The Role of Oxidative Stress Markers in Predicting Acute Thrombotic Occlusion of Haemodialysis Vascular Access and Progressive Stenotic Dysfunction Demanding Angioplasty
Abstract
1. Introduction
2. Materials and Methods
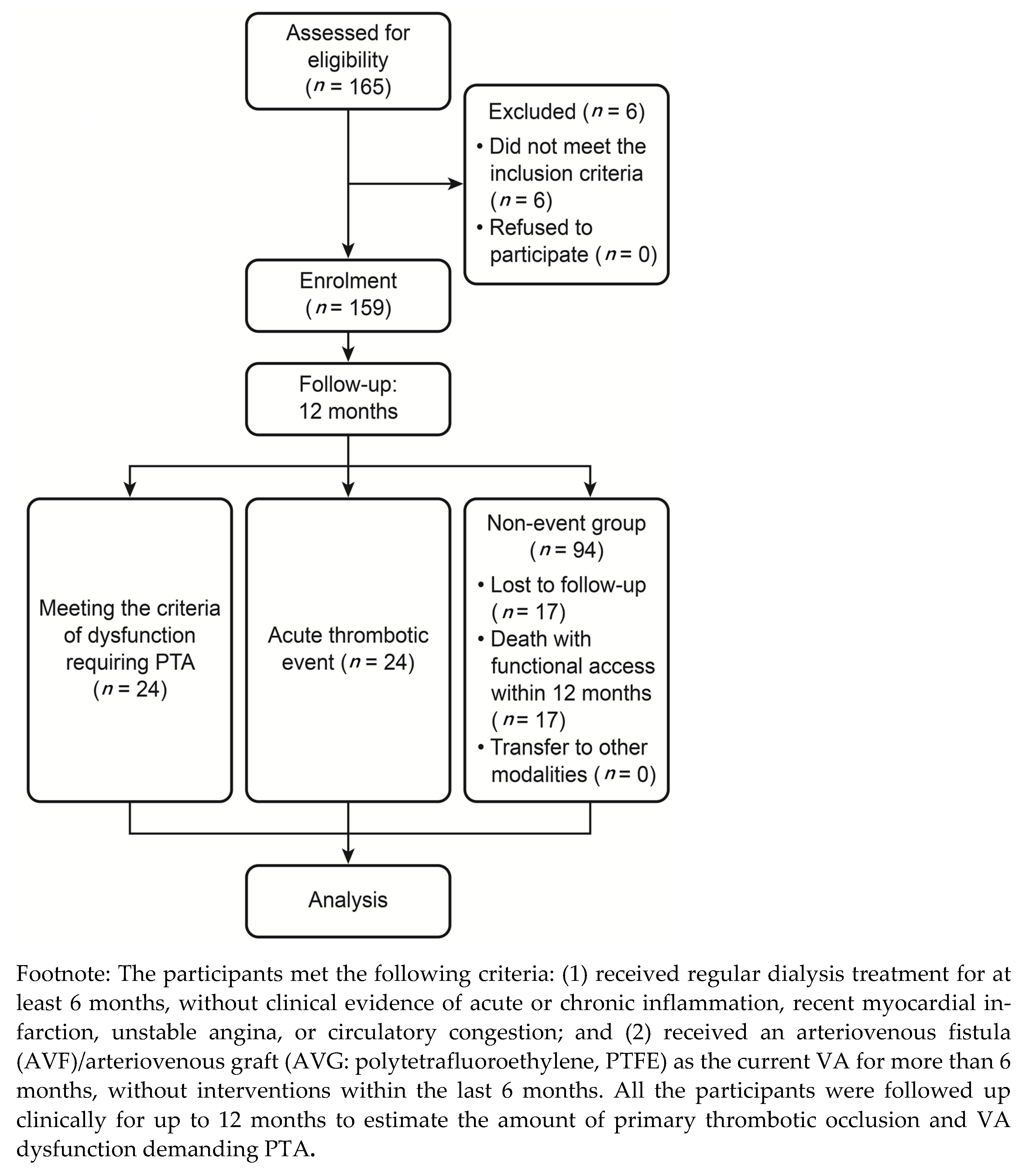
2.1. Study Participants
2.2. Study Protocol
2.3. Laboratory Methods
2.4. Follow-Up and Definitions
2.5. Statistical Analysis
3. Results
3.1. Patient Characteristics
3.2. Clinical Follow-Up
3.3. Baseline Parameters and Acute Thrombotic Occlusion
3.4. Baseline Parameters and Progressive Dysfunction of Vascular Access
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wu, C.K.; Wu, C.L.; Lin, C.H.; Leu, J.G.; Kor, C.T.; Tarng, D.C. Association of vascular access flow with short-term and long-term mortality in chronic haemodialysis patients: A retrospective cohort study. BMJ Open 2017, 7, e017035. [Google Scholar] [CrossRef][Green Version]
- Roy-Chaudhury, P.; Sukhatme, V.P.; Cheung, A.K. Hemodialysis vascular access dysfunction: A cellular and molecular viewpoint. J. Am. Soc. Nephrol. 2006, 17, 1112–1127. [Google Scholar] [CrossRef]
- Chang, C.J.; Ko, P.J.; Hsu, L.A.; Ko, Y.S.; Ko, Y.L.; Chen, C.F.; Huang, C.C.; Hsu, T.S.; Lee, Y.S.; Pang, J.H. Highly increased cell proliferation activity in the restenotic hemodialysis vascular access after percutaneous transluminal angioplasty: Implication in prevention of restenosis. Am. J. Kidney Dis. 2004, 43, 74–84. [Google Scholar] [CrossRef]
- Weiss, M.F.; Scivittaro, V.; Anderson, J.M. Oxidative stress and increased expression of growth factors in lesions of failed hemodialysis access. Am. J. Kidney Dis. 2001, 37, 970–980. [Google Scholar] [CrossRef]
- Chen, S.C.; Chang, J.M.; Hwang, S.J.; Tsai, J.C.; Wang, C.S.; Mai, H.C.; Lin, F.H.; Su, H.M.; Chen, H.C. Significant correlation between ankle-brachial index and vascular access failure in hemodialysis patients. Clin. J. Am. Soc. Nephrol. 2009, 4, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Stracke, S.; Konner, K.; Köstlin, I.; Friedl, R.; Jehle, P.M.; Hombach, V.; Keller, F.; Waltenberger, J. Increased expression of TGF-beta1 and IGF-I in inflammatory stenotic lesions of hemodialysis fistulas. Kidney Int. 2002, 61, 1011–1019. [Google Scholar] [CrossRef]
- Wu, T.C.; Chen, Y.H.; Leu, H.B.; Chen, Y.L.; Lin, F.Y.; Lin, S.J.; Chen, J.W. Carvedilol, a pharmacological antioxidant, inhibits neointimal matrix metalloproteinase-2 and-9 in experimental atherosclerosis. Free Radic. Biol. Med. 2007, 43, 1508–1522. [Google Scholar] [CrossRef]
- Hu, Y.; Baker, A.H.; Zou, Y.; Newby, A.C.; Xu, Q. Local gene transfer of tissue inhibitor of metalloproteinase-2 influences vein graft remodeling in a mouse model. Arterioscler. Thromb. Vasc. Biol. 2001, 21, 1275–1280. [Google Scholar] [CrossRef]
- Sho, E.; Sho, M.; Singh, T.M.; Nanjo, H.; Komatsu, M.; Xu, C.; Masuda, H.; Zarins, C.K. Arterial enlargement in response to high flow requires early expression of matrix metalloproteinases to degrade extracellular matrix. Exp. Mol. Pathol. 2002, 73, 142–153. [Google Scholar] [CrossRef]
- Zoccali, C.; Bode-Böger, S.; Mallamaci, F.; Benedetto, F.; Tripepi, G.; Malatino, L.; Cataliotti, A.; Bellanuova, I.; Fermo, I.; Frölich, J.; et al. Plasma concentration of asymmetrical dimethylarginine and mortality in patients with end-stage renal disease: A prospective study. Lancet 2001, 358, 2113–2117. [Google Scholar] [CrossRef]
- Wu, C.C.; Wen, S.C.; Yang, C.W.; Pu, S.Y.; Tsai, K.C.; Chen, J.W. Plasma ADMA predicts restenosis of arteriovenous fistula. J. Am. Soc. Nephrol. 2009, 20, 213–222. [Google Scholar] [CrossRef]
- Jacobi, J.; Tsao, P.S. Asymmetrical dimethylarginine in renal disease: Limits of variation or variation limits? A systematic review. Am. J. Nephrol. 2008, 28, 224–237. [Google Scholar] [CrossRef]
- May, M.; Batkai, S.; Zörner, A.A.; Tsikas, D.; Jordan, J.; Engeli, S. Clinical evaluation of extracellular ADMA concentrations in human blood and adipose tissue. Int. J. Mol. Sci. 2014, 15, 1189–1200. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.N.; Tain, Y.L. Developmental origins of kidney disease: Why oxidative stress matters? Antioxidants 2021, 10, 33. [Google Scholar] [CrossRef] [PubMed]
- Schnyder, G.; Roffi, M.; Flammer, Y.; Pin, R.; Hess, O.M. Effect of homocysteine-lowering therapy with folic acid, vitamin B12, and vitamin B6 on clinical outcome after percutaneous coronary intervention: The Swiss Heart study: A randomized controlled trial. JAMA 2002, 288, 973–979. [Google Scholar] [CrossRef]
- Sidawy, A.N.; Gray, R.; Besarab, A.; Henry, M.; Ascher, E.; Silva, M., Jr.; Miller, A.; Scher, L.; Trerotola, S.; Gregory, R.T.; et al. Recommended standards for reports dealing with arteriovenous hemodialysis accesses. J. Vasc. Surg. 2002, 35, 603–610. [Google Scholar] [CrossRef]
- Brahmbhatt, A.; Misra, S. The biology of hemodialysis vascular access failure. Semin. Interv. Radiol. 2016, 33, 15–20. [Google Scholar] [CrossRef]
- Zimmermann, A.; Senner, S.; Eckstein, H.H.; Pelisek, J. Histomorphological evaluation of atherosclerotic lesions in patients with peripheral artery occlusive disease. Adv. Med. Sci. 2015, 60, 236–239. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Patel, S.; Hanisch, J.J.; Santana, J.M.; Hashimoto, T.; Bai, H.; Kudze, T.; Foster, T.R.; Guo, J.; Yatsula, B.; et al. Future research directions to improve fistula maturation and reduce access failure. Semin. Vasc. Surg. 2016, 29, 153–171. [Google Scholar] [CrossRef]
- Pearson, T.A.; Mensah, G.A.; Alexander, R.W.; Anderson, J.L.; Cannon, R.O., 3rd; Criqui, M.; Fadl, Y.Y.; Fortmann, S.P.; Hong, Y.; Myers, G.L.; et al. Markers of inflammation and cardiovascular disease: Application to clinical and public health practice: A statement for healthcare professionals from the Centers for Disease Control and Prevention and the American Heart Association. Circulation 2003, 107, 499–511. [Google Scholar] [CrossRef]
- Vascular Access Work Group. Clinical practice guidelines for vascular access. Am. J. Kidney Dis. 2006, 48, S248–S273. [Google Scholar] [CrossRef] [PubMed]
- Adragao, T.; Pires, A.; Branco, P.; Castro, R.; Oliveira, A.; Nogueira, C.; Bordalo, J.; Curto, J.D.; Prata, M.M. Ankle--brachial index, vascular calcifications and mortality in dialysis patients. Nephrol. Dial. Transplant. 2012, 27, 318–325. [Google Scholar] [CrossRef]
- Lok, C.E.; Huber, T.S.; Lee, T.; Shenoy, S.; Yevzlin, A.S.; Abreo, K.; Allon, M.; Asif, A.; Astor, B.C.; Glickman, M.H.; et al. KDOQI clinical practice guideline for vascular access: 2019 update. Am. J. Kidney Dis. 2020, 75, S1–S164. [Google Scholar] [CrossRef] [PubMed]
- Wan, Q.; Yang, S.; Li, L.; Chu, F. Effects of far infrared therapy on arteriovenous fistulas in hemodialysis patients: A meta-analysis. Ren. Fail. 2017, 39, 613–622. [Google Scholar] [CrossRef]
- Lee, T.; Misra, S. New Insights into dialysis vascular access: Molecular targets in arteriovenous fistula and arteriovenous graft failure and their potential to improve vascular access outcomes. Clin. J. Am. Soc. Nephrol. 2016, 11, 1504–1512. [Google Scholar] [CrossRef]
- Chen, B.; Wang, P.; Brem, A.; Dworkin, L.; Liu, Z.; Gong, R. Mineralocorticoid receptor: A hidden culprit for hemodialysis vascular access dysfunction. EBioMedicine 2019, 39, 621–627. [Google Scholar] [CrossRef]
- Shiu, Y.T.; Rotmans, J.I.; Geelhoed, W.J.; Pike, D.B.; Lee, T. Arteriovenous conduits for hemodialysis: How to better modulate the pathophysiological vascular response to optimize vascular access durability. Am. J. Physiol. Ren. Physiol. 2019, 316, F794–F806. [Google Scholar] [CrossRef]
- Dowsett, L.; Duluc, L.; Higgins, E.; Leiper, J. Asymmetric dimethylarginine is a posititve regulator of endothelial nitric oxide production by modulating calcium sensing receptor signalling. J. Hypertens. 2019, 37, e70. [Google Scholar] [CrossRef]
- O’Hare, A.M.; Hsu, C.Y.; Bacchetti, P.; Johansen, K.L. Peripheral vascular disease risk factors among patients undergoing hemodialysis. J. Am. Soc. Nephrol. 2002, 13, 497–503. [Google Scholar]
- Chen, S.C.; Chang, J.M.; Hwang, S.J.; Chen, J.H.; Lin, F.H.; Su, H.M.; Chen, H.C. Comparison of ankle-brachial index and brachial-ankle pulse wave velocity between patients with chronic kidney disease and hemodialysis. Am. J. Nephrol. 2009, 29, 374–380. [Google Scholar] [CrossRef]
- Arroyo, D.; Betriu, A.; Valls, J.; Gorriz, J.L.; Pallares, V.; Abajo, M.; Gracia, M.; Valdivielso, J.M.; Fernandez, E. Factors influencing pathological ankle-brachial index values along the chronic kidney disease spectrum: The NEFRONA study. Nephrol. Dial. Transplant. 2017, 32, 513–520. [Google Scholar] [CrossRef] [PubMed]
- Boelaert, J.; Schepers, E.; Glorieux, G.; Eloot, S.; Vanholder, R.; Lynen, F. Determination of asymmetric and symmetric dimethylarginine in serum from patients with chronic kidney disease: UPLC-MS/MS versus ELISA. Toxins 2016, 8, 149. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Acute Thrombosis (n = 24) | Non-Thrombosis (n = 135) | p |
|---|---|---|---|
| Age (years), mean ± SD | 68.38 ± 12.08 | 64.33 ± 12.77 | 0.145 |
| Sex (%) | |||
| Female | 13 (54.2%) | 63 (46.7%) | 0.648 |
| Male | 11 (45.8%) | 72 (53.3%) | |
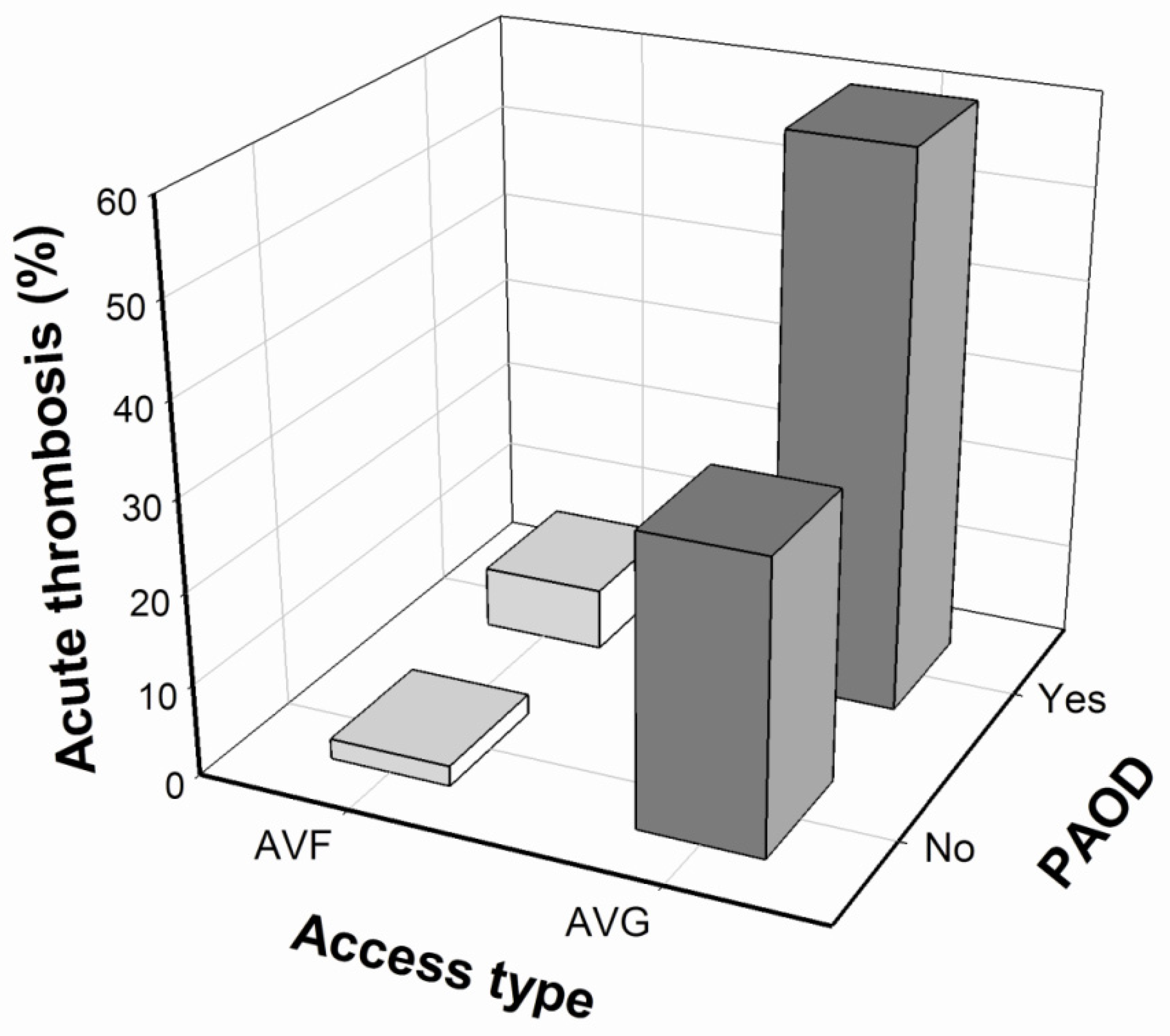
| Access type (%) | |||
| AVF | 3 (12.5%) | 103 (76.3%) | <0.001 * |
| AVG | 21 (87.5%) | 32 (23.7%) | |
| Duration of access use (months), mean ± SD | 3.67 ± 2.68 | 10.91 ± 2.83 | <0.001 * |
| Risk factors (%) | |||
| Hypertension | 20 (83.3%) | 109 (80.7%) | 0.987 |
| Diabetes mellitus | 13 (54.2%) | 58 (43.0%) | 0.427 |
| Current smoking | 1 (4.2%) | 12 (8.9%) | 0.693 |
| CAD | 13 (54.2%) | 46 (34.1%) | 0.099 |
| PAOD | 10 (41.7%) | 20 (14.8%) | 0.005 * |
| Plasma biochemical data, mean ± SD | |||
| LDL-C (mg/dL) | 96.67 ± 35.30 | 90.75 ± 34.23 | 0.439 |
| HDL-C (mg/dL) | 44.38 ± 14.03 | 42.49 ± 15.92 | 0.406 |
| TG (mg/dL) | 189.92 ± 150.24 | 152.36 ± 89.78 | 0.437 |
| Calcium (mg/dL) | 9.15 ± 0.99 | 9.27 ± 1.02 | 0.597 |
| Phosphate (mg/dL) | 5.38 ± 1.67 | 4.69 ± 1.52 | 0.043 * |
| Albumin (mg/dL) | 3.80 ± 0.42 | 3.85 ± 0.33 | 0.530 |
| Creatinine (mg/dL) | 10.06 ± 3.22 | 10.44 ± 2.57 | 0.519 |
| Kt/V | 1.35 ± 0.22 | 1.41 ± 0.29 | 0.359 |
| Medications (%) | |||
| Anti-platelet agents | 19 (79.2%) | 69 (51.1%) | 0.020 * |
| Aspirin | 18 (75.0%) | 55 (40.7%) | 0.004 * |
| Dipyridamole | 3 (12.5%) | 6 (4.4%) | 0.321 |
| Cilostazol | 5 (20.8%) | 16 (11.9%) | 0.137 |
| Coumadin | 1 (4.2%) | 4 (3.0%) | 0.564 |
| Nitrates | 10 (41.7%) | 54 (40.0%) | 1.000 |
| β-blockers | 8 (33.3%) | 48 (35.6%) | 1.000 |
| Calcium antagonists | 16 (66.7%) | 74 (54.8%) | 0.392 |
| ACEI/ARB | 9 (37.5%) | 37 (27.4%) | 0.447 |
| Diuretics | 9 (37.5%) | 39 (28.9%) | 0.545 |
| Lipid-lowering agents | |||
| Statin | 3 (12.5%) | 12 (8.9%) | 0.703 |
| Fibrate | 1 (4.2%) | 3 (2.2%) | 0.484 |
| Plasma biomarkers, mean ± SD | |||
| Hs-CRP (mg/dL) | 0.94 ± 1.15 | 1.15 ± 2.12 | 0.255 |
| Homocysteine (μmol/L) | 29.984 ± 15.00 | 26.92 ± 10.44 | 0.319 |
| ADMA (μmol/mL) | 0.67 ± 0.28 | 0.58 ± 0.25 | 0.102 |
| MMP-2 (ng/mL) | 760.28 ± 180.73 | 855.10 ± 208.99 | 0.053 |
| MMP-9 (ng/mL) | 62.12 ± 41.57 | 62.53 ± 41.01 | 0.886 |
| NO (μM) | 219.39 ± 196.25 | 257.23 ± 299.59 | 0.436 |
| MCP-1 (pg/mL) | 319.91 ± 118.32 | 338.64 ± 177.82 | 0.635 |
| TGF-β (pg/mL) | 3.23 ± 4.79 | 6.09 ± 8.25 | 0.117 |
| TNF-α (pg/mL) | 20.09 ± 66.09 | 39.64 ± 82.99 | 0.296 |
| IL-1β (pg/mL) | 66.91 ± 43.14 | 57.47 ± 44.02 | 0.352 |
| Parameter | Univariate Analysis | Multivariate Analysis | ||
|---|---|---|---|---|
| HR (95% Cl) | p | HR (95% Cl) | p | |
| Age (years) | 1.03 (0.996–1.07) | 0.084 | ||
| Sex | ||||
| Female | 1.00 | |||
| Male | 0.75 (0.33–1.66) | 0.472 | ||
| Access type (AVF/AVG) | ||||
| AVF | 1.00 | |||
| AVG | 19.05 (5.67–63.998) | <0.001 * | 16.93 (3.828–50.935) | <0.001 * |
| Risk factors (%) | ||||
| Hypertension | 1.17 (0.40–3.43) | 0.773 | ||
| Diabetes mellitus | 1.56 (0.697–3.47) | 0.281 | ||
| Current smoking | 0.47 (0.6–3.49) | 0.461 | ||
| CAD | 2.16 (0.97–4.83) | 0.060 | ||
| PAOD | 3.63 (1.61–8.18) | 0.002 * | 2.35 (0.901–6.476) | 0.047 |
| Plasma biochemical data | ||||
| LDL-C > 130 mg/dL | 0.87 (0.26–2.93) | 0.826 | ||
| HDL-C < 50 mg/dL | 1.00 (0.398–2.52) | 0.997 | ||
| TG > 200 mg/dL | 1.15 (0.46–2.89) | 0.769 | ||
| Calcium (mg/dL) | 0.92 (0.61–1.38) | 0.687 | ||
| Phosphate (mg/dL) | 1.32 (1.01–1.71) | 0.043 * | 1.266 (0.945–1.696) | 0.114 |
| Albumin (mg/dL) | 0.56 (0.17–1.83) | 0.334 | ||
| Creatinine (mg/dL) | 0.92 (0.78–1.08) | 0.290 | ||
| Kt/V | 0.39 (0.08–1.92) | 0.248 | ||
| Medications | ||||
| Anti-platelet agents | 3.26 (1.22–8.74) | 0.019 * | 0.741 (0.078–6.994) | 0.793 |
| Aspirin | 3.78 (1.50–9.52) | 0.005 * | 2.172 (0.274–17.202) | 0.462 |
| Dipyridamole | 1.88 (0.70–5.04) | 0.209 | ||
| Cilostazol | 3.19 (0.95–10.69) | 0.061 | ||
| Coumadin | 1.53 (0.21–11.32) | 0.678 | ||
| Nitrates | 1.15 (0.51–2.60) | 0.732 | ||
| β-blockers | 0.89 (0.38–2.08) | 0.790 | ||
| Calcium antagonists | 1.54 (0.66–3.59) | 0.321 | ||
| ACEI/ARB | 1.61 (0.70–3.67) | 0.261 | ||
| Diuretics | 1.37 (0.6–3.14) | 0.453 | ||
| Lipid-lowering agents | ||||
| Statin | 1.30 (0.39–4.34) | 0.675 | ||
| Fibrate | 1.88 (0.25–13.95) | 0.537 | ||
| Plasma biomarkers | ||||
| Hs-CRP > 0.5 mg/dL | 1.61 (0.72–3.58) | 0.248 | ||
| Homocysteine (μmol/L) | 1.02 (0.99–1.05) | 0.189 | ||
| ADMA (μmol/mL) | 3.45 (0.78–15.14) | 0.101 | ||
| MMP-2 (ng/mL) | 0.998 (0.006–1.00) | 0.045 * | 0.998 (0.996–1.001) | 0.142 |
| MMP-9 (ng/mL) | 1.00 (0.99–1.01) | 0.932 | ||
| NO (μM) | 0.999 (0.997–1.00) | 0.493 | ||
| MCP-1 (pg/mL) | 0.999 (0.996–1.002) | 0.594 | ||
| TGF-β (pg/mL) | 0.936 (0.858–1.021) | 0.138 | ||
| TNF-α (pg/mL) | 0.996 (0.988–1.004) | 0.310 | ||
| IL-1β (pg/mL) | 1.004 (0.995–1.013) | 0.390 | ||
| Dysfunction (n = 24) | Non-Dysfunction (n = 135) | p | |
|---|---|---|---|
| Age (years), mean ± SD | 62.21 ± 10.48 | 65.42 ± 13.04 | 0.156 |
| Sex (%) | 0.648 | ||
| Female | 13 (54.2%) | 63 (46.7%) | |
| Male | 11 (45.8%) | 72 (53.3%) | |
| Access type (%) | 0.060 | ||
| AVF | 12 (50.0%) | 94 (69.6%) | |
| AVG | 12 (50.0%) | 41 (30.4%) | |
| Duration of access use (months), mean ± SD | 4.18 ± 3.08 | 11.1 ± 2.69 | <0.001 * |
| Risk factors (%) | |||
| Hypertension | 21 (87.5%) | 108 (80.0%) | 0.572 |
| Diabetes mellitus | 12 (50.0%) | 59 (43.7%) | 0.727 |
| Current smoking | 2(8.3%) | 11 (8.1%) | 1.00 |
| CAD | 10 (41.7%) | 49 (36.3%) | 0.785 |
| PAOD | 3 (12.5%) | 27 (20.0%) | 0.572 |
| Plasma biochemical data, mean ± SD | |||
| LDL-C (mg/dL) | 100.88 ± 24.75 | 89.98 ± 35.64 | 0.086 |
| HDL-C (mg/dL) | 43.96 ± 17.13 | 42.57 ± 15.39 | 0.902 |
| TG (mg/dL) | 152.29 ± 93.42 | 159.05 ± 103.17 | 0.916 |
| Calcium (mg/dL) | 9.57 ± 1.06 | 9.19 ± 1.00 | 0.058 |
| Phosphate (mg/dL) | 5.07 ± 1.58 | 4.74 ± 1.55 | 0.510 |
| Albumin (mg/dL) | 3.88 ± 0.34 | 3.83 ± 0.35 | 0.520 |
| Creatinine (mg/dL) | 11.07 ± 2.48 | 10.26 ± 2.69 | 0.186 |
| Kt/V | 1.38 ± 0.19 | 1.41 ± 0.30 | 0.778 |
| Medications (%) | |||
| Anti-platelet agents | 15 (62.5%) | 73 (54.1%) | 0.444 |
| Aspirin | 11 (45.8%) | 62 (45.9%) | 0.993 |
| Dipyridamole | 1 (4.2%) | 8 (5.9%) | 1.00 |
| Cilostazol | 4 (16.7%) | 17 (12.6%) | 0.527 |
| Coumadin | 2 (8.3%) | 3 (2.2%) | 0.164 |
| Nitrates | 10 (41.7%) | 54 (40.0%) | 0.878 |
| β-blockers | 8 (33.3%) | 48 (35.6%) | 0.834 |
| Calcium antagonists | 13 (54.2%) | 77 (57.0%) | 0.794 |
| ACEI/ARB | 7 (29.2%) | 39 (28.9%) | 0.978 |
| Diuretics | 15 (62.5%) | 33 (24.4%) | <0.001 * |
| Lipid-lowering agents | |||
| Statin | 5 (20.8%) | 10 (7.4%) | 0.054 |
| Fibrate | 0 (0.0%) | 4 (3.0%) | 1.00 |
| Plasma biomarkers, mean ± SD | |||
| Hs-CRP (mg/dL) | 1.09 ± 1.76 | 1.13 ± 2.05 | 0.191 |
| Homocysteine (μmol/L) | 26.27 ± 6.53 | 27.58 ± 11.90 | 0.874 |
| ADMA (μmol/mL) | 0.7740 ± 0.2735 | 0.5642 ± 0.2426 | 0.001 * |
| MMP-2 (ng/mL) | 793.63 ± 221.14 | 848.72 ± 204.57 | 0.336 |
| MMP-9 (ng/mL) | 81.87 ± 47.16 | 59.13 ± 39.04 | 0.021 * |
| NO (μM) | 357.84 ± 516.03 | 232.62 ± 220.21 | 0.051 |
| MCP-1 (pg/mL) | 324.48 ± 106.73 | 337.96 ± 179.54 | 0.728 |
| TGF-β (pg/mL) | 5.83 ± 6.80 | 5.64 ± 8.10 | 0.918 |
| TNF-α (pg/mL) | 14.09 ± 37.08 | 40.88 ± 85.86 | 0.016 * |
| IL-1β (pg/mL) | 73.17 ± 39.82 | 56.27 ± 44.22 | 0.089 |
| Parameter | Univariate Analysis | Multivariate Analysis | ||
|---|---|---|---|---|
| HR (95% Cl) | p | HR (95% Cl) | p | |
| Age (years) | 0.98 (0.95–1.02) | 0.312 | ||
| Sex | 0.506 | |||
| Female | 1.00 | |||
| Male | 0.76 (0. 34–1.70) | |||
| Access type (AVF/AVG) | ||||
| AVF | 1.00 | 1.00 | ||
| AVG | 2.28 (1.03–5.08) | 0.043* | 2.39 (0.91–6.28) | 0.076 |
| Risk factors | ||||
| Hypertension (%) | 1.65 (0.49–5.53) | 0.418 | ||
| Diabetes mellitus (%) | 1.36 (0.61–3.04) | 0.448 | ||
| Current smoking (%) | 0.99 (0.23–4.20) | 0.986 | ||
| CAD (%) | 1.26 (0.56–2.83) | 0.581 | ||
| PAOD (%) | 0.57 (0.17–1.90) | 0.357 | ||
| Plasma biochemical data | ||||
| LDL-C (mg/dL) | 1.01 (0.99–1.02) | 0.193 | ||
| HDL-C (mg/dL) | 1.00 (0.98–1.03) | 0.794 | ||
| TG (mg/dL) | 1.00 (0.99–1.00) | 0.735 | ||
| Calcium (mg/dL) | 1.44 (1.00–2.10) | 0.053 | ||
| Phosphate (mg/dL) | 1.14 (0.87–1.48) | 0.354 | ||
| Albumin (mg/dL) | 1.17 (0.35–3.86) | 0.799 | ||
| Creatinine (mg/dL) | 1.09 (0.94–1.26) | 0.258 | ||
| Kt/V | 0.71 (0.16–3.13) | 0.652 | ||
| Medications | ||||
| Anti-platelet agents | 1.35 (0.59–3.07) | 0.482 | ||
| Aspirin | 0.96 (0.43–2.14) | 0.918 | ||
| Dipyridamole | 1.41 (0.48–4.12) | 0.532 | ||
| Cilostazol | 0.77 (0.10–5.67) | 0.793 | ||
| Coumadin | 3.06 (0.72–13.03) | 0.13 | ||
| Nitrates | 1.10 (0.49–2.47) | 0.821 | ||
| β-blockers | 0.89 (0.38–2.07) | 0.777 | ||
| Calcium antagonists | 0.81 (0.36–1.80) | 0.603 | ||
| ACEI/ARB | 0.99 (0.41–2.38) | 0.973 | ||
| Diuretic | 4.23 (1.85–9.66) | 0.001 * | 1.80 (0.63–5.16) | 0.277 |
| Lipid-lowering agents | ||||
| Statin | 2.80 (1.05–7.50) | 0.040 * | 1.16 (0.35–3.83) | 0.810 |
| Fibrate | 0.05 (<0.0001–3221.32) | 0.592 | ||
| Plasma biomarkers | ||||
| Hs-CRP (mg/dL) | 1.01 (0.83–1.42) | 0.907 | ||
| Homocysteine (μmol/L) | 0.99 (0.95–1.03) | 0.634 | ||
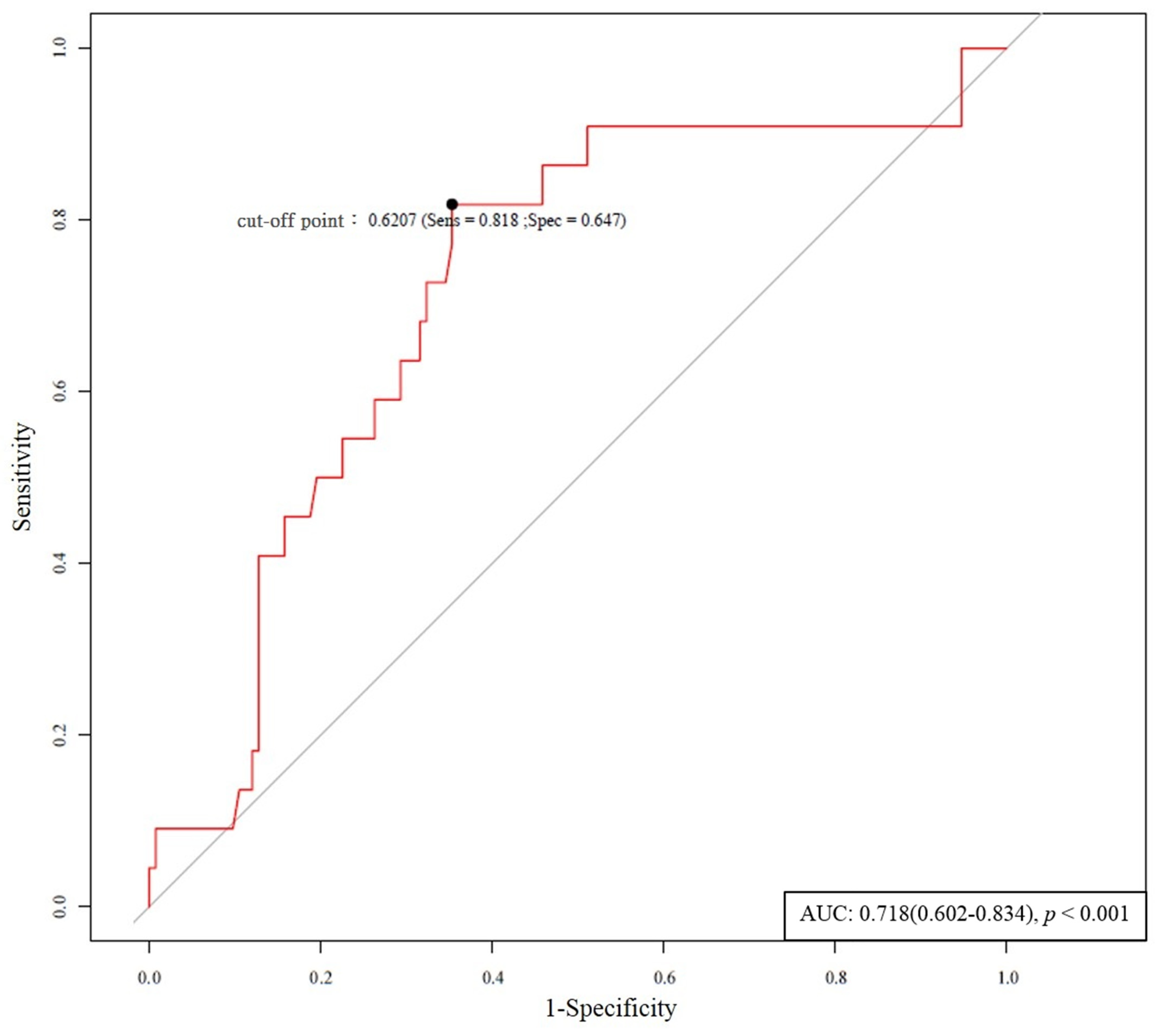
| ADMA (μmol/mL) | 13.90 (3.20 – 60.45) | <0.001 * | 4.36 (1.14–16.65) | 0.031 *,a |
| MMP-2 (ng/mL) | 0.999 (0.997–1.001) | 0.471 | ||
| MMP-9 (ng/mL) | 1.01 (1.00–1.02) | 0.011 * | 1.00 (0.99–1.01) | 0.602 |
| NO (μM) | 1.00 (1.000–1.002) | 0.089 | ||
| MCP-1 (pg/mL) | 1.00 (0.997–1.002) | 0.711 | ||
| TGF-β (pg/mL) | 1.01 (0.96–1.06) | 0.860 | ||
| TNF-α (pg/mL) | 0.99 (0.98–1.00) | 0.204 | ||
| IL-1β (pg/mL) | 1.01 (0.999–1.015) | 0.086 | ||
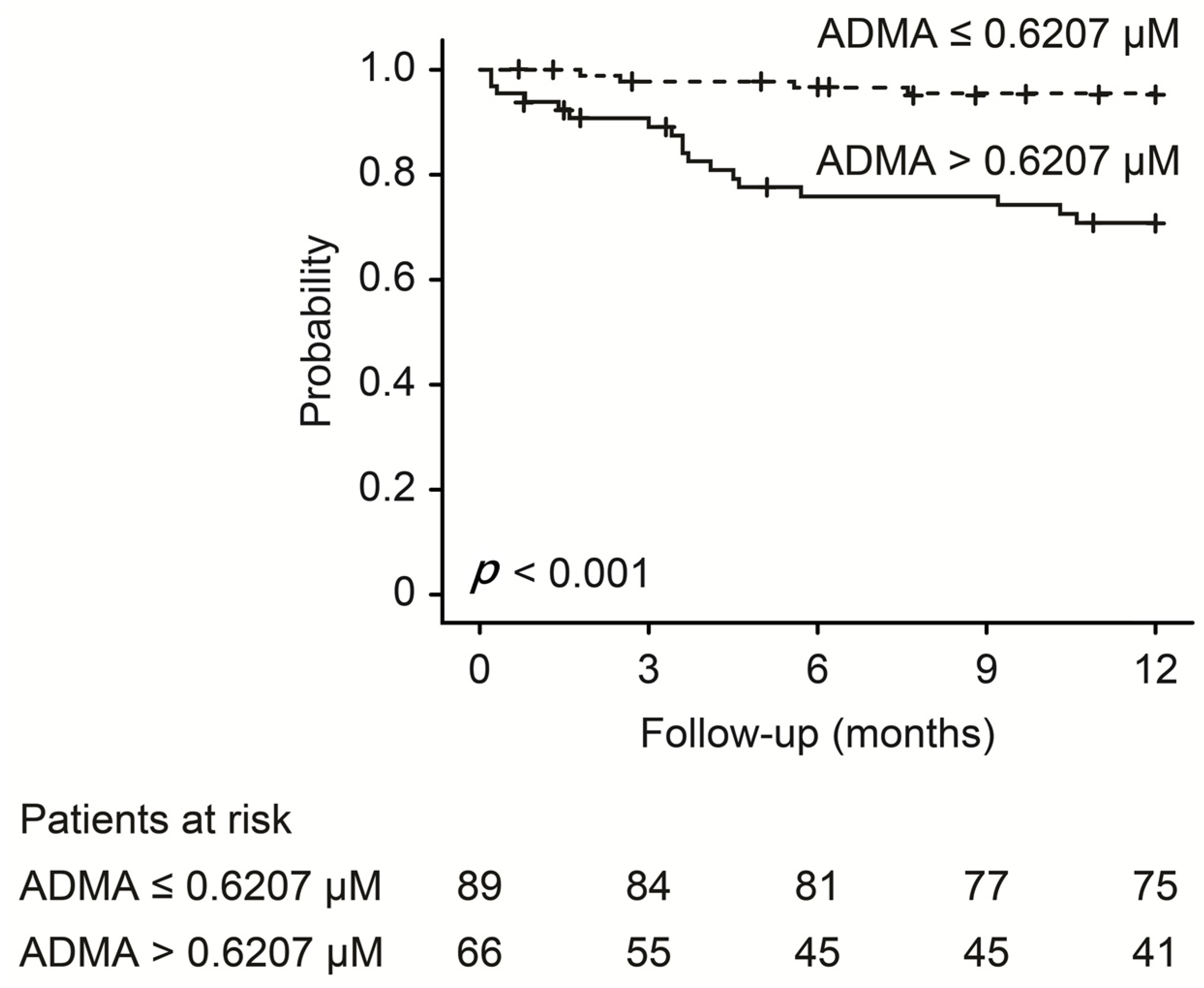
| Characteristics | ADMA > 0.6207 μM (n = 65) | ADMA ≤ 0.6207 μM (n = 90) | p |
|---|---|---|---|
| Age (years), mean ± SD | 64.29 ± 13.19 | 65.29 ± 12.61 | 0.758 |
| Sex (%) | |||
| Female | 35 (53.8%) | 38 (42.2%) | 0.205 |
| Male | 30 (46.2%) | 52 (57.8%) | |
| Access type (AVF/AVG) | |||
| AVF | 41 (63.1%) | 63 (70.0%) | 0.464 |
| AVG | 24 (36.9%) | 27 (30.0%) | |
| Duration of access use (months) | 9.02 ± 4.40 | 10.86 ± 2.90 | 0.001 * |
| Risk factors | |||
| Hypertension (%) | 57 (87.7%) | 68 (75.6%) | 0.093 |
| Diabetes mellitus (%) | 31 (47.7%) | 39 (43.3%) | 0.708 |
| Current smoking (%) | 6 (9.2%) | 7 (7.8%) | 0.977 |
| CAD (%) | 26 (40.0%) | 32 (35.6%) | 0.692 |
| PAOD (%) | 10 (15.4%) | 19 (21.1%) | 0.488 |
| Plasma biochemical data | |||
| LDL-C (mg/dL) | 104.02 ± 35.37 | 80.93 ± 28.16 | <0.001 * |
| HDL-C (mg/dL) | 41.55 ± 15.23 | 43.76 ± 15.88 | 0.390 |
| TG (mg/dL) | 159.86 ± 116.27 | 156.99 ± 90.07 | 0.863 |
| Calcium (mg/dL) | 9.60 ± 0.88 | 8.98 ± 1.05 | <0.001 * |
| Phosphate (mg/dL) | 4.76 ± 1.64 | 4.83 ± 1.52 | 0.757 |
| Albumin (mg/dL) | 3.85 ± 0.39 | 3.82 ± 0.31 | 0.549 |
| Creatinine (mg/dL) | 10.58 ± 2.98 | 10.24 ± 2.48 | 0.549 |
| Kt/V | 1.44 ± 0.23 | 1.37 ± 0.32 | 0.170 |
| Medications | |||
| Anti-platelet agents | 33 (50.8%) | 52 (57.8%) | 0.483 |
| Aspirin | 29 (44.6%) | 41 (45.6%) | 1.000 |
| Dipyridamole | 3 (4.6%) | 5 (5.6%) | 0.667 |
| Cilostazol | 7 (10.8%) | 13 (14.4%) | 1.000 |
| Coumadin | 4 (6.2%) | 1 (1.1%) | 0.196 |
| Nitrates | 28 (43.1%) | 35 (38.9%) | 0.720 |
| β-blockers | 27 (41.5%) | 26 (28.9%) | 0.142 |
| Calcium antagonists | 39 (60.0%) | 47 (52.2%) | 0.425 |
| ACEI/ARB | 26 (40.0%) | 18 (20.0%) | 0.011 * |
| Diuretics | 37 (56.9%) | 8 (8.9%) | <0.001 * |
| Lipid-lowering agents | |||
| Statin | 9 (13.8%) | 4 (4.4%) | 0.073 |
| Fibrate | 1 (1.5%) | 3 (3.3%) | 0.855 |
| Plasma biomarkers | |||
| Hs-CRP (mg/dL) | 1.23 ± 2.29 | 1.06 ± 1.82 | 0.620 |
| Homocysteine (μmol/L) | 31.18 ± 13.37 | 24.45 ± 8.42 | <0.001 * |
| MMP-2 (ng/mL) | 823.46 ± 223.15 | 847.86 ± 188.02 | 0.462 |
| MMP-9 (ng/mL) | 87.69 ± 44.83 | 44.85 ± 27.08 | <0.001 * |
| NO (μM) | 280.37 ± 268.54 | 232.96 ± 302.88 | 0.315 |
| MCP-1 (pg/mL) | 336.32 ± 114.16 | 331.40 ± 200.91 | 0.865 |
| TGF-β (pg/mL) | 6.32 ± 9.74 | 5.38 ± 6.56 | 0.515 |
| TNF-α (pg/mL) | 12.46 ± 37.64 | 54.73 ± 97.68 | <0.001 * |
| IL-1β (pg/mL) | 90.48 ± 41.61 | 37.93 ± 32.18 | <0.001 * |
| Events at follow-up (n %) | 18 (27.7%) | 4 (4.4%) | <0.001 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chan, J.-S.; Hsiao, P.-J.; Chiang, W.-F.; Roy-Chaudhury, P. The Role of Oxidative Stress Markers in Predicting Acute Thrombotic Occlusion of Haemodialysis Vascular Access and Progressive Stenotic Dysfunction Demanding Angioplasty. Antioxidants 2021, 10, 569. https://doi.org/10.3390/antiox10040569
Chan J-S, Hsiao P-J, Chiang W-F, Roy-Chaudhury P. The Role of Oxidative Stress Markers in Predicting Acute Thrombotic Occlusion of Haemodialysis Vascular Access and Progressive Stenotic Dysfunction Demanding Angioplasty. Antioxidants. 2021; 10(4):569. https://doi.org/10.3390/antiox10040569
Chicago/Turabian StyleChan, Jenq-Shyong, Po-Jen Hsiao, Wen-Fang Chiang, and Prabir Roy-Chaudhury. 2021. "The Role of Oxidative Stress Markers in Predicting Acute Thrombotic Occlusion of Haemodialysis Vascular Access and Progressive Stenotic Dysfunction Demanding Angioplasty" Antioxidants 10, no. 4: 569. https://doi.org/10.3390/antiox10040569
APA StyleChan, J.-S., Hsiao, P.-J., Chiang, W.-F., & Roy-Chaudhury, P. (2021). The Role of Oxidative Stress Markers in Predicting Acute Thrombotic Occlusion of Haemodialysis Vascular Access and Progressive Stenotic Dysfunction Demanding Angioplasty. Antioxidants, 10(4), 569. https://doi.org/10.3390/antiox10040569





