A Leaf Extract of Harrisonia abyssinica Ameliorates Neurobehavioral, Histological and Biochemical Changes in the Hippocampus of Rats with Aluminum Chloride-Induced Alzheimer’s Disease
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material and Extraction
2.2. HPLC-PDA-ESI-MS/MS
2.3. Total Phenolic Content and In Vitro Antioxidant Activities
2.4. Animals
2.5. Experimental Design
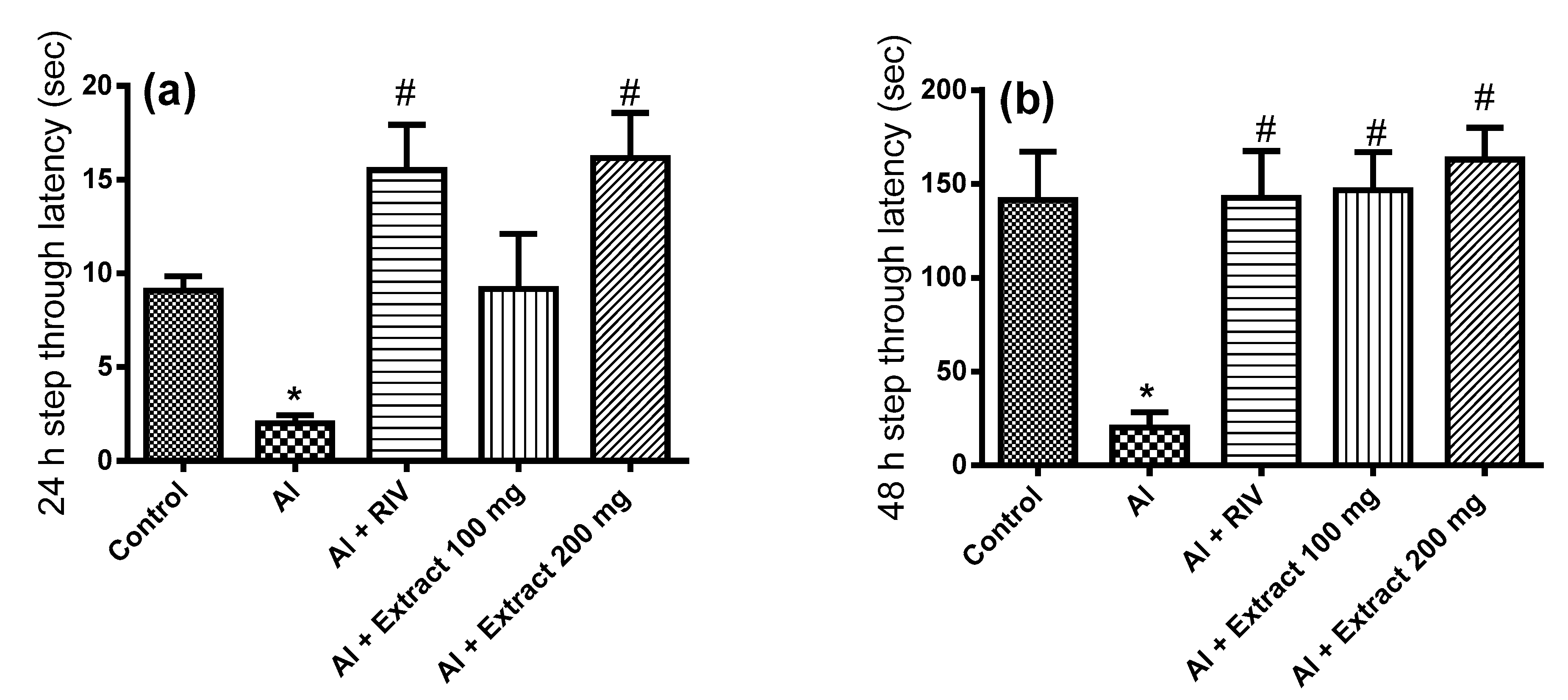
2.6. Behavioral Passive Avoidance Test
2.7. Biochemical Analysis
2.7.1. Quantification of Neurotransmitters in the Hippocampus
2.7.2. Estimation of TNF-α, MDA, IL-1 Beta and Caspase-3 in the Hippocampus
2.7.3. Estimation of Acetylcholine Esterase (AChE) Activity and Glutamate in the Hippocampus
2.7.4. Estimation of Extracellular Regulated Kinase (ERK) Level in the Hippocampus
2.8. Bax and Bcl2 Gene Expression Using RT-PCR
2.9. Hematoxylin and Eosin Staining of the Hippocampus
2.10. Congo Red Staining of the Hippocampus
2.11. Molecular Docking
2.12. Statistical Analysis
3. Results
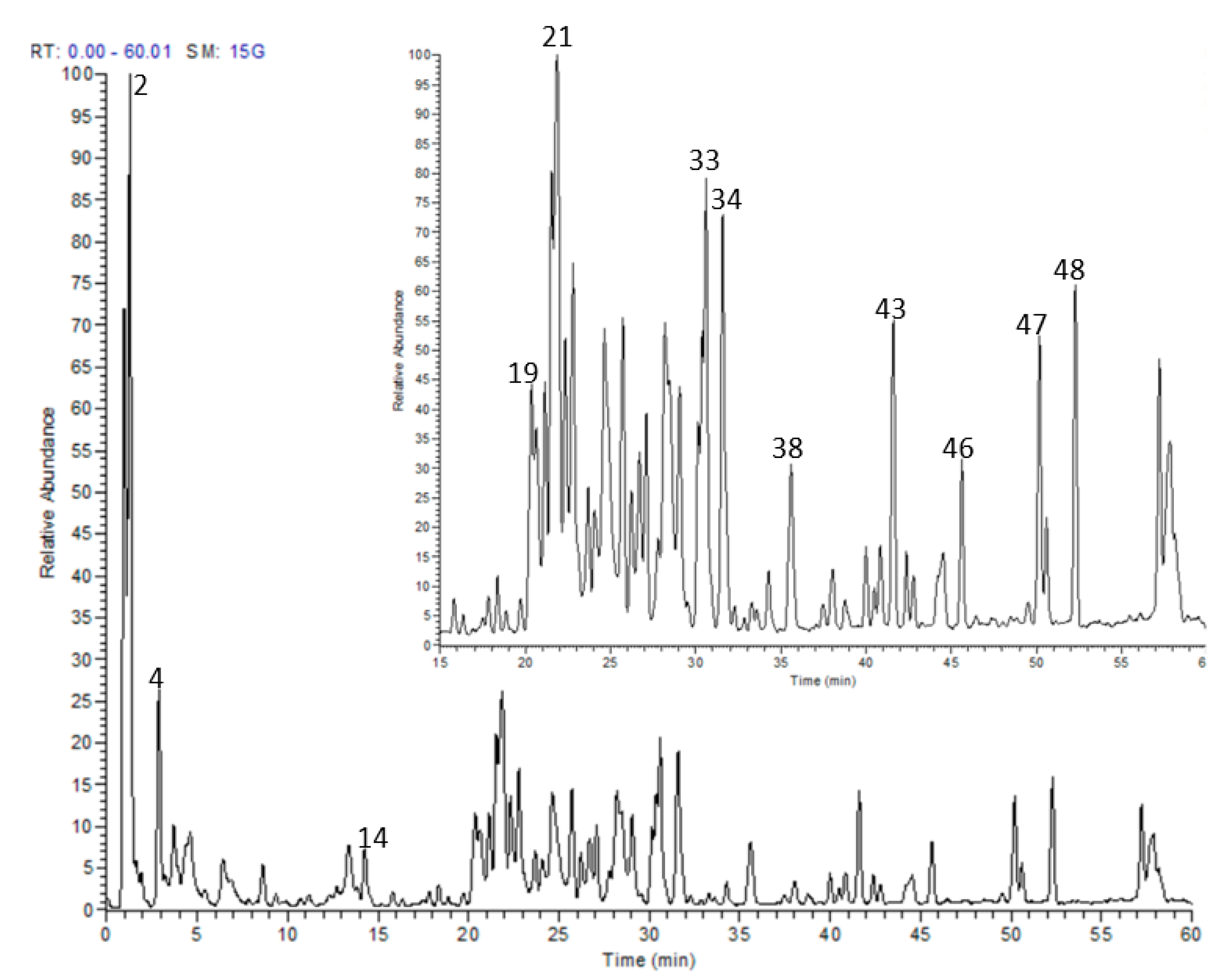
3.1. HPLC-MS/MS and 1HNMR
| No. | Rt | [M-H]− | MS/MS | Tentatively Identified Compound |
|---|---|---|---|---|
| 1 | 1.43 | 191 | 85, 127, 173 | Quinic acid |
| 2 | 1.63 | 343 | 169, 191 | Theogallin |
| 3 | 1.93 | 169 | 125 | Gallic acid |
| 4 | 2.97 | 353 | 179, 191 | Chlorogenic acid |
| 5 | 3.07 | 495 | 169, 343 | 3,4-di-O-galloylquinic acid |
| 6 | 3.65 | 337 | 119, 163 | p-coumaroylquinic acid |
| 7 | 4.89 | 647 | 343, 495 | 3,4,5-tri-O-galloylquinic acid |
| 8 | 6.97 | 477 | 169, 313, 433 | Coumaroyl galloylglucose |
| 9 | 8.64 | 183 | 125, 169, 183 | Methylgallate a |
| 10 | 9.35 | 301 | 229, 257, 301 | Ellagic acid |
| 11 | 11.21 | 953 | 301, 463, 633, 935 | Chebulagic acid b |
| 12 | 12.73 | 951 | 301, 613, 933 | Geraniin |
| 13 | 13.52 | 633 | 301, 463 | Galloyl-HHDP-hexoside |
| 14 | 14.62 | 453 | 169, 313, 327 | Pyrogallol-O-methylgalloyl glucose |
| 15 | 15.85 | 635 | 331, 483 | 1,2,6-trigalloyl glucose |
| 16 | 17.80 | 635 | 313, 465, 483 | 1,3,6-trigalloyl glucose |
| 17 | 18.87 | 537 | 169, 271, 313 | Sinapoyl galloyl glucose |
| 18 | 19.67 | 551 | 169, 271, 533 | Gallic acid glucoside derivative |
| 19 | 20.30 | 615 | 301, 463 | Quercetin 3-O-(6″-O-galloyl) glucoside |
| 20 | 21.04 | 449 | 179, 449 | Myricetin 3-O- pentoside |
| 21 | 21.81 | 615 | 179, 301, 463 | Quercetin 7-(6’’-galloylglucoside) |
| 22 | 22.37 | 431 | 269, 311, 341 | Isovitexin |
| 23 | 23.10 | 609 | 179, 301 | Rutin |
| 24 | 24.18 | 463 | 179, 255, 301 | Quercetin 3-O-glucoside |
| 25 | 23.93 | 539 | 169, 271, 313 | Gallic acid glucoside derivative |
| 26 | 24.59 | 599 | 169, 285, 447 | Kaempferol 3-O-(6’’-galloyl) glucopyranoside |
| 27 | 25.58 | 433 | 179, 301 | Quercetin 3-O-pentoside |
| 28 | 26.76 | 433 | 151, 179, 301 | Quercetin 5-O-pentoside |
| 29 | 27.12 | 447 | 151, 285 | Kaempferol 3-O-glucoside |
| 30 | 28.47 | 447 | 151, 285 | Kaempferol 5-O-glucoside |
| 31 | 29.04 | 599 | 169, 285, 313, 447 | Kaempferol galloyl glucoside |
| 32 | 30.13 | 417 | 179, 285 | Kaempferol 3-O-pentoside |
| 33 | 31.42 | 583 | 169, 341, 431, 565 | Isovitexin 2”-O-gallate |
| 34 | 31.62 | 431 | 269 | Apigenin 7-glucoside |
| 35 | 33.34 | 447 | 151, 300, 315 | Isorhamnetin 3-O-glucoside |
| 36 | 33.61 | 461 | 299 | Diosmetin-7-O-glucoside |
| 37 | 34.26 | 583 | 179, 301, 431, 463 | Quercetin 3-(6’’-p-hydroxybenzoylgalactoside) |
| 38 | 35.52 | 625 | 179, 301, 463 | Quercetin 3-O-(6’’-O-caffeoyl)-glucoside |
| 39 | 38.08 | 567 | 255, 285, 447 | Kaempferol 3-(6”-p-hydroxybenzoylgalactoside) |
| 40 | 38.76 | 609 | 285, 323, 447 | Kaempferol 3-O-(6’’-O-caffeoyl)-galactoside |
| 41 | 39.98 | 567 | 255, 285, 447 | Kaempferol 3-(6”-p-hydroxybenzoyl glucoside) |
| 42 | 40.81 | 609 | 179, 301, 463 | Quercetin 3-(3-p-coumaroylglucoside) |
| 43 | 41.56 | 609 | 179, 301, 463 | Quercetin 3-O-β-(6″-O-coumaroylglucoside) |
| 44 | 42.85 | 301 | 151, 179, 301 | Quercetin |
| 45 | 44.57 | 593 | 285, 447 | Kaempferol 3-(6′′-caffeoylglucoside) |
| 46 | 45.62 | 593 | 285, 447 | Kaempferol 3-(6′′-caffeoylrahmnoside) |
| 47 | 50.18 | 269 | 107, 149, 269 | Apigenin |
| 48 | 52.30 | 285 | 151, 285 | Kaempferol |
| 49 | 57.21 | 439 | 163, 377, 395 | p-coumaric acid derivative |
3.2. Total Phenolic Content and Antioxidant Activity In Vitro
3.3. Effect of H. abyssinica Treatment on Passive Avoidance Behavioral Test in AD Rats
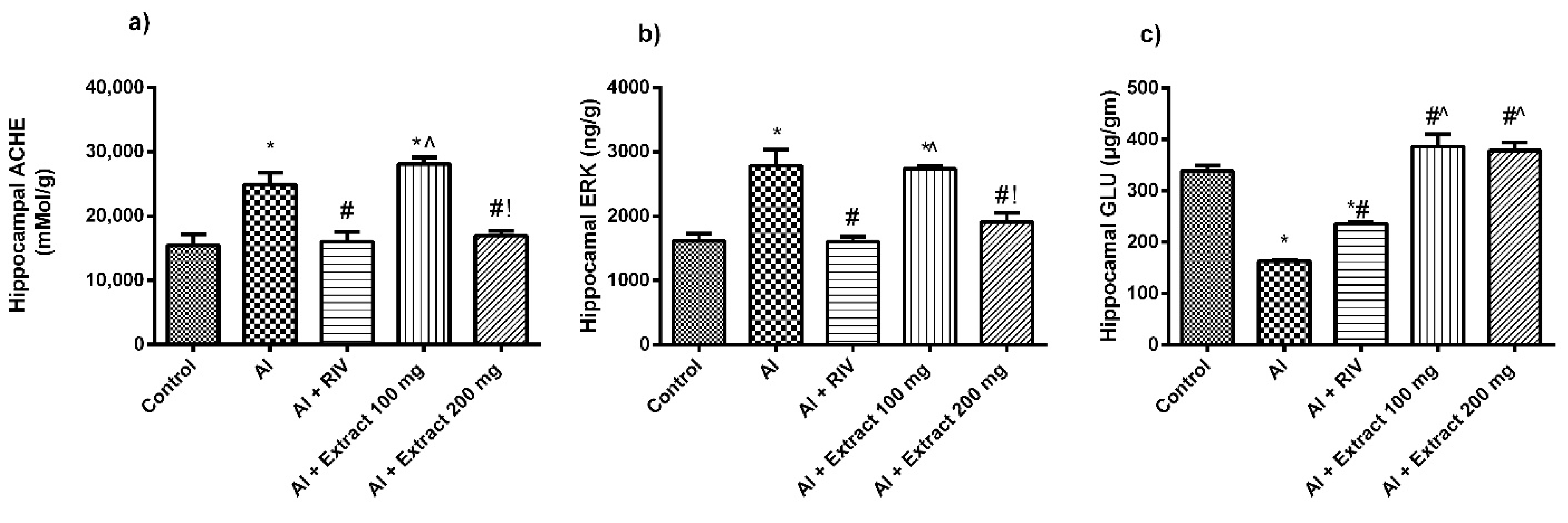
3.4. Effect of H. abyssinica Treatment on AChE Activity, ERK and Glutamate Levels in the Hippocampus
3.5. Effect of H. abyssinica Treatment on MDA, TNF-α, IL-1β and Caspase-3 Levels
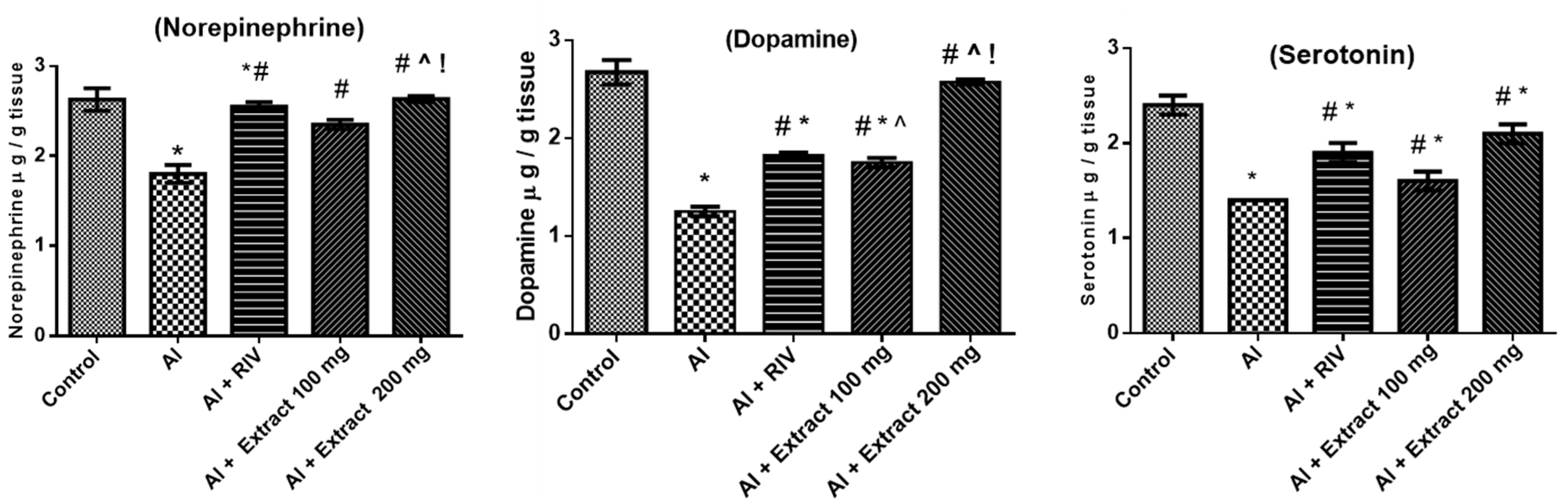
3.6. Effect of H. abyssinica Treatment on Norepinephrine, Dopamine and Serotonin Levels
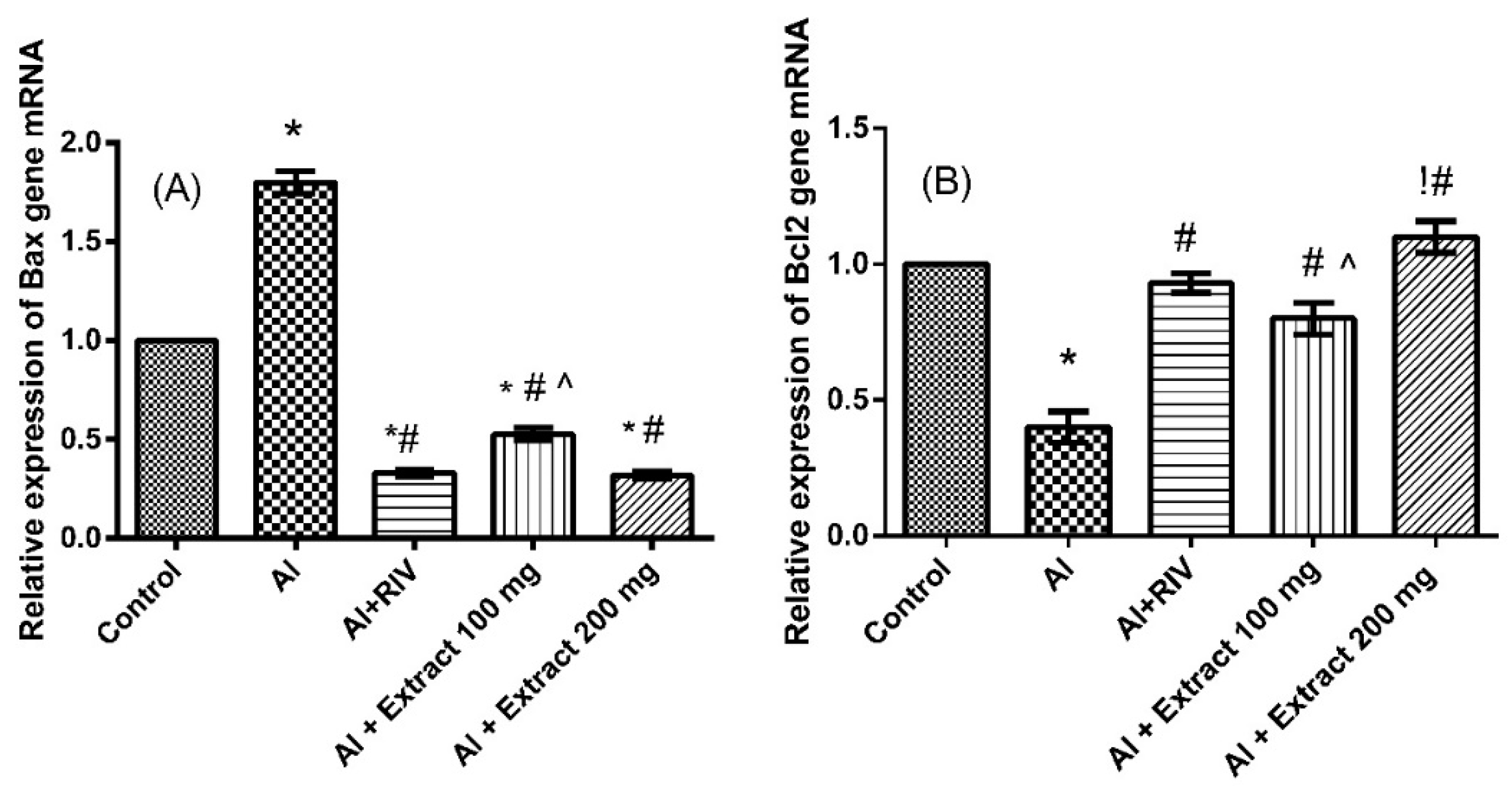
3.7. Effect of H. abyssinica Treatment on mRNA Expression Levels of Bax and Bcl-2 Gene in the Hippocampus
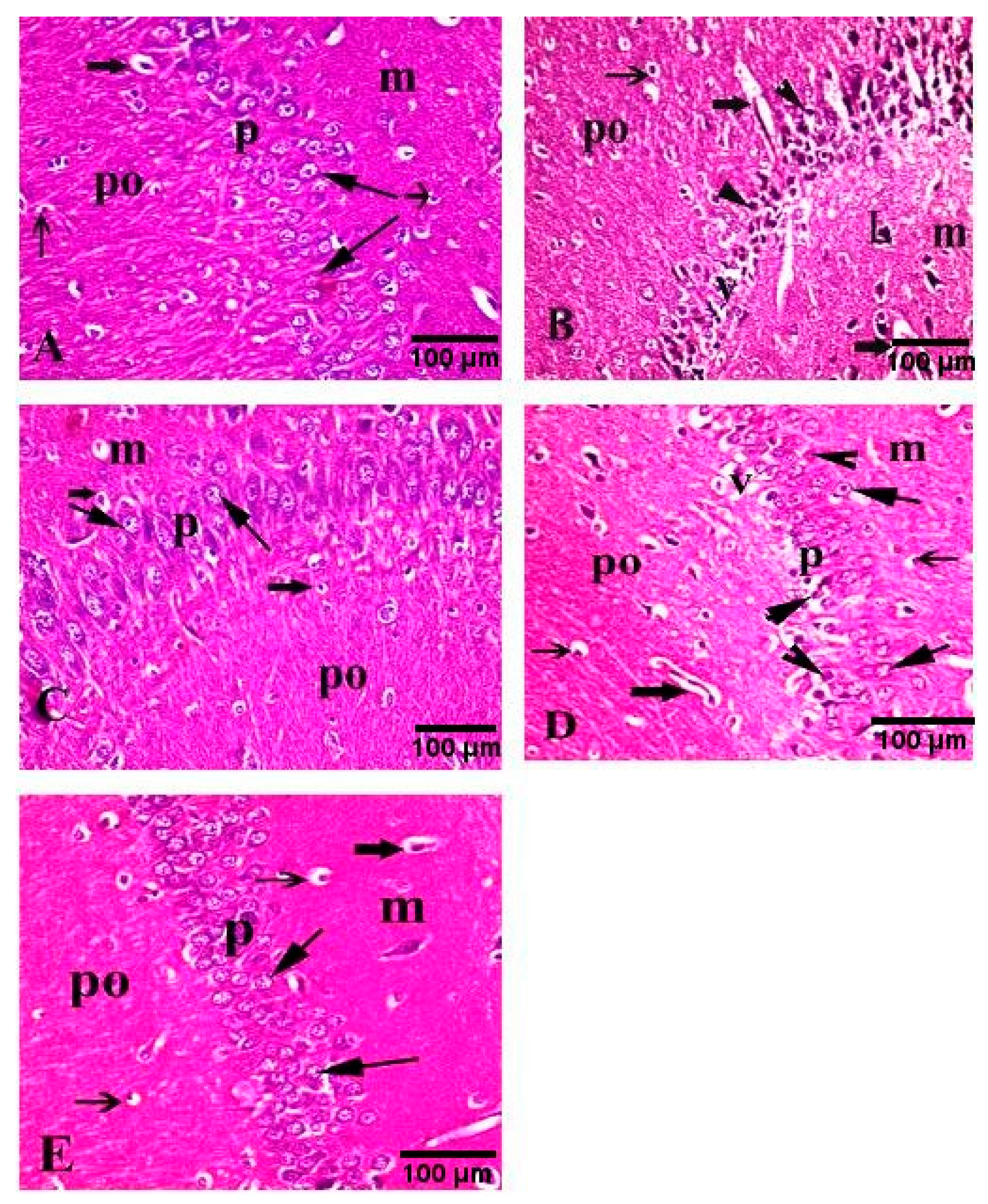
3.8. Effect of H. abyssinica Treatment on the Histopathology of the Hippocampus
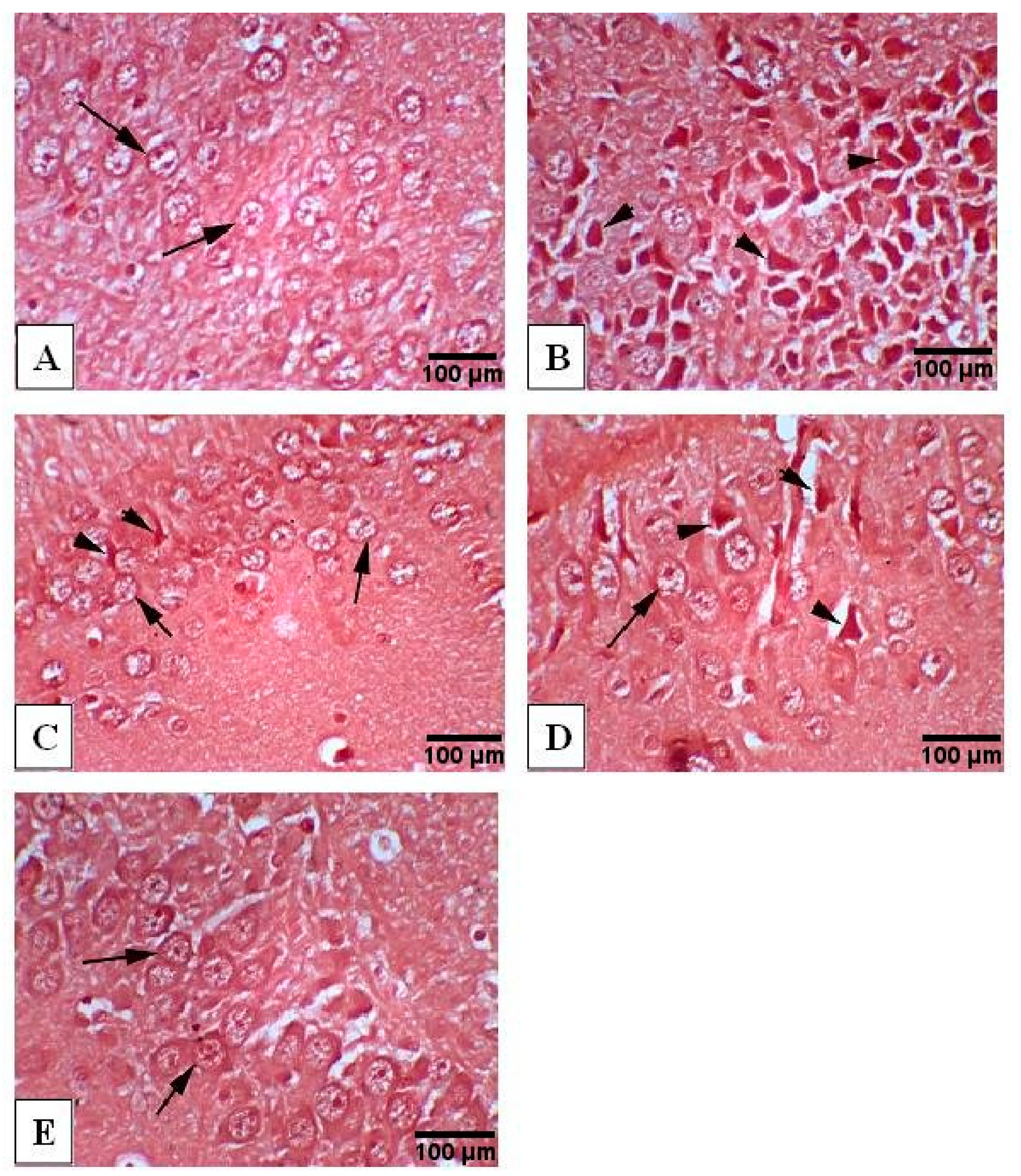
3.9. Congo Red Staining of Hippocampus
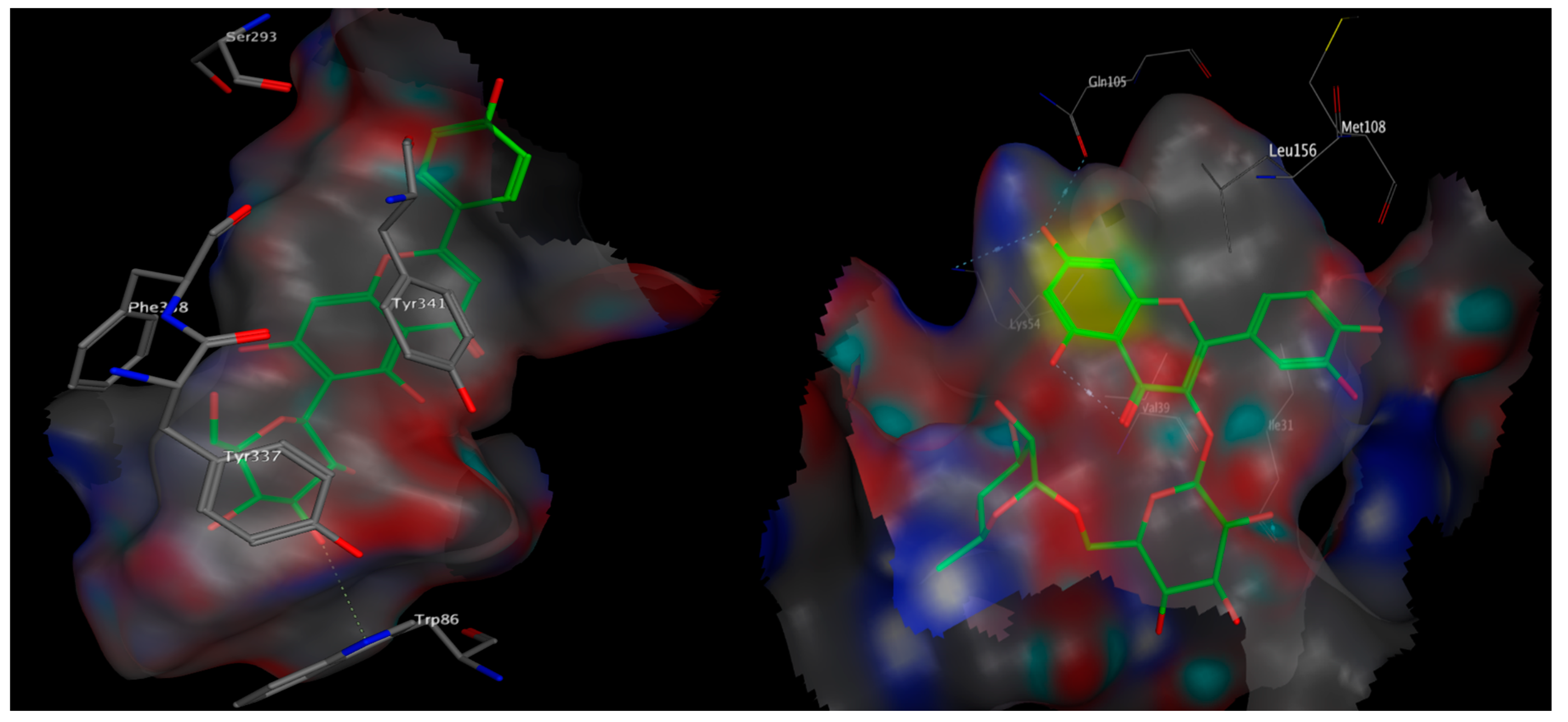
3.10. Molecular Docking
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Qu, Z.; Zhang, J.; Yang, H.; Huo, L.; Gao, J.; Chen, H.; Gao, W. Protective Effect of Tetrahydropalmatine against D-Galactose Induced Memory Impairment in Rat. Physiol. Behav. 2016, 154, 114–125. [Google Scholar] [CrossRef] [PubMed]
- Mattson, M.P. Pathways towards and Away from Alzheimer’s Disease. Nature 2004, 430, 631–639. [Google Scholar] [CrossRef] [PubMed]
- Carreiras, M.; Mendes, E.; Perry, M.; Francisco, A.; Marco-Contelles, J. The Multifactorial Nature of Alzheimer’s Disease for Developing Potential Therapeutics. Curr. Top. Med. Chem. 2014, 13, 1745–1770. [Google Scholar] [CrossRef]
- Bhuiyan, F.R.; Howlader, S.; Raihan, T.; Hasan, M. Plants Metabolites: Possibility of Natural Therapeutics against the COVID-19 Pandemic. Front. Med. 2020, 7, 444. [Google Scholar] [CrossRef] [PubMed]
- Balde, A.M.; Pieters, L.; De Bruyne, T.; Geerts, S.; Vanden Berghe, D.; Vlietinck, A. Biological Investigations on Harrisonia abyssinica. Phytomedicine 1995, 1, 299–302. [Google Scholar] [CrossRef]
- Rugutt, J.K.; Rugutt, K.J.; Berner, D.K. Limonoids from Nigerian Harrisonia a Byssinica and Their Stimulatory Activity against Striga h Ermonthica Seeds. J. Nat. Prod. 2001, 64, 1434–1438. [Google Scholar] [CrossRef]
- Rajab, M.S.; Fronczek, F.R.; Mulholland, D.A.; Rugutt, J.K. 11β,12β-Diacetoxyharrisonin, a Tetranortriterpenoid from Harrisonia abyssinica. Phytochemistry 1999, 52, 127–133. [Google Scholar] [CrossRef]
- Baldé, A.M.; Apers, S.; Claeys, M.; Pieters, L.; Vlietinck, A.J. Cycloabyssinone, a New Cycloterpene from Harrisonia abyssinica. Fitoterapia 2001, 72, 438–440. [Google Scholar] [CrossRef]
- Mayaka, R.; Langat, M.; Omolo, J.; Cheplogoi, P. Antimicrobial Prenylated Acetophenones from Berries of Harrisonia abyssinica. Planta Med. 2012, 78, 383–386. [Google Scholar] [CrossRef]
- Lee, W.J.; Moon, J.S.; Kim, S.I.; Kim, Y.T.; Nash, O.; Bahn, Y.-S.; Kim, S.U. Inhibition of the Calcineurin Pathway by Two Tannins, Chebulagic Acid and Chebulanin, Isolated from Harrisonia abyssinica Oliv. J. Microbiol. Biotechnol. 2014, 24, 1377–1381. [Google Scholar] [CrossRef]
- Nibret, E.; Ashour, M.L.; Rubanza, C.D.; Wink, M. Screening of Some Tanzanian Medicinal Plants for Their Trypanocidal and Cytotoxic Activities: Trypanocidal and Cytotoxic Activities of Tanzanian Plants. Phytother. Res. 2010, 24, 945–947. [Google Scholar] [CrossRef]
- El-Hawary, S.S.; Sobeh, M.; Badr, W.K.; Abdelfattah, M.A.O.; Ali, Z.Y.; El-Tantawy, M.E.; Rabeh, M.A.; Wink, M. HPLC-PDA-MS/MS Profiling of Secondary Metabolites from Opuntia Ficus-Indica Cladode, Peel and Fruit Pulp Extracts and Their Antioxidant, Neuroprotective Effect in Rats with Aluminum Chloride Induced Neurotoxicity. Saudi J. Biol. Sci. 2020, 27, 2829–2838. [Google Scholar] [CrossRef]
- Ghareeb, M.A.; Sobeh, M.; Rezq, S.; El-Shazly, A.M.; Mahmoud, M.F.; Wink, M. HPLC-ESI-MS/MS Profiling of Polyphenolics of a Leaf Extract from Alpinia zerumbet (Zingiberaceae) and Its Anti-Inflammatory, Anti-Nociceptive, and Antipyretic Activities In Vivo. Molecules 2018, 23, 3238. [Google Scholar] [CrossRef] [PubMed]
- Ashour, M.L.; Youssef, F.S.; Gad, H.A.; El-Readi, M.Z.; Bouzabata, A.; Abuzeid, R.M.; Sobeh, M.; Wink, M. Evidence for the Anti-Inflammatory Activity of Bupleurum marginatum (Apiaceae) Extracts Using In Vitro and In Vivo Experiments Supported by Virtual Screening. J. Pharm. Pharmacol. 2018, 70, 952–963. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, Y.U.; Yu, Q.I.; Meng, F. Effect of Ligustrazine on a Rat Model with Hepatic Preneoplasia. Microbiology 2017, 65, 3. [Google Scholar]
- Narayanan, S.N.; Kumar, R.S.; Potu, B.K.; Nayak, S.; Bhat, P.G.; Mailankot, M. Effect of Radio-Frequency Electromagnetic Radiations (RF-EMR) on Passive Avoidance Behaviour and Hippocampal Morphology in Wistar Rats. Upsala J. Med. Sci. 2010, 115, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Ellman, G.L.; Callaway, E. Erythrocyte Cholinesterase-Levels in Mental Patients. Nature 1961, 192, 1216. [Google Scholar] [CrossRef] [PubMed]
- El-raey, M.A.; Mohamed, T.K.; El-kashak, W.A.; Fayad, W.O. Phenolic Constituents and Biological Activities of Acalypha Wilkesiana Forma Tricolor Muell Arg Seeds. Int. J. Pharm. Phytochem. Res. 2016, 8, 386–392. [Google Scholar]
- Bene, K.; Sinan, K.I.; Zengin, G.; Diuzheva, A.; Jekő, J.; Cziáky, Z.; Aumeeruddy, M.Z.; Xiao, J.; Mahomoodally, M.F. A Multidirectional Investigation of Stem Bark Extracts of Four African Plants: HPLC-MS/MS Profiling and Biological Potentials. J. Pharm. Biomed. Anal. 2019, 168, 217–224. [Google Scholar] [CrossRef]
- Colizzi, C. The Protective Effects of Polyphenols on Alzheimer’s Disease: A Systematic Review. Alzheimer’s Dement. Transl. Res. Clin. Interv. 2019, 5, 184–196. [Google Scholar] [CrossRef]
- Ahmed, M.Q.; Alenazi, F.S.; Fazaludeen, M.F.; Shahid, S.M.A.; Kausar, M.A. Pathology and Management of Alzheimer’s Disease: A Review. Int. J. Pharm. Res. Allied Sci. 2018, 7, 2. [Google Scholar]
- Auti, S.T.; Kulkarni, Y.A. Neuroprotective Effect of Cardamom Oil against Aluminum Induced Neurotoxicity in Rats. Front. Neurol. 2019, 10, 399. [Google Scholar] [CrossRef]
- Van Wyk, B.-E.; Wink, M. Phytomedicines, Herbal Drugs, and Poisons; The University of Chicago Press: Chicago, IL, USA; Kew Publishing: Royal Botanic Gardens, Kew, UK, 2015; ISBN 978-0-226-20491-8. [Google Scholar]
- Sun, J.; Nan, G. The Extracellular Signal-Regulated Kinase 1/2 Pathway in Neurological Diseases: A Potential Therapeutic Target. Int. J. Mol. Med. 2017, 39, 1338–1346. [Google Scholar] [CrossRef]
- Elsawi, S.A.; Aly, H.F.; Elbatanony, M.M.; Maamoun, A.A.; Mowawad, D.M. Phytochemical Evaluation of Lagerstroemia indica (L.) Pers Leaves as Anti-Alzheimer’s. J. Mater. Environ. Sci. 2018, 9, 2575–2586. [Google Scholar]
- Olennikov, D.N.; Kashchenko, N.I.; Chirikova, N.K.; Akobirshoeva, A.; Zilfikarov, I.N.; Vennos, C. Isorhamnetin and Quercetin Derivatives as Anti-Acetylcholinesterase Principles of Marigold (Calendula officinalis) Flowers and Preparations. Int. J. Mol. Sci. 2017, 18, 1685. [Google Scholar] [CrossRef] [PubMed]
- Schroeter, H.; Bahia, P.; Spencer, J.P.; Sheppard, O.; Rattray, M.; Cadenas, E.; Rice-Evans, C.; Williams, R.J. (-) Epicatechin Stimulates ERK-dependent Cyclic AMP Response Element Activity and Up-regulates GluR2 in Cortical Neurons. J. Neurochem. 2007, 101, 1596–1606. [Google Scholar] [CrossRef] [PubMed]
- Taïr, K.; Kharoubi, O.; Taïr, O.A.; Hellal, N.; Benyettou, I.; Aoues, A. Aluminium-Induced Acute Neurotoxicity in Rats: Treatment with Aqueous Extract of Arthrophytum (Hammada scoparia). J. Acute Dis. 2016, 5, 470–482. [Google Scholar] [CrossRef]
- Amri, Z.; Ghorbel, A.; Turki, M.; Akrout, F.M.; Ayadi, F.; Elfeki, A.; Hammami, M. Effect of Pomegranate Extracts on Brain Antioxidant Markers and Cholinesterase Activity in High Fat-High Fructose Diet Induced Obesity in Rat Model. BMC Complement. Altern. Med. 2017, 17, 339. [Google Scholar] [CrossRef] [PubMed]
- Kujawska, M.; Jourdes, M.; Kurpik, M.; Szulc, M.; Szaefer, H.; Chmielarz, P.; Kreiner, G.; Krajka-Kuźniak, V.; Mikołajczak, P.Ł.; Teissedre, P.-L. Neuroprotective Effects of Pomegranate Juice against Parkinson’s Disease and Presence of Ellagitannins-Derived Metabolite—Urolithin a—In the Brain. Int. J. Mol. Sci. 2020, 21, 202. [Google Scholar] [CrossRef] [PubMed]
- Yaseen, A.A.; Al-Okbi, S.Y.; Hussein, A.M.; Mohamed, D.A.; Mohammad, A.A.; Fouda, K.A.; Mehaya, F.M. Potential Protection from Alzheimer’s Disease by Wheat Germ and Rice Bran Nano-Form in Rat Model. J. Appl. Pharm. Sci. 2019, 9, 67–76. [Google Scholar]
- Zhang, H.; Wei, M.; Lu, X.; Sun, Q.; Wang, C.; Zhang, J.; Fan, H. Aluminum Trichloride Caused Hippocampal Neural Cells Death and Subsequent Depression-like Behavior in Rats via the Activation of IL-1β/JNK Signaling Pathway. Sci. Total Environ. 2020, 715, 136942. [Google Scholar] [CrossRef] [PubMed]
- Essa, M.M.; Vijayan, R.K.; Castellano-Gonzalez, G.; Memon, M.A.; Braidy, N.; Guillemin, G.J. Neuroprotective Effect of Natural Products against Alzheimer’s Disease. Neurochem. Res. 2012, 37, 1829–1842. [Google Scholar] [CrossRef] [PubMed]
- Doungue, H.T.; Kengne, A.P.N.; Kuate, D. Neuroprotective Effect and Antioxidant Activity of Passiflora Edulis Fruit Flavonoid Fraction, Aqueous Extract, and Juice in Aluminum Chloride-Induced Alzheimer’s Disease Rats. Nutrire 2018, 43, 23. [Google Scholar] [CrossRef]
- Ano, Y.; Ayabe, T.; Kutsukake, T.; Ohya, R.; Takaichi, Y.; Uchida, S.; Yamada, K.; Uchida, K.; Takashima, A.; Nakayama, H. Novel Lactopeptides in Fermented Dairy Products Improve Memory Function and Cognitive Decline. Neurobiol. Aging 2018, 72, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Kinawy, A.A. The Potential Roles of Aluminum Chloride and Sodium Fluoride on the Neurotoxicity of the Cerebral Cortex, Hippocampus, and Hypothalamus of Male Rat Offspring. J. Basic Appl. Zool. 2019, 80, 17. [Google Scholar] [CrossRef]
- Foster, H.D. How Aluminum Causes Alzheimer’s Disease: The Implications for Prevention and Treatment of Foster’s Multiple Antagonist Hypothesis. J. Orthomol. Med. 2000, 15, 21–51. [Google Scholar]
- Wink, M. Chapter 1 Molecular Modes of Action of Cytotoxic Alkaloids: From DNA Intercalation, Spindle Poisoning, Topoisomerase Inhibition to Apoptosis and Multiple Drug Resistance. In The Alkaloids: Chemistry and Biology; Elsevier: Amsterdam, The Netherlands, 2007; Volume 64, pp. 1–47. ISBN 978-0-12-373911-7. [Google Scholar]
- Wink, M. Modes of Action of Herbal Medicines and Plant Secondary Metabolites. Medicines 2015, 2, 251–286. [Google Scholar] [CrossRef] [PubMed]
- Garlapati, P.K.; Raghavan, A.K.; Shivanna, N. Phytochemicals Having Neuroprotective Properties from Dietary Sources and Medicinal Herbs. Phcogn. J. 2014, 7, 1–17. [Google Scholar] [CrossRef]
- Lichtenegger, A.; Muck, M.; Eugui, P.; Harper, D.J.; Augustin, M.; Leskovar, K.; Hitzenberger, C.K.; Woehrer, A.; Baumann, B. Assessment of Pathological Features in Alzheimer’s Disease Brain Tissue with a Large Field-of-View Visible-Light Optical Coherence Microscope. Neurophotonics 2018, 5, 035002. [Google Scholar] [CrossRef]
- Peixoto, H.; Roxo, M.; Silva, E.; Valente, K.; Braun, M.; Wang, X.; Wink, M. Bark Extract of the Amazonian Tree Endopleura uchi (Humiriaceae) Extends Lifespan and Enhances Stress Resistance in Caenorhabditis elegans. Molecules 2019, 24, 915. [Google Scholar] [CrossRef]
- Heiner, F.; Feistel, B.; Wink, M. Sideritis scardica Extracts Inhibit Aggregation and Toxicity of Amyloid-β in Caenorhabditis elegans Used as a Model for Alzheimer’s Disease. PeerJ 2018, 6, e4683. [Google Scholar] [CrossRef] [PubMed]
| Sequence | Accession Number | |
|---|---|---|
| Bax | Forward 5’GAACCATCATGGGCTGGACA3’ Reverse 5’TGAGGTTTATTGGCGCCTCC3’ | XM_032915032.1 |
| Bcl2 | Forward 5’GAACTGGGGGAGGATTGTGG3’ Reverse 5’ACTTCACTTGTGGCCCAGAT3’ | XM_034943915.1 |
| GAPDH | Forward 5’GACAGTCAGCCGCATCTTCT3’ Reverse 5’GCGCCCAATACGACCAAATC3’ | XM_003819132.3 |
| Sample | DPPH | FRAP | TPC |
|---|---|---|---|
| EC50, µg/mL | mM FeSO4/mg Extract | mg GAE/ g Extract | |
| Extract | 9.6 | 18.24 | 274 |
| Ascorbic acid | 2.92 | - | - |
| Quercetin | - | 24.04 | - |
| Parameters | TNF-α | Caspase-3 | IL-1β | MDA |
|---|---|---|---|---|
| mg/g Tissue | pg/g Tissue | ng/g Tissue | nmol/g Tissue | |
| Control | 1.5 ± 0.06 | 22.00 ± 3 | 6.00 ± 0.3 | 170 ± 1.0 |
| AlCl3 | 2.6 ± 0.05 * | 158 ± 3.5 * | 11.5 ± 0.5 * | 305 ± 2.0 * |
| AlCl3 + RIV | 1.9 ± 0.02 # | 51.5 ± 2.5 # | 8.00 ± 0.17 # | 124.5 ± 3.5 # |
| AlCl3 + Extract 100 mg | 3.2 ± 0.15 #*! | 91.7 ± 2.2 *#^ | 8.80 ± 0.7 # | 210 ± 0.02 #^ |
| AlCl3 + Extract 200 mg | 2.1 ± 0.12 # | 68 ± 2 *# | 7.30 ± 1.05 # | 178 ± 1.8 # |
| Compound Name | Docking Score (kcal/mol) | |
|---|---|---|
| ERK2 | AchE | |
| Quercetin | −18.09 | −19.75 * |
| Quercetin 3-glucoside | −22.50 | −29.45 |
| Quercetin 3-xyloside | −25.24 | −29.53 |
| Quercetin 3-rutinoside | −34.09 | −26.31 |
| Quercetin 3-(6′-p-coumaroylglucoside) | −27.19 | −31.20 |
| Quercetin 3-(6′-caffeoylglucoside) | −27.85 | −29.03 |
| Quercetin 3-(6′-galloylglucoside) | −28.32 | −21.01 |
| Quercetin 3-(6′-p-hydroxybenzoylgalactoside) | −27.50 | −21.16 |
| Kaempferol | −16.74 | −17.69 * |
| Kaempferol 3-glucoside | −22.47 | −25.25 |
| Kaempferol 3-xyloside | −22.54 | −22.87 |
| Kaempferol 3-(6′-caffeoylglucoside) | −27.37 | −29.88 |
| Kaempferol 3-(6′-galloylglucoside) | −27.36 | −27.76 |
| Kaempferol 3-(6′-p-hydroxybenzoylglucoside) | −29.19 | −25.19 |
| Apigenin | −13.52 | −16.72 |
| Apigenin 7-glucoside | −21.20 | −26.56 |
| Apigenin 6-glucoside | −19.42 | −26.97 |
| Apigenin 6-(2′-galloylglucoside) | −30.03 | −30.86 |
| Co-crystallized inhibitor | −17.10 | −18.38 |
| EGCG (natural reference inhibitor) | −22.52 | |
| Quercetin 3-(2′,6′-diacetylglucoside) (natural reference inhibitor) | −33.01 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anwar, H.M.; Georgy, G.S.; Hamad, S.R.; Badr, W.K.; El Raey, M.A.; Abdelfattah, M.A.O.; Wink, M.; Sobeh, M. A Leaf Extract of Harrisonia abyssinica Ameliorates Neurobehavioral, Histological and Biochemical Changes in the Hippocampus of Rats with Aluminum Chloride-Induced Alzheimer’s Disease. Antioxidants 2021, 10, 947. https://doi.org/10.3390/antiox10060947
Anwar HM, Georgy GS, Hamad SR, Badr WK, El Raey MA, Abdelfattah MAO, Wink M, Sobeh M. A Leaf Extract of Harrisonia abyssinica Ameliorates Neurobehavioral, Histological and Biochemical Changes in the Hippocampus of Rats with Aluminum Chloride-Induced Alzheimer’s Disease. Antioxidants. 2021; 10(6):947. https://doi.org/10.3390/antiox10060947
Chicago/Turabian StyleAnwar, Hend Mohamed, Gehan S. Georgy, Sherin Ramadan Hamad, Wafaa K. Badr, Mohamed A. El Raey, Mohamed A. O. Abdelfattah, Michael Wink, and Mansour Sobeh. 2021. "A Leaf Extract of Harrisonia abyssinica Ameliorates Neurobehavioral, Histological and Biochemical Changes in the Hippocampus of Rats with Aluminum Chloride-Induced Alzheimer’s Disease" Antioxidants 10, no. 6: 947. https://doi.org/10.3390/antiox10060947
APA StyleAnwar, H. M., Georgy, G. S., Hamad, S. R., Badr, W. K., El Raey, M. A., Abdelfattah, M. A. O., Wink, M., & Sobeh, M. (2021). A Leaf Extract of Harrisonia abyssinica Ameliorates Neurobehavioral, Histological and Biochemical Changes in the Hippocampus of Rats with Aluminum Chloride-Induced Alzheimer’s Disease. Antioxidants, 10(6), 947. https://doi.org/10.3390/antiox10060947








