Novel Copper-Zinc-Manganese Ternary Metal Oxide Nanocomposite as Heterogeneous Catalyst for Glucose Sensor and Antibacterial Activity
Abstract
1. Introduction
2. Experimentation
2.1. Materials
2.2. Methods
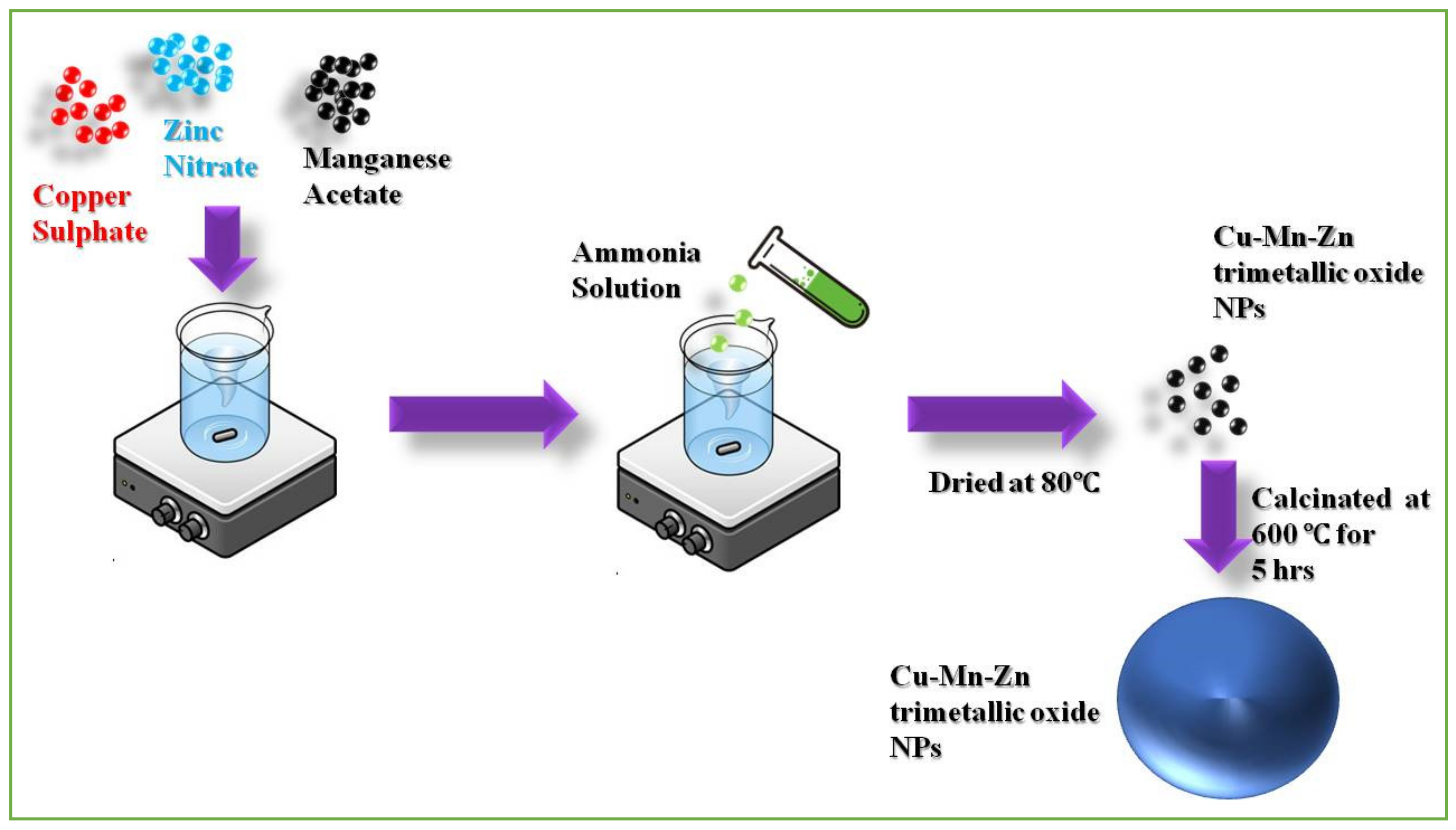
Synthesis of Cu-Zn-Mn Mixed Trimetal Oxide
2.3. Characterization
2.4. Electrochemical Measurement
2.5. Photocatalytic Degradation of Dye Solution
2.6. Antioxidant Activity
DPPH Scavenging Activity
2.7. Antibacterial Activity
3. Results and Discussion
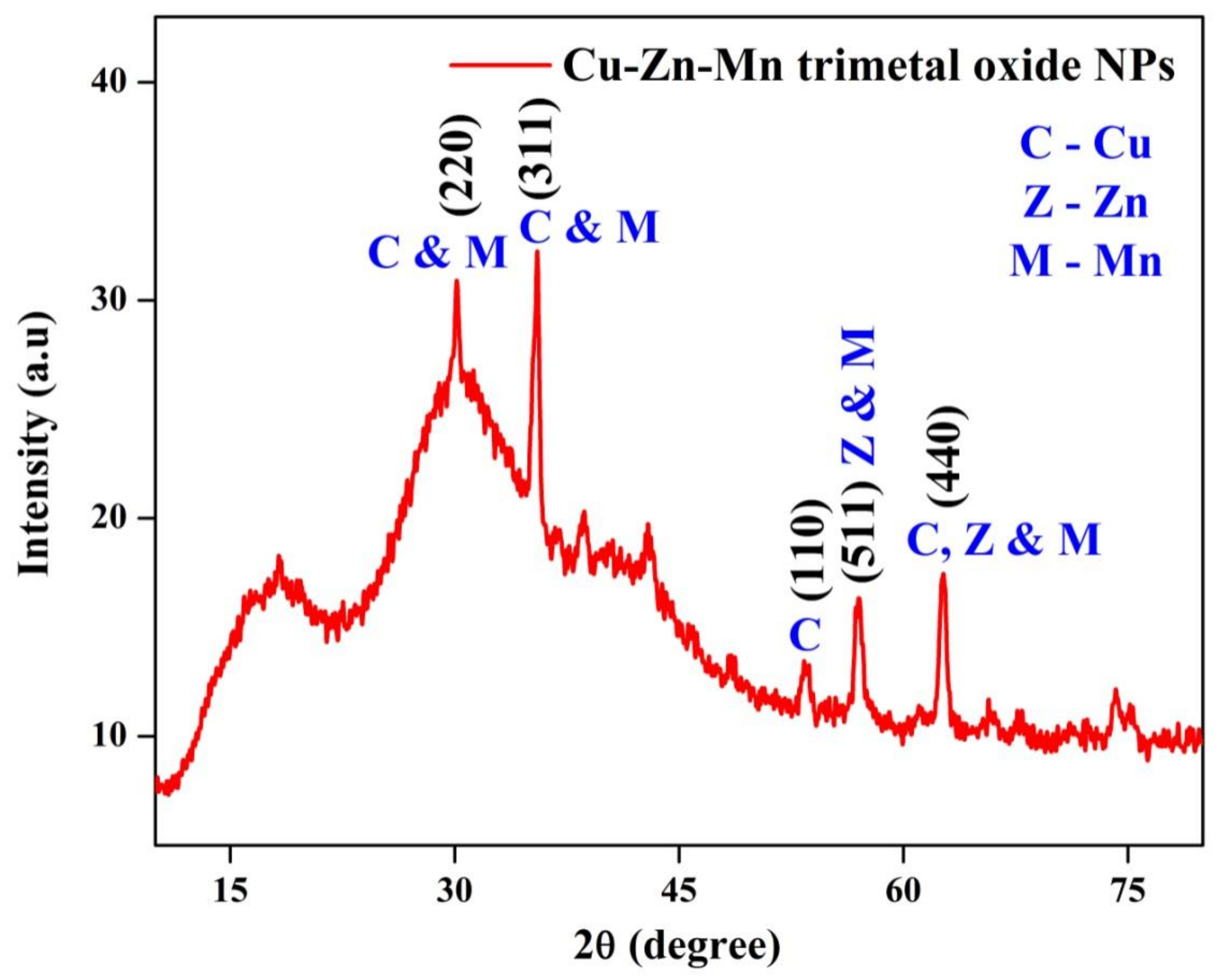
3.1. XRD Analysis
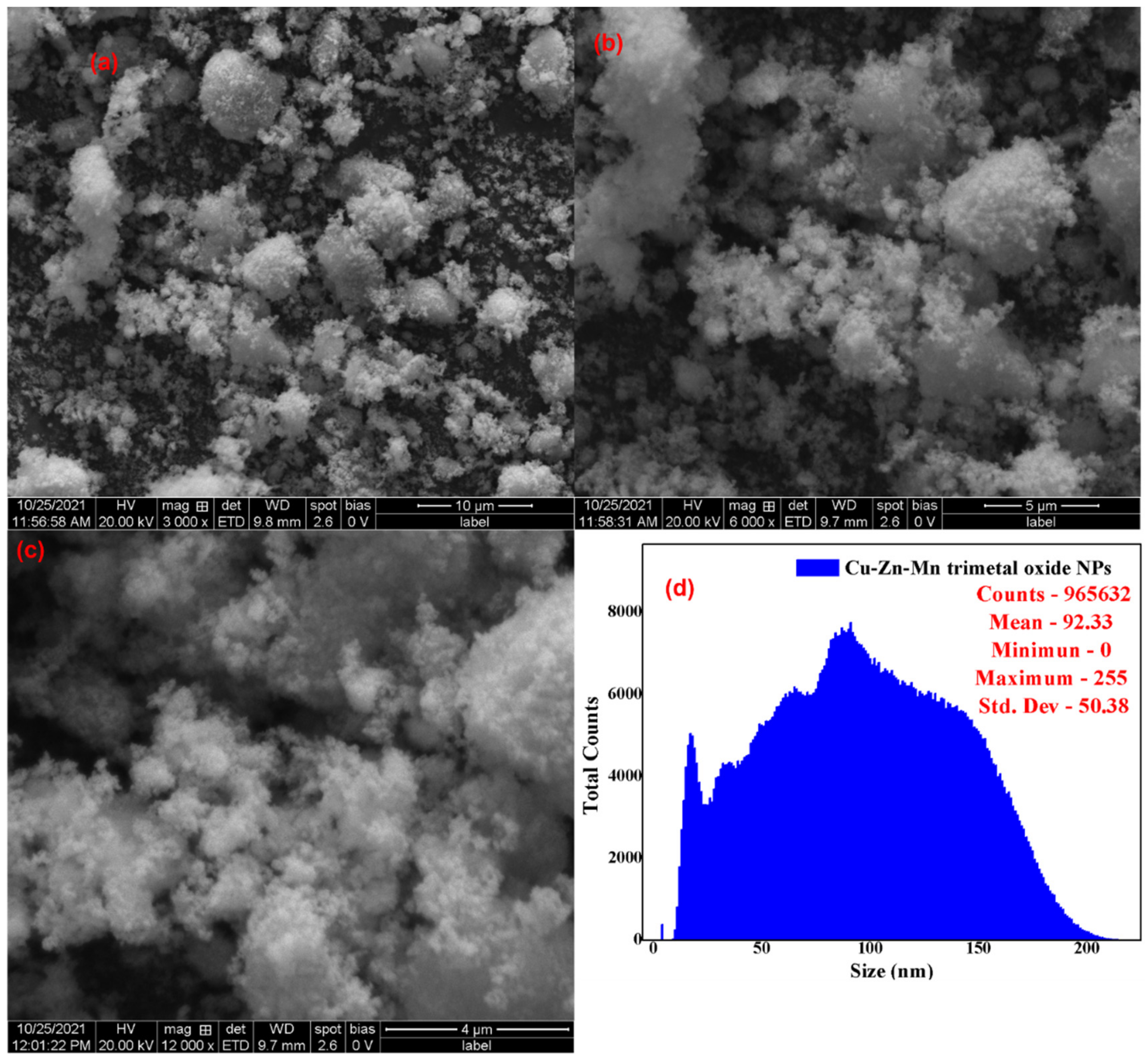
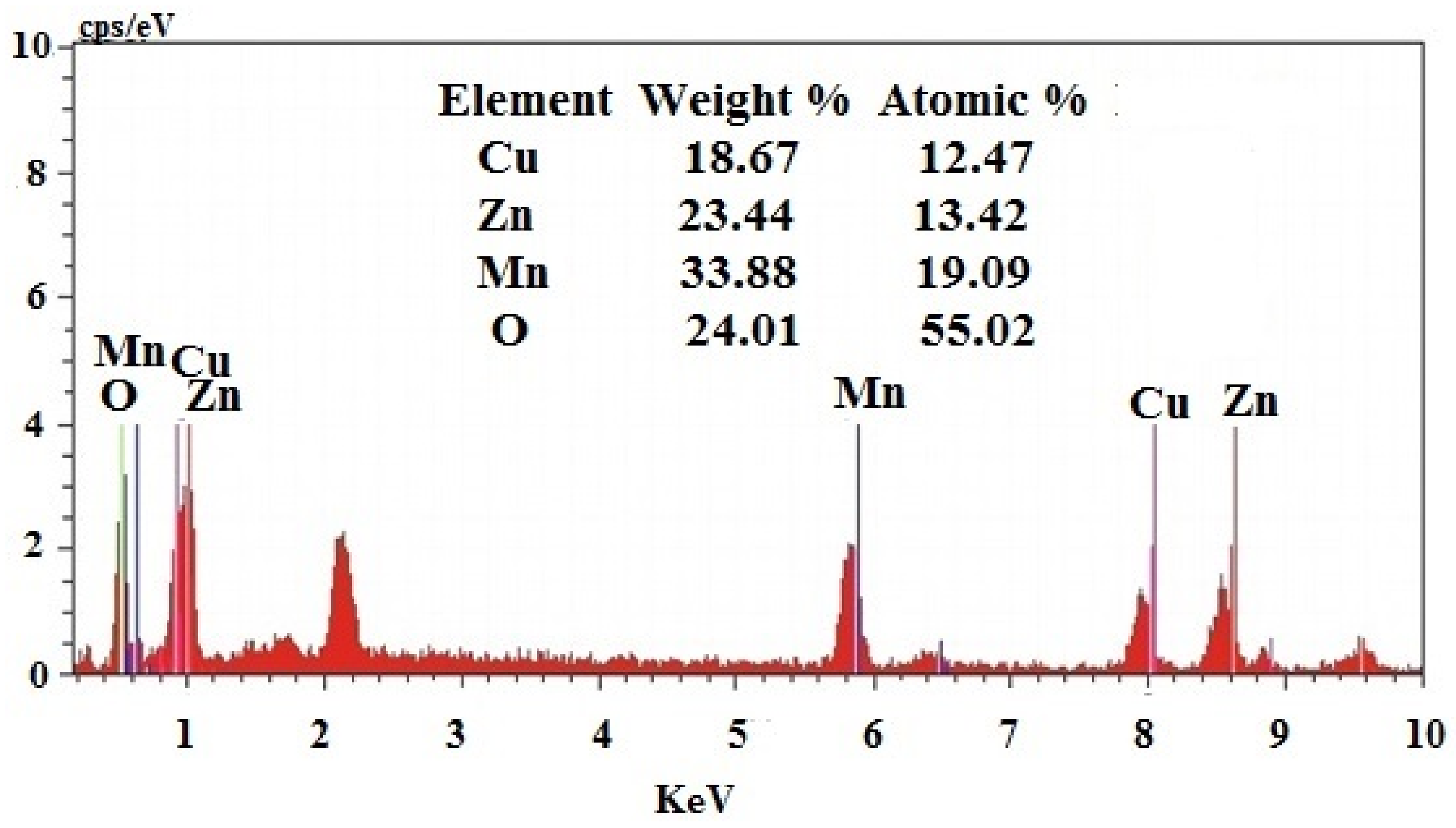
3.2. SEM-EDAX Analysis
3.3. HR-TEM—SAED Analysis
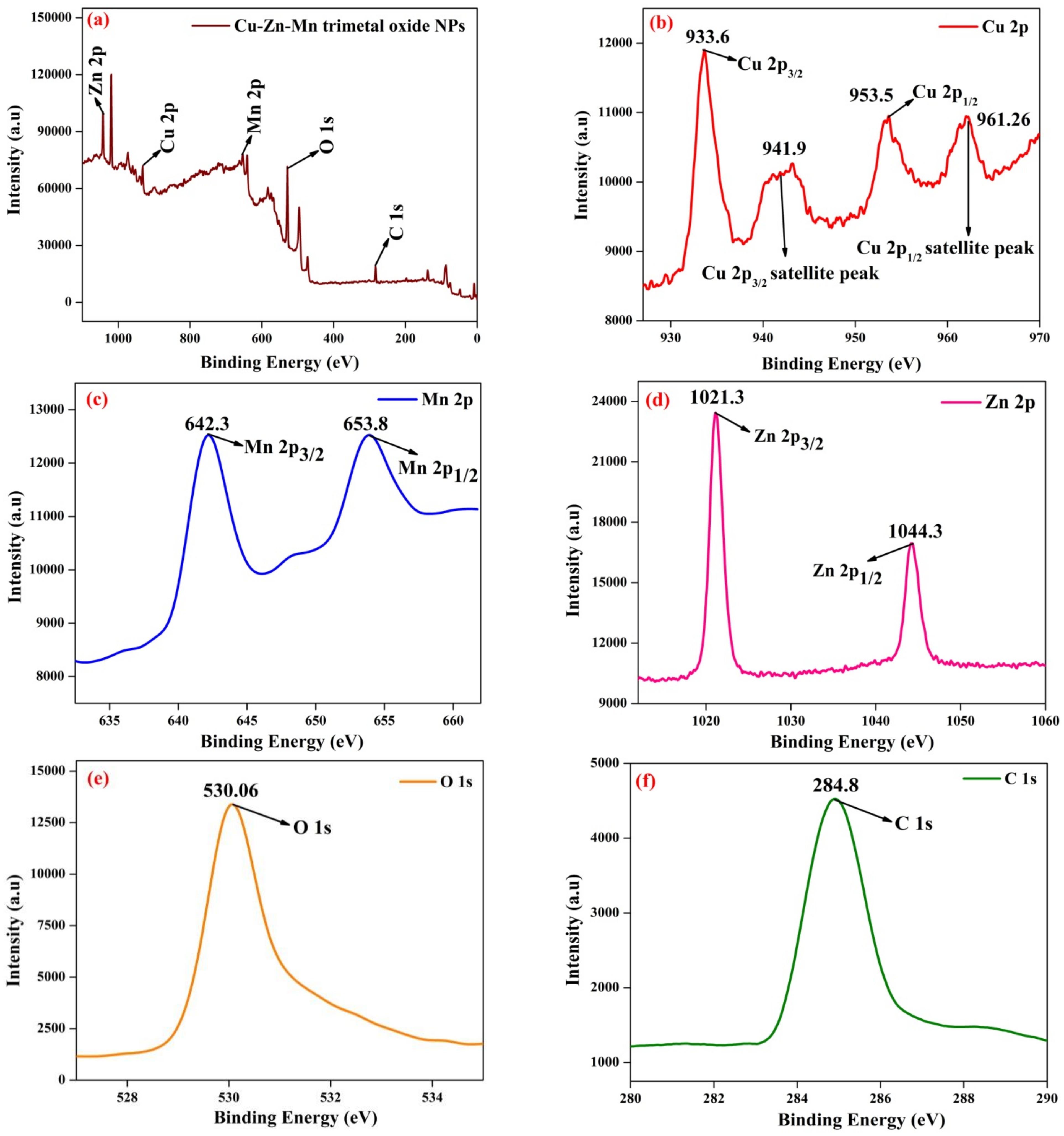
3.4. XPS Analysis
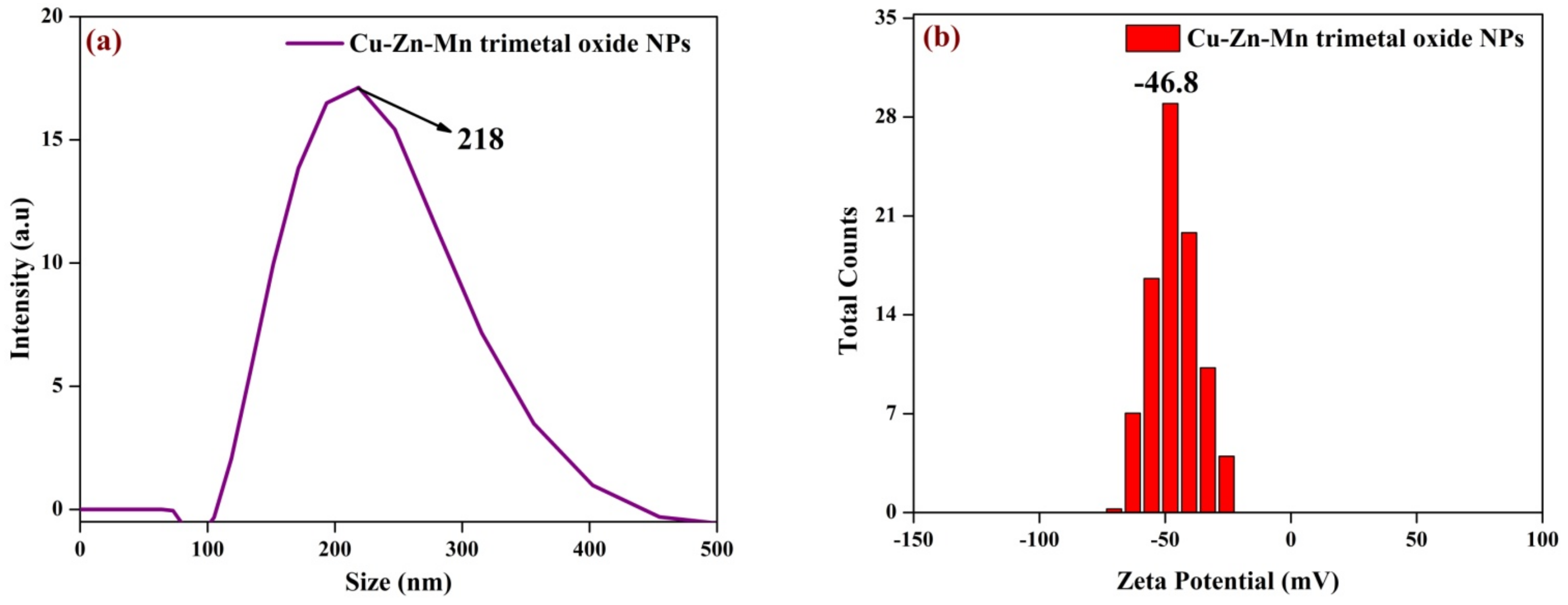
3.5. DLS Analysis
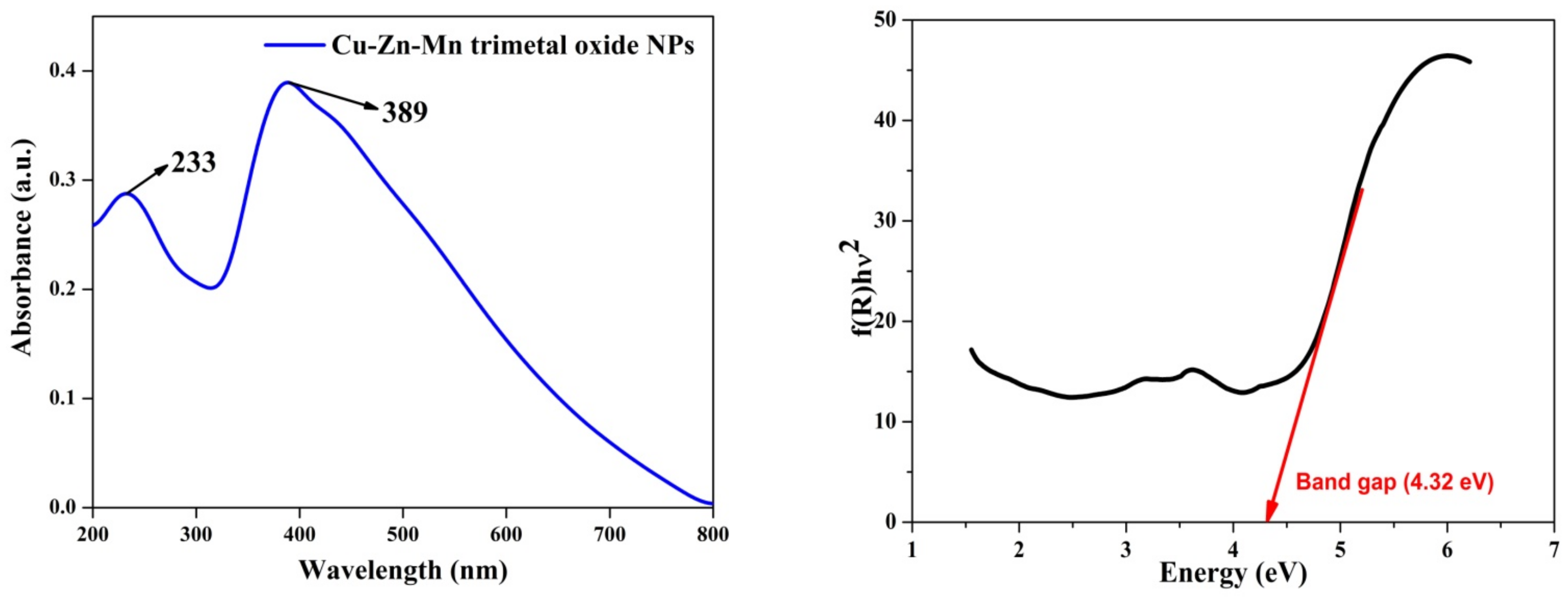
3.6. UV-Vis Analysis
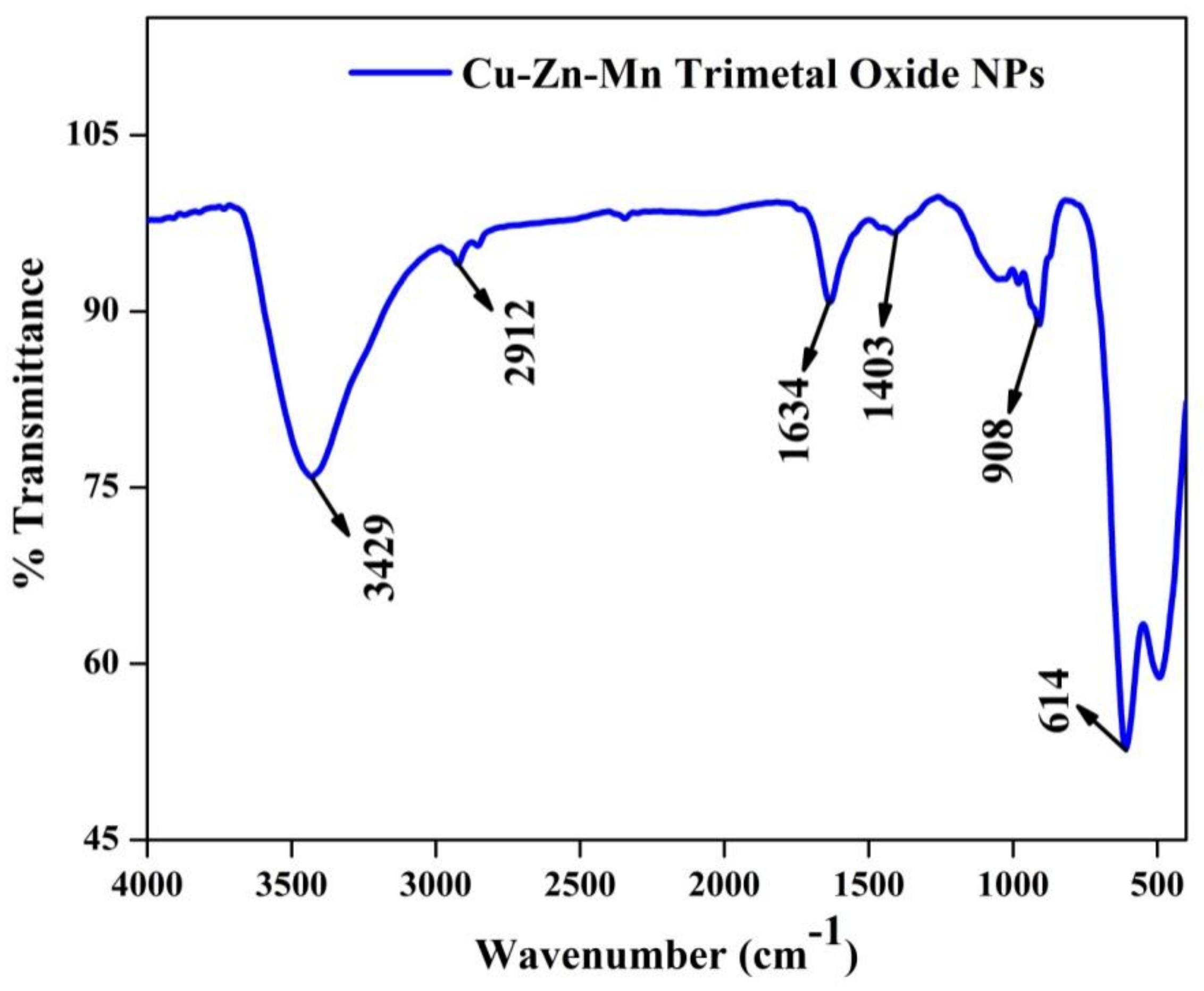
3.7. FT-IR Analysis
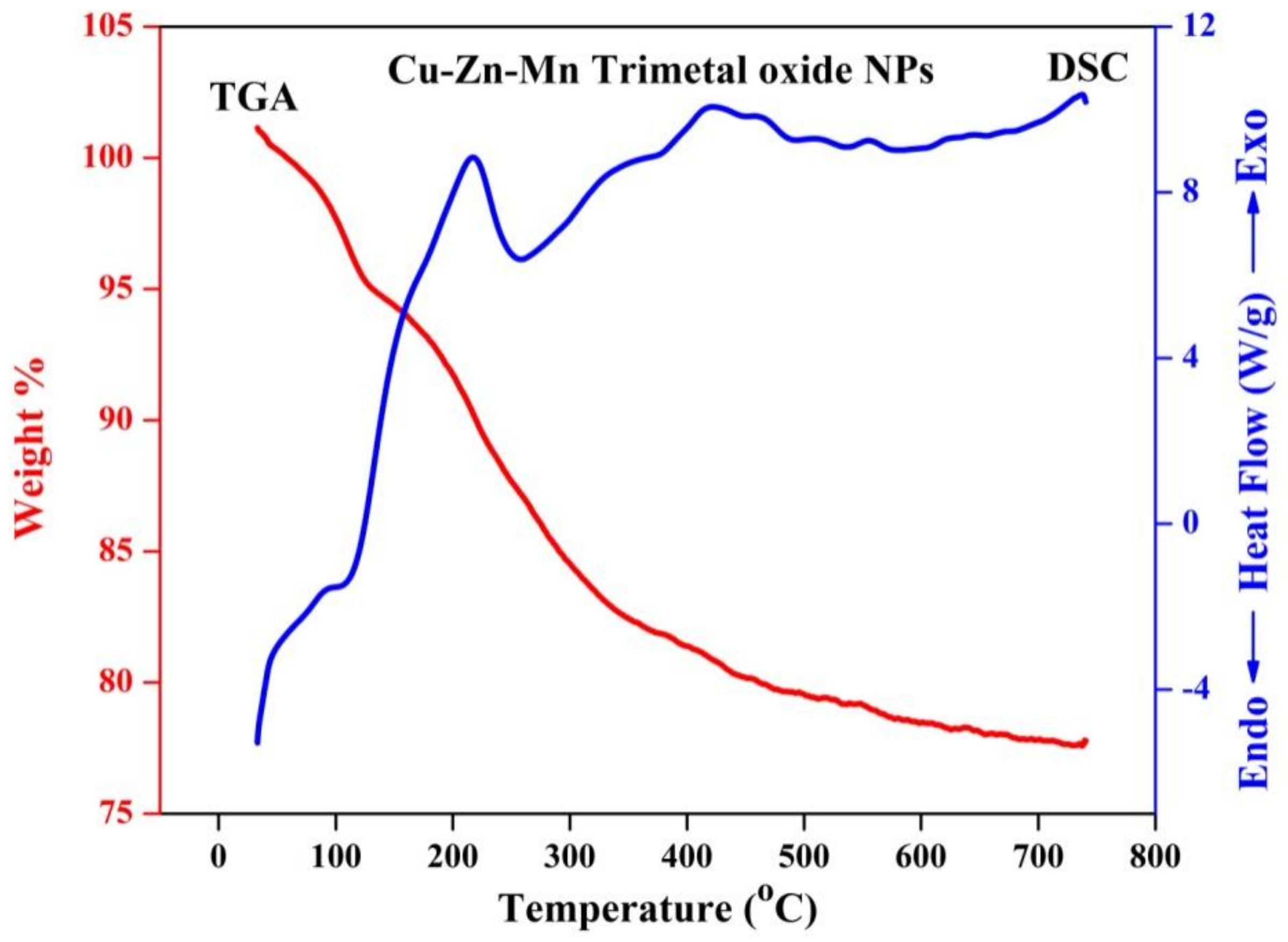
3.8. TG-DSC Analysis
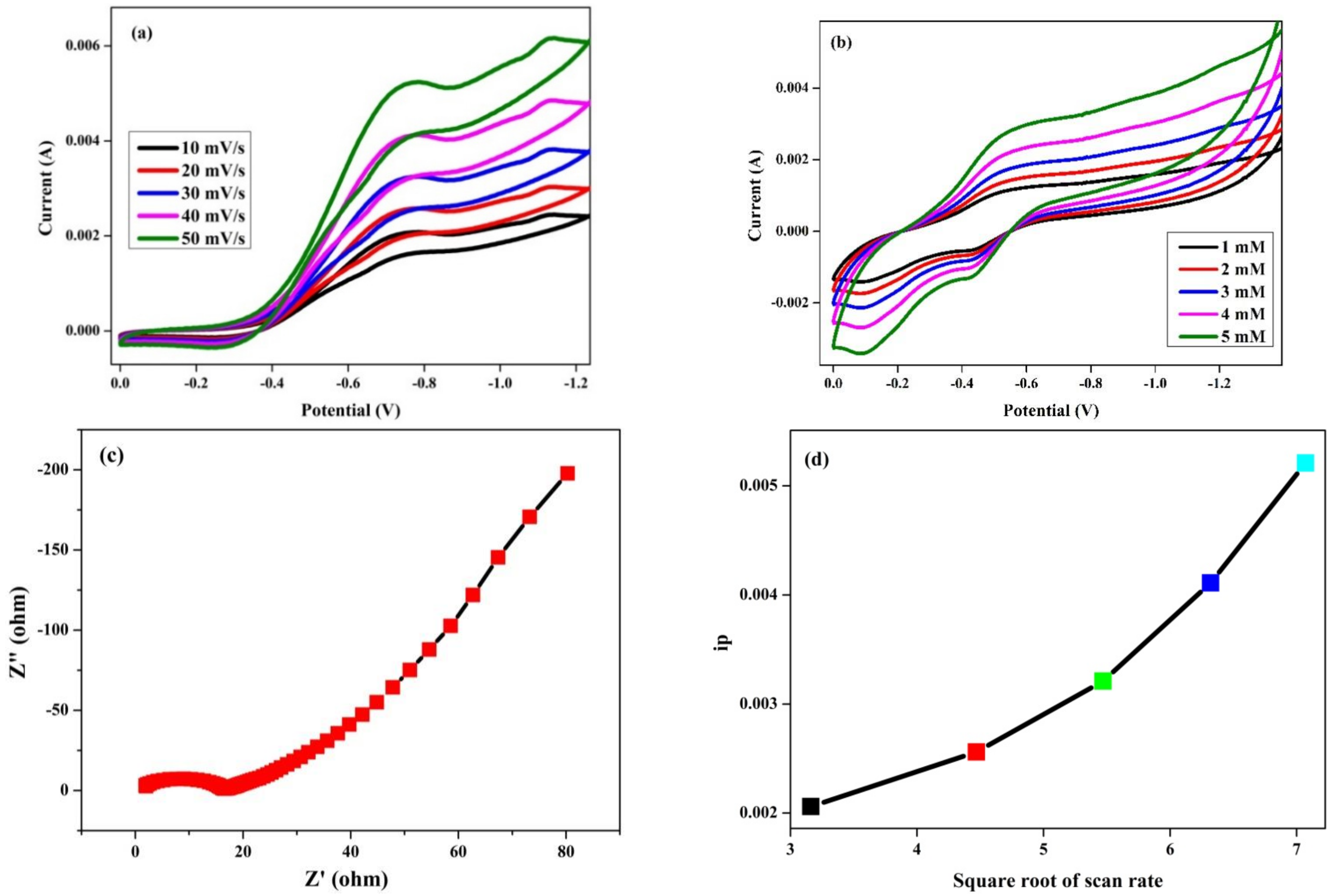
3.9. Glucose Sensor
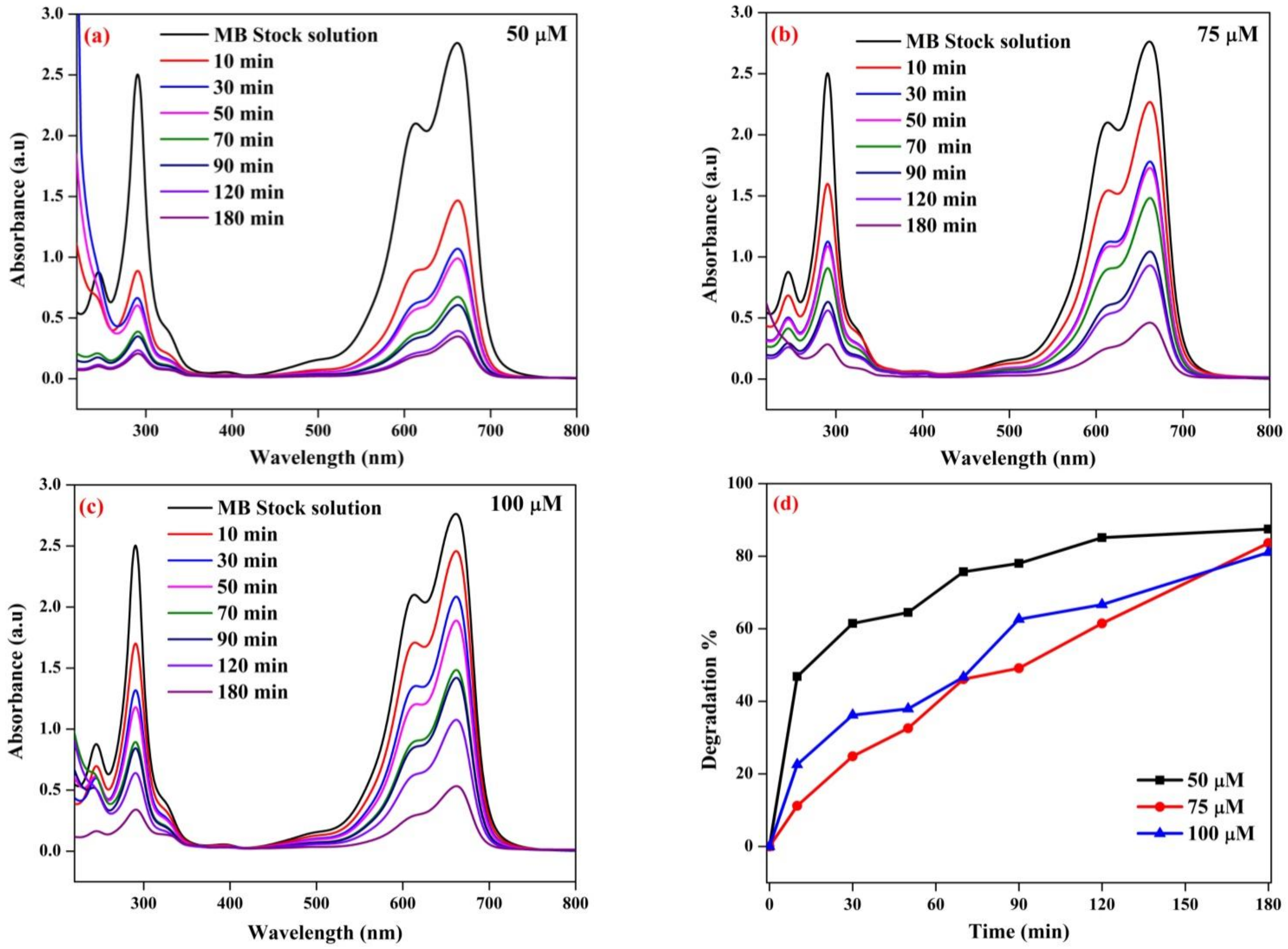
3.10. Photocatalytic Degradation of MB Dye
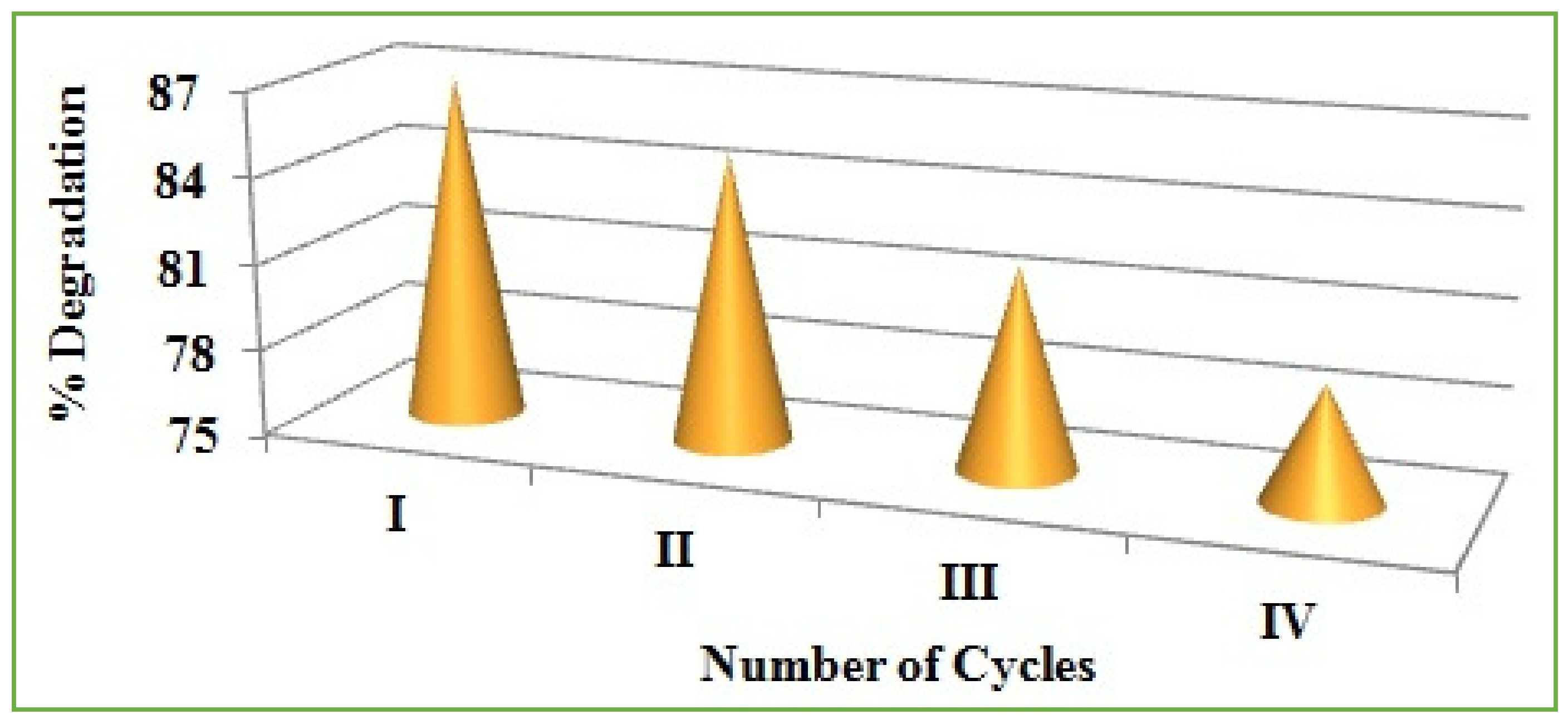
Regeneration and Reusability
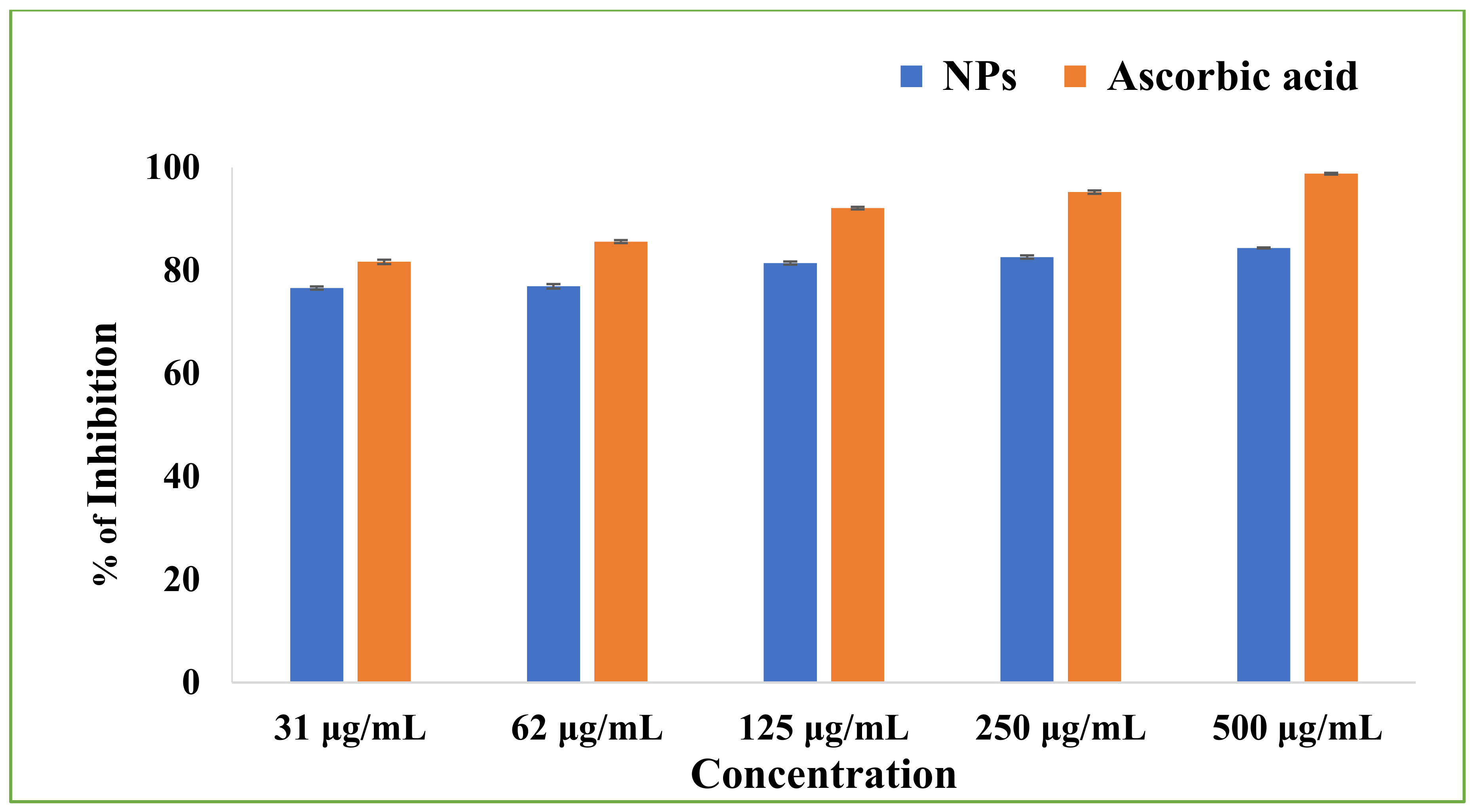
3.11. Antioxidant Activity
3.12. Antibacterial Activity
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hoo, X.F.; Abdul Razak, K.; Ridhuan, N.S.; Mohamad Nor, N.; Zakaria, N.D. Electrochemical glucose biosensor based on ZnO nanorods modified with gold nanoparticles. J. Mater. Sci. Mater. Electron. 2019, 30, 7460–7470. [Google Scholar] [CrossRef]
- Steiner, M.; Duerkop, A.; Wolfbeis, O. Optical methods for sensing glucose. Chem. Soc. Rev. 2011, 40, 4805–4839. [Google Scholar] [CrossRef]
- Coster, S.; Gulliford, M.C.; Seed, P.T.; Powrie, J.K.; Swaminathan, R. Monitoring blood glucose control in diabetes mellitus: A systematic review. Health Technol. Assess. 2000, 4, 1–93. [Google Scholar] [CrossRef]
- Demiate, I.M.; Konkel, F.E.; Pedroso, R.A. Enzymatic determination of starch in doce de leite using dialysis. Food Sci. Technol. 2001, 21, 339–342. [Google Scholar] [CrossRef][Green Version]
- Xavier, J.; Lemaitre, R.N.; Rea, T.D.; Sotoodehnia, N.; Empana, J.; Siscovick, D.S. Diabetes, glucose level, and risk of sudden cardiac death. Eur. Heart J. 2005, 26, 2142–2147. [Google Scholar]
- Mislovicova, D.; Michalkova, E.; Vikartovska, A. Immobilized glucose oxidase on different supports for biotransformation removal of glucose from oligosaccharide mixtures. Process Biochem. 2007, 42, 704–709. [Google Scholar] [CrossRef]
- Beitollahi, H.; Tajik, S.; di Bartolomeo, A. Application of MnO2 nanorod-ionic liquid modified carbon paste electrode for the voltammetric determination of sulfanilamide. Micromachines 2022, 13, 598. [Google Scholar] [CrossRef]
- Tajik, S.; Afshar, A.A.; Shamsaddini, S.; Askari, M.B.; Dourandish, Z.; Nejad, F.G.; Beitollahi, H.; di Bartolomeo, A. Fe3O4@MoS2/rGO nanocomposite/ionic liquid modified carbon paste electrode for electrochemical sensing of Dasatinib in the presence of doxorubicin. Ind. Eng. Chem. Res. 2022. [Google Scholar] [CrossRef]
- Alam, M.W. Electrochemical sensing of dextrose and photocatalytic activities by nickel ferrite nanoparticles synthesized by probe sonication method. Curr. Nanosci. 2021, 17, 893–903. [Google Scholar] [CrossRef]
- Li, M.; Zhao, Z.; Liu, X.; Xiong, Y.; Han, C.; Zhang, Y.; Bo, X.; Guo, L. Novel bamboo leaf shaped CuO nanorod@hollow carbon fibers derived from plant biomass for efficient and non-enzymatic glucose detection. Analyst 2015, 140, 6412–6420. [Google Scholar] [CrossRef]
- Yin, H.; Zhu, J.; Chen, J.; Gong, J.; Nie, Q. Hierarchical CuCo2O4/C microspheres assembled with nanoparticle-stacked nanosheets for sensitive non-enzymatic glucose detection. J. Mater. Sci. 2018, 53, 11951–11961. [Google Scholar] [CrossRef]
- Zhang, D.; Zhang, X.; Bu, Y.; Zhang, J.; Zhang, R. Copper cobalt sulfide structures derived from MOF precursors with enhanced electrochemical glucose sensing properties. Nanomaterials 2022, 12, 1394. [Google Scholar] [CrossRef]
- Dourandish, Z.; Tajik, S.; Beitollahi, H.; Jahani, P.M.; Nejad, F.G.; Sheikhshoaie, I.; di Bartolomeo, A. A comprehensive review of metal organic framework: Synthesis, characterization, and investigation of their application in electrochemical biosensors for biomedical analysis. Sensors 2022, 22, 2238. [Google Scholar] [CrossRef]
- Iqbal, T.; Irfan, F.; Afsheen, S.; Zafar, M.; Naeem, S.; Raza, A. Synthesis and characterization of Ag-TiO2 nanocomposites to study their effect on seed germination. Appl. Nanosci. 2021, 11, 2043–2057. [Google Scholar] [CrossRef]
- Hoseinpour, V.; Ghaemi, N. Novel ZnO-MnO2-Cu2O triple nanocomposite: Facial synthesis, characterization, antibacterial activity and visible light photocatalytic performance for dyes degradation-A comparative study. Mater. Res. Express 2018, 5, 085012. [Google Scholar] [CrossRef]
- Tedla, H.; Diaz, I.; Kebede, T.; Taddesse, A.M. Synthesis, characterization and photocatalytic activity of zeolite supported ZnO/Fe2O3/MnO2 nanocomposites. J. Environ. Chem. Eng. 2015, 3, 1586–1591. [Google Scholar] [CrossRef]
- Pranesh, S.; Nagaraju, J. Nano sized ZnO/MnO2/Gd2O3 ternary heterostructuctures for enhanced photocatalysis. Curr. Nanomater. 2020, 5, 36–46. [Google Scholar] [CrossRef]
- Zou, X.; Dong, Y.; Ke, J.; Ge, H.; Chen, D.; Sun, H.; Cui, Y. Cobalt monoxide/tunsten trioxide p-n heterojunction boosting charge separation for efficient visible-light driven gaseous toluene degradation. Chem. Eng. J. 2020, 400, 125919. [Google Scholar] [CrossRef]
- Sharma, G.; Garica-Penas, A.; Kumar, A.; Naushad, M.; Mola, G.T.; Alshehri, S.M.; Ahmed, J.; Alhokbany, N.; Stadler, F.J. Fe/La/Zn nanocomposite with graphene oxide for photodegradation of phenylhydrazine. J. Mol. Liq. 2019, 285, 362–374. [Google Scholar] [CrossRef]
- Zhang, L.; Jiang, Y.; Ding, Y.; Povey, M.; York, D. Investigation into the antibacterial behaviour of suspensions of ZnO nanoparticles (ZnO nanofluids). J. Nanopart. Res. 2007, 9, 479–489. [Google Scholar] [CrossRef]
- Al-Hazmi, F.; Alnowaiser, F.; Al-Ghamdi, A.A.; Al-Ghamdi, A.A.; Aly, M.M.; Al-Tuwirqi, R.M.; El-Tantawy, F. A new large–scale synthesis of magnesium oxide nanowires: Structural and antibacterial properties. Superlattices Microstruct. 2012, 52, 200–209. [Google Scholar] [CrossRef]
- Lv, M.; Su, S.; He, Y.; Huang, Q.; Hu, W.; Li, D.; Fan, C.; Lee, S.T. Long-term antimicrobial effect of silicon nanowires decorated with silver nanoparticles. Adv. Mater. 2010, 22, 5463–5467. [Google Scholar] [CrossRef]
- Flores-González, M.; Talavera-Rojas, M.; Soriano-Vargas, E.; Rodríguez-González, V. Practical mediated-assembly synthesis of silver nanowires using commercial Camellia sinensis extracts and their antibacterial properties. New J. Chem. 2018, 42, 2133–2139. [Google Scholar] [CrossRef]
- Ringwal, S.; Bartwal, A.S.; Semwal, A.R.; Sati, S.C. Review on green synthesized nanocomposites and their biological activities. J. Mt. Res. 2021, 16, 181–186. [Google Scholar] [CrossRef]
- Moradiya, M.A.; Khiriya, P.K.; Tripathi, G.K.; Bundela, P.; Khare, P.S. The investigation of Microwave assisted greener synthesis of Ag/Ti/Zn trimetallic nanoparticles and carbon quantum dots nanocomposites in the application of solar cell. Lett. Appl. NanoBioSci. 2022, 11, 3102–3110. [Google Scholar]
- Vaseghi, Z.; Tavakoli, O.; Nematollahzadeh, A. Rapid biosynthesis of novel Cu/Cr/Ni trimetallic oxide nanoparticles with antimicrobial activity. J. Environ. Chem. Eng. 2018, 6, 1898–1911. [Google Scholar] [CrossRef]
- Noushin, A.; Varasteh-Moradi, A.; Sayyed-Alangi, S.Z.; Hossaini, Z. Green synthesis and evaluation of antioxidant and antimicrobial activity of new dihydropyrroloazepines: Using bio-Ag/CdO/ZnO@MWCNTs nanocomposites as a reusable organometallic catalyst. Appl. Organimetallic Chem. 2021, 35, 6295. [Google Scholar] [CrossRef]
- Moongraksathum, B.; Shang, J.-Y.; Chen, Y.-W. Photocatalytic Antibacterial Effectiveness of Cu-Doped TiO2 Thin Film Prepared via the Peroxo Sol-Gel Method. Catalysts 2018, 8, 352. [Google Scholar] [CrossRef]
- Rajaramanan, T.; Shanmugaratnam, S.; Gurunanthanan, V.; Yohi, S.; Velauthapillai, D.; Ravirajan, P.; Senthilnanthanan, M. Cost Effective Solvothermal Method to Synthesize Zn-Doped TiO2 Nanomaterials for Photovoltaic and Photocatalytic Degradation Applications. Catalysts 2021, 11, 690. [Google Scholar] [CrossRef]
- Maharubin, S.; Nayak, C.; Phatak, O.; Kurhade, A.; Singh, M.; Zhou, Y.; Tan, G. Polyvinylchloride coated with silver nanoparticles and zinc oxide nanowires for antimicrobial applications. Mater. Lett. 2019, 249, 108–111. [Google Scholar] [CrossRef]
- Marie, M.; Manoharan, A.; Kuchuk, A.; Ang, S.; Manasreh, M.O. Vertically grown zinc oxide nanorods functionalized with ferric oxide for in vivo and non-enzymatic glucose detection. Nanotechnology 2017, 29, 115501. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, R.; Tripathy, N.; Ahn, M.S.; Bhat, K.S.; Mahmoudi, T.; Wang, Y.; Yoo, J.; Kwon, D.; Yang, H.; Hahn, Y. Highly efficient non-enzymatic glucose sensor based on CuO modified vertically grown ZnO nanorods on electrode. Sci. Rep. 2017, 7, 5715. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; de Zhang, W.; Ye, J. Non enzymatic electrochemical glucose sensor based on MnO2/MWNTs nanocomposite. Electrochem. Commun. 2008, 10, 1268–1271. [Google Scholar] [CrossRef]
- Raha, S.; Mohanta, D.; Ahmaruzzaman, M. Novel CuO/Mn3O4/ZnO nanocomposite with superior photocatalytic activity for removal of rabeprazole from water. Sci. Rep. 2021, 11, 15187. [Google Scholar] [CrossRef] [PubMed]
- Ogunyemi, S.O.; Zhang, M.; Abdallah, Y.; Ahmed, T.; Qiu, W.; Ali, M.; Yan, C.; Yang, Y.; Chen, J.; Li, B. The bio-synthesis of three metal oxide nanoparticles (ZnO, MnO2, and MgO) and their antibacterial activity against the bacterial leaf blight pathogen. Front. Microbiol. 2020, 11, 588326. [Google Scholar] [CrossRef] [PubMed]
- Yan, Q.; Cai, Z. Issues in preparation of metal-lignin nanocomposites by co-precipitation method. J. Inorg. Organomet. Polym. Mater. 2021, 31, 978–996. [Google Scholar] [CrossRef]
- Alam, M.W.; Al Qahtani, H.S.; Aamir, M.; Abuzir, A.; Khan, M.S.; Albuhulayqah, M.; Mushtaq, S.; Zaidi, N.; Ramya, A. Phyto Synthesis of Manganese-Doped Zinc Nanoparticles Using Carica papaya Leaves: Structural Properties and Its Evaluation for Catalytic, Antibacterial and Antioxidant Activities. Polymers 2022, 14, 1827. [Google Scholar] [CrossRef]
- Burygin, G.L.; Khlebtsov, B.N.; Shantrokha, A.N.; Dykman, L.A.; Bogatyrev, V.A.; Khlebtsov, N.G. On the enhanced antibacterial activity of antibiotics mixed with gold nanoparticles. Nanoscale Res. Lett. 2009, 4, 794–801. [Google Scholar] [CrossRef]
- Juma, A.O.; Arbab, E.A.A.; Muiva, C.M.; Lepodise, L.M.; Mola, G.T. Synthesis and characterization of CuO-NiO-ZnO mixed metal oxide nanocomposite. J. Alloys Compd. 2017, 723, 866–872. [Google Scholar] [CrossRef]
- Enhessari, M.; Salehabadi, A.; Maarofian, K.; Khanahmadzadeh, S. Synthesis and physicochemical properties of CuMn2O4 nanoparticles; a potential semiconductor for photoelectric devices. Int. J. Bio-Inorg. Hybrid Nanomater. 2016, 5, 115–120. [Google Scholar]
- Sanchez, J.S.; Pendashteh, A.; Palma, J.; Anderson, M.; Marcilla, R. Porous NiCoMn trimetal oxide/graphene nanocomposites for high performance hybrid energy storage devices. Electrochim. Acta 2018, 279, 44–56. [Google Scholar] [CrossRef]
- Guo, H.; Zhu, Q.; Wu, X.; Jiang, Y.; Xie, X.; Xu, A. Oxygen deficient ZnO1-x nanosheets with high visible light photocatalytic activity. Nanoscale 2015, 7, 7216–7223. [Google Scholar] [CrossRef] [PubMed]
- Kosova, N.V.; Devyatkina, E.T.; Kaichev, V.V. Mixed layered Ni-Mn-Co hydroxides: Crystal structure, electronic state of ions, and thermal decomposition. J. Power Source 2007, 174, 735–740. [Google Scholar] [CrossRef]
- Pal, S.; Maiti, S.; Maiti, U. Chattopadhyay, Low temperature solution processed ZnO/CuO heterojunction photocatalyst for visible light induced photo-degradation of organic pollutants. Cryst. Eng. Comm. 2015, 17, 1464–1476. [Google Scholar] [CrossRef]
- Wu, Z.S.; Ren, W.; Wen, L.; Gao, L.; Zhao, J.; Chen, Z.; Zhou, G.; Li, F.; Cheng, H.M. Graphene anchored with Co3O4 nanoparticles as anode of lithium ion capacity and cyclic performance. ACS Nano 2010, 4, 3187–3194. [Google Scholar] [CrossRef]
- Jerng, S.; Yu, D.S.; Lee, J.H.; Kim, C.; Yoon, S.; Chun, S. Graphitic carbon growth on crystalline and amorphous oxide substrates using molecular beam epitaxy. Nanoscale Res. Lett. 2011, 6, 565–571. [Google Scholar] [CrossRef]
- Freitas, C.; Muller, R.H. Effect of light and temperature on zeta potential and physical stability in solid lipid nanoparticle (SLN (TM)) dispersions. Int. J. Pharm. 1998, 168, 221–229. [Google Scholar] [CrossRef]
- Vanaja, A.; Suresh, M.; Jeevanandam, J.; Gousia, S.; Pavan, D.; Balaji, D.; Murthy, N.B. Copper-doped zinc oxide nanoparticles for the fabrication of white LEDs. Prot. Met. Phys. Chem. Surf. 2019, 55, 481–486. [Google Scholar] [CrossRef]
- Khalid, A.; Ahmad, P.; Alharthi, A.I.; Muhammad, S.; Khandaker, M.U.; Rehman, M. Mohammad Rashed Iqbal Faruque, Israf Ud Din, Mshari A. Alotaibi, Khalid Alzimami, David A. Bradley, Structural, optical and antibacterial efficacy of pure and zinc doped copper oxide against pathogenic bacteria. Nanomaterials 2021, 11, 451. [Google Scholar] [CrossRef]
- Abel, M.J.; Pramothkumar, A.; Senthilkumar, N.; Jothivenkatachalam, K.; Inbaraj, P.F.H.; Prince, J.J. Flake-like CuMn2O4 nanoparticles synthesized via co-precipitation method for photocatalytic activity. Phys. B Condens. Matter 2019, 572, 117–124. [Google Scholar]
- Devi, S.M.; Nivetha, A.; Prabha, I. Role of citric acid/glycine-reinforced nanometal oxide for the enhancement of physio-chemical specifications in catalytic properties. J. Supercond. Nov. Magn. 2020, 33, 3893–3901. [Google Scholar] [CrossRef]
- Shanmugam, V.; Jeyaperumal, K.S. Investigations of visible light driven Sn and Cu doped ZnO hybrid nanoparticles for photocatalytic performance and antibacterial activity. Appl. Surf. Sci. 2018, 449, 617–630. [Google Scholar] [CrossRef]
- Ahmad, M.M.; Kotb, H.M.; Mushtaq, S.; Waheed-Ur-Rehman, M.; Maghanga, C.M.; Alam, M.W. Green synthesis of Mn+Cu bimetallic nanoparticles using vinca rosea extract and their antioxidant, antibacterial, and catalytic activities. Crystals 2022, 12, 72. [Google Scholar] [CrossRef]
- Liu, X.; Wang, C.; Zheng, Z.; Liu, W. Synthesis and Characterization of ultra-fine bimetallic Ag-Cu nanoparticles as die attach materials. In Proceedings of the International Conference on Electronic Packaging Technology (ICEPT) 2016, Wuhan, China, 16–19 August 2016; pp. 206–208. [Google Scholar]
- Alam, M.W.; Aamir, M.; Farhan, M.; Albuhulayqah, M.; Ahmad, M.M.; Ravikumar, C.R.; Kumar, V.G.D.; Murthy, H.C.A. Green synthesis of Ni-Cu-Zn based nanosized metal oxides for photocatalytic and sensor applications. Crystals 2021, 11, 1467. [Google Scholar] [CrossRef]
- Lin, J.; Luo, Z.; Liu, J.; Li, P. Photocatalytic degradation of methylene blue in aqueous solution by using ZnO-SnO2 nanocomposites. Mater. Sci. Semicond. Process. 2018, 87, 24–41. [Google Scholar] [CrossRef]
- Koli, P.B.; Kapadnis, K.H.; Deshpande, U.G.; Patil, M.R. Fabrication and characterization of pure and modified Co3O4 nanocatalyst and their application for photocatalytic degradation of eosine blue dye: A comparative study. J. Nanostruct. Chem. 2018, 8, 453–463. [Google Scholar] [CrossRef]
- Alzahrani, K.E.; Niazy, A.A.; Alswieleh, A.M.; Wahab, R.; El-Toni, A.M.; Alghamdi, H.S. Antibacterial activity of trimetal (CuZnFe) oxide nanoparticles. Int. J. Nanomed. 2018, 13, 77–87. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alam, M.W.; Al Qahtani, H.S.; Souayeh, B.; Ahmed, W.; Albalawi, H.; Farhan, M.; Abuzir, A.; Naeem, S. Novel Copper-Zinc-Manganese Ternary Metal Oxide Nanocomposite as Heterogeneous Catalyst for Glucose Sensor and Antibacterial Activity. Antioxidants 2022, 11, 1064. https://doi.org/10.3390/antiox11061064
Alam MW, Al Qahtani HS, Souayeh B, Ahmed W, Albalawi H, Farhan M, Abuzir A, Naeem S. Novel Copper-Zinc-Manganese Ternary Metal Oxide Nanocomposite as Heterogeneous Catalyst for Glucose Sensor and Antibacterial Activity. Antioxidants. 2022; 11(6):1064. https://doi.org/10.3390/antiox11061064
Chicago/Turabian StyleAlam, Mir Waqas, Hassan S. Al Qahtani, Basma Souayeh, Waqar Ahmed, Hind Albalawi, Mohd Farhan, Alaaedeen Abuzir, and Sumaira Naeem. 2022. "Novel Copper-Zinc-Manganese Ternary Metal Oxide Nanocomposite as Heterogeneous Catalyst for Glucose Sensor and Antibacterial Activity" Antioxidants 11, no. 6: 1064. https://doi.org/10.3390/antiox11061064
APA StyleAlam, M. W., Al Qahtani, H. S., Souayeh, B., Ahmed, W., Albalawi, H., Farhan, M., Abuzir, A., & Naeem, S. (2022). Novel Copper-Zinc-Manganese Ternary Metal Oxide Nanocomposite as Heterogeneous Catalyst for Glucose Sensor and Antibacterial Activity. Antioxidants, 11(6), 1064. https://doi.org/10.3390/antiox11061064









