To Synthesize Hydroxyapatite by Modified Low Temperature Method Loaded with Bletilla striata Polysaccharide as Antioxidant for the Prevention of Sarcopenia by Intramuscular Administration
Abstract
1. Introduction
2. Materials and Methods
2.1. Bletilla Striata Polysaccharide (BSP) Extraction and Purification
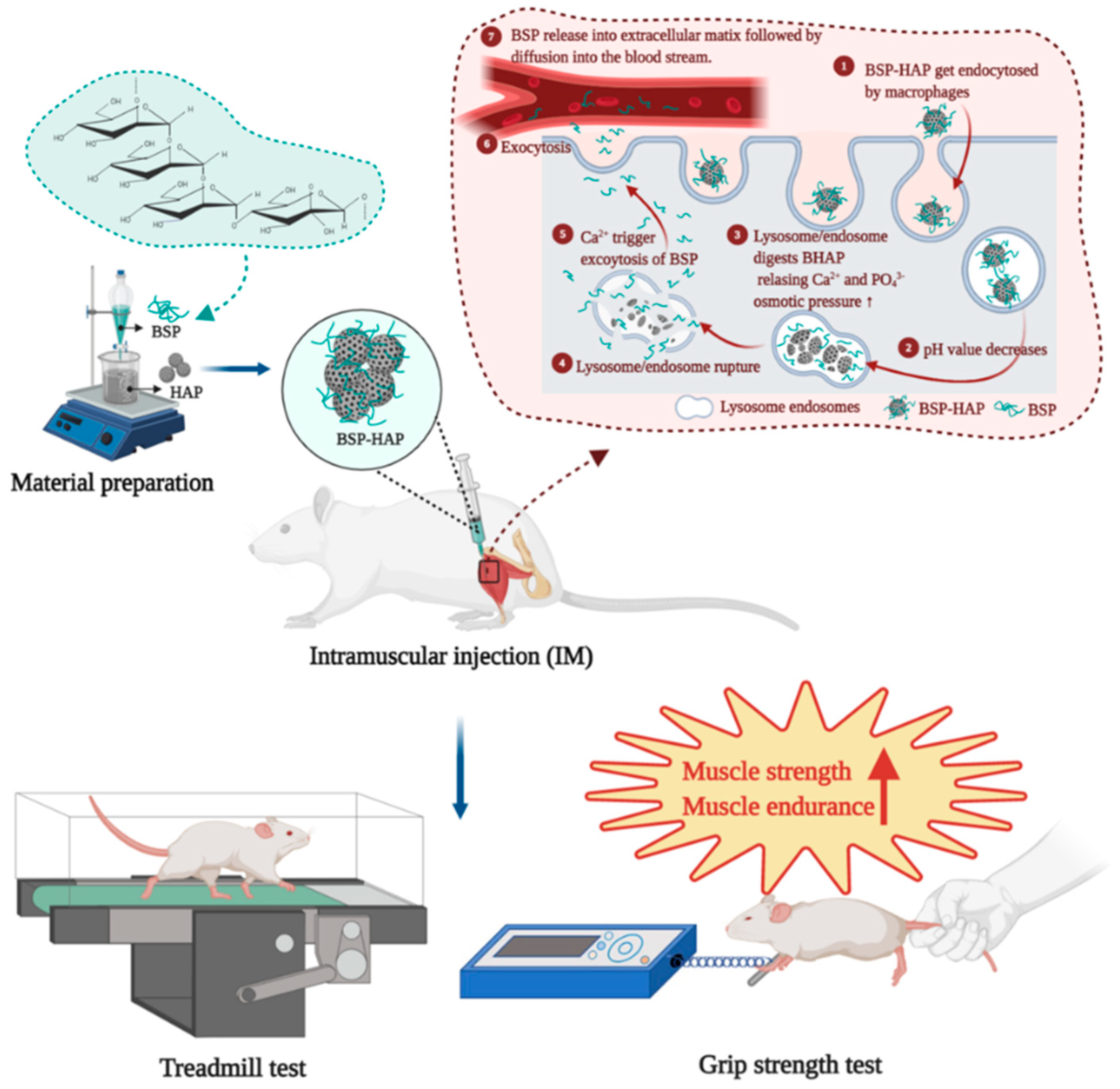
2.2. Preparation of the BSP-HAP
2.3. Material Characterization
2.4. BSP Loading Efficiency and Release Profile
2.5. The Evaluation of Cell Viability and Cytotoxicity
2.6. Detection of Cellular ROS Generation
2.7. Animal Model with Lipopolysaccharide-Induced Sarcopenia Model
2.8. Treadmill Test and Grip Strength Test
2.9. Fat-to-Muscle Ratio Determination by Magnetic Resonance Imaging (MRI)
2.10. Blood Biochemical Analysis
2.11. Statistical Analysis
3. Results
3.1. Molecular Structure, Functional Groups and Crystal Structure Identification
3.2. Morphology, Particle Size and Chemical Composition of BSP-HAP
3.3. Determination of the Loading Efficiency of BSP
3.4. Release Profile of BSP from BSP-HAP
3.5. Cytotoxicity Assay and Cell Viability Assay
3.6. Antioxidant Effect of BSP-HAP
3.7. Muscle Endurance Analysis by Treadmill Test and Grip Strength Measurement
3.8. The Ratio of Fat Mass to Lean Mass in Rat Muscle by MRI
3.9. Safety of BSP-HAP in Vivo by Serological Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kim, T.N.; Choi, K.M. Sarcopenia: Definition, epidemiology, and pathophysiology. J. Bone Metab. 2013, 20, 1–10. [Google Scholar] [CrossRef]
- Argiles, J.M.; Busquets, S.; Stemmler, B.; Lopez-Soriano, F.J. Cachexia and sarcopenia: Mechanisms and potential targets for intervention. Curr. Opin. Pharmacol. 2015, 22, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Dalle, S.; Rossmeislova, L.; Koppo, K. The Role of Inflammation in Age-Related Sarcopenia. Front. Physiol. 2017, 8, 1045. [Google Scholar] [CrossRef]
- Damiano, S.; Muscariello, E.; La Rosa, G.; Di Maro, M.; Mondola, P.; Santillo, M. Dual Role of Reactive Oxygen Species in Muscle Function: Can Antioxidant Dietary Supplements Counteract Age-Related Sarcopenia? Int. J. Mol. Sci. 2019, 20, 3815. [Google Scholar] [CrossRef] [PubMed]
- Bian, A.L.; Hu, H.Y.; Rong, Y.D.; Wang, J.; Wang, J.X.; Zhou, X.Z. A study on relationship between elderly sarcopenia and inflammatory factors IL-6 and TNF-alpha. Eur. J. Med. Res. 2017, 22. [Google Scholar] [CrossRef] [PubMed]
- Meng, S.J.; Yu, L.J. Oxidative stress, molecular inflammation and sarcopenia. Int. J. Mol. Sci. 2010, 11, 1509–1526. [Google Scholar] [CrossRef]
- Luk, H.Y.; Appell, C.; Chyu, M.C.; Chen, C.H.; Wang, C.Y.; Yang, R.S.; Shen, C.L. Impacts of Green Tea on Joint and Skeletal Muscle Health: Prospects of Translational Nutrition. Antioxidants 2020, 9, 1050. [Google Scholar] [CrossRef]
- Denison, H.J.; Cooper, C.; Sayer, A.A.; Robinson, S.M. Prevention and optimal management of sarcopenia: A review of combined exercise and nutrition interventions to improve muscle outcomes in older people. Clin. Interv. Aging 2015, 10, 859–869. [Google Scholar] [CrossRef]
- Khor, S.C.; Karim, N.A.; Ngah, W.Z.W.; Yusof, Y.A.M.; Makpol, S. Vitamin E in Sarcopenia: Current Evidences on Its Role in Prevention and Treatment. Oxid. Med. Cell. Longev. 2014, 2014. [Google Scholar] [CrossRef]
- Borst, S.E. Interventions for sarcopenia and muscle weakness in older people. Age Ageing 2004, 33, 548–555. [Google Scholar] [CrossRef] [PubMed]
- Moon, J.J.; Park, S.G.; Ryu, S.M.; Park, C.H. New Skeletal Muscle Mass Index in Diagnosis of Sarcopenia. J. Bone Metab. 2018, 25, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Sun, J.; Luo, Y.; Xue, W.; Diao, H.; Dong, L.; Chen, J.; Zhang, J. A polysaccharide isolated from the medicinal herb Bletilla striata induces endothelial cells proliferation and vascular endothelial growth factor expression in vitro. Biotechnol. Lett. 2006, 28, 539–543. [Google Scholar] [CrossRef]
- Zhang, C.; He, Y.; Chen, Z.; Shi, J.; Qu, Y.; Zhang, J. Effect of Polysaccharides from Bletilla striata on the Healing of Dermal Wounds in Mice. Evid. Based Complement. Altern. Med. 2019, 2019, 9212314. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.Y.; Chen, S.H.; Chen, C.H.; Chou, P.Y.; Yang, C.C.; Lin, F.H. Polysaccharide Extracted from Bletilla striata Promotes Proliferation and Migration of Human Tenocytes. Polymers 2020, 12, 2567. [Google Scholar] [CrossRef]
- Yue, L.; Wang, W.; Wang, Y.; Du, T.; Shen, W.; Tang, H.; Wang, Y.; Yin, H. Bletilla striata polysaccharide inhibits angiotensin II-induced ROS and inflammation via NOX4 and TLR2 pathways. Int. J. Biol. Macromol. 2016, 89, 376–388. [Google Scholar] [CrossRef] [PubMed]
- Thacker, M.; Tseng, C.L.; Chang, C.Y.; Jakfar, S.; Chen, H.Y.; Lin, F.H. Mucoadhesive Bletilla striata Polysaccharide-Based Artificial Tears to Relieve Symptoms and Inflammation in Rabbit with Dry Eyes Syndrome. Polymers 2020, 12, 1465. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Kao, W.J. Drug release kinetics and transport mechanisms of non-degradable and degradable polymeric delivery systems. Expert Opin. Drug Del. 2010, 7, 429–444. [Google Scholar] [CrossRef]
- Shyong, Y.J.; Wang, M.H.; Tseng, H.C.; Cheng, C.; Chang, K.C.; Lin, F.H. Mesoporous Hydroxyapatite as Olanzapine Carrier Provides a Long-Acting Effect in Antidepression Treatment. J. Med. Chem. 2015, 58, 8463–8474. [Google Scholar] [CrossRef]
- Itokazu, M.; Yang, W.Y.; Aoki, T.; Ohara, A.; Kato, N. Synthesis of antibiotic-loaded interporous hydroxyapatite blocks by vacuum method and in vitro drug release testing. Biomaterials 1998, 19, 817–819. [Google Scholar] [CrossRef]
- Shyong, Y.J.; Wang, M.H.; Kuo, L.W.; Su, C.F.; Kuo, W.T.; Chang, K.C.; Lin, F.H. Mesoporous hydroxyapatite as a carrier of olanzapine for long-acting antidepression treatment in rats with induced depression. J. Control. Release 2017, 255, 62–72. [Google Scholar] [CrossRef]
- Agrawal, A.; Suryakumar, G.; Rathor, R. Role of defective Ca2+ signaling in skeletal muscle weakness: Pharmacological implications. J. Cell Commun. Signal. 2018, 12, 645–659. [Google Scholar] [CrossRef] [PubMed]
- Barbiera, A.; Pelosi, L.; Sica, G.; Scicchitano, B.M. Nutrition and microRNAs: Novel Insights to Fight Sarcopenia. Antioxidants 2020, 9, 951. [Google Scholar] [CrossRef]
- Lai, Y.L.; Lin, Y.Y.; Sadhasivam, S.; Kuan, C.Y.; Chi, C.Y.; Dong, G.C.; Lin, F.H. Efficacy of Bletilla striata polysaccharide on hydrogen peroxide-induced apoptosis of osteoarthritic chondrocytes. J. Polym. Res. 2018, 25. [Google Scholar] [CrossRef]
- Huang, Y.-C.; Lin, P.-W.; Qiu, W.-J.; Yang, T.-I. Amphiphilic Polymer-Assisted Synthesis of Hydroxyapatite Particles and Their Influence on the Rheological and Mechanical Properties of Thermosensitive Hydrogels. Biomed. Eng. Appl. Basis Commun. 2016, 28. [Google Scholar] [CrossRef]
- Hsiao, M.Y.; Lin, P.C.; Lin, A.C.; Wu, Y.W.; Chen, W.S.; Lin, F.H. Oxidized Hyaluronic Acid/Adipic Acid Dihydrazide Hydrogel as Drug-Carrier for Cytoprotective Medications-Preliminary Results. Biomed. Eng. App. Bas. C 2019, 31. [Google Scholar] [CrossRef]
- Yang, C.C.; Wang, C.X.; Kuan, C.Y.; Chi, C.Y.; Chen, C.Y.; Lin, Y.Y.; Chen, G.S.; Hou, C.H.; Lin, F.H. Using C-doped TiO2 Nanoparticles as a Novel Sonosensitizer for Cancer Treatment. Antioxidants 2020, 9, 880. [Google Scholar] [CrossRef]
- Buford, T.W.; Anton, S.D.; Judge, A.R.; Marzetti, E.; Wohlgemuth, S.E.; Carter, C.S.; Leeuwenburgh, C.; Pahor, M.; Manini, T.M. Models of accelerated sarcopenia: Critical pieces for solving the puzzle of age-related muscle atrophy. Ageing Res. Rev. 2010, 9, 369–383. [Google Scholar] [CrossRef] [PubMed]
- Wan, J.; Chen, D.W.; Yu, B.; Luo, Y.H.; Mao, X.B.; Zheng, P.; Yu, J.; Luo, J.Q.; He, J. Leucine Protects Against Skeletal Muscle Atrophy in Lipopolysaccharide-Challenged Rats. J. Med. Food 2017, 20, 93–101. [Google Scholar] [CrossRef]
- Ko, I.G.; Jeong, J.W.; Kim, Y.H.; Jee, Y.S.; Kim, S.E.; Kim, S.H.; Jin, J.J.; Kim, C.J.; Chung, K.J. Aerobic Exercise Affects Myostatin Expression in Aged Rat Skeletal Muscles: A Possibility of Antiaging Effects of Aerobic Exercise Related With Pelvic Floor Muscle and Urethral Rhabdosphincter. Int. Neurourol. J. 2014, 18, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Kemmochi, Y.; Ohta, T.; Motohashi, Y.; Kaneshige, A.; Katsumi, S.; Kakimoto, K.; Yasui, Y.; Anagawa-Nakamura, A.; Toyoda, K.; Taniai-Riya, E.; et al. Pathophysiological analyses of skeletal muscle in obese type 2 diabetes SDT fatty rats. J. Toxicol. Pathol. 2018, 31, 113–123. [Google Scholar] [CrossRef] [PubMed]
- Carroll, M.F.; Schade, D.S. A practical approach to hypercalcemia. Am. Fam. Physician 2003, 67, 1959–1966. [Google Scholar]
- Baird, M.F.; Graham, S.M.; Baker, J.S.; Bickerstaff, G.F. Creatine-kinase- and exercise-related muscle damage implications for muscle performance and recovery. J. Nutr. Metab. 2012, 2012, 960363. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, D.; Chen, S.J.; Wang, Y.; Jiang, H.X.; Yin, H.P. A new glucomannan from Bletilla striata: Structural and anti-fibrosis effects. Fitoterapia 2014, 92, 72–78. [Google Scholar] [CrossRef] [PubMed]
- Oberbek, P.; Bolek, T.; Chlanda, A.; Hirano, S.; Kusnieruk, S.; Rogowska-Tylman, J.; Nechyporenko, G.; Zinchenko, V.; Swieszkowski, W.; Puzyn, T. Characterization and influence of hydroxyapatite nanopowders on living cells. Beilstein J. Nanotechnol. 2018, 9, 3079–3094. [Google Scholar] [CrossRef]
- Romanick, M.; Thompson, L.V.; Brown-Borg, H.M. Murine models of atrophy, cachexia, and sarcopenia in skeletal muscle. BBA Mol. Basis Dis. 2013, 1832, 1410–1420. [Google Scholar] [CrossRef]
- Wilson, D.; Jackson, T.; Sapey, E.; Lord, J.M. Frailty and sarcopenia: The potential role of an aged immune system. Ageing Res. Rev. 2017, 36, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Ishida, J.; Saitoh, M.; Doehner, W.; von Haehling, S.; Anker, M.; Anker, S.D.; Springer, J. Animal models of cachexia and sarcopenia in chronic illness: Cardiac function, body composition changes and therapeutic results. Int. J. Cardiol. 2017, 238, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Scharf, G.; Heineke, J. Finding good biomarkers for sarcopenia. J. Cachexia Sarcopeni Muscle 2012, 3, 145–148. [Google Scholar] [CrossRef]
- Sumien, N.; Shetty, R.A.; Gonzales, E.B. Creatine, Creatine Kinase, and Aging. Subcell. Biochem. 2018, 90, 145–168. [Google Scholar] [CrossRef]








| Sample | C Atomic % | O Atomic % | P Atomic % | Ca Atomic % |
|---|---|---|---|---|
| HAP | 17.92 | 53.78 | 10.62 | 17.69 |
| BSP-HAP | 34.20 | 49.03 | 7.24 | 9.53 |
| Sample | Result |
|---|---|
| BSP-HAP | BET Surface Area: 60.7645 ± 0.7091 m2/g |
| Correlation Coefficient: 0.9994673 | |
| Langmuir Surface Area: 96.2750 ± 2.4493 m2/g | |
| Correlation Coefficient: 0.997421 |
| Control | LPS | LPS-BSP-HAP | Reference | |
|---|---|---|---|---|
| Total protein (g/dL) | 7.0 ± 0.3 | 7.4 ± 0.1 | 6.9 ± 0.2 | 6.6 ± 1.0 |
| Creatine kinase (U/L) | 130.9 ± 23.4 | 958.3 ± 200.6 * | 148.5 ± 13.4 # | 131.3 ± 66.4 |
| LDH (U/L) | 397.0 ± 77.6 | 759.4 ± 85.0 * | 329.3 ± 95.5 # | 500.0 ± 200.0 |
| Calcium (mg/dL) | 9.7 ± 0.1 | 9.8 ± 0.1 | 9.7 ± 0.2 | 9.5 ± 0.9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liang, Y.-J.; Hong, J.-Y.; Yang, I.-H.; Zhou, X.-R.; Lin, Y.-W.; Lin, T.-C.; Hou, C.-H.; Lin, F.-H. To Synthesize Hydroxyapatite by Modified Low Temperature Method Loaded with Bletilla striata Polysaccharide as Antioxidant for the Prevention of Sarcopenia by Intramuscular Administration. Antioxidants 2021, 10, 488. https://doi.org/10.3390/antiox10030488
Liang Y-J, Hong J-Y, Yang I-H, Zhou X-R, Lin Y-W, Lin T-C, Hou C-H, Lin F-H. To Synthesize Hydroxyapatite by Modified Low Temperature Method Loaded with Bletilla striata Polysaccharide as Antioxidant for the Prevention of Sarcopenia by Intramuscular Administration. Antioxidants. 2021; 10(3):488. https://doi.org/10.3390/antiox10030488
Chicago/Turabian StyleLiang, Ya-Jyun, Jia-Yu Hong, I-Hsuan Yang, Xin-Ran Zhou, Yi-Wen Lin, Tzu-Chieh Lin, Chun-Han Hou, and Feng-Huei Lin. 2021. "To Synthesize Hydroxyapatite by Modified Low Temperature Method Loaded with Bletilla striata Polysaccharide as Antioxidant for the Prevention of Sarcopenia by Intramuscular Administration" Antioxidants 10, no. 3: 488. https://doi.org/10.3390/antiox10030488
APA StyleLiang, Y.-J., Hong, J.-Y., Yang, I.-H., Zhou, X.-R., Lin, Y.-W., Lin, T.-C., Hou, C.-H., & Lin, F.-H. (2021). To Synthesize Hydroxyapatite by Modified Low Temperature Method Loaded with Bletilla striata Polysaccharide as Antioxidant for the Prevention of Sarcopenia by Intramuscular Administration. Antioxidants, 10(3), 488. https://doi.org/10.3390/antiox10030488







