Exploring the Nutritional Composition and Bioactive Compounds in Different Cocoa Powders
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Samples
2.3. Ultrasound Bath Extraction
2.4. Determination of Total Phenolic Content by Folin–Ciocalteu
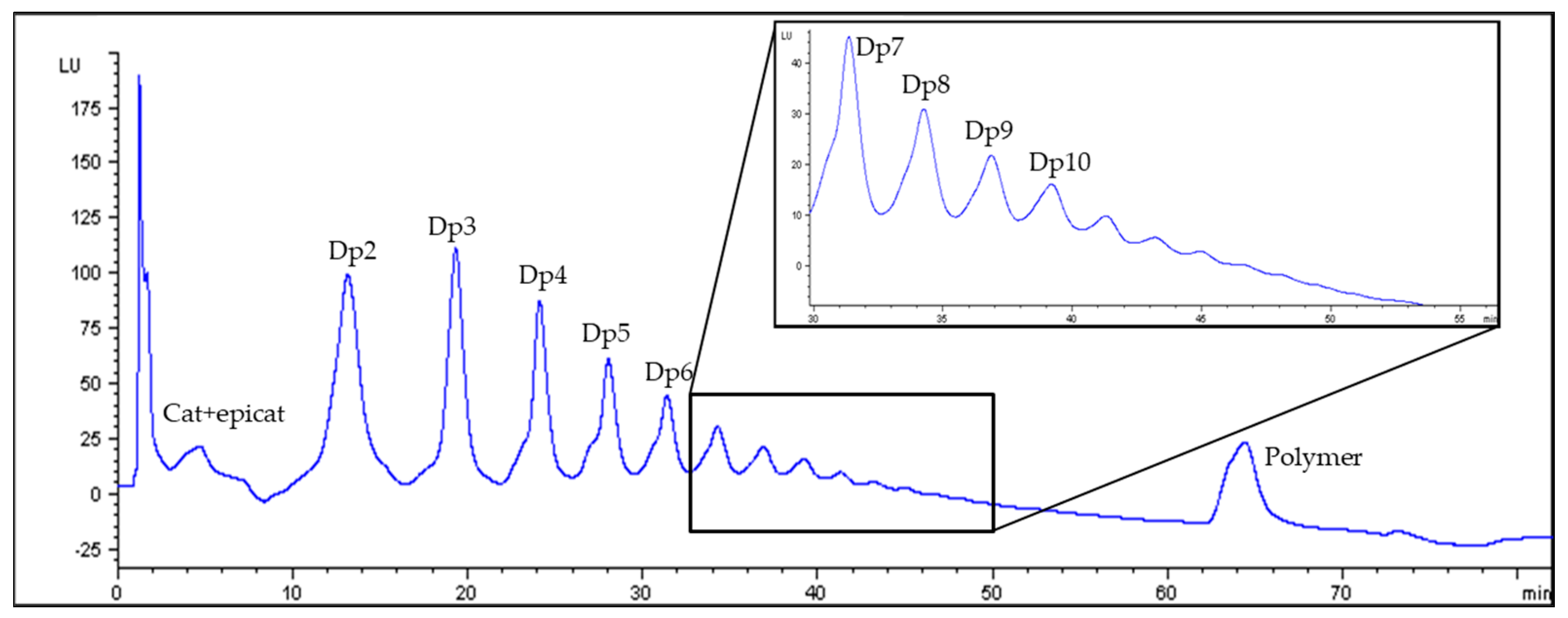
2.5. Determination of Flavan-3-Ols by HPLC-FLD
2.6. Determination of Methylxanthynes by HPLC-DAD
2.7. Determination of Antioxidant Activity: DPPH, ABTS and FRAP Assays
2.8. Statistical Analysis
3. Results and Discussion
3.1. Nutritional Evaluation of the Cocoas
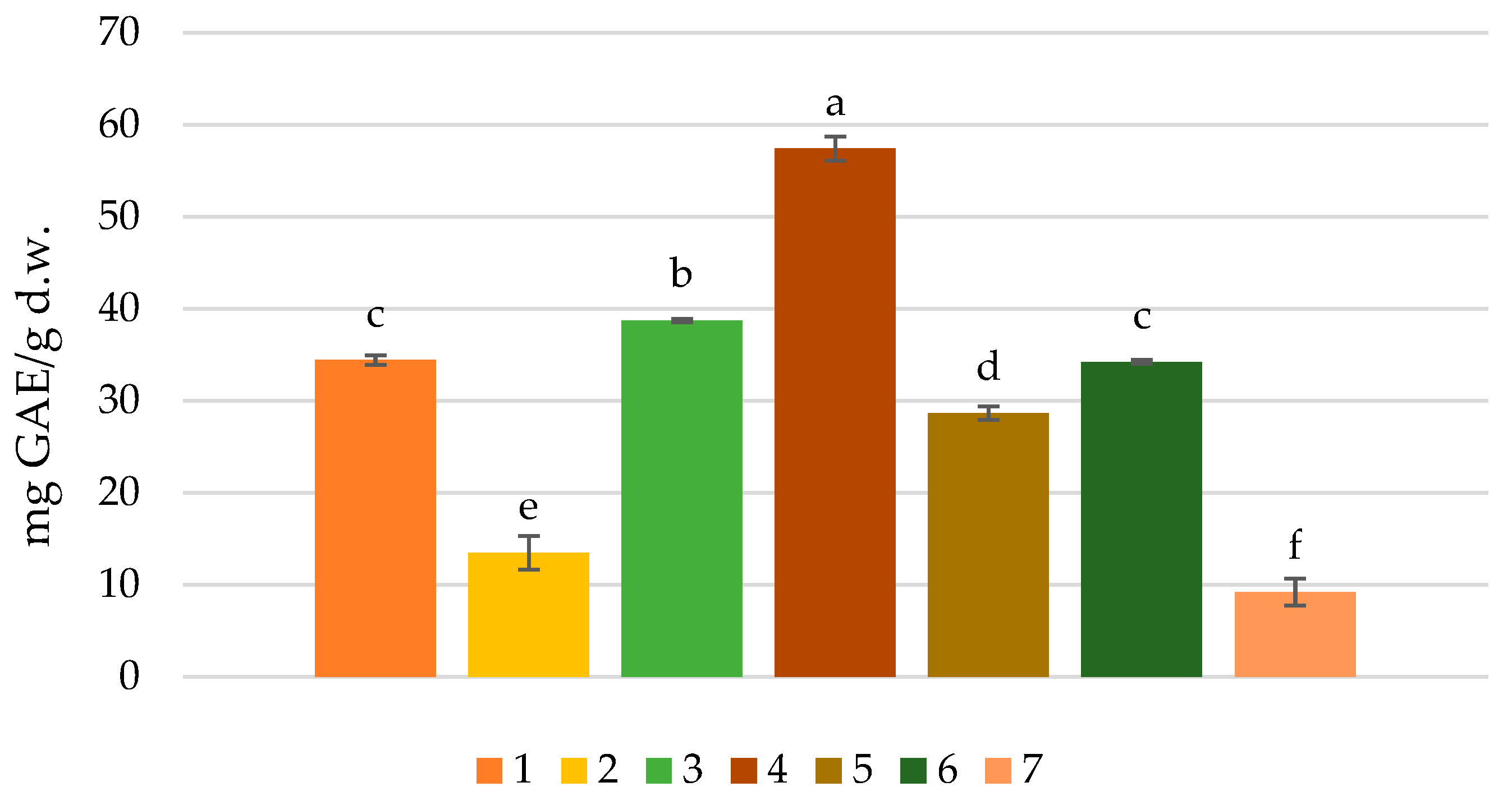
3.2. Total Phenolic Content of Cocoas
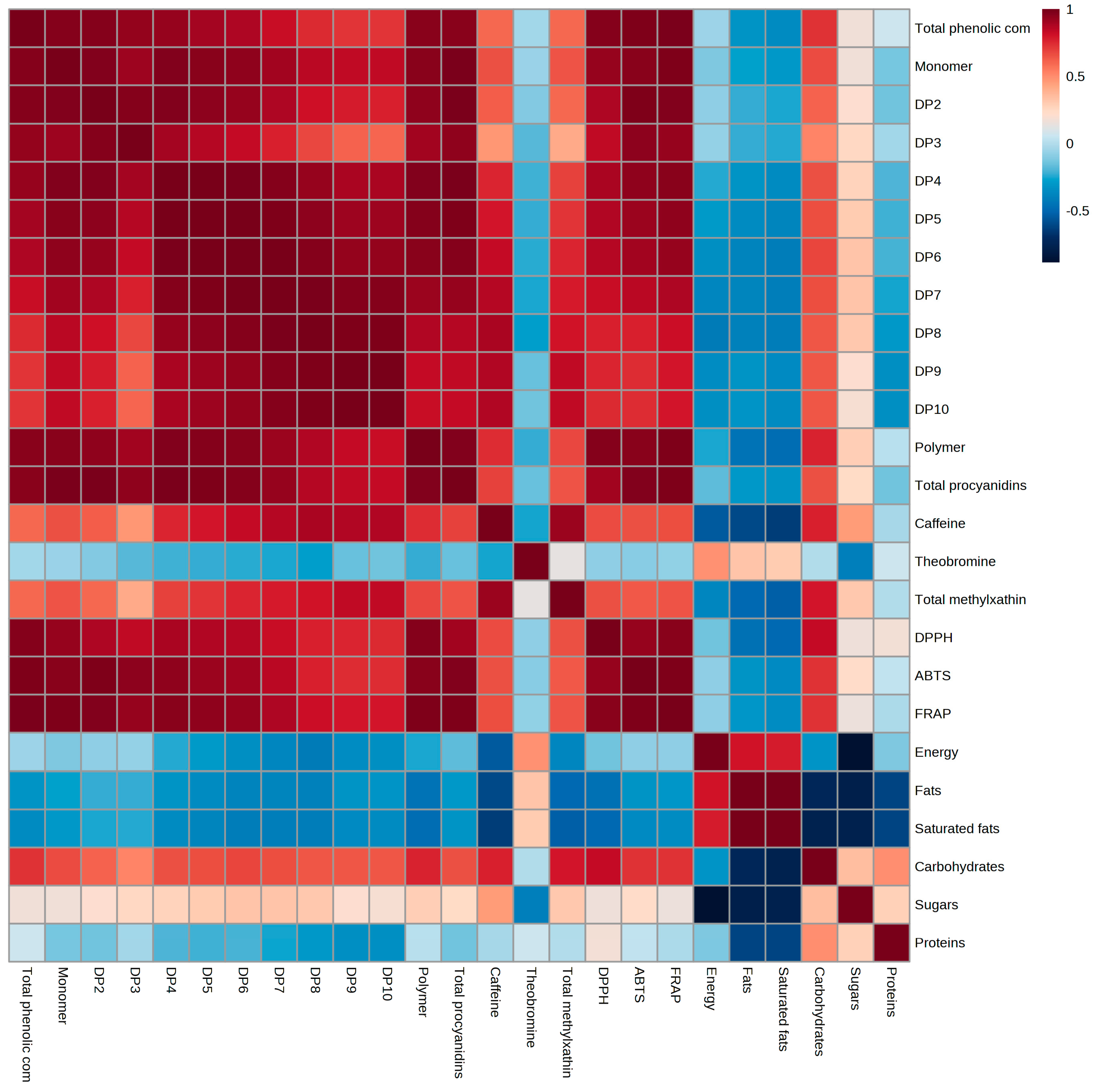
3.3. Procyanidin Content of Cocoas
3.4. Methyxanthines Content of Cocoas
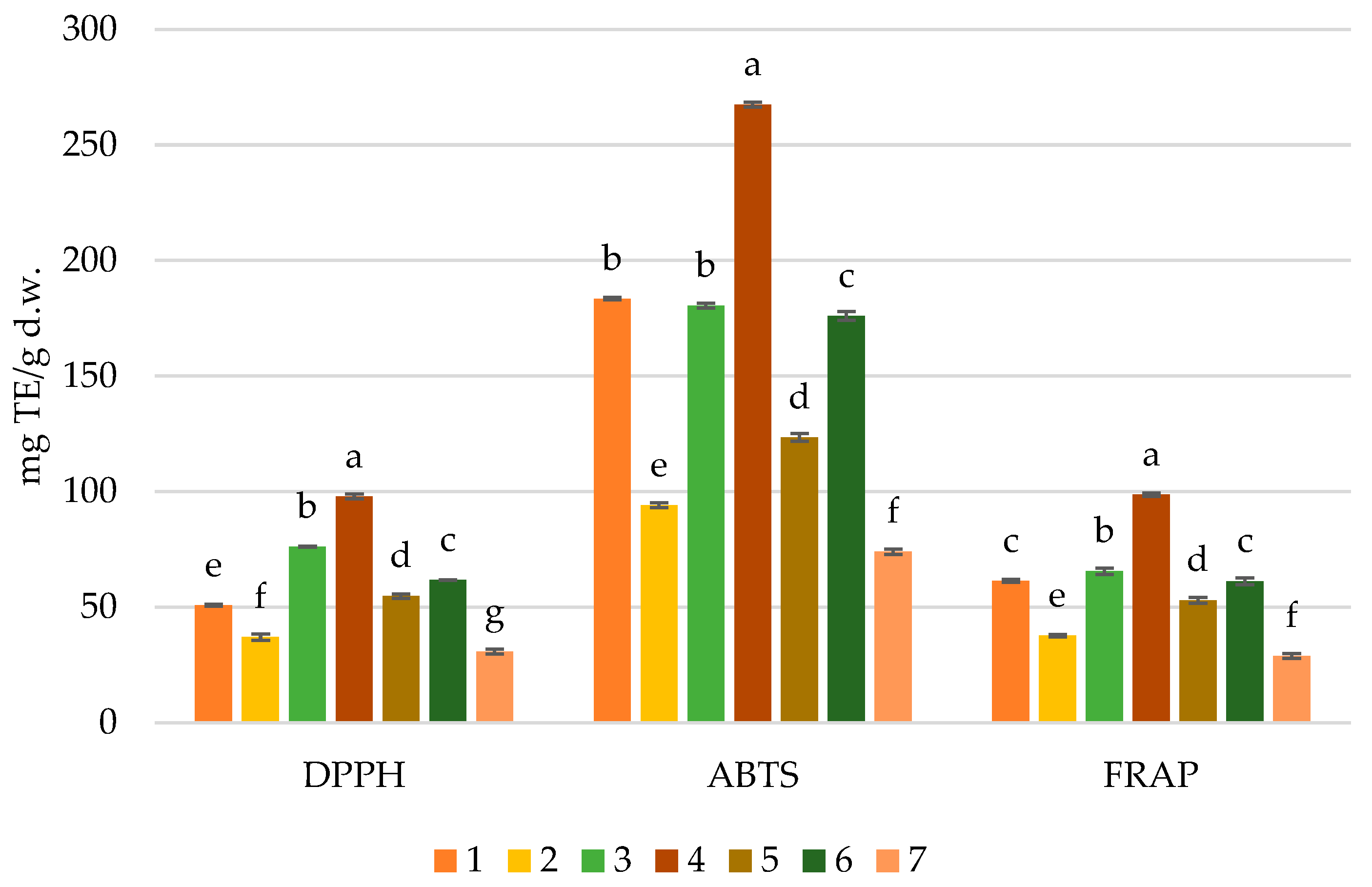
3.5. Antioxidant Activity of Cocoas by DPPH, ABST and FRAP
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Statista. Available online: https://www.statista.com/ (accessed on 4 May 2022).
- Cocoa Producing Countries 2020. Available online: https://worldpopulationreview.com/country-rankings/cocoa-producing-countries (accessed on 30 January 2023).
- Global Consumption of Cocoa by Rezgion 2016|Statista. Available online: https://www.statista.com/statistics/263150/consumption-of-cocoa-by-continent/ (accessed on 30 January 2023).
- Global Trade Magazine—THE Magazine for U.S. Companies Doing Business Globally. Available online: https://www.globaltrademag.com/ (accessed on 30 January 2023).
- Cinar, Z.Ö.; Atanassova, M.; Tumer, T.B.; Caruso, G.; Antika, G.; Sharma, S.; Sharifi-Rad, J.; Pezzani, R. Cocoa and cocoa bean shells role in human health: An updated review. J. Food Compos. Anal. 2021, 103, 104115. [Google Scholar] [CrossRef]
- Gu, L.; House, S.E.; Wu, X.; Ou, B.; Prior, R.L. Procyanidin and catechin contents and antioxidant capacity of cocoa and chocolate products. J. Agric. Food Chem. 2006, 54, 4057–4061. [Google Scholar] [CrossRef] [PubMed]
- Nehlig, A. The neuroprotective effects of cocoa flavanol and its influence on cognitive performance. Br. J. Clin. Pharmacol. 2013, 75, 716–727. [Google Scholar] [CrossRef] [PubMed]
- Valverde García, D.; Pérez Esteve, É.; Barat Baviera, J.M. Changes in cocoa properties induced by the alkalization process: A review. Compr. Rev. Food Sci. Food Saf. 2020, 19, 2200–2221. [Google Scholar] [CrossRef]
- Cova, I.; Leta, V.; Mariani, C.; Pantoni, L.; Pomati, S. Exploring cocoa properties: Is theobromine a cognitive modulator? Psychopharmacology 2019, 236, 561–572. [Google Scholar] [CrossRef]
- Franco, R.; Oñatibia-Astibia, A.; Martínez-Pinilla, E. Health benefits of methylxanthines in cacao and chocolate. Nutrients 2013, 5, 4159–4173. [Google Scholar] [CrossRef]
- Cheng, C.M.; Jalil, A.M.M.; Ismail, A. Phenolic and theobromine contents of commercial dark, milk and white chocolates on the Malaysian market. Molecules 2009, 14, 200–209. [Google Scholar] [CrossRef]
- Miller, K.B.; Hurst, W.J.; Flannigan, N.; Ou, B.; Lee, C.Y.; Smith, N.; Stuart, D.A. Survey of commercially available chocolate- and cocoa-containing products in the United States. 2. Comparison of flavan-3-ol content with nonfat cocoa solids, total polyphenols, and percent cacao. J. Agric. Food Chem. 2009, 57, 9169–9180. [Google Scholar] [CrossRef]
- Carrillo, L.C.; Londoño-Londoño, J.; Gil, A. Comparison of polyphenol, methylxanthines and antioxidant activity in Theobroma cacao beans from different cocoa-growing areas in Colombia. Food Res. Int. 2014, 60, 273–280. [Google Scholar] [CrossRef]
- Sorrenti, V.; Fortinguerra, S.; Caudullo, G.; Buriani, A. Deciphering the role of polyphenols in sports performance: From nutritional genomics to the gut microbiota toward phytonutritional epigenomics. Nutrients 2020, 12, 1265. [Google Scholar] [CrossRef]
- Urbańska, B.; Kowalska, J. Comparison of the total polyphenol content and antioxidant activity of chocolate obtained from roasted and unroasted cocoa beans from different regions of the world. Antioxidants 2019, 8, 283. [Google Scholar] [CrossRef] [PubMed]
- Álvarez, C.O.; Pérez, E.E.; Lares, M.D.C. Beneficio del cacao Criollo venezolano: Variaciones en composición proximal, metilxantinas y polifenoles. Rivar 2022, 9, 113–128. [Google Scholar] [CrossRef]
- Li, Y.; Feng, Y.; Zhu, S.; Luo, C.; Ma, J.; Zhong, F. The effect of alkalization on the bioactive and flavor related components in commercial cocoa powder. J. Food Compos. Anal. 2012, 25, 17–23. [Google Scholar] [CrossRef]
- Sumiyoshi, E.; Matsuzaki, K.; Sugimoto, N.; Tanabe, Y.; Hara, T.; Katakura, M.; Miyamoto, M.; Mishima, S.; Shido, O. Sub-chronic consumption of dark chocolate enhances cognitive function and releases nerve growth factors: A parallel-group randomized trial. Nutrients 2019, 11, 2800. [Google Scholar] [CrossRef]
- Brickman, A.M.; Khan, U.A.; Provenzano, F.A.; Yeung, L.K.; Suzuki, W.; Schroeter, H.; Wall, M.; Sloan, R.P.; Small, S.A. Enhancing dentate gyrus function with dietary flavanols improves cognition in older adults. Nat. Neurosci. 2014, 17, 1798–1803. [Google Scholar] [CrossRef]
- Neshatdoust, S.; Saunders, C.; Castle, S.M.; Vauzour, D.; Williams, C.; Butler, L.; Lovegrove, J.A.; Spencer, J.P.E. High-flavonoid intake induces cognitive improvements linked to changes in serum brain-derived neurotrophic factor: Two randomised, controlled trials. Nutr. Health Aging 2016, 4, 81. [Google Scholar] [CrossRef]
- Fajardo Daza, J.A.; Ibarra, C.A.; Arturo Perdomo, D.; Herrera Ruales, F.C. Optimization of ultrasound assisted extraction of polyphenols in cocoa beans. Vitae 2020, 27, 1–8. [Google Scholar] [CrossRef]
- González-Barrio, R.; Nuñez-Gomez, V.; Cienfuegos-Jovellanos, E.; García-Alonso, F.J.; Periago-Castón, M.J. Improvement of the Flavanol Profile and the Antioxidant Capacity of Chocolate Using a Phenolic Rich Cocoa Powder. Foods 2020, 9, 189. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. [14] Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. In Oxidants and Antioxidants Part A; Methods in Enzymology; Academic Press: Cambridge, MA, USA, 1999; Volume 299, pp. 152–178. [Google Scholar]
- Gómez-Caravaca, A.M.; López-Cobo, A.; Verardo, V.; Segura-Carretero, A.; Fernández-Gutiérrez, A. HPLC-DAD-q-TOF-MS as a powerful platform for the determination of phenolic and other polar compounds in the edible part of mango and its by-products (peel, seed, and seed husk). Electrophoresis 2016, 37, 1072–1084. [Google Scholar] [CrossRef]
- Robbins, R.J.; Leonczak, J.; Johnson, J.C.; Li, J.; Kwik-Uribe, C.; Prior, R.L.; Gu, L. Method performance and multi-laboratory assessment of a normal phase high pressure liquid chromatography-fluorescence detection method for the quantitation of flavanols and procyanidins in cocoa and chocolate containing samples. J. Chromatogr. A 2009, 1216, 4831–4840. [Google Scholar] [CrossRef]
- Alañón, M.E.; Castle, S.M.; Siswanto, P.J.; Cifuentes-Gómez, T.; Spencer, J.P.E. Assessment of flavanol stereoisomers and caffeine and theobromine content in commercial chocolates. Food Chem. 2016, 208, 177–184. [Google Scholar] [CrossRef]
- Razola-Díaz, M.D.C.; Gómez-Caravaca, A.M.; Guerra-Hernández, E.J.; Garcia-Villanova, B.; Verardo, V. New Advances in the Phenolic Composition of Tiger Nut (Cyperus esculentus L.) by-Products. Foods 2022, 11, 343. [Google Scholar] [CrossRef] [PubMed]
- Razola-Díaz, M.D.C.; Gómez-Caravaca, A.M.; López de Andrés, J.; Voltes-Martínez, A.; Zamora, A.; Pérez-Molina, G.M.; Castro, D.J.; Marchal, J.A.; Verardo, V. Evaluation of Phenolic Compounds and Pigments Content in Yellow Bell Pepper Wastes. Antioxidants 2022, 11, 557. [Google Scholar] [CrossRef] [PubMed]
- Razola-Díaz, M.D.C.; Aznar-Ramos, M.J.; Guerra-Hernández, E.J.; García-Villanova, B.; Gómez-Caravaca, A.M.; Verardo, V. Establishment of a Sonotrode Ultrasound-Assisted Extraction of Phenolic Compounds from Apple Pomace. Foods 2022, 11, 3809. [Google Scholar] [CrossRef] [PubMed]
- Razola-Díaz, M.D.C.; Guerra-Hernández, E.J.; Rodríguez-Pérez, C.; Gómez-Caravaca, A.M.; García-Villanova, B.; Verardo, V. Optimization of Ultrasound-Assisted Extraction via Sonotrode of Phenolic Compounds from Orange By-Products. Foods 2021, 10, 1120. [Google Scholar] [CrossRef] [PubMed]
- Granato, D.; de Araújo Calado, V.Ô.M.; Jarvis, B. Observations on the use of statistical methods in Food Science and Technology. Food Res. Int. 2014, 55, 137–149. [Google Scholar] [CrossRef]
- Samaniego, I.; Espín, S.; Quiroz, J.; Ortiz, B.; Carrillo, W.; García-Viguera, C.; Mena, P. Effect of the growing area on the methylxanthines and flavan-3-ols content in cocoa beans from Ecuador. J. Food Compos. Anal. 2020, 88, 103448. [Google Scholar] [CrossRef]
- Borja Fajardo, J.G.; Horta Tellez, H.B.; Peñaloza Atuesta, G.C.; Sandoval Aldana, A.P.; Mendez Arteaga, J.J. Antioxidant activity, total polyphenol content and methylxantine ratio in four materials of Theobroma cacao L. from Tolima, Colombia. Heliyon 2022, 8, e09402. [Google Scholar] [CrossRef]
- Elwers, S.; Zambrano, A.; Rohsius, C.; Lieberei, R. Differences between the content of phenolic compounds in Criollo, Forastero and Trinitario cocoa seed (Theobroma cacao L.). Eur. Food Res. Technol. 2009, 229, 937–948. [Google Scholar] [CrossRef]
- Todorovic, V.; Milenkovic, M.; Vidovic, B.; Todorovic, Z.; Sobajic, S. Correlation between Antimicrobial, Antioxidant Activity, and Polyphenols of Alkalized/Nonalkalized Cocoa Powders. J. Food Sci. 2017, 82, 1020–1027. [Google Scholar] [CrossRef]
- Jayeola, C.O.; Oluwadun, A.O. Mycoflora and nutritional components of cocoa powder samples in South West Nigeria. Afr. J. Agric. Res. 2010, 5, 2694–2698. [Google Scholar]
- Joel, N.; Pius, B.; Deborah, A.; Chris, U. Production and quality evaluation of cocoa products (plain cocoa powder and chocolate). Am. J. Food. Nutr. 2013, 3, 31–38. [Google Scholar] [CrossRef]
- Ondo, S.E.; Ryu, G.H. Physicochemical and antioxidant properties of extruded cornmeal with natural cocoa powder. Food Sci. Biotechnol. 2013, 22, 167–175. [Google Scholar] [CrossRef]
- Redgwell, R.J.; Trovato, V.; Curti, D. Cocoa bean carbohydrates: Roasting-induced changes and polymer interactions. Food Chem. 2003, 80, 511–516. [Google Scholar] [CrossRef]
- Barišić, V.; Icyer, N.C.; Akyil, S.; Toker, O.S.; Flanjak, I.; Ačkar, Đ. Cocoa based beverages—Composition, nutritional value, processing, quality problems and new perspectives. Trends Food Sci. Technol. 2023, 132, 65–75. [Google Scholar] [CrossRef]
- OCU. Organización de Consumidores y Usuarios. Available online: https://www.ocu.org/ (accessed on 30 January 2023).
- Veit, M.; van Asten, R.; Olie, A.; Prinz, P. The role of dietary sugars, overweight, and obesity in type 2 diabetes mellitus: A narrative review. Eur. J. Clin. Nutr. 2022, 76, 1497–1501. [Google Scholar] [CrossRef]
- EUR-Lex-32000L0036-EN-EUR-Lex. Available online: https://eur-lex.europa.eu/legal-content/ES/ALL/?uri=celex:32000L0036 (accessed on 30 January 2023).
- Ilesanmi Adeyeye, E. Proximate, Mineral and Antinutrient Compositions of Natural Cocoa Cake, Cocoa Liquor and Alkalized Cocoa Powders. J. Adv. Pharm. Sci. Technol. 2016, 1, 12–28. [Google Scholar] [CrossRef]
- Petit, J.; Michaux, F.; Jacquot, C.; Chávez Montes, E.; Dupas, J.; Girard, V.; Gianfrancesco, A.; Scher, J.; Gaiani, C. Storage-induced caking of cocoa powder. J. Food Eng. 2017, 199, 42–53. [Google Scholar] [CrossRef]
- Alasti, F.M.; Asefi, N.; Maleki, R.; SeiiedlouHeris, S.S. The influence of three different types and dosage of alkaline on the inherent properties in cocoa powder. J. Food Sci. Technol. 2020, 57, 2561–2571. [Google Scholar] [CrossRef]
- Panda, A.; Alvarenga, N.; Lita da Silva, J.; Partidário, A.; Lageiro, M.; Roseiro, C.; Dias, J. Influence of cocoa origin on the nutritional characterization of chocolate. Eur. Food Res. Technol. 2022, 248, 2569–2577. [Google Scholar] [CrossRef]
- Ramos-Escudero, F.; Casimiro-Gonzales, S.; Fernández-Prior, Á.; Cancino Chávez, K.; Gómez-Mendoza, J.; Fuente-Carmelino, L.D.L.; Muñoz, A.M. Colour, fatty acids, bioactive compounds, and total antioxidant capacity in commercial cocoa beans (Theobroma cacao L.). LWT 2021, 147, 111629. [Google Scholar] [CrossRef]
- Grassia, M.; Salvatori, G.; Roberti, M.; Planeta, D.; Cinquanta, L. Polyphenols, methylxanthines, fatty acids and minerals in cocoa beans and cocoa products. J. Food Meas. Charact. 2019, 13, 1721–1728. [Google Scholar] [CrossRef]
- Kobori, K.; Maruta, Y.; Mineo, S.; Shigematsu, T.; Hirayama, M. Polyphenol-retaining decaffeinated cocoa powder obtained by supercritical carbon dioxide extraction and its antioxidant activity. Foods 2013, 2, 462–477. [Google Scholar] [CrossRef]
- Acosta-Otálvaro, E.; Valencia-Gallego, W.; Mazo-Rivas, J.C.; García-Viguera, C. Cocoa extract with high content of flavan 3-ols, procyanidins and methylxanthines. J. Food Sci. Technol. 2022, 59, 1152–1161. [Google Scholar] [CrossRef]
- Toro-Uribe, S.; Ibañez, E.; Decker, E.A.; Villamizar-Jaimes, A.R.; López-Giraldo, L.J. Food-safe process for high recovery of flavonoids from cocoa beans: Antioxidant aND HPLC-DAD-ESIMS/ MS analysis. Antioxidants 2020, 9, 364. [Google Scholar] [CrossRef] [PubMed]
- Quiroz-Reyes, C.N.; Fogliano, V. Design cocoa processing towards healthy cocoa products: The role of phenolics and melanoidins. J. Funct. Foods 2018, 45, 480–490. [Google Scholar] [CrossRef]
- Piggott, C.O.; Connolly, A.; FitzGerald, R.J. Application of ultrafiltration in the study of phenolic isolates and melanoidins from pale and black brewers’ spent grain. Int. J. Food Sci. Technol. 2014, 49, 2252–2259. [Google Scholar] [CrossRef]
- Cooper, K.A.; Campos-Giménez, E.; Alvarez, D.J.; Rytz, A.; Nagy, K.; Williamson, G. Predictive relationship between polyphenol and nonfat cocoa solids content of chocolate. J. Agric. Food Chem. 2008, 56, 260–265. [Google Scholar] [CrossRef]
- Belščak, A.; Komes, D.; Horžić, D.; Ganić, K.K.; Karlović, D. Comparative study of commercially available cocoa products in terms of their bioactive composition. Food Res. Int. 2009, 42, 707–716. [Google Scholar] [CrossRef]
- Botella-Martínez, C.; Lucas-Gonzalez, R.; Ballester-Costa, C.; Pérez-álvarez, J.Á.; Fernández-López, J.; Delgado-Ospina, J.; Chaves-López, C.; Viuda-Martos, M. Ghanaian cocoa (Theobroma cacao L.) bean shells coproducts: Effect of particle size on chemical composition, bioactive compound content and antioxidant activity. Agronomy 2021, 11, 401. [Google Scholar] [CrossRef]
- Pico-Hernández, S.M.; Murillo-Méndez, C.J.; López-Giraldo, L.J. Extraction, separation, and evaluation of antioxidant effect of the different fractions of polyphenols from cocoa beans. Rev. Colomb. Quim. 2020, 49, 19–27. [Google Scholar] [CrossRef]
- Sioriki, E.; Tuenter, E.; Van de Walle, D.; Lemarcq, V.; Cazin, C.S.J.; Nolan, S.P.; Pieters, L.; Dewettinck, K. The effect of cocoa alkalization on the non-volatile and volatile mood-enhancing compounds. Food Chem. 2022, 381, 132082. [Google Scholar] [CrossRef] [PubMed]
- Sioriki, E.; Lemarcq, V.; Alhakim, F.; Triharyogi, H.; Tuenter, E.; Cazin, C.S.J.; Nolan, S.P.; Pieters, L.; Van de Walle, D.; Dewettinck, K. Impact of alkalization conditions on the phytochemical content of cocoa powder and the aroma of cocoa drinks. LWT 2021, 145, 1–9. [Google Scholar] [CrossRef]
- Stanley, T.H.; Smithson, A.T.; Neilson, A.P.; Anantheswaran, R.C.; Lambert, J.D. Analysis of Cocoa Proanthocyanidins Using Reversed Phase High-Performance Liquid Chromatography and Electrochemical Detection: Application to Studies on the Effect of Alkaline Processing. J. Agric. Food Chem. 2015, 63, 5970–5975. [Google Scholar] [CrossRef]
- Oracz, J.; Nebesny, E.; Zyzelewicz, D.; Budryn, G.; Luzak, B. Bioavailability and metabolism of selected cocoa bioactive compounds: A comprehensive review. Crit. Rev. Food Sci. Nutr. 2020, 60, 1947–1985. [Google Scholar] [CrossRef] [PubMed]
- Goya, L.; Kongor, J.E.; de Pascual-Teresa, S. From Cocoa to Chocolate: Effect of Processing on Flavanols and Methylxanthines and Their Mechanisms of Action. Int. J. Mol. Sci. 2022, 23, 14365. [Google Scholar] [CrossRef] [PubMed]
- Steinberg, F.M.; Holt, R.R.; Schmitz, H.H.; Keen, C.L. Cocoa procyanidin chain length does not determine ability to protect LDL from oxidation when monomer units are controlled. J. Nutr. Biochem. 2002, 13, 645–652. [Google Scholar] [CrossRef]
- Baba, S.; Osakabe, N.; Kato, Y.; Natsume, M.; Yasuda, A.; Kido, T.; Fukuda, K.; Muto, Y.; Kondo, K. Continuous intake of polyphenolic compounds containing cocoa powder reduces LDL oxidative susceptibility and has beneficial effects on plasma HDL-cholesterol concentrations in humans. Am. J. Clin. Nutr. 2007, 85, 709–717. [Google Scholar] [CrossRef]
- Quelal-Vásconez, M.A.; Lerma-García, M.J.; Pérez-Esteve, É.; Arnau-Bonachera, A.; Barat, J.M.; Talens, P. Changes in methylxanthines and flavanols during cocoa powder processing and their quantification by near-infrared spectroscopy. LWT 2020, 117, 108598. [Google Scholar] [CrossRef]
- Peláez, P.; Bardón, I.; Camasca, P. Methylxanthine and catechin content of fresh and fermented cocoa beans, dried cocoa beans, and cocoa liquor. Sci. Agropecu. 2016, 7, 355–365. [Google Scholar] [CrossRef]
- Sansone, R.; Ottaviani, J.I.; Rodriguez-Mateos, A.; Heinen, Y.; Noske, D.; Spencer, J.P.; Crozier, A.; Merx, M.W.; Kelm, M.; Schroeter, H.; et al. Methylxanthines enhance the effects of cocoa flavanols on cardiovascular function: Randomized, double-masked controlled studies. Am. J. Clin. Nutr. 2017, 105, 352–360. [Google Scholar] [CrossRef] [PubMed]
- Ali, F.; Ranneh, Y.; Ismail, A.; Esa, N.M. Identification of phenolic compounds in polyphenols-rich extract of Malaysian cocoa powder using the HPLC-UV-ESI—MS/MS and probing their antioxidant properties. J. Food Sci. Technol. 2015, 52, 2103–2111. [Google Scholar] [CrossRef] [PubMed]
| Sample | Origin | Alkalized |
|---|---|---|
| 1 | Venezuela | N |
| 2 | Ivory Coast | A |
| 3 | Ivory Coast | N |
| 4 | Peru | N |
| 5 | Dominican Republic | N |
| 6 | West Africa | N |
| 7 | West Africa | A |
| Per 100 g of Product | Energy (kJ) | Fat (g) | Saturated Fat (g) | Carbohydrates (g) | Sugars (g) | Protein (g) | Salt (g) |
|---|---|---|---|---|---|---|---|
| Average | 1457.6 | 16.1 | 9.8 | 16.3 | 1.1 | 21.5 | 0.03 |
| SD | 158.4 | 5.84 | 3.60 | 7.99 | 0.73 | 3.26 | 0.02 |
| Min. | 1249.8 | 11.0 | 6.5 | 9.0 | 0.0 | 19.0 | 0.00 |
| Max. | 1634.4 | 23.0 | 14.0 | 28.0 | 2.0 | 28.0 | 0.06 |
| CV (%) | 10.9 | 36.2 | 36.6 | 49.0 | 67.9 | 15.2 | 80.9 |
| 1 (µg CE/g d.w.) | 2 (µg CE/g d.w.) | 3 (µg CE/g d.w.) | 4 (µg CE/g d.w.) | 5 (µg CE/g d.w.) | 6 (µg CE/g d.w.) | 7 (µg CE/g d.w.) | |
|---|---|---|---|---|---|---|---|
| Cat + Epicat | 4536.90 ± 11.81 b | 2238.23 ± 11.90 f | 4288.26 ± 12.33 c | 7816.56 ± 16.66 a | 4132.57 ± 10.13 e | 4201.90 ± 10.37 d | 1999.86 ± 9.85 g |
| Dp2 | 5018.78 ± 12.39 b | 1607.62 ± 10.41 f | 3744.19 ± 9.41 d | 7972.94 ± 17.33 a | 3196.20 ± 8.22 e | 4476.44 ± 11.02 c | 928.93 ± 13.33 g |
| Dp3 | 2311.63 ± 12.86 c | 589.02 ± 2.15 f | 1731.81 ± 6.74 d | 3141.32 ± 6.48 a | 1538.89 ± 3.71 e | 2462.54 ± 7.51 b | 233.43 ± 2.63 g |
| Dp4 | 1191.32 ± 18.37 c | 250.11 ± 2.26 f | 902.74 ± 2.91 d | 3268.16 ± 6.91 a | 743.68 ± 2.19 e | 1403.49 ± 3.29 b | 28.94 ± 3.58 g |
| Dp5 | 612.22 ± 7.47 c | 93.72 ± 1.82 f | 447.36 ± 1.68 d | 2018.99 ± 4.67 a | 351.58 ± 1.23 e | 796.08 ± 1.49 b | <LOQ |
| Dp6 | 228.95 ± 1.44 c | 2.38 ± 0.76 f | 180.24 ± 0.95 d | 996.17 ± 2.10 a | 113.11 ± 0.32 e | 339.24 ± 0.99 b | <LOQ |
| Dp7 | 124.31 ± 0.69 c | <LOQ | 79.71 ± 0.57 d | 795.09 ± 2.06 a | 43.24 ± 0.45 e | 221.48 ± 1.25 b | <LOQ |
| Dp8 | 1.16 ± 1.52 c | <LOQ | <LOQ | 330.58 ± 1.00 a | <LOQ | 63.11 ± 0.42 b | <LOQ |
| Dp9 | <LOQ | <LOQ | <LOQ | 112.03 ± 0.65 a | <LOQ | 1.78 ± 0.32 b | <LOQ |
| Dp10 | <LOQ | <LOQ | <LOQ | 28.94 ± 0.56 | <LOQ | <LOQ | <LOD |
| Polymers | 729.98 ± 3.99 d | 252.95 ± 2.86 f | 1023.22 ± 1.35 c | 2094.28 ± 3.95 a | 608.21 ± 1.38 e | 1132.00 ± 2.73 b | 105.18 ± 6.25 g |
| Sum of Procyanidins | 14,756.84 ± 70.56 c | 5035.55 ± 32.17 f | 12,397.53 ± 35.95 d | 28,575.06 ± 62.37 a | 10,727.47 ± 27.62 e | 15,101.75 ± 39.40 b | 3298.89 ± 35.64 g |
| Sample | Caffeine (mg/g) | Theobromine (mg/g) | Total Methylxanthines (mg/g) | T/C Ratio |
|---|---|---|---|---|
| 1 | 9.56 ± 0.21 c | 15.14 ± 0.38 a | 24.70 ± 0.59 b–d | 1.58 |
| 2 | 11.23 ± 0.16 b,c | 12.30 ± 1.87 a,b | 23.53 ± 1.89 b–d | 1.10 |
| 3 | 13.78 ± 0.60 b | 14.69 ± 3.41 a | 28.47 ± 3.61 b | 1.07 |
| 4 | 27.06 ± 1.34 a | 12.09 ± 2.06 a,b | 39.15 ± 2.12 a | 0.45 |
| 5 | 5.41 ± 0.22 d | 14.04 ± 0.76 a | 19.44 ± 0.87 d | 2.60 |
| 6 | 13.62 ± 0.06 b | 8.87 ± 0.44 b | 22.49 ± 0.49 c,d | 0.65 |
| 7 | 14.24 ± 0.57 b | 13.29 ± 0.08 a,b | 27.54 ± 2.93 b,c | 0.93 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Razola-Díaz, M.d.C.; Aznar-Ramos, M.J.; Verardo, V.; Melgar-Locatelli, S.; Castilla-Ortega, E.; Rodríguez-Pérez, C. Exploring the Nutritional Composition and Bioactive Compounds in Different Cocoa Powders. Antioxidants 2023, 12, 716. https://doi.org/10.3390/antiox12030716
Razola-Díaz MdC, Aznar-Ramos MJ, Verardo V, Melgar-Locatelli S, Castilla-Ortega E, Rodríguez-Pérez C. Exploring the Nutritional Composition and Bioactive Compounds in Different Cocoa Powders. Antioxidants. 2023; 12(3):716. https://doi.org/10.3390/antiox12030716
Chicago/Turabian StyleRazola-Díaz, María del Carmen, María José Aznar-Ramos, Vito Verardo, Sonia Melgar-Locatelli, Estela Castilla-Ortega, and Celia Rodríguez-Pérez. 2023. "Exploring the Nutritional Composition and Bioactive Compounds in Different Cocoa Powders" Antioxidants 12, no. 3: 716. https://doi.org/10.3390/antiox12030716
APA StyleRazola-Díaz, M. d. C., Aznar-Ramos, M. J., Verardo, V., Melgar-Locatelli, S., Castilla-Ortega, E., & Rodríguez-Pérez, C. (2023). Exploring the Nutritional Composition and Bioactive Compounds in Different Cocoa Powders. Antioxidants, 12(3), 716. https://doi.org/10.3390/antiox12030716









