Abstract
Bee pollen is a hive product made up of flower pollen grains, nectar, and bee salivary secretions that beekeepers can collect without damaging the hive. Bee pollen, also called bee-collected pollen, contains a wide range of nutritious elements, including proteins, carbs, lipids, and dietary fibers, as well as bioactive micronutrients including vitamins, minerals, phenolic, and volatile compounds. Because of this composition of high quality, this product has been gaining prominence as a functional food, and studies have been conducted to show and establish its therapeutic potential for medical and food applications. In this context, this work aimed to provide a meticulous summary of the most relevant data about bee pollen, its composition—especially the phenolic compounds—and its biological and/or therapeutic properties as well as the involved molecular pathways.
1. Introduction
The pollen grain is the male flower’s reproductive organ; in other words, it carries the organ that carries the male gametes to their progenitor cells. It is produced and spread by higher plants as part of their reproductive process [1]. Bee pollen is the final result of the agglutination of pollen grains harvested by worker bees, held together by nectar and/or honey, and gland secretions, and collected at the hive entrance [2]. It is among the most important bee products gaining popularity as a functional food due to its high concentration of bioactive compounds known for their benefits for both mental and physical health, such as proteins, dietary fibers, lipids, carbohydrates, and minerals [3]. Furthermore, bee pollen is known as “the perfectly complete food” due to its strong antioxidant potential and the presence of antioxidant compounds such as polyphenols, flavonoids, carotenoids, and vitamins (A, C, and E) giving this product a high antioxidant potential and making it the latest trend in dietary supplements [4]. Because of this high load of natural bioactive molecules, various scientific studies have reported that bee pollen possesses a wide spectrum of biological properties such as antioxidant [5], hypoglycemic [6], anti-inflammatory [7], antibacterial [8], and anticancer [9]. As a result of all of this, the German Federal Ministry of Health has formally acknowledged bee pollen as a drug. [10]. This paper aims to provide a comprehensive overview of bee pollen in terms of bee pollen harvesting, chemical and nutritional content, and, finally, its biological and therapeutic characteristics. Furthermore, we intended to unveil for the first time the proposed mechanism of action and the involved biomolecular pathways of the various bioactive compounds of bee pollen that are responsible for the improvement of oxidative stress and associated health conditions.
2. Methodology
The following online databases were employed to collect the literature data for this paper: Science Direct, Google Scholar, Web of Science, Pub-Med, and Scopus, using the keywords: “bee pollen”; “bee-collected pollen”; “chemical composition of bee pollen”; “therapeutic effect of bee pollen”; “functional effect of bee pollen”, “protective effect of bee pollen”, and “nutritional value of bee pollen”. After collecting and reviewing all selected articles, their general ideas were summarized and used in this review.
3. Bee Pollen: From Flowers to the Hive
It is commonly known that nectar and pollen grains are the main sources of nutrients required for the survival and health of bee colonies. Nectar is mainly produced by the nectariferous glands of plants and serves as the raw material for honey production [11], whereas pollen grains represent the plant’s male gametophyte. They take the form of fine dust with tiny particles that vary in color based on the floral origin [12]. Flower nectar provides bees with carbon and nitrogen, while pollen grains provide other dietary components such as lipids, proteins, vitamins, and minerals [13,14].
The flower–bee interaction is a mutualistic relationship in which the flowers reproduce sexually and the bees feed on nectar and pollen [14]. In this respect, plants adopt many techniques to be pollinated and reproduce, including the coloration of their petals and the emission of scents known as pheromones [15]. Worker honeybees perform hundreds of flights to blooms to collect the necessary amount of pollen and nectar [16]. The honeybees’ behavior of gathering nectar and pollen is known as “foraging” and it is highly vital and necessary for the survival of bee colonies. Foraging is a behavior that develops in worker bees between the ages of one and two weeks [17]. Young foraging bees conduct many scouting flights to become acquainted, and at the age of 21 days, they will leave their hives to seek and gather nectar, honeydew, pollen, water, and many essential elements, as well as the resin used to maintain the hive’s asepsis [18,19]. Foraging bees use their proboscis to collect nectar and water by pumping and capillarity; the liquids are stored in the foragers’ crops until they are discharged to the other workers in the hive [20], who then use their hind legs coated with short stiff hairs called “scopae” to squeeze the collected pollen grains into pollen balls using their saliva and honey, which they finally place into their pollen baskets [16]. The majority of bees have developed specialized mechanisms for transporting pollen to their nests and have adjusted their grooming behaviors to transfer pollen from their bodies to the hive [21].
When the foraging bees arrive at the hive, they cover the pollen balls with saliva, then compact the alveoli with a layer of pollen balls and honey, and finally cap them with a layer of wax [22].
Lactic fermentation occurs at this stage due to the participation of lactic bacteria strains that proliferate inside the hives [14]. These bacteria are Pseudomonas, which consumes oxygen and creates an anaerobic environment; Lactobacillus, which converts carbohydrates into lactic acid; and finally, Saccharomyces, which ensures the metabolism of the rest of the sugars that exist in the medium. These reaction chains reduce the environment’s pH, prevent pollen germination, and improve bee pollen absorption capacity and nutritional value [23]. When bee-collected pollen is completely fermented, it becomes “bee bread,” which provides additional proteins for bees, particularly during the period of royal jelly production, as well as nutrition for larvae, and future workers who are fed a diet of pollen, honey, and a small quantity of royal jelly [18].
4. Chemical Composition
Bee pollen is one of the magical superfoods due to its extremely wide range of nutritional compounds and microelements. However, this composition may be affected by botanical origin, harvesting season, and storage methods (freeze-drying duration). Considering this large variability, its nutritional and chemical composition has been extensively studied, summarized, and standardized. In this section, we assembled the main macro- and micronutrients of bee-collected pollen.
4.1. Main Compound
4.1.1. Water
Several studies have succeeded in quantifying the water content in bee pollen samples, the results obtained being largely variable and dependent on storage conditions (fresh or dried bee pollen), botanical, and geographical origins [24].
In fresh bee-collected pollen, water content varies between 20 and 30% [2]. However, this high humidity is considered a favorable environment for bacterial and fungal growth [25,26,27]. As a result, the bee pollen freezing process must begin immediately after harvesting [28]. Other researchers prefer the nitrogen processing of fresh bee pollen to preserve the optimal microbiological and nutritional properties [29]. Meanwhile, the maximum water content allowed in dried bee pollen depends on the country and must not exceed 4% according to the Brazilian legislation, 6% in Poland and Switzerland, 8% in Argentina, and 10% in Bulgaria [2,24,30,31,32,33]. Thus, the moisture content can be used as a bee pollen quality criterion.
4.1.2. Protein Content
The production of protein for human consumption as well as defining the need for protein, evaluating its quality to meet human needs, and managing the consequences of variations in dietary protein intake are considered major public health issues. Consequently, it is necessary to ensure a sufficient daily supply of protein of good biological quality, since the human body is unable to store it. Therefore, bee pollen, which has high protein content, could guarantee an ideal diet in terms of protein requirement. In addition, pollen is the principal source of protein for bees, providing the necessary elements for their longevity, organ development, larva growth, and body size [34,35]. Proteins also provide essential substances for royal jelly production in the hive [36].The protein content is highly variable between plant species and harvesting geographic areas [37] and varies between 10 and 40 % of pollen dry weight [2].
4.1.3. Amino Acids
Amino acids are crucial not only for protein synthesis but also for the biosynthesis of hormones as well as other molecules with a biological role. Nutritionally, two kinds of amino acids are distinguished, namely, the essential amino acids that the human organism cannot synthesize, and non-essential amino acids that our body has all the machinery to synthesize. In the same context, bee-collected pollen is often considered the “most natural perfect food” because it is a great source of all essential amino acids needed in honey bee and human nutrition [38,39]. This content varies strongly from species to species and depends on botanical and geographical origin, climatic conditions, and nutrient availability in the plant [37,40,41]. Therefore, the amino acid amount can be used as an indicator of freshness, storage, and drying process adequacy [3,42].
The total amino acid content in bee pollen has been quantified by many researchers, and it generally ranges between 108.1 and 287.7 mg/g of bee pollen [43]. Concerning the amino acid profile, De-Melo and Almeida-Muradian have reported twenty-five amino acids, eight of which are essential (valine, leucine, isoleucine, lysine, phenylalanine, threonine, histidine, and methionine). Tryptophan is usually undetectable because of the hydrolysis method employed in the determination of the amino acids. However, tryptophan was detected in Chinese, Slovenian, Spanish, and Italian bee pollen using specific high-performance liquid chromatography methods [44,45,46]. The remaining amino acids are non-essential, such as aspartic acid, alanine, glycine, glutamine, arginine, asparagine, glutamic acid, serine, tyrosine, cystine, cysteine, γ-aminobutyric acid (GABA), ornithine, proline, and homoserine [3]. It has been reported that proline is the most abundant amino acid in dried bee pollen from many countries, while glutamic acid is the main amino acid in freshly collected bee pollen [3,44,47].
4.1.4. Carbohydrates
Bee pollen is composed of pollen mixed with nectar, and the bee’s salivary secretions. Carbohydrates are the major fraction of bee pollen (13–55%), they are mainly polysaccharides and cell wall material [2]. Carbohydrates can be affected by botanical and geographical origin, harvesting methods, and conditioning processes such as high temperature when drying fresh bee pollen [48].
- Sugar
Sugar content is the most important quality parameter in bee pollen characterization studies and should not be less than 40 % [2]. Nevertheless, sugars are generally neglected or included in the total carbohydrates which also regroups the dietary fiber and starch [49]. The sugar composition of bee pollen has been assessed by many studies either as reducing sugar [50,51,52,53,54] or as individual sugars [41,44,49,55,56] and all showed a predominance of glucose and fructose as monosaccharides which represented the major amount of sugar fraction. Sucrose, maltose, trehalose, turanose, and melezitose have been identified in previous studies [49,57]. The sugar content and profile can be considerably influenced by nectar added by bees during the packaging and storage of bee-collected pollen [50,58]. Floral source, drying process, and extraction methods can also greatly affect the sugar content [48,56]
Mannitol is a polyol previously identified and isolated in high concentrations from bee pollen collected by stingless bee Melipona subnitida from Jandaíra, Brazil, Tetragonula biroi Friese from the Philippines, and Trigona from Malaysia [41,59,60]. According to these authors, the significant amount of mannitol did not depend on the floral origin, and it is supposed that the previous stingless bee species are capable of converting the glucose and fructose mainly found in flowers into mannitol via their salivary enzymes.
On this basis, sugars can be considered as an additional parameter for establishing quality standards for bee pollen.
- Dietary fibers
Dietary fibers describe the soluble and insoluble fraction of fibers from plant-based foods, which include hemicellulose, cellulose, lignin, oligosaccharides, pectins, gums, and waxes; these compounds are resistant to digestive enzymes, thus they are neither hydrolyzed nor absorbed in the intestinal tract [61,62]. Many recent studies have supported the crucial physiological role of dietary fibers in the human body; indeed, they are involved in type 2 diabetes management by the selective promotion of certain gut microbiota [63,64]. A high-fiber diet was found to be effective in many conditions such as obesity-related disorders, cardiovascular diseases, constipation, inflammatory bowel diseases, and colon cancers [65,66,67,68,69]. Regarding this, bee pollen can be a good source of dietary fiber, especially crude fibers. Despite the importance of bee pollen in the human diet, few characterization studies have focused on the determination of the dietary fiber content of bee pollen. According to Compos et al., total dietary fiber should range between 0.3 and 20 g/100 g of bee pollen dry weight [2]. Dietary fiber content has been reported by a few studies. For instance, a recent study carried out on Slovenian bee pollen showed a range of 10–21.4 g/100 g dry weight bee pollen with 73–82% of crude fiber [49]. Brazilian researchers reported an average of 3.6 ± 1.4 g/100 g of dry weight bee pollen [54]. Colombian bee pollen has also been characterized, and the results showed an average of 14.5 ± 3.5 g/100 g dry weight [70], while El-Kazafy recorded that different Egyptian bee pollen showed a range of 0.15 ± 0.01 and 1.70 ± 0.02 g/100 g dry weight bee pollen [47]. Dietary fiber content may vary according to the botanical origin and methods used during hydrolysis.
4.1.5. Lipid and Fat Content
Physiologically, the human body uses a variety of biosynthetic pathways to synthesize lipids; however, some important lipids cannot be obtained through biosynthesis and must be obtained from food. Essential fatty acids (especially omega-3 fatty acids) are involved in many biological functions and play an important role in the prevention of inflammatory and cardiovascular diseases and hormone-dependent tumors. [71,72]. Indeed, bee pollen can be a great source of these compounds since they are crucial for royal jelly production [73]. The lipidic fraction is most attractive for bees, and thus plants with high lipid concentration pollen are more frequently visited [37]. According to Campos et al., lipid content ranges between 1–13 g/100 g [2], while De-Melo and Almeida-Muradian, reported that the total lipid fraction can reach 22 g/100 g [3]. In the same context, Thakur et al. and De-Melo et al. have reported a huge variation of lipid content in monofloral bee pollen from different countries; Brassica napus bee pollen from Brazil, China, India, and Greece showed a total lipid content of 7.4%, 6.6 %, 12.38%, and 7.76% respectively; Cistus bee pollen from Italy, Spain, and Greece showed a total lipid content of −1.9%, 7.2%, and 3.80% respectively. Cocos nucifera bee pollen from India and Brazil showed 10.43% and 4.6–5.1% total lipid content [3,73].
The lipid profile of bee pollen has been barely investigated while most research studies have focused on the protein, carbohydrate, and antioxidant content. According to Ares et al., carotenoids, steroids, and fatty acids are the main constituents of bee pollen’s total lipid fraction [52]. A study conducted by Li et al. on three monofloral bee pollen samples from China suggested the presence of nine lipid classes, including triglycerides and fatty acids [74].
The fatty acid profile of bee pollen varies between saturated fatty acids, which include mainly the myristic, stearic, and palmitic acids, and unsaturated fatty acids, which include the oleic, α-linolenic (omega-3), and linoleic (omega-6) acids. This fraction is the most dominant in bee pollen [4,44,73,75,76,77]. Other lipid classes such as phospholipids, triterpenes (oleanolic and ursolic acids), and plant sterols (β-sitosterol) have been isolated from bee pollen in smaller amounts [4,36,76]. All these studies have reported that the lipid content in the studied bee pollen depends on botanical origin, harvesting season, drying, storage, and beekeeping methods.
4.2. Micronutrients
Micronutrients include minerals and vitamins that are not involved in the energetic balance but are essential for all chemical reactions and for the maintenance of life. Micronutrients are required in small amounts by the body for its growth and development from birth to old age. In a recent report (2020), most food and health organizations estimated that more than two billion people globally suffer from micronutrient deficiency occurring due to an insufficient intake or impaired absorption of vitamins and minerals [78,79]. Generally, micronutrient deficiency is considered a global health concern for all ages. During pregnancy, this deficiency has a devastating effect on both the mother and her fetus, being associated with anemia, hypertension, gestational diabetes, thyroid disorders, obstetric complications, and failure in the growth and development of the fetus, among other conditions [80,81,82,83,84]. During childhood, micronutrient insufficiency may affect the mental and physical development of the children and increase their vulnerability to and exacerbation of diseases such as impaired host defense and infections, developmental disabilities, autism, ocular disorders, and general loss of energy and potential [85,86,87]. The elderly population is also vulnerable to micronutrient deficiencies, which can lead to many age-related diseases such as mild cognitive decline, high risk of type 2 diabetes, cardiovascular diseases, acute respiratory infections especially coronavirus infection, and immune function impairments [88,89,90,91]. Based on what has been stated above, it is clear that maintaining a micronutrient-rich diet may provide enough protection against all of these pathologies. This protection can be guaranteed by consuming a large variety of well-balanced and rich natural products such as bee pollen.
4.2.1. Minerals
There are around twenty minerals that are essential in the human diet and are classified as macro-elements and oligo-elements, also known as trace elements. Mineral deficiency in the human body causes several metabolic problems and severe developmental defects in pregnancy, and significantly affects the individual’s wellness and economic output [92]. Bee pollen is a good source of essential minerals for the development of bees as well as humans, which represent 2–6 % of its content, with about 25 elements [2]. This makes bee pollen an interesting value-added product.
Potassium (K) is the principal mineral element found in high concentrations in bee pollen (400–2000 mg/100 g of bee pollen), and 15 g of bee pollen covers up to 25% of the recommended daily intake (RDI) of this element (2000 mg/day). Phosphorus (P) is the second element mainly present in bee pollen (0.80–6 mg/100 g of bee pollen), covering 16% of the RDI (1000 mg/day) of 15 g of bee pollen. The third important element is magnesium (Mg; 20–300 mg/100 g of bee pollen) which covers up to 23% of the RDI of this element (350 mg/day) of 15 g of bee pollen. Calcium (Ca) is also widely present in pollen (20–300 mg/100 g of pollen) and covers 7% of the RDI (1100 mg/day) of calcium. These elements are known for their crucial role in bone tissue formation by maintaining the proper osmotic pressure of blood as well as cellular fluids. Iron (Fe), zinc (Zn), copper (Cu), and manganese (Mn) are also microelements present in large quantities in bee pollen, covering up to 37%, 79%, 36%, and 85% of the respective RDIs. These trace elements play an important role in blood formation and also in the growth, development, and reproduction process [2,28,93,94].
There are other trace elements such as cobalt (Co), selenium (Se), molybdenum (Mo), and boron (B) which have been identified in bee pollen from different countries. Adequate intake of these trace elements is necessary to support bone and brain health, they play a key role in the maintenance of vitamin structure, reproduction, thyroid hormone metabolism, DNA synthesis, and protection against oxidative damage and infections [3,95,96,97].
Sodium (Na) is also a macro-element present in bee pollen; however, its content remains below 2 g/kg with a high K/Na ratio, and this ratio makes bee pollen beneficial and safe for daily diets with a good electrolyte balance [3]. The mineral content of bee pollen is recommended as a distinct marker of its floral and geographical origin as well as its quality [5,73].
4.2.2. Vitamins
Vitamins are a class of nutrients or organic compounds essential for the body not synthesized by humans, except for vitamins D, K, and biotin (B7), where vitamin D is synthesized in the body by irradiating skin sterols with UV rays, while biotin and vitamin K are present in certain foods but can also be synthesized by the human intestinal flora. Therefore, the essential vitamins must be daily ingested from food to prevent metabolic disorders related to vitamin deficiencies due to their major role in the synthesis of vital cofactors, enzymes, and metabolic reactions based on coenzymes [98,99,100]. Bee pollen is considered a “vitamin bomb” due to the presence of almost all vitamins with an average of 0.02–0.7% of its total content, with a higher amount of water-soluble than fat-soluble vitamins [4,101]. Table 1 summarizes the different vitamins identified in different samples of bee pollen with different floral species and geographical origins.

Table 1.
Summary of the different vitamins isolated from bee pollen.
- water-soluble vitamins
B vitamin group (thiamin (B1), riboflavin (B2), PP vitamin or niacin (B3), pantothenic acid (B5), pyridoxine (B6), biotin (B7), folic acid (B9), and cobalamin (B12)) are the most commonly identified class of water-soluble vitamins in bee pollen (Table 2) [29,75,104,105,107]. Vitamin C or L-ascorbic acid is marginally identified because of its deterioration by thermal pretreatments [28]. Water-soluble vitamins, such as B-complex, are not normally stored in the body in significant amounts, necessitating a daily intake of these vitamins. This group of vitamins plays a key role in host immunity, dermatology, and cellular energy production (B1, B2); they facilitate the production of amino acids and improve their metabolism (B6); and help the body to convert carbohydrates into glucose (B3 or PP). Water-soluble vitamin deficiencies triggered by malnutrition can be the origin of certain metabolic and nervous pathologies [108]. Regarding its high content of water-soluble vitamins, bee pollen could be one of their potential sources.
- Fat-soluble vitamins
Bee pollen contains fat-soluble vitamins such as vitamin D, K, E, and A (β-Carotene) with low and variable amounts depending on the botanical origin and the season of collection [73,109,110].
The family of vitamins E is commonly called tocochromanols (tocopherols and tocotrienols), and pollen contains in particular the group of tocopherols (α tocopherol, β-tocopherol, γ-tocopherol, and δ tocopherol) with a dominance of α and γ-tocopherol [56,111]. Vitamin K2 (Menaquinone-4), and two types of vitamin A (β-carotene and retinol) were also detected by Bayram et al. [102]. Although there are no identification studies of vitamin D listed in the international literature, Campos, Komosinska-Vassev, and Khalifa et al. cited the presence of vitamin D in their review articles [36,110,112]. Fat-soluble vitamins are involved in a multitude of physiological processes such as vision, bone health, immune function, and coagulation [113].
4.2.3. Carotenoids
Carotenoids are a highly diversified group of yellow- to red-colored polyenes responsible for the colors in many plant-derived products and play an important role in human health [94]. In bee pollen, β-carotene is the most frequently identified of this class. It is an antioxidant provitamin with good effects on human health (anti-tumor, anti-leukemic, and beneficial against cardiovascular diseases) [114]. Besides β-carotene, other carotenoids such as lutein and cryptoxanthin, zeaxanthin, β-cryptoxanthin, and α-carotene have been identified [60,115]. The carotenoid content varies according to the botanical origin, harvest period (climate conditions), drying, and storage condition [2,52,107].
4.3. Pollen Probiotics
Throughout history, bee pollen has been considered a complete food with many therapeutic virtues, and for this reason, it has been the subject of numerous and diverse biochemical and microbiological studies. Many studies have been concerned with the anti-microbial activities of bee pollen, but little is known about its microbiome.
P. Percie du Sert has reported that bees raise lactic ferments in the nectar stored inside the hive, and this bacteria-rich nectar will be used during their flight to stick pollen grains on their forelegs, which explains the presence of lactic acid bacteria in freshly harvested pollen [29]. Several studies have demonstrated that honey bees and bumblebees seem to have a simple intestinal bacterial fauna which includes acidophilic bacteria, mainly from the Lactobacillus family such as Lactobacillus kunkeii, Lactobacillus plantarum, Lactobacillus fermentum; lactobacillus kunkeii; Lactobacillus plantarum; Lactobacillus fermentum; Lactococcus slactis; Pediococcus acidi lactici; Pediococcus pentosaceus; Lactobacillus ingluviei; and Weissella cibaria [116,117,118,119]. It is now quite clear that the fresh bee pollen bacterial community comes from the specific bacterial fauna of the bee intestinal gut.
Previous in vitro studies have reported the beneficial effect of the isolated probiotic strains of fresh pollen against pathogenic Gram-positive and Gram-negative bacteria through the high production of bacteriocins, such as organic acids, which can be of major importance in fighting human infectious diseases [116,120,121]. Lactobacillus strains isolated from fresh bee pollen can survive under human digestive tract conditions such as low pH, and bile salts. In the same context, the high hydrophobicity and autoaggregation of fresh bee pollen lactobacillus strains are necessary characteristics for the bacterial adhesion to the host system, and its protection through biofilm formation over the host intestinal tissue, making these bacteria promising candidates for use as novel probiotics in the food and pharmaceutical industries [122]. In a recent Turkish study, another bacterial strain known as “fructophilic lactic acid bacteria (FLAB)” was isolated from fresh bee pollen and bee bread samples. FLAB are lactic acid bacteria (LAB) that prefer fructose over glucose as a carbon source and have been isolated from ecological fructose-rich niches including flowers and fruits, as well as the gastrointestinal tracts (GIT) of several fructose-based diet insects such as honeybees [123]. Several studies have shown the beneficial uses of probiotics in humans against type 2 diabetes, obesity, inflammation, tumors, allergy, metabolic disorders, and infectious diseases [124,125,126,127]. Due to this large bacterial population, bee pollen may have a promising role in the food and pharmaceutical industries.
4.4. Phenolic Profile and In Vitro Antioxidant Potential of Bee Pollen
4.4.1. Volatile Compounds of Bee Pollen
The volatile content of bee pollen is rarely studied; indeed, two recent studies on samples from Lithuania, China, and Spain showed the presence of 42 different volatile compounds, mainly nonanal, dodecane, tridecane, hexane, 6-methyl-5-hepten2-one, methyl butanoic acid, limonene, and styrene [113,128]. These compounds are mainly found in flowers and participate in the attracting behavior of pollinators [129]. In other studies (from Greece and Poland), the results showed the presence of aldehydes, ketones, terpenoids, and minor amounts of furfural [115,130]. Bee pollen’s aromatic profile is related to the botanical and geographical origins as well as climatic conditions and bee species [28,113].
4.4.2. Phenolic Profile of Bee Pollen
Bioactive compounds of bee pollen constitute an important quality criterion. Bee pollen as a natural product has gained great scientific interest due to its beneficial properties [131]. Interestingly, bioactive compounds may counteract the installation and/or development of different pathologies [132]. Therefore, the determination of the phenolic profile of bee pollen is considered the first step toward the standardization and prediction of the usefulness of this beehive product. The analysis of their composition revealed that the polyphenolic content presented an average of 3% to 5% of their composition, depending on the botanical origin of the bee pollen [133].
Phenolic acids represent an average of 0.19% of bee pollen, and their properties are mainly related to their structure [132]. Phenolic acids could be divided into benzoic acids, phenylacetic acid, and cinnamic acids, which are of the greatest interest and exert a good antioxidant activity as compared to the other phenolic acids groups. The main molecules of phenolic acids are chlorogenic acid, gallic acid, cinnamic acid, ferulic acid [103], hydroxycinnamic acid, and coumaric acid [134]. Flavonoids, on the other hand, are the most important group of polyphenols found in bee pollen with an average of 0.25% and 1.4% of its total composition; they are an excellent indicator of bee pollen quality [134]. Flavonoids are mostly found in bee pollen as glycosides (flavonoids associated with sugar units), with flavonol glycosides being the most abundant. However, the presence of glycoside bonds decreases the antioxidant activity of flavonols because of the steric effects, the reason why the content of bee pollen in free flavonoids is a good quality criterion [135,136]. The main flavonols identified in bee pollen are quercetin, kaempferol, and rutin. The main flavones are represented by apigenin, chrysin, and luteolin, and flavanones are represented by naringenin and pinocembrin, while genistein is the major isoflavone identified in bee pollen [132]. Additionally, resveratrol, the most important stilbene has been isolated by Ares et al. [137]. Table 2 summarizes the different isolated phenolic compounds with the different extraction techniques.

Table 2.
Summary of the different methods used to determine the different active molecules of pollen.
Table 2.
Summary of the different methods used to determine the different active molecules of pollen.
| Country | Floral Origin | Techniques | Phenolic Compounds Name | References |
|---|---|---|---|---|
| Turkey | ND | HPLC-PDA detector | gallic acid, 3,4-hydroxybenzoic acid, (+)-catechin1,2-dihydroxy-benzene, syringic acid, caffeic acid, rutin trihydrate, p-coumaric acid, trans-ferulic acid, apigenin 7 glucoside, resveratrol, quercetin, trans-cinnamic acid, naringenin, kaempferol, isorhamnetin | [131] |
| Turkey | Castanea spp. | HPLC-DAD | rosmarinic acid, vitexin, hyperoside, pinocembrin, trans-chalcone, apigenin, protocatechuic acid, galangin | [133] |
| Portugal | Monofloral: Rubus spp., Cystisus spp., Quercus spp., Prunus spp., Leontondon spp., Cistus spp., and Trifolium spp. Heterofloral: Castanea sativa and Echium spp. and ii) Erica spp., and Eucalyptus spp. | UHPLC-DAD-ESI-MS | coumaroyl quinic acid, myricetin-O-rutinoside, luteolin-O-dihexoside, quercetin-O-dihexoside, myricetin-O-hexoside, myricetin-O-(malonyl)rutinoside, isorhamnetin-O-dihexoside, quercetin-O-hexosyl-pentoside, quercetin-O-rutinoside isomer 1, quercetin-O-rutinoside isomer 2, luteolin-di-O-hexosyl-rhamosíde, quercetin-O-(malonyl)rutinoside, isorhamnetin-O-rutinoside, hydroxybenzoyl myricetin, quercetin-O-(malonyl)hexoside, quercetin derivative, quercetin-O-rhamnoside, isorhamnetin-O-(malonyl)hexoside isomer 1, luteolin-O-(malonyl)hexoside, myricetin, isorhamnetin-O-(malonyl)hexoside isomer 2, myricetin-O-dihydroferuloyl protocatechuic acid, myricetin-O-acetyl hydroxybenzoyl protocatechuic acid-isomer 1, myricetin-O-acetyl hydroxybenzoyl protocatechuic acid isomer 2, quercetin-O-acetyl hydroxybenzoyl protocatechuic acid isomer 1, myricetin-O-acetyl hydroxybenzoyl hydroxybenzoic acid isomer 2, quercetin-O-acetyl hydroxybenzoyl hydroxybenzoic acid isomer 1, quercetin-O-acetyl hydroxybenzoyl hydroxybenzoic acid isomer 2, O-dihydroxy benzoyl acetyl malonyl coumaric acid flavonoid derivative | [103] |
| China | Rosa rugosa | UPLC-ESI-QTOF-MS/MS | isorhamnetin 3-O-diglucoside, sorhamnetin-3-O-coumaroyl glucoside, isorhamnetin-3-O-6-O-acetyl-β-D-glucopyranosy, kaempferol-3-O-neohesperidoside, N′,N″,N‴-Tricaffeoyl spermidine, N′,N″,N‴-Dicaffeoyl p-coumaroyl spermidine, N′,N″,N‴-Di-p-coumaroyl caffeoyl spermidine, N′,N″,N‴-Tri-p-coumaroyl spermidine | [134] |
| Chile | Brassica rapa and Eschscholzia californica | HAPLC-DAD | syringic acid, coumaric acid, sinapic acid, ferulic acid, cinnamic acid, abscisic acid, catechin, myricetin, quercetin, apigenin, kaempferol, naringenin, rhamnetin | [135] |
| Brazil | Eucalyptus marginata; Corymbia calophylla | HPLC | gallic acid, 4-hydroxyphenylacetic acid, rutin, resveratrol, myricetin, quercetin-3-O-glucopyranoside, kaempferol-3-O-glucoside, kaempferol-3-O-rutinoside, naringenin, quercetin, phloretine, kaempferol | [136] |
| Morocco | Coriandrum sativum | HPLC/DAD/ESI-MSn | myricetin-3-O-rutinoside, quercetin-diglucoside, quercetin-3-O-rutinoside, kaempferol-3-O-rutinoside, isorhamnetin-3-O-rutinoside, isorhamnetin-O-pentosylhexoside, kaempferol-diglucuronide, isorhamnetin-3-O-glucoside, quercetin-3-O-rhamnoside, ellagic acid, N1-p-coumaroyl-N5, N10-dicaffeoylspermidinea, N1, N10-di-p-coumaroyl-N5-caffeoylspermidine, luteolin, quercetin-3-methyl-ether, N1, N5-di-p-coumaroyl-N10-caffeoylspermidine, N1, N5, N10-tri-pcoumaroylspermidine, N1, N5, N10-tri-pcoumaroylspermidine, N1, N5, N10-tri-pcoumaroylspermidine, N1, N5, N10-tri-pcoumaroylspermidine | [138] |
| Romania | Hedera, Helianthus, Cistus, Cornus, Brassica, Gledistia, Hedysarum, Trifolium, Castanea, lamium, Magnolia, Fraxinus, Papaver, Crataegus, Prunus, Rubus, and Cordiandrum | HPLC-DAD | gallic acid, protocatechuic acid, p-hydroxybenzoic acid, vanillic acid, caffeic acid, chlorogenic acid, p-coumaric acid, rosmarinic acid, myricetin, luteolin, quercetin, kaempferol | [139] |
| Italy | Cistus ladanifer, Echium, Achillea, Quercus ilex, Rubus, Pinaceae, Filipendula, Trifolium incarnatum, Trifolium pratense, Trifolium repens, Prunus, Pyrus, Malus, and Oxalis | UHPLC-ESI-QTOF-MS | cyanidin 3-O-xyloside/arabinoside, delphinidin 3-O-(60 ’-p-coumaroyl-glucoside), petunidin 3-O-arabinoside, pelargonidin 3-O-glucoside, delphinidin 3-O-glucoside, delphinidin 3-O-glucosyl-glucoside, delphinidin 3-O-rutinoside, cyanidin 3-O-sophoroside, naringin 60-malonate, naringin 40-O-glucoside, naringenin 7-O-glucoside, apigenin 7-O-(60′-malonyl-apiosyl-glucoside), tetramethylscutellarein, luteolin 7-O-glucuronide, apigenin 6-C-glucoside, kaempferol 3-O-glucuronide, quercetin 3-O-rutinoside, kamepferol 3,7-O-diglucoside, quercetin 3-O-galactoside 7-O-rhamnoside, quercetin 3-O-rhamnosyl-galactoside, kaempferol 3-O-sophoroside, 3,7-Dimethylquercetin, dihydroquercetin, formononetin, genistin, gallic acid ethyl ester, syringic acid, caffeic acid 4-O-glucoside, caffeoyl glucose, feruloyl glucose, caffeic acid, sesamol, hydroxytyrosol 4-O-glucoside, curcumin, and carnosic acid | [140] |
| Colombia | Cistus ladanifer; Echium Achillea; Taraxacum; Carduus; Cirsium; Vicia; Quercus ilex; Rubus; Pinaceae; Filipendula; Trifolium incarnatum, Trifolium pratense; Trifolium repens; Prunus, Pyrus; Malus and Oxalis | UHPLC-DAD | Caffeic acid, ferulic acid, S-N1,5,10-tri-ferulic acid isorhamnetin, kaempferol, luteolin, myricetin, p-coumaric acid, SP-N1,5,10,14-tetra-p-coumaric acid, pinobanskin, quercetin, spermidine, spermine, 4-methyl gallic acid, apigenin, amentoflavone, N1-caffeoyl-N5,10-di-p-coumaroyl-spermidine, and N1,10-di-pcoumaroyl-N5-caffeoyl-spermidine. | [44] |
ND: Not determined.
4.4.3. In Vitro Antioxidant Activity of Bee Pollen
Oxidative stress is involved in the development of several pathologies such as diabetes, Alzheimer’s, cancer, atherosclerotic, and other disorders [141]. The use of natural products such as beehive products including honey, pollen, royal jelly, and propolis as a source of antioxidant molecules has been supported and suggested to protect human cells from the effects of oxidative stress by numerous scientific studies [142]. Moreover, the antioxidant activities of bee pollen have been evaluated in several works using well-known techniques such as DPPH, ABTS, β- carotene, FRAP, CUPRAC, NO, and TAC assays [132,138,140,143,144,145,146,147,148,149,150,151,152,153,154,155,156,157,158,159,160,161,162,163,164,165,166,167,168,169,170]. Table 3 summarizes all studies that have evaluated the antioxidant activity of bee pollen. It presents the pollen origin, the type of extract, the methods used, and the main results. Indeed, El Ghouizi et al. [138] evaluated in vitro the antioxidant activity of the aqueous extract of Moroccan fresh bee pollen and revealed an important scavenging capacity against DPPH and FRAP with IC50 values of 0.39 ± 0.13 mg/mL and 0.54 ± 0.53 mg/mL, respectively.

Table 3.
Summary of the different studies of the antioxidant activity of bee pollen.
In Brazil, several authors have investigated bee pollen samples from several botanical and geographical origins for their antioxidant proprieties using four antioxidant assays (DPPH, ORAC, β-carotene, and FRAP) and the results revealed that the bee pollen extracts exhibited important antioxidant activity in all tests with a significant difference among them, and a potential correlation between this activity and polyphenolic composition, which in turn varied depending on the geographical and botanical origins of the plant visited by the bees [145,150,151,168,171].
In Egypt, authors have investigated the antioxidant activity of ethanol, methanol, petroleum ether, dichloromethane, and ethyl acetate extracts of bee pollen samples from monofloral sources (Zea mays and Trifolium alexandrinum L) by two in vitro methods (ABTS and DPPH). The results showed an interesting anti-DPPH activity of ethanol extract with a percentage activity of 90%, while the methanol extract revealed a strong activity using the ABTS test with a percentage activity of 76.51%. Based on the findings, the high antioxidant activity of ethanol extract could be related to its major compounds including catechin, quercetin, and caffeic and gallic acid [144,162]. These results are confirmed by studies on Chinese bee pollen. In those works, authors investigated the antioxidant capacity of aqueous, ethanol, and methanol extracts using DPPH, ABTS, superoxide-scavenging activity, and reducing power and showed that ethanol extract of bee pollen has a good anti-DPPH effect with IC50 = 1.28 ± 0.03 mg/mL, and an important anti-ABTS (1.06 ± 0.02 mmol TE g−1) and reduction power activity (70.55%). The methanol extract showed also an antioxidant effect with an anti-DPPH IC50 value of 1.72 mg/mL, and IC50 = 3.48 mg/mL for superoxide-scavenging activity [157,161,169]. Methanol extracts of twenty-two bee pollen samples from different floral origins in Portugal also showed an interesting antioxidant effect, which manifests in scavenger activity of the free DPPH• and β-carotene in bleaching assays with mean values of 3.0 ± 0.7 mg/mL and 4.6 mg/mL, respectively [155].
On the other hand, several authors have reported the antioxidant activity of Turkish bee pollen extracts based on different geographical origins [160,167], bee races (Apis mellifera caucasica, Apis mellifera anatoliaca, Apis mellifera syriaca, and Apis mellifera carnica) [166], and extraction methods as well as the storage conditions [152]. The authors showed important antiradical activity of bee pollen which is highly affected by chemical composition, plant origins, geographical origin, and storage conditions.
Other research works reported a variable antioxidant effect of different bee pollen extracts including those from Algeria, Greece, Italy, Korea, Spain, Slovakia, Malaysia, Serbia, Thailand, and Poland, and this variability can be significantly related to a variety of botanical and geographical origins [132,140,143,147,148,153,154,156,158,159,163,164,165,170].
5. Therapeutic Properties of Bee Pollen against Oxidative Stress-Related Diseases
The use of bee pollen in traditional medicine dates to ancient times and is attested by books of Arab and Jewish doctors such as Ibn al-Beithar and Maimonides in the early 1100s, where they described bee pollen as an aphrodisiac, sedative, and effective for the stomach, the heart, and intestines [172,173].
5.1. Antioxidative Properties
Oxidative stress is caused by an imbalance between free radical production and antioxidant defense systems, resulting in an accumulation of reactive oxygen species (ROS). In turn, ROS interact with various cytoplasmic components such as proteins, membrane lipids, and DNA [174]. As a result, ROS induce serious cell damage and participate in the development of many chronic illnesses such as diabetes and associated complications, arthritis, Parkinson, and Alzheimer’s [175]. Bee pollen is one of the natural antioxidant-rich products mainly used against oxidative stress and related pathologies. In this context, Kawther and coworkers have proved that the administration of bee pollen extract (250 mg/kg b.w) attenuated oxidative stress induced by protein [176]. It has been demonstrated that the anti-oxidative activity of bee pollen is attributed to its content of secondary metabolites including, vitamin E, vitamin C, carotenoids, and phenolic compounds [149]. Thanks to its phenolic hydroxyl group, the flavonoids present in bee pollen can scavenge ROS and free radicals, and inactivate electrophiles [164]. As reported in Table 4, several studies have reported the antioxidative properties of various phenolic compounds and their involved mechanisms. Pari et al. reported that the sub-chronic administration of Caffeic acid (6 mg/kg b.w) improved the oxidative stress caused by alcohol-induced toxicity in rats by increasing non-enzymic antioxidant defense systems, and by preventing lipid peroxidation [177]. Likewise, cinnamic acid occurs in the antioxidative process by modulating lipid metabolism and boosting GSH, SOD, and CAT enzyme activities as well as scavenging and decreasing ROS production [178]. In addition, the oral administration of rutin at a dose of 50 and 100 mg/kg/b.w for 20 days enhances the production of antioxidant enzymes, decreases serum toxicity markers, and downregulates COX, 2p38-, MAPK, i-NOS, and the NF-κB signaling pathway [179]. Furthermore, a recent study showed that quercetin weakened oxidative stress and decreased the expression of TNFα, IL-1β, and IL-6 [180]. Similarly, treatment with luteolin minimized the oxidative stress through multiple mechanisms: (a) up-regulation of the Nrf-2 pathway, (b) enhancement of HO-1 expression, (c) increase in GSH, SOD, and GPX activities, and (d) decrease in MDA levels [181]. Pinocembrin decreased oxidative stress, apoptotic, and inflammatory markers [182] Figure 1.
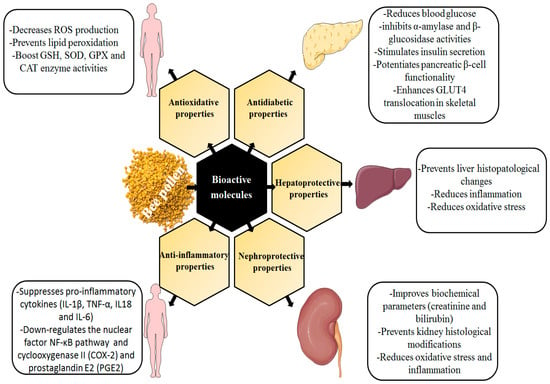
Figure 1.
Schematic presentation of the therapeutic properties of bee pollen.
5.2. Antidiabetic and Anti-Hyperglycemic Properties
Diabetes mellitus is an endocrine disorder characterized either by insufficient insulin secretion and/or its defective utilization [183]. The antidiabetic/anti-hyperglycemic activity of bee pollen has been previously studied. According to Nema et al., bee pollen administration at 100 mg/kg body weight/day for 4 weeks lowered blood glucose and prevented pituitary–testicular axis dysfunction [6]. Furthermore, bee pollen exhibited a potent anti-hyperglycemic activity in patients with insulin-independent diabetes mellitus (T2DM) [184]. Moreover, thanks to its bioactive constituents, bee pollen exerts its anti-hyperglycemic effect by modulating glucose uptake and inhibiting α-amylase and β-glucosidase activities, leading to the management of diabetes and its serious complications [185]. The anti-diabetic properties of many individual phenolic compounds present in bee pollen have already been investigated. Adisakwattana and coworkers enunciated that cinnamic acid administered orally at a dose of 50 mg/kg/day for 5 weeks stimulates insulin and adiponectin secretions, increases hepatic glycolysis, improves glucose uptake, potentiates pancreatic β-cell functionality, and decreases protein glycation [186]. Rutin regulates glycemia and ensures its anti-diabetic effect through the inhibition of the polyol signaling pathway as well as via the modulation of lipid metabolism and the prevention of lipid peroxidation [187]. Apigenin facilitates and enhances GLUT4 translocation in skeletal muscles either by up-regulating the AMP-activated protein kinase pathway or by activating the insulin signaling pathway, which leads to glucose uptake and thus hypoglycemia. A paper published by Alkhalidy et al. explored the anti-diabetic effect of kaempferol against streptozotocin-induced diabetes in rats and found that chronic administration of kaempferol (50 mg/kg-b.w) reduced hepatic glucose production, increased hexokinase activity, decreased hepatic pyruvate carboxylase activity, and inhibited the gluconeogenesis pathway [188], as shown in Figure 1.
5.3. Hepatoprotective Properties
Bee pollen extract has been found to possess a potent hepato-protective effect. Cheng and coworkers reported that bee pollen extract administration increased tissue catalase SOD and GSH-Px activity, and prevents liver histological changes induced by carbon tetrachloride treatment in mice. This suggests the potential role of bee pollen in preventing hepatocellular changes associated with exposure to xenobiotics. The hepatoprotective capacity of bee pollen is largely attributed to its rich content of natural antioxidants such as phenols and flavonoids [189]. The hepatoprotective ability of phenolic compounds has been explored in numerous studies (Table 3). Malayeri et al. showed that the co-administration of a single dose of naringenin (50 mg/kg/b.w) boosted enzymatic and non-enzymatic antioxidant activities, and reduced NO, TNF-α, and IL-6 levels [190]. Yang and coworkers indicated that ferulic acid exhibits its protective role against CCL4-caused acute oxidative liver damage in rats via the up-regulation of p-JNK, p-p38 MAPK, and Bcl-2 signaling pathways and thus, decreased the expression of pro-inflammatory mediators of hepatic-toxicity TNF-α and IL-1β [191]. Owumi et al. recently emphasized that protocatechuic acid protects against methotrexate-induced liver dysfunction via enhancing enzyme antioxidant defense mechanisms and decreasing oxidative stress and free radical production, which was confirmed by biochemical analysis and histopathological investigations [192]. Vanderson et al. demonstrated that caffeic acid treatment improves oxidative stress and kidney dysfunction mediated by ethanol in a rat model. This was attributed to the down-regulation of CYP2E1 and the protection of DNA against oxidative damage [193]. Ebrahimi and coworkers proved that ellagic acid treatment reduced oxidative damage and liver ultrastructure changes in methotrexate-induced mitochondrial dysfunction and liver toxicity in rats [194] (see Figure 1).
5.4. Nephroprotective Properties
Owing to its rich content of various bioactive molecules, bee pollen has been found to have a potent nephroprotective activity. In a rat model, it was reported that bee pollen extract improved biochemical parameters (creatinine and bilirubin), increased the antioxidant defense system (SOD, CAT, and GSH), lowered oxidative stress biomarkers (MDA and iNOS), and prevented kidney histological effects induced by cisplatin. Intraperitoneal administration of apigenin reduced COXI, COXII, and MDA levels, and increased kidney GSH levels [195]. Recently, Owumi et al. reported that protocatechuic acid exhibits its reno-protective activity by increasing the activity of antioxidant enzymes (SOD, CAT, GSH, and GPX) and decreasing the renal (RNOS and LPO) levels, as well as reducing the inflammation biomarkers NO, TNF-α, and IL-1β levels in renal tissue [192]. Another study found that naringenin (100 mg/kg/b.w) reduced oxidative stress and prevented lipid peroxidation in rats after cyclosporine treatment [196]. According to Chowdhury and colleagues, ferulic acid prevented hyperglycemia-induced kidney damage and oxidative stress in rats; this was related to the modification of AGEs, MAPKs (p38 and JNK), and the NF-B signaling pathways by this acid [197]. Mohammed and coworkers evidenced that ellagic acid stimulated the expression of SIRT1, decreased P53 protein levels, reduced ROS production, and enhanced enzymatic and non-enzymatic antioxidant systems [198]. This was demonstrated through improved biochemical values (creatinine, urea, and uric acid), kidney histopathological tissue, and renal biomarker stress (MDA, GSH, CAT). Another study showed that pinocembrin treatment mitigates gentamicin-induced inflammation and renal toxicity via the modulation of Nrf2/HO-1 and NQO1 pathways [199]. This could suggest its potent ability to reduce oxidative stress and inflammation-related nephrocellular dysfunctions. Shanmugam et al. reported that oral administration of Kaempferol (100 mg/kg/day/b.w) exerts its nephroprotective action by inhibiting RhoA/Rho kinase-mediated inflammatory pathway [200] Figure 1.
5.5. Anti-Inflammatory Properties
Numerous scientific studies have indicated that bee pollen has a potent anti-inflammatory impact. Indeed, it has been shown that flavonoids and phenolic acids play a major role in the anti-inflammatory activity of bee pollen extracts. As indicated in Table 3, several individual phenolic components have shown anti-inflammatory effects through different signaling pathways. Indeed, phenolic acids including caffeic acid, ferulic acid, and cinnamic acid are documented as potent inhibitors of tumor necrosis factor (TNF) and thus down-regulation of the nuclear factor NF-κB pathway (proinflammatory signaling pathway) [201,202,203]. In addition, ellagic acid has been found to inhibit nitric oxide (NO), TNF-α, and IL-6, and induce the down-regulation of cyclooxygenase II (COX-2) and prostaglandin E2 (PGE2) [204]. The anti-inflammatory activity of bee pollen is also linked to Galangin, chrysin, quercetin, resveratrol, kaempferol, and other flavonoid molecules. Choi and coworkers have shown that galangin inhibits the expression of iNOS, COX-2, and the release of pro-inflammatory cytokines such as IL-1β and TNF-α [205]. In the same context, the findings of Li, Zhipeng, et al. showed that chrysin improves inflammatory reaction through the inhibition of NO, prostaglandin E2, and the NF-κB signaling pathway [206]. Quercetin and resveratrol exhibit their anti-inflammatory actvity via the down-regulation of the NF-κB pathway and the inhibition of COX-1 and COX-2 activities [207,208]. Park and coworkers reported that Kaempferol ensures its protective effect in aged kidney tissues via the suppression of pro-inflammatory cytokines (IL-1β, TNF-α, IL-18, and IL-6) [209]. These data revealed that kaempferol could have the ability to attenuate age-related chronic inflammatory reactions Figure 1.

Table 4.
Summary of the different pharmacological properties of different phenolic compounds found in bee pollen.
Table 4.
Summary of the different pharmacological properties of different phenolic compounds found in bee pollen.
| Molecules | Dosage, Route, and Exposure Duration | Pharmacological Properties | Involved Mechanisms | References |
|---|---|---|---|---|
| Caffeic acid | 6 mg/kg/day, orally for 45 days. | Anti-oxidative properties | ↑Non-enzymic antioxidants, ↓lipid peroxidation, and ↓TBARS level. | [177] |
| Cinnamic acid | 20 mg/kg/day, i.p for 40 days. | ↓lipid peroxidation, ↓ROS production ↑GSH, ↑SOD, and ↑CAT levels. | [178] | |
| Ferulic acid | 25 mg/kg/day, orally for 10 days. | ↓lipid peroxidation, ↓ROS levels, and ↓N-acetyl-β-glucosminidase activity. | [210] | |
| Ellagic acid | 10 and 30 mg/kg/day for 30 days. | Enhances the concentration of enzymatic antioxidant levels (SOD, CAT, and GPx), and ↓ MDA, ↓TNF-α, and ↓IL-1β. | [211] | |
| Quercetin | 50 mg/kg/day, i.p for 21 days. | Increases GSH level, SOD, GR, G, P, and CAT activity, and decreases the expression of TNFα, IL-1β, and IL-6. | [180] | |
| Kaempferol | 100 mg/ kg/day, i.p for 6 weeks. | Inhibits the activity of ASK1/MAPK signaling pathways (JNK1/2 and p38). | [212] | |
| Galangin | 8 mg/kg/day, i.p for 45 days. | ↓lipid peroxidation, ↑enzymatic and non-enzymatic antioxidants. | [213] | |
| Chrysin | 30 mg/kg/day, orally, for 14 days. | ↑GSH, ↓TBARS, ↓XO, and ↓NADPH levels | [214] | |
| Protocatechuic acid | 100 mg/kg/day, i.p for 7 days. | Prevents lipid peroxidation and the formation of NO, and enhances antioxidant enzymes. | [215] | |
| Apigenin | 0.625, 1.25, and 2.5 mg/mL. | ↓oxidative stress, GSH level ↑SOD activity, ↓IL-6, and ↓NF-κB levels. | [216] | |
| Luteolin | 100 and 200 mg/kg/ day, orally for 28 days. | ↓MDA, ↑GSH, ↑SOD ↑GPX ↑Nrf2, and ↑HO-1 Expressions. | [181] | |
| Rutin | 50 and 100 mg/kg/day, orally for 20 days. | ↑Production of antioxidant enzymes, ↓serum toxicity markers, and downregulation of (COX, 2p38-, MAPK, i-NOS, and NF-κB). | [179] | |
| Naringenin | 50 mg/kg/day, orally for 8 weeks. | Minimizes oxidative stress and enhances CAT, SOD GSH, and GPx levels. | [217] | |
| Pinocembrin | 10 mg/kg/day, orally for 7 days. | Decreases oxidative stress, and apoptotic and inflammatory markers. | [182] | |
| Caffeic acid | 100 mg/kg/day, orally for 4 weeks. | Antidiabetic and anti-hyperglycemic properties | IL-6, ↓ IL-1β, ↓ TNF-α, ↓ MCP-1, ↓HbA1c, ↓ UGA, ↓ sorbitol, ↓ fructose, and ↑AMPKα2. | [218] |
| Ferulic acid | 10 mg/kg/day, orally for 15 days. | Down-regulation of NF- κB pathway. | [219] | |
| Cinnamic acid | 50 mg/kg/day, orally for 5 weeks. | ↑insulin secretion, ↑hepatic glycolysis, ↑adiponectin secretion ↑glucose uptake, ↑pancreatic β-cell functionality, and ↓protein glycation. | [186] | |
| Ellagic acid | 250 mg/kg/day, orally for 28 days. | ↑ insulin secretion, ↑β-cell number, ↑plasma total antioxidants, and ↑glucose intolerance. | [220] | |
| Quercetin | 10 and 30 mg/kg/day, i.p for 14 days. | ↑GLUTs, ↑IR-P, ↑GLUT4, ↑Glucose uptake, ↑pancreatic cell-β generation, ↑glucokinase activity, ↓α-glucosidase activity. | [221] | |
| Kaempferol | 50 mg/kg/day, orally for 12 weeks. 200 mg/kg/day, orally for 14 days. | ↓hepatic glucose production, ↑hexokinase activity, ↓hepatic pyruvate carboxylase activity, and gluconeogenesis. ↑GLP-1 and insulin release, ↑ cAMP, and Ca2+ intracellular levels. | [188,222] | |
| Galangin | 4, 8, and 16 mg/kg/day, orally for 45 days. | Inhibition of DPP-4, ↓oxidative stress, and ↑antioxidant status. | [213] | |
| Chrysin | 40 mg/kg/day, orally for 16 weeks. | Inhibition of the TNF-α pathway, ↓secretion of pro-inflammatory cytokines, and ↓glucose and lipid peroxidation levels. | [223] | |
| Protocatechuic acid | 50 and 100 mg/kg/day, orally for 7 days. | ↑insulin sensitivity, ↓insulin resistance, ↓gluconeogenesis, and ↑glucose uptake. | [224] | |
| Apigenin | 1.5 mg/kg/day, i.p for 28 days. | Enhances GLUT4 translocation. | [225] | |
| Luteolin | 10 mg/kg/day, orally for 24 weeks. | Reduces oxidative stress and inhibits the STAT3 pathway. | [226] | |
| Rutin | 90 mg/kg/day, orally for 10 weeks. | Inhibition of polyol pathway, oxidative stress, and lipid peroxidation. | [187] | |
| Naringenin | 50 and 100 mg/kg/day, orally for 6 weeks. | Improvement of glucose and lipid metabolism, and ↓insulin resistance. | [227] | |
| Pinocembrin | 50 mg/ kg/day, orally for 10 days. | ↓ NF-κB and TNF-α levels. | [228] | |
| Resveratrol | 12 mg/kg/day, orally for 1 5 days. | Down-regulation of NF- κB pathway. | [219] | |
| Caffeic acid | 100 mg/kg/day, orally for 4 days. | Hepato-protective properties | Downregulation of CYP2E1 and the protection of DNA against oxidative damage. | [193] |
| Cinnamic acid | 20 mg/kg/day, orally for 10 days. | ↓NF-kB and ↓iNOS activities. | [229] | |
| Ellagic acid | 5 and 10 mg/kg/day, orally for 10 days | Up-Regulation of Nrf2 and HO-1 expression and inhibition of NF-κB signaling pathway. | [194] | |
| Quercetin | 20, 40, and 80 mg/kg/day, orally for 7 days. | Modulation of the expression of nuclear orphan receptors (CAR, PXR) and cytochrome P450 enzymes (CYP1A2, CYP2E1, CYP2D22, CYP3A11). | [230] | |
| Kaempferol | 20 mg/kg, twice a day, orally for 28 days. | ↓CYP2E1 activity and ↓ROS production. | [231] | |
| Galangin | 15, 3,0, and 60 mg/kg/day, orally for 15 days. | Activation of Nrf2 and HO-1 signaling pathway. | [232] | |
| Chrysin | 25 or 50 mg/kg, orally for 6 days. | Decreases the expression of COX-2, iNOS. | [233] | |
| Ferulic acid | 25, 50, and 100 mg/kg/day, orally for 7 days. | ↓ the expression TNF-α and IL-1β, upregulation of p-JNK, p-p38 MAPK, and Bcl-2. | [191] | |
| Protocatechuic acid | 25 and 50 mg/kg/day, i.p for 7 days. | ↓ oxidant species ↑antioxidant enzymes | [192] | |
| Apigenin | 10 mg/kg/day, orally for 3 weeks. | Enhances antioxidant defense mechanisms and decreases lipid peroxidation. | [234] | |
| Luteolin | 100 mg/kg/day, i.p for 7 days. | Modulation of Nrf2/HO-1 pathway and ↓oxidative stress. | [235] | |
| Rutin | 20 mg/kg/day, orally for 15 days. | ↑Antioxidant profile and regulation of Na+/K+ ATPase activity. | [236] | |
| Naringenin | 50 mg/kg/day, orally for 10 days. | ↑the enzymatic and non-enzymatic antioxidant levels, ↓NO, TNF-α, and IL-6 levels. | [190] | |
| Pinocembrin | 50 and 75 mg/kg/day, i.p for 10 days. | Modulation of Nrf2/HO-1 and NQO1 pathways. | [199] | |
| Resveratrol | 50 and 100 mg/kg/day, orally for 28 days | Modulation of SIRT1 and p53 pathways. | [237] | |
| Caffeic acid | 100 mg/kg/day, orally for 14 days. | Nephroprotective properties | Enhances the antioxidant defense system and reduces lipid peroxidation. | [238] |
| Ferulic acid | 50 mg/kg/day, orally for 8 weeks. | Modulation of AGEs, MAPKs (p38 and JNK), and NF-κB pathways, and ↓oxidative stress. | [197] | |
| Cinnamic acid | 50 mg/kg /day, orally for 7 days. | antioxidant expression GSH levels, SOD, CAT, and GPx activities. | [239] | |
| Ellagic acid | 10 mg/kg/day, orally for 30 days. | Stimulates the expression of SIRT1, ↓P53 protein level, ↓ROS production, and ↑enzymatic and non-enzymatic antioxidant system. | [198] | |
| Quercetin | 10 mg/kg/day, i.p for 10 weeks. | ↑antioxidant expression and ↓lipid peroxidation. | [240] | |
| Kaempferol | 100 mg/kg/day, orally for 28 days. | Inhibits RhoA/Rho Kinase mediated inflammatory pathway. | [241] | |
| Chrysin | 30 and 100 mg/kg, ip for 26 days. | ↑iNOS and PKC Levels, and ↓AGEs and RAGE. | [242] | |
| Protocatechuic acid | 25 and 50 mg/kg/day, i.p for 7 days. | ↓ oxidant species ↑antioxidant enzymes. | [192] | |
| Apigenin | 3 mg/kg/day, i.p for 7 days. | Reduces COXI and COXII, MDA levels and increases GSH level. | [195] | |
| Luteolin | 10 and 20 mg/kg/day, orally for 4 weeks. | Inhibition of RIP140/NF-κB pathway. | [243] | |
| Rutin | 100 mg/kg/day, orally for 14 days. | Suppresses NF-κB activation and TGF-β1/Smad3 signaling. | [244] | |
| Naringenin | 100 mg/kg/day, orally for 45 days. | ↓ oxidative stress and lipid peroxidation levels. | [196] | |
| Pinocembrin | 50 and 75 mg/kg/day, i.p for 10 days. | Modulation of Nrf2/HO-1 and NQO1 pathways. | [199] | |
| Resveratrol | 20 mg/kg/day, orally for 40 weeks. | Modulation of the NF-κB signaling pathway. | [245] | |
| Caffeic acid | 50 mg/kg/day, orally for 21 days. | Anti-inflammatory properties | inhibition of NO, prostaglandin E2, and NF-κB signaling pathways. | [201] |
| Ferulic acid | 100 mg/kg/day, orally for 6 weeks. | Inhibition of NADPH oxidase and NF-κB pathway. | [202] | |
| Cinnamic acid | 60 mg/kg/day, orally for 21 days. | Down-regulation of the NLRP3, NF- κB, and ASK1/MAPK signaling pathways. | [203] | |
| Ellagic acid | 1, 3, 10, and 30 mg/kg, i.p for 5 h. | Suppression of NF-κB pathway and NO, TNF-α, IL-6, COX-2 activity, and PGE2. | [246] | |
| Quercetin | 1 mg/kg/day, orally for 15 days. | Down-regulation of the NF-κB pathway. | [208] | |
| Kaempferol | 2 and 4 mg/kg/day for 10 days. | Decreases the synthesis of IL-1β, TNF-α, IL-18, and IL-6. | [209] | |
| Galangin | 50 mg/kg per day, orally for 4 days. | Inhibits the expression of iNOS, COX-2, and pro-inflammatory cytokines. | [205] | |
| Chrysin | 40 mg/kg/day, orally for 16 weeks. | inhibition of NO, prostaglandin E2, and NF-κB signaling pathways. | [206] | |
| Protocatechuic acid | 20 mg/kg/day, orally for 8 weeks. | ↓IL-1β, ↓IL-6, and ↓TNF-α synthesis pathways. | [247] | |
| Apigenin | 20 and 40 mg/kg/day, orally for 28 days. | ↓TNF-α and IL-6 production. | [248] | |
| Luteolin | 100 mg/kg, i.p for 6 h. | ↑HO-1 expression, ↑IL-10, ↓TNF-α, and ↓IL-6 levels. | [249] | |
| Rutin | 30 mg/kg/day, orally for 14 days. | Inhibition of p38-MAPK pathway. | [250] | |
| Naringenin | 5, 10, and 20 mg/kg/day, for 16 days. | Up-regulation of Nrf-2/HO-1pathway and ↓NF-kB mRNA expression. | [251] | |
| Pinocembrin | 50 mg/kg/day, i.p for 24 days. | Down-regulation of NF-kB pathway. | [252] | |
| Resveratrol | 10 or 50 mg/kg/day, orally for 28 days. | Inhibition of COX-1 and COX-2 activities | [207] |
↑ represent increases; ↓ represent decreases.
5.6. Other Beneficial Effects of Bee Pollen
Bee pollen has a broad spectrum of pharmacological effects and provides a promising area for researchers interested in the therapeutic effects of natural products, particularly hive products. The rich composition of probiotics, proteins, macro-, and micronutrients in bee pollen has been related to its positive effect on morphological development (thickness of epithelium) and functioning (absorption) of the small intestine, leading to the proper functioning of the gastrointestinal tract [217,253].
Bee pollen is also known to modulate the secretory activity (release of IGF-I growth factor and progesterone and estradiol steroid hormones) and apoptotic activity of the ovary in rats. In postmenopausal women with breast cancer, bee pollen can improve menopausal-related symptoms when used in association with antihormonal treatment; it is also beneficial for women who suffer from post-menopausal disorders [254].
Studies have demonstrated the positive effects of the phenolic and probiotic content of bee pollen on preventing metabolic syndrome by reducing body and liver weight gain, decreasing fasting blood glucose, and lipid accumulation in serum and liver, which can be explained through the regulation of intestinal microbiota [255].
In many cultures, bee pollen has long been used by women to maintain their beauty and whiten their skin. Since more than 70% of bee pollen composition is active, (proteins, carbohydrates, lipids/fatty acids, phenolic compounds, and vitamins), the cosmeceutical properties of bee pollen in the laboratory have been studied and researchers have demonstrated that pollen can boost protective mechanisms against skin aging (polyphenols, vitamin E, C), skin dryness (sugars and fatty acids), ultraviolet radiation (carotenoids), oxidative damage (polyphenols), and inflammation and melanogenesis, which are involved in human skin damage [256,257]. These scientific pieces of evidence are turning cosmetologists’ attention toward introducing bee pollen into their beauty products and formulations, and guaranteeing better quality and functionality.
6. Conclusions
As has been shown so far, pollen grains are microscopic vegetal cells produced and dispersed during the process of plant reproduction. In the hive, pollen grains transform into bee bread through the fermentation process and become accessible for human consumption because of their complete composition of macronutrients such as proteins, carbohydrates, lipids, and micronutrients such as minerals, vitamins, and phenolic compounds. This diversified composition affords a wide range of pharmacological and biological properties to the bee pollen. Nonetheless, due to the significant variability of its composition, which is affected by several factors, bee pollen application in phytomedicine remains quite limited. On this basis, scientists and professionals should pay closer attention to a few key aspects: (i) standardization should be expanded to include the phenolic composition and nutritional value of different types of bee-collected pollen, especially monofloral pollen; (ii) more quality-control research is needed to encourage beekeepers to produce clean, safe, and economically valuable bee pollen; (iii) as bee pollen is partially digested by human digestive enzymes, more pharmacological and biochemical studies are necessary to enhance the bioavailability of bee pollen bioactive compounds and capitalize on bee pollen’s biological importance; (iv) considering the techno-functional value of bee pollen as a superfood, it can be potentially used as a good ingredient in the food and pharmaceutical industries for the production of novel bee pollen-enriched food products or dietary supplements; (v) eventually, considering bee pollen’s techno-functional value and biological properties, more clinical trials should be conducted to investigate the beneficial effect of this superfood on human health and to encourage the food and pharmaceutical industries to develop and manufacture novel bee pollen-enriched food products and dietary supplements.
Author Contributions
Conceptualization, A.E.G. and M.B.; writing—original draft preparation, A.E.G., M.B., D.O., H.L. and N.E.M.; writing—review and editing, C.H. and B.L.; supervision, C.H. and B.L. All authors have read and agreed to the published version of the manuscript.
Funding
This study was partially funded by the SNAMOPEQ laboratory, FSDM, USMBA, Fez, Morocco.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data are available upon request.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Knox, R.B. The Pollen Grain. In Embryology of Angiosperms; Johri, B.M., Ed.; Springer: Berlin/Heidelberg, Germany, 1984; pp. 197–271. ISBN 978-3-642-69302-1. [Google Scholar]
- Campos, M.G.R.; Bogdanov, S.; de Almeida-Muradian, L.B.; Szczesna, T.; Mancebo, Y.; Frigerio, C.; Ferreira, F. Pollen Composition and Standardisation of Analytical Methods. J. Apic. Res. 2008, 47, 154–161. [Google Scholar] [CrossRef]
- De-Melo, A.A.M.; de Almeida-Muradian, L.B. Chemical Composition of Bee Pollen. In Bee Products-Chemical and Biological Properties; Alvarez-Suarez, J.M., Ed.; Springer International Publishing: Cham, Switzerland, 2017; pp. 221–259. ISBN 978-3-319-59688-4. [Google Scholar]
- Kieliszek, M.; Piwowarek, K.; Kot, A.M.; Błażejak, S.; Chlebowska-Śmigiel, A.; Wolska, I. Pollen and Bee Bread as New Health-Oriented Products: A Review. Trends Food Sci. Technol. 2018, 71, 170–180. [Google Scholar] [CrossRef]
- Asmae, E.G.; Nawal, E.M.; Bakour, M.; Lyoussi, B. Moroccan Monofloral Bee Pollen: Botanical Origin, Physicochemical Characterization, and Antioxidant Activities. J. Food Qual. 2021, 2021, 1–10. [Google Scholar] [CrossRef]
- Mohamed, N.A.; Ahmed, O.M.; Hozayen, W.G.; Ahmed, M.A. Ameliorative Effects of Bee Pollen and Date Palm Pollen on the Glycemic State and Male Sexual Dysfunctions in Streptozotocin-Induced Diabetic Wistar Rats. Biomed. Pharm. 2018, 97, 9–18. [Google Scholar] [CrossRef]
- Maruyama, H.; Sakamoto, T.; Araki, Y.; Hara, H. Anti-Inflammatory Effect of Bee Pollen Ethanol Extract from Cistus Sp. of Spanish on Carrageenan-Induced Rat Hind Paw Edema. BMC Complement. Altern. Med. 2010, 10, 30. [Google Scholar] [CrossRef]
- Kaškonienė, V.; Adaškevičiūtė, V.; Kaškonas, P.; Mickienė, R.; Maruška, A. Antimicrobial and Antioxidant Activities of Natural and Fermented Bee Pollen. Food Biosci. 2020, 34, 100532. [Google Scholar] [CrossRef]
- Wang, B.; Diao, Q.; Zhang, Z.; Liu, Y.; Gao, Q.; Zhou, Y.; Li, S. Antitumor Activity of Bee Pollen Polysaccharides from Rosa Rugosa. Mol. Med. Rep. 2013, 7, 1555–1558. [Google Scholar] [CrossRef]
- Prieto, P.; Pineda, M.; Aguilar, M. Spectrophotometric Quantitation of Antioxidant Capacity through the Formation of a Phosphomolybdenum Complex: Specific Application to the Determination of Vitamin E. Anal. Biochem. 1999, 269, 337–341. [Google Scholar] [CrossRef]
- Roy, R.; Schmitt, A.J.; Thomas, J.B.; Carter, C.J. Nectar Biology: From Molecules to Ecosystems. Plant Sci. 2017, 262, 148–164. [Google Scholar] [CrossRef]
- Wodehouse, R.P. Pollen Grains. In Their Structure, Identification and Significance in Science and Medicine, 1st ed.; McGraw-Hill Book Company: New York, NY, USA, 1935. [Google Scholar]
- Dolezal, A.G.; Toth, A.L. Feedbacks between Nutrition and Disease in Honey Bee Health. Curr. Opin. Insect Sci. 2018, 26, 114–119. [Google Scholar] [CrossRef]
- Nepi, M.; Grasso, D.A.; Mancuso, S. Nectar in Plant–Insect Mutualistic Relationships: From Food Reward to Partner Manipulation. Front. Plant Sci. 2018, 9, 1063. [Google Scholar] [CrossRef]
- Glover, B.J. Pollinator Attraction: The Importance of Looking Good and Smelling Nice. Curr. Biol. 2011, 21, R307–R309. [Google Scholar] [CrossRef]
- Garbuzov, M.; Ratnieks, F.L.W. Quantifying Variation among Garden Plants in Attractiveness to Bees and Other Flower-Visiting Insects. Funct. Ecol. 2014, 28, 364–374. [Google Scholar] [CrossRef]
- Page, R.E.; Scheiner, R.; Erber, J.; Amdam, G.V. The Development and Evolution of Division of Labor and Foraging Specialization in a Social Insect (Apis Mellifera L.). Curr. Top. Dev. Biol. 2006, 74, 253–286. [Google Scholar] [CrossRef]
- Abou-Shaara, H.F. The Foraging Behaviour of Honey Bees, Apis Mellifera: A Review. Vet. Med. 2014, 59, 1–10. [Google Scholar] [CrossRef]
- Vance, J.T.; Williams, J.B.; Elekonich, M.M.; Roberts, S.P. The Effects of Age and Behavioral Development on Honey Bee (Apis Mellifera) Flight Performance. J. Exp. Biol. 2009, 212, 2604–2611. [Google Scholar] [CrossRef]
- Seeley, T.D. The Wisdom of the Hive: The Social Physiology of Honey Bee Colonies; Harvard University Press: Cambridge, MA, USA; London, UK, 1995. [Google Scholar]
- Thorp, R.W. The Collection of Pollen by Bees. In Pollen and Pollination; Dafni, A., Hesse, M., Pacini, E., Eds.; Springer: Vienna, Austria, 2000; pp. 211–223. ISBN 978-3-7091-6306-1. [Google Scholar]
- Nicolson, S.W.; Da Silva Das Neves, S.; Human, H.; Pirk, C.W.W. Digestibility and Nutritional Value of Fresh and Stored Pollen for Honey Bees ( Apis Mellifera Scutellata ). J. Insect Physiol. 2018, 107, 302–308. [Google Scholar] [CrossRef]
- Dozuotų, B.D.T.I.J.; Kūrimas, F. Investigation of Bee Bread and Development of Its Dosage Forms. Proteins 2014, 24, 20.30–21.70. [Google Scholar]
- Radev, Z. Water Content of Honey Bee Collected Pollen from 50 Plants from Bulgaria. Mod. Concepts Dev. Agron. 2018, 3, 314–316. [Google Scholar] [CrossRef]
- Mauriello, G.; De Prisco, A.; Di Prisco, G.; La Storia, A.; Caprio, E. Microbial Characterization of Bee Pollen from the Vesuvius Area Collected by Using Three Different Traps. PLoS ONE 2017, 12, e0183208. [Google Scholar] [CrossRef]
- Kaèániová, M.; Fikselová, M.; Hašèík, P.; Kòazovická, V.; Nô, J.; Fatrcová-Šrámková, K. Changes in microflora of bee pollen treated with uv light and freezing during storage. Ecol. Chem. Eng. A 2010, 17, 89–94. [Google Scholar]
- Mekki, I. Microbial Contamination of Bee Pollen and Impact of Preservation Methods; Univérsité libre de Tunis: Tunis, Tunisia, 2019. [Google Scholar]
- Campos, M.G.R.; Frigerio, C.; Lopes, J.; Bogdanov, S. What Is the Future of Bee-Pollen? J. ApiProd. ApiMed. Sci. 2010, 2, 131–144. [Google Scholar] [CrossRef]
- Percie du Sert, P. Les pollens apicoles. Phytothérapie 2009, 7, 75–82. [Google Scholar] [CrossRef]
- Bogdanov, S. Quality and Standards of Pollen and Beeswax. APIACTA 2004, 38, 334–341. [Google Scholar]
- Martins, M.C.T.; Morgano, M.A.; Vicente, E.; Baggio, S.R.; Rodriguez-Amaya, D.B. Physicochemical composition of bee pollen from eleven brazilian states. J. Apic. Sci. 2011, 55, 11. [Google Scholar]
- De Melo, I.L.P.; de Almeida-Muradian, L.B. Comparison of Methodologies for Moisture Determination on Dried Bee Pollen Samples. Food Sci. Technol. 2011, 31, 194–197. [Google Scholar] [CrossRef]
- Coronel, B.B.; Grasso, D.; Pereira, S.C.; Fernández, G. Caracterización bromatológica del polen apícola argentino. Ciencia Docencia y Tecnología 2004, 38, 145–181. [Google Scholar]
- Vanderplanck, M.; Leroy, B.; Wathelet, B.; Wattiez, R.; Michez, D. Standardized Protocol to Evaluate Pollen Polypeptides as Bee Food Source. Apidologie 2014, 45, 192–204. [Google Scholar] [CrossRef]
- Frias, B.E.D.; Barbosa, C.D.; Lourenço, A.P. Pollen Nutrition in Honey Bees (Apis Mellifera): Impact on Adult Health. Apidologie 2016, 47, 15–25. [Google Scholar] [CrossRef]
- Komosinska-Vassev, K.; Olczyk, P.; Kaźmierczak, J.; Mencner, L.; Olczyk, K. Bee Pollen: Chemical Composition and Therapeutic Application. Evid. -Based Complement. Altern. Med. 2015, 2015, 1–6. [Google Scholar] [CrossRef]
- Negrão, A.F.; Orsi, R.O. Harvesting Season and Botanical Origin Interferes in Production and Nutritional Composition of Bee Pollen. An. Acad. Bras. Ciênc. 2018, 90, 325–332. [Google Scholar] [CrossRef]
- Hendriksma, H.P.; Pachow, C.D.; Nieh, J.C. Effects of Essential Amino Acid Supplementation to Promote Honey Bee Gland and Muscle Development in Cages and Colonies. J. Insect Physiol. 2019, 117, 103906. [Google Scholar] [CrossRef]
- Pizzorno, J.E.; Murray, M.T. Textbook of Natural Medicine–E-Book; Elsevier Health Sciences: Amsterdam, The Netherlands, 2020; ISBN 978-0-323-52380-6. [Google Scholar]
- Kaur, R.; Nagpal, A.K.; Katnoria, J.K. Amino Acids Composition of Pollen Grains of Some Medicinally Important Plant Species. J. Chem. Pharm. Res. 2015, 7, 122–127. [Google Scholar]
- Da Silva, G.R.; da Natividade, T.B.; Camara, C.A.; da Silva, E.M.S.; dos Santos, F.d.A.R.; Silva, T.M.S. Identification of Sugar, Amino Acids and Minerals from the Pollen of Jandaíra Stingless Bees (Melipona Subnitida). Food Nutr. Sci. 2014, 2014. [Google Scholar] [CrossRef]
- Canale, A.; Benelli, G.; Castagna, A.; Sgherri, C.; Poli, P.; Serra, A.; Mele, M.; Ranieri, A.; Signorini, F.; Bientinesi, M.; et al. Microwave-Assisted Drying for the Conservation of Honeybee Pollen. Materials 2016, 9, 363. [Google Scholar] [CrossRef]
- Alvarez-Suarez, J.M. Bee Products-Chemical and Biological Properties; Springer: New York, NY, USA, 2017; ISBN 978-3-319-59689-1. [Google Scholar]
- Gardana, C.; Del Bo’, C.; Quicazán, M.C.; Corrrea, A.R.; Simonetti, P. Nutrients, Phytochemicals and Botanical Origin of Commercial Bee Pollen from Different Geographical Areas. J. Food Compos. Anal. 2018, 73, 29–38. [Google Scholar] [CrossRef]
- Lilek, N.; Gonzales, A.P.; Boži, J.; Borovšak, A.K.; Bertoncelj, J. Chemical Composition and Content of Free Tryptophan in Slovenian Bee Pollen. J. Food Nutr. Res. 2015, 54, 11. [Google Scholar]
- Yang, K.; Wu, D.; Ye, X.; Liu, D.; Chen, J.; Sun, P. Characterization of Chemical Composition of Bee Pollen in China. J. Agric. Food Chem. 2013, 61, 708–718. [Google Scholar] [CrossRef]
- Taha, E.-K.A.; Al-Kahtani, S.; Taha, R. Protein Content and Amino Acids Composition of Bee-Pollens from Major Floral Sources in Al-Ahsa, Eastern Saudi Arabia. Saudi J. Biol. Sci. 2017. [Google Scholar] [CrossRef]
- Isik, A.; Ozdemir, M.; Doymaz, I.; Isik, A.; Ozdemir, M.; Doymaz, I. Effect of Hot Air Drying on Quality Characteristics and Physicochemical Properties of Bee Pollen. Food Sci. Technol. 2019, 39, 224–231. [Google Scholar] [CrossRef]
- Bertoncelj, J.; Polak, T.; Pucihar, T.; Lilek, N.; Borovšak, A.K.; Korošec, M. Carbohydrate Composition of Slovenian Bee Pollens. Int. J. Food Sci. Technol. 2018, 53, 1880–1888. [Google Scholar] [CrossRef]
- Anjos, O.; Paula, V.; Delgado, T.; Estevinho, L. Influence of the Storage Conditions on the Quality of Bee Pollen. Zemdirb. Agric. 2019, 106, 87–94. [Google Scholar] [CrossRef]
- Wu, W.; Wang, K.; Qiao, J.; Dong, J.; Li, Z.; Zhang, H. Improving Nutrient Release of Wall-Disrupted Bee Pollen with a Combination of Ultrasonication and High Shear Technique. J. Sci. Food Agric. 2019, 99, 564–575. [Google Scholar] [CrossRef]
- Ares, A.M.; Valverde, S.; Bernal, J.L.; Nozal, M.J.; Bernal, J. Extraction and Determination of Bioactive Compounds from Bee Pollen. J. Pharm. Biomed. Anal. 2018, 147, 110–124. [Google Scholar] [CrossRef]
- de Arruda, V.A.S.; Vieria dos Santos, A.; Figueiredo Sampaio, D.; da Silva Araújo, E.; de Castro Peixoto, A.L.; Estevinho, M.L.F.; Bicudo de Almeida-Muradian, L. Microbiological Quality and Physicochemical Characterization of Brazilian Bee Pollen. J. Apic. Res. 2017, 56, 231–238. [Google Scholar] [CrossRef]
- Bárbara, M.S.; Machado, C.S.; Sodré, G.D.S.; Dias, L.G.; Estevinho, L.M.; De Carvalho, C.A.L. Microbiological Assessment, Nutritional Characterization and Phenolic Compounds of Bee Pollen from Mellipona Mandacaia Smith, 1983. Molecules 2015, 20, 12525–12544. [Google Scholar] [CrossRef]
- Kalaycıoğlu, Z.; Kaygusuz, H.; Döker, S.; Kolaylı, S.; Erim, F.B. Characterization of Turkish Honeybee Pollens by Principal Component Analysis Based on Their Individual Organic Acids, Sugars, Minerals, and Antioxidant Activities. LWT 2017, 84, 402–408. [Google Scholar] [CrossRef]
- Sattler, J.A.G.; de Melo, I.L.P.; Granato, D.; Araújo, E.; da Silva de Freitas, A.; Barth, O.M.; Sattler, A.; de Almeida-Muradian, L.B. Impact of Origin on Bioactive Compounds and Nutritional Composition of Bee Pollen from Southern Brazil: A Screening Study. Food Res. Int. 2015, 77, 82–91. [Google Scholar] [CrossRef]
- Szczęsna, T. Study on the Sugar Composition of Honeybee-Collected Pollen. Available online: http://agro.icm.edu.pl/agro/element/bwmeta1.element.dl-catalog-2bfe8051-7503-4faa-b7c0-552c257dd9a3 (accessed on 19 October 2020).
- Liolios, V.; Tananaki, C.; Dimou, M.; Kanelis, D.; Rodopoulou, M.-A.; Thrasyvoulou, A. Exploring the Sugar Profile of Unifloral Bee Pollen Using High Performance Liquid Chromatography. J. Food Nutr. Res. 2018, 57, 11. [Google Scholar]
- Belina-Aldemita, M.D.; Opper, C.; Schreiner, M.; D’Amico, S. Nutritional Composition of Pot-Pollen Produced by Stingless Bees (Tetragonula Biroi Friese) from the Philippines. J. Food Compos. Anal. 2019, 82, 103215. [Google Scholar] [CrossRef]
- Omar, W.A.W.; Yahaya, N.; Ghaffar, Z.A.; Fadzilah, N.H. GC-MS Analysis of Chemical Constituents in Ethanolic Bee Pollen Extracts from Three Species of Malaysian Stingless Bee. J. Apic. Sci. 2018, 62, 275–284. [Google Scholar] [CrossRef]
- Li, Y.O.; Komarek, A.R. Dietary Fibre Basics: Health, Nutrition, Analysis, and Applications. Food Qual. Saf. 2017, 1, 47–59. [Google Scholar] [CrossRef]
- Trowell, H.; Burkitt, D.; Heaton, K. Dietary Fibre, Fibre-Depleted Foods and Disease; Academic Press: Orlando, FL, USA, 1985. [Google Scholar]
- Makki, K.; Deehan, E.C.; Walter, J.; Bäckhed, F. The Impact of Dietary Fiber on Gut Microbiota in Host Health and Disease. Cell Host Microbe 2018, 23, 705–715. [Google Scholar] [CrossRef]
- Zhao, L.; Zhang, F.; Ding, X.; Wu, G.; Lam, Y.Y.; Wang, X.; Fu, H.; Xue, X.; Lu, C.; Ma, J.; et al. Gut Bacteria Selectively Promoted by Dietary Fibers Alleviate Type 2 Diabetes. Science 2018, 359, 1151–1156. [Google Scholar] [CrossRef]
- Bishehsari, F.; Engen, P.A.; Preite, N.Z.; Tuncil, Y.E.; Naqib, A.; Shaikh, M.; Rossi, M.; Wilber, S.; Green, S.J.; Hamaker, B.R.; et al. Dietary Fiber Treatment Corrects the Composition of Gut Microbiota, Promotes SCFA Production, and Suppresses Colon Carcinogenesis. Genes 2018, 9, 102. [Google Scholar] [CrossRef]
- Dayib, M.; Larson, J.; Slavin, J. Dietary Fibers Reduce Obesity-Related Disorders: Mechanisms of Action. Curr. Opin. Clin. Nutr. Metab. Care 2020, 23, 445–450. [Google Scholar] [CrossRef]
- McRae, M.P. Dietary Fiber Is Beneficial for the Prevention of Cardiovascular Disease: An Umbrella Review of Meta-Analyses. J. Chiropr. Med. 2017, 16, 289–299. [Google Scholar] [CrossRef]
- Pituch-Zdanowska, A.; Banaszkiewicz, A.; Albrecht, P. The Role of Dietary Fibre in Inflammatory Bowel Disease. Prz. Gastroenterol. 2015, 10, 135–141. [Google Scholar] [CrossRef]
- Merenkova, S.P.; Zinina, O.V.; Stuart, M.; Okuskhanova, E.K.; Androsova, N.V. Effects of dietary fiber on human health: A review. Human. Sport. Med. 2020, 20, 106–113. [Google Scholar] [CrossRef]
- Fuenmayor, B.C.; Zuluaga, D.C.; Díaz, M.C.; de Quicazán, C.M.; Cosio, M.; Mannino, S. Evaluation of the Physicochemical and Functional Properties of Colombian Bee Pollen. Revista MVZ Córdoba 2014, 19, 4003–4014. [Google Scholar] [CrossRef]
- Calder, P.C. Omega-3 Fatty Acids and Inflammatory Processes: From Molecules to Man. Biochem. Soc. Trans. 2017, 45, 1105–1115. [Google Scholar] [CrossRef]
- La Guardia, M.; Giammanco, S.; Di Majo, D.; Tabacchi, C.; Tripoli, E.; Giammanco, M. Omega 3 Fatty Acids: Biological Activity and Effects on Human Health. Panminerva. Med. 2005, 47, 245–257. [Google Scholar]
- Thakur, M.; Nanda, V. Composition and Functionality of Bee Pollen: A Review. Trends Food Sci. Technol. 2020, 98, 82–106. [Google Scholar] [CrossRef]
- Li, Q.; Liang, X.; Zhao, L.; Zhang, Z.; Xue, X.; Wang, K.; Wu, L. UPLC-Q-Exactive Orbitrap/MS-Based Lipidomics Approach To Characterize Lipid Extracts from Bee Pollen and Their in Vitro Anti-Inflammatory Properties. J. Agric. Food Chem. 2017, 65, 6848–6860. [Google Scholar] [CrossRef]
- AL-Kahtani, S.N. Fatty Acids and B Vitamins Contents in Honey Bee Collected Pollen in Relation to Botanical Origin. Sci. J. King Faisal Univ. 2017, 18, 41–48. [Google Scholar]
- Conte, G.; Benelli, G.; Serra, A.; Signorini, F.; Bientinesi, M.; Nicolella, C.; Mele, M.; Canale, A. Lipid Characterization of Chestnut and Willow Honeybee-Collected Pollen: Impact of Freeze-Drying and Microwave-Assisted Drying. J. Food Compos. Anal. 2017, 55, 12–19. [Google Scholar] [CrossRef]
- Dong, J.; Yang, Y.; Wang, X.; Zhang, H. Fatty Acid Profiles of 20 Species of Monofloral Bee Pollen from China. J. Apic. Res. 2015, 54, 503–511. [Google Scholar] [CrossRef]
- Bailey, R.L.; West, K.P., Jr.; Black, R.E. The Epidemiology of Global Micronutrient Deficiencies. ANM 2015, 66, 22–33. [Google Scholar] [CrossRef]
- FAO; IFAD; UNICEF; WFP; WHO. The State of Food Security and Nutrition in the World 2020. In Transforming Food Systems for Affordable Healthy Diets; FAO: Rome, Italy, 2020. [Google Scholar] [CrossRef]
- Gernand, A.D.; Schulze, K.J.; Stewart, C.P.; West, K.P.; Christian, P. Micronutrient Deficiencies in Pregnancy Worldwide: Health Effects and Prevention. Nat. Rev. Endocrinol. 2016, 12, 274–289. [Google Scholar] [CrossRef]
- Kangalgil, M.; Sahinler, A.; Kırkbir, I.B.; Ozcelik, A.O. Associations of Maternal Characteristics and Dietary Factors with Anemia and Iron-Deficiency in Pregnancy. J. Gynecol. Obstet. Hum. Reprod. 2021, 50, 102137. [Google Scholar] [CrossRef]
- Khayat, S.; Fanaei, H.; Ghanbarzehi, A. Minerals in Pregnancy and Lactation: A Review Article. J. Clin. Diagn. Res. 2017, 11, QE01–QE05. [Google Scholar] [CrossRef]
- Looman, M.; Schoenaker, D.A.J.M.; Soedamah-Muthu, S.S.; Mishra, G.D.; Geelen, A.; Feskens, E.J.M. Pre-Pregnancy Dietary Micronutrient Adequacy Is Associated with Lower Risk of Developing Gestational Diabetes in Australian Women. Nutr. Res. 2019, 62, 32–40. [Google Scholar] [CrossRef]
- Moreno-Reyes, R. Micronutrient Deficiencies and the Thyroid in Pregnancy. Placenta 2017, 51, 102. [Google Scholar] [CrossRef]
- Ibrahim, M.K.; Zambruni, M.; Melby, C.L.; Melby, P.C. Impact of Childhood Malnutrition on Host Defense and Infection. Clin. Microbiol. Rev. 2017, 30, 919–971. [Google Scholar] [CrossRef]
- Ritchie, H.; Roser, M. Micronutrient Deficiency. Our World in Data, August 2017. [Google Scholar]
- Tan, M.; Yang, T.; Zhu, J.; Li, Q.; Lai, X.; Li, Y.; Tang, T.; Chen, J.; Li, T. Maternal Folic Acid and Micronutrient Supplementation Is Associated with Vitamin Levels and Symptoms in Children with Autism Spectrum Disorders. Reprod. Toxicol. 2020, 91, 109–115. [Google Scholar] [CrossRef]
- Gorji, A.; Khaleghi Ghadiri, M. Potential Roles of Micronutrient Deficiency and Immune System Dysfunction in the Coronavirus Disease 2019 (COVID-19) Pandemic. Nutrition 2021, 82, 111047. [Google Scholar] [CrossRef]
- Hoffman, R. Micronutrient Deficiencies in the Elderly–Could Ready Meals Be Part of the Solution? J. Nutr. Sci. 2017, 6, e2. [Google Scholar] [CrossRef]
- Song, E.K.; Moser, D.K.; Kang, S.-M.; Lennie, T.A. Association of Depressive Symptoms and Micronutrient Deficiency With Cardiac Event–Free Survival in Patients With Heart Failure. J. Card. Fail. 2015, 21, 945–951. [Google Scholar] [CrossRef]
- Wang, M.X.; Koh, J.; Pang, J. Association between Micronutrient Deficiency and Acute Respiratory Infections in Healthy Adults: A Systematic Review of Observational Studies. Nutr. J. 2019, 18, 80. [Google Scholar] [CrossRef]
- Shankar, A.H. 140-Mineral Deficiencies. In Hunter’s Tropical Medicine and Emerging Infectious Disease, 9th ed.; Magill, A.J., Hill, D.R., Solomon, T., Ryan, E.T., Eds.; W.B. Saunders: London, UK, 2013; pp. 1003–1010. ISBN 978-1-4160-4390-4. [Google Scholar]
- Bogdanov, S. Pollen: Nutrition, Functional Properties, Health. Bee Prod. Sci. 2012, 20–350. [Google Scholar]
- Li, Q.-Q.; Wang, K.; Marcucci, M.C.; Sawaya, A.C.H.F.; Hu, L.; Xue, X.-F.; Wu, L.-M.; Hu, F.-L. Nutrient-Rich Bee Pollen: A Treasure Trove of Active Natural Metabolites. J. Funct. Foods 2018, 49, 472–484. [Google Scholar] [CrossRef]
- Adaškevičiūtė, V.; Kaškonienė, V.; Kaškonas, P.; Barčauskaitė, K.; Maruška, A. Comparison of Physicochemical Properties of Bee Pollen with Other Bee Products. Biomolecules 2019, 9, 819. [Google Scholar] [CrossRef]
- Altunatmaz, S.S.; Tarhan, D.; Aksu, F.; Barutçu, U.B.; Or, M.E. Mineral Element and Heavy Metal (Cadmium, Lead and Arsenic) Levels of Bee Pollen in Turkey. Food Sci. Technol. 2017, 37, 136–141. [Google Scholar] [CrossRef]
- Sattler, J.A.G.; De-Melo, A.A.M.; Nascimento, K.S.D.; Melo, I.L.P.D.; Mancini-Filho, J.; Sattler, A.; Almeida-Muradian, L.B.D. Essential Minerals and Inorganic Contaminants (Barium, Cadmium, Lithium, Lead and Vanadium) in Dried Bee Pollen Produced in Rio Grande Do Sul State, Brazil. Food Sci. Technol. 2016, 36, 505–509. [Google Scholar] [CrossRef]
- Mellidou, I.; Georgiadou, E.C.; Kaloudas, D.; Kalaitzis, P.; Fotopoulos, V.; Kanellis, A.K. Chapter 17-Vitamins. In Postharvest Physiology and Biochemistry of Fruits and Vegetables; Yahia, E.M., Ed.; Woodhead Publishing: Cambridge, UK, 2019; pp. 359–383. ISBN 978-0-12-813278-4. [Google Scholar]
- Ravisankar, P.; Reddy, A.A.; Nagalakshmi, B.; Sai, O.; Kumar, B.V.; Anvith, P.S. The Comprehensive Review on Fat Soluble Vitamins. IOSR J. Pharm. 2015, 5, 12–28. [Google Scholar]
- Yoshii, K.; Hosomi, K.; Sawane, K.; Kunisawa, J. Metabolism of Dietary and Microbial Vitamin B Family in the Regulation of Host Immunity. Front. Nutr. 2019, 6, 48. [Google Scholar] [CrossRef]
- Farag, S.; El-Rayes, T. Effect of Bee-Pollen Supplementation on Performance, Carcass Traits and Blood Parameters of Broiler Chickens. Asian J. Anim. Vet. Adv. 2016, 11, 168–177. [Google Scholar] [CrossRef]
- Ecem Bayram, N. Vitamin, Mineral, Polyphenol, Amino Acid Profile of Bee Pollen from Rhododendron Ponticum (Source of “Mad Honey”): Nutritional and Palynological Approach. Food Meas. 2021, 15, 2659–2666. [Google Scholar] [CrossRef]
- Estevinho, L.M.; Dias, T.; Anjos, O. Influence of the Storage Conditions (Frozen vs. Dried) in Health-Related Lipid Indexes and Antioxidants of Bee Pollen. Eur. J. Lipid Sci. Technol. 2019, 121, 1800393. [Google Scholar] [CrossRef]
- Barbieri, D.; Gabriele, M.; Summa, M.; Colosimo, R.; Leonardi, D.; Domenici, V.; Pucci, L. Antioxidant, Nutraceutical Properties, and Fluorescence Spectral Profiles of Bee Pollen Samples from Different Botanical Origins. Antioxidants 2020, 9, 1001. [Google Scholar] [CrossRef]
- de Arruda, V.A.S.; Pereira, A.A.S.; de Freitas, A.S.; Barth, O.M.; de Almeida-Muradian, L.B. Dried Bee Pollen: B Complex Vitamins, Physicochemical and Botanical Composition. J. Food Compos. Anal. 2013, 29, 100–105. [Google Scholar] [CrossRef]
- de Melo, I.L.P.; de Almeida-Muradian, L.B. Stability of Antioxidants Vitamins in Bee Pollen Samples. Quím. Nova 2010, 33, 514–518. [Google Scholar] [CrossRef]
- De-Melo, A.A.M.; Estevinho, M.L.M.F.; Sattler, J.A.G.; Souza, B.R.; da Freitas, A.S.; Barth, O.M.; Almeida-Muradian, L.B. Effect of Processing Conditions on Characteristics of Dehydrated Bee-Pollen and Correlation between Quality Parameters. LWT-Food Sci. Technol. 2016, 65, 808–815. [Google Scholar] [CrossRef]
- Chawla, J.; Kvarnberg, D. Chapter 59-Hydrosoluble Vitamins. In Handbook of Clinical Neurology; Biller, J., Ferro, J.M., Eds.; Neurologic Aspects of Systemic Disease Part II; Elsevier: Amsterdam, The Netherlands, 2014; Volume 120, pp. 891–914. [Google Scholar]
- Aylanc, V.; Falcão, S.I.; Ertosun, S.; Vilas-Boas, M. From the Hive to the Table: Nutrition Value, Digestibility and Bioavailability of the Dietary Phytochemicals Present in the Bee Pollen and Bee Bread. Trends Food Sci. Technol. 2021, 109, 464–481. [Google Scholar] [CrossRef]
- Khalifa, S.A.M.; Elashal, M.H.; Yosri, N.; Du, M.; Musharraf, S.G.; Nahar, L.; Sarker, S.D.; Guo, Z.; Cao, W.; Zou, X.; et al. Bee Pollen: Current Status and Therapeutic Potential. Nutrients 2021, 13, 1876. [Google Scholar] [CrossRef]
- Oliveira, K.C.L.S.; Moriya, M.; Azedo, R.A.B.; de Almeida-Muradian, L.B.; Teixeira, E.W.; Alves, M.L.T.M.F.; Moreti, A.C. de C.C. Relationship between Botanical Origin and Antioxidants Vitamins of Bee-Collected Pollen. Quím. Nova 2009, 32, 1099–1102. [Google Scholar] [CrossRef]
- Campos, M.G.; Cunha, A.; Markham, K.R. Bee-Pollen: Composition, Properties, and Applications. In Bee Products; Mizrahi, A., Lensky, Y., Eds.; Springer: Boston, MA, USA, 1997; pp. 93–100. ISBN 978-1-4757-9373-4. [Google Scholar]
- Kaškonienė, V.; Ruočkuvienė, G.; Kaškonas, P.; Akuneca, I.; Maruška, A. Chemometric Analysis of Bee Pollen Based on Volatile and Phenolic Compound Compositions and Antioxidant Properties. Food Anal. Methods 2015, 8, 1150–1163. [Google Scholar] [CrossRef]
- Milani, A.; Basirnejad, M.; Shahbazi, S.; Bolhassani, A. Carotenoids: Biochemistry, Pharmacology and Treatment. Br. J. Pharmacol. 2017, 174, 1290–1324. [Google Scholar] [CrossRef]
- Karabagias, I.K.; Karabagias, V.K.; Karabournioti, S.; Badeka, A.V. Aroma Identification of Greek Bee Pollen Using HS-SPME/GC–MS. Eur. Food Res. Technol. 2021, 247, 1781–1789. [Google Scholar] [CrossRef]
- Belhadj, H.; Harzallah, D.; Bouamra, D.; Khennouf, S.; Dahamna, S.; Ghadbane, M. Phenotypic and Genotypic Characterization of Some Lactic Acid Bacteria Isolated from Bee Pollen: A Preliminary Study. Biosci. Microbiota Food Health 2014, 33, 11–23. [Google Scholar] [CrossRef]
- Friedle, C.; D’Alvise, P.; Schweikert, K.; Wallner, K.; Hasselmann, M. Changes of Microorganism Composition in Fresh and Stored Bee Pollen from Southern Germany. Environ. Sci. Pollut. Res. 2021, 28, 47251–47261. [Google Scholar] [CrossRef]
- Gilliam, M. microbiology of pollen and bee BREAD: The yeasts. Apidologie 1979, 10, 43–53. [Google Scholar] [CrossRef]
- Tajabadi, N.; Mardan, M.; Saari, N.; Mustafa, S.; Bahreini, R.; Manap, M.Y.A. Identification of Lactobacillus Plantarum, Lactobacillus Pentosus and Lactobacillus Fermentum from Honey Stomach of Honeybee. Braz. J. Microbiol. 2013, 44, 717–722. [Google Scholar] [CrossRef]
- Belhadj, H.; Harzallah, D.; Khennouf, S.; Dahamna, S.; Bouharati, S.; Baghiani, A. Isolation, identification and antimicrobial activity of lactic acid bacteria from algerian honeybee collected pollen. Acta Hortic. 2010, 51–58. [Google Scholar] [CrossRef]
- Vásquez, A.; Olofsson, T.C. The Lactic Acid Bacteria Involved in the Production of Bee Pollen and Bee Bread. J. Apic. Res. 2009, 48, 189–195. [Google Scholar] [CrossRef]
- Belhamra, Z.; Harzallah, D.; Naili, O.; Belhadj, H. Probiotic Potential of Lactobacillus Strains Isolated from Fresh Bee Pollen. Pharm. Lett. 2016, 8, 357–365. [Google Scholar]
- Filannino, P.; Di Cagno, R.; Addante, R.; Pontonio, E.; Gobbetti, M. Metabolism of Fructophilic Lactic Acid Bacteria Isolated from the Apis Mellifera L. Bee Gut: Phenolic Acids as External Electron Acceptors. Appl. Environ. Microbiol. 2016, 82, 6899–6911. [Google Scholar] [CrossRef]
- George Kerry, R.; Patra, J.K.; Gouda, S.; Park, Y.; Shin, H.-S.; Das, G. Benefaction of Probiotics for Human Health: A Review. J. Food Drug Anal. 2018, 26, 927–939. [Google Scholar] [CrossRef]
- Goldin, B.R.; Gorbach, S.L. Clinical Indications for Probiotics: An Overview. Clin. Infect. Dis. 2008, 46, S96–S100. [Google Scholar] [CrossRef]
- Petrof, E.O. Probiotics and Gastrointestinal Disease: Clinical Evidence and Basic Science. Anti-Inflamm. Anti-Allergy Agents Med. Chem. (Former. Curr. Med. Chem. -Anti-Inflamm. Anti-Allergy Agents) 2009, 8, 260–269. [Google Scholar] [CrossRef]
- Sun, Z.; Sun, X.; Li, J.; Li, Z.; Hu, Q.; Li, L.; Hao, X.; Song, M.; Li, C. Using Probiotics for Type 2 Diabetes Mellitus Intervention: Advances, Questions, and Potential. Crit. Rev. Food Sci. Nutr. 2020, 60, 670–683. [Google Scholar] [CrossRef]
- Kaškonienė, V.; Kaškonas, P.; Maruška, A. Volatile Compounds Composition and Antioxidant Activity of Bee Pollen Collected in Lithuania. Chem. Pap. 2015, 69, 291–299. [Google Scholar] [CrossRef]
- Klatt, B.K.; Burmeister, C.; Westphal, C.; Tscharntke, T.; von Fragstein, M. Flower Volatiles, Crop Varieties and Bee Responses. PLoS ONE 2013, 8, e72724. [Google Scholar] [CrossRef]
- Starowicz, M.; Hanus, P.; Lamparski, G.; Sawicki, T. Characterizing the Volatile and Sensory Profiles, and Sugar Content of Beeswax, Beebread, Bee Pollen, and Honey. Molecules 2021, 26, 3410. [Google Scholar] [CrossRef]
- Özcan, M.M.; Aljuhaimi, F.; Babiker, E.E.; Uslu, N.; Ceylan, D.A.; Ghafoor, K.; Özcan, M.M.; Dursun, N.; Ahmed, I.M.; Jamiu, F.G.; et al. Determination of Antioxidant Activity, Phenolic Compound, Mineral Contents and Fatty Acid Compositions of Bee Pollen Grains Collected from Different Locations. J. Apic. Sci. 2019, 63, 69–79. [Google Scholar] [CrossRef]
- Rzepecka-Stojko, A.; Stojko, J.; Kurek-Górecka, A.; Górecki, M.; Kabała-Dzik, A.; Kubina, R.; Moździerz, A.; Buszman, E. Polyphenols from Bee Pollen: Structure, Absorption, Metabolism and Biological Activity. Molecules 2015, 20, 21732–21749. [Google Scholar] [CrossRef]
- Şahin, S.; Karkar, B. The Antioxidant Properties of the Chestnut Bee Pollen Extract and Its Preventive Action against Oxidatively Induced Damage in DNA Bases. J. Food Biochem. 2019, 43, e12888. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, J.; Zhou, Q.; Wang, L.; Huang, W.; Wang, R. Effect of Ultrasonic and Ball-milling Treatment on Cell Wall, Nutrients, and Antioxidant Capacity of Rose (Rosa Rugosa) Bee Pollen, and Identification of Bioactive Components. J. Sci. Food Agric. 2019, 99, 5350–5357. [Google Scholar] [CrossRef]
- Bridi, R.; Atala, E.; Pizarro, P.N.; Montenegro, G. Honeybee Pollen Load: Phenolic Composition and Antimicrobial Activity and Antioxidant Capacity. J. Nat. Prod. 2019, 82, 559–565. [Google Scholar] [CrossRef]
- Santa Bárbara, M.F.; Moreira, M.M.; Machado, C.S.; Chambó, E.D.; Pascoal, A.; de Carvalho, C.A.L.; da Silva Sodré, G.; Delerue-Matos, C.; Estevinho, L.M. Storage Methods, Phenolic Composition, and Bioactive Properties of Apis Mellifera and Trigona Spinipes Pollen. J. Apic. Res. 2021, 60, 99–107. [Google Scholar] [CrossRef]
- Ares, A.M.; Soto, M.E.; Nozal, M.J.; Bernal, J.L.; Higes, M.; Bernal, J. Determination of Resveratrol and Piceid Isomers in Bee Pollen by Liquid Chromatography Coupled to Electrospray Ionization-Mass Spectrometry. Food Anal. Methods 2015, 8, 1565–1575. [Google Scholar] [CrossRef]
- El Ghouizi, A.; El Menyiy, N.; Falcão, S.I.; Vilas-Boas, M.; Lyoussi, B. Chemical Composition, Antioxidant Activity, and Diuretic Effect of Moroccan Fresh Bee Pollen in Rats. Vet World 2020, 13, 1251–1261. [Google Scholar] [CrossRef]
- Oroian, M.; Ursachi, F.; Dranca, F. Ultrasound-Assisted Extraction of Polyphenols from Crude Pollen. Antioxidants 2020, 9, 322. [Google Scholar] [CrossRef]
- Rocchetti, G.; Castiglioni, S.; Maldarizzi, G.; Carloni, P.; Lucini, L. UHPLC-ESI-QTOF-MS Phenolic Profiling and Antioxidant Capacity of Bee Pollen from Different Botanical Origin. Int. J. Food Sci. Technol. 2019, 54, 335–346. [Google Scholar] [CrossRef]
- Denisow, B.; Denisow-Pietrzyk, M. Biological and Therapeutic Properties of Bee Pollen: A Review: Bee Pollen and Medicine. J. Sci. Food Agric. 2016, 96, 4303–4309. [Google Scholar] [CrossRef]
- El-Guendouz, S.; Lyoussi, B.; Miguel, M.G. Insight into the Chemical Composition and Biological Properties of Mediterranean Royal Jelly. J. Apic. Res. 2020, 59, 890–909. [Google Scholar] [CrossRef]
- Aličić, D.; Flanjak, I.; Ačkar, Đ.; Jašić, M.; Babić, J.; Šubarić, D. Physicochemical Properties and Antioxidant Capacity of Bee Pollen Collected in Tuzla Canton (B&H). J. Cent. Eur. Agric. 2020, 21, 42–50. [Google Scholar] [CrossRef]
- AbdElsalam, E.; Foda, H.S.; Abdel-Aziz, M.S.; Abd, F.K. Antioxidant and Antimicrobial Activities of Egyptian Bee Pollen. Middle East J. Appl. Sci. 2018, 8, 1248–1255. [Google Scholar]
- de Florio Almeida, J.; dos Reis, A.S.; Heldt, L.F.S.; Pereira, D.; Bianchin, M.; de Moura, C.; Oviedo, M.V.P.; Haminiuk, C.W.I.; Ribeiro, I.S.; da Luz, C.F.P.; et al. Lyophilized Bee Pollen Extract: A Natural Antioxidant Source to Prevent Lipid Oxidation in Refrigerated Sausages. LWT-Food Sci. Technol. 2017, 76, 299–305. [Google Scholar] [CrossRef]
- Anjos, O.; Fernandes, R.; Cardoso, S.M.; Delgado, T.; Farinha, N.; Paula, V.; Estevinho, L.M.; Carpes, S.T. Bee Pollen as a Natural Antioxidant Source to Prevent Lipid Oxidation in Black Pudding. LWT 2019, 111, 869–875. [Google Scholar] [CrossRef]
- Atsalakis, E.; Chinou, I.; Makropoulou, M.; Karabournioti, S.; Graikou, K. Evaluation of Phenolic Compounds in Cistus Creticus Bee Pollen from Greece. Antioxidant and Antimicrobial Properties. Nat. Prod. Commun. 2017, 12, 1934578X1701201141. [Google Scholar] [CrossRef]
- Bleha, R.; Shevtsova, T.; Kruzik, V.; Brindza, J.; Sinica, A. Morphology, Physicochemical Properties and Antioxidant Capacity of Bee Pollens. Czech J. Food Sci. 2019, 37, 1–8. [Google Scholar] [CrossRef]
- Carpes, S.T.; Begnini, R.; de Alencar, S.M.; Masson, M.L. Study of Preparations of Bee Pollen Extracts, Antioxidant and Antibacterial Activity. Ciênc. Agrotec. 2007, 31, 1818–1825. [Google Scholar] [CrossRef]
- De-Melo, A.A.M.; Estevinho, L.M.; Moreira, M.M.; Delerue-Matos, C.; de Freitas, A.D.S.; Barth, O.M.; Almeida-Muradian, L.B. de A Multivariate Approach Based on Physicochemical Parameters and Biological Potential for the Botanical and Geographical Discrimination of Brazilian Bee Pollen. Food Biosci. 2018, 25, 91–110. [Google Scholar] [CrossRef]
- Duarte, A.W.F.; dos Vasconcelos, M.R.S.; Oda-Souza, M.; de Oliveira, F.F.; López, A.M.Q. Honey and Bee Pollen Produced by Meliponini (Apidae) in Alagoas, Brazil: Multivariate Analysis of Physicochemical and Antioxidant Profiles. Food Sci. Technol. 2018, 38, 493–503. [Google Scholar] [CrossRef]
- Dulger Altiner, D.; Sandίkcί Altunatmaz, S.; Sabuncu, M.; Aksu, F.; Sahan, Y. In-Vitro Bioaccessibility of Antioxidant Properties of Bee Pollen in Turkey. Food Sci. Technol. 2020, 41, 133–141. [Google Scholar] [CrossRef]
- Fatrcová-Šramková, K.; Nôžková, J.; Máriássyová, M.; Kačániová, M. Biologically Active Antimicrobial and Antioxidant Substances in the Helianthus Annuus L. Bee Pollen. J. Environ. Sci. Health Part B 2016, 51, 176–181. [Google Scholar] [CrossRef]
- Fatrcová-Šramková, K.; Nôžková, J.; Kačániová, M.; Máriássyová, M.; Rovná, K.; Stričík, M. Antioxidant and Antimicrobial Properties of Monofloral Bee Pollen. J. Environ. Sci. Health Part B 2013, 48, 133–138. [Google Scholar] [CrossRef]
- Feás, X.; Vázquez-Tato, M.P.; Estevinho, L.; Seijas, J.A.; Iglesias, A. Organic Bee Pollen: Botanical Origin, Nutritional Value, Bioactive Compounds, Antioxidant Activity and Microbiological Quality. Molecules 2012, 17, 8359–8377. [Google Scholar] [CrossRef]
- Futui, W.; Thongwai, N. Antimicrobial and Antioxidant Activities, Total Phenolic and Flavonoid Contents of Bee Pollen Crude Extracts. Int. J. Biosci. Biochem. Bioinform 2020, 10, 42–48. [Google Scholar] [CrossRef]
- Jin, T.-Y.; Saravanakumar, K.; Wang, M.-H. In Vitro and in Vivo Antioxidant Properties of Water and Methanol Extracts of Linden Bee Pollen. Biocatal. Agric. Biotechnol. 2018, 13, 186–189. [Google Scholar] [CrossRef]
- Kostić, A.Ž.; Milinčić, D.D.; Gašić, U.M.; Nedić, N.; Stanojević, S.P.; Tešić, Ž.L.; Pešić, M.B. Polyphenolic Profile and Antioxidant Properties of Bee-Collected Pollen from Sunflower (Helianthus Annuus L.) Plant. LWT 2019, 112, 108244. [Google Scholar] [CrossRef]
- Leja, M.; Mareczek, A.; Wyżgolik, G.; Klepacz-Baniak, J.; Czekońska, K. Antioxidative Properties of Bee Pollen in Selected Plant Species. Food Chem. 2007, 100, 237–240. [Google Scholar] [CrossRef]
- Mayda, N.; Özkök, A.; Ecem Bayram, N.; Gerçek, Y.C.; Sorkun, K. Bee Bread and Bee Pollen of Different Plant Sources: Determination of Phenolic Content, Antioxidant Activity, Fatty Acid and Element Profiles. Food Meas. 2020, 14, 1795–1809. [Google Scholar] [CrossRef]
- Mejías, E.; Montenegro, G. The Antioxidant Activity of C Hilean Honey and Bee Pollen Produced in the L Laima V Olcano’s Zones. J. Food Qual. 2012, 35, 315–322. [Google Scholar] [CrossRef]
- Mohdaly, A.A.A.; Mahmoud, A.A.; Roby, M.H.H.; Smetanska, I.; Ramadan, M.F. Phenolic Extract from Propolis and Bee Pollen: Composition, Antioxidant and Antibacterial Activities. J. Food Biochem. 2015, 39, 538–547. [Google Scholar] [CrossRef]
- Nurdianah, H.F.; Firdaus, A.A.; Azam, O.E.; Adnan, W.W. Antioxidant Activity of Bee Pollen Ethanolic Extracts from Malaysian Stingless Bee Measured Using DPPH-HPLC Assay. Int. Food Res. J. 2016, 23, 403. [Google Scholar]
- Pascoal, A.; Rodrigues, S.; Teixeira, A.; Feás, X.; Estevinho, L.M. Biological Activities of Commercial Bee Pollens: Antimicrobial, Antimutagenic, Antioxidant and Anti-Inflammatory. Food Chem. Toxicol. 2014, 63, 233–239. [Google Scholar] [CrossRef]
- Rebiai, A.; Lanez, T. Chemical Composition and Antioxidant Activity of Apis Mellifera Bee Pollen from Northwest Algeria. J. Fundam. Appl. Sci. 2015, 4, 155. [Google Scholar] [CrossRef]
- Saral, Ö.; Kilicarslan, M.; Şahin, H.; Yildiz, O.; Dincer, B. Evaluation of Antioxidant Activity of Bee Products of Different Bee Races in Turkey. Turk. J. Vet. Anim. Sci. 2019, 43, 441–447. [Google Scholar] [CrossRef]
- Ulusoy, E.; Kolayli, S. Phenolic Composition and Antioxidant Properties of Anzer Bee Pollen. J. Food Biochem. 2014, 38, 73–82. [Google Scholar] [CrossRef]
- dos Vasconcelos, M.R.S.; Duarte, A.W.F.; Gomes, E.P.; da Silva, S.C.; López, A.M.Q. Physicochemical Composition and Antioxidant Potential of Bee Pollen from Different Botanical Sources in Alagoas, Brazil. Ciência e Agrotecnologia 2017, 41, 447–458. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, X.; Wang, K.; Li, C. Antioxidant and Tyrosinase Inhibitory Properties of Aqueous Ethanol Extracts from Monofloral Bee Pollen. J. Apic. Sci. 2015, 59, 119–128. [Google Scholar] [CrossRef]
- Zou, Y.; Hu, J.; Huang, W.; Zhu, L.; Shao, M.; Dordoe, C.; Ahn, Y.-J.; Wang, D.; Zhao, Y.; Xiong, Y. The Botanical Origin and Antioxidant, Anti-BACE1 and Antiproliferative Properties of Bee Pollen from Different Regions of South Korea. BMC Complement. Med. Ther. 2020, 20, 236. [Google Scholar] [CrossRef]
- Carpes, S.T.; Prado, A.; Moreno, I.A.M.; Mourão, G.B.; de Alencar, S.M.; Masson, M.L. Avaliação do potencial antioxidante do pólen apícola produzido na região sul do Brasil. Química Nova 2008, 31, 1660–1664. [Google Scholar] [CrossRef]
- Crane, E. The Past and Present Importance of Bee Products to Man. In Bee Products: Properties, Applications, and Apitherapy; Mizrahi, A., Lensky, Y., Eds.; Springer: Boston, MA, USA, 1997; pp. 1–13. ISBN 978-1-4757-9371-0. [Google Scholar]
- Mizrahi, A.; Lensky, Y. (Eds.) Bee Products: Properties, Applications, and Apitherapy; Springer: New York, NY, USA, 1997; ISBN 978-0-306-45502-5. [Google Scholar]
- Arivarasu, N.A.; Priyamvada, S.; Mahmood, R. Oral Administration of Caffeic Acid Ameliorates the Effect of Cisplatin on Brush Border Membrane Enzymes and Antioxidant System in Rat Intestine. Exp. Toxicol. Pathol. 2013, 65, 21–25. [Google Scholar] [CrossRef]
- Derochette, S.; Franck, T.; Mouithys-Mickalad, A.; Deby-Dupont, G.; Neven, P.; Serteyn, D. Intra- and Extracellular Antioxidant Capacities of the New Water-Soluble Form of Curcumin (NDS27) on Stimulated Neutrophils and HL-60 Cells. Chem. Biol. Interact 2013, 201, 49–57. [Google Scholar] [CrossRef]
- Aabed, K.; Shafi Bhat, R.; Moubayed, N.; Al-Mutiri, M.; Al-Marshoud, M.; Al-Qahtani, A.; Ansary, A. Ameliorative Effect of Probiotics (Lactobacillus Paracaseii and Protexin®) and Prebiotics (Propolis and Bee Pollen) on Clindamycin and Propionic Acid-Induced Oxidative Stress and Altered Gut Microbiota in a Rodent Model of Autism. Cell Mol. Biol. (Noisy-le-grand) 2019, 65, 1–7. [Google Scholar] [CrossRef]
- Pari, L.; Karthikesan, K. Protective Role of Caffeic Acid against Alcohol-induced Biochemical Changes in Rats. Fundam. Clin. Pharmacol. 2007, 21, 355–361. [Google Scholar] [CrossRef]
- Hemmati, A.A.; Alboghobeish, S.; Ahangarpour, A. Effects of Cinnamic Acid on Memory Deficits and Brain Oxidative Stress in Streptozotocin-Induced Diabetic Mice. Korean J. Physiol. Pharmacol. 2018, 22, 257–267. [Google Scholar] [CrossRef]
- Nafees, S.; Rashid, S.; Ali, N.; Hasan, S.K.; Sultana, S. Rutin Ameliorates Cyclophosphamide Induced Oxidative Stress and Inflammation in Wistar Rats: Role of NFκB/MAPK Pathway. Chem. Biol. Interact. 2015, 231, 98–107. [Google Scholar] [CrossRef]
- Ince, E. The Protective Effect of Quercetin in the Alcohol-Induced Liver and Lymphoid Tissue Injuries in Newborns. Mol. Biol. Rep. 2020, 47, 451–459. [Google Scholar] [CrossRef]
- Alekhya Sita, G.J.; Gowthami, M.; Srikanth, G.; Krishna, M.M.; Rama Sireesha, K.; Sajjarao, M.; Nagarjuna, K.; Nagarjuna, M.; Chinnaboina, G.K.; Mishra, A. Protective Role of Luteolin against Bisphenol A-induced Renal Toxicity through Suppressing Oxidative Stress, Inflammation, and Upregulating Nrf2/ARE/HO-1 Pathway. IUBMB Life 2019, 71, 1041–1047. [Google Scholar] [CrossRef]
- Saad, M.A.; Salam, R.M.A.; Kenawy, S.A.; Attia, A.S. Pinocembrin Attenuates Hippocampal Inflammation, Oxidative Perturbations and Apoptosis in a Rat Model of Global Cerebral Ischemia Reperfusion. Pharmacol. Rep. 2015, 67, 115–122. [Google Scholar] [CrossRef]
- Afsharpour, F.; Javadi, M.; Hashemipour, S.; Koushan, Y. Propolis Supplementation Improves Glycemic and Antioxidant Status in Patients with Type 2 Diabetes: A Randomized, Double-Blind, Placebo-Controlled Study. Complement. Ther. Med. 2019, 43, 283–288. [Google Scholar] [CrossRef]
- Senyuk, B.; Boreyko, L.; Yurnyuk, S. Correction of clinical and biochemical parameters using bee pollen in patients with diabetes. Unity Sci. Int. Sci. Period. J. 2016, 137–139. [Google Scholar]
- Daudu, O.M. Bee Pollen Extracts as Potential Antioxidants and Inhibitors of α-Amylase and α-Glucosidase Enzymes In Vitro Assessment. J. Apic. Sci. 2019, 63, 315–325. [Google Scholar] [CrossRef]
- Adisakwattana, S. Cinnamic Acid and Its Derivatives: Mechanisms for Prevention and Management of Diabetes and Its Complications. Nutrients 2017, 9, 163. [Google Scholar] [CrossRef]
- Lu, Q.; Hao, M.; Wu, W.; Zhang, N.; Isaac, A.T.; Yin, J.; Zhu, X.; Du, L.; Yin, X. Antidiabetic Cataract Effects of GbE, Rutin and Quercetin Are Mediated by the Inhibition of Oxidative Stress and Polyol Pathway. Acta Biochim. Pol. 2018, 65, 35–41. [Google Scholar] [CrossRef]
- Alkhalidy, H.; Moore, W.; Wang, Y.; Luo, J.; McMillan, R.P.; Zhen, W.; Zhou, K.; Liu, D. The Flavonoid Kaempferol Ameliorates Streptozotocin-Induced Diabetes by Suppressing Hepatic Glucose Production. Molecules 2018, 23, 2338. [Google Scholar] [CrossRef]
- Cheng, N.; Ren, N.; Gao, H.; Lei, X.; Zheng, J.; Cao, W. Antioxidant and Hepatoprotective Effects of Schisandra Chinensis Pollen Extract on CCl4-Induced Acute Liver Damage in Mice. Food Chem. Toxicol. 2013, 55, 234–240. [Google Scholar] [CrossRef]
- Malayeri, A.; Badparva, R.; Mombeini, M.A.; Khorsandi, L.; Goudarzi, M. Naringenin: A Potential Natural Remedy against Methotrexate-Induced Hepatotoxicity in Rats. Drug Chem. Toxicol. 2022, 45, 491–498. [Google Scholar] [CrossRef]
- Yang, C.; Li, L.; Ma, Z.; Zhong, Y.; Pang, W.; Xiong, M.; Fang, S.; Li, Y. Hepatoprotective Effect of Methyl Ferulic Acid against Carbon Tetrachloride-Induced Acute Liver Injury in Rats. Exp. Ther. Med. 2018, 15, 2228–2238. [Google Scholar] [CrossRef]
- Owumi, S.; Ajijola, I.; Agbeti, O. Hepatorenal Protective Effects of Protocatechuic Acid in Rats Administered with Anticancer Drug Methotrexate. Hum. Exp. Toxicol. 2019, 38, 1254–1265. [Google Scholar] [CrossRef]
- Bispo, V.S.; Dantas, L.S.; CHAVES, A.B.; Pinto, I.F.; SILVA, R.P.; Otsuka, F.A.; Santos, R.B.; Santos, A.C.; Trindade, D.J.; Matos, H.R. Reduction of the DNA Damages, Hepatoprotective Effect and Antioxidant Potential of the Coconut Water, Ascorbic and Caffeic Acids in Oxidative Stress Mediated by Ethanol. Anais da Academia Brasileira de Ciências 2017, 89, 1095–1109. [Google Scholar] [CrossRef]
- Ebrahimi, R.; Sepand, M.R.; Seyednejad, S.A.; Omidi, A.; Akbariani, M.; Gholami, M.; Sabzevari, O. Ellagic Acid Reduces Methotrexate-Induced Apoptosis and Mitochondrial Dysfunction via up-Regulating Nrf2 Expression and Inhibiting the IĸBα/NFĸB in Rats. DARU J. Pharm. Sci. 2019, 27, 721–733. [Google Scholar] [CrossRef]
- Hassan, S.M.; Khalaf, M.M.; Sadek, S.A.; Abo-Youssef, A.M. Protective Effects of Apigenin and Myricetin against Cisplatin-Induced Nephrotoxicity in Mice. Pharm. Biol. 2017, 55, 766–774. [Google Scholar] [CrossRef]
- Said, S.E.; Eldin, H.O.H.; Meghawry, E.K.A.E. Renoprotective effects of naringenin and olive oil against cyclosporine- induced nephrotoxicity in rats. Iran. J. Toxicol. 2016, 10, 27–37. [Google Scholar]
- Chowdhury, S.; Ghosh, S.; Das, A.K.; Sil, P.C. Ferulic Acid Protects Hyperglycemia-Induced Kidney Damage by Regulating Oxidative Insult, Inflammation and Autophagy. Front. Pharmacol. 2019, 10, 27. [Google Scholar] [CrossRef]
- Mohammed, E.T.; Hashem, K.S.; Abdelazem, A.Z.; Foda, F.A.M.A. Prospective Protective Effect of Ellagic Acid as a SIRT1 Activator in Iron Oxide Nanoparticle-Induced Renal Damage in Rats. Biol. Trace. Elem. Res. 2020, 198, 177–188. [Google Scholar] [CrossRef]
- Promsan, S.; Jaikumkao, K.; Pongchaidecha, A.; Chattipakorn, N.; Chatsudthipong, V.; Arjinajarn, P.; Pompimon, W.; Lungkaphin, A. Pinocembrin Attenuates Gentamicin-Induced Nephrotoxicity in Rats. Can. J. Physiol. Pharmacol. 2016, 94, 808–818. [Google Scholar] [CrossRef]
- Shanmugam, K.R.; Shanmugam, B.; Subbaiah, G.V.; Ravi, S.; Reddy, K.S. Medicinal Plants and Bioactive Compounds for Diabetes Management: Important Advances in Drug Discovery. Curr. Pharm. Des. 2021, 27, 763–774. [Google Scholar] [CrossRef]
- Shin, K.-M.; Kim, I.-T.; Park, Y.-M.; Ha, J.; Choi, J.-W.; Park, H.-J.; Lee, Y.S.; Lee, K.-T. Anti-Inflammatory Effect of Caffeic Acid Methyl Ester and Its Mode of Action through the Inhibition of Prostaglandin E2, Nitric Oxide and Tumor Necrosis Factor-α Production. Biochem. Pharmacol. 2004, 68, 2327–2336. [Google Scholar] [CrossRef]
- Bumrungpert, A.; Lilitchan, S.; Tuntipopipat, S.; Tirawanchai, N.; Komindr, S. Ferulic Acid Supplementation Improves Lipid Profiles, Oxidative Stress, and Inflammatory Status in Hyperlipidemic Subjects: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. Nutrients 2018, 10, 713. [Google Scholar] [CrossRef]
- Abozaid, O.A.R.; Moawed, F.S.M.; Ahmed, E.S.A.; Ibrahim, Z.A. Cinnamic Acid Nanoparticles Modulate Redox Signal and Inflammatory Response in Gamma Irradiated Rats Suffering from Acute Pancreatitis. Biochim. et Biophys. Acta (BBA)-Mol. Basis Dis. 2020, 1866, 165904. [Google Scholar] [CrossRef]
- Khanal, L.; Yadav, P.; Baral, P.; Shah, R.; Rauniar, G.P. Effect of Local Bee Honey on Dihydrofolate Reductase Enzyme Inhibitor-Induced Mucositis: A Histological Study on Albino Wistar Rats. Indian J. Dent. Res. 2019, 30, 708. [Google Scholar] [CrossRef]
- Choi, M.-J.; Lee, E.-J.; Park, J.-S.; Kim, S.-N.; Park, E.-M.; Kim, H.-S. Anti-Inflammatory Mechanism of Galangin in Lipopolysaccharide-Stimulated Microglia: Critical Role of PPAR-γ Signaling Pathway. Biochem. Pharmacol. 2017, 144, 120–131. [Google Scholar] [CrossRef]
- Li, Z.; Chu, S.; He, W.; Zhang, Z.; Liu, J.; Cui, L.; Yan, X.; Li, D.; Chen, N. A20 as a Novel Target for the Anti-Neuroinflammatory Effect of Chrysin via Inhibition of NF-ΚB Signaling Pathway. Brain Behav. Immun. 2019, 79, 228–235. [Google Scholar] [CrossRef]
- Chen, X.; Lu, J.; An, M.; Ma, Z.; Zong, H.; Yang, J. Anti-inflammatory Effect of Resveratrol on Adjuvant Arthritis Rats with Abnormal Immunological Function via the Reduction of Cyclooxygenase-2 and Prostaglandin E2. Mol. Med. Rep. 2014, 9, 2592–2598. [Google Scholar] [CrossRef]
- Comalada, M.; Camuesco, D.; Sierra, S.; Ballester, I.; Xaus, J.; Gálvez, J.; Zarzuelo, A. In Vivo Quercitrin Anti-Inflammatory Effect Involves Release of Quercetin, Which Inhibits Inflammation through down-Regulation of the NF-ΚB Pathway. Eur. J. Immunol. 2005, 35, 584–592. [Google Scholar] [CrossRef]
- Park, M.J.; Lee, E.K.; Heo, H.-S.; Kim, M.-S.; Sung, B.; Kim, M.K.; Lee, J.; Kim, N.D.; Anton, S.; Choi, J.S.; et al. The Anti-Inflammatory Effect of Kaempferol in Aged Kidney Tissues: The Involvement of Nuclear Factor-ΚB via Nuclear Factor-Inducing Kinase/IκB Kinase and Mitogen-Activated Protein Kinase Pathways. J. Med. Food 2009, 12, 351–358. [Google Scholar] [CrossRef]
- Kelainy, E.G.; Ibrahim Laila, I.M.; Ibrahim, S.R. The Effect of Ferulic Acid against Lead-Induced Oxidative Stress and DNA Damage in Kidney and Testes of Rats. Environ. Sci. Pollut. Res. 2019, 26, 31675–31684. [Google Scholar] [CrossRef]
- Karimi, M.Y.; Fatemi, I.; Kalantari, H.; Mombeini, M.A.; Mehrzadi, S.; Goudarzi, M. Ellagic Acid Prevents Oxidative Stress, Inflammation, and Histopathological Alterations in Acrylamide-Induced Hepatotoxicity in Wistar Rats. J. Diet. Suppl. 2020, 17, 651–662. [Google Scholar] [CrossRef]
- Feng, H.; Cao, J.; Zhang, G.; Wang, Y. Kaempferol Attenuates Cardiac Hypertrophy via Regulation of ASK1/MAPK Signaling Pathway and Oxidative Stress. Planta Med. 2017, 83, 837–845. [Google Scholar] [CrossRef]
- Aloud, A.A.; Veeramani, C.; Govindasamy, C.; Alsaif, M.A.; El Newehy, A.S.; Al-Numair, K.S. Galangin, a Dietary Flavonoid, Improves Antioxidant Status and Reduces Hyperglycemia-Mediated Oxidative Stress in Streptozotocin-Induced Diabetic Rats. Redox Rep. 2017, 22, 290–300. [Google Scholar] [CrossRef]
- El Khashab, I.H.; Abdelsalam, R.M.; Elbrairy, A.I.; Attia, A.S. Chrysin Attenuates Global Cerebral Ischemic Reperfusion Injury via Suppression of Oxidative Stress, Inflammation and Apoptosis. Biomed. Pharmacother. 2019, 112, 108619. [Google Scholar] [CrossRef]
- Al Olayan, E.M.; Aloufi, A.S.; AlAmri, O.D.; El-Habit, O.H.; Abdel Moneim, A.E. Protocatechuic Acid Mitigates Cadmium-Induced Neurotoxicity in Rats: Role of Oxidative Stress, Inflammation and Apoptosis. Sci. Total Environ. 2020, 723, 137969. [Google Scholar] [CrossRef]
- Pan, X.; Shao, Y.; Wang, F.; Cai, Z.; Liu, S.; Xi, J.; He, R.; Zhao, Y.; Zhuang, R. Protective Effect of Apigenin Magnesium Complex on H2O2-Induced Oxidative Stress and Inflammatory Responses in Rat Hepatic Stellate Cells. Pharm. Biol. 2020, 58, 553–560. [Google Scholar] [CrossRef]
- Han, X.-Y.; Du, W.-L.; Huang, Q.-C.; Xu, Z.-R.; Wang, Y.-Z. Changes in Small Intestinal Morphology and Digestive Enzyme Activity with Oral Administration of Copper-Loaded Chitosan Nanoparticles in Rats. Biol. Trace. Elem. Res. 2012, 145, 355–360. [Google Scholar] [CrossRef]
- Chen, L.; Gnanaraj, C.; Arulselvan, P.; El-Seedi, H.; Teng, H. A Review on Advanced Microencapsulation Technology to Enhance Bioavailability of Phenolic Compounds: Based on Its Activity in the Treatment of Type 2 Diabetes. Trends Food Sci. Technol. 2019, 85, 149–162. [Google Scholar] [CrossRef]
- Ramar, M.; Manikandan, B.; Raman, T.; Priyadarsini, A.; Palanisamy, S.; Velayudam, M.; Munusamy, A.; Marimuthu Prabhu, N.; Vaseeharan, B. Protective Effect of Ferulic Acid and Resveratrol against Alloxan-Induced Diabetes in Mice. Eur. J. Pharmacol. 2012, 690, 226–235. [Google Scholar] [CrossRef]
- Fatima, N.; Hafizur, R.M.; Hameed, A.; Ahmed, S.; Nisar, M.; Kabir, N. Ellagic Acid in Emblica Officinalis Exerts Anti-Diabetic Activity through the Action on β-Cells of Pancreas. Eur. J. Nutr. 2017, 56, 591–601. [Google Scholar] [CrossRef]
- Shi, G.-J.; Li, Y.; Cao, Q.-H.; Wu, H.-X.; Tang, X.-Y.; Gao, X.-H.; Yu, J.-Q.; Chen, Z.; Yang, Y. In Vitro and in Vivo Evidence That Quercetin Protects against Diabetes and Its Complications: A Systematic Review of the Literature. Biomed. Pharmacother. 2019, 109, 1085–1099. [Google Scholar] [CrossRef]
- Sharma, D.; Kumar Tekade, R.; Kalia, K. Kaempferol in Ameliorating Diabetes-Induced Fibrosis and Renal Damage: An in Vitro and in Vivo Study in Diabetic Nephropathy Mice Model. Phytomedicine 2020, 76, 153235. [Google Scholar] [CrossRef]
- Vinayagam, R.; Xu, B. Antidiabetic Properties of Dietary Flavonoids: A Cellular Mechanism Review. Nutr. Metab. 2015, 12, 60. [Google Scholar] [CrossRef]
- El-Sonbaty, Y.A.; Suddek, G.M.; Megahed, N.; Gameil, N.M. Protocatechuic Acid Exhibits Hepatoprotective, Vasculoprotective, Antioxidant and Insulin-like Effects in Dexamethasone-Induced Insulin-Resistant Rats. Biochimie 2019, 167, 119–134. [Google Scholar] [CrossRef]
- Hossain, C.M.; Ghosh, M.K.; Satapathy, B.S.; Dey, N.S.; Mukherjee, B. Apigenin Causes Biochemical Modulation, GLUT4 and Cd38 Alterations to Improve Diabetes and to Protect Damages of Some Vital Organs in Experimental Diabetes. Am. J. Pharmacol. Toxicol. 2014, 9, 39–52. [Google Scholar] [CrossRef]
- Zhang, M.; He, L.; Liu, J.; Zhou, L. Luteolin Attenuates Diabetic Nephropathy through Suppressing Inflammatory Response and Oxidative Stress by Inhibiting STAT3 Pathway. Exp. Clin. Endocrinol. Diabetes 2021, 129, 729–739. [Google Scholar] [CrossRef]
- Ren, B.; Qin, W.; Wu, F.; Wang, S.; Pan, C.; Wang, L.; Zeng, B.; Ma, S.; Liang, J. Apigenin and Naringenin Regulate Glucose and Lipid Metabolism, and Ameliorate Vascular Dysfunction in Type 2 Diabetic Rats. Eur. J. Pharmacol. 2016, 773, 13–23. [Google Scholar] [CrossRef]
- Pei, B.; Sun, J. Pinocembrin Alleviates Cognition Deficits by Inhibiting Inflammation in Diabetic Mice. J. Neuroimmunol. 2018, 314, 42–49. [Google Scholar] [CrossRef]
- Aref, A.; Tohamy, A.; E Abdel Moneim, A.; Sayed, R. Cinnamic Acid Attenuates Cisplatin-Induced Hepatotoxicity and Nephrotoxicity. J. Basic Environ. Sci. 2016, 1–9. [Google Scholar]
- Zhao, L.; Chen, F.; Zhang, Y.; Yue, L.; Guo, H.; Ye, G.; Shi, F.; Lv, C.; Jing, B.; Tang, H.; et al. Involvement of P450s and Nuclear Receptors in the Hepatoprotective Effect of Quercetin on Liver Injury by Bacterial Lipopolysaccharide. Immunopharmacol. Immunotoxicol. 2020, 42, 211–220. [Google Scholar] [CrossRef]
- Wang, M.; Sun, J.; Jiang, Z.; Xie, W.; Zhang, X. Hepatoprotective Effect of Kaempferol Against Alcoholic Liver Injury in Mice. Am. J. Chin. Med. 2015, 43, 241–254. [Google Scholar] [CrossRef]
- Aladaileh, S.H.; Abukhalil, M.H.; Saghir, S.A.M.; Hanieh, H.; Alfwuaires, M.A.; Almaiman, A.A.; Bin-Jumah, M.; Mahmoud, A.M. Galangin Activates Nrf2 Signaling and Attenuates Oxidative Damage, Inflammation, and Apoptosis in a Rat Model of Cyclophosphamide-Induced Hepatotoxicity. Biomolecules 2019, 9, 346. [Google Scholar] [CrossRef]
- Pingili, R.B.; Pawar, A.K.; Challa, S.R.; Kodali, T.; Koppula, S.; Toleti, V. A Comprehensive Review on Hepatoprotective and Nephroprotective Activities of Chrysin against Various Drugs and Toxic Agents. Chem. -Biol. Interact. 2019, 308, 51–60. [Google Scholar] [CrossRef]
- Rašković, A.; Gigov, S.; Čapo, I.; Kusturica, M.P.; Milijašević, B.; Kojić-Damjanov, S.; Martić, N. Antioxidative and Protective Actions of Apigenin in a Paracetamol-Induced Hepatotoxicity Rat Model. Eur. J. Drug Metab. Pharmacokinet. 2017, 42, 849–856. [Google Scholar] [CrossRef]
- Yan, Y.; Jun, C.; Lu, Y.; Jiangmei, S. Combination of Metformin and Luteolin Synergistically Protects Carbon Tetrachloride-Induced Hepatotoxicity: Mechanism Involves Antioxidant, Anti-Inflammatory, Antiapoptotic, and Nrf2/HO-1 Signaling Pathway. BioFactors 2019, 45, 598–606. [Google Scholar] [CrossRef]
- Reddy, M.K.; Reddy, A.G.; Kumar, B.K.; Madhuri, D.; Boobalan, G.; Reddy, M.A. Protective Effect of Rutin in Comparison to Silymarin against Induced Hepatotoxicity in Rats. Vet World 2017, 10, 74–80. [Google Scholar] [CrossRef]
- Wang, Y.; Jiang, Y.; Fan, X.; Tan, H.; Zeng, H.; Wang, Y.; Chen, P.; Huang, M.; Bi, H. Hepato-Protective Effect of Resveratrol against Acetaminophen-Induced Liver Injury Is Associated with Inhibition of CYP-Mediated Bioactivation and Regulation of SIRT1–P53 Signaling Pathways. Toxicol. Lett. 2015, 236, 82–89. [Google Scholar] [CrossRef]
- Olayinka, E.T.; Ola, O.S.; Ore, A.; Adeyemo, O.A. Ameliorative Effect of Caffeic Acid on Capecitabine-Induced Hepatic and Renal Dysfunction: Involvement of the Antioxidant Defence System. Medicines 2017, 4, 78. [Google Scholar] [CrossRef]
- El-Sayed, E.-S.M.; Abd El-Raouf, O.M.; Fawzy, H.M.; Manie, M.F. Comparative Study of the Possible Protective Effects of Cinnamic Acid and Cinnamaldehyde on Cisplatin-Induced Nephrotoxicity in Rats. J. Biochem. Mol. Toxicol. 2013, 27, 508–514. [Google Scholar] [CrossRef]
- Kocahan, S.; Dogan, Z.; Erdemli, M.E.; Taskin, E. Protective Effect of Quercetin Against Oxidative Stressinduced Toxicity Associated With Doxorubicin and Cyclophosphamide in Rat Kidney and Liver Tissue. Iran. J. Kidney Dis. 2017, 11, 124. [Google Scholar]
- Vijayaprakash, S.; Langeswaran, K.; Gowtham Kumar, S.; Revathy, R.; Balasubramanian, M.P. Nephro-Protective Significance of Kaempferol on Mercuric Chloride Induced Toxicity in Wistar Albino Rats. Biomed. Aging Pathol. 2013, 3, 119–124. [Google Scholar] [CrossRef]
- Farkhondeh, T.; Samarghandian, S.; Roshanravan, B. Impact of Chrysin on the Molecular Mechanisms Underlying Diabetic Complications. J. Cell. Physiol. 2019, 234, 17144–17158. [Google Scholar] [CrossRef]
- Chen, L.; Tian, G.; Tang, W.; Luo, W.; Liu, P.; Ma, Z. Protective Effect of Luteolin on Streptozotocin-Induced Diabetic Renal Damage in Mice via the Regulation of RIP140/NF-KB Pathway and Insulin Signalling Pathway. J. Funct. Foods 2016, 22, 93–100. [Google Scholar] [CrossRef]
- Wang, B.; Liu, D.; Zhu, Q.; Li, M.; Chen, H.; Guo, Y.; Fan, L.; Yue, L.; Li, L.; Zhao, M. Rutin Ameliorates Kidney Interstitial Fibrosis in Rats with Obstructive Nephropathy. Int. Immunopharmacol. 2016, 35, 77–84. [Google Scholar] [CrossRef]
- Xue, H.-Y.; Yuan, L.; Cao, Y.-J.; Fan, Y.-P.; Chen, X.-L.; Huang, X.-Z. Resveratrol Ameliorates Renal Injury in Spontaneously Hypertensive Rats by Inhibiting Renal Micro-Inflammation. Biosci. Rep. 2016, 36, e00339. [Google Scholar] [CrossRef]
- Mansouri, M.T.; Hemmati, A.A.; Naghizadeh, B.; Mard, S.A.; Rezaie, A.; Ghorbanzadeh, B. A Study of the Mechanisms Underlying the Anti-Inflammatory Effect of Ellagic Acid in Carrageenan-Induced Paw Edema in Rats. Indian J. Pharm. 2015, 47, 292–298. [Google Scholar] [CrossRef]
- Lin, C.-Y.; Huang, C.-S.; Huang, C.-Y.; Yin, M.-C. Anticoagulatory, Antiinflammatory, and Antioxidative Effects of Protocatechuic Acid in Diabetic Mice. J. Agric. Food Chem. 2009, 57, 6661–6667. [Google Scholar] [CrossRef]
- Darabi, P.; Khazali, H.; Mehrabani Natanzi, M. Therapeutic Potentials of the Natural Plant Flavonoid Apigenin in Polycystic Ovary Syndrome in Rat Model: Via Modulation of pro-Inflammatory Cytokines and Antioxidant Activity. Gynecol. Endocrinol. 2020, 36, 582–587. [Google Scholar] [CrossRef]
- Xiong, J.; Wang, K.; Yuan, C.; Xing, R.; Ni, J.; Hu, G.; Chen, F.; Wang, X. Luteolin Protects Mice from Severe Acute Pancreatitis by Exerting HO-1-Mediated Anti-Inflammatory and Antioxidant Effects. Int. J. Mol. Med. 2017, 39, 113–125. [Google Scholar] [CrossRef] [PubMed]
- Alhoshani, A.R.; Hafez, M.M.; Husain, S.; Al-sheikh, A.M.; Alotaibi, M.R.; Al Rejaie, S.S.; Alshammari, M.A.; Almutairi, M.M.; Al-Shabanah, O.A. Protective Effect of Rutin Supplementation against Cisplatin-Induced Nephrotoxicity in Rats. BMC Nephrol. 2017, 18, 194. [Google Scholar] [CrossRef]
- Fan, R.; Pan, T.; Zhu, A.-L.; Zhang, M.-H. Anti-Inflammatory and Anti-Arthritic Properties of Naringenin via Attenuation of NF-ΚB and Activation of the Heme Oxygenase (HO)-1/Related Factor 2 Pathway. Pharmacol. Rep. 2017, 69, 1021–1029. [Google Scholar] [CrossRef] [PubMed]
- Gu, X.; Zhang, Q.; Du, Q.; Shen, H.; Zhu, Z. Pinocembrin Attenuates Allergic Airway Inflammation via Inhibition of NF-ΚB Pathway in Mice. Int. Immunopharmacol. 2017, 53, 90–95. [Google Scholar] [CrossRef]
- Fazayeli-Rad, A.R.; Afzali, N.; Farhangfar, H.; Asghari, M.R. Effect of Bee Pollen on Growth Performance, Intestinal Morphometry and Immune Status of Broiler Chicks. Eur. Poult. Sci. 2015, 79, 86. [Google Scholar] [CrossRef]
- Münstedt, K.; Voss, B.; Kullmer, U.; Schneider, U.; Hübner, J. Bee Pollen and Honey for the Alleviation of Hot Flushes and Other Menopausal Symptoms in Breast Cancer Patients. Mol. Clin. Oncol. 2015, 3, 869–874. [Google Scholar] [CrossRef]
- Yan, S.; Wang, K.; Wang, X.; Ou, A.; Wang, F.; Wu, L.; Xue, X. Effect of Fermented Bee Pollen on Metabolic Syndrome in High-Fat Diet-Induced Mice. Food Sci. Hum. Wellness 2021, 10, 345–355. [Google Scholar] [CrossRef]
- Kurek-Górecka, A.; Górecki, M.; Rzepecka-Stojko, A.; Balwierz, R.; Stojko, J. Bee Products in Dermatology and Skin Care. Molecules 2020, 25, 556. [Google Scholar] [CrossRef]
- Xi, X.; Li, J.; Guo, S.; Li, Y.; Xu, F.; Zheng, M.; Cao, H.; Cui, X.; Guo, H.; Han, C. The Potential of Using Bee Pollen in Cosmetics: A Review. J. Oleo Sci. 2018, 67, 1071–1082. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).