Microbial Phenolic Metabolites in Urine Are Inversely Linked to Certain Features of Metabolic Syndrome in Spanish Adolescents
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Assessment of Cardiometabolic Parameters
2.3. Determination of Urinary Phenolic Metabolites
2.3.1. Reagents and Standards
2.3.2. Urine Sample Collection and Treatment for MPM Analysis
2.3.3. Chromatographic Conditions
2.3.4. Creatinine Analysis
2.4. Assessment of Covariates
2.5. Statistical Analysis
3. Results
3.1. Participant Characteristics
3.2. Association between MPM and Cardiometabolic Health Parameters
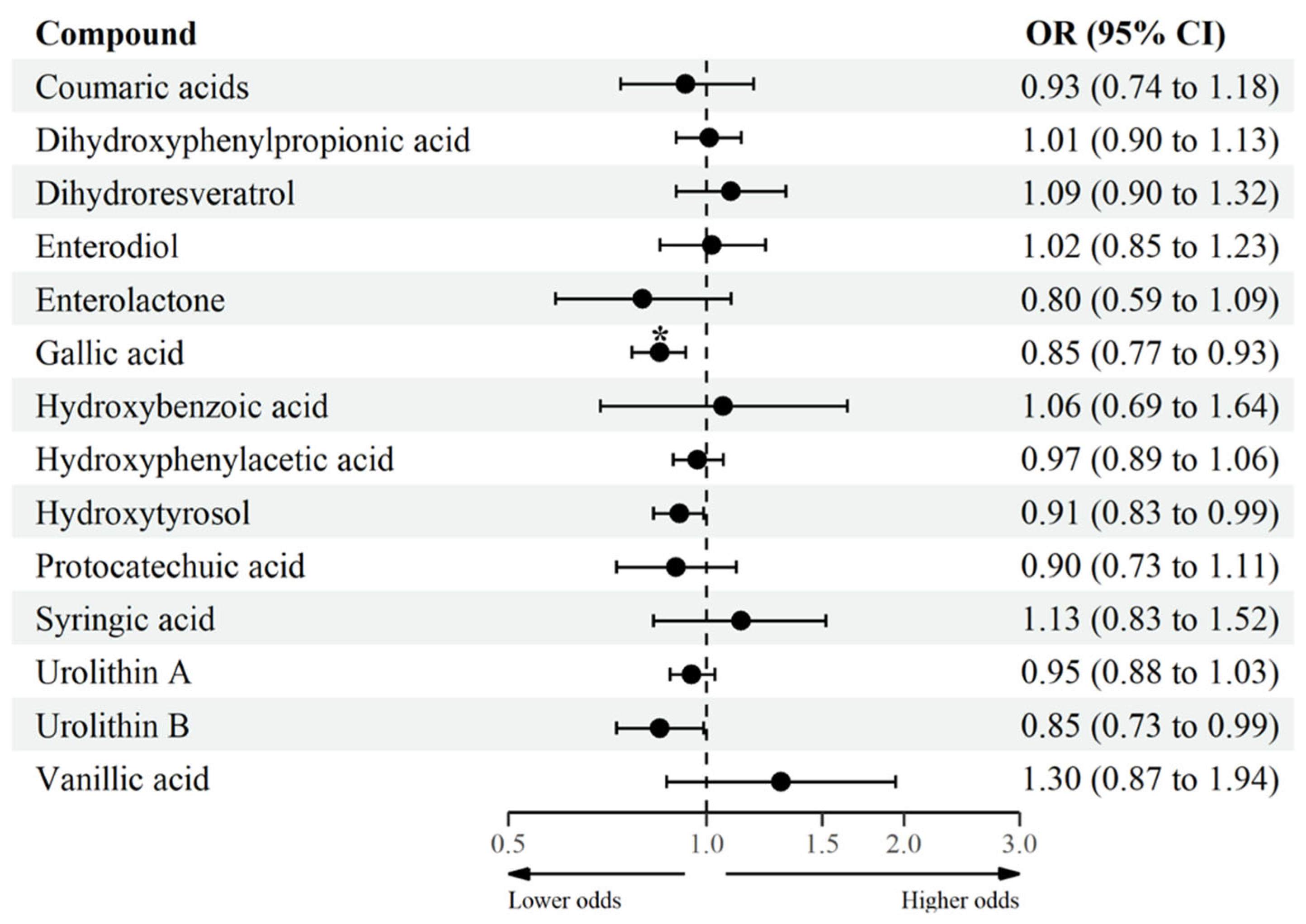
3.3. Association between MPM and MetS and Its Clinical Features
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BG | blood glucose |
| CI | confidence interval |
| DBP | diastolic blood pressure |
| FDR | false discovery rate |
| HDL-c | high-density lipoprotein cholesterol |
| HPLC-LTQ-Orbitrap-HRMS | High-performance liquid chromatography coupled to a linear trap quadrupole Orbitrap high-resolution mass spectrometer |
| LOQ | limit of quantitation |
| MAP | mean arterial pressure |
| MAD | mean absolute deviation method |
| MetS | metabolic syndrome |
| MS | mass spectra |
| MPM | microbial phenolic metabolites |
| OR | odds ratio |
| SBP | systolic blood pressure |
| TG | triglycerides |
| UM | Urinary metabotypes |
| WC | waist circumference |
References
- Zimmet, P.; Alberti, G.; Kaufman, F.; Tajima, N.; Silink, M.; Arslanian, S.; Wong, G.; Bennett, P.; Shaw, J.; Caprio, S. The Metabolic Syndrome in Children and Adolescents. Lancet 2007, 369, 2059–2061. [Google Scholar] [CrossRef]
- Magliano, D.J.; Zimmet, P.; Shaw, J.E. Classification of Diabetes Mellitus and Other Categories of Glucose Intolerance. In International Textbook of Diabetes Mellitus; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2015; pp. 1–16. [Google Scholar] [CrossRef]
- Magnussen, C.G.; Koskinen, J.; Chen, W.; Thomson, R.; Schmidt, M.D.; Srinivasan, S.R.; Kivimäki, M.; Mattsson, N.; Kähönen, M.; Laitinen, T.; et al. Pediatric Metabolic Syndrome Predicts Adulthood Metabolic Syndrome, Subclinical Atherosclerosis, and Type 2 Diabetes Mellitus but Is No Better Than Body Mass Index Alone. Circulation 2010, 122, 1604–1611. [Google Scholar] [CrossRef] [PubMed]
- Noubiap, J.J.; Nansseu, J.R.; Lontchi-Yimagou, E.; Nkeck, J.R.; Nyaga, U.F.; Ngouo, A.T.; Tounouga, D.N.; Tianyi, F.L.; Foka, A.J.; Ndoadoumgue, A.L.; et al. Global, Regional, and Country Estimates of Metabolic Syndrome Burden in Children and Adolescents in 2020: A Systematic Review and Modelling Analysis. Lancet Child Adolesc. Health 2022, 6, 158–170. [Google Scholar] [CrossRef]
- Després, J.-P.; Lemieux, I.; Bergeron, J.; Pibarot, P.; Mathieu, P.; Larose, E.; Rodés-Cabau, J.; Bertrand, O.F.; Poirier, P. Abdominal Obesity and the Metabolic Syndrome: Contribution to Global Cardiometabolic Risk. Arter. Thromb. Vasc. Biol. 2008, 28, 1039–1049. [Google Scholar] [CrossRef] [PubMed]
- Casas, L.; Fernández, M.F.; Llop, S.; Guxens, M.; Ballester, F.; Olea, N.; Irurzun, M.B.; Rodríguez, L.S.M.; Riaño, I.; Tardón, A.; et al. Urinary Concentrations of Phthalates and Phenols in a Population of Spanish Pregnant Women and Children. Environ. Int. 2011, 37, 858–866. [Google Scholar] [CrossRef]
- Güil-Oumrait, N.; Cano-Sancho, G.; Montazeri, P.; Stratakis, N.; Warembourg, C.; Lopez-Espinosa, M.J.; Vioque, J.; Santa-Marina, L.; Jimeno-Romero, A.; Ventura, R.; et al. Prenatal Exposure to Mixtures of Phthalates and Phenols and Body Mass Index and Blood Pressure in Spanish Preadolescents. Environ. Int. 2022, 169, 107527. [Google Scholar] [CrossRef]
- Martínez-González, M.A.; Gea, A.; Ruiz-Canela, M. The Mediterranean Diet and Cardiovascular Health. Circ. Res. 2019, 124, 779–798. [Google Scholar] [CrossRef]
- Hurtado-Barroso, S.; Quifer-Rada, P.; de Alvarenga, J.F.R.; Pérez-Fernández, S.; Tresserra-Rimbau, A.; Lamuela-Raventos, R.M. Changing to a Low-Polyphenol Diet Alters Vascular Biomarkers in Healthy Men after Only Two Weeks. Nutrients 2018, 10, 1766. [Google Scholar] [CrossRef]
- Tresserra-Rimbau, A.; Rimm, E.B.; Medina-Remón, A.; Martínez-González, M.A.; de la Torre, R.; Corella, D.; Salas-Salvadó, J.; Gómez-Gracia, E.; Lapetra, J.; Arós, F.; et al. Inverse Association between Habitual Polyphenol Intake and Incidence of Cardiovascular Events in the PREDIMED Study. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 639–647. [Google Scholar] [CrossRef]
- Zhang, H.; Tsao, R. Dietary Polyphenols, Oxidative Stress and Antioxidant and Anti-Inflammatory Effects. Curr. Opin. Food Sci. 2016, 8, 33–42. [Google Scholar] [CrossRef]
- Loo, Y.T.; Howell, K.; Chan, M.; Zhang, P.; Ng, K. Modulation of the Human Gut Microbiota by Phenolics and Phenolic Fiber-Rich Foods. Compr. Rev. Food Sci. Food Saf. 2020, 19, 1268–1298. [Google Scholar] [CrossRef]
- Marhuenda-Muñoz, M.; Laveriano-Santos, E.P.; Tresserra-Rimbau, A.; Lamuela-Raventós, R.M.; Martínez-Huélamo, M.; Vallverdú-Queralt, A. Microbial Phenolic Metabolites: Which Molecules Actually Have an Effect on Human Health? Nutrients 2019, 11, 2725. [Google Scholar] [CrossRef] [PubMed]
- Mosele, J.I.; Macià, A.; Motilva, M.J. Metabolic and Microbial Modulation of the Large Intestine Ecosystem by Non-Absorbed Diet Phenolic Compounds: A Review. Molecules 2015, 20, 17429–17468. [Google Scholar] [CrossRef] [PubMed]
- Teng, H.; Chen, L. Polyphenols and Bioavailability: An Update. Crit. Rev. Food Sci. Nutr. 2019, 59, 2040–2051. [Google Scholar] [CrossRef] [PubMed]
- López-Yerena, A.; Domínguez-López, I.; Vallverdú-Queralt, A.; Pérez, M.; Jáuregui, O.; Escribano-Ferrer, E.; Lamuela-Raventós, R.M. Metabolomics Technologies for the Identification and Quantification of Dietary Phenolic Compound Metabolites: An Overview. Antioxidants 2021, 10, 846. [Google Scholar] [CrossRef] [PubMed]
- Selma, M.V.; González-Sarrías, A.; Salas-Salvadó, J.; Andrés-Lacueva, C.; Alasalvar, C.; Örem, A.; Tomás-Barberán, F.A.; Espín, J.C. The Gut Microbiota Metabolism of Pomegranate or Walnut Ellagitannins Yields Two Urolithin-Metabotypes That Correlate with Cardiometabolic Risk Biomarkers: Comparison between Normoweight, Overweight-Obesity and Metabolic Syndrome. Clin. Nutr. 2018, 37, 897–905. [Google Scholar] [CrossRef]
- Romo-Vaquero, M.; Cortés-Martín, A.; Loria-Kohen, V.; Ramírez-de-Molina, A.; García-Mantrana, I.; Collado, M.C.; Espín, J.C.; Selma, M.V. Deciphering the Human Gut Microbiome of Urolithin Metabotypes: Association with Enterotypes and Potential Cardiometabolic Health Implications. Mol. Nutr. Food Res. 2019, 63, 1800958. [Google Scholar] [CrossRef]
- Fernandez-Jimenez, R.; Santos-Beneit, G.; Tresserra-Rimbau, A.; Bodega, P.; de Miguel, M.; de Cos-Gandoy, A.; Rodríguez, C.; Carral, V.; Orrit, X.; Haro, D.; et al. Rationale and Design of the School-Based SI! Program to Face Obesity and Promote Health among Spanish Adolescents: A Cluster-Randomized Controlled Trial. Am. Heart J. 2019, 215, 27–40. [Google Scholar] [CrossRef]
- Laveriano-Santos, E.P.; Marhuenda-Muñoz, M.; Vallverdú-Queralt, A.; Martínez-Huélamo, M.; Tresserra-Rimbau, A.; Miliarakis, E.; Arancibia-Riveros, C.; Jáuregui, O.; Ruiz-León, A.M.; Castro-Baquero, S.; et al. Identification and Quantification of Urinary Microbial Phenolic Metabolites by HPLC-ESI-LTQ-Orbitrap-HRMS and Their Relationship with Dietary Polyphenols in Adolescents. Antioxidants 2022, 11, 1167. [Google Scholar] [CrossRef]
- Sharma, A.K.; Metzger, D.L.; Daymont, C.; Hadjiyannakis, S.; Rodd, C.J. LMS Tables for Waist-Circumference and Waist-Height Ratio Z-Scores in Children Aged 5–19 y in NHANES III: Association with Cardio-Metabolic Risks. Pediatr. Res. 2015, 78, 723–729. [Google Scholar] [CrossRef]
- National High Blood Pressure Education Program Working Group on High Blood Pressure in Children and Adolescents The Fourth Report on the Diagnosis, Evaluation, and Treatment of High Blood Pressure in Children and Adolescents. Pediatrics 2004, 114, 555–576. [CrossRef]
- Shafiee, G.; Kelishadi, R.; Heshmat, R.; Qorbani, M.; Motlagh, M.E.; Aminaee, T.; Ardalan, G.; Taslimi, M.; Poursafa, P.; Larijani, B. First Report on the Validity of a Continuous Metabolic Syndrome Score as an Indicator for Metabolic Syndrome in a National Sample of Paediatric Population—The CASPIAN-III Study. Endokrynol. Pol. 2013, 64, 278–284. [Google Scholar] [CrossRef] [PubMed]
- Medina-Remón, A.; Barrionuevo-González, A.; Zamora-Ros, R.; Andres-Lacueva, C.; Estruch, R.; Martínez-González, M.-Á.; Diez-Espino, J.; Lamuela-Raventos, R.M. Rapid Folin–Ciocalteu Method Using Microtiter 96-Well Plate Cartridges for Solid Phase Extraction to Assess Urinary Total Phenolic Compounds, as a Biomarker of Total Polyphenols Intake. Anal. Chim. Acta 2009, 634, 54–60. [Google Scholar] [CrossRef]
- Juton, C.; Castro-Barquero, S.; Casas, R.; Freitas, T.; Ruiz-León, A.M.; Crovetto, F.; Domenech, M.; Crispi, F.; Vieta, E.; Gratacós, E.; et al. Reliability and Concurrent and Construct Validity of a Food Frequency Questionnaire for Pregnant Women at High Risk to Develop Fetal Growth Restriction. Nutrients 2021, 13, 1629. [Google Scholar] [CrossRef] [PubMed]
- Moreiras, O.; Carvajal, H.; Cabrera, L.; Cuadrado, C. Tablas de Composición de Alimentos: Guía de Prácticas, 19th ed.; Editorial Pirámide: Madrid, Spain, 2018; ISBN 978-84-368-3947-0. [Google Scholar]
- Farrán, A.; Zamora, R.C.P. Tablas de Composición de Alimentos del CESNID; McGraw-Hill Interamericana, Edicions Universitat de Barcelona: Barcelona, Spain, 2003. [Google Scholar]
- Chandler, J.L.; Brazendale, K.; Beets, M.W.; Mealing, B.A. Classification of Physical Activity Intensities Using a Wrist-Worn Accelerometer in 8-12-Year-Old Children. Pediatr. Obes. 2016, 11, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, N.; Sanchez, C.E.; Vera, J.A.; Perez, W.; Thalabard, J.-C.; Rieu, M. A Physical Activity Questionnaire: Reproducibility and Validity. J. Sport. Sci. Med. 2007, 6, 505–518. [Google Scholar]
- Bull, F.C.; Al-Ansari, S.S.; Biddle, S.; Borodulin, K.; Buman, M.P.; Cardon, G.; Carty, C.; Chaput, J.-P.; Chastin, S.; Chou, R.; et al. World Health Organization 2020 Guidelines on Physical Activity and Sedentary Behaviour. Br. J. Sport. Med. 2020, 54, 1451–1462. [Google Scholar] [CrossRef]
- Tanner, J.M.; Whitehouse, R.H. Clinical Longitudinal Standards for Height, Weight, Height Velocity, Weight Velocity, and Stages of Puberty. Arch. Dis. Child 1976, 51, 170–179. [Google Scholar] [CrossRef]
- INE: Instituto Nacional de Estadística Encuesta Anual de Estructura Salarial. Available online: https://www.ine.es/jaxiT3/Datos.htm?t=9948 (accessed on 19 June 2022).
- Rusiman, M.S.; Nor, S.N.M.; Suparman, S.; Razali, S.N.A.M. Robust Method in Multiple Linear Regression Model on Diabetes Patients. Math. Stat. 2020, 8, 36–39. [Google Scholar] [CrossRef]
- Benjamini, Y.; Hochberg, Y. Controlling the False Discovery Rate: A Practical and Powerful Approach to Multiple Testing. J. R. Stat. Soc. Ser. B (Methodol.) 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Kosuru, R.Y.; Roy, A.; Das, S.K.; Bera, S. Gallic Acid and Gallates in Human Health and Disease: Do Mitochondria Hold the Key to Success? Mol. Nutr. Food Res. 2018, 62, 1700699. [Google Scholar] [CrossRef] [PubMed]
- Esteban-Torres, M.; Santamaría, L.; Cabrera-Rubio, R.; Plaza-Vinuesa, L.; Crispie, F.; de Las Rivas, B.; Cotter, P.; Muñoz, R. A Diverse Range of Human Gut Bacteria Have the Potential To Metabolize the Dietary Component Gallic Acid. Appl. Environ. Microbiol. 2018, 84, e01558-18. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Moon, J.H.; Kim, Y.M.; Huh, J.Y. Epigallocatechin Exerts Anti-Obesity Effect in Brown Adipose Tissue. Chem. Biodivers. 2019, 16, e1900347. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.S.; Zhang, J.; Zhang, L.; Huang, J.; Wang, Y. Mechanisms of Body Weight Reduction and Metabolic Syndrome Alleviation by Tea. Mol. Nutr. Food Res. 2016, 60, 160–174. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, M.P.; Sugita, M.; Fukuzawa, Y.; Okubo, T. Physiological Effects of Epigallocatechin-3-Gallate (EGCG) on Energy Expenditure for Prospective Fat Oxidation in Humans: A Systematic Review and Meta-Analysis. J. Nutr. Biochem. 2017, 43, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Auvichayapat, P.; Prapochanung, M.; Tunkamnerdthai, O.; Sripanidkulchai, B.; Auvichayapat, N.; Thinkhamrop, B.; Kunhasura, S.; Wongpratoom, S.; Sinawat, S.; Hongprapas, P. Effectiveness of Green Tea on Weight Reduction in Obese Thais: A Randomized, Controlled Trial. Physiol. Behav. 2008, 93, 486–491. [Google Scholar] [CrossRef]
- Eid, H.M.; Wright, M.L.; Anil Kumar, N.V.; Qawasmeh, A.; Hassan, S.T.S.; Mocan, A.; Nabavi, S.M.; Rastrelli, L.; Atanasov, A.G.; Haddad, P.S. Significance of Microbiota in Obesity and Metabolic Diseases and the Modulatory Potential by Medicinal Plant and Food Ingredients. Front. Pharm. 2017, 8, 387. [Google Scholar] [CrossRef]
- Bento-Silva, A.; Koistinen, V.M.; Mena, P.; Bronze, M.R.; Hanhineva, K.; Sahlstrøm, S.; Kitrytė, V.; Moco, S.; Aura, A.M. Factors Affecting Intake, Metabolism and Health Benefits of Phenolic Acids: Do We Understand Individual Variability? Eur. J. Nutr. 2019, 59, 1275–1293. [Google Scholar] [CrossRef]
- Kasprzak-Drozd, K.; Oniszczuk, T.; Stasiak, M.; Oniszczuk, A. Beneficial Effects of Phenolic Compounds on Gut Microbiota and Metabolic Syndrome. Int. J. Mol. Sci. 2021, 22, 3715. [Google Scholar] [CrossRef]
- Vishnu Prasad, C.N.; Anjana, T.; Banerji, A.; Gopalakrishnapillai, A. Gallic Acid Induces GLUT4 Translocation and Glucose Uptake Activity in 3T3-L1 Cells. FEBS Lett. 2010, 584, 531–536. [Google Scholar] [CrossRef]
- Punithavathi, V.R.; Prince, P.S.M.; Kumar, R.; Selvakumari, J. Antihyperglycaemic, Antilipid Peroxidative and Antioxidant Effects of Gallic Acid on Streptozotocin Induced Diabetic Wistar Rats. Eur. J. Pharm. 2011, 650, 465–471. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Simbo, S.Y.; Fang, C.; McAlister, L.; Roque, A.; Banerjee, N.; Talcott, S.T.; Zhao, H.; Kreider, R.B.; Mertens-Talcott, S.U. Açaí (Euterpe Oleracea Mart.) Beverage Consumption Improves Biomarkers for Inflammation but Not Glucose- or Lipid-Metabolism in Individuals with Metabolic Syndrome in a Randomized, Double-Blinded, Placebo-Controlled Clinical Trial. Food Funct. 2018, 9, 3097–3103. [Google Scholar] [CrossRef] [PubMed]
- Hasheminezhad, S.H.; Boozari, M.; Iranshahi, M.; Yazarlu, O.; Sahebkar, A.; Hasanpour, M.; Iranshahy, M. A Mechanistic Insight into the Biological Activities of Urolithins as Gut Microbial Metabolites of Ellagitannins. Phytother. Res. 2021, 36, 112–146. [Google Scholar] [CrossRef] [PubMed]
- Li, K.; Xiao, Y.; Bian, J.; Han, L.; He, C.; El-Omar, E.; Gong, L.; Wang, M. Ameliorative Effects of Gut Microbial Metabolite Urolithin A on Pancreatic Diseases. Nutrients 2022, 14, 2549. [Google Scholar] [CrossRef]
- García-Muñoz, C.; Hernández, L.; Pérez, A.; Vaillant, F. Diversity of Urinary Excretion Patterns of Main Ellagitannins’ Colonic Metabolites after Ingestion of Tropical Highland Blackberry (Rubus adenotrichus) Juice. Food Res. Int. 2014, 55, 161–169. [Google Scholar] [CrossRef]
- Istas, G.; Feliciano, R.P.; Weber, T.; Garcia-Villalba, R.; Tomas-Barberan, F.; Heiss, C.; Rodriguez-Mateos, A. Plasma Urolithin Metabolites Correlate with Improvements in Endothelial Function after Red Raspberry Consumption: A Double-Blind Randomized Controlled Trial. Arch. Biochem. Biophys. 2018, 651, 43–51. [Google Scholar] [CrossRef]
- García-Conesa, M.T.; Chambers, K.; Combet, E.; Pinto, P.; Garcia-Aloy, M.; Andrés-Lacueva, C.; de Pascual-Teresa, S.; Mena, P.; Ristic, A.K.; Hollands, W.J.; et al. Meta-Analysis of the Effects of Foods and Derived Products Containing Ellagitannins and Anthocyanins on Cardiometabolic Biomarkers: Analysis of Factors Influencing Variability of the Individual Responses. Int. J. Mol. Sci. 2018, 19, 694. [Google Scholar] [CrossRef]
- González-Sarrías, A.; Larrosa, M.; Tomás-Barberán, F.A.; Dolara, P.; Espín, J.C. NF-KappaB-Dependent Anti-Inflammatory Activity of Urolithins, Gut Microbiota Ellagic Acid-Derived Metabolites, in Human Colonic Fibroblasts. Br. J. Nutr. 2010, 104, 503–512. [Google Scholar] [CrossRef]
- Giménez-Bastida, J.A.; González-Sarrías, A.; Larrosa, M.; Tomás-Barberán, F.; Espín, J.C.; García-Conesa, M.T. Ellagitannin Metabolites, Urolithin A Glucuronide and Its Aglycone Urolithin A, Ameliorate TNF-α-Induced Inflammation and Associated Molecular Markers in Human Aortic Endothelial Cells. Mol. Nutr. Food Res. 2012, 56, 784–796. [Google Scholar] [CrossRef]
- Spigoni, V.; Mena, P.; Cito, M.; Fantuzzi, F.; Bonadonna, R.C.; Brighenti, F.; Cas, A.D.; Rio, D. del Effects on Nitric Oxide Production of Urolithins, Gut-Derived Ellagitannin Metabolites, in Human Aortic Endothelial Cells. Molecules 2016, 21, 1009. [Google Scholar] [CrossRef]
- Cortés-Martín, A.; Colmenarejo, G.; Selma, M.V.; Espín, J.C. Genetic Polymorphisms, Mediterranean Diet and Microbiota-Associated Urolithin Metabotypes Can Predict Obesity in Childhood-Adolescence. Sci. Rep. 2020, 10, 7850. [Google Scholar] [CrossRef] [PubMed]
- Bugger, H.; Abel, E.D. Molecular Mechanisms of Diabetic Cardiomyopathy. Diabetologia 2014, 57, 660. [Google Scholar] [CrossRef]
- Sala, R.; Mena, P.; Savi, M.; Brighenti, F.; Crozier, A.; Miragoli, M.; Stilli, D.; del Rio, D. Urolithins at Physiological Concentrations Affect the Levels of Pro-Inflammatory Cytokines and Growth Factor in Cultured Cardiac Cells in Hyperglucidic Conditions. J. Funct. Foods 2015, 15, 97–105. [Google Scholar] [CrossRef]
- Williamson, G.; Kay, C.D.; Crozier, A. The Bioavailability, Transport, and Bioactivity of Dietary Flavonoids: A Review from a Historical Perspective. Compr. Rev. Food Sci. Food Saf. 2018, 17, 1054–1112. [Google Scholar] [CrossRef] [PubMed]
- Frankenfeld, C.L. Cardiometabolic Risk Factors Are Associated with High Urinary Enterolactone Concentration, Independent of Urinary Enterodiol Concentration and Dietary Fiber Intake in Adults. J. Nutr. 2014, 144, 1445–1453. [Google Scholar] [CrossRef] [PubMed]
- Gibney, E.R.; Milenkovic, D.; Combet, E.; Ruskovska, T.; Greyling, A.; González-Sarrías, A.; de Roos, B.; Tomás-Barberán, F.; Morand, C.; Rodriguez-Mateos, A. Factors Influencing the Cardiometabolic Response to (Poly)Phenols and Phytosterols: A Review of the COST Action POSITIVe Activities. Eur. J. Nutr. 2019, 58, 37–47. [Google Scholar] [CrossRef]
- Ávila-Román, J.; Soliz-Rueda, J.R.; Bravo, F.I.; Aragonès, G.; Suárez, M.; Arola-Arnal, A.; Mulero, M.; Salvadó, M.J.; Arola, L.; Torres-Fuentes, C.; et al. Phenolic Compounds and Biological Rhythms: Who Takes the Lead? Trends Food Sci. Technol. 2021, 113, 77–85. [Google Scholar] [CrossRef]



| Characteristics | Total |
|---|---|
| Female, n (%) | 304 (54) |
| Age, years | 12.0 ± 0.4 |
| WC, cm | 73.6 ± 11.4 |
| SBP, mm Hg | 105.3 ± 11.2 |
| DBP, mm Hg | 62.5 ± 9.3 |
| BG, mg/dL | 102.9 ± 14.2 |
| TG, mg/dL | 87.9 ± 47.2 |
| HDL-c, mg/dL | 62.4 ± 16.5 |
| MetS score | 0.01 ± 3.04 |
| MetS, n (%) | 16 (3) |
| Clinical features | |
| Abdominal obesity, n (%) | 122 (22) |
| High blood pressure, n (%) | 2 (0.4) |
| Low HDL-c, n (%) | 32 (6) |
| High TG, n (%) | 43 (8) |
| High BG, n (%) | 159 (28) |
| Total energy intake, kcal/day | 2492.1 ± 586.4 |
| Moderate-to-vigorous physical activity, n (%) | |
| <60 min/day | 156 (28) |
| ≥60 min/day | 404 (72) |
| Household income, n (%) | |
| Low | 173 (32) |
| Medium | 144 (27) |
| High | 216 (41) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Laveriano-Santos, E.P.; Quifer-Rada, P.; Marhuenda-Muñoz, M.; Arancibia-Riveros, C.; Vallverdú-Queralt, A.; Tresserra-Rimbau, A.; Ruiz-León, A.M.; Casas, R.; Estruch, R.; Bodega, P.; et al. Microbial Phenolic Metabolites in Urine Are Inversely Linked to Certain Features of Metabolic Syndrome in Spanish Adolescents. Antioxidants 2022, 11, 2191. https://doi.org/10.3390/antiox11112191
Laveriano-Santos EP, Quifer-Rada P, Marhuenda-Muñoz M, Arancibia-Riveros C, Vallverdú-Queralt A, Tresserra-Rimbau A, Ruiz-León AM, Casas R, Estruch R, Bodega P, et al. Microbial Phenolic Metabolites in Urine Are Inversely Linked to Certain Features of Metabolic Syndrome in Spanish Adolescents. Antioxidants. 2022; 11(11):2191. https://doi.org/10.3390/antiox11112191
Chicago/Turabian StyleLaveriano-Santos, Emily P., Paola Quifer-Rada, María Marhuenda-Muñoz, Camila Arancibia-Riveros, Anna Vallverdú-Queralt, Anna Tresserra-Rimbau, Ana María Ruiz-León, Rosa Casas, Ramon Estruch, Patricia Bodega, and et al. 2022. "Microbial Phenolic Metabolites in Urine Are Inversely Linked to Certain Features of Metabolic Syndrome in Spanish Adolescents" Antioxidants 11, no. 11: 2191. https://doi.org/10.3390/antiox11112191
APA StyleLaveriano-Santos, E. P., Quifer-Rada, P., Marhuenda-Muñoz, M., Arancibia-Riveros, C., Vallverdú-Queralt, A., Tresserra-Rimbau, A., Ruiz-León, A. M., Casas, R., Estruch, R., Bodega, P., de Miguel, M., de Cos-Gandoy, A., Martínez-Gómez, J., Santos-Beneit, G., Fernández-Alvira, J. M., Fernández-Jiménez, R., & Lamuela-Raventós, R. M. (2022). Microbial Phenolic Metabolites in Urine Are Inversely Linked to Certain Features of Metabolic Syndrome in Spanish Adolescents. Antioxidants, 11(11), 2191. https://doi.org/10.3390/antiox11112191











