Abstract
The maintenance of redox homeostasis is associated with a healthy status while the disruption of this mechanism leads to the development of various pathological conditions. Bioactive molecules such as carbohydrates accessible to the microbiota (MACs), polyphenols, and polyunsaturated fatty acids (PUFAs) are food components best characterized for their beneficial effect on human health. In particular, increasing evidence suggests that their antioxidant ability is involved in the prevention of several human diseases. Some experimental data indicate that the activation of the nuclear factor 2-related erythroid 2 (Nrf2) pathway—the key mechanism in the maintenance of redox homeostasis—is involved in the beneficial effects exerted by the intake of PUFAs and polyphenols. However, it is known that the latter must be metabolized before becoming active and that the intestinal microbiota play a key role in the biotransformation of some ingested food components. In addition, recent studies, indicating the efficacy of the MACs, polyphenols, and PUFAs in increasing the microbial population with the ability to yield biologically active metabolites (e.g., polyphenol metabolites, short-chain fatty acids (SCFAs)), support the hypothesis that these factors are responsible for the antioxidant action on the physiology of the host. The underlying mechanisms through which MACs, polyphenols, and PUFAs might influence the redox status have not been fully elucidated, but based on the efficacy of SCFAs as Nrf2 activators, their contribution to the antioxidant efficacy of dietary bioactives cannot be excluded. In this review, we aimed to summarize the main mechanisms through which MACs, polyphenols, and PUFAs can modulate the host’s redox homeostasis through their ability to directly or indirectly activate the Nrf2 pathway. We discuss their probiotic effects and the role played by the alteration of the metabolism/composition of the gut microbiota in the generation of potential Nrf2-ligands (e.g., SCFAs) in the host’s redox homeostasis.
1. Introduction
Oxidative eustress represents the physiological exposure to low doses of endogenous oxidant species, produced by cells to address specific targets via the redox network to maintain cell homeostasis [1]. The alteration of the redox status is largely involved in cells and consequent organ dysfunction, which often leads to a wide variety of chronic and age-related human diseases. One of the molecular pathways responsible for the preservation of this equilibrium state is the Nrf2/Keap1 pathway. Among several pathways regulated by Nrf2, there is the maintenance of proteostasis, whose dysfunction determines cell death by autophagy or apoptosis caused by events such as protein misfolding and aggregation [2].
The intestine is an essential organ involved in human nutrition, and increasing evidence indicates that the interplay between gut commensal bacteria (microbiota)—the complex microbial community that colonizes the human gut—and its composition is influenced by the host’s genotype, environment, and diet. In particular, food nutrients play a key role in human metabolism and health via the modulation of multiple mechanisms, including energy metabolism, intestinal homeostasis, antioxidant homeostasis, and immune responses [3]. In particular, the metabolic activity of gut microbes is essential for maintaining host health, and alterations in its composition induce metabolic shifts that may have adverse effects. Under healthy conditions, the preponderance of potentially beneficial bacterial species such as Firmicutes and Bacteroides over potentially pathogenic ones such as Proteobacteria is called eubiosis, and it has been associated with a healthy status of the host organism. On the contrary, the perturbation or disruption of this composition, known as dysbiosis, has been associated with several metabolic or immune disorders [4].
The consensus on microbiota-mediated healthy effects on the host is based on the microbe-induced biotransformation of food components into bioactive metabolites. Bioactive molecules exhibit, in combination with food components, the ability to modulate the metabolic pathways of the host or to modify the composition and metabolism of the microbiota. Among them, non-digestible fibers, also known as microbiota-accessible carbohydrates (MACs), polyphenols, and PUFAs, are the best-characterized food components influencing the composition and metabolism of the microbiota [5].
Analogously, the regulation/activation of the Nrf2 pathway can be exerted by endogenous ROS (Reactive Oxygen Species) or exogenous molecules, such as dietary bioactive molecules that may need to undergo structural transformations by the gut microbiota before performing their bioactivity. The link between Nrf2 and gut microbiota health may be hypothesized on the basis of the association of Nrf2 dysfunction with the alteration of the composition of the microbiota due to ageing or pathological conditions. Unfortunately, there is only one study that supports this connection [6] and, as far as we know, the molecules responsible for this crosstalk have not yet been indicated.
In this review, we first discuss the role of microbial by-products, such as short-chain fatty acids (SCFAs), on Nrf2-mediated oxidoreductive homeostasis (redox status). Then, we provide a comprehensive and updated overview of the interplay of MACs, polyphenols, and PUFAs on the composition/metabolism of the gut microbiota and their direct action on specific molecular targets or their indirect action—via the modulation of the composition of the microbiota—which, downstream, modulates the host’s redox status via the production of SCFAs (Figure 1).
Figure 1.
Possible role of SCFAs in the modulation of Nrf2-mediated redox homeostasis.
2. Importance of the Nrf2 Pathway and Its Link with Gut Microbiota
Nrf2 is a redox-sensitive transcription factor, and it is the master regulator of oxidoreductive and immune homeostasis. In the cell cytoplasm, Nrf2 is associated with the inhibitory protein Kelch-such as ECH-associated Protein 1(Keap1), which, owing to the presence of specific cysteine residues, acts as a sensor of endogenous and exogenous prooxidants [7]. In particular, under a mild increase in oxidative stress, specific cysteine residues in Keap1 allow newly synthesized Nrf2 to escape Keap1-mediated ubiquitination and to activate the transcription of Nrf2-target genes (more than 200) involved in fundamental biochemical pathways (e.g., mitochondrial functions, oxidoreductive and immune homeostasis). Due to its pleiotropic activity, Nrf2 has been indicated as a therapeutical target for a variety of human diseases [8], and the activation of this molecular mechanism has been mainly involved in the cytoprotective activity of dietary antioxidants, including plant polyphenols [9], dietary PUFAs, and some of their metabolites [10].
As for SCFAs, bioactive polyphenol metabolites can affect specific pathways determining the modulation of specific target genes. In particular, their antioxidant activity is achieved through the activation of the Nrf2 pathway [11]. However, based on their low bioavailability and their biotransformation by the intestinal microbiota, it is probable that their antioxidant ability is mediated by some of their metabolites or by other molecules of bacterial origin (e.g., SCFAs). These fatty acids may be at the crossroad between the diet and the organism’s redox status.
This hypothesis is consistent with a recent result showing the positive correlation between Nrf2 activation in the brain with the levels of SCFA-producing bacteria (e.g., Roseburia, Oscillibacter, Faecalibaculum) in mice treated with several Nrf2 activators [6]. In this framework, an overview of the modulatory effects of MACs, PUFAs, and polyphenols to increase the level of SCFA-producing bacteria along with data reporting the efficacy of SCFAs as Nrf2 ligands will be summarized in the following sections.
3. Composition of the Gut Microbiota
The gut microbiota comprise thousands of bacterial species, mainly those of Bacteroidetes (9–42%), Firmicutes (30–52%), and Actinobacteria (1–13%) [12,13], whose composition is influenced by the environment and host genotype, as well as by age and diet. In adults, bacterial cells can reach gut concentrations of up to 1014 cells, representing the largest number and highest concentration of microorganisms found in the human body [14]. The great variety of bacterial species characterizing the microbiota results in the expression of a large number of genes. It has been estimated that the whole genomic content of gut microbiota exceeds that of humans by one hundred times, suggesting that the genome of the microbiota displays a metabolic potential capacity to influence the physiology of the host [15]. Recent advances in high-throughput sequencing technologies have allowed us to easily identify the genomes of ecosystem samples, contributing to the comprehension of the role of the gut microbiome in health and disease (Human Microbiome Project) [16].
The homeostatic condition of the intestinal microbiota (eubiosis) mainly depends on the balanced diversity of these microbial populations and the controlled growth of potentially pathogenic bacteria. A reduction in the diversity of the intestinal population (dysbiosis) can directly affect the epithelial and mucosal functions, leading to an inflammatory environment in the gastrointestinal tract [17] as well as other human pathologies such as non-alcoholic fatty liver disease and neurodegenerative disorders. The activity of the gut microbiota is essential in the host metabolism, protecting against infections from pathogens and intervening in energy homeostasis and the immune response by coordinating specific gene expression in response to different host and environmental signals [18]. In particular, the Firmicutes to Bacteroidetes ratio (F/B ratio) has been extensively examined in the human and mouse gut microbiota, and it has been demonstrated that the F/B ratio is associated with metabolic diseases [19], inflammatory diseases [20], neuropsychiatric disorders, and cancer [21,22,23].
4. Effect of SCFAs on the Composition of the Gut Microbiota
The beneficial effects associated with the diversity of the microbial population arise from the metabolic activities of specific microbial populations. Under eubiotic conditions, the commensal relationship between the microbiota and the host mainly consists of the capacity of bacteria to generate bioactive metabolites, starting from the ingested food, which exhibits the ability to modulate different metabolic pathways of the host [24]. For example, the production of carboxylic acids with aliphatic tails with fewer than six carbon atoms such as acetate (C2), propionate (C3), and butyrate (C4), resulting from the anaerobic fermentation of dietary plant polysaccharides, is the most relevant metabolic activity of enteric microbiota. These molecules are collectively referred to as Short-chain Fatty Acids (SCFAs) [25].
The growth of anaerobic SCFA-producing bacteria is favored by the low oxygen concentrations in the intestine where the two most abundant populations, namely, Bacteroidetes and Firmicutes, mainly produce acetate/propionate and butyrate, respectively [26]. Interestingly, due to butyrate generation during acetate metabolism, their coexistence can be consequential to mutual metabolic gain, thus resulting from the utilization of acetate produced by Bacteroidetes and Firmicutes to produce butyrate and propionate [27]. This example strongly supports the concept that the production of SCFAs is finely tuned by the balance of the bacterial species present in the gut.
The homeostatic condition of the intestinal microbiota can be restored by the level of SCFAs, and many studies in vivo describe the link between gut dysbiosis and the production of SCFAs (Table 1).

Table 1.
Studies reporting the link between gut dysbiosis/production of SCFAs in several human diseases. An increase or decrease in the levels considered is indicated by upward (↑) or downward arrow (↓), respectively.
It can be assumed that butyrate, being a fundamental nutrient for colonocytes, satisfies the metabolic demands of the colon epithelium [42], and it also modulates the expression of tight junction proteins, thus preserving the intestinal barrier whose integrity is a crucial part of the overall immune response [43]. In addition, local O2 consumption during butyrate uptake and its metabolism by the intestinal epithelium stabilizes the hypoxia-inducible factor (HIF)—a transcription factor that coordinates barrier protection—which promotes the creation of an anaerobic environment. This “physiological hypoxia” stimulates the growth of SCFA-producers (anaerobic bacteria) [44], indirectly regulating the functionality of the intestinal barrier [45]. In addition, SCFAs have been shown to display an inhibitory effect on the growth of potentially pathogenic bacteria such as Salmonella typhimurium [46] or Clostridium difficile [47].
4.1. Effect of SCFAs on Gut Homeostasis
Acetate, propionate, and butyrate in the colon are present in the molar ratio 60:25:15, although proportions can vary depending on factors such as diet, microbiota composition, the site of fermentation, and the genotype of the host [48]. These are the predominant SCFAs present in the proximal regions of the large intestine in humans and rodents, and they are present at mM levels [49,50,51].
Acetate, propionate, and butyrate reach the highest concentrations (70–140 mM) in the proximal colon [48] where they enhance mucin secretion by increasing the expression of the MUC2 gene [52], with a concentration gradient decreasing from the lumen to the periphery [53]. When these SCFAs are absorbed into hepatic portal circulation and the lacteal lymphatic system, they reach total concentrations ranging from 375 μM to 148 μM in the portal and hepatic blood respectively, or 79 μM in peripheral blood [48,54]. Butyrate and propionate, mostly metabolized by hepatocytes, were reported in a range of 1–15 μM in the systemic circulation, while acetate ranged between 100 and 200 μM [55,56]. However, the small amounts of SCFAs present in the bloodstream are sufficient to elicit a wide range of biological functions in different tissues.
A study on a mouse model of induced colitis demonstrated that SCFAs preserve gut homeostasis by acting on the inflammasome pathway through the upregulation of interleukin (IL)-18 [57]. Accordingly, low levels of butyrate and propionate-producing bacteria were found in patients suffering from inflammatory bowel diseases (IBD) such as ulcerative colitis or Crohn’s disease [31,32]. Several in vivo analyses have indicated that SCFAs regulate gut motility by stimulating mucosal receptors [58] or by increasing the release of the Peptide YY from gut endocrine cells, thus favoring intestine motility [59]. Other studies demonstrated that SCFAs act in preventing colonic diseases, by enhancing the absorption of minerals and decreasing the cholesterol concentration [60,61]. Experiments using germ-free animals reported that their reduced gut motility can be restored by the infusion of SCFAs [62].
Convincing evidence supports the idea that the beneficial effect of SCFAs extends beyond the colon. In fact, SCFAs participate in different physiological processes in the human body, being able to improve gut physiology, modulate the host’s glucose and lipid metabolism, and affect immune function [63,64]. In particular, upon their transport from the intestinal lumen into the blood compartment of the host, SCFAs are absorbed by the liver for gluconeogenesis or by muscle to generate energy [65]. Among SCFAs, acetate is the primary substrate for cholesterol synthesis [66], while propionate inhibits cholesterol synthesis by reducing serum lipids and has a protective effect against colon cancer [64,67]. Notably, SCFAs modulate brain functions by acting on the production of neuroactive metabolites [68]. For example, butyrate and propionate can be transferred from the gut to the brain where they act as signalling molecules through the monocarboxylate transporters that are highly expressed in the blood–brain barrier [69]. Finally, butyrate has been reported to play a protective role against carcinogenesis in colon cancer cells by enhancing the expression of cell cycle inhibitory genes [70].
4.2. Signaling Mechanisms Induced by SCFAs
The detailed description of the mechanisms involved in the signalling activity of SCFAs does not fall within the scope of this work and, due to its complexity, only a synthetic presentation is reported herein. Besides the relevant role in intestinal health, SCFAs may play their signalling role via the activation of several biochemical pathways: G-protein-coupled receptors (GPCRs), histone deacetylases (HDACs), and Nrf2 [71,72,73] (Figure 2).
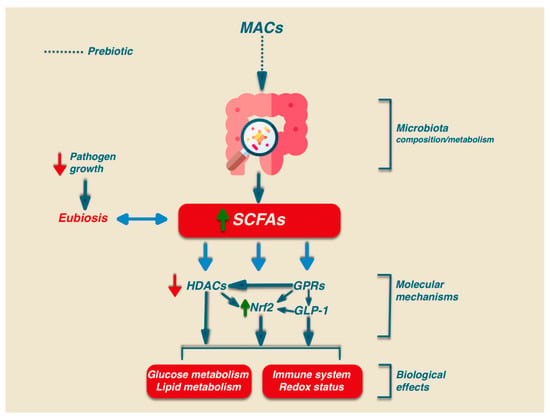
Figure 2.
Biological effects of MACs. The intake of MACs can modulate the composition and metabolism of the microbiota, leading to an increased production of SCFAs. SCFAs exert biological effects by modulating specific signaling pathways. HDACs: histone deacetylases; GPCRs: G-protein-coupled receptors; GLP-1: glucagon like peptide 1; Nrf2: Nuclear factor 2-related erythroid 2.
In humans, there are at least six GPRs that are sensitive to SCFAs, but among them, only GPR41, GPR43, and GPR109A are involved in SCFA-mediated signaling. GPR41 and GPR43 are the best-studied SCFA receptors [74] and are activated by acetate, propionate, and, to a lesser extent, also by butyrate. GPR41 is expressed in colon cells, in the blood vessels, and in the sympathetic nervous system, while GPR43 is mainly expressed in enteroendocrine L cells, lymphocytes, neutrophils, and monocytes [75]. GPR109A has a high affinity for niacin that can be activated by butyrate, and it is expressed only in human immune cells and colonocytes. In addition, GPR109A is highly expressed in adipocytes. The activation of this receptor in adipocytes has been linked to lipolysis and a decrease in plasma free fatty acids [76]. Activated GPCR receptors can regulate different signaling via the activation of many cellular functions such as the mitogen-activated protein kinase (MAPK) family of serine-threonine kinases, including extracellular signal-regulated kinase (ERK), c-jun N-terminal kinase (JNK), p38, and ERK5, through an intricate network of signaling. The activation of GPR43 also stimulates the phospholipase-C determining intracellular Ca2+ release and the activation of protein kinase C [77].
HDACs are a group of enzymes that affect gene transcription or alter protein activity by removing the acetyl group on the lysine ϵ-amino group of the target protein. The inhibition of HDACs is relevant for immune and inflammatory regulation by modulating either innate immunity through regulation of the Toll-Like Receptor (TLR) and Interferon (IFN) signaling pathways or by regulating antigen presentation and B and T lymphocytes to achieve adaptive immunity [78,79]. In particular, the inhibitory effect of HDACs on SCFAs, mainly due to propionate and butyrate, results in an anti-inflammatory effect through the promotion of regulatory T cell (Treg) development as well as CD4+ T cell IL-10 production [80,81].
Interestingly, the functional link existing between this signaling pathway and gut microbiota homeostasis has been indicated by (a) the modulatory ability of SCFAs in the Nrf2 pathway [72], (b) the age-dependent decline in the concentration of SCFAs in the gut [82], and (c) the positive association between microbiota diversity and Nrf2 efficacy [83]. In addition, the link between the production of SCFAs and the Nrf2 pathway was indicated in a recent study showing the ability of Clostridium butyricum pretreatment to increase the SCFA contents in the cecum of Enterotoxigenic Escherichia coli K88 (ETEC K88)-infected mice. In particular, the data indicated that such improvement was associated with the amelioration of the oxidative damage induced by ETEC K88 infection through the activation of the Nrf2 pathway [84]. A summary of the differential ability of microbial SCFAs in activating different receptors involved in the Nrf2 pathway is shown in Table 2.

Table 2.
A brief summary of SCFAs produced by the gut microbial population and of their response to different receptors; adapted from [26,72]. Low or high affinity is denoted by + or ++, respectively.
Finally, the interplay existing between different SCFAs further strengthens the complexity of their mechanism of action [85,86] (Figure 2).
5. Dietary Bioactive Molecules and the Gut Microbiota Composition
Diet has a fundamental role in determining the composition of the gut microbiota. MACs, PUFAs, and polyphenols are the most well-characterized food components able to modulate the composition and metabolism of microbiota. All of these food components can exert prebiotic effects that result in the modulation of bacterial strains producing SCFAs. These compounds can affect specific molecular mechanisms that result directly in beneficial effects on the host’s health or indirectly allow the gut microbiota to produce active/antioxidant metabolites. A detailed description of the complex network involved in the SCFA-mediated signaling activity and its therapeutic relevance have been presented elsewhere by [87] and will not be further examined herein.
5.1. Effect of Microbiota-Accessible Carbohydrates (MACs) on Gut Homeostasis
Non-digestible polysaccharides such as resistant starch, inulin, cellulose, guar gum, and pectin—collectively known as MACs—are the main energetic source of gut bacteria. MACs exert a modulatory role in the gut microbial composition, maintaining gut homeostasis. An MAC-rich diet in humans is associated with an increased content of colonic and fecal SCFAs (Table 3). On the contrary, a high-fat and high-sucrose diet can determine dysbiosis onset, which represents the first step in the development of an increased susceptibility to inflammatory diseases such as IBD or non-alcoholic fatty liver disease or colon cancer [88].
After ingestion, intact MACs reach the colon where they are metabolized by microbial enzymes, i.e., glycoside hydrolases and polysaccharide lyases. These enzymes, degrading the glycosidic bonds, convert MACs into monosaccharides [89]. The digestion of MACs by gut bacteria yields SCFAs that can be easily absorbed by gut epithelial cells, exerting healthy effects on human health [90]. It is important to underline that food intake has a crucial role in the fine-tuning of SCFA production by gut bacterial species. This implies that dietary changes affecting the MAC content can exert a prebiotic effect, altering the composition and metabolism of the gut microbiota. As previously mentioned, the eubiotic condition is associated with “good health,” while low microbial diversity and dysbiosis have been correlated with diseases highly prevalent in Western society, such as obesity, type 2 diabetes, or IBD [91]. Several studies have shown a positive correlation between the intake of a vegetable diet enriched in fiber and microbiota diversity and the enrichment of SCFA-producing bacteria in human populations [92,93].

Table 3.
The effect of MACs on the composition of microbiota and the production of SCFAs. An increase or decrease in the levels considered is indicated by upward (↑) or downward arrow (↓), respectively.
Table 3.
The effect of MACs on the composition of microbiota and the production of SCFAs. An increase or decrease in the levels considered is indicated by upward (↑) or downward arrow (↓), respectively.
| Treatment | Model | Microbiota Alteration/SCFA Production | Ref. |
|---|---|---|---|
| Metanalysis | Studies investigating the effect of dietary fiber on gut microbiota | ↑ Bifidobacterium ↑ SCFAs | [29] |
| Dietary Fiber | European children (Low-fiber diet) vs. Rural African village (High-fiber diet) | ↑ Bacteroidetes ↑ SCFAs | [92] |
| African Americans vs. Rural native Africans | ↑ Prevotella ↑ SCFAs | [94] | |
| Inulin | Mice with hyperuricemia vs. wild-type mice | ↑ microbial diversity ↑ SCFA-producing bacteria (Akkermansia and Ruminococcus). ↑ acetate, propionate, and butyrate | [95] |
| Nonalcoholic Fatty Liver Disease rat model | ↑ Bifidobacterium, Phascolarctobacterium, Blautia ↓ Acetate ↑ Propionate and Butyrate | [96] |
5.2. Effect of Polyphenols on Gut Homeostasis
Polyphenols are dietary bioactive compounds derived from plants and present in fruits and vegetables. These compounds are chemically characterized by the presence of at least one phenyl ring and one or more hydroxyl substituents. Dietary polyphenols are characterized by poor absorption and extensive metabolism [97].
Most of the dietary-ingested polyphenols are present as glycosylated ester or polymer forms, and after ingestion, they are recognized as xenobiotics. Their structural complexity determines that they reach the large intestine without modifications. In the gut, polyphenols—through microbic metabolism—are converted into low-molecular-weight metabolites and then absorbed from epithelial cells to reach the plasma.
Small polyphenols can be easily absorbed after de-glycosylation by bacterial enzymes. Then, they are converted into soluble metabolites through Phase I (oxidation, reduction, and hydrolysis) and Phase II reactions (conjugation) [98]. On the contrary, complex polyphenols (oligomeric and polymeric) need to be transformed by specific gut enzymes to increase their bioavailability and plasma levels. As glycosides, polyphenols are first converted into aglycones through specific enzymatic transformations. Subsequently, they undergo additional modifications including C-ring cleavage, decarboxylation, dehydroxylation, and demethylation [99]. The released aglycones, after absorption into the small intestine, can be further metabolized by enterocytes and hepatocytes. In the liver cells, polyphenols undergo Phase II biotransformation with the production of glucuronide and sulfation metabolites, followed by distribution to organs and excretion in urine [100]. As a consequence, the final metabolites are quite different from the parent molecules present in the ingested food. In addition, their residence time in the plasma is extended compared to parental compounds, thus allowing them to exert biological effects [101].
Polyphenols are present in low concentrations in foods, as compared to macronutrients, and their low bioavailability results in a lower amount absorbed by the body. The strong discrepancy between the biological activity of polyphenols and their concentration has led to the formulation of the low bioavailability/high bioactivity paradox [102,103].
The biotransformation of polyphenols by gut microbiota and the capacity of polyphenols to modulate gut microbiota have been reported [104,105]. Consequently, less than 5% of the total polyphenolic intake is absorbed and reaches the plasma unchanged while their microbial metabolites predominate in the plasma [106].
For example, resveratrol, widely distributed in grapes, berries, and peanuts, is transformed by the gut microbiota into different metabolites: dihydroresveratrol (DHR), which is partly absorbed and further metabolized to two conjugated forms: monosulfate (DHR) and monoglucuronide (DHR). An analysis of the bioavailability of these microbial metabolites indicates that upon daily intake of 500 mg of pure trans-resveratrol, the metabolite concentration in plasma increased from 3 to 13 μM [107].
Curcumin, found in turmeric (Curcuma longa (Linn.)), is converted into active metabolites in a two-step reaction: the first produces dihydro curcumin (DHC) from curcumin, and then DHC is converted into tetrahydro curcumin (THC). It has been reported that supplementation with 1 g of turmeric acid and curcumin was not detectable in plasma while the concentration of the active metabolites persisted in blood for at least 8 h, ranging from 2 to 47 nM [108].
Bioactive polyphenol metabolites can affect specific pathways determining the modulation of specific target genes. Quercetin, a glycoside present in many fruits and vegetables, is transformed into active glucuronated, sulfated, and methylated metabolites in the enterocytes of the small intestine. Quercetin that is not absorbed is metabolized by specific bacterial enzymes able to cleave the quercetin C-ring, producing DOPAC (3,4-Dihydroxyphenylacetic acid) PCA (3,4-dihydroxybenzoic acid), and 3-OPAC (3-hydroxyphenyl acetic acid). These metabolites may exert their effect by directly modulating different signaling pathways (Figure 3), such as the activation of the Nrf2 pathway that is involved in their antioxidant activity [11].
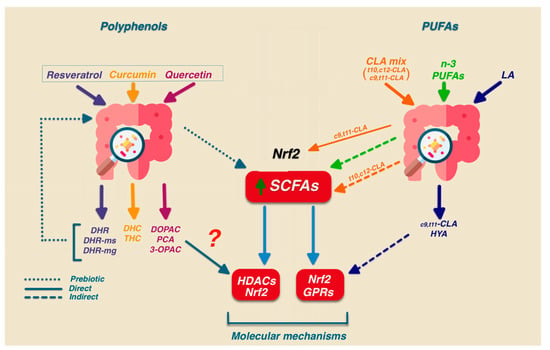
Figure 3.
Antioxidant effects of polyphenols and PUFAs. Polyphenols and PUFAs can act indirectly or directly. Indirect effects act by favoring the growth of a specific microbial population, while direct effects affect the physiology of the host by modulating the Nrf2 pathway. DHR: Dihydroresveratrol DHR-ms: Dihydroresveratrol monosulfate; DHR-mg: Dihydroresveratrol monoglucuronide forms; DHC: Dihydrocurcumin; THC: tetrahydro curcumin; DOPAC 3,4-Dihydroxyphenylacetic acid; PCA: 3,4-dihydroxybenzoic acid; 3-OPAC: 3-hydroxyphenylacetic acid; HYA: 10-hydroxy-cis-12-octadecenoic acid.
As for SCFAs, bioactive polyphenol metabolites can affect specific pathways determining the modulation of specific target genes. For example, their antioxidant activity is achieved through the activation of the Nrf2 pathway. Several in vitro and in vivo studies demonstrated that polyphenols can activate the Nrf2 pathway [11]; however, owing to their low bioavailability and their bio-transformation by intestinal microbiota, whether polyphenols exert their modulatory effect directly or indirectly—through their metabolites—remains to be established.
Different microbial genera including Bacteroides, Enterococcus, and Eubacteria play a key role in determining the metabolic fate of polyphenols [109]. In addition, some polyphenol bio-transformations require the presence of specific bacteria. For example, gut bacteria involved in resveratrol metabolism are Slackia equolifaciens and Adlercreutzia equolifaciens. Curcumin is converted into active metabolites mainly by Escherichia coli, but it can also be metabolized by Blautia sp. [110], and quercetin can be converted into active metabolites by specific bacterial strains such as Eubacterium ramulus, Clostridium orbiscindens, Eubacterium oxidoreducens, and Butyrovibrio spp. that can cleave the C-ring [111]. The importance of gut bacteria for polyphenol metabolism is further indicated by the fact that germ-free animals are not able to produce phenolic metabolites [112]. However, individual differences in gut microbiota composition can account for individual variations in absorption, metabolism, and polyphenol bioactivity. Thus, the identification of bacterial species responsible for polyphenol metabolism is crucial to unraveling the health-promoting effects on the host [113]. Individual differences in microbiota compositions can influence the metabolic fate of ingested polyphenols [114]. This can explain the reason why a similar daily intake of polyphenols results in different health effects.
In addition, their conversion into bioactive metabolites can have beneficial effects on the host’s health. In particular, the intestinal microbiota conversion of dietary polyphenols promotes the proliferation of SCFA-producing bacteria such as Bifidobacteria and decreases the ratio of Firmicutes to Bacteroidetes. Polyphenols can alter the composition of the microbiota, thus determining changes in polyphenol metabolism and bioavailability, and bioactive metabolites can have beneficial effects on the host’s health (Table 4).

Table 4.
Effects of the dietary supplementation of polyphenols on the composition of microbiota. An increase or decrease in the levels considered is indicated by upward (↑) or downward arrow (↓), respectively.
Figure 3 shows that the conversion of dietary polyphenols promotes the proliferation of SCFA-producing bacteria such as Bifidobacteria and decreases the ratio of Firmicutes to Bacteroidetes. This bidirectional interaction also accounts for polyphenol’s antioxidant effects.
5.3. Effect of Polyunsaturated Fatty Acids (PUFAs) and Conjugated Linoleic Acid (CLA) on Gut Homeostasis
n-3 PUFAs are a major component of fish fat and are widely ingested through food or supplements; they are involved in many biochemical processes in the human body and are well-known modulators of SCFAs produced by the gut microbiome [107].
Before the incorporation of PUFAs into the membrane of the target tissues from systemic circulation, dietary fats are emulsified in the stomach before they enter the small intestine, where they are cleaved off to form free fatty acids and 2-monoacylglyceride. In the enterocytes, PUFAs are re-esterified to triacyl glycerides, and, upon incorporation into chylomicrons, are transferred to the lymph and blood circulation. The concentrations of CLA and n-3 PUFAs in human blood plasma are markedly different—8 nm and 20 μM, respectively—but their significant increase can be induced by the dietary supplementation of CLAmix or fish oil [131,132]. Notably, since the biological activities of these PUFAs depend on their incorporation and metabolism in the target organ, the marked differences observed in the blood may not reflect the efficacy of their biological action [133].
Dietary lipids have been recognized to alter the gut microbiota composition by acting as substrates for bacterial metabolic processes or by modulating the growth of propionate- and butyrate-producing bacteria (e.g., Bacteroides, Clostridium, and Roseburia) [134]. A recent review article reported that the concentration of n-3 PUFAs in the blood is positively correlated with the abundance of human gut microbes, thus indicating that n-3 PUFAs could directly modulate the diversity and the abundance of gut microbiota [135]. In particular, dietary n-3 PUFAs regulate gut microbiota homeostasis, and their deficiency may induce dysbiosis [136]. Several clinical data indicate the ability of n-3 PUFAs to restore gut eubiosis in aged people or in those with several pathological conditions by increasing the abundance of butyrate-producing bacteria [137,138]. Other in vivo studies have reported that dietary n-3 PUFAs can increase the growth of lipopolysaccharide (LPS)-suppressing bacteria (Bifidobacteria) and limit that of LPS-producing bacteria (Proteobacteria) [139], whose presence can determine acute inflammatory responses by triggering the release pro-inflammatory cytokines. Interestingly, dietary lipids also have a relevant role in neural development, the differentiation of nerve cells, and the plasticity of the nervous system. Their importance is supported by in vivo and clinical trial studies that clearly show the crucial role of dietary n-3 PUFAs in brain development, ageing, and neurodegeneration (Table 5).

Table 5.
Effects of PUFA dietary supplementation on microbiota composition and SCFA production. An increase or decrease in the levels considered is indicated by upward (↑) or downward arrow (↓), respectively.
Conjugated Linoleic Acid (C18:2, CLA) is another PUFA that is attracting scientific interest due to its multiple beneficial effects, which, similarly to n-3 PUFAs, may be independent or dependent on the modulation of gut microbiota metabolism. The term CLA is the collective name generally used to indicate a further subclass of PUFAs without a methyl group between adjacent double bonds (conjugated diene of Linoleic Acid C18:2, LA). In particular, this generally refers to geometric and positional isomers of LA mainly present in dairy products and meat from ruminants (cis9, trans11 and trans10, cis12); the concentration of the former isomer typically ranges from 3 to 7 mg/g of fat. Commercially available dietary CLA supplement (CLAmix) is composed of an equimolar mixture of these two isomers. Dietary supplementation with the trans10, cis12 isomer is associated with the modulation of lipid metabolism and glucose tolerance, while the cis9, trans11 isomer has preeminent anti-oxidant and anti-inflammatory effects. The intake of an equimolar mixture of the two CLA isomers was suggested to effectively activate many biological pathways owing to the combined action of its isomers [149]. Moreover, dietary trans10, cis12 CLA exhibits the ability to increase the population of SCFA-producing bacteria and the cecal concentration of SCFAs (isobutyrate, acetate, and propionate) [150]. Similarly, trans10, cis12 CLA supplementation in a mouse model of obesity increased the level of butyrate-producing bacteria (Butyrivibrio, Roseburia, and Lactobacillus), leading to a significant increase in butyrate in the feces and in acetate in the plasma [151]. In this context, owing to the activity of the gut–brain axis, dysbiosis has been associated with the onset and progression of several neurological disorders [152]. In addition, the dysregulation of SCFA production has been linked to psychiatric illnesses and immune, metabolic, and neurodegenerative diseases [153].
In other studies, dietary LA may be indeed metabolized by several gut bacteria and converted into cis9, trans11 or some of its precursors (trans-11-18:1 or 10-hydroxy-18:1; 10-Oxo-trans-11-octadecenoic) [154]. These C18 fatty acids, similarly to SCFAs, exhibit the ability to activate the Nrf2 pathway [155,156,157,158]. Interestingly, some CLA by-products—such as 10-hydroxy-cis-12-octadecenoic acid (HYA) generated during Linoleic Acid metabolism—exhibit anti-inflammatory and antioxidant effects likely through GPR120-dependent activation [159,160] (Figure 3). Based on these data, it can be stated that the involvement of specific receptors/transporters in the CLA-mediated activation of Nrf2 needs further investigation. The possibility that CLA metabolites generated by gut microbiota may contribute to the biological activity of dietary CLA has been hypothesized [161]. However, different results have been presented, and the independence between the anti-inflammatory effect of CLA and the activity of the intestinal microbiota has recently been demonstrated [162].
6. Conclusions
In this review, we summarized the antioxidant mechanisms underlying dietary MACs, polyphenols, and PUFAs. The literature data reported in the field suggest that these molecules can exert their modulatory activity on the human redox status by acting in two main different ways: direct or indirect.
In particular, the antioxidant activity exhibited by PUFAs may be, at least in part, a consequence of their direct ability to target the Nrf2 pathway. In addition, based on the role of SCFAs as Nrf2 ligands, dietary MACs, polyphenols, and PUFAs may also have prebiotic activity, favoring the growth of SCFA-producing microbial populations, which are characteristic of eubiosis conditions.
Polyphenols and PUFA metabolites also exhibit an indirect effect, in which their biological activities can contribute to or enhance the effects triggered by the parent ingested molecules.
The key modulatory role played by SCFAs and polyphenol metabolites on the physiology of the host is mirrored by the network of molecular mechanisms underlying their direct activities, involving Nrf2 and HDAC signaling pathways and GPRs for SCFAs. Such complexity is further accentuated by the reciprocal interactions between these pathways.
SCFA production also represents important crossroads of the biological activities exerted by polyphenols and PUFAs.
The interplay among n-3 PUFA and CLA and gut microbiota is herein summarized for the first time, and the evidence of their direct and indirect actions on the physiology of the host likely contributes to expanding the nutritional and therapeutic importance of these dietary molecules.
In conclusion, the shared molecular pathways activated by polyphenols, PUFAs, and SCFAs support the role of Nrf2, HDACs, and GPRs as pharmacological targets. Since the activation of these pathways triggers a downstream signaling cascade, this can explain why dietary bioactive molecules can exert antioxidant/beneficial effects even when present in a low plasma concentration. A better understanding of the mechanisms underlying the prebiotic effects of polyphenols and PUFAs is an issue to be further explored that will shed light on the knowledge of their beneficial effects on human health.
Author Contributions
Conceptualization, P.B. and S.C.; writing and original draft preparation, R.C. and F.D.M.; writing, review and editing, P.B., S.C. and R.C.; visualization, R.C.; supervision, P.B., S.C. and S.F. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by CNR project NUTR-AGE (DSB.AD004.271) and by PON-MUR ALIFUN (ARS01_00783). This study was also carried out within the Agritech National Research Center and received funding from the European Union Next-GenerationEU (PIANO NAZIONALE DI RIPRESA E RESILIENZA (PNRR)-MISSIONE 4 COMPONENTE 2, INVESTIMENTO 1.4-D.D. 1032 17/06/2022, CN00000022). This manuscript reflects only the authors’ views and opinions; neither the European Union nor the European Commission can be considered responsible for them.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
The Ph.D. fellowship in Biology awarded to R.C. is supported by CNR/IRCCS Neuromed/Università degli Studi di Napoli Federico II “Dottorati Industriali” (XXXVI Cycle). We thank Chiara Nobile and Valentina Brasiello for their editing assistance. We thank Maria Rosaria Aletta for her bibliographic assistance.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Sies, H. Oxidative eustress: On constant alert for redox homeostasis. Redox Biol. 2021, 41, 101867. [Google Scholar] [CrossRef]
- Kurtishi, A.; Rosen, B.; Patil, K.S.; Alves, G.W.; Møller, S.G. Cellular Proteostasis in Neurodegeneration. Mol. Neurobiol. 2019, 56, 3676–3689. [Google Scholar] [CrossRef]
- Gentile, C.L.; Weir, T.L. The gut microbiota at the intersection of diet and human health. Science 2018, 362, 776–780. [Google Scholar] [CrossRef] [PubMed]
- Kataoka, K. The intestinal microbiota and its role in human health and disease. J. Med. Investig. 2016, 63, 27–37. [Google Scholar] [CrossRef]
- Rinninella, E.; Cintoni, M.; Raoul, P.; Lopetuso, L.R.; Scaldaferri, F.; Pulcini, G.; Miggiano, G.A.D.; Gasbarrini, A.; Mele, M.C. Food Components and Dietary Habits: Keys for a Healthy Gut Microbiota Composition. Nutrients 2019, 11, 2393. [Google Scholar] [CrossRef]
- Sadovnikova, I.S.; Gureev, A.P.; Ignatyeva, D.A.; Gryaznova, M.V.; Chernyshova, E.V.; Krutskikh, E.P.; Novikova, A.G.; Popov, V.N. Nrf2/ARE Activators Improve Memory in Aged Mice via Maintaining of Mitochondrial Quality Control of Brain and the Modulation of Gut Microbiome. Pharmaceuticals 2021, 14, 607. [Google Scholar] [CrossRef]
- Suzuki, T.; Yamamoto, M. Stress-sensing mechanisms and the physiological roles of the Keap1-Nrf2 system during cellular stress. J. Biol. Chem. 2017, 292, 16817–16824. [Google Scholar] [CrossRef]
- Cuadrado, A.; Manda, G.; Hassan, A.; Alcaraz, M.J.; Barbas, C.; Daiber, A.; Ghezzi, P.; León, R.; López, M.G.; Oliva, B.; et al. Transcription Factor NRF2 as a Therapeutic Target for Chronic Diseases: A Systems Medicine Approach. Pharmacol. Rev. 2018, 70, 348–383. [Google Scholar] [CrossRef] [PubMed]
- Qin, S.; Hou, D.X. Multiple regulations of Keap1/Nrf2 system by dietary phytochemicals. Mol. Nutr. Food Res. 2016, 60, 1731–1755. [Google Scholar] [CrossRef]
- Abrescia, P.; Treppiccione, L.; Rossi, M.; Bergamo, P. Modulatory role of dietary polyunsaturated fatty acids in Nrf2-mediated redox homeostasis. Prog. Lipid Res. 2020, 80, 101066. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Jiang, Z.; Lu, H.; Xu, Z.; Tong, R.; Shi, J.; Jia, G. Recent Advances of Natural Polyphenols Activators for Keap1-Nrf2 Signaling Pathway. Chem. Biodivers. 2019, 16, e1900400. [Google Scholar] [CrossRef]
- Bourlioux, P.; Koletzko, B.; Guarner, F.; Braesco, V. The intestine and its microflora are partners for the protection of the host: Report on the Danone Symposium “The Intelligent Intestine,” held in Paris, June 14, 2002. Am. J. Clin. Nutr. 2003, 78, 675–683. [Google Scholar] [CrossRef] [PubMed]
- Rajoka, M.; Shi, J.; Mehwish, H.; Zhu, J.; Li, Q.; Shao, D.; Huang, Q.; Yang, H. Interaction between diet composition and gut microbiota and its impact on gastrointestinal tract health. Food Sci. Hum. Wellness 2017, 6, 121–130. [Google Scholar] [CrossRef]
- Zhu, B.; Wang, X.; Li, L. Human gut microbiome: The second genome of human body. Protein Cell 2010, 1, 718–725. [Google Scholar] [CrossRef]
- Xu, J.; Mahowald, M.A.; Ley, R.E.; Lozupone, C.A.; Hamady, M.; Martens, E.C.; Henrissat, B.; Coutinho, P.M.; Minx, P.; Latreille, P.; et al. Evolution of symbiotic bacteria in the distal human intestine. PLoS Biol. 2007, 5, e156. [Google Scholar] [CrossRef]
- Consortium, H.M.P. Structure, function and diversity of the healthy human microbiome. Nature 2012, 486, 207–214. [Google Scholar] [CrossRef]
- Zhou, Y.; He, Y.; Liu, L.; Zhou, W.; Wang, P.; Hu, H.; Nie, Y.; Chen, Y. Alterations in Gut Microbial Communities Across Anatomical Locations in Inflammatory Bowel Diseases. Front. Nutr. 2021, 8, 615064. [Google Scholar] [CrossRef] [PubMed]
- Clemente, J.; Ursell, L.; Parfrey, L.; Knight, R. The Impact of the Gut Microbiota on Human Health: An Integrative View. Cell 2012, 148, 1258–1270. [Google Scholar] [CrossRef]
- Tilg, H. Obesity, metabolic syndrome, and microbiota: Multiple interactions. J Clin. Gastroenterol. 2010, 44 (Suppl. 1), S16–S18. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, D.; Jia, H.; Feng, Q.; Wang, D.; Liang, D.; Wu, X.; Li, J.; Tang, L.; Li, Y.; et al. The oral and gut microbiomes are perturbed in rheumatoid arthritis and partly normalized after treatment. Nat. Med. 2015, 21, 895–905. [Google Scholar] [CrossRef] [PubMed]
- Scheithauer, T.P.M.; Rampanelli, E.; Nieuwdorp, M.; Vallance, B.A.; Verchere, C.B.; van Raalte, D.H.; Herrema, H. Gut Microbiota as a Trigger for Metabolic Inflammation in Obesity and Type 2 Diabetes. Front. Immunol. 2020, 11, 571731. [Google Scholar] [CrossRef]
- Angelucci, F.; Cechova, K.; Amlerova, J.; Hort, J. Antibiotics, gut microbiota, and Alzheimer’s disease. J. Neuroinflamm. 2019, 16, 108. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.X.; Schwabe, R.F. The gut microbiome and liver cancer: Mechanisms and clinical translation. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 527–539. [Google Scholar] [CrossRef]
- Descamps, H.C.; Herrmann, B.; Wiredu, D.; Thaiss, C.A. The path toward using microbial metabolites as therapies. EBioMedicine 2019, 44, 747–754. [Google Scholar] [CrossRef]
- Koh, A.; De Vadder, F.; Kovatcheva-Datchary, P.; Bäckhed, F. From Dietary Fiber to Host Physiology: Short-Chain Fatty Acids as Key Bacterial Metabolites. Cell 2016, 165, 1332–1345. [Google Scholar] [CrossRef]
- Louis, P.; Flint, H.J. Formation of propionate and butyrate by the human colonic microbiota. Environ. Microbiol. 2017, 19, 29–41. [Google Scholar] [CrossRef]
- Mahowald, M.; Rey, F.; Seedorf, H.; Turnbaugh, P.; Fulton, R.; Wollam, A.; Shah, N.; Wang, C.; Magrini, V.; Wilson, R.; et al. Characterizing a model human gut microbiota composed of members of its two dominant bacterial phyla. Proc. Natl. Acad. Sci. USA 2009, 106, 5859–5864. [Google Scholar] [CrossRef]
- Zhao, L.; Zhang, F.; Ding, X.; Wu, G.; Lam, Y.Y.; Wang, X.; Fu, H.; Xue, X.; Lu, C.; Ma, J.; et al. Gut bacteria selectively promoted by dietary fibers alleviate type 2 diabetes. Science 2018, 359, 1151–1156. [Google Scholar] [CrossRef]
- Ojo, O.; Feng, Q.Q.; Ojo, O.O.; Wang, X.H. The Role of Dietary Fibre in Modulating Gut Microbiota Dysbiosis in Patients with Type 2 Diabetes: A Systematic Review and Meta-Analysis of Randomised Controlled Trials. Nutrients 2020, 12, 3239. [Google Scholar] [CrossRef]
- Imhann, F.; Vich Vila, A.; Bonder, M.J.; Fu, J.; Gevers, D.; Visschedijk, M.C.; Spekhorst, L.M.; Alberts, R.; Franke, L.; van Dullemen, H.M.; et al. Interplay of host genetics and gut microbiota underlying the onset and clinical presentation of inflammatory bowel disease. Gut 2018, 67, 108–119. [Google Scholar] [CrossRef]
- Machiels, K.; Joossens, M.; Sabino, J.; De Preter, V.; Arijs, I.; Eeckhaut, V.; Ballet, V.; Claes, K.; Van Immerseel, F.; Verbeke, K.; et al. A decrease of the butyrate-producing species Roseburia hominis and Faecalibacterium prausnitzii defines dysbiosis in patients with ulcerative colitis. Gut 2014, 63, 1275–1283. [Google Scholar] [CrossRef]
- Takahashi, K.; Nishida, A.; Fujimoto, T.; Fujii, M.; Shioya, M.; Innaeda, H.; Lnatorni, O.; Bamba, S.; Andoh, A.; Sugimoto, M. Reduced Abundance of Butyrate-Producing Bacteria Species in the Fecal Microbial Community in Crohn’s Disease. Digestion 2016, 93, 59–65. [Google Scholar] [CrossRef]
- Rau, M.; Rehman, A.; Dittrich, M.; Groen, A.K.; Hermanns, H.M.; Seyfried, F.; Beyersdorf, N.; Dandekar, T.; Rosenstiel, P.; Geier, A. Fecal SCFAs and SCFA-producing bacteria in gut microbiome of human NAFLD as a putative link to systemic T-cell activation and advanced disease. United Eur. Gastroenterol. J. 2018, 6, 1496–1507. [Google Scholar] [CrossRef]
- Tsai, M.C.; Liu, Y.Y.; Lin, C.C.; Wang, C.C.; Wu, Y.J.; Yong, C.C.; Chen, K.D.; Chuah, S.K.; Yao, C.C.; Huang, P.Y.; et al. Gut Microbiota Dysbiosis in Patients with Biopsy-Proven Nonalcoholic Fatty Liver Disease: A Cross-Sectional Study in Taiwan. Nutrients 2020, 12, 820. [Google Scholar] [CrossRef]
- Boursier, J.; Mueller, O.; Barret, M.; Machado, M.; Fizanne, L.; Araujo-Perez, F.; Guy, C.D.; Seed, P.C.; Rawls, J.F.; David, L.A.; et al. The severity of nonalcoholic fatty liver disease is associated with gut dysbiosis and shift in the metabolic function of the gut microbiota. Hepatology 2016, 63, 764–775. [Google Scholar] [CrossRef]
- Hirayama, M.; Ohno, K. Parkinson’s Disease and Gut Microbiota. Ann. Nutr. Metab. 2021, 77 (Suppl. 2), 28–35. [Google Scholar] [CrossRef]
- Chen, S.J.; Chen, C.C.; Liao, H.Y.; Lin, Y.T.; Wu, Y.W.; Liou, J.M.; Wu, M.S.; Kuo, C.H.; Lin, C.H. Association of Fecal and Plasma Levels of Short-Chain Fatty Acids with Gut Microbiota and Clinical Severity in Patients with Parkinson Disease. Neurology 2022, 98, e848–e858. [Google Scholar] [CrossRef]
- Yang, X.; Ai, P.; He, X.; Mo, C.; Zhang, Y.; Xu, S.; Lai, Y.; Qian, Y.; Xiao, Q. Parkinson’s Disease Is Associated with Impaired Gut-Blood Barrier for Short-Chain Fatty Acids. Mov. Disord. 2022, 37, 1634–1643. [Google Scholar] [CrossRef]
- Vogt, N.M.; Kerby, R.L.; Dill-McFarland, K.A.; Harding, S.J.; Merluzzi, A.P.; Johnson, S.C.; Carlsson, C.M.; Asthana, S.; Zetterberg, H.; Blennow, K.; et al. Gut microbiome alterations in Alzheimer’s disease. Sci. Rep. 2017, 7, 13537. [Google Scholar] [CrossRef]
- Verhaar, B.J.H.; Hendriksen, H.M.A.; de Leeuw, F.A.; Doorduijn, A.S.; van Leeuwenstijn, M.; Teunissen, C.E.; Barkhof, F.; Scheltens, P.; Kraaij, R.; van Duijn, C.M.; et al. Gut Microbiota Composition Is Related to AD Pathology. Front. Immunol. 2021, 12, 794519. [Google Scholar] [CrossRef]
- Fernando, W.M.A.D.; Martins, I.J.; Morici, M.; Bharadwaj, P.; Rainey-Smith, S.R.; Lim, W.L.F.; Martins, R.N. Sodium Butyrate Reduces Brain Amyloid-β Levels and Improves Cognitive Memory Performance in an Alzheimer’s Disease Transgenic Mouse Model at an Early Disease Stage. J. Alzheimer’s Dis. 2020, 74, 91–99. [Google Scholar] [CrossRef]
- Barcenilla, A.; Pryde, S.; Martin, J.; Duncan, S.; Stewart, C.; Henderson, C.; Flint, H. Phylogenetic relationships of butyrate-producing bacteria from the human gut. Appl. Environ. Microbiol. 2000, 66, 1654–1661. [Google Scholar] [CrossRef]
- Ohata, A.; Usami, M.; Miyoshi, M. Short-chain fatty acids alter tight junction permeability in intestinal monolayer cells via lipoxygenase activation. Nutrition 2005, 21, 838–847. [Google Scholar] [CrossRef]
- Rivera-Chávez, F.; Zhang, L.F.; Faber, F.; Lopez, C.A.; Byndloss, M.X.; Olsan, E.E.; Xu, G.; Velazquez, E.M.; Lebrilla, C.B.; Winter, S.E.; et al. Depletion of Butyrate-Producing Clostridia from the Gut Microbiota Drives an Aerobic Luminal Expansion of Salmonella. Cell Host Microbe 2016, 19, 443–454. [Google Scholar] [CrossRef]
- Colgan, S.P.; Taylor, C.T. Hypoxia: An alarm signal during intestinal inflammation. Nat. Rev. Gastroenterol. Hepatol. 2010, 7, 281–287. [Google Scholar] [CrossRef]
- Bohnhoff, M.; Miller, C.P.; Martin, W.R. Resistance of the mouse’s intestinal tract to experimental salmonella infection. II. factors responsible for its loss following streptomycin treatment. J. Exp. Med. 1964, 120, 817–828. [Google Scholar] [CrossRef]
- Rolfe, R.D. Role of volatile fatty acids in colonization resistance to Clostridium difficile. Infect. Immun. 1984, 45, 185–191. [Google Scholar] [CrossRef]
- Cummings, J.H.; Pomare, E.W.; Branch, W.J.; Naylor, C.P.; Macfarlane, G.T. Short chain fatty acids in human large intestine, portal, hepatic and venous blood. Gut 1987, 28, 1221–1227. [Google Scholar] [CrossRef]
- Lin, H.V.; Frassetto, A.; Kowalik, E.J.; Nawrocki, A.R.; Lu, M.M.; Kosinski, J.R.; Hubert, J.A.; Szeto, D.; Yao, X.; Forrest, G.; et al. Butyrate and propionate protect against diet-induced obesity and regulate gut hormones via free fatty acid receptor 3-independent mechanisms. PLoS ONE 2012, 7, e35240. [Google Scholar] [CrossRef]
- Bergman, E.N. Energy contributions of volatile fatty acids from the gastrointestinal tract in various species. Physiol. Rev. 1990, 70, 567–590. [Google Scholar] [CrossRef]
- Macfarlane, G.T.; Macfarlane, S. Fermentation in the human large intestine: Its physiologic consequences and the potential contribution of prebiotics. J. Clin. Gastroenterol. 2011, 45, S120–S127. [Google Scholar] [CrossRef]
- Gaudier, E.; Rival, M.; Buisine, M.P.; Robineau, I.; Hoebler, C. Butyrate enemas upregulate Muc genes expression but decrease adherent mucus thickness in mice colon. Physiol. Res. 2009, 58, 111–119. [Google Scholar] [CrossRef]
- Morrison, D.J.; Preston, T. Formation of short chain fatty acids by the gut microbiota and their impact on human metabolism. Gut Microbes 2016, 7, 189–200. [Google Scholar] [CrossRef]
- Hamer, H.M.; Jonkers, D.; Venema, K.; Vanhoutvin, S.; Troost, F.J.; Brummer, R.J. Review article: The role of butyrate on colonic function. Aliment. Pharmacol. Ther. 2008, 27, 104–119. [Google Scholar] [CrossRef] [PubMed]
- Meesters, R.; van Eijk, H.; ten Have, G.; de Graaf, A.; Venema, K.; van Rossum, B.; Deutz, N. Application of liquid chromatography-mass spectrometry to measure the concentrations and study the synthesis of short chain fatty acids following stable isotope infusions. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2007, 854, 57–62. [Google Scholar] [CrossRef]
- Moreau, N.; Goupry, S.; Antignac, J.; Monteau, F.; Le Bizec, B.; Champ, M.; Martin, L.; Dumon, H. Simultaneous measurement of plasma concentrations and C-13-enrichment of short-chain fatty acids, lactic acid and ketone bodies by gas chromatography coupled to mass spectrometry. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2003, 784, 395–403. [Google Scholar] [CrossRef] [PubMed]
- Macia, L.; Tan, J.; Vieira, A.T.; Leach, K.; Stanley, D.; Luong, S.; Maruya, M.; Ian McKenzie, C.; Hijikata, A.; Wong, C.; et al. Metabolite-sensing receptors GPR43 and GPR109A facilitate dietary fibre-induced gut homeostasis through regulation of the inflammasome. Nat. Commun. 2015, 6, 6734. [Google Scholar] [CrossRef]
- Yajima, T. Contractile effect of short-chain fatty acids on the isolated colon of the rat. J. Physiol. 1985, 368, 667–678. [Google Scholar] [CrossRef]
- Plaisancié, P.; Dumoulin, V.; Chayvialle, J.A.; Cuber, J.C. Luminal peptide YY-releasing factors in the isolated vascularly perfused rat colon. J. Endocrinol. 1996, 151, 421–429. [Google Scholar] [CrossRef] [PubMed]
- Fåk, F.; Jakobsdottir, G.; Kulcinskaja, E.; Marungruang, N.; Matziouridou, C.; Nilsson, U.; Stålbrand, H.; Nyman, M. The physico-chemical properties of dietary fibre determine metabolic responses, short-chain Fatty Acid profiles and gut microbiota composition in rats fed low- and high-fat diets. PLoS ONE 2015, 10, e0127252. [Google Scholar] [CrossRef]
- Kilner, J.; Corfe, B.; McAuley, M.; Wilkinson, S. A deterministic oscillatory model of microtubule growth and shrinkage for differential actions of short chain fatty acids. Mol. Biosyst. 2016, 12, 93–101. [Google Scholar] [CrossRef]
- Vincent, A.D.; Wang, X.Y.; Parsons, S.P.; Khan, W.I.; Huizinga, J.D. Abnormal absorptive colonic motor activity in germ-free mice is rectified by butyrate, an effect possibly mediated by mucosal serotonin. Am. J. Physiol. Gastrointest. Liver Physiol. 2018, 315, G896–G907. [Google Scholar] [CrossRef] [PubMed]
- Gao, Z.; Yin, J.; Zhang, J.; Ward, R.E.; Martin, R.J.; Lefevre, M.; Cefalu, W.T.; Ye, J. Butyrate improves insulin sensitivity and increases energy expenditure in mice. Diabetes 2009, 58, 1509–1517. [Google Scholar] [CrossRef] [PubMed]
- Fushimi, T.; Suruga, K.; Oshima, Y.; Fukiharu, M.; Tsukamoto, Y.; Goda, T. Dietary acetic acid reduces serum cholesterol and triacylglycerols in rats fed a cholesterol-rich diet. Br. J. Nutr. 2006, 95, 916–924. [Google Scholar] [CrossRef] [PubMed]
- De Vadder, F.; Kovatcheva-Datchary, P.; Goncalves, D.; Vinera, J.; Zitoun, C.; Duchampt, A.; Backhed, F.; Mithieux, G. Microbiota-Generated Metabolites Promote Metabolic Benefits via Gut-Brain Neural Circuits. Cell 2014, 156, 84–96. [Google Scholar] [CrossRef]
- Ukai, M.; Tomura, A.; Ito, M. Cholesterol-synthesis in germfree and conventional rats. J. Nutr. 1976, 106, 1175–1183. [Google Scholar] [CrossRef]
- Wang, G.; Yu, Y.; Wang, Y.; Wang, J.; Guan, R.; Sun, Y.; Shi, F.; Gao, J.; Fu, X. Role of SCFAs in gut microbiome and glycolysis for colorectal cancer therapy. J. Cell. Physiol. 2019, 234, 17023–17049. [Google Scholar] [CrossRef]
- Reigstad, C.S.; Salmonson, C.E.; Rainey, J.F.; Szurszewski, J.H.; Linden, D.R.; Sonnenburg, J.L.; Farrugia, G.; Kashyap, P.C. Gut microbes promote colonic serotonin production through an effect of short-chain fatty acids on enterochromaffin cells. FASEB J. 2015, 29, 1395–1403. [Google Scholar] [CrossRef]
- Vijay, N.; Morris, M.E. Role of monocarboxylate transporters in drug delivery to the brain. Curr. Pharm. Des. 2014, 20, 1487–1498. [Google Scholar] [CrossRef]
- Daly, K.; Shirazi-Beechey, S. Microarray analysis of butyrate regulated genes in colonic epithelial cells. DNA Cell Biol. 2006, 25, 49–62. [Google Scholar] [CrossRef]
- Campos-Perez, W.; Martinez-Lopez, E. Effects of short chain fatty acids on metabolic and inflammatory processes in human health. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2021, 1866, 158900. [Google Scholar] [CrossRef] [PubMed]
- González-Bosch, C.; Boorman, E.; Zunszain, P.A.; Mann, G.E. Short-chain fatty acids as modulators of redox signaling in health and disease. Redox Biol. 2021, 47, 102165. [Google Scholar] [CrossRef] [PubMed]
- Visekruna, A.; Luu, M. The Role of Short-Chain Fatty Acids and Bile Acids in Intestinal and Liver Function, Inflammation, and Carcinogenesis. Front. Cell Dev. Biol. 2021, 9, 703218. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.J.; Goldsworthy, S.M.; Barnes, A.A.; Eilert, M.M.; Tcheang, L.; Daniels, D.; Muir, A.I.; Wigglesworth, M.J.; Kinghorn, I.; Fraser, N.J.; et al. The Orphan G protein-coupled receptors GPR41 and GPR43 are activated by propionate and other short chain carboxylic acids. J. Biol. Chem. 2003, 278, 11312–11319. [Google Scholar] [CrossRef]
- Karaki, S.; Mitsui, R.; Hayashi, H.; Kato, I.; Sugiya, H.; Iwanaga, T.; Furness, J.B.; Kuwahara, A. Short-chain fatty acid receptor, GPR43, is expressed by enteroendocrine cells and mucosal mast cells in rat intestine. Cell Tissue Res. 2006, 324, 353–360. [Google Scholar] [CrossRef]
- Ahmed, K.; Tunaru, S.; Offermanns, S. GPR109A, GPR109B and GPR81, a family of hydroxy-carboxylic acid receptors. Trends Pharmacol. Sci. 2009, 30, 557–562. [Google Scholar] [CrossRef]
- Dorsam, R.T.; Gutkind, J.S. G-protein-coupled receptors and cancer. Nat. Rev. Cancer 2007, 7, 79–94. [Google Scholar] [CrossRef]
- Schotterl, S.; Brennenstuhl, H.; Naumann, U. Modulation of immune responses by histone deacetylase inhibitors. Crit. Rev. Oncog. 2015, 20, 139–154. [Google Scholar] [CrossRef]
- Weiss, U.; Möller, M.; Husseini, S.A.; Manderscheid, C.; Häusler, J.; Geisslinger, G.; Niederberger, E. Inhibition of HDAC Enzymes Contributes to Differential Expression of Pro-Inflammatory Proteins in the TLR-4 Signaling Cascade. Int. J. Mol. Sci. 2020, 21, 8943. [Google Scholar] [CrossRef]
- Smith, P.M.; Howitt, M.R.; Panikov, N.; Michaud, M.; Gallini, C.A.; Bohlooly-Y, M.; Glickman, J.N.; Garrett, W.S. The microbial metabolites, short-chain fatty acids, regulate colonic Treg cell homeostasis. Science 2013, 341, 569–573. [Google Scholar] [CrossRef]
- Sun, M.; Wu, W.; Chen, L.; Yang, W.; Huang, X.; Ma, C.; Chen, F.; Xiao, Y.; Zhao, Y.; Yao, S.; et al. Microbiota-derived short-chain fatty acids promote Th1 cell IL-10 production to maintain intestinal homeostasis. Nat. Commun. 2018, 9, 3555. [Google Scholar] [CrossRef]
- Salazar, N.; Arboleya, S.; Fernández-Navarro, T.; de Los Reyes-Gavilán, C.G.; Gonzalez, S.; Gueimonde, M. Age-Associated Changes in Gut Microbiota and Dietary Components Related with the Immune System in Adulthood and Old Age: A Cross-Sectional Study. Nutrients 2019, 11, 1765. [Google Scholar] [CrossRef]
- Matsumaru, D.; Motohashi, H. The KEAP1-NRF2 System in Healthy Aging and Longevity. Antioxidants 2021, 10, 1929. [Google Scholar] [CrossRef]
- Li, H.; Shang, Z.; Liu, X.; Qiao, Y.; Wang, K.; Qiao, J. Alleviates Enterotoxigenic. Front. Immunol. 2021, 12, 771826. [Google Scholar] [CrossRef]
- Dong, W.; Jia, Y.; Liu, X.; Zhang, H.; Li, T.; Huang, W.; Chen, X.; Wang, F.; Sun, W.; Wu, H. Sodium butyrate activates NRF2 to ameliorate diabetic nephropathy possibly via inhibition of HDAC. J. Endocrinol. 2017, 232, 71–83. [Google Scholar] [CrossRef]
- Wu, J.; Jiang, Z.; Zhang, H.; Liang, W.; Huang, W.; Li, Y.; Wang, Z.; Wang, J.; Jia, Y.; Liu, B.; et al. Sodium butyrate attenuates diabetes-induced aortic endothelial dysfunction via P300-mediated transcriptional activation of Nrf2. Free Radic. Biol. Med. 2018, 124, 454–465. [Google Scholar] [CrossRef]
- Parada Venegas, D.; De la Fuente, M.K.; Landskron, G.; González, M.J.; Quera, R.; Dijkstra, G.; Harmsen, H.J.M.; Faber, K.N.; Hermoso, M.A. Short Chain Fatty Acids (SCFAs)-Mediated Gut Epithelial and Immune Regulation and Its Relevance for Inflammatory Bowel Diseases. Front. Immunol. 2019, 10, 277. [Google Scholar] [CrossRef]
- Sun, S.; Araki, Y.; Hanzawa, F.; Umeki, M.; Kojima, T.; Nishimura, N.; Ikeda, S.; Mochizuki, S.; Oda, H. High sucrose diet-induced dysbiosis of gut microbiota promotes fatty liver and hyperlipidemia in rats. J. Nutr. Biochem. 2021, 93, 108621. [Google Scholar] [CrossRef]
- El Kaoutari, A.; Armougom, F.; Gordon, J.I.; Raoult, D.; Henrissat, B. The abundance and variety of carbohydrate-active enzymes in the human gut microbiota. Nat. Rev. Microbiol. 2013, 11, 497–504. [Google Scholar] [CrossRef] [PubMed]
- Topping, D.L.; Clifton, P.M. Short-chain fatty acids and human colonic function: Roles of resistant starch and nonstarch polysaccharides. Physiol. Rev. 2001, 81, 1031–1064. [Google Scholar] [CrossRef] [PubMed]
- Martinez, K.B.; Leone, V.; Chang, E.B. Western diets, gut dysbiosis, and metabolic diseases: Are they linked? Gut Microbes 2017, 8, 130–142. [Google Scholar] [CrossRef] [PubMed]
- De Filippo, C.; Cavalieri, D.; Di Paola, M.; Ramazzotti, M.; Poullet, J.; Massart, S.; Collini, S.; Pieraccini, G.; Lionetti, P. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc. Natl. Acad. Sci. USA 2010, 107, 14691–14696. [Google Scholar] [CrossRef] [PubMed]
- Schnorr, S.; Candela, M.; Rampelli, S.; Centanni, M.; Consolandi, C.; Basaglia, G.; Turroni, S.; Biagi, E.; Peano, C.; Severgnini, M.; et al. Gut microbiome of the Hadza hunter-gatherers. Nat. Commun. 2014, 5, 3654. [Google Scholar] [CrossRef] [PubMed]
- Ou, J.; Carbonero, F.; Zoetendal, E.G.; DeLany, J.P.; Wang, M.; Newton, K.; Gaskins, H.R.; O’Keefe, S.J. Diet, microbiota, and microbial metabolites in colon cancer risk in rural Africans and African Americans. Am. J. Clin. Nutr. 2013, 98, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Yu, Y.; Li, H.; Ding, X.; Li, X.; Jing, X.; Chen, J.; Liu, G.; Lin, Y.; Jiang, C.; et al. Inulin supplementation ameliorates hyperuricemia and modulates gut microbiota in Uox-knockout mice. Eur. J. Nutr. 2021, 60, 2217–2230. [Google Scholar] [CrossRef]
- Yang, Z.; Su, H.; Lv, Y.; Tao, H.; Jiang, Y.; Ni, Z.; Peng, L.; Chen, X. Inulin intervention attenuates hepatic steatosis in rats via modulating gut microbiota and maintaining intestinal barrier function. Food Res. Int. 2023, 163, 112309. [Google Scholar] [CrossRef]
- Williamson, G.; Clifford, M. Role of the small intestine, colon and microbiota in determining the metabolic fate of polyphenols. Biochemical. Pharmacol. 2017, 139, 24–39. [Google Scholar] [CrossRef]
- Manach, C.; Williamson, G.; Morand, C.; Scalbert, A.; Remesy, C. Bioavailability and bioefficacy of polyphenols in humans. I. Review of 97 bioavailability studies. Am. J. Clin. Nutr. 2005, 81, 230S–242S. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Mateos, A.; Vauzour, D.; Krueger, C.; Shanmuganayagam, D.; Reed, J.; Calani, L.; Mena, P.; Del Rio, D.; Crozier, A. Bioavailability, bioactivity and impact on health of dietary flavonoids and related compounds: An update. Arch. Toxicol. 2014, 88, 1803–1853. [Google Scholar] [CrossRef]
- Cassidy, A.; Minihane, A. The role of metabolism (and the microbiome) in defining the clinical efficacy of dietary flavonoids. Am. J. Clin. Nutr. 2017, 105, 10–22. [Google Scholar] [CrossRef]
- Aura, A. Microbial metabolism of dietary phenolic compounds in the colon. Phytochem. Rev. 2008, 7, 407–429. [Google Scholar] [CrossRef]
- Heleno, S.; Martins, A.; Queiroz, M.; Ferreira, I. Bioactivity of phenolic acids: Metabolites versus parent compounds: A review. Food Chem. 2015, 173, 501–513. [Google Scholar] [CrossRef] [PubMed]
- Koudoufio, M.; Desjardins, Y.; Feldman, F.; Spahis, S.; Delvin, E.; Levy, E. Insight into Polyphenol and Gut Microbiota Crosstalk: Are Their Metabolites the Key to Understand Protective Effects against Metabolic Disorders? Antioxidants 2020, 9, 982. [Google Scholar] [CrossRef] [PubMed]
- Ozdal, T.; Sela, D.A.; Xiao, J.; Boyacioglu, D.; Chen, F.; Capanoglu, E. The Reciprocal Interactions between Polyphenols and Gut Microbiota and Effects on Bioaccessibility. Nutrients 2016, 8, 78. [Google Scholar] [CrossRef]
- Catalkaya, G.; Venema, K.; Lucini, L.; Rocchetti, G.; Delmas, D.; Daglia, M.; De Filippis, A.; Xiao, H.H.; Quiles, J.L.; Xiao, J.; et al. Interaction of dietary polyphenols and gut microbiota:Microbial metabolism of polyphenols, influence on the gut microbiota, and implications on host health. Food Front. 2020, 1, 109–133. [Google Scholar] [CrossRef]
- Carbonell-Capella, J.M.; Buniowska, M.; Barba, F.J.; Esteve, M.J.; Frígola, A. Analytical Methods for Determining Bioavailability and Bioaccessibility of Bioactive Compounds from Fruits and Vegetables: A Review. Compr. Rev. Food Sci. Food Saf. 2014, 13, 155–171. [Google Scholar] [CrossRef]
- Wightman, E.L.; Haskell-Ramsay, C.F.; Reay, J.L.; Williamson, G.; Dew, T.; Zhang, W.; Kennedy, D.O. The effects of chronic trans-resveratrol supplementation on aspects of cognitive function, mood, sleep, health and cerebral blood flow in healthy, young humans. Br. J. Nutr. 2015, 114, 1427–1437. [Google Scholar] [CrossRef] [PubMed]
- Mahale, J.; Singh, R.; Howells, L.; Britton, R.; Khan, S.; Brown, K. Detection of Plasma Curcuminoids from Dietary Intake of Turmeric-Containing Food in Human Volunteers. Mol. Nutr. Food Res. 2018, 62, e1800267. [Google Scholar] [CrossRef]
- Marin, L.; Miguelez, E.; Villar, C.; Lombo, F. Bioavailability of Dietary Polyphenols and Gut Microbiota Metabolism: Antimicrobial Properties. BioMed Res. Int. 2015, 2015, 905215. [Google Scholar] [CrossRef]
- Burapan, S.; Kim, M.; Han, J. Curcuminoid Demethylation as an Alternative Metabolism by Human Intestinal Microbiota. J. Agric. Food Chem. 2017, 65, 3305–3310. [Google Scholar] [CrossRef]
- Chiou, Y.; Wu, J.; Huang, Q.; Shahidi, F.; Wang, Y.; Ho, C.; Pan, M. Metabolic and colonic microbiota transformation may enhance the bioactivities of dietary polyphenols. J. Funct. Foods 2014, 7, 3–25. [Google Scholar] [CrossRef]
- Griffiths, L.A.; Barrow, A. Metabolism of flavonoid compounds in germ-free rats. Biochem. J. 1972, 130, 1161–1162. [Google Scholar] [CrossRef] [PubMed]
- Hehemann, J.H.; Correc, G.; Barbeyron, T.; Helbert, W.; Czjzek, M.; Michel, G. Transfer of carbohydrate-active enzymes from marine bacteria to Japanese gut microbiota. Nature 2010, 464, 908–912. [Google Scholar] [CrossRef] [PubMed]
- van Duynhoven, J.; Vaughan, E.E.; Jacobs, D.M.; Kemperman, R.A.; van Velzen, E.J.; Gross, G.; Roger, L.C.; Possemiers, S.; Smilde, A.K.; Doré, J.; et al. Metabolic fate of polyphenols in the human superorganism. Proc. Natl. Acad. Sci. USA 2011, 108 (Suppl. 1), 4531–4538. [Google Scholar] [CrossRef]
- Wu, L.; Lyu, Y.; Srinivasagan, R.; Wu, J.; Ojo, B.; Tang, M.; El-Rassi, G.D.; Metzinger, K.; Smith, B.J.; Lucas, E.A.; et al. Astaxanthin-Shifted Gut Microbiota Is Associated with Inflammation and Metabolic Homeostasis in Mice. J. Nutr. 2020, 150, 2687–2698. [Google Scholar] [CrossRef] [PubMed]
- Terasaki, M.; Uehara, O.; Ogasa, S.; Sano, T.; Kubota, A.; Kojima, H.; Tanaka, T.; Maeda, H.; Miyashita, K.; Mutoh, M. Alteration of fecal microbiota by fucoxanthin results in prevention of colorectal cancer in AOM/DSS mice. Carcinogenesis 2021, 42, 210–219. [Google Scholar] [CrossRef]
- Xia, H.; Liu, C.; Li, C.C.; Fu, M.; Takahashi, S.; Hu, K.Q.; Aizawa, K.; Hiroyuki, S.; Wu, G.; Zhao, L.; et al. Dietary Tomato Powder Inhibits High-Fat Diet-Promoted Hepatocellular Carcinoma with Alteration of Gut Microbiota in Mice Lacking Carotenoid Cleavage Enzymes. Cancer Prev. Res. 2018, 11, 797–810. [Google Scholar] [CrossRef]
- Wang, X.; Liu, F.; Cui, Y.; Yin, Y.; Li, S.; Li, X. Apple Polyphenols Extracts Ameliorate High Carbohydrate Diet-Induced Body Weight Gain by Regulating the Gut Microbiota and Appetite. J. Agric. Food Chem. 2022, 70, 196–210. [Google Scholar] [CrossRef]
- Cladis, D.P.; Simpson, A.M.R.; Cooper, K.J.; Nakatsu, C.H.; Ferruzzi, M.G.; Weaver, C.M. Blueberry polyphenols alter gut microbiota & phenolic metabolism in rats. Food Funct. 2021, 12, 2442–2456. [Google Scholar] [CrossRef]
- Guo, X.; Xu, Y.; Geng, R.; Qiu, J.; He, X. Curcumin Alleviates Dextran Sulfate Sodium-Induced Colitis in Mice Through Regulating Gut Microbiota. Mol. Nutr. Food Res. 2022, 66, e2100943. [Google Scholar] [CrossRef]
- Zhang, F.; Zhou, Y.; Chen, H.; Jiang, H.; Zhou, F.; Lv, B.; Xu, M. Curcumin Alleviates DSS-Induced Anxiety-Like Behaviors via the Microbial-Brain-Gut Axis. Oxid. Med. Cell. Longev. 2022, 2022, 6244757. [Google Scholar] [CrossRef] [PubMed]
- Park, J.M.; Shin, Y.; Kim, S.H.; Jin, M.; Choi, J.J. Dietary Epigallocatechin-3-Gallate Alters the Gut Microbiota of Obese Diabetic. J. Med. Food 2020, 23, 1033–1042. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.; Li, Y.L.; Zhang, X.; Wang, K.B.; Huang, J.A.; Liu, Z.H.; Zhu, M.Z. Polyphenols from Fu Brick Tea Reduce Obesity via Modulation of Gut Microbiota and Gut Microbiota-Related Intestinal Oxidative Stress and Barrier Function. J. Agric. Food Chem. 2021, 69, 14530–14543. [Google Scholar] [CrossRef] [PubMed]
- Xiang, J.Y.; Chi, Y.Y.; Han, J.X.; Kong, P.; Liang, Z.; Wang, D.; Xiang, H.; Xie, Q. Litchi chinensis seed prevents obesity and modulates the gut microbiota and mycobiota compositions in high-fat diet-induced obese zebrafish. Food Funct. 2022, 13, 2832–2845. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.; Wang, L.; Liu, M.; Wu, X.; Lu, Q.; Liu, R. The underlying mechanism of A-type procyanidins from peanut skin on DSS-induced ulcerative colitis mice by regulating gut microbiota and metabolism. J. Food Biochem. 2022, 46, e14103. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; Shen, X.; Chu, Q.; Zheng, X. Pomegranate fruit pulp polyphenols reduce diet-induced obesity with modulation of gut microbiota in mice. J. Sci. Food Agric. 2022, 102, 1968–1977. [Google Scholar] [CrossRef]
- Liu, D.; Ji, Y.; Wang, K.; Guo, Y.; Wang, H.; Zhang, H.; Li, L.; Li, H.; Cui, S.W. Purple sweet potato anthocyanin extract regulates redox state related to gut microbiota homeostasis in obese mice. J. Food Sci. 2022, 87, 2133–2146. [Google Scholar] [CrossRef]
- Chen, C.; Wang, H.; Hong, T.; Huang, X.; Xia, S.; Zhang, Y.; Chen, X.; Zhong, Y.; Nie, S. Effects of tea polysaccharides in combination with polyphenols on dextran sodium sulfate-induced colitis in mice. Food Chem. X 2022, 13, 100190. [Google Scholar] [CrossRef]
- Luo, L.; Zhang, J.; Liu, M.; Qiu, S.; Yi, S.; Yu, W.; Liu, T.; Huang, X.; Ning, F. Monofloral Triadica Cochinchinensis Honey Polyphenols Improve Alcohol-Induced Liver Disease by Regulating the Gut Microbiota of Mice. Front. Immunol. 2021, 12, 673903. [Google Scholar] [CrossRef]
- Logan, I.E.; Shulzhenko, N.; Sharpton, T.J.; Bobe, G.; Liu, K.; Nuss, S.; Jones, M.L.; Miranda, C.L.; Vasquez-Perez, S.; Pennington, J.M.; et al. Xanthohumol Requires the Intestinal Microbiota to Improve Glucose Metabolism in Diet-Induced Obese Mice. Mol. Nutr. Food Res. 2021, 65, e2100389. [Google Scholar] [CrossRef]
- Zlatanos, S.N.; Laskaridis, K.; Sagredos, A. Conjugated linoleic acid content of human plasma. Lipids Health Dis. 2008, 7, 34. [Google Scholar] [CrossRef] [PubMed]
- Garg, M.L.; Leitch, J.; Blake, R.J.; Garg, R. Long-chain n-3 polyunsaturated fatty acid incorporation into human atrium following fish oil supplementation. Lipids 2006, 41, 1127–1132. [Google Scholar] [CrossRef] [PubMed]
- Murru, E.; Carta, G.; Manca, C.; Saebo, A.; Santoni, M.; Mostallino, R.; Pistis, M.; Banni, S. Dietary Phospholipid-Bound Conjugated Linoleic Acid and Docosahexaenoic Acid Incorporation Into Fetal Liver and Brain Modulates Fatty Acid and. Front. Nutr. 2022, 9, 834066. [Google Scholar] [CrossRef] [PubMed]
- Schoeler, M.; Caesar, R. Dietary lipids, gut microbiota and lipid metabolism. Rev. Endocr. Metab. Disord. 2019, 20, 461–472. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Wang, Y.; Gao, H.; Li, D.; Jiang, R.; Ge, L.; Tong, C.; Xu, K. Associations among Dietary Omega-3 Polyunsaturated Fatty Acids, the Gut Microbiota, and Intestinal Immunity. Mediat. Inflamm. 2021, 2021, 8879227. [Google Scholar] [CrossRef]
- Robertson, R.C.; Seira Oriach, C.; Murphy, K.; Moloney, G.M.; Cryan, J.F.; Dinan, T.G.; Ross, R.P.; Stanton, C. Deficiency of essential dietary n-3 PUFA disrupts the caecal microbiome and metabolome in mice. Br. J. Nutr. 2017, 118, 959–970. [Google Scholar] [CrossRef]
- Menni, C.; Zierer, J.; Pallister, T.; Jackson, M.A.; Long, T.; Mohney, R.P.; Steves, C.J.; Spector, T.D.; Valdes, A.M. Omega-3 fatty acids correlate with gut microbiome diversity and production of N-carbamylglutamate in middle aged and elderly women. Sci. Rep. 2017, 7, 11079. [Google Scholar] [CrossRef]
- Noriega, B.S.; Sanchez-Gonzalez, M.A.; Salyakina, D.; Coffman, J. Understanding the Impact of Omega-3 Rich Diet on the Gut Microbiota. Case Rep. Med. 2016, 2016, 3089303. [Google Scholar] [CrossRef]
- Kaliannan, K.; Wang, B.; Li, X.Y.; Kim, K.J.; Kang, J.X. A host-microbiome interaction mediates the opposing effects of omega-6 and omega-3 fatty acids on metabolic endotoxemia. Sci. Rep. 2015, 5, 11276. [Google Scholar] [CrossRef]
- Younge, N.; Yang, Q.; Seed, P.C. Enteral High Fat-Polyunsaturated Fatty Acid Blend Alters the Pathogen Composition of the Intestinal Microbiome in Premature Infants with an Enterostomy. J. Pediatr. 2017, 181, 93–101.e6. [Google Scholar] [CrossRef]
- Mokkala, K.; Röytiö, H.; Munukka, E.; Pietilä, S.; Ekblad, U.; Rönnemaa, T.; Eerola, E.; Laiho, A.; Laitinen, K. Gut Microbiota Richness and Composition and Dietary Intake of Overweight Pregnant Women Are Related to Serum Zonulin Concentration, a Marker for Intestinal Permeability. J. Nutr. 2016, 146, 1694–1700. [Google Scholar] [CrossRef] [PubMed]
- Watson, H.; Mitra, S.; Croden, F.C.; Taylor, M.; Wood, H.M.; Perry, S.L.; Spencer, J.A.; Quirke, P.; Toogood, G.J.; Lawton, C.L.; et al. A randomised trial of the effect of omega-3 polyunsaturated fatty acid supplements on the human intestinal microbiota. Gut 2018, 67, 1974–1983. [Google Scholar] [CrossRef] [PubMed]
- Balfegó, M.; Canivell, S.; Hanzu, F.A.; Sala-Vila, A.; Martínez-Medina, M.; Murillo, S.; Mur, T.; Ruano, E.G.; Linares, F.; Porras, N.; et al. Effects of sardine-enriched diet on metabolic control, inflammation and gut microbiota in drug-naïve patients with type 2 diabetes: A pilot randomized trial. Lipids Health Dis. 2016, 15, 78. [Google Scholar] [CrossRef] [PubMed]
- Mujico, J.R.; Baccan, G.C.; Gheorghe, A.; Díaz, L.E.; Marcos, A. Changes in gut microbiota due to supplemented fatty acids in diet-induced obese mice. Br. J. Nutr. 2013, 110, 711–720. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Hougen, H.; Vollmer, A.C.; Hiebert, S.M. Gut bacteria profiles of Mus musculus at the phylum and family levels are influenced by saturation of dietary fatty acids. Anaerobe 2012, 18, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Patterson, E.; O’ Doherty, R.M.; Murphy, E.F.; Wall, R.; O’ Sullivan, O.; Nilaweera, K.; Fitzgerald, G.F.; Cotter, P.D.; Ross, R.P.; Stanton, C. Impact of dietary fatty acids on metabolic activity and host intestinal microbiota composition in C57BL/6J mice. Br. J. Nutr. 2014, 111, 1905–1917. [Google Scholar] [CrossRef]
- Caesar, R.; Tremaroli, V.; Kovatcheva-Datchary, P.; Cani, P.D.; Bäckhed, F. Crosstalk between Gut Microbiota and Dietary Lipids Aggravates WAT Inflammation through TLR Signaling. Cell Metab. 2015, 22, 658–668. [Google Scholar] [CrossRef] [PubMed]
- Guinane, C.M.; Cotter, P.D. Role of the gut microbiota in health and chronic gastrointestinal disease: Understanding a hidden metabolic organ. Ther. Adv. Gastroenterol. 2013, 6, 295–308. [Google Scholar] [CrossRef]
- Pariza, M.W. Perspective on the safety and effectiveness of conjugated linoleic acid. Am. J. Clin. Nutr. 2004, 79, 1132S–1136S. [Google Scholar] [CrossRef]
- Marques, T.M.; Wall, R.; O’Sullivan, O.; Fitzgerald, G.F.; Shanahan, F.; Quigley, E.M.; Cotter, P.D.; Cryan, J.F.; Dinan, T.G.; Ross, R.P.; et al. Dietary trans-10, cis-12-conjugated linoleic acid alters fatty acid metabolism and microbiota composition in mice. Br. J. Nutr. 2015, 113, 728–738. [Google Scholar] [CrossRef]
- den Hartigh, L.J.; Gao, Z.; Goodspeed, L.; Wang, S.; Das, A.K.; Burant, C.F.; Chait, A.; Blaser, M.J. Obese Mice Losing Weight Due to trans-10,cis-12 Conjugated Linoleic Acid Supplementation or Food Restriction Harbor Distinct Gut Microbiota. J. Nutr. 2018, 148, 562–572. [Google Scholar] [CrossRef] [PubMed]
- Chidambaram, S.B.; Essa, M.M.; Rathipriya, A.G.; Bishir, M.; Ray, B.; Mahalakshmi, A.M.; Tousif, A.H.; Sakharkar, M.K.; Kashyap, R.S.; Friedland, R.P.; et al. Gut dysbiosis, defective autophagy and altered immune responses in neurodegenerative diseases: Tales of a vicious cycle. Pharmacol. Ther. 2021, 231, 107988. [Google Scholar] [CrossRef] [PubMed]
- Custers; Emma, E.M.; Kiliaan; Amanda, J. Dietary lipids from body to brain. Prog. Lipid Res. 2022, 85, 101144. [Google Scholar] [CrossRef]
- Gorissen, L.; Raes, K.; Weckx, S.; Dannenberger, D.; Leroy, F.; De Vuyst, L.; De Smet, S. Production of conjugated linoleic acid and conjugated linolenic acid isomers by Bifidobacterium species. Appl. Microbiol. Biotechnol. 2010, 87, 2257–2266. [Google Scholar] [CrossRef]
- Bergamo, P.; Luongo, D.; Maurano, F.; Mazzarella, G.; Stefanile, R.; Rossi, M. Conjugated linoleic acid enhances glutathione synthesis and attenuates pathological signs in MRL/MpJ-Fas(lpr) mice. J. Lipid Res. 2006, 47, 2382–2391. [Google Scholar] [CrossRef]
- Bergamo, P.; Cocca, E.; Monaco, A.; Cozzolino, V.; Boscaino, F.; Ferrandino, I.; Maurano, F.; Rossi, M. Protective effect of Rumenic acid rich cow’s milk against colitis is associated with the activation of Nrf2 pathway in a murine model. Prostaglandins Leukot. Essent. Fat. Acids 2017, 125, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Bergamo, P.; Luongo, D.; Miyamoto, J.; Cocca, E.; Kishino, S.; Ogawa, J.; Tanabe, S.; Rossi, M. Immunomodulatory activity of a gut microbial metabolite of dietary linoleic acid, 10-hydroxy-cis-12-octadecenoic acid, associated with improved antioxidant/detoxifying defences. J. Funct. Foods 2014, 11, 192–202. [Google Scholar] [CrossRef]
- Furumoto, H.; Nanthirudjanar, T.; Kume, T.; Izumi, Y.; Park, S.B.; Kitamura, N.; Kishino, S.; Ogawa, J.; Hirata, T.; Sugawara, T. 10-Oxo-trans-11-octadecenoic acid generated from linoleic acid by a gut lactic acid bacterium Lactobacillus plantarum is cytoprotective against oxidative stress. Toxicol. Appl. Pharmacol. 2016, 296, 1–9. [Google Scholar] [CrossRef]
- Miyamoto, J.; Mizukure, T.; Park, S.B.; Kishino, S.; Kimura, I.; Hirano, K.; Bergamo, P.; Rossi, M.; Suzuki, T.; Arita, M.; et al. A gut microbial metabolite of linoleic acid, 10-hydroxy-cis-12-octadecenoic acid, ameliorates intestinal epithelial barrier impairment partially via GPR40-MEK-ERK pathway. J. Biol. Chem. 2015, 290, 2902–2918. [Google Scholar] [CrossRef]
- Yokoji-Takeuchi, M.; Takahashi, N.; Yamada-Hara, M.; Sulijaya, B.; Tsuzuno, T.; Aoki-Nonaka, Y.; Tabeta, K.; Kishino, S.; Ogawa, J.; Yamazaki, K. A bacterial metabolite induces Nrf2-mediated anti-oxidative responses in gingival epithelial cells by activating the MAPK signaling pathway. Arch. Oral Biol. 2020, 110, 104602. [Google Scholar] [CrossRef]
- Druart, C.; Neyrinck, A.M.; Dewulf, E.M.; De Backer, F.C.; Possemiers, S.; Van de Wiele, T.; Moens, F.; De Vuyst, L.; Cani, P.D.; Larondelle, Y.; et al. Implication of fermentable carbohydrates targeting the gut microbiota on conjugated linoleic acid production in high-fat-fed mice. Br. J. Nutr. 2013, 110, 998–1011. [Google Scholar] [CrossRef] [PubMed]
- Fleck, A.K.; Hucke, S.; Teipel, F.; Eschborn, M.; Janoschka, C.; Liebmann, M.; Wami, H.; Korn, L.; Pickert, G.; Hartwig, M.; et al. Dietary conjugated linoleic acid links reduced intestinal inflammation to amelioration of CNS autoimmunity. Brain 2021, 144, 1152–1166. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).