyphenol Recovery Using Innovative Pretreatments and Extraction Techniques: An Updated Review. Separations 2023, 10, 587. [Google Scholar] [CrossRef]
- Vardakas, A.; Kechagias, A.; Penov, N.; Giannakas, A.E. Optimization of Enzymatic Assisted Extraction of Bioactive Compounds from Olea Europaea Leaves. Biomass 2024, 4, 647–657. [Google Scholar] [CrossRef]
- Vardakas, A.T.; Shikov, V.T.; Dinkova, R.H.; Mihalev, K.M. Optimisation of the Enzyme-Assisted Extraction of Polyphenols from Saffron (Crocus sativus L.) Petals. Acta Sci. Pol. Technol. Aliment. 2021, 20, 359–367. [Google Scholar] [CrossRef]
- IOC Standards, Methods and Guides, Int. Olive Counc. Determination of Biophenols in Olive Oils by HPLC. 2017. Available online: https://www.internationaloliveoil.org/wp-content/uploads/2019/11/COI-T.20-Doc.-No-29-Rev-1-2017.pdf (accessed on 1 May 2025).
- Kechagias, A.; Salmas, C.E.; Chalmpes, N.; Leontiou, A.A.; Karakassides, M.A.; Giannelis, E.P.; Giannakas, A.E. Laponite vs. Montmorillonite as Eugenol Nanocarriers for Low Density Polyethylene Active Packaging Films. Nanomaterials 2024, 14, 1938. [Google Scholar] [CrossRef] [PubMed]
- Elshikh, M.; Ahmed, S.; Funston, S.; Dunlop, P.; McGaw, M.; Marchant, R.; Banat, I.M. Resazurin-Based 96-Well Plate Microdilution Method for the Determination of Minimum Inhibitory Concentration of Biosurfactants. Biotechnol. Lett. 2016, 38, 1015–1019. [Google Scholar] [CrossRef]
- Boukhalfa, N.; Boutahala, M.; Djebri, N. Synthesis and Characterization of ZnAl-Layered Double Hydroxide and Organo-K10 Montmorillonite for the Removal of Diclofenac from Aqueous Solution. Adsorpt. Sci. Technol. 2017, 35, 20–36. [Google Scholar] [CrossRef]
- Ahmed, A.A.A.; Talib, Z.A.; Hussein, M.Z. bin Thermal, Optical and Dielectric Properties of Zn–Al Layered Double Hydroxide. Appl. Clay Sci. 2012, 56, 68–76. [Google Scholar] [CrossRef]
- Smalenskaite, A.; Pavasaryte, L.; Yang, T.C.K.; Kareiva, A. Undoped and Eu3+ Doped Magnesium-Aluminium Layered Double Hydroxides: Peculiarities of Intercalation of Organic Anions and Investigation of Luminescence Properties. Materials 2019, 12, 736. [Google Scholar] [CrossRef]
- Khan, A.I.; O’Hare, D. Intercalation Chemistry of Layered Double Hydroxides: Recent Developments and Applications. J. Mater. Chem. 2002, 12, 3191–3198. [Google Scholar] [CrossRef]
- Agatonovic-Kustrin, S.; Gegechkori, V.; Petrovich, D.S.; Ilinichna, K.T.; Morton, D.W. HPTLC and FTIR Fingerprinting of Olive Leaves Extracts and ATR-FTIR Characterisation of Major Flavonoids and Polyphenolics. Molecules 2021, 26, 6892. [Google Scholar] [CrossRef] [PubMed]
- Nasir, G.A.; Mohammed, A.K.; Samir, H.F. Biosynthesis and Characterization of Silver Nanoparticles Using Olive Leaves Extract and Sorbitol. Iraqi J. Biotechnol. 2016, 15, 1. [Google Scholar]
- Chalmpes, N.; Bourlinos, A.B.; Talande, S.; Bakandritsos, A.; Moschovas, D.; Avgeropoulos, A.; Karakassides, M.A.; Gournis, D. Nanocarbon from Rocket Fuel Waste: The Case of Furfuryl Alcohol-Fuming Nitric Acid Hypergolic Pair. Nanomaterials 2021, 11, 1. [Google Scholar] [CrossRef] [PubMed]
- Kouloumpis, A.; Vourdas, N.; Zygouri, P.; Chalmpes, N.; Potsi, G.; Kostas, V.; Spyrou, K.; Stathopoulos, V.N.; Gournis, D.; Rudolf, P. Controlled Deposition of Fullerene Derivatives within a Graphene Template by Means of a Modified Langmuir-Schaefer Method. J. Colloid Interface Sci. 2018, 524, 388–398. [Google Scholar] [CrossRef]
- Shameli, K.; Ahmad, M.B.; Jazayeri, S.D.; Shabanzadeh, P.; Sangpour, P.; Jahangirian, H.; Gharayebi, Y. Investigation of Antibacterial Properties Silver Nanoparticles Prepared via Green Method. Chem. Cent. J. 2012, 6, 73. [Google Scholar] [CrossRef]
- Khalil, M.M.H.; Ismail, E.H.; El-Baghdady, K.Z.; Mohamed, D. Green Synthesis of Silver Nanoparticles Using Olive Leaf Extract and Its Antibacterial Activity. Arab. J. Chem. 2014, 7, 1131–1139. [Google Scholar] [CrossRef]
- Chalmpes, N.; Patila, M.; Kouloumpis, A.; Alatzoglou, C.; Spyrou, K.; Subrati, M.; Polydera, A.C.; Bourlinos, A.B.; Stamatis, H.; Gournis, D. Graphene Oxide–Cytochrome c Multilayered Structures for Biocatalytic Applications: Decrypting the Role of Surfactant in Langmuir–Schaefer Layer Deposition. ACS Appl. Mater. Interfaces 2022, 14, 26204–26215. [Google Scholar] [CrossRef]
- Chalmpes, N.; Moschovas, D.; Tantis, I.; Bourlinos, A.B.; Bakandritsos, A.; Fotiadou, R.; Patila, M.; Stamatis, H.; Avgeropoulos, A.; Karakassides, M.A.; et al. Carbon Nanostructures Derived through Hypergolic Reaction of Conductive Polymers with Fuming Nitric Acid at Ambient Conditions. Molecules 2021, 26, 1595. [Google Scholar] [CrossRef]
- Mahjoubi, F.Z.; Khalidi, A.; Abdennouri, M.; Barka, N. Zn–Al Layered Double Hydroxides Intercalated with Carbonate, Nitrate, Chloride and Sulphate Ions: Synthesis, Characterisation and Dye Removal Properties. J. Taibah Univ. Sci. 2017, 11, 90–100. [Google Scholar] [CrossRef]
- Chen, X.; Li, H.; Xu, J.; Jaber, F.; Musharavati, F.; Zalnezhad, E.; Bae, S.; Hui, K.S.; Hui, K.N.; Liu, J. Synthesis and Characterization of a NiCo2O4@NiCo2O4 Hierarchical Mesoporous Nanoflake Electrode for Supercapacitor Applications. Nanomaterials 2020, 10, 1292. [Google Scholar] [CrossRef]
- Hui, L.; Xue, Y.; Huang, B.; Yu, H.; Zhang, C.; Zhang, D.; Jia, D.; Zhao, Y.; Li, Y.; Liu, H.; et al. Overall Water Splitting by Graphdiyne-Exfoliated and -Sandwiched Layered Double-Hydroxide Nanosheet Arrays. Nat. Commun. 2018, 9, 5309. [Google Scholar] [CrossRef]
- Wang, Q.; O’Hare, D. Recent Advances in the Synthesis and Application of Layered Double Hydroxide (LDH) Nanosheets. Chem. Rev. 2012, 112, 4124–4155. [Google Scholar] [CrossRef] [PubMed]
- Mohanty, U.A.; Sahoo, D.P.; Paramanik, L.; Parida, K. A Critical Review on Layered Double Hydroxide (LDH)-Derived Functional Nanomaterials as Potential and Sustainable Photocatalysts. Sustain. Energy Fuels 2023, 7, 1145–1186. [Google Scholar] [CrossRef]
- Ahmed, A.A.A.; Talib, Z.A.; bin Hussein, M.Z.; Zakaria, A. Zn–Al Layered Double Hydroxide Prepared at Different Molar Ratios: Preparation, Characterization, Optical and Dielectric Properties. J. Solid State Chem. 2012, 191, 271–278. [Google Scholar] [CrossRef]
- Jakobek, L.; Ištuk, J.; Barron, A.R.; Matić, P. Bioactive Phenolic Compounds from Apples during Simulated In Vitro Gastrointestinal Digestion: Kinetics of Their Release. Appl. Sci. 2023, 13, 8434. [Google Scholar] [CrossRef]
- Li, H.; Gao, Z.; Xu, J.; Sun, W.; Wu, J.; Zhu, L.; Gao, M.; Zhan, X. Encapsulation of Polyphenols in pH-Responsive Micelles Self-Assembled from Octenyl-Succinylated Curdlan Oligosaccharide and Its Effect on the Gut Microbiota. Colloids Surf. B Biointerfaces 2022, 219, 112857. [Google Scholar] [CrossRef] [PubMed]
- Tarchi, I.; Olewnik-Kruszkowska, E.; Aït-Kaddour, A.; Bouaziz, M. Innovative Process for the Recovery of Oleuropein-Rich Extract from Olive Leaves and Its Biological Activities: Encapsulation for Activity Preservation with Concentration Assessment Pre and Post Encapsulation. ACS Omega 2025, 10, 6135–6146. [Google Scholar] [CrossRef]
- Medfai, W.; Oueslati, I.; Dumas, E.; Harzalli, Z.; Viton, C.; Mhamdi, R.; Gharsallaoui, A. Physicochemical and Biological Characterization of Encapsulated Olive Leaf Extracts for Food Preservation. Antibiotics 2023, 12, 987. [Google Scholar] [CrossRef]
- Gajic, I.; Kabic, J.; Kekic, D.; Jovicevic, M.; Milenkovic, M.; Mitic Culafic, D.; Trudic, A.; Ranin, L.; Opavski, N. Antimicrobial Susceptibility Testing: A Comprehensive Review of Currently Used Methods. Antibiotics 2022, 11, 427. [Google Scholar] [CrossRef]
- Balouiri, M.; Sadiki, M.; Ibnsouda, S.K. Methods for in Vitro Evaluating Antimicrobial Activity: A Review. J. Pharm. Anal. 2016, 6, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Paciulli, M.; Grimaldi, M.; Rinaldi, M.; Cavazza, A.; Flamminii, F.; Mattia, C.D.; Gennari, M.; Chiavaro, E. Microencapsulated Olive Leaf Extract Enhances Physicochemical Stability of Biscuits. Future Foods 2023, 7, 100209. [Google Scholar] [CrossRef]
- Oliveira, F.M.; Oliveira, R.M.; Gehrmann Buchweitz, L.T.; Pereira, J.R.; Cristina dos Santos Hackbart, H.; Nalério, É.S.; Borges, C.D.; Zambiazi, R.C. Encapsulation of Olive Leaf Extract (Olea europaea L.) in Gelatin/Tragacanth Gum by Complex Coacervation for Application in Sheep Meat Hamburger. Food Control 2022, 131, 108426. [Google Scholar] [CrossRef]
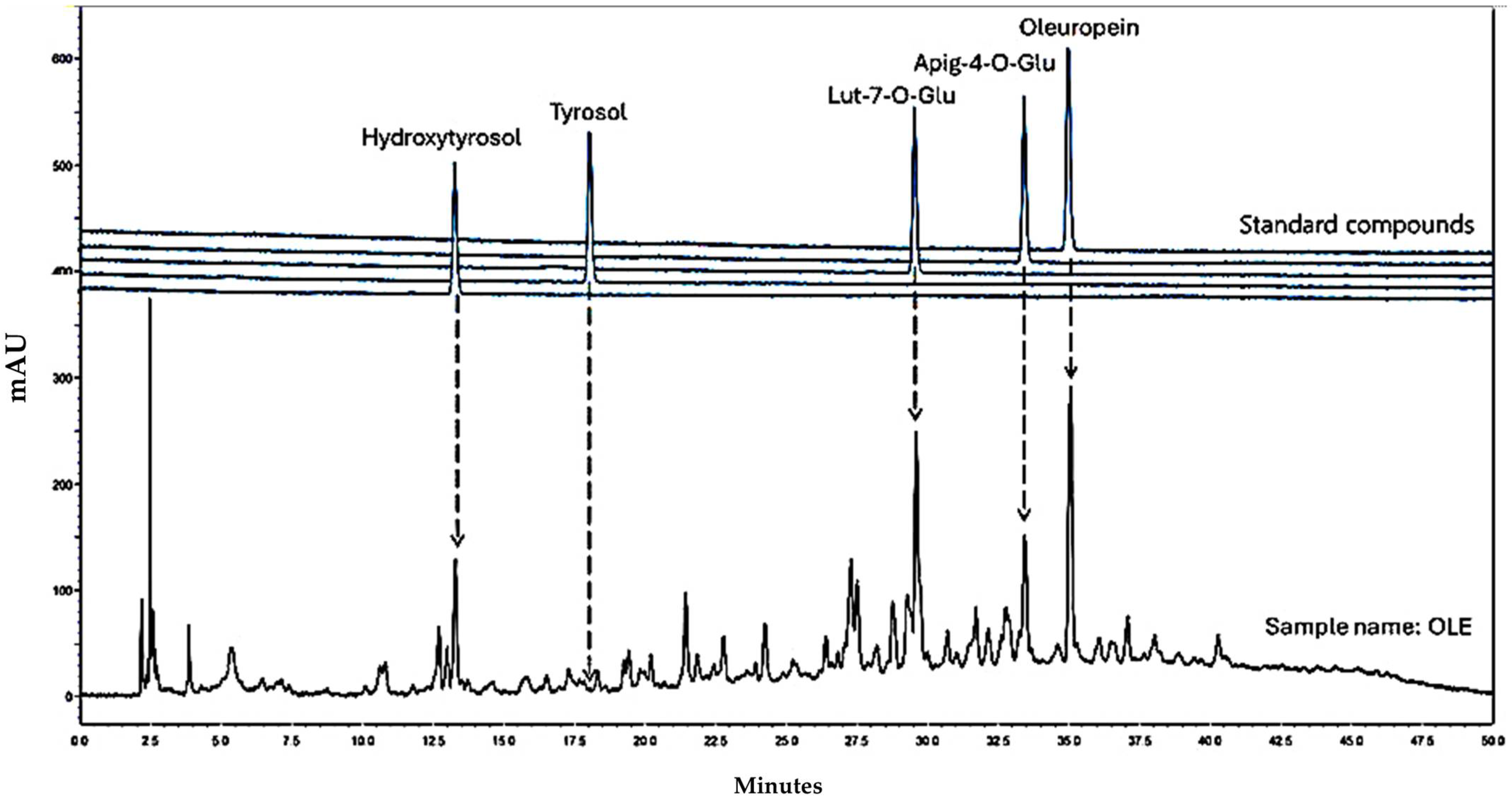
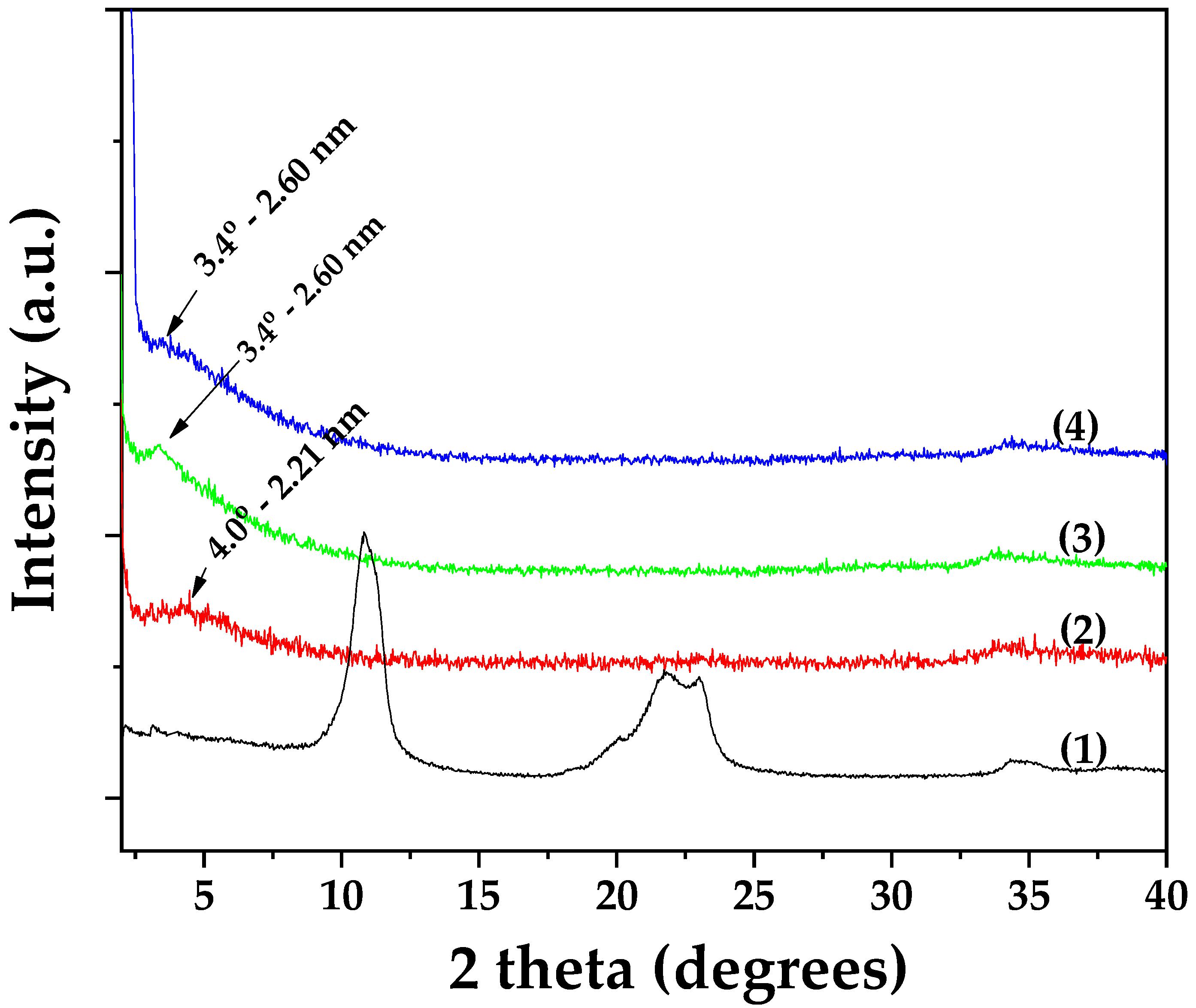


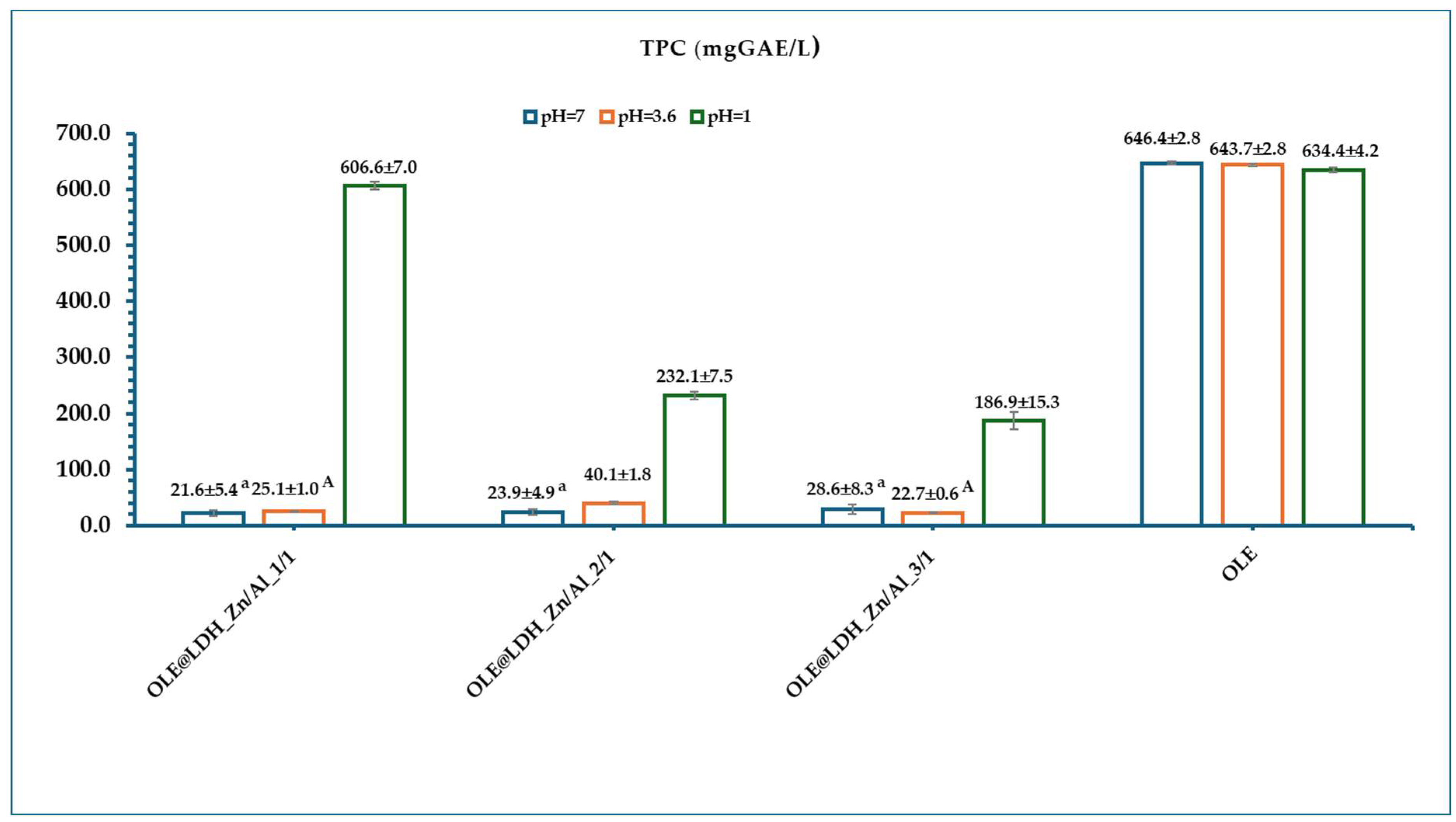
| Sample Name | Extraction Yield (mg/L) | HT (mg/L) | lut-7-ο-glu (mg/L) | apig-4-o-glu (mg/L) | oleur (mg/L) |
|---|---|---|---|---|---|
| OLE | 24.00 ± 0.02 | 0.53 ± 0.02 | 0.70 ± 0.02 | 0.18 ± 0.01 | 4.24 ± 0.03 |
| Sample | EC50,DPPH (mg/mL) | EC50,ABTS (mg/mL) | EC50,FRAP (mg/mL) |
|---|---|---|---|
| OLE@LDH_Zn/Al_1/1 | 27.88 ± 1.82 a | 25.70 ± 0.76 a | 39.42 ± 2.16 a |
| OLE@LDH_Zn/Al_2/1 | 35.62 ± 4.80 a | 35.33 ± 3.24 a | 52.71 ± 5.01 |
| OLE@LDH_Zn/Al_3/1 | 69.07 ± 10.50 | 67.82 ± 8.59 | 98.68 ± 4.96 |
| OLE | 30.56 ± 0.48 a | 28.25 ± 0.92 a | 41.96 ± 1.37 a |
| Bacteria | Sample | MIC * (mg/mL) | MBC * (mg/mL) | ZOI * (mm) |
|---|---|---|---|---|
| E. coli | ||||
| 1/1 # | 3.12 | 12.5 | 5 ± 1 a | |
| 2/1 # | 3.12 | 6.25 | 6 ± 1 a | |
| 3/1 # | 3.12 | 6.25 | 6 ± 1 a | |
| S. aureus | ||||
| 1/1 # | 1.56 | 6.25 | 3 ± 1 b | |
| 2/1 # | 0.78 | 6.25 | 4 ± 1 b | |
| 3/1 # | 1.56 | 3.12 | 4 ± 1 b | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kechagias, A.; Leontiou, A.A.; Vardakas, A.; Stathopoulos, P.; Xenaki, M.; Stathopoulou, P.; Proestos, C.; Giannelis, E.P.; Chalmpes, N.; Salmas, C.E.; et al. Antioxidant Nanohybrid Materials Derived via Olive Leaf Extract Incorporation in Layered Double Hydroxide: Preparation, Characterization, and Evaluation for Applications. Antioxidants 2025, 14, 1010. https://doi.org/10.3390/antiox14081010
Kechagias A, Leontiou AA, Vardakas A, Stathopoulos P, Xenaki M, Stathopoulou P, Proestos C, Giannelis EP, Chalmpes N, Salmas CE, et al. Antioxidant Nanohybrid Materials Derived via Olive Leaf Extract Incorporation in Layered Double Hydroxide: Preparation, Characterization, and Evaluation for Applications. Antioxidants. 2025; 14(8):1010. https://doi.org/10.3390/antiox14081010
Chicago/Turabian StyleKechagias, Achilleas, Areti A. Leontiou, Alexios Vardakas, Panagiotis Stathopoulos, Maria Xenaki, Panagiota Stathopoulou, Charalampos Proestos, Emmanuel P. Giannelis, Nikolaos Chalmpes, Constantinos E. Salmas, and et al. 2025. "Antioxidant Nanohybrid Materials Derived via Olive Leaf Extract Incorporation in Layered Double Hydroxide: Preparation, Characterization, and Evaluation for Applications" Antioxidants 14, no. 8: 1010. https://doi.org/10.3390/antiox14081010
APA StyleKechagias, A., Leontiou, A. A., Vardakas, A., Stathopoulos, P., Xenaki, M., Stathopoulou, P., Proestos, C., Giannelis, E. P., Chalmpes, N., Salmas, C. E., & Giannakas, A. E. (2025). Antioxidant Nanohybrid Materials Derived via Olive Leaf Extract Incorporation in Layered Double Hydroxide: Preparation, Characterization, and Evaluation for Applications. Antioxidants, 14(8), 1010. https://doi.org/10.3390/antiox14081010













