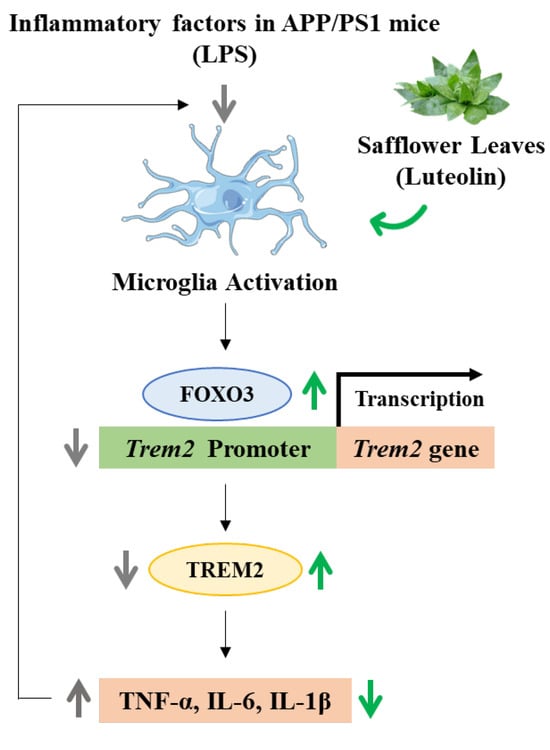
Luteolin in Safflower Leaves Suppresses Microglial Inflammation Through FOXO3-Mediated Trem2 Transcription
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Cell Culture
2.3. Cell Viability
2.4. Calcein AM Staining
2.5. Immunofluorescence (IF)
2.6. Multiplex Immunohistochemistry (mIHC)
2.7. Western Blot
2.8. Real-Time PCR
2.9. Transfection
2.10. Dual-Luciferase Reporter Assay
2.11. Statistical Analysis
3. Results
3.1. Safflower Leaves Inhibited Microglial Activation in APP/PS1 Mice
3.2. Luteolin Served as the Major Active Flavonoid in Safflower Leaves
3.3. Luteolin Inhibited Microglial Inflammatory Response and Increased TREM2 in LPS-Activated N9 Cells
3.4. Trem2 Knockdown Attenuated Luteolin’s Anti-Inflammatory Effects
3.5. Luteolin Promoted Trem2 Transcription Through Increasing FOXO3
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AD | Alzheimer’s disease |
| TREM2 | triggering receptor expressed on myeloid cells 2 |
| LPS | lipopolysaccharides |
| FOXO3 | forkhead box protein O3 |
| TNF-α | tumor necrosis factor-α |
| IL-1β | interleukin-1β |
| NO | nitric oxide |
| CD33 | cluster of differentiation 33 |
| ABCA7 | ATP-binding cassette sub-family A member 7 |
| SHIP1 | Src homology 2 domain-containing inositol phosphatase 1 |
| APOE | apolipoprotein E |
| CNS | central nervous system |
| DPPH | 1,1-diphenyl-2-picrylhydrazyl |
| SLP | safflower leaves powder |
| WT | wild-type |
| PFA | paraformaldehyde |
| FBS | fetal bovine serum |
| H-DMEM | high glucose-Dulbecco’s modified eagle medium |
| MTT | 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| DMSO | dimethyl sulfoxide |
| IF | immunofluorescence |
| PBS | phosphate-buffered saline |
| BSA | bovine serum albumin |
| DAPI | 4′,6-diamidino-2-phenylindole |
| CD68 | cluster of differentiation 68 |
| mIHC | multiplex immunohistochemistry |
| Iba1 | ionized calcium-binding adaptor molecule 1 |
| PMSF | phenylmethylsulfonyl fluoride |
| SYK | spleen tyrosine kinase |
| TLR4 | toll-like receptor 4 |
| ERK | extracellular signal-regulated kinase |
| TBST | Tris-buffered saline with Tween 20 |
| SEM | standard error of the mean |
| ANOVA | analysis of variance |
| IL-6 | interleukin-6 |
| IC50 | half maximal inhibitory concentration |
| CI | combination index |
| NOS2 | nitric oxide synthase 2 |
| COX-2 | cyclooxygenase-2 |
| Aβ | amyloid-β |
| JNK | c-Jun N-terminal kinase |
References
- Wang, G.; Qi, J.; Liu, X.; Ren, R.; Lin, S.; Hu, Y.; Li, H.; Xie, X.; Wang, J.; Li, J.; et al. China Alzheimer Report 2024. J. Diagn. Concepts Pract. 2024, 23, 219–256. (In Chinese) [Google Scholar] [CrossRef]
- Calsolaro, V.; Edison, P. Neuroinflammation in Alzheimer’s disease: Current evidence and future directions. Alzheimer’s Dement. 2016, 12, 719–732. [Google Scholar] [CrossRef]
- Wang, C.; Zong, S.; Cui, X.; Wang, X.; Wu, S.; Wang, L.; Liu, Y.; Lu, Z. The effects of microglia-associated neuroinflammation on Alzheimer’s disease. Front. Immunol. 2023, 14, 1117172. [Google Scholar] [CrossRef]
- Nizami, S.; Hall-Roberts, H.; Warrier, S.; Cowley, S.A.; Di Daniel, E. Microglial inflammation and phagocytosis in Alzheimer’s disease: Potential therapeutic targets. Br. J. Pharmacol. 2019, 176, 3515–3532. [Google Scholar] [CrossRef]
- Sochocka, M.; Zwolińska, K.; Leszek, J. The infectious etiology of Alzheimer’s disease. Curr. Neuropharmacol. 2017, 15, 996–1009. [Google Scholar] [CrossRef] [PubMed]
- Bruno, F.; Abondio, P.; Bruno, R.; Ceraudo, L.; Paparazzo, E.; Citrigno, L.; Luiselli, D.; Bruni, A.C.; Passarino, G.; Colao, R.; et al. Alzheimer’s disease as a viral disease: Revisiting the infectious hypothesis. Ageing Res. Rev. 2023, 91, 102068. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.L.; Zhu, Z.Q. Current status of sevoflurane anesthesia in association with microglia inflammation and neurodegenerative diseases. Ibrain 2024, 10, 217–224. [Google Scholar] [CrossRef]
- Hashioka, S.; Wu, Z.; Klegeris, A. Glia-driven neuroinflammation and systemic inflammation in Alzheimer’s disease. Curr. Neuropharmacol. 2021, 19, 908–924. [Google Scholar] [CrossRef]
- Swardfager, W.; Lanctôt, K.; Rothenburg, L.; Wong, A.; Cappell, J.; Herrmann, N. A meta-analysis of cytokines in Alzheimer’s disease. Biol. Psychiatry 2010, 68, 930–941. [Google Scholar] [CrossRef]
- Decourt, B.; Lahiri, D.K.; Sabbagh, M.N. Targeting tumor necrosis factor alpha for Alzheimer’s disease. Curr. Alzheimer Res. 2017, 14, 412–425. [Google Scholar] [CrossRef] [PubMed]
- Malik, M.; Parikh, I.; Vasquez, J.B.; Smith, C.; Tai, L.; Bu, G.; LaDu, M.J.; Fardo, D.W.; Rebeck, G.W.; Estus, S. Genetics ignite focus on microglial inflammation in Alzheimer’s disease. Mol. Neurodegener. 2015, 10, 52. [Google Scholar] [CrossRef]
- Tagliatti, E.; Desiato, G.; Mancinelli, S.; Bizzotto, M.; Gagliani, M.C.; Faggiani, E.; Hernández-Soto, R.; Cugurra, A.; Poliseno, P.; Miotto, M.; et al. Trem2 expression in microglia is required to maintain normal neuronal bioenergetics during development. Immunity 2024, 57, 86–105.e9. [Google Scholar] [CrossRef]
- Stefaniak, O.; Dobrzyńska, M.; Drzymała-Czyż, S.; Przysławski, J. Diet in the prevention of Alzheimer’s disease: Current knowledge and future research requirements. Nutrients 2022, 14, 4564. [Google Scholar] [CrossRef]
- Paramesha, M.; Ramesh, C.K.; Krishna, V.; Kumar, Y.S.R.; Parvathi, K.M.M. Hepatoprotective and in vitro antioxidant effect of Carthamus tinctorious L., var Annigeri-2-, an oil-yielding crop, against CCl4-induced liver injury in rats. Pharmacogn. Mag. 2011, 7, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Emongor, V. Safflower (Carthamus tinctorius L.) the underutilized and neglected crop: A review. Asian J. Plant Sci. 2010, 9, 299–306. [Google Scholar] [CrossRef]
- Dajue, L.; Heller, J.; Engels, J. Safflower Carthamus tinctorius L. In Promoting the Conservation and Use of Underutilized and Neglected Crops. 7; International Plant Genetic Resources Institute: Rome, Italy, 1996; pp. 17–25. [Google Scholar]
- Ishida, S. Safflower Blended Light Yellow Colored Black Tea, Contains Black Tea Leaf and Safflower Petals. 2001231451-A, 2001-08-28 2001. Available online: https://webofscience.clarivate.cn/wos/alldb/full-record/DIIDW:2001609928 (accessed on 8 December 2025).
- He, T. Safflower Tea Is Prepared from Safflower Tender Bud, Leaf, Flower and Fruit of Plant. 1471830-A, 2004-02-04 2004. Available online: https://webofscience.clarivate.cn/wos/alldb/full-record/DIIDW:2004317275 (accessed on 8 December 2025).
- Hu, Z.; Jin, X.; Wei, G.; Xu, J.; Xu, X.; Zhang, X. Safflower Tea Comprises Safflower Tea Leaves, Medlar, Codonopsis pilosula, Astragalus mongholicus, Red Date and Rose. 102422955-A, 2012-04-25 2012. Available online: https://webofscience.clarivate.cn/wos/alldb/full-record/DIIDW:2012F22088 (accessed on 8 December 2025).
- Lee, S.H.; Lillehoj, H.S.; Heckert, R.A.; Cho, S.M.; Tuo, W.; Lillehoj, E.P.; Chun, H.K.; Park, H.J. Immune enhancing properties of safflower leaf (Carthamus tinctorius) on chicken lymphocytes and macrophages. J. Poult. Sci. 2008, 45, 147–151. [Google Scholar] [CrossRef]
- Lee, S.H.; Lillehoj, H.S.; Cho, S.M.; Park, D.W.; Hong, Y.H.; Chun, H.K.; Lillehoj, E.P. Immunomodulatory effects of dietary safflower leaf in chickens. Korean Soc. Community Living Sci. 2007, 18, 715–724. [Google Scholar]
- Hiramatsu, M.; Igarashi, K.; Suzuki, J.; Murakami, A.; Oikawa, A.; Takahashi, T. Composition and radical scavenging activity of polyphenols present in petals and young leaves of Mogami-benibana (Safflower, Carthamus tinctrius Linne). Curr. Top. Nutraceutical Res. 2017, 15, 27–32. [Google Scholar]
- Golkar, P.; Arzani, A.; Rezaei, A.M.; Yarali, Z.; Yousefi, M. Genetic variation of leaf antioxidants and chlorophyll content in safflower. Afr. J. Agric. Res. 2009, 4, 1475–1482. [Google Scholar]
- Zhang, Q.; Yan, Y. The role of natural flavonoids on neuroinflammation as a therapeutic target for Alzheimer’s disease: A narrative review. Neural Regen. Res. 2023, 18, 2582–2591. [Google Scholar] [CrossRef] [PubMed]
- Abbas, H.; Sayed, N.S.E.; Youssef, N.; ME Gaafar, P.; Mousa, M.R.; Fayez, A.M.; Elsheikh, M.A. Novel luteolin-loaded chitosan decorated nanoparticles for brain-targeting delivery in a sporadic Alzheimer’s disease mouse model: Focus on antioxidant, anti-inflammatory, and amyloidogenic pathways. Pharmaceutics 2022, 14, 1003. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Zhang, S.; Peng, Y.; Wang, Y.; Gao, P.; Hu, Y.; Wang, Z.; Noda, M.; Hiramatsu, M.; Liu, J.; et al. Safflower leaf ameliorates cognitive impairment through moderating excessive astrocyte activation in APP/PS1 mice. Food Funct. 2021, 12, 11704–11716. [Google Scholar] [CrossRef]
- Hopperton, K.E.; Mohammad, D.; Trépanier, M.O.; Giuliano, V.; Bazinet, R.P. Markers of microglia in post-mortem brain samples from patients with Alzheimer’s disease: A systematic review. Mol. Psychiatry 2018, 23, 177–198. [Google Scholar] [CrossRef]
- Xu, M.; Zhang, D.F.; Luo, R.; Wu, Y.; Zhou, H.; Kong, L.L.; Bi, R.; Yao, Y.G. A systematic integrated analysis of brain expression profiles reveals YAP1 and other prioritized hub genes as important upstream regulators in Alzheimer’s disease. Alzheimer’s Dement. 2018, 14, 215–229. [Google Scholar] [CrossRef]
- Das, D.; Nanda, M.; Banjare, P.; Lanjhiyana, S. Exploration of multitargeted antialzheimer’s activity of safflower leaves phytoconstituents: In silico molecular docking approach. Eur. J. Med. Chem. Rep. 2024, 10, 100119. [Google Scholar] [CrossRef]
- Kure, A.; Nakagawa, K.; Kondo, M.; Kato, S.; Kimura, F.; Watanabe, A.; Shoji, N.; Hatanaka, S.; Tsushida, T.; Miyazawa, T. Metabolic fate of luteolin in rats: Its relationship to anti-inflammatory effect. J. Agric. Food Chem. 2016, 64, 4246–4254. [Google Scholar] [CrossRef]
- Ma, W.; Ren, F.C.; Wang, X.R.; Li, N. Anti-Inflammatory Effect of Xanthones from Hypericum beanii on Macrophage RAW 264.7 Cells through Reduced NO Production and TNF-α, IL-1β, IL-6, and COX-2 Expression. Molecules 2024, 29, 3705. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Gao, X.; Zhao, K.; Chen, H.; Xu, M.; Wang, K. Effect of TREM2 on release of inflammatory factor from LPS-stimulated microglia and its possible mechanism. Ann. Clin. Lab. Sci. 2019, 49, 249–256. [Google Scholar]
- Goshi, N.; Morgan, R.K.; Lein, P.J.; Seker, E. A primary neural cell culture model to study neuron, astrocyte, and microglia interactions in neuroinflammation. J. Neuroinflamm. 2020, 17, 155. [Google Scholar] [CrossRef]
- Kim, J.; Sullivan, O.; Lee, K.; Jao, J.; Tamayo, J.; Madany, A.M.; Wong, B.; Ashwood, P.; Ciernia, A.V. Repeated LPS induces training and tolerance of microglial responses across brain regions. J. Neuroinflamm. 2024, 21, 233. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Boska, M.; Zheng, Y.; Liu, J.; Fox, H.S.; Xiong, H. Minocycline attenuation of rat corpus callosum abnormality mediated by low-dose lipopolysaccharide-induced microglia activation. J. Neuroinflamm. 2021, 18, 100. [Google Scholar] [CrossRef]
- Kim, J.; Lee, H.J.; Park, S.K.; Park, J.H.; Jeong, H.R.; Lee, S.; Lee, H.; Seol, E.; Hoe, H.S. Donepezil regulates LPS and Aβ-stimulated neuroinflammation through MAPK/NLRP3 inflammasome/STAT3 signaling. Int. J. Mol. Sci. 2021, 22, 10637. [Google Scholar] [CrossRef]
- Ulland, T.K.; Colonna, M. TREM2—A key player in microglial biology and Alzheimer disease. Nat. Rev. Neurol. 2018, 14, 667–675. [Google Scholar] [CrossRef]
- Paradowska-Gorycka, A.; Jurkowska, M. Structure, expression pattern and biological activity of molecular complex TREM-2/DAP12. Hum. Immunol. 2013, 74, 730–737. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; He, Y.L.; Xu, N.; Wang, X.F.; Song, B.; Tang, B.Q.; Lee, S.M. A natural small molecule aspidosperma-type alkaloid, hecubine, as a new TREM2 activator for alleviating lipopolysaccharide-induced neuroinflammation in vitro and in vivo. Redox Biol. 2024, 70, 103057. [Google Scholar] [CrossRef] [PubMed]
- Perugorria, M.J.; Esparza-Baquer, A.; Oakley, F.; Labiano, I.; Korosec, A.; Jais, A.; Mann, J.; Tiniakos, D.; Santos-Laso, A.; Arbelaiz, A.; et al. Non-parenchymal TREM-2 protects the liver from immune-mediated hepatocellular damage. Gut 2019, 68, 533–546. [Google Scholar] [CrossRef]
- Xu, M.; Yang, Y.; Peng, J.; Zhang, Y.; Wu, B.; He, B.; Jia, Y.; Yan, T. Effects of Alpinae Oxyphyllae Fructus on microglial polarization in a LPS-induced BV2 cells model of neuroinflammation via TREM2. J. Ethnopharmacol. 2023, 302, 115914. [Google Scholar] [CrossRef]
- Zhou, J.; Yu, W.; Zhang, M.; Tian, X.; Li, Y.; Lü, Y. Imbalance of microglial TLR4/TREM2 in LPS-treated APP/PS1 transgenic mice: A potential link between Alzheimer’s disease and systemic inflammation. Neurochem. Res. 2019, 44, 1138–1151. [Google Scholar] [CrossRef] [PubMed]
- Calderaro, A.; Patanè, G.T.; Tellone, E.; Barreca, D.; Ficarra, S.; Misiti, F.; Laganà, G. The neuroprotective potentiality of flavonoids on Alzheimer’s disease. Int. J. Mol. Sci. 2022, 23, 14835. [Google Scholar] [CrossRef]
- Kempuraj, D.; Thangavel, R.; Kempuraj, D.D.; Ahmed, M.E.; Selvakumar, G.P.; Raikwar, S.P.; Zaheer, S.A.; Iyer, S.S.; Govindarajan, R.; Chandrasekaran, P.N.; et al. Neuroprotective effects of flavone luteolin in neuroinflammation and neurotrauma. Biofactors 2021, 47, 190–197. [Google Scholar] [CrossRef]
- Shishtar, E.; Rogers, G.T.; Blumberg, J.B.; Au, R.; Jacques, P.F. Long-term dietary flavonoid intake and risk of Alzheimer disease and related dementias in the Framingham Offspring Cohort. Am. J. Clin. Nutr. 2020, 112, 343–353. [Google Scholar] [CrossRef]
- Che, D.N.; Cho, B.O.; Kim, J.S.; Shin, J.Y.; Kang, H.J.; Jang, S.I. Effect of luteolin and apigenin on the production of Il-31 and Il-33 in lipopolysaccharides-activated microglia cells and their mechanism of action. Nutrients 2020, 12, 811. [Google Scholar] [CrossRef]
- Jang, S.; Kelley, K.W.; Johnson, R.W. Luteolin reduces IL-6 production in microglia by inhibiting JNK phosphorylation and activation of AP-1. Proc. Natl. Acad. Sci. USA 2008, 105, 7534–7539. [Google Scholar] [CrossRef]
- Rezai-Zadeh, K.; Ehrhart, J.; Bai, Y.; Sanberg, P.R.; Bickford, P.; Tan, J.; Shytle, R.D. Apigenin and luteolin modulate microglial activation via inhibition of STAT1-induced CD40 expression. J. Neuroinflamm. 2008, 5, 41. [Google Scholar] [CrossRef]
- Caltagirone, C.; Cisari, C.; Schievano, C.; Di Paola, R.; Cordaro, M.; Bruschetta, G.; Esposito, E.; Cuzzocrea, S. Co-ultramicronized palmitoylethanolamide/luteolin in the treatment of cerebral ischemia: From rodent to man. Transl. Stroke Res. 2016, 7, 54–69. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Li, X.; Wang, Z.; Cao, Y.; Han, S.; Li, N.; Cai, J.; Cheng, S.; Liu, Q. Protective effects of luteolin against amyloid beta-induced oxidative stress and mitochondrial impairments through peroxisome proliferator-activated receptor γ-dependent mechanism in Alzheimer’s disease. Redox Biol. 2023, 66, 102848. [Google Scholar] [CrossRef]
- Rezai-Zadeh, K.; Douglas Shytle, R.; Bai, Y.; Tian, J.; Hou, H.; Mori, T.; Zeng, J.; Obregon, D.; Town, T.; Tan, J. Flavonoid-mediated presenilin-1 phosphorylation reduces Alzheimer’s disease beta-amyloid production. J. Cell. Mol. Med. 2009, 13, 574–588. [Google Scholar] [CrossRef] [PubMed]
- Owona, B.A.; Njayou, F.N.; Mkounga, P.; Moundipa, P.F. Khaya grandifoliola active fraction as a source of therapeutic compounds for Alzheimer’s disease treatment: In silico validation of identified compounds. Silico Pharmacol. 2022, 10, 11. [Google Scholar] [CrossRef] [PubMed]
- Shippy, D.C.; Ulland, T.K. Lipid metabolism transcriptomics of murine microglia in Alzheimer’s disease and neuroinflammation. Sci. Rep. 2023, 13, 14800. [Google Scholar] [CrossRef]
- Shippy, D.C.; Ulland, T.K. Genome-wide identification of murine interferon genes in microglial-mediated neuroinflammation in Alzheimer’s disease. J. Neuroimmunol. 2023, 375, 578031. [Google Scholar] [CrossRef]
- Xie, D.; Deng, T.; Zhai, Z.; Sun, T.; Xu, Y. The cellular model for Alzheimer’s disease research: PC12 cells. Front. Mol. Neurosci. 2022, 15, 1016559. [Google Scholar] [CrossRef] [PubMed]
- Malvaso, A.; Gatti, A.; Negro, G.; Calatozzolo, C.; Medici, V.; Poloni, T.E. Microglial senescence and activation in healthy aging and Alzheimer’s disease: Systematic review and neuropathological scoring. Cells 2023, 12, 2824. [Google Scholar] [CrossRef]
- Zhang, X.; Chen, X.; Zhang, L.; Sun, Y.; Liang, Y.; Li, H.; Zhang, Y. Role of trigger receptor 2 expressed on myeloid cells in neuroinflammation—neglected multidimensional regulation of microglia. Neurochem. Int. 2023, 171, 105639. [Google Scholar] [CrossRef]
- Hou, J.; Chen, Y.; Grajales-Reyes, G.; Colonna, M. TREM2 dependent and independent functions of microglia in Alzheimer’s disease. Mol. Neurodegener. 2022, 17, 84. [Google Scholar] [CrossRef]
- Zhong, L.; Zhang, Z.L.; Li, X.; Liao, C.; Mou, P.; Wang, T.; Wang, Z.; Wang, Z.; Wei, M.; Xu, H.; et al. TREM2/DAP12 complex regulates inflammatory responses in microglia via the JNK signaling pathway. Front. Aging Neurosci. 2017, 9, 204. [Google Scholar] [CrossRef]
- Wang, H.; Ma, J.; Li, X.; Peng, Y.; Wang, M. FDA compound library screening Baicalin upregulates TREM2 for the treatment of cerebral ischemia-reperfusion injury. Eur. J. Pharmacol. 2024, 969, 176427. [Google Scholar] [CrossRef]
- Teter, B.; Morihara, T.; Lim, G.P.; Chu, T.; Jones, M.R.; Zuo, X.; Paul, R.M.; Frautschy, S.A.; Cole, G.M. Curcumin restores innate immune Alzheimer’s disease risk gene expression to ameliorate Alzheimer pathogenesis. Neurobiol. Dis. 2019, 127, 432–448. [Google Scholar] [CrossRef]
- Cao, G.; Lin, M.; Gu, W.; Su, Z.; Duan, Y.; Song, W.; Liu, H.; Zhang, F. The rules and regulatory mechanisms of FOXO3 on inflammation, metabolism, cell death and aging in hosts. Life Sci. 2023, 328, 121877. [Google Scholar] [CrossRef] [PubMed]
- Bernardo, V.S.; Torres, F.F.; da Silva, D.G.H. FoxO3 and oxidative stress: A multifaceted role in cellular adaptation. J. Mol. Med. 2023, 101, 83–99. [Google Scholar] [CrossRef]
- Du, S.; Jin, F.; Maneix, L.; Gedam, M.; Xu, Y.; Catic, A.; Wang, M.C.; Zheng, H. FoxO3 deficiency in cortical astrocytes leads to impaired lipid metabolism and aggravated amyloid pathology. Aging Cell 2021, 20, e13432. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Garcia, M.A.; Lara-Ureña, N.; March-Diaz, R.; Ortega-de San Luis, C.; Quiñones-Cañete, S.; Mora-Romero, B.; Barba-Reyes, J.M.; Cabello-Rivera, D.; Romero-Molina, C.; Heras-Garvin, A.; et al. Inactivation of the PHD3-FOXO3 axis blunts the type I interferon response in microglia and ameliorates Alzheimer’s disease progression. Sci. Adv. 2025, 11, eadu2244. [Google Scholar] [CrossRef]
- Miao, J.; Liu, X.; Liao, Y.; Li, Y.; Kuang, Y.; Zheng, J.; Li, Z.; Lan, J. Paeonol enhances macrophage phagocytic function by modulating lipid metabolism through the P53-TREM2 axis. Front. Pharmacol. 2023, 14, 1214756. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Zheng, Y.; Huang, L.; Ye, J.; Ye, Y.; Luo, H.; Chen, X.; Yao, W.; Chen, J.; Zhang, J.C. Nrf2 regulates the arginase 1(+) microglia phenotype through the initiation of TREM2 transcription, ameliorating depression-like behavior in mice. Transl. Psychiatry 2022, 12, 459. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Hu, S.; Liu, L. Phosphorylation and acetylation modifications of FOXO3a: Independently or synergistically? Oncol. Lett. 2017, 13, 2867–2872. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.K.; Webb, A.E. Regulation of FOXO factors in mammalian cells. Curr. Top. Dev. Biol. 2018, 127, 165–192. [Google Scholar] [CrossRef]
- Schoch, K.M.; Ezerskiy, L.A.; Morhaus, M.M.; Bannon, R.N.; Sauerbeck, A.D.; Shabsovich, M.; Jafar-Nejad, P.; Rigo, F.; Miller, T.M. Acute Trem2 reduction triggers increased microglial phagocytosis, slowing amyloid deposition in mice. Proc. Natl. Acad. Sci. USA 2021, 118, e2100356118. [Google Scholar] [CrossRef]





Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, T.; Zhang, S.; Ma, J.; Atiakshin, D.; Han, S.; Noda, M.; Hiramatsu, M.; Liu, J.; Peng, Y.; Long, J. Luteolin in Safflower Leaves Suppresses Microglial Inflammation Through FOXO3-Mediated Trem2 Transcription. Antioxidants 2025, 14, 1495. https://doi.org/10.3390/antiox14121495
Zhang T, Zhang S, Ma J, Atiakshin D, Han S, Noda M, Hiramatsu M, Liu J, Peng Y, Long J. Luteolin in Safflower Leaves Suppresses Microglial Inflammation Through FOXO3-Mediated Trem2 Transcription. Antioxidants. 2025; 14(12):1495. https://doi.org/10.3390/antiox14121495
Chicago/Turabian StyleZhang, Tiantian, Shuangxi Zhang, Jiayang Ma, Dmitrii Atiakshin, Shujun Han, Mami Noda, Midori Hiramatsu, Jiankang Liu, Yunhua Peng, and Jiangang Long. 2025. "Luteolin in Safflower Leaves Suppresses Microglial Inflammation Through FOXO3-Mediated Trem2 Transcription" Antioxidants 14, no. 12: 1495. https://doi.org/10.3390/antiox14121495
APA StyleZhang, T., Zhang, S., Ma, J., Atiakshin, D., Han, S., Noda, M., Hiramatsu, M., Liu, J., Peng, Y., & Long, J. (2025). Luteolin in Safflower Leaves Suppresses Microglial Inflammation Through FOXO3-Mediated Trem2 Transcription. Antioxidants, 14(12), 1495. https://doi.org/10.3390/antiox14121495