Ruta graveolens: Boost Melanogenic Effects and Protection against Oxidative Damage in Melanocytes
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Plant Material
2.3. Equipment
2.4. Extraction and Isolation
2.5. Cell Treatments and Culture
2.6. Cell Viability Assay
2.7. Assay for Relative Intracellular Melanin Concentration and Tyrosinase Activity
2.8. Western Blotting
2.9. Cytotoxicity and Oxidative Injury Assessment in 4-TBP-Induced PIG3V Melanocytes
2.10. ROS Assessment in 4-TBP-Induced PIG3V Melanocytes
2.11. Cytokine IL-6 Secretion Assessment in 4-TBP-Induced PIG3V Melanocytes
2.12. Evaluation of UPR Modulators in 4-TBP-Induced PIG3V Melanocytes
2.13. Statistical Analysis
3. Results
3.1. Identification of the Components
3.2. Melanogenic Effects of R. graveolens Fractions in B16 Cells
3.3. Effects on Relative Melanin Content and Tyrosinase Activity of R. graveolens Fractions in B16 Cells
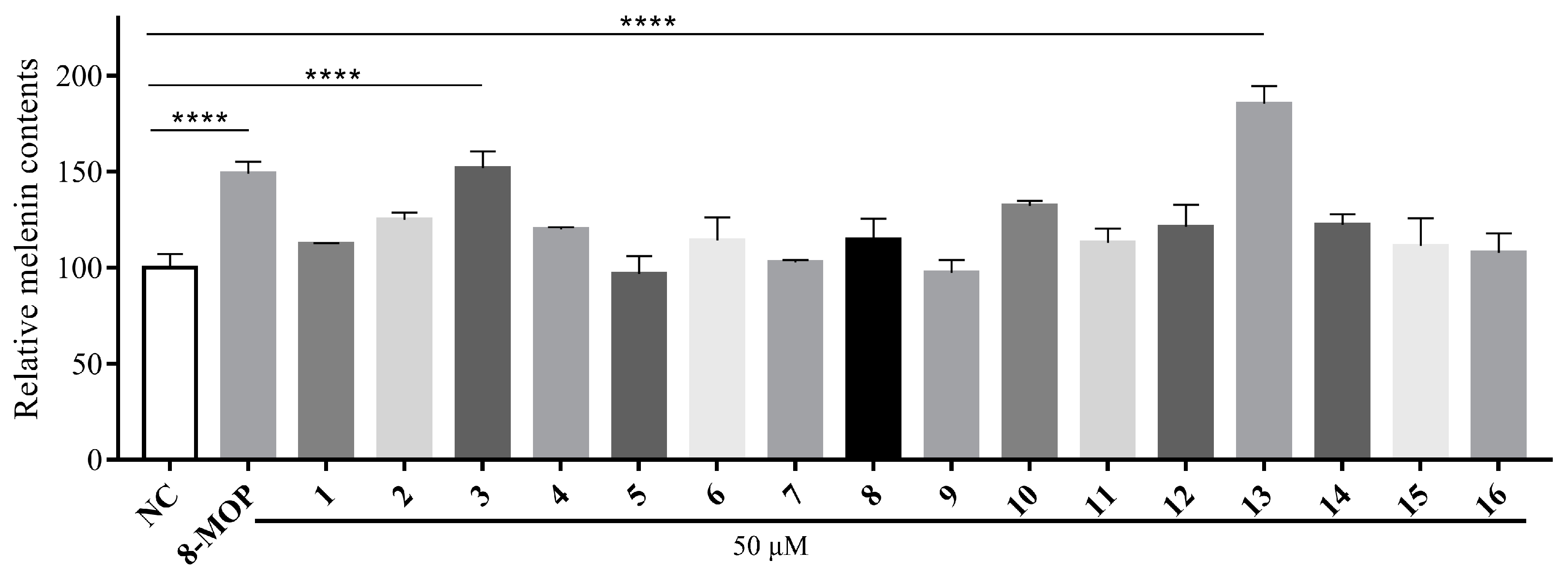
3.4. Melanogenic Effects of Compounds 1–16 in B16 Cells
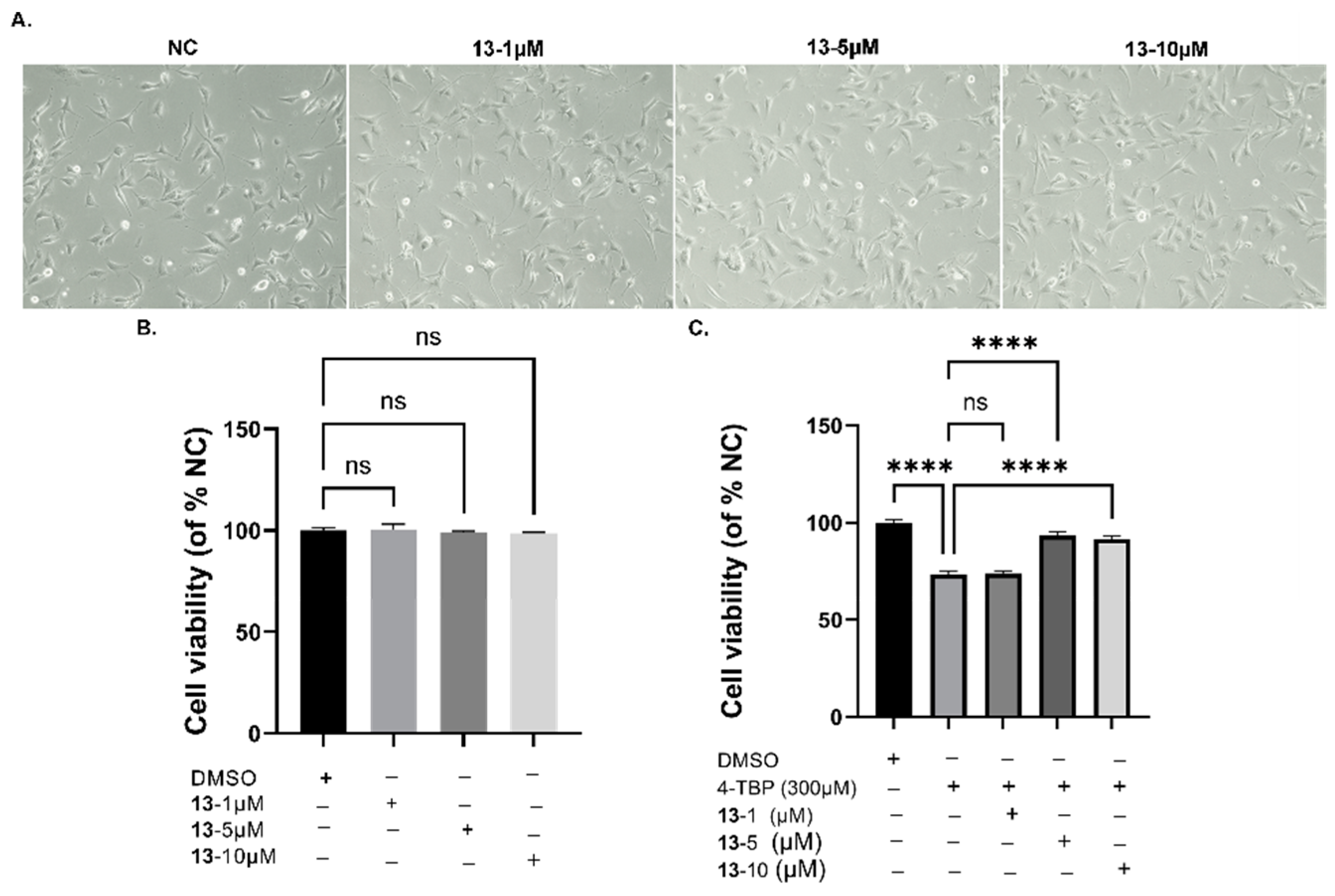
3.5. Cell Toxicity of Compound 13
3.6. Effect on Melanin Content and Tyrosinase Activity of 13
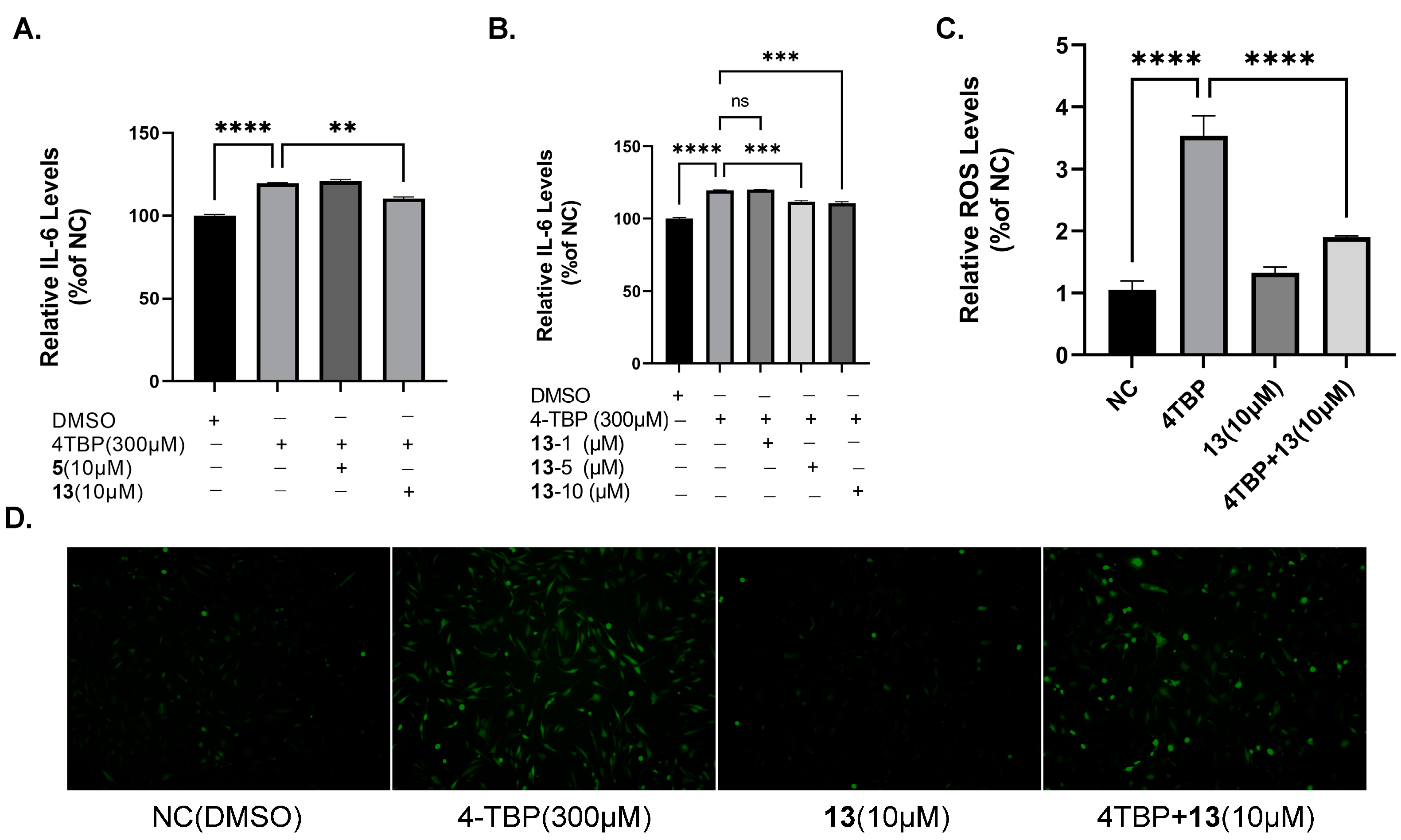
3.7. Protective Effects of Compound 13 on 4-TBP-Induced Oxidative Injury in PIG3V Melanocytes
3.8. Compound 13 Inhibited ROS Generation and Cytokine Expression Induced by 4-TBP in PIG3V Melanocytes
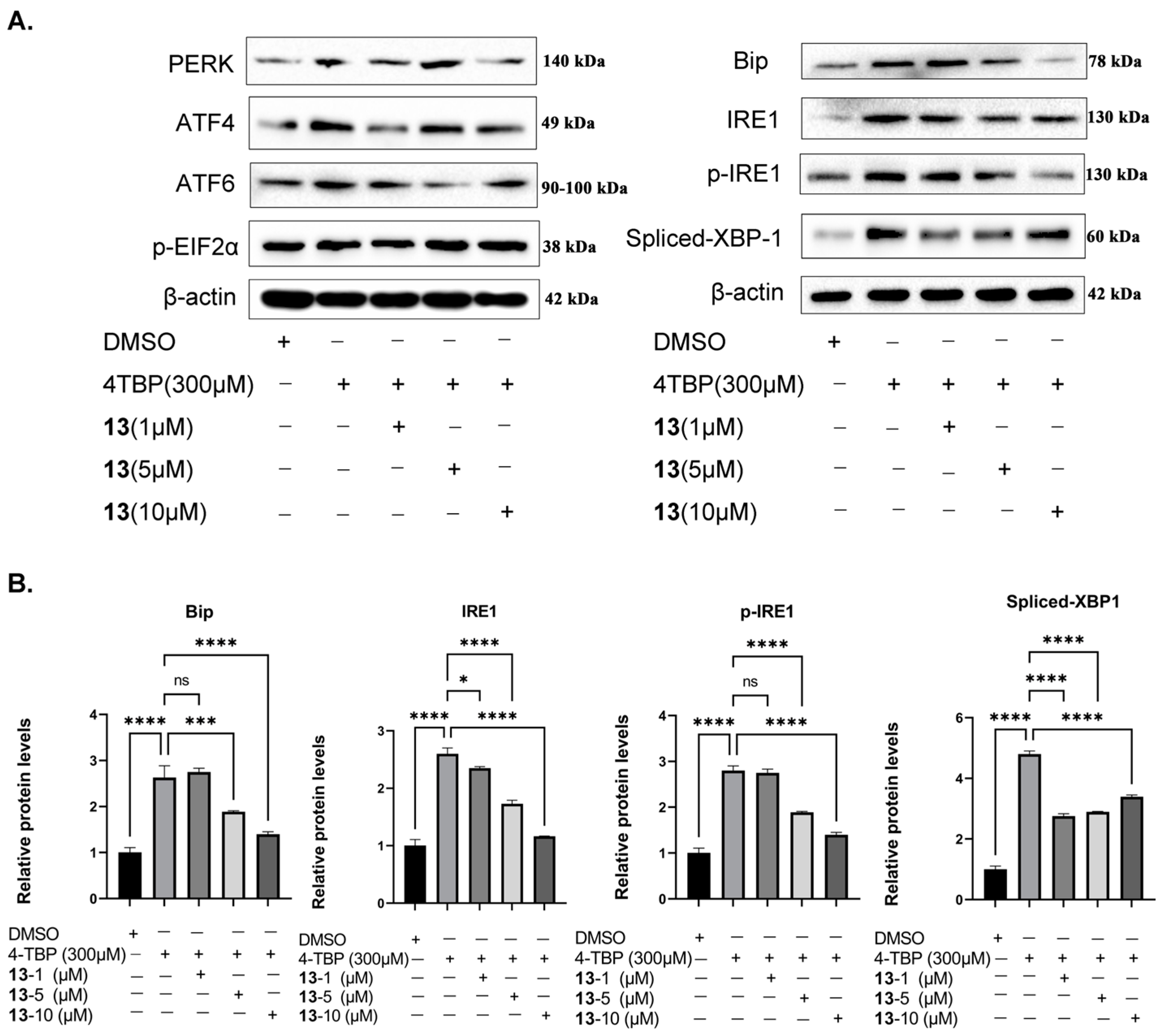
3.9. Compound 13 Inhibited 4-TBP-Induced Upregulation of Key UPR Proteins
3.10. Compound 13 Inhibited 4-TBP-Induced Production of IL-6 by Interfering with UPR
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Frisoli, M.L.; Essien, K.; Harris, J.E. Vitiligo: Mechanisms of Pathogenesis and Treatment. Annu. Rev. Immunol. 2020, 38, 621–648. [Google Scholar] [CrossRef] [PubMed]
- Niu, C.; Aisa, H.A. Upregulation of melanogenesis and tyrosinase activity: Potential Agents for Vitiligo. Molecules 2017, 22, 1303. [Google Scholar] [CrossRef] [PubMed]
- Lambert, M.W.; Maddukuri, S.; Karanfilian, K.M.; Elias, M.L.; Lambert, W.C. The physiology of melanin deposition in health and disease. Clin. Dermatol. 2019, 37, 402–417. [Google Scholar] [CrossRef]
- D’Orazio, J.; Jarrett, S.; Amaro-Ortiz, A.; Scott, T. UV radiation and the skin. Int. J. Mol. Sci. 2013, 14, 12222–12248. [Google Scholar] [CrossRef]
- Baroni, A.; Buommino, E.; De Gregorio, V.; Ruocco, E.; Ruocco, V.; Wolf, R. Structure and function of the epidermis related to barrier properties. Clin. Dermatol. 2012, 30, 257–262. [Google Scholar] [CrossRef]
- Wang, Y.; Li, S.; Li, C. Perspectives of new advances in the pathogenesis of vitiligo: From oxidative stress to autoimmunity. Med. Sci. Monit. 2019, 25, 1017–1023. [Google Scholar] [CrossRef]
- Passeron, T.; Ortonne, J.P. Activation of the unfolded protein response in vitiligo: The missing link? J. Investig. Dermatol. 2012, 132, 2502–2504. [Google Scholar] [CrossRef]
- Hariharan, V.; Klarquist, J.; Reust, M.J.; Koshoffer, A.; McKee, M.D.; Boissy, R.E.; Le Poole, I.C. Monobenzyl ether of hydroquinone and 4-tertiary butyl phenol activate markedly different physiological responses in melanocytes: Relevance to skin depigmentation. J. Investig. Dermatol. 2010, 130, 211–220. [Google Scholar] [CrossRef]
- Le Poole, I.C.; Boissy, R.E.; Sarangarajan, R.; Chen, J.; Forristal, J.J.; Sheth, P.; Westerhof, W.; Babcock, G.; Das, P.K.; Saelinger, C.B. PIG3V, an immortalized human vitiligo melanocyte cell line, expresses dilated endoplasmic reticulum. In Vitro Cell Dev. Biol. Anim. 2000, 36, 309–319. [Google Scholar] [CrossRef]
- Toosi, S.; Orlow, S.J.; Manga, P. Vitiligo-inducing phenols activate the unfolded protein response in melanocytes resulting in upregulation of IL6 and IL8. J. Investig. Dermatol. 2012, 132, 2601–2609. [Google Scholar] [CrossRef] [PubMed]
- Park, K.; Lee, S.E.; Shin, K.O.; Uchida, Y. Insights into the role of endoplasmic reticulum stress in skin function and associated diseases. FEBS J. 2019, 286, 413–425. [Google Scholar] [CrossRef]
- Jadeja, S.D.; Mayatra, J.M.; Vaishnav, J.; Shukla, N.; Begum, R. A concise review on the role of endoplasmic reticulum stress in the development of autoimmunity in vitiligo pathogenesis. Front. Immunol. 2020, 11, 624566. [Google Scholar] [CrossRef] [PubMed]
- Laddha, N.C.; Dwivedi, M.; Mansuri, M.S.; Gani, A.R.; Ansarullah, M.; Ramachandran, A.V.; Dalai, S.; Begum, R. Vitiligo: Interplay between oxidative stress and immune system. Exp. Dermatol. 2013, 22, 245–250. [Google Scholar] [CrossRef]
- Kadowaki, D.; Anraku, M.; Sakaya, M.; Hirata, S.; Maruyama, T.; Otagiri, M. Olmesartan protects endothelial cells against oxidative stress-mediated cellular injury. Clin. Exp. Nephrol. 2015, 19, 1007–1014. [Google Scholar] [CrossRef]
- Begum, M.E.; Sen, D. DOR agonist (SNC-80) exhibits anti-parkinsonian effect via downregulating UPR/oxidative stress signals and inflammatory response in vivo. Neurosci. Lett. 2018, 678, 29–36. [Google Scholar] [CrossRef]
- Tarique, M.; Siddiqui, H.H.; Khushtar, M.; Rahman, M.A. Protective effect of hydro-alcoholic extract of Ruta graveolens Linn. leaves on indomethacin and pylorus ligation-induced gastric ulcer in rats. J. Ayurveda Integr. Med. 2016, 7, 38–43. [Google Scholar] [CrossRef]
- Grigorjev, C.A.; Brizuela, N.Y. [Effects of ruta SSP on the activity of the smooth gastrointestinal muscle isolated of rat]. Rev. Fac. Cien Med. Univ. Nac. Cordoba 2010, 67, 77–80. [Google Scholar] [PubMed]
- Swapna, T.S.; Chippy, C.L. In Vitro Propagation and Phytochemcial Screening of Ruta graveolens; LAP LAMBERT Academic Publishing: Saarbrücken, Germany, 2017. [Google Scholar]
- Ratheesh, M.; Helen, A. Anti-inflammatory activity of Ruta graveolens Linn on carrageenan induced paw edema in wistar male rats. Afr. J. Biotechnol. 2006, 6, 10. [Google Scholar]
- Toserkani, A.; Jalali, M.R.; Najafzadeh, H. Changes of lipid profiles, glucose, and hemogram after administration of Ruta graveolens extract in diabetic rats. Comp. Clin. Pathol. 2011, 21, 1587–1592. [Google Scholar] [CrossRef]
- Meepagala, K.M.; Schrader, K.K.; Wedge, D.E.; Duke, S.O. Algicidal and antifungal compounds from the roots of Ruta graveolens and synthesis of their analogs. Phytochemistry 2005, 66, 2689–2695. [Google Scholar] [CrossRef]
- Khouri, N.A.; El-Akawi, Z. Antiandrogenic activity of Ruta graveolens L in male Albino rats with emphasis on sexual and aggressive behavior. Neuro Endocrinol. Lett. 2005, 26, 823–829. [Google Scholar]
- Flávia, S.; Barbosa, G.; Leão, D.; Leite; Alves, S.; Fonseca, A.; De, V.; Ávila, A.; Costa, C. Insecticide effects of Ruta graveolens, Copaifera langsdorffii and Chenopodium ambrosioides against pests and natural enemies in commercial tomato plantation. Acta Scientiarum. Agron. 2011, 33, 37–43. [Google Scholar] [CrossRef]
- Ainiwaer, P.; Nueraihemaiti, M.; Li, Z.; Zang, D.; Jiang, L.; Li, Y.; Aisa, H.A. Chemical constituents of Ruta graveolens L. and their melanogenic effects and action mechanism. Fitoterapia 2022, 156, 105094. [Google Scholar] [CrossRef] [PubMed]
- Win, N.N.; Woo, S.Y.; Ngwe, H.; Wong, C.P.; Ito, T.; Okamoto, Y.; Tanaka, M.; Imagawa, H.; Asakawa, Y.; Abe, I.; et al. Tetrahydrofuran lignans: Melanogenesis inhibitors from Premna integrifolia wood collected in myanmar. Fitoterapia 2018, 127, 308–313. [Google Scholar] [CrossRef]
- Tuerxuntayi, A.; Liu, Y.Q.; Tulake, A.; Kabas, M.; Eblimit, A.; Aisa, H.A. Kaliziri extract upregulates tyrosinase, TRP-1, TRP-2 and MITF expression in murine B16 melanoma cells. BMC Complement. Altern. Med. 2014, 14, 166. [Google Scholar] [CrossRef]
- Wang, Y.; Duan, T.; Hong, M.; Zhou, Y.; Huang, H.; Xiao, X.; Zheng, J.; Zhou, H.; Lu, Z. quantitative proteomic analysis uncovers inhibition of melanin synthesis by silk fibroin via MITF/tyrosinase axis in B16 melanoma cells. Life Sci. 2021, 284, 119930. [Google Scholar] [CrossRef]
- Bowman, R.M.; Grundon, M.F. Quinoline Alkaloids. Part V1II.l The synthesis and nuclear magnetic resonance spectra of (±)-platydesmine, (±)-isobalfourodine, and related compounds. J. Chem. Soc. 1966, 10, 1504–1507. [Google Scholar]
- Sampaio, O.M.; Vieira, L.C.C.; Bellete, B.S.; King-Diaz, B.; Lotina-Hennsen, B.; da Silva, M.; Veiga, T.A.M. Evaluation of alkaloids isolated from Ruta graveolens as photosynthesis inhibitors. Molecules 2018, 23, 2693. [Google Scholar] [CrossRef] [PubMed]
- Tran, N.M.A.; Do, T.H.T.; Truong, L.H.; Le, D.T.; Phan, M.N.; Pham, N.K.T.; Mai, D.T.; Nguyen, T.P. Acridone alkaloids from the rhizomes of Luvunga scandens (Roxb.) Buch. Ham. Nat. Prod. Res. 2019, 33, 2176–2181. [Google Scholar] [CrossRef]
- Kitamura, S.; Hashizume, K.; Iida, T.; Miyashita, E.; Shirahata, K.; Kase, H. Studies on lipoxygenase inhibitors. II. KF8940 (2-n-heptyl-4-hydroxyquinoline-N-oxide), a potent and selective inhibitor of 5-lipoxygenase, produced by Pseudomonas methanica. J. Antibiot. 1986, 39, 1160–1166. [Google Scholar] [CrossRef]
- Kostova, I.; Ivanova, A.; Mikhova, B.; Klaiber, I. Alkaloids and coumarins from Ruta graveolens. Monatshefte Chem./Chem. Mon. 1999, 130, 703–707. [Google Scholar] [CrossRef]
- Oliva, A.; Meepagala, K.M.; Wedge, D.E.; Harries, D.; Hale, A.L.; Aliotta, G.; Duke, S.O. Natural fungicides from Ruta graveolens L. leaves, including a new quinolone alkaloid. J. Agric. Food Chem. 2003, 51, 890–896. [Google Scholar] [CrossRef]
- Singh, S.; Nerella, S.; Pabbaraja, S.; Mehta, G. Access to 2-alkyl/aryl-4-(1 H )-quinolones via orthogonal “NH3” insertion into o -Haloaryl Ynones: Total synthesis of bioactive pseudanes, graveoline, graveolinine, and waltherione F. Org. Lett. 2020, 22, 1575–1579. [Google Scholar] [CrossRef]
- Van Nam, V.; Huong, D.T.M.; Dao, P.T.; Anh, N.M.; Minh, L.T.H.; Van Cuong, P. Secondary metabolites produced by marine actinomycete Micromonospora sp. A258. Vietnam. J. Chem. 2019, 57, 707–711. [Google Scholar] [CrossRef]
- Zhou, Y.; Wu, J.; Zou, K. Xylogranatinin, a new pyrido[1, 2-a]pyrazine alkaloid from the fruit of a Chinese mangrove Xylocarpus granatum. Chem. Nat. Compd. 2007, 43, 426–428. [Google Scholar] [CrossRef]
- Bergenthal, D.; Mester, I.; Rózsa, Z.; Reisch, J. 13C-NMR-spektren einiger acridon-alkaloide. Phytochemistry 1979, 18, 161–163. [Google Scholar] [CrossRef]
- Bhattacharyya, J.; Serur, L. The minor alkaloids of Monnieria-Trifolia L. Heterocycles 1983, 20, 1063–1066. [Google Scholar] [CrossRef]
- Nganou, B.K.; Mbaveng, A.T.; Fobofou, S.A.T.; Fankam, A.G.; Bitchagno, G.T.M.; Simo Mpetga, J.D.; Wessjohann, L.A.; Kuete, V.; Efferth, T.; Tane, P. Furoquinolines and dihydrooxazole alkaloids with cytotoxic activity from the stem bark of Araliopsis soyauxii. Fitoterapia 2019, 133, 193–199. [Google Scholar] [CrossRef]
- Cardoso-Lopes, E.M.; Maier, J.A.; da Silva, M.R.; Regasini, L.O.; Simote, S.Y.; Lopes, N.P.; Pirani, J.R.; Bolzani Vda, S.; Young, M.C. Alkaloids from stems of Esenbeckia leiocarpa Engl. (Rutaceae) as potential treatment for alzheimer disease. Molecules 2010, 15, 9205–9213. [Google Scholar] [CrossRef]
- Jurd, L.; Wong, R. Quinoline alkaloids from the Heartwood of balfourodendron riedelianum. Aust. J. Chem. 1983, 36, 1615–1622. [Google Scholar] [CrossRef]
- Parhoodeh, P.; Rahmani, M.; Hashim, N.M.; Sukari, M.A.; Ee, G. Alkaloid constituents of Haplophyllum laeviusculum (Rutaceae). Sains Malays. 2012, 41, 47–52. [Google Scholar]
- Kuzovkina, I.; Al’terman, I.; Schneider, B. Specific accumulation and revised structures of acridone alkaloid glucosides in the tips of transformed roots of Ruta graveolens. Phytochemistry 2004, 65, 1095–1100. [Google Scholar] [CrossRef] [PubMed]
- Orhan Püsküllü, M.; Tekiner, B.; Suzen, S. Recent studies of antioxidant quinoline derivatives. Mini Rev. Med. Chem. 2013, 13, 365–372. [Google Scholar] [CrossRef] [PubMed]
- Ratheesh, M.; Sindhu, G.; Helen, A. Anti-inflammatory effect of quinoline alkaloid skimmianine isolated from Ruta graveolens L. Inflamm. Res. 2013, 62, 367–376. [Google Scholar] [CrossRef] [PubMed]
- Colucci-D’Amato, L.; Cimaglia, G. Ruta graveolens as a potential source of neuroactive compounds to promote and restore neural functions. J. Tradit. Complement. Med. 2020, 10, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Pollio, A.; De Natale, A.; Appetiti, E.; Aliotta, G.; Touwaide, A. Continuity and change in the Mediterranean medical tradition: Ruta spp. (rutaceae) in Hippocratic medicine and present practices. J. Ethnopharmacol. 2008, 116, 469–482. [Google Scholar] [CrossRef]
- Zhou, F.; Lu, X.-y.; He, F.; Wu, T.; Aisa, H.A.; Maiwulanjiang, M. Effects of traditional artiri La Li formulation on experimental models of vitiligo and mechanisms of melanogenesis. J. Ethnopharmacol. 2023, 302, 115884. [Google Scholar] [CrossRef] [PubMed]
- Tuna, A.; Ozturk, G.; Gerceker, T.B.; Karaca, E.; Onay, H.; Guvenc, S.M.; Cogulu, O. Superoxide dismutase 1 and 2 gene polymorphism in turkish vitiligo patients. Balkan J. Med. Genet. 2017, 20, 67–74. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pfaffenbach, K.T.; Nivala, A.M.; Reese, L.; Ellis, F.; Wang, D.; Wei, Y.; Pagliassotti, M.J. Rapamycin inhibits postprandial-mediated X-box-binding protein-1 splicing in rat liver. J. Nutr. 2010, 140, 879–884. [Google Scholar] [CrossRef]
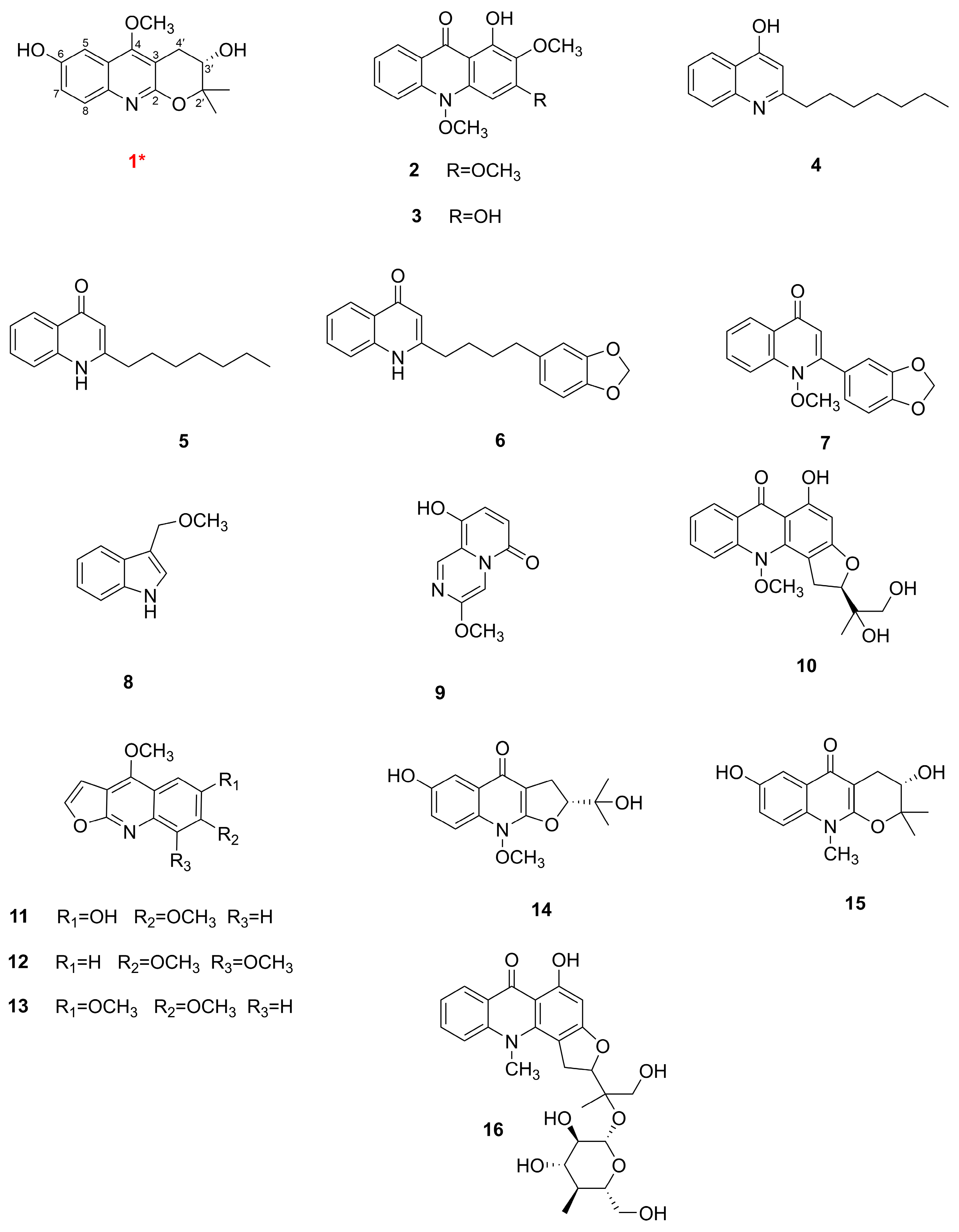
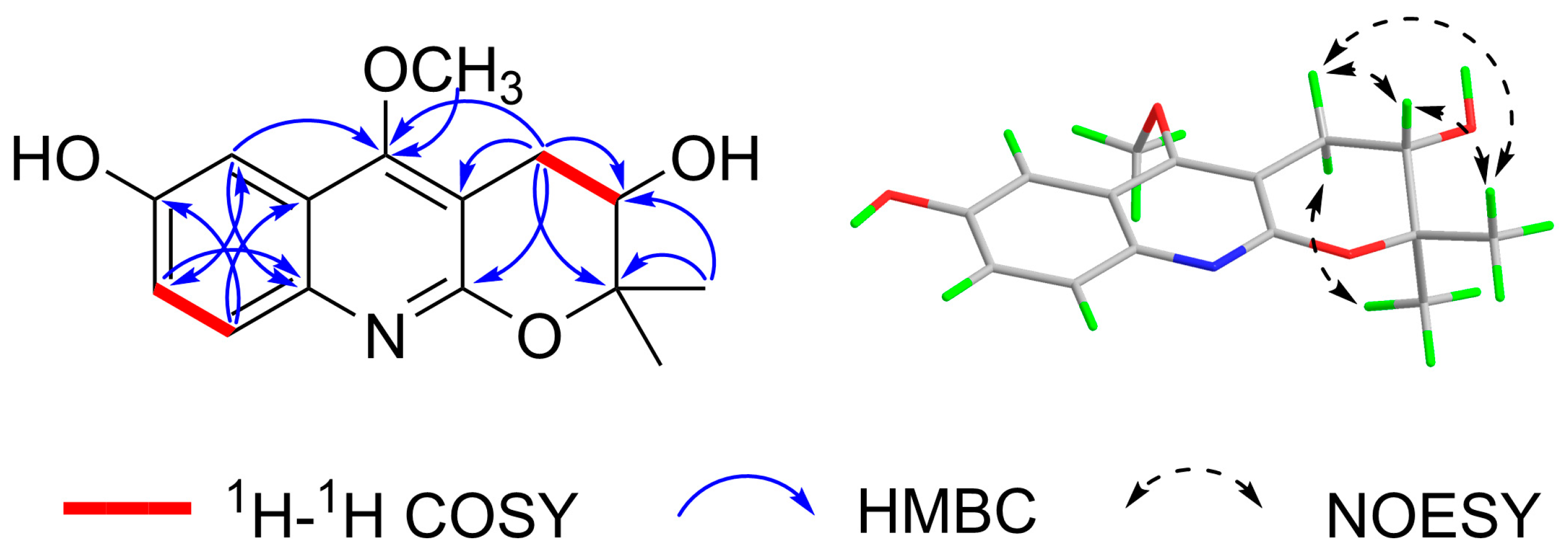
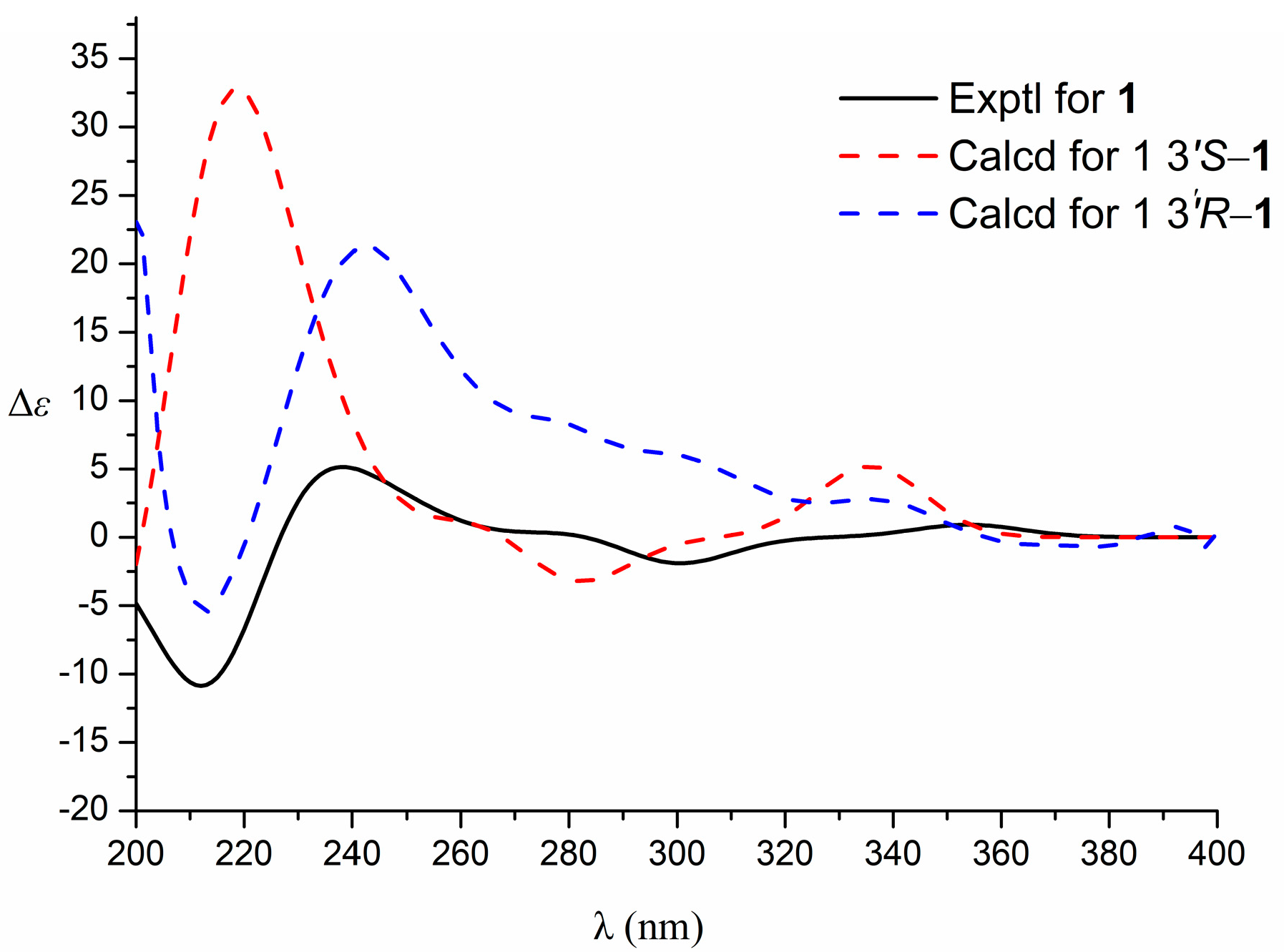





| No. | 1 | |
|---|---|---|
| 1H | 13C | |
| 2 | 159.6 | |
| 3 | 109.5 | |
| 4 | 164.0 | |
| 5 | 7.23 (1H, d, J = 2.8 Hz) | 104.4 |
| 6 | 155.3 | |
| 7 | 7.19 (1H, dd, J = 9.0, 2.8 Hz) | 122.9 |
| 8 | 7.56 (1H, d, J = 9.0 Hz) | 128.5 |
| 4a | 122.7 | |
| 8a | 142.5 | |
| 2′ | 80.4 | |
| 3′ | 3.90 (1H, dd, J = 6.6, 5.1 Hz) | 69.2 |
| 4′α | 2.94 (1H, dd, J = 17.1, 6.6 Hz) | |
| 4′β | 3.21 (1H, dd, J = 17.1, 5.1 Hz) | |
| 2′α-CH3 | 1.41(3H, s) | 22.2 |
| 2′β-CH3 | 1.44(3H, s) | 26.1 |
| 4-OCH3 | 4.01(3H, s) | 61.6 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ainiwaer, P.; Li, Z.; Zang, D.; Jiang, L.; Zou, G.; Aisa, H.A. Ruta graveolens: Boost Melanogenic Effects and Protection against Oxidative Damage in Melanocytes. Antioxidants 2023, 12, 1580. https://doi.org/10.3390/antiox12081580
Ainiwaer P, Li Z, Zang D, Jiang L, Zou G, Aisa HA. Ruta graveolens: Boost Melanogenic Effects and Protection against Oxidative Damage in Melanocytes. Antioxidants. 2023; 12(8):1580. https://doi.org/10.3390/antiox12081580
Chicago/Turabian StyleAiniwaer, Pazilaiti, Zuopeng Li, Deng Zang, Lan Jiang, Guoan Zou, and Haji Akber Aisa. 2023. "Ruta graveolens: Boost Melanogenic Effects and Protection against Oxidative Damage in Melanocytes" Antioxidants 12, no. 8: 1580. https://doi.org/10.3390/antiox12081580
APA StyleAiniwaer, P., Li, Z., Zang, D., Jiang, L., Zou, G., & Aisa, H. A. (2023). Ruta graveolens: Boost Melanogenic Effects and Protection against Oxidative Damage in Melanocytes. Antioxidants, 12(8), 1580. https://doi.org/10.3390/antiox12081580







