Levels of Plasma Coenzyme Q10 Are Associated with Physical Capacity and Cardiovascular Risk in the Elderly
Abstract
:1. Introduction
2. Materials and Methods
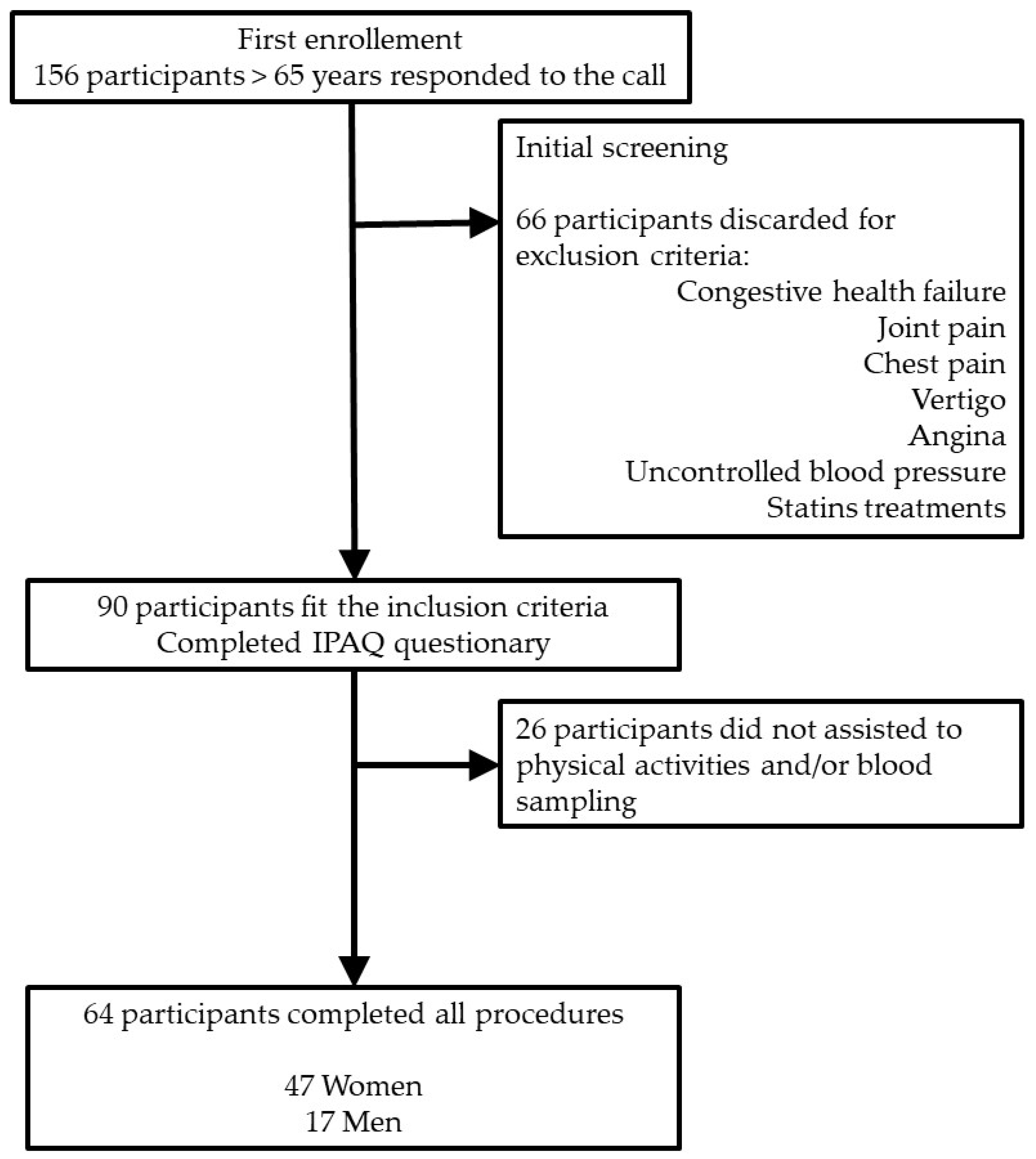
2.1. Study Design and Subjects
2.2. Participants
2.3. Anthropometric Determinations
2.4. Determination of the Physical Capacity
2.5. Determination of the Level of Physical Activity (IPAQ)
2.6. Blood Sampling
2.7. CoQ10 Determination in Plasma
2.8. Determination of Other Parameters in Plasma
2.9. Statistical Analysis
3. Results
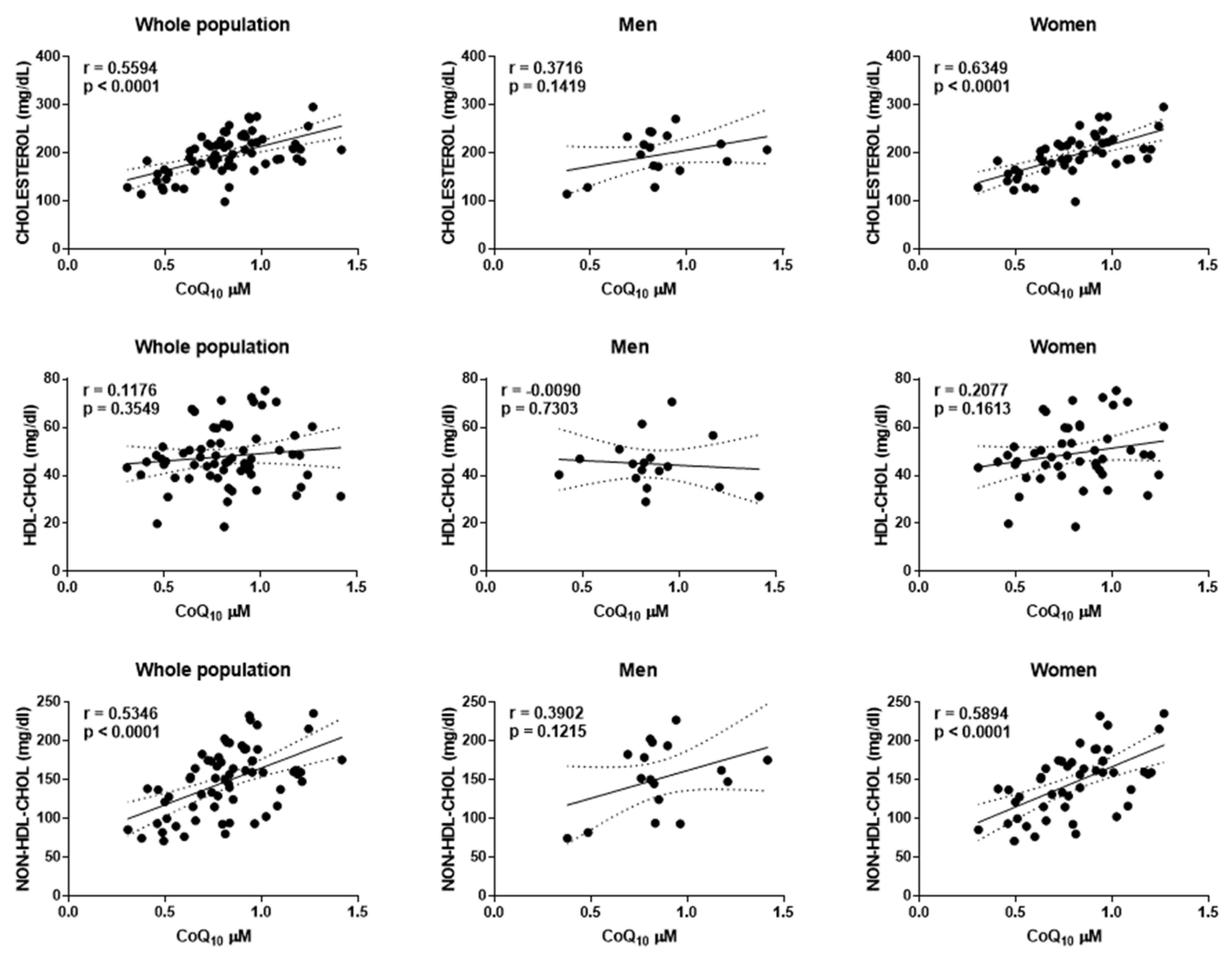
3.1. Biochemical Characterization of the Participants
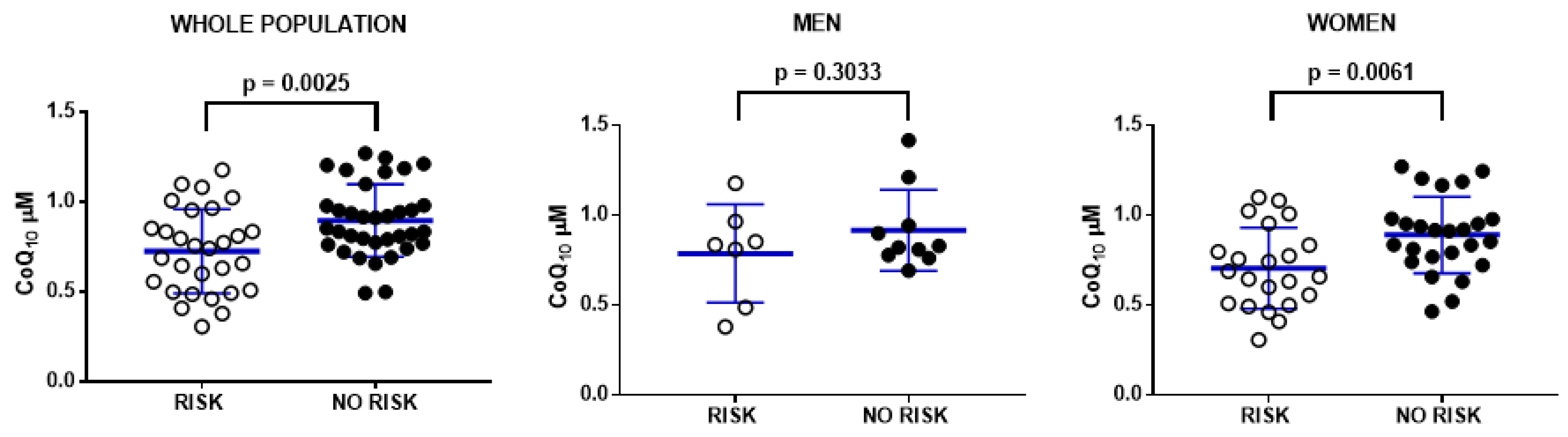
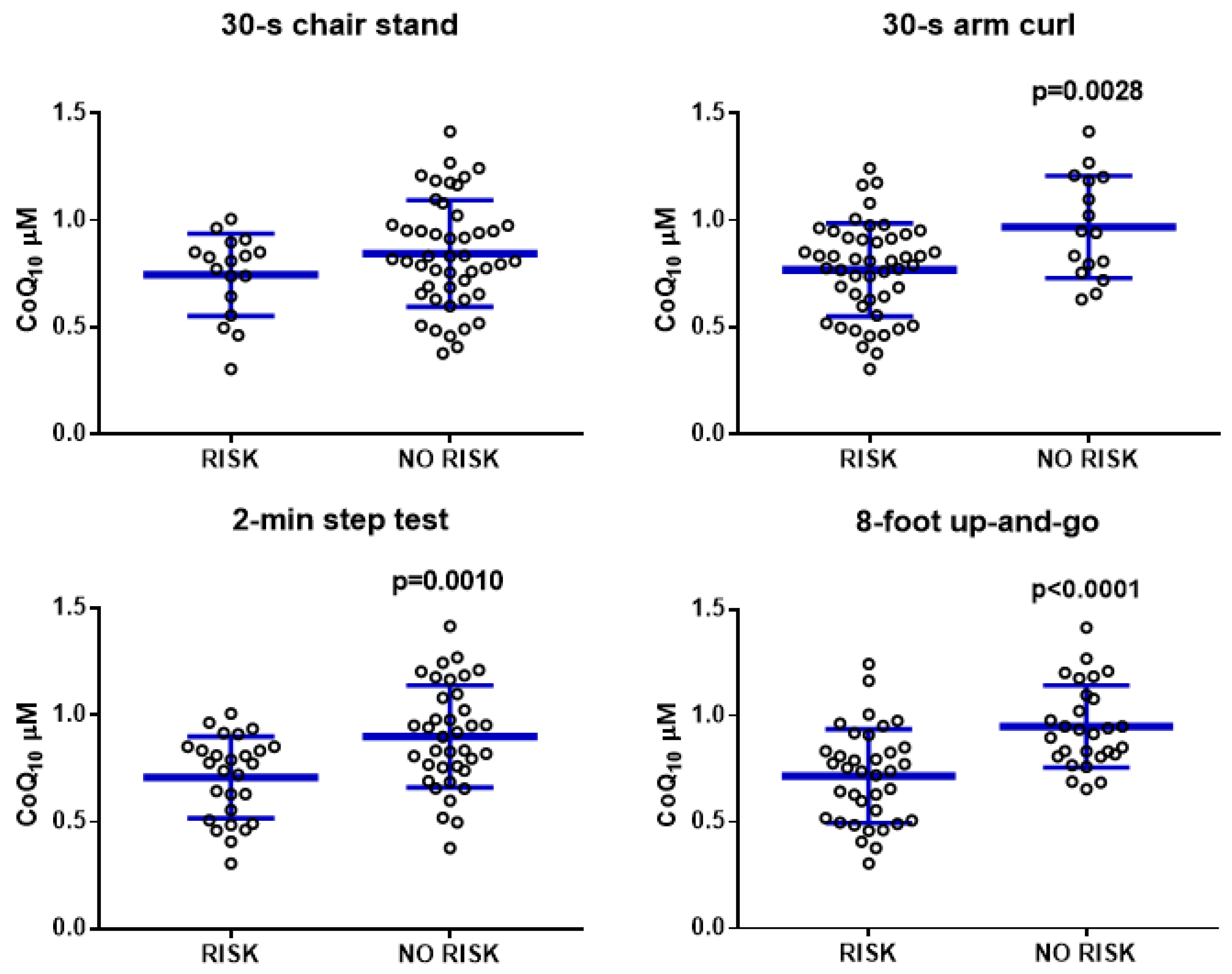
3.2. CoQ10 Levels in Plasma Correlates with Physical Activity
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- López-Lluch, G.; Lluch, L. The Important Role of CoQ10 in Aging. Antioxidants 2019, 8, 570. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- López-Lluch, G.; Rodríguez-Aguilera, J.C.; Santos-Ocaña, C.; Navas, P. Is coenzyme Q a key factor in aging? Mech. Ageing Dev. 2010, 131, 225–235. [Google Scholar] [CrossRef] [PubMed]
- Barroso, M.P.; Gómez-Díaz, C.; Villalba, J.M.; Burón, M.I.; López-Lluch, G.; Navas, P. Plasma Membrane Ubiquinone Controls Ceramide Production and Prevents Cell Death Induced by Serum Withdrawal. J. Bioenerg. Biomembr. 1997, 29, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Crane, F.L.; Navas, P. The diversity of coenzyme Q function. Mol. Asp. Med. 1997, 18, 1–6. [Google Scholar] [CrossRef]
- López-Lluch, G.; Barroso, M.P.; Martín, S.F.; Fernández-Ayala, D.J.M.; Gómez-Díaz, C.; Villalba, J.M.; Navas, P. Role of plasma membrane coenzyme Q on the regulation of apoptosis. BioFactors 1999, 9, 171–177. [Google Scholar] [CrossRef]
- Villalba, J.; Navarro, F.; Gómez-Díaz, C.; Arroyo, A.; Bello, R.; Navas, P. Role of cytochrome b5 reductase on the antioxidant function of coenzyme Q in the plasma membrane. Mol. Asp. Med. 1997, 18, 7–13. [Google Scholar] [CrossRef]
- Yubero-Serrano, E.M.; Delgado-Casado, N.; Gomez-Delgado, F.; Perez-Martinez, P.; Tasset-Cuevas, I.; Santos-Gonzalez, M.; Caballero-Villarraso, J.; Garcia-Rios, A.; Marín, C.; Gutierrez-Mariscal, F.M.; et al. Postprandial antioxidant effect of the Mediterranean diet supplemented with coenzyme Q10 in elderly men and women. Age 2010, 33, 579–590. [Google Scholar] [CrossRef] [Green Version]
- De Barcelos, I.P.; Haas, R.H. CoQ10 and Aging. Biology 2019, 8, 28. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hernández-Camacho, J.D.; García-Corzo, L.; Fernández-Ayala, D.J.M.; Navas, P.; López-Lluch, G. Coenzyme Q at the Hinge of Health and Metabolic Diseases. Antioxidants 2021, 10, 1785. [Google Scholar] [CrossRef]
- Santos-Ocaña, C.; Cascajo, M.V.; Alcázar-Fabra, M.; Staiano, C.; López-Lluch, G.; Brea-Calvo, G.; Navas, P. Cellular Models for Primary CoQ Deficiency Pathogenesis Study. Int. J. Mol. Sci. 2021, 22, 10211. [Google Scholar] [CrossRef]
- del Pozo-Cruz, J.; Rodríguez-Bies, E.; Ballesteros-Simarro, M.; Navas-Enamorado, I.; Tung, B.T.; Navas, P.; López-Lluch, G. Physical activity affects plasma coenzyme Q10 levels differently in young and old humans. Biogerontology 2014, 15, 199–211. [Google Scholar] [CrossRef] [PubMed]
- del Pozo-Cruz, J.; Rodríguez-Bies, E.; Navas-Enamorado, I.; Cruz, B.D.P.; Navas, P.; López-Lluch, G. Relationship between functional capacity and body mass index with plasma coenzyme Q10 and oxidative damage in community-dwelling elderly-people. Exp. Gerontol. 2014, 52, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Niklowitz, P.; Scherer, J.; Döring, F.; Paulussen, M.; Menke, T. Oxidized proportion of muscle coenzyme Q10 increases with age in healthy children. Pediatr. Res. 2015, 78, 365–370. [Google Scholar] [CrossRef]
- Vásquez-Alvarez, S.; Bustamante-Villagomez, S.K.; Vazquez-Marroquin, G.; Porchia, L.M.; Pérez-Fuentes, R.; Torres-Rasgado, E.; Herrera-Fomperosa, O.; Montes-Arana, I.; Gonzalez-Mejia, M.E. Metabolic Age, an Index Based on Basal Metabolic Rate, Can Predict Individuals That are High Risk of Developing Metabolic Syndrome. High Blood Press. Cardiovasc. Prev. 2021, 28, 263–270. [Google Scholar] [CrossRef] [PubMed]
- Daďová, K.; Petr, M.; Šteffl, M.; Sontáková, L.; Chlumský, M.; Matouš, M.; Štich, V.; Štěpán, M.; Šiklová, M. Effect of Calanus Oil Supplementation and 16 Week Exercise Program on Selected Fitness Parameters in Older Women. Nutrients 2020, 12, 481. [Google Scholar] [CrossRef] [Green Version]
- Rikli, R.E.; Jones, C.J. Senior Fitness Test Manual; Human Kinetics: Champaign, IL, USA, 2001. [Google Scholar]
- Bohannon, R.W.; Crouch, R.H. Two-Minute Step Test of Exercise Capacity: Systematic Review of Procedures, Performance, and Clinimetric Properties. J. Geriatr. Phys. Ther. 2019, 42, 105–112. [Google Scholar] [CrossRef]
- Schoene, D.; Wu, S.M.-S.; Mikolaizak, A.S.; Menant, J.C.; Smith, S.; Delbaere, K.; Lord, S.R. Discriminative Ability and Predictive Validity of the Timed Up and Go Test in Identifying Older People Who Fall: Systematic Review and Meta-Analysis. J. Am. Geriatr. Soc. 2013, 61, 202–208. [Google Scholar] [CrossRef]
- Craig, C.L.; Marshall, A.L.; Sjöström, M.; Bauman, A.E.; Booth, M.L.; Ainsworth, B.E.; Pratt, M.; Ekelund, U.; Yngve, A.; Sallis, J.F.; et al. International Physical Activity Questionnaire: 12-Country Reliability and Validity. Med. Sci. Sports Exerc. 2003, 35, 1381–1395. [Google Scholar] [CrossRef] [Green Version]
- Rodríguez-Aguilera, J.C.; Cortés, A.B.; Fernández-Ayala, D.J.M.; Navas, P. Biochemical Assessment of Coenzyme Q10 Deficiency. J. Clin. Med. 2017, 6, 27. [Google Scholar] [CrossRef] [Green Version]
- Friedewald, W.T.; Levy, R.I.; Fredrickson, D.S. Estimation of the Concentration of Low-Density Lipoprotein Cholesterol in Plasma, Without Use of the Preparative Ultracentrifuge. Clin. Chem. 1972, 18, 499–502. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences; Lawrence Erlbaum Associates: Hillsdale, NJ, USA, 1988. [Google Scholar]
- Lu, W.; Resnick, H.E.; Jablonski, K.A.; Jones, K.L.; Jain, A.K.; Howard, W.J.; Robbins, D.C.; Howard, B.V. Non-HDL Cholesterol as a Predictor of Cardiovascular Disease in Type 2 Diabetes. Diabetes Care 2003, 26, 16–23. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rikli, R.E.; Jones, C.J. Development and Validation of Criterion-Referenced Clinically Relevant Fitness Standards for Maintaining Physical Independence in Later Years. Gerontologist 2013, 53, 255–267. [Google Scholar] [CrossRef] [PubMed]
- Hanlon, P.; Nicholl, B.I.; Jani, B.D.; Lee, D.; McQueenie, R.; Mair, F.S. Frailty and pre-frailty in middle-aged and older adults and its association with multimorbidity and mortality: A prospective analysis of 493 737 UK Biobank participants. Lancet Public Health 2018, 3, e323–e332. [Google Scholar] [CrossRef]
- Linnane, A.W.; Zhang, C.; Yarovaya, N.; Kopsidas, G.; Kovalenko, S.; Papakostopoulos, P.; Eastwood, H.; Graves, S.; Richardson, M. Human aging and global function of coenzyme Q10. Ann. N. Y. Acad. Sci. 2002, 959, 396–411. [Google Scholar] [CrossRef] [PubMed]
- Andreux, P.A.; Van Diemen, M.P.J.; Heezen, M.R.; Auwerx, J.; Rinsch, C.; Groeneveld, G.J.; Singh, A. Mitochondrial function is impaired in the skeletal muscle of pre-frail elderly. Sci. Rep. 2018, 8, 8548. [Google Scholar] [CrossRef] [PubMed]
- Rygiel, K.A.; Dodds, R.M.; Patel, H.P.; Syddall, H.E.; Westbury, L.D.; Granic, A.; Cooper, C.; Cliff, J.; Rocha, M.; Turnbull, D.M.; et al. Mitochondrial respiratory chain deficiency in older men and its relationship with muscle mass and performance. JCSM Clin. Rep. 2017, 2, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Pilotto, A.; Custodero, C.; Maggi, S.; Polidori, M.C.; Veronese, N.; Ferrucci, L. A multidimensional approach to frailty in older people. Ageing Res. Rev. 2020, 60, 101047. [Google Scholar] [CrossRef]
- Romanello, V. The Interplay between Mitochondrial Morphology and Myomitokines in Aging Sarcopenia. Int. J. Mol. Sci. 2020, 22, 91. [Google Scholar] [CrossRef]
- Lass, A.; Kwong, L.; Sohal, P.R.S. Mitochondrial coenzyme Q content and aging. BioFactors 1999, 9, 199–205. [Google Scholar] [CrossRef]
- Chen, H.-C.; Huang, C.-C.; Lin, T.-J.; Hsu, M.-C.; Hsu, Y.-J. Ubiquinol Supplementation Alters Exercise Induced Fatigue by Increasing Lipid Utilization in Mice. Nutrients 2019, 11, 2550. [Google Scholar] [CrossRef] [Green Version]
- Beyer, R.E.; Burnett, B.-A.; Cartwright, K.J.; Edington, D.W.; Falzon, M.J.; Kreitman, K.R.; Kuhn, T.W.; Ramp, B.J.; Rhee, S.Y.S.; Rosenwasser, M.J.; et al. Tissue coenzyme Q (ubiquinone) and protein concentrations over the life span of the laboratory rat. Mech. Ageing Dev. 1985, 32, 267–281. [Google Scholar] [CrossRef] [Green Version]
- Sugiyama, S.; Yamada, K.; Ozawa, T. Preservation of mitochondrial respiratory function by coenzyme Q10 in aged rat skeletal muscle. Biochem. Mol. Biol. Int. 1995, 37, 1111–1120. [Google Scholar] [PubMed]
- Chang, P.-S.; Yen, C.-H.; Huang, Y.-Y.; Chiu, C.-J.; Lin, P.-T. Associations between Coenzyme Q10 Status, Oxidative Stress, and Muscle Strength and Endurance in Patients with Osteoarthritis. Antioxidants 2020, 9, 1275. [Google Scholar] [CrossRef] [PubMed]
- Trevisson, E.; DiMauro, S.; Navas, P.; Salviati, L. Coenzyme Q deficiency in muscle. Curr. Opin. Neurol. 2011, 24, 449–456. [Google Scholar] [CrossRef] [PubMed]
- Du, J.; Wang, T.; Huang, P.; Cui, S.; Gao, C.; Lin, Y.; Fu, R.; Shen, J.; He, Y.; Tan, Y.; et al. Clinical correlates of decreased plasma coenzyme Q10 levels in patients with multiple system atrophy. Park. Relat. Disord. 2018, 57, 58–62. [Google Scholar] [CrossRef] [PubMed]
- Mortensen, A.L.; Rosenfeldt, F.; Filipiak, K.J. Effect of coenzyme Q10 in Europeans with chronic heart failure: A sub-group analysis of the Q-SYMBIO randomized double-blind trial. Cardiol. J. 2013, 26, 147–156. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mortensen, S.A.; Rosenfeldt, F.; Kumar, A.; Dolliner, P.; Filipiak, K.J.; Pella, D.; Alehagen, U.; Steurer, G.; Littarru, G.P. The Effect of Coenzyme Q 10 on Morbidity and Mortality in Chronic Heart Failure. JACC Hear. Fail. 2014, 2, 641–649. [Google Scholar] [CrossRef]
- Raizner, A.E.; Quiñones, M.A. Coenzyme Q10 for Patients With Cardiovascular Disease. J. Am. Coll. Cardiol. 2021, 77, 609–619. [Google Scholar] [CrossRef] [PubMed]
- Martelli, A.; Testai, L.; Colletti, A.; Cicero, A.F.G. Coenzyme Q10: Clinical Applications in Cardiovascular Diseases. Antioxidants 2020, 9, 341. [Google Scholar] [CrossRef] [Green Version]
- Nilsson, M.I.; Tarnopolsky, M.A. Mitochondria and Aging—The Role of Exercise as a Countermeasure. Biology 2019, 8, 40. [Google Scholar] [CrossRef] [Green Version]
- Turunen, M.; Olsson, J.; Dallner, G. Metabolism and function of coenzyme Q. Biochim. Biophys. Acta (BBA)-Biomembr. 2004, 1660, 171–199. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sabbatinelli, J.; Orlando, P.; Galeazzi, R.; Silvestri, S.; Cirilli, I.; Marcheggiani, F.; Dludla, P.V.; Giuliani, A.; Bonfigli, A.R.; Mazzanti, L.; et al. Ubiquinol Ameliorates Endothelial Dysfunction in Subjects with Mild-to-Moderate Dyslipidemia: A Randomized Clinical Trial. Nutrients 2020, 12, 1098. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, S.-Y.; Kwon, O.S.; Andtbacka, R.H.I.; Hyngstrom, J.R.; Reese, V.; Murphy, M.P.; Richardson, R.S. Age-related endothelial dysfunction in human skeletal muscle feed arteries: The role of free radicals derived from mitochondria in the vasculature. Acta Physiol. 2018, 222, e12893. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lönnrot, K.; Pörsti, I.; Alho, H.; Wu, X.; Hervonen, A.; Tolvanen, J.-P. Control of arterial tone after long-term coenzyme Q10supplementation in senescent rats. J. Cereb. Blood Flow Metab. 1998, 124, 1500–1506. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, S.-Y.; Pekas, E.J.; Headid, R.J., 3rd; Son, W.-M.; Wooden, T.K.; Song, J.; Layec, G.; Yadav, S.K.; Mishra, P.K.; Pipinos, I.I. Acute mitochondrial antioxidant intake improves endothelial function, antioxidant enzyme activity, and exercise tolerance in patients with peripheral artery disease. Am. J. Physiol. Heart Circ. Physiol. 2020, 319, H456–H467. [Google Scholar] [CrossRef] [PubMed]
- Fischer, A.; Onur, S.; Niklowitz, P.; Menke, T.; Laudes, M.; Rimbach, G.; Döring, F. Coenzyme Q10 Status as a Determinant of Muscular Strength in Two Independent Cohorts. PLoS ONE 2016, 11, e0167124. [Google Scholar] [CrossRef] [PubMed]
- Bailey, D.M.; McEneny, J.; Mathieu-Costello, O.; Henry, R.R.; James, P.E.; Mccord, J.M.; Pietri, S.; Young, I.S.; Richardson, R.S. Sedentary aging increases resting and exercise-induced intramuscular free radical formation. J. Appl. Physiol. 2010, 109, 449–456. [Google Scholar] [CrossRef] [Green Version]
- Sánchez-Cuesta, A.; Cortés-Rodríguez, A.B.; Navas-Enamorado, I.; Lekue, J.A.; Viar, T.; Axpe, M.; Navas, P.; López-Lluch, G. High coenzyme Q10 plasma levels improve stress and damage markers in professional soccer players during competition. Int. J. Vitam. Nutr. Res. 2020, 1–12. [Google Scholar] [CrossRef]
- Belviranlı, M.; Okudan, N. Effect of Coenzyme Q10 Alone and in Combination with Exercise Training on Oxidative Stress Biomarkers in Rats. Int. J. Vitam. Nutr. Res. 2018, 88, 126–136. [Google Scholar] [CrossRef]
- Ochoa, J.J.; Quiles, J.L.; Frias, M.L.; Huertas, J.R.; Mataix, J. Effect of Lifelong Coenzyme Q10 Supplementation on Age-Related Oxidative Stress and Mitochondrial Function in Liver and Skeletal Muscle of Rats Fed on a Polyunsaturated Fatty Acid (PUFA)-Rich Diet. J. Gerontol. Ser. A 2007, 62, 1211–1218. [Google Scholar] [CrossRef] [Green Version]
- Rodríguez-Bies, E.; Navas, P.; López-Lluch, G. Age-Dependent Effect of Every-Other-Day Feeding and Aerobic Exercise in Ubiquinone Levels and Related Antioxidant Activities in Mice Muscle. J. Gerontol. Ser. A 2015, 70, 33–43. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Andreani, C.; Bartolacci, C.; Guescini, M.; Battistelli, M.; Stocchi, V.; Orlando, F.; Provinciali, M.; Amici, A.; Marchini, C.; Tiano, L.; et al. Combination of Coenzyme Q10 Intake and Moderate Physical Activity Counteracts Mitochondrial Dysfunctions in a SAMP8 Mouse Model. Oxidative Med. Cell. Longev. 2018, 2018, 8936251. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guescini, M.; Tiano, L.; Genova, M.L.; Polidori, E.; Silvestri, S.; Orlando, P.; Fimognari, C.; Calcabrini, C.; Stocchi, V.; Sestili, P. The Combination of Physical Exercise with Muscle-Directed Antioxidants to Counteract Sarcopenia: A Biomedical Rationale for Pleiotropic Treatment with Creatine and Coenzyme Q10. Oxidative Med. Cell. Longev. 2017, 2017, 7083049. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aaseth, J.; Alexander, J.; Alehagen, U. Coenzyme Q10 supplementation—In ageing and disease. Mech. Ageing Dev. 2021, 197, 111521. [Google Scholar] [CrossRef] [PubMed]
- López-Lluch, G. Coenzyme Q homeostasis in aging: Response to non-genetic interventions. Free Radic. Biol. Med. 2021, 164, 285–302. [Google Scholar] [CrossRef] [PubMed]
- Picca, A.; Beli, R.; Calvani, R.; Coelho-Júnior, H.J.; Landi, F.; Bernabei, R.; Bucci, C.; Guerra, F.; Marzetti, E. Older Adults with Physical Frailty and Sarcopenia Show Increased Levels of Circulating Small Extracellular Vesicles with a Specific Mitochondrial Signature. Cells 2020, 9, 973. [Google Scholar] [CrossRef] [Green Version]
- Habiballa, L.; Salmonowicz, H.; Passos, J.F. Mitochondria and cellular senescence: Implications for musculoskeletal ageing. Free Radic. Biol. Med. 2019, 132, 3–10. [Google Scholar] [CrossRef]
- Marcheggiani, F.; Kordes, S.; Cirilli, I.; Orlando, P.; Silvestri, S.; Vogelsang, A.; Möller, N.; Blatt, T.; Weise, J.M.; Damiani, E.; et al. Anti-ageing effects of ubiquinone and ubiquinol in a senescence model of human dermal fibroblasts. Free Radic. Biol. Med. 2021, 165, 282–288. [Google Scholar] [CrossRef]
- Olivieri, F.; Lazzarini, R.; Babini, L.; Prattichizzo, F.; Rippo, M.R.; Tiano, L.; Di Nuzzo, S.; Graciotti, L.; Festa, R.; Brugè, F.; et al. Anti-inflammatory effect of ubiquinol-10 on young and senescent endothelial cells via miR-146a modulation. Free Radic. Biol. Med. 2013, 63, 410–420. [Google Scholar] [CrossRef]
- Rong, Y.-D.; Bian, A.-L.; Hu, H.-Y.; Ma, Y.; Zhou, X.-Z. Study on relationship between elderly sarcopenia and inflammatory cytokine IL-6, anti-inflammatory cytokine IL-10. BMC Geriatr. 2018, 18, 308. [Google Scholar] [CrossRef] [Green Version]
- Huo, J.; Xu, Z.; Hosoe, K.; Kubo, H.; Miyahara, H.; Dai, J.; Mori, M.; Sawashita, J.; Higuchi, K. Coenzyme Q10 Prevents Senescence and Dysfunction Caused by Oxidative Stress in Vascular Endothelial Cells. Oxidative Med. Cell. Longev. 2018, 2018, 3181759. [Google Scholar] [CrossRef] [PubMed]
- Hargreaves, I.; Heaton, R.A.; Mantle, D. Disorders of Human Coenzyme Q10 Metabolism: An Overview. Int. J. Mol. Sci. 2020, 21, 6695. [Google Scholar] [CrossRef] [PubMed]
- Beaulieu, S.; Vachon, A.; Plourde, M. Women have higher levels of CoQ10 than men when supplemented with a single dose of CoQ10 with monoglycerides omega-3 or rice oil and followed for 48 h: A crossover randomised triple blind controlled study. J. Nutr. Sci. 2022, 1, 1–9. [Google Scholar] [CrossRef]
- Brivio, P.; Paladini, M.S.; Racagni, G.; Riva, M.A.; Calabrese, F.; Molteni, R. From Healthy Aging to Frailty: In Search of the Underlying Mechanisms. Curr. Med. Chem. 2019, 26, 3685–3701. [Google Scholar] [CrossRef] [PubMed]
| Parameter | Total Population (n = 64) | MEN (n = 17) | WOMEN (n = 47) |
|---|---|---|---|
| Mean ± SD | Mean ± SD | Mean ± SD | |
| Age | |||
| From 65 to 75 | 32 (50.0%) | 11 (64.7%) | 21 (44.68%) |
| More than 76 | 32 (50.0%) | 6 (35.3%) | 26 (55.32%) |
| Pharmacological treatments | |||
| 59 (92.2%) | 16 (94.1%) | 43 (91.5%) | |
| Hypertension | 30 (46.9%) | 10 (58.8%) | 20 (42.6%) |
| Diabetes | 8 (12.5%) | 5 (29.4%) | 3 (6.4%) |
| Others | 47 (73.4%) | 11 (64.7%) | 35 (74.5%) |
| Smoking habits | |||
| Never | 38 (59.3%) | 1 (5.9%) | 37 (78.7%) |
| Before | 20 (31.3%) | 10 (58.8%) | 10 (21.3%) |
| During study | 6 (9.4%) | 6 (35.3%) | 0 (0.0%) |
| Drinking habits | |||
| Never | 19 (29.7%) | 0 (0.0%) | 19 (40.4%) |
| Before | 13 (20.31%) | 9 (52.9%) | 4 (2.5%) |
| During study | 32 (50.0%) | 8 (47.1%) | 24 (51.1%) |
| Parameter | Total Population (n = 64) | MEN (n = 17) | WOMEN (n = 47) | Significance | |||
|---|---|---|---|---|---|---|---|
| Mean ± SD | Range | Mean ± SD | Range | Mean ± SD | Range | p | |
| Age (y) | 77.0 ± 8.0 | 65–99 | 73.4 ± 6.3 | 65–84 | 78.3 ± 8.2 | 66–90 | 0.0381 * |
| Metabolic age (y) | 76.5 ± 11.0 | 48–90 | 76.4 ± 13.1 | 48–90 | 76.5 ± 10.2 | 55–90 | 0.9803 |
| DIA (y) | 0.5 ± 12.6 | −20–30 | −3 ± 16.1 | −20–30 | 1.8 ± 11.0 | −15–15 | 0.1847 |
| BP Systolic | 134 ± 17 | 89–167 | 134 ± 17 | 89–164 | 134 ± 17.6 | 94–167 | 0.9617 |
| BP Diastolic | 72.9± 9.6 | 48–93 | 70.4 ± 10.6 | 48–93 | 74.8± 12.4 | 52–132 | 0.1976 |
| Height (cm) | 154.1 ± 7.4 | 138–173 | 160.6 ± 5.6 | 149–173 | 151.8 ± 6.6 | 138–167 | <0.0001 * |
| Weigth (kg) | 70.6 ± 8.6 | 49.2–90.7 | 74.2 ± 7.9 | 60.0–88.4 | 69.2 ± 8.6 | 49.2–90.7 | 0.0426 * |
| BMI | 29.8 ± 3.9 | 22.6–40.8 | 28.7 ± 3.1 | 23.4–35.5 | 30.2 ± 4.5 | 19.9–40.9 | 0.2277 |
| Fat (%) | 40.4 ± 7.8 | 21.7–61.7 | 32.5 ± 5.4 | 21.7–40.1 | 43.3 ± 6.4 | 24.1–61.7 | <0.0001 * |
| Muscle (kg) | 39.7 ± 6.6 | 25–61 | 47.4 ± 5.5 | 38.4–61 | 36.9 ± 4.5 | 25–49.6 | <0.0001 * |
| Visceral fat (kg) | 14.7 ± 4.0 | 7–30.4 | 18.8 ± 4.2 | 12–30.4 | 13.2 ± 2.3 | 7–19 | <0.0001 * |
| Parameter | Total Population (n = 64) | MEN (n = 17) | WOMEN (n = 47) | Significance | |||
|---|---|---|---|---|---|---|---|
| Mean ± SD | Range | Mean ± SD | Range | Mean ± SD | Range | p | |
| Total CoQ10 (μM) | 0.818 ± 0.238 | 0.306–1.416 | 0.862 ± 0.247 | 0.378–1.416 | 0.806 ± 0.236 | 0.306–1.269 | 0.3859 |
| Chol (mg/dL) | 196.1 ± 42.9 | 99–296 | 197.0 ± 45.1 | 115–271 | 195.8 ± 42.6 | 99–296 | 0.9214 |
| HDL-chol (mg/dL) | 48.0 ± 12.3 | 18.6–75.3 | 44.8 ± 10.7 | 29–70.6 | 49.1 ± 12.8 | 18.6–75.3 | 0.2166 |
| LDL-chol (mg/dL) | 128 ± 42 | 51–215 | 132 ± 46 | 58.1–205 | 126.5 ± 40.9 | 51–215 | 0.6451 |
| VLDL-chol (mg/dL) | 20.2 ± 6.7 | 14–41 | 20.2 ± 6.9 | 14–36 | 20.2 ± 6.6 | 14–41 | 0.9931 |
| Non-HDL-chol (mg/dL) | 148 ± 42.2 | 71.1–236 | 152 ± 45.4 | 74.7–227.3 | 146 ± 41.4 | 71.1–235 | 0.6458 |
| Chol/HDL-chol | 4.3 ± 1.3 | 2.3–7.9 | 4.6 ± 1.3 | 2.3–6.6 | 4.2 ± 1.3 | 2.3–7.9 | 0.3239 |
| TGs (mg/dL) | 100.8 ± 33.3 | 69.9–205 | 100.9 ± 34.6 | 69.9–180 | 100.8 ± 33.2 | 69.9–205 | 0.9917 |
| CoQ10/Chol (nmol/mmol) | 163.2 ± 44.5 | 85.6–316.3 | 172.7 ± 50.3 | 114–264 | 159.8 ± 42.2 | 85.6–316 | 0.3059 |
| CoQ10/HDL-chol (nmol/mmol/ | 705 ± 303 | 273–1746 | 791 ± 346 | 363–1746 | 674 ± 284 | 273–1684 | 0.1744 |
| CoQ10/LDL-chol (nmol/mmol) | 265 ± 92.6 | 127–538 | 275 ± 105 | 158–538 | 261 ± 88.5 | 127–538 | 0.5821 |
| CoQ10/VLDL-chol (nmol/mmol) | 1690 ± 638 | 596–3219 | 1773 ± 644 | 881–2990 | 1661 ± 640 | 596–3219 | 0.5383 |
| CoQ10/non-HDL-chol (nmol/mmol) | 222 ± 67.4 | 113.8–398.7 | 231 ± 75.4 | 145–399 | 219 ± 64.8 | 114–390 | 0.5436 |
| oxLDL (U/L) | 75.6 ± 17.1 | 34.6–105 | 77.8 ± 19.5 | 34.6–109 | 75.5 ± 16.4 | 46.4–105 | 0.6316 |
| CK (U/L) | 69.8 ± 36.8 | 24.3–226.0 | 86.0 ± 49.6 | 24.3–226.0 | 64.0 ± 29.4 | 24.3–159 | 0.0335 * |
| GGT (U/L) | 18.1 ± 23.4 | 0.49–144 | 29.1 ± 36.1 | 0.5–144 | 13.8 ± 15.1 | 0.5–82.4 | 0.0196 * |
| GOT (U/L) | 21.2 ± 6.5 | 5–37.4 | 18.0 ± 6.7 | 5–29.2 | 22.4 ± 6.0 | 12–37.4 | 0.0153 * |
| GPT (U/L) | 15.1 ± 5.1 | 6.5–32.8 | 16.7 ± 5.6 | 6.5–25.2 | 14.5 ± 4.8 | 7.5–32.8 | 0.122 |
| BILIRRUBIN (mg/dL) | 0.76 ± 0.19 | 0.49–1.28 | 0.72 ± 0.20 | 0.49–1.28 | 0.78 ± 0.19 | 0.5–1.3 | 0.3505 |
| CREATININE (mg/dL) | 1.16 ± 0.28 | 0.63–2.18 | 1.28 ± 0.43 | 0.49–2.18 | 1.09 ± 0.22 | 0.49–1.86 | 0.0302 * |
| URIC ACID (mg/dL) | 5.75 ± 1.75 | 1.99–10.1 | 6.25 ± 1.79 | 3.23–9.70 | 5.57 ± 1.71 | 1.99–10.1 | 0.1719 |
| UREA (mg/dL | 35.5 ± 14.2 | 19–87.5 | 34.1 ± 15.7 | 19–76.5 | 36.0 ± 13.7 | 19–87.5 | 0.6314 |
| GLUCOSE (mg/dL) | 113.6 ± 34.8 | 38.5–218 | 123.2 ± 46.1 | 48.9–218 | 110.2 ± 29.5 | 38.5–205 | 0.189 |
| Parameter | Total Population (n = 64) | MEN (n = 17) | WOMEN (n = 47) | |||
|---|---|---|---|---|---|---|
| Pearson r | p | Pearson r | p | Pearson r | p | |
| Chol (mg/dL) | 0.56 | <0.0001 | 0.372 | 0.1419 | 0.635 | <0.0001 |
| HDL-chol (mg/dL) | 0.118 | 0.3549 | −0.09 | 0.7303 | 0.208 | 0.1613 |
| LDL-chol (mg/dL) | 0.509 | <0.0001 | 0.347 | 0.172 | 0.571 | <0.0001 |
| VLDL.chol (mg/dL) | 0.172 | 0.1725 | 0.253 | 0.3266 | 0.156 | 0.2942 |
| Non-HDL-chol (mg/dL) | 0.535 | <0.0001 | 0.39 | 0.1215 | 0.589 | <0.0001 |
| TC/HDL-chol | 0.328 | <0.0080 | 0.466 | 0.0593 | 0.265 | 0.0719 |
| oxLDL (U/L) | 0.073 | 0.5642 | −0.005 | 0.9856 | −0.001 | 0.9953 |
| TGs (mg/dL) | 0.182 | 0.1495 | 0.253 | 0.3266 | 0.157 | 0.2934 |
| CK (U/L) | −0.009 | 0.9444 | 0.000 | 0.9995 | −0.066 | 0.6586 |
| GGT (U/L) | −0.006 | 0.9634 | 0.001 | 0.9693 | −0.089 | 0.5517 |
| GOT (U/L) | 0.18 | 0.154 | 0.331 | 0.1937 | 0.18 | 0.154 |
| GPT (U/L) | 0.128 | 0.3131 | 0.110 | 0.6738 | 0.109 | 0.4662 |
| BILIRRUBIN (mg/dL) | −0.009 | 0.9444 | −0.065 | 0.8045 | 0.158 | 0.2876 |
| CREATININE (mg/dL) | −0.013 | 0.313 | 0.164 | 0.5295 | −0.095 | 0.5259 |
| URIC ACID (mg/dL) | −0.058 | 0.6466 | 0.031 | 0.9057 | −0.121 | 0.4172 |
| UREA (mg/Dl | 0.085 | 0.505 | 0.397 | 0.115 | −0.035 | 0.8153 |
| GLUCOSE (mg/dL) | −0.143 | 0.2589 | −0.199 | 0.4433 | −0.15 | 0.3143 |
| Parameter | Total Population (n = 64) | MEN (n = 17) | WOMEN (n = 47) | Significance | |||
|---|---|---|---|---|---|---|---|
| Mean ± SD | Range | Mean ± SD | Range | Mean ± SD | Range | p | |
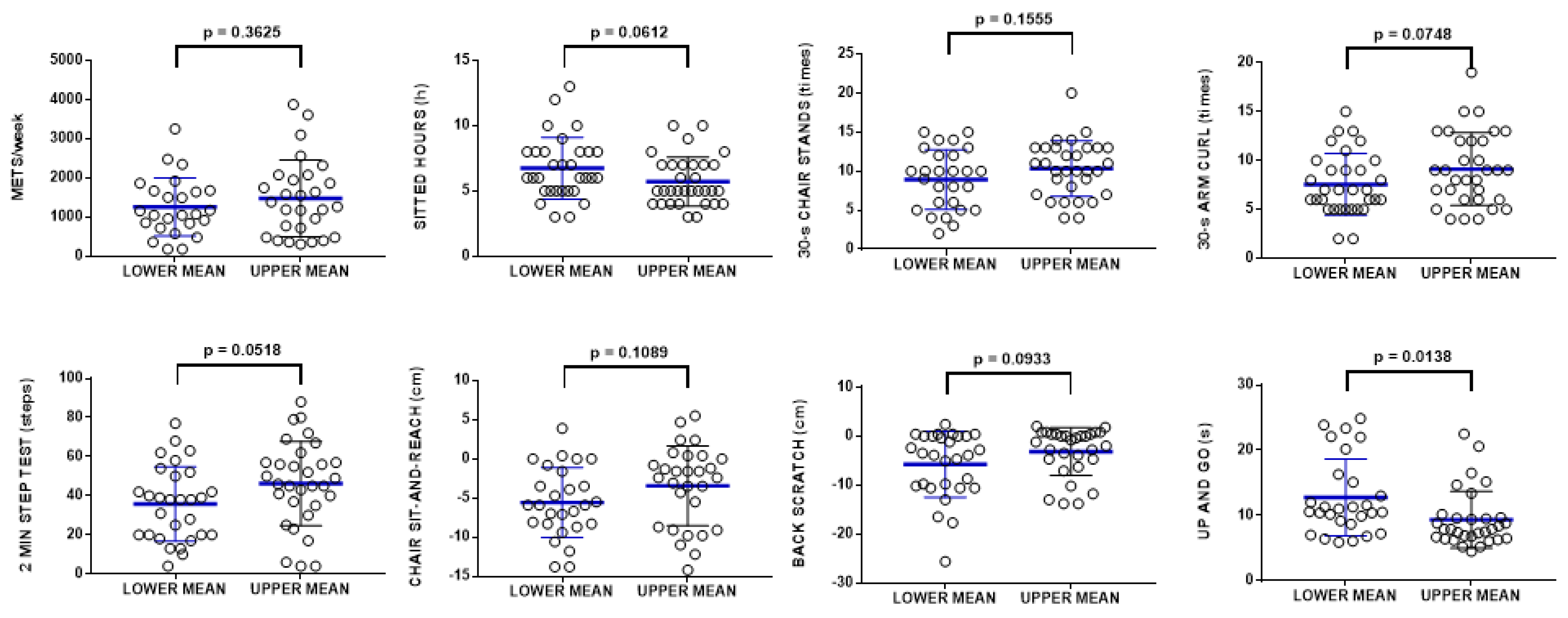
| METS/week | 1374 ± 874 | 180–3879 | 2129 ± 857 | 918–3879 | 1103 ± 714 | 180–3252 | <0.0001 * |
| 30 s CHAIR STAND (times) | 9.67 ± 3.72 | 2–20 | 10.57 ± 3.78 | 4–15 | 9.39 ± 3.70 | 2–20 | 0.3033 |
| 30 s ARM CURL (times) | 8.34 ± 3.51 | 2–19 | 8.47 ± 3.20 | 4–15 | 8.30 ± 3.65 | 2–19 | 0.8636 |
| 2 MIN STEP TEST (steps) | 41.2 ± 20.9 | 4–88 | 48.0 ± 27.0 | 4–88 | 39.0 ± 18.3 | 4–79 | 0.1492 |
| CHAIR SIT-AND-REACH (cm) | −4.48 ± 6.89 | −14.2–5.5 | −3.70 ± 5.09 | −12.2–3.9 | −4.68 ± 4.87 | −14.2–5.5 | 0.5403 |
| BACK SCRATCH (cm) | −4.43 ± 5.96 | −25.6–2.4 | −2.85 ± 4.31 | −13–1.8 | −4.82 ± 6.31 | −25.6–2.4 | 0.2806 |
| UP AND GO (s) | 10.9 ± 5.4 | 4.5–24.9 | 10.3 ± 5.7 | 5.2–22.5 | 11.2 ± 5.3 | 4.5–24.9 | 0.5796 |
| SITTING TIME (h) | 6.22 ± 2.17 | 3–13 | 5.35 ± 1.84 | 3–10 | 6.54 ± 2.22 | 3–13 | 0.0529 |
| Parameter | Total Population (n = 64) | MEN (n = 17) | WOMEN (n = 47) | |||
|---|---|---|---|---|---|---|
| Pearson r | p | Pearson r | p | Pearson r | p | |
| METS/week | 0.148 | 0.2902 | 0.523 | 0.0551 | −0.023 | 0.8883 |
| 30 s CHAIR STAND (times) | 0.372 | 0.0041 | 0.191 | 0.5138 | 0.344 | 0.02 |
| 30 s ARM CURL (times) | 0.45 | 0.0002 | 0.344 | 0.1766 | 0.428 | 0.0027 |
| 2 MIN STEP TEST (steps) | 0.473 | 0.0001 | 0.416 | 0.1233 | 0.451 | 0.0019 |
| CHAIR SIT-AND-REACH (cm) | 0.214 | 0.114 | −0.079 | 0.8087 | 0.325 | 0.0314 |
| BACK SCRATCH (cm) | 0.128 | 0.3334 | 0.1628 | 0.5782 | 0.158 | 0.2929 |
| UP AND GO (s) | −0.513 | <0.0001 | −0.363 | 0.1838 | −0.514 | 0.0004 |
| SITTING TIME (h) | −0.405 | 0.001 | −0.157 | 0.5473 | −0.399 | 0.006 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
de la Bella-Garzón, R.; Fernández-Portero, C.; Alarcón, D.; Amián, J.G.; López-Lluch, G. Levels of Plasma Coenzyme Q10 Are Associated with Physical Capacity and Cardiovascular Risk in the Elderly. Antioxidants 2022, 11, 279. https://doi.org/10.3390/antiox11020279
de la Bella-Garzón R, Fernández-Portero C, Alarcón D, Amián JG, López-Lluch G. Levels of Plasma Coenzyme Q10 Are Associated with Physical Capacity and Cardiovascular Risk in the Elderly. Antioxidants. 2022; 11(2):279. https://doi.org/10.3390/antiox11020279
Chicago/Turabian Stylede la Bella-Garzón, Rocío, Cristina Fernández-Portero, David Alarcón, Josué G. Amián, and Guillermo López-Lluch. 2022. "Levels of Plasma Coenzyme Q10 Are Associated with Physical Capacity and Cardiovascular Risk in the Elderly" Antioxidants 11, no. 2: 279. https://doi.org/10.3390/antiox11020279
APA Stylede la Bella-Garzón, R., Fernández-Portero, C., Alarcón, D., Amián, J. G., & López-Lluch, G. (2022). Levels of Plasma Coenzyme Q10 Are Associated with Physical Capacity and Cardiovascular Risk in the Elderly. Antioxidants, 11(2), 279. https://doi.org/10.3390/antiox11020279






