Fighting Bisphenol A-Induced Male Infertility: The Power of Antioxidants
Abstract
1. Introduction
2. BPA: What Is This?
3. BPA-Induced Alterations in Testicular Structure, Function, and Semen Parameters
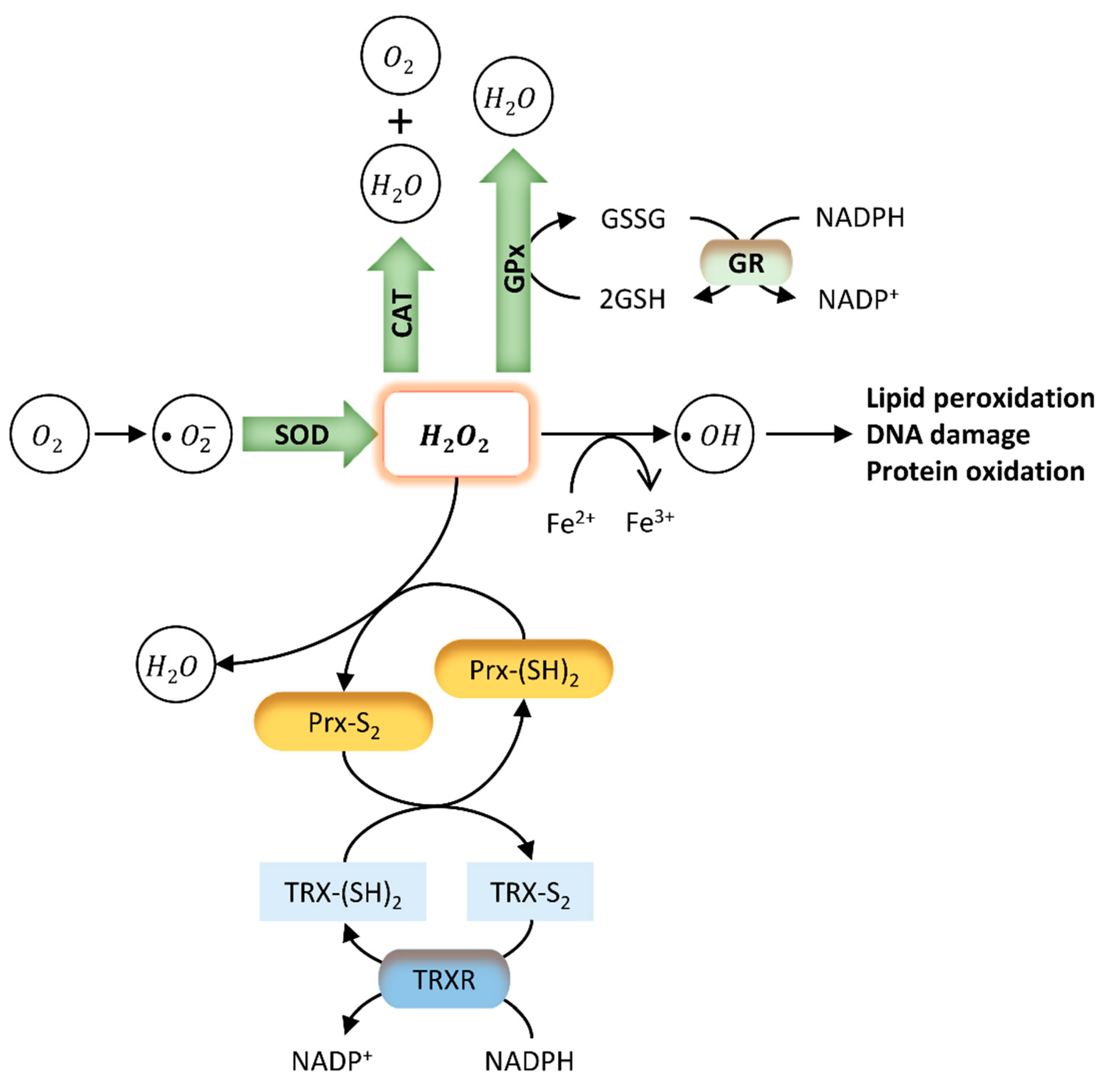
4. Impact of BPA Exposure on Oxidative Stress in Testis and Sperm
5. Ameliorative Effects of Antioxidants in BPA-Induced Reproductive Toxicity
5.1. Melatonin
5.2. Vitamins
5.3. Natural Extracts
5.4. Other Antioxidants
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kasonga, T.K.; Coetzee, M.A.; Kamika, I.; Ngole-Jeme, V.M.; Momba, M.N.B. Endocrine-disruptive chemicals as contaminants of emerging concern in wastewater and surface water: A review. J. Environ. Manag. 2021, 277, 111485. [Google Scholar] [CrossRef]
- Kelly, M.; Connolly, L.; Dean, M. Public Awareness and Risk Perceptions of Endocrine Disrupting Chemicals: A Qualitative Study. Int. J. Environ. Res. Public Health 2020, 17, 7778. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Community Strategy for Endocrine Disrupters—A Range of Substances Suspected of Interfering with the Hormone Systems of Humans and Wildlife; Commission of the European Communities: Brussels, Belgium, 1999.
- Fuhrman, V.F.; Tal, A.; Arnon, S. Why endocrine disrupting chemicals (EDCs) challenge traditional risk assessment and how to respond. J. Hazard. Mater. 2015, 286, 589–611. [Google Scholar] [CrossRef] [PubMed]
- Vandenberg, L.N.; Colborn, T.; Hayes, T.B.; Heindel, J.J.; Jacobs, D.R.J.; Lee, D.H.; Shioda, T.; Soto, A.M.; vom Saal, F.S.; Welshons, W.V.; et al. Hormones and Endocrine-Disrupting Chemicals: Low-Dose Effects and Nonmonotonic Dose Responses. Endocr. Rev. 2012, 33, 378–455. [Google Scholar] [CrossRef]
- Diamanti-Kandarakis, E.; Bourguignon, J.-P.; Giudice, L.C.; Hauser, R.; Prins, G.S.; Soto, A.M.; Zoeller, R.T.; Gore, A.C. Endocrine-Disrupting Chemicals: An Endocrine Society Scientific Statement. Endocr. Rev. 2009, 30, 293–342. [Google Scholar] [CrossRef]
- Balabanič, D.; Rupnik, M.; Klemenčič, A.K. Negative impact of endocrine-disrupting compounds on human reproductive health. Reprod. Fertil. Dev. 2011, 23, 403–416. [Google Scholar] [CrossRef]
- Chiang, C.; Mahalingam, S.; Flaws, J.A. Environmental Contaminants Affecting Fertility and Somatic Health. Semin. Reprod. Med. 2017, 35, 241–249. [Google Scholar] [CrossRef] [PubMed]
- Giwercmanz, A.; Rylander, L.; Giwercman, Y.L. Influence of endocrine disruptors on human male fertility. Reprod. Biomed. Online 2007, 15, 633–642. [Google Scholar] [CrossRef]
- Manikkam, M.; Tracey, R.; Guerrero-Bosagna, C.; Skinner, M.K. Plastics Derived Endocrine Disruptors (BPA, DEHP and DBP) Induce Epigenetic Transgenerational Inheritance of Obesity, Reproductive Disease and Sperm Epimutations. PLoS ONE 2013, 8, e55387. [Google Scholar] [CrossRef]
- Sharma, A.; Mollier, J.; Brocklesby, R.W.K.; Caves, C.; Jayasena, C.N.; Minhas, S. Endocrine-disrupting chemicals and male reproductive health. Reprod. Med. Biol. 2020, 19, 243–253. [Google Scholar] [CrossRef] [PubMed]
- Calafat, A.M.; Ye, X.; Wong, L.-Y.; Reidy, J.A.; Needham, L.L. Exposure of the U.S. Population to Bisphenol A and 4-tertiary-Octylphenol: 2003–2004. Environ. Health Perspect. 2008, 116, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Lang, I.A.; Galloway, T.S.; Scarlett, A.; Henley, W.E.; Depledge, M.; Wallace, R.B.; Melzer, D. Association of Urinary Bisphenol A Concentration with Medical Disorders and Laboratory Abnormalities in Adults. JAMA 2008, 300, 1303–1310. [Google Scholar] [CrossRef]
- Vitku, J.; Sosvorova, L.; Chlupacova, T.; Hampl, R.; Hill, M.; Sobotka, V.; Heracek, J.; Bicikova, M.; Starka, L. Differences in Bisphenol A and Estrogen Levels in the Plasma and Seminal Plasma of Men with Different Degrees of Infertility. Physiol. Res. 2015, 64, S303–S311. [Google Scholar] [CrossRef] [PubMed]
- Vandenberg, L.N.; Hauser, R.; Marcus, M.; Olea, N.; Welshons, W.V. Human exposure to bisphenol A (BPA). Reprod. Toxicol. 2007, 24, 139–177. [Google Scholar] [CrossRef] [PubMed]
- Völkel, W.; Bittner, N.; Dekant, W. Quantitation of Bisphenol a and Bisphenol a Glucuronide in Biological Samples by High Performance Liquid Chromatography-Tandem Mass Spectrometry. Drug Metab. Dispos. 2005, 33, 1748–1757. [Google Scholar] [CrossRef] [PubMed]
- Rochester, J.R. Bisphenol A and human health: A review of the literature. Reprod. Toxicol. 2013, 42, 132–155. [Google Scholar] [CrossRef] [PubMed]
- De Toni, L.; Ponce, M.D.R.; Petre, G.C.; Rtibi, K.; Di Nisio, A.; Foresta, C. Bisphenols and Male Reproductive Health: From Toxicological Models to Therapeutic Hypotheses. Front. Endocrinol. 2020, 11, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Cariati, F.; D’Uonno, N.; Borrillo, F.; Iervolino, S.; Galdiero, G.; Tomaiuolo, R. Bisphenol A: An emerging threat to male fertility. Reprod. Biol. Endocrinol. 2019, 17, 1–8. [Google Scholar] [CrossRef]
- Radwan, M.; Wielgomas, B.; Dziewirska, E.; Radwan, P.; Kałużny, P.; Klimowska, A.; Hanke, W.; Jurewicz, J. Urinary Bisphenol A Levels and Male Fertility. Am. J. Men Health 2018, 12, 2144–2151. [Google Scholar] [CrossRef]
- Anjum, S.; Rahman, S.; Kaur, M.; Ahmad, F.; Rashid, H.; Ansari, R.A.; Raisuddin, S. Melatonin ameliorates bisphenol A-induced biochemical toxicity in testicular mitochondria of mouse. Food Chem. Toxicol. 2011, 49, 2849–2854. [Google Scholar] [CrossRef] [PubMed]
- Kaur, S.; Saluja, M.; Bansal, M.P. Bisphenol A induced oxidative stress and apoptosis in mice testes: Modulation by selenium. Andrology 2018, 50, e12834. [Google Scholar] [CrossRef]
- Hulak, M.; Gazo, I.; Shaliutina, A.; Linhartova, P. In vitro effects of bisphenol A on the quality parameters, oxidative stress, DNA integrity and adenosine triphosphate content in sterlet (Acipenser ruthenus) spermatozoa. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2013, 158, 64–71. [Google Scholar] [CrossRef]
- D’Cruz, S.C.; Jubendradass, R.; Mathur, P.P. Bisphenol A Induces Oxidative Stress and Decreases Levels of Insulin Receptor Substrate 2 and Glucose Transporter 8 in Rat Testis. Reprod. Sci. 2011, 19, 163–172. [Google Scholar] [CrossRef]
- Wu, H.-J.; Liu, C.; Duan, W.-X.; Xu, S.-C.; He, M.-D.; Chen, C.-H.; Wang, Y.; Zhou, Z.; Yu, Z.-P.; Zhang, L.; et al. Melatonin ameliorates bisphenol A-induced DNA damage in the germ cells of adult male rats. Mutat. Res. Toxicol. Environ. Mutagen. 2013, 752, 57–67. [Google Scholar] [CrossRef] [PubMed]
- Othman, A.I.; Edrees, G.M.; El-Missiry, M.A.; Ali, D.A.; Aboel-Nour, M.; Dabdoub, B.R. Melatonin controlled apoptosis and protected the testes and sperm quality against bisphenol A-induced oxidative toxicity. Toxicol. Ind. Health 2016, 32, 1537–1549. [Google Scholar] [CrossRef]
- Aydoğan, M.; Korkmaz, A.; Barlas, N.; Kolankaya, D. Pro-oxidant effect of vitamin C coadministration with bisphenol A, nonylphenol, and octylphenol on the reproductive tract of male rats. Drug Chem. Toxicol. 2009, 33, 193–203. [Google Scholar] [CrossRef]
- Yuan, C.; Wang, L.; Zhu, L.; Ran, B.; Xue, X.; Wang, Z. N-acetylcysteine alleviated bisphenol A-induced testicular DNA hypermethylation of rare minnow (Gobiocypris rarus) by increasing cysteine contents. Ecotoxicol. Environ. Saf. 2019, 173, 243–250. [Google Scholar] [CrossRef]
- Carneiro, M.F.H.; Shin, N.; Karthikraj, R.; Barbosa, F.; Kannan, K.; Colaiácovo, M.P. Antioxidant CoQ10 Restores Fertility by Rescuing Bisphenol A-Induced Oxidative DNA Damage in the Caenorhabditis elegans Germline. Genetics 2020, 214, 381–395. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Chen, C.; Jiang, Z.; Wang, M.; Jiang, H.; Zhang, X. Protective effect of Cordyceps militaris extract against bisphenol A induced reproductive damage. Syst. Biol. Reprod. Med. 2016, 62, 249–257. [Google Scholar] [CrossRef]
- Grami, D.; Rtibi, K.; Selmi, S.; Jridi, M.; Sebai, H.; Marzouki, L.; Sabovic, I.; Foresta, C.; De Toni, L. Aqueous extract of Eruca Sativa protects human spermatozoa from mitochondrial failure due to bisphenol A exposure. Reprod. Toxicol. 2018, 82, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Grami, D.; Rtibi, K.; Hammami, I.; Selmi, S.; De Toni, L.; Foresta, C.; Marzouki, L.; Sebai, H. Protective Action of Eruca sativa Leaves Aqueous Extracts Against Bisphenol A-Caused In Vivo Testicular Damages. J. Med. Food 2020, 23, 600–610. [Google Scholar] [CrossRef]
- Tomza-Marciniak, A.; Stępkowska, P.; Kuba, J.; Pilarczyk, B. Effect of bisphenol A on reproductive processes: A review of in vitro, in vivo and epidemiological studies. J. Appl. Toxicol. 2018, 38, 51–80. [Google Scholar] [CrossRef]
- Fenichel, P.; Chevalier, N.; Brucker-Davis, F. Bisphenol A: An endocrine and metabolic disruptor. In Annales d’Endocrinologie; Elsevier: Amsterdam, The Netherlands, 2013; pp. 211–220. [Google Scholar]
- Almeida, S.; Raposo, A.; Almeida-González, M.; Carrascosa, C. Bisphenol A: Food Exposure and Impact on Human Health. Compr. Rev. Food Sci. Food Saf. 2018, 17, 1503–1517. [Google Scholar] [CrossRef]
- López-Cervantes, J.; Paseiro-Losada, P. Determination of bisphenol A in, and its migration from, PVC stretch film used for food packaging. Food Addit. Contam. 2003, 20, 596–606. [Google Scholar] [CrossRef]
- Tan, B.; Mustafa, A. Leaching of Bisphenol A from New and Old Babies’ Bottles, and New Babies’ Feeding Teats. Asia Pac. J. Public Health 2003, 15, 118–123. [Google Scholar] [CrossRef]
- Wilson, N.K.; Chuang, J.C.; Lyu, C.; Menton, R.; Morgan, M.K. Aggregate exposures of nine preschool children to persistent organic pollutants at day care and at home. J. Expo. Sci. Environ. Epidemiol. 2003, 13, 187–202. [Google Scholar] [CrossRef]
- Wilson, N.K.; Chuang, J.C.; Morgan, M.K.; Lordo, R.A.; Sheldon, L.S. An observational study of the potential exposures of preschool children to pentachlorophenol, bisphenol-A, and nonylphenol at home and daycare. Environ. Res. 2007, 103, 9–20. [Google Scholar] [CrossRef]
- Hines, C.J.; Christianson, A.L.; Jackson, M.V.; Ye, X.; Pretty, J.R.; Arnold, J.; Calafat, A.M. An Evaluation of the Relationship among Urine, Air, and Hand Measures of Exposure to Bisphenol A (BPA) in US Manufacturing Workers. Ann. Work. Expo. Health 2018, 62, 840–851. [Google Scholar] [CrossRef]
- Porras, S.P.; Heinälä, M.; Santonen, T. Bisphenol A exposure via thermal paper receipts. Toxicol. Lett. 2014, 230, 413–420. [Google Scholar] [CrossRef]
- Biedermann, S.; Tschudin, P.; Grob, K. Transfer of bisphenol A from thermal printer paper to the skin. Anal. Bioanal. Chem. 2010, 398, 571–576. [Google Scholar] [CrossRef]
- Liao, C.; Kannan, K. Widespread Occurrence of Bisphenol A in Paper and Paper Products: Implications for Human Exposure. Environ. Sci. Technol. 2011, 45, 9372–9379. [Google Scholar] [CrossRef]
- Thayer, K.A.; Doerge, D.R.; Hunt, D.; Schurman, S.H.; Twaddle, N.C.; Churchwell, M.I.; Garantziotis, S.; Kissling, G.E.; Easterling, M.R.; Bucher, J.R.; et al. Pharmacokinetics of bisphenol A in humans following a single oral administration. Environ. Int. 2015, 83, 107–115. [Google Scholar] [CrossRef]
- Pottenger, L.H.; Domoradzki, J.Y.; Markham, D.A.; Hansen, S.C.; Cagen, S.Z.; Waechter, J.M. The Relative Bioavailability and Metabolism of Bisphenol A in Rats Is Dependent upon the Route of Administration. Toxicol. Sci. 2000, 54, 3–18. [Google Scholar] [CrossRef]
- Tominaga, T.; Negishi, T.; Hirooka, H.; Miyachi, A.; Inoue, A.; Hayasaka, I.; Yoshikawa, Y. Toxicokinetics of bisphenol A in rats, monkeys and chimpanzees by the LC–MS/MS method. Toxicology 2006, 226, 208–217. [Google Scholar] [CrossRef]
- Völkel, W.; Kiranoglu, M.; Fromme, H. Determination of free and total bisphenol A in human urine to assess daily uptake as a basis for a valid risk assessment. Toxicol. Lett. 2008, 179, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Gould, J.C.; Leonard, L.S.; Maness, S.C.; Wagner, B.L.; Conner, K.; Zacharewski, T.; Safe, S.; McDonnell, D.P.; Gaido, K.W. Bisphenol A interacts with the estrogen receptor α in a distinct manner from estradiol. Mol. Cell. Endocrinol. 1998, 142, 203–214. [Google Scholar] [CrossRef]
- Kuiper, G.G.J.M.; Lemmen, J.G.; Carlsson, B.; Corton, J.C.; Safe, S.H.; Van Der Saag, P.T.; Van Der Burg, B.; Gustafsson, J.-Å. Interaction of Estrogenic Chemicals and Phytoestrogens with Estrogen Receptor β. Endocrinology 1998, 139, 4252–4263. [Google Scholar] [CrossRef]
- Li, L.; Wang, Q.; Zhang, Y.; Niu, Y.; Yao, X.; Liu, H. The Molecular Mechanism of Bisphenol A (BPA) as an Endocrine Disruptor by Interacting with Nuclear Receptors: Insights from Molecular Dynamics (MD) Simulations. PLoS ONE 2015, 10, e0120330. [Google Scholar] [CrossRef]
- Amjad, S.; Rahman, S.; Pang, M.-G. Role of Antioxidants in Alleviating Bisphenol A Toxicity. Biomolecules 2020, 10, 1105. [Google Scholar] [CrossRef]
- Dong, S.; Terasaka, S.; Kiyama, R. Bisphenol A induces a rapid activation of Erk1/2 through GPR30 in human breast cancer cells. Environ. Pollut. 2011, 159, 212–218. [Google Scholar] [CrossRef]
- Wozniak, A.L.; Bulayeva, N.N.; Watson, C.S. Xenoestrogens at Picomolar to Nanomolar Concentrations Trigger Membrane Estrogen Receptor-α–Mediated Ca2+ Fluxes and Prolactin Release in GH3/B6 Pituitary Tumor Cells. Environ. Health Perspect. 2005, 113, 431–439. [Google Scholar] [CrossRef]
- Matsushima, A.; Kakuta, Y.; Teramoto, T.; Koshiba, T.; Liu, X.; Okada, H.; Tokunaga, T.; Kawabata, S.; Kimura, M.; Shimohigashi, Y. Structural Evidence for Endocrine Disruptor Bisphenol A Binding to Human Nuclear Receptor ERR. J. Biochem. 2007, 142, 517–524. [Google Scholar] [CrossRef]
- Okada, H.; Tokunaga, T.; Liu, X.; Takayanagi, S.; Matsushima, A.; Shimohigashi, Y. Direct Evidence Revealing Structural Elements Essential for the High Binding Ability of Bisphenol A to Human Estrogen-Related Receptor-γ. Environ. Health Perspect. 2008, 116, 32–38. [Google Scholar] [CrossRef]
- Richter, C.A.; Birnbaum, L.S.; Farabollini, F.; Newbold, R.R.; Rubin, B.S.; Talsness, C.E.; Vandenbergh, J.G.; Walser-Kuntz, D.R.; Saal, F.S.V. In vivo effects of bisphenol A in laboratory rodent studies. Reprod. Toxicol. 2007, 24, 199–224. [Google Scholar] [CrossRef] [PubMed]
- European Union. Commission Regulation (EU) No 10/2011 of 14 January 2011; Official Journal of the European Union: Brussels, Belgium, 2011; pp. 1–89.
- European Commission. Commission Regulation (EU) 2018/832 of 12 February 2018 on the Use of Bisphenol A in Varnishes and Coatings Intended to Come into Contact with Food and Amending Regulation (EU) No 10/2011 as Regards the Use of That Substance in Plastic Food Contact Materials; Official Journal of the European Union: Brussels, Belgium, 2018; p. 7.
- National Toxicology Program—NTP. NTP Research Report on the CLARITY-BPA Core Study: A Perinatal and Chronic Extended-Dose-Range Study of Bisphenol A in Rats; National Toxicology Program: Research Triangle Park, NC, USA, 2018. [Google Scholar]
- Schug, T.T.; Heindel, J.J.; Camacho, L.; Delclos, K.B.; Howard, P.; Johnson, A.F.; Aungst, J.; Keefe, D.; Newbold, R.; Walker, N.J.; et al. A new approach to synergize academic and guideline-compliant research: The CLARITY-BPA research program. Reprod. Toxicol. 2013, 40, 35–40. [Google Scholar] [CrossRef]
- Camacho, L.; Lewis, S.; VanLandingham, M.; Olson, G.; Davis, K.; Patton, R.; Twaddle, N.; Doerge, D.; Churchwell, M.; Bryant, M.; et al. A two-year toxicology study of bisphenol A (BPA) in Sprague-Dawley rats: CLARITY-BPA core study results. Food Chem. Toxicol. 2019, 132, 110728. [Google Scholar] [CrossRef]
- Heindel, J.J.; Newbold, R.R.; Bucher, J.R.; Camacho, L.; Delclos, K.B.; Lewis, S.M.; VanLandingham, M.; Churchwell, M.I.; Twaddle, N.C.; McLellen, M.; et al. NIEHS/FDA CLARITY-BPA research program update. Reprod. Toxicol. 2015, 58, 33–44. [Google Scholar] [CrossRef]
- Dere, E.; Anderson, L.M.; Huse, S.M.; Spade, D.J.; McDonnell-Clark, E.; Madnick, S.J.; Hall, S.J.; Camacho, L.; Lewis, S.M.; VanLandingham, M.M.; et al. Effects of continuous bisphenol A exposure from early gestation on 90-day old rat testes function and sperm molecular profiles: A CLARITY-BPA consortium study. Toxicol. Appl. Pharmacol. 2018, 347, 1–9. [Google Scholar] [CrossRef]
- Morrissey, R.E.; George, J.D.; Price, C.J.; Tyl, R.W.; Marr, M.C.; Kimmel, C.A. The Developmental Toxicity of Bisphenol A in Rats and Mice. Toxicol. Sci. 1987, 8, 571–582. [Google Scholar] [CrossRef]
- Saunders, P.T.K.; Majdic, G.; Parte, P.; Millar, M.R.; Fisher, J.S.; Turner, K.J.; Sharpe, R.M. Fetal and Perinatal Influence of Xenoestrogens on Testis Gene Expression. Results Probl. Cell Differ. 1997, 424, 99–110. [Google Scholar] [CrossRef]
- Hart, R.J.; Doherty, D.A.; Keelan, J.A.; Minaee, N.S.; Thorstensen, E.B.; Dickinson, J.E.; Pennell, C.E.; Newnham, J.P.; McLachlan, R.; Norman, R.J.; et al. The impact of antenatal Bisphenol A exposure on male reproductive function at 20–22 years of age. Reprod. Biomed. Online 2018, 36, 340–347. [Google Scholar] [CrossRef]
- Vitku, J.; Chlupacova, T.; Sosvorova, L.; Hampl, R.; Hill, M.; Heracek, J.; Bicikova, M.; Starka, L. Development and validation of LC–MS/MS method for quantification of bisphenol A and estrogens in human plasma and seminal fluid. Talanta 2015, 140, 62–67. [Google Scholar] [CrossRef]
- Zhang, T.; Sun, H.; Kannan, K. Blood and Urinary Bisphenol A Concentrations in Children, Adults, and Pregnant Women from China: Partitioning between Blood and Urine and Maternal and Fetal Cord Blood. Environ. Sci. Technol. 2013, 47, 4686–4694. [Google Scholar] [CrossRef]
- Lan, H.-C.; Wu, K.-Y.; Lin, I.-W.; Yang, Z.-J.; Chang, A.-A.; Hu, M.-C. Bisphenol A disrupts steroidogenesis and induces a sex hormone imbalance through c-Jun phosphorylation in Leydig cells. Chemosphere 2017, 185, 237–246. [Google Scholar] [CrossRef]
- Akingbemi, B.T.; Sottas, C.M.; Koulova, A.I.; Klinefelter, G.R.; Hardy, M.P. Inhibition of Testicular Steroidogenesis by the Xenoestrogen Bisphenol A Is Associated with Reduced Pituitary Luteinizing Hormone Secretion and Decreased Steroidogenic Enzyme Gene Expression in Rat Leydig Cells. Endocrinology 2004, 145, 592–603. [Google Scholar] [CrossRef]
- Xi, W.; Lee, C.; Yeung, W.; Giesy, J.P.; Wong, M.; Zhang, X.; Hecker, M.; Wong, C.K. Effect of perinatal and postnatal bisphenol A exposure to the regulatory circuits at the hypothalamus–pituitary–gonadal axis of CD-1 mice. Reprod. Toxicol. 2011, 31, 409–417. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.; Chen, F.; Wang, X.; Bai, Y.; Zhou, R.; Li, Y.; Chen, L.; Bai, Y. Exposure of preimplantation embryos to low-dose bisphenol A impairs testes development and suppresses histone acetylation of StAR promoter to reduce production of testosterone in mice. Mol. Cell. Endocrinol. 2016, 427, 101–111. [Google Scholar] [CrossRef] [PubMed]
- Ben Maamar, M.; Lesné, L.; Desdoits-Lethimonier, C.; Coiffec, I.; Lassurguère, J.; Lavoué, V.; Deceuninck, Y.; Antignac, J.-P.; Le Bizec, B.; Perdu, E.; et al. An Investigation of the Endocrine-Disruptive Effects of Bisphenol A in Human and Rat Fetal Testes. PLoS ONE 2015, 10, e0117226. [Google Scholar] [CrossRef]
- Lv, Y.; Li, L.; Fang, Y.; Chen, P.; Wu, S.; Chen, X.; Ni, C.; Zhu, Q.; Huang, T.; Lian, Q.; et al. In utero exposure to bisphenol A disrupts fetal testis development in rats. Environ. Pollut. 2019, 246, 217–224. [Google Scholar] [CrossRef]
- Hanaoka, T.; Kawamura, N.; Hara, K.; Tsugane, S. Urinary bisphenol A and plasma hormone concentrations in male workers exposed to bisphenol A diglycidyl ether and mixed organic solvents. Occup. Environ. Med. 2002, 59, 625–628. [Google Scholar] [CrossRef] [PubMed]
- Meeker, J.D.; Yang, T.; Ye, X.; Calafat, A.M.; Hauser, R. Urinary Concentrations of Parabens and Serum Hormone Levels, Semen Quality Parameters, and Sperm DNA Damage. Environ. Health Perspect. 2011, 119, 252–257. [Google Scholar] [CrossRef]
- Mendiola, J.; Jørgensen, N.; Andersson, A.M.; Calafat, A.M.; Ye, X.; Redmon, J.B.; Drobnis, E.Z.; Wang, C.; Sparks, A.; Thurston, S.W.; et al. Are environmental levels of bisphenol A associated with reproductive function in fertile men? Environ. Health Perspect. 2010, 118, 1286–1291. [Google Scholar] [CrossRef] [PubMed]
- Galloway, T.; Cipelli, R.; Guralnik, J.; Ferrucci, L.; Bandinelli, S.; Corsi, A.M.; Money, C.; McCormack, P.; Melzer, D. Daily Bisphenol A Excretion and Associations with Sex Hormone Concentrations: Results from the InCHIANTI Adult Population Study. Environ. Health Perspect. 2010, 118, 1603–1608. [Google Scholar] [CrossRef]
- Lassen, T.H.; Frederiksen, H.; Jensen, T.K.; Petersen, J.H.; Joensen, U.N.; Main, K.M.; Skakkebaek, N.E.; Juul, A.; Jørgensen, N.; Andersson, A.-M. Urinary Bisphenol A Levels in Young Men: Association with Reproductive Hormones and Semen Quality. Environ. Health Perspect. 2014, 122, 478–484. [Google Scholar] [CrossRef]
- Santiago, J.; Patrício, D.; Silva, J.V. Testicular Signaling: Team Work in Sperm Production. In Tissue-Specific Cell Signaling; Silva, J.V., Freitas, M.J., Fardilha, M., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 225–255. ISBN 978-3-030-44435-8. [Google Scholar]
- Gualtieri, A.F.; Iwachow, M.A.; Venara, M.; Rey, R.A.; Schteingart, H.F. Bisphenol A effect on glutathione synthesis and recycling in testicular Sertoli cells. J. Endocrinol. Investig. 2011, 34, e102–e109. [Google Scholar] [CrossRef]
- Wang, C.; Qi, S.; Liu, C.; Yang, A.; Fu, W.; Quan, C.; Duan, P.; Yu, T.; Yang, K. Mitochondrial Dysfunction and Ca2+ Overload in Injured Sertoli Cells Exposed to Bisphenol A. Environ. Toxicol. 2016, 32, 823–831. [Google Scholar] [CrossRef] [PubMed]
- Salian, S.; Doshi, T.; Vanage, G. Neonatal exposure of male rats to Bisphenol A impairs fertility and expression of sertoli cell junctional proteins in the testis. Toxicology 2009, 265, 56–67. [Google Scholar] [CrossRef] [PubMed]
- De Freitas, A.T.A.G.; Ribeiro, M.A.; Pinho, C.F.; Peixoto, A.R.; Domeniconi, R.F.; Scarano, W.R. Regulatory and junctional proteins of the blood-testis barrier in human Sertoli cells are modified by monobutyl phthalate (MBP) and bisphenol A (BPA) exposure. Toxicol. In Vitro 2016, 34, 1–7. [Google Scholar] [CrossRef]
- Akarca-Dizakar, S.Ö.; Erdoğan, D.; Peker, T.; Coşkun Akçay, N.; Türkoğlu, I.; Eşmekaya, M.A.; Ömeroğlu, S. Effects of co-administered melatonin, fructose and bisphenol A (BPA) on rat epididymis and sperm characteristics. Biotech. Histochem. 2020, 95, 18–26. [Google Scholar] [CrossRef]
- Olukole, S.G.; Lanipekun, D.O.; Ola-Davies, E.O.; Oke, B.O. Maternal exposure to environmentally relevant doses of bisphenol A causes reproductive dysfunction in F1 adult male rats: Protective role of melatonin. Environ. Sci. Pollut. Res. 2019, 26, 28940–28950. [Google Scholar] [CrossRef]
- Jiang, Z.; Wang, J.; Li, X.; Zhang, X. Echinacoside and Cistanche tubulosa (Schenk) R. wight ameliorate bisphenol A-induced testicular and sperm damage in rats through gonad axis regulated steroidogenic enzymes. J. Ethnopharmacol. 2016, 193, 321–328. [Google Scholar] [CrossRef]
- Alboghobeish, S.; Mahdavinia, M.; Zeidooni, L.; Samimi, A.; Oroojan, A.A.; Alizadeh, S.; Dehghani, M.A.; Ahangarpour, A.; Khorsandi, L. Efficiency of naringin against reproductive toxicity and testicular damages induced by bisphenol A in rats. Iran. J. Basic Med. Sci. 2019, 22, 315–523. [Google Scholar] [CrossRef]
- Jahan, S.; Ain, Q.U.; Ullah, H. Therapeutic effects of quercetin against bisphenol A induced testicular damage in male Sprague Dawley rats. Syst. Biol. Reprod. Med. 2016, 62, 114–124. [Google Scholar] [CrossRef]
- Kaur, S.; Sadwal, S. Studies on the phytomodulatory potential of fenugreek (Trigonella foenum-graecum) on bisphenol-A induced testicular damage in mice. Andrology 2019, 52, e13492. [Google Scholar] [CrossRef]
- Liu, X.; Wang, Z.; Liu, F. Chronic exposure of BPA impairs male germ cell proliferation and induces lower sperm quality in male mice. Chemosphere 2021, 262, 127880. [Google Scholar] [CrossRef]
- Salian, S.; Doshi, T.; Vanage, G. Perinatal exposure of rats to Bisphenol A affects the fertility of male offspring. Life Sci. 2009, 85, 742–752. [Google Scholar] [CrossRef]
- Al-Hiyasat, A.S.; Darmani, H.; Elbetieha, A.M. Effects of bisphenol A on adult male mouse fertility. Eur. J. Oral Sci. 2002, 110, 163–167. [Google Scholar] [CrossRef] [PubMed]
- Wisniewski, P.; Romano, R.M.; Kizys, M.M.; Oliveira, K.C.; Kasamatsu, T.; Giannocco, G.; Chiamolera, M.I.; Dias-Da-Silva, M.R.; Romano, M.A. Adult exposure to bisphenol A (BPA) in Wistar rats reduces sperm quality with disruption of the hypothalamic–pituitary–testicular axis. Toxicology 2015, 329, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Dobrzyńska, M.M.; Radzikowska, J. Genotoxicity and reproductive toxicity of bisphenol A and X-ray/bisphenol A combination in male mice. Drug Chem. Toxicol. 2011, 36, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Tainaka, H.; Takahashi, H.; Umezawa, M.; Tanaka, H.; Nishimune, Y.; Oshio, S.; Takeda, K. Evaluation of the testicular toxicity of prenatal exposure to bisphenol A based on microarray analysis combined with MeSH annotation. J. Toxicol. Sci. 2012, 37, 539–548. [Google Scholar] [CrossRef]
- Tiwari, D.; Vanage, G. Mutagenic effect of Bisphenol A on adult rat male germ cells and their fertility. Reprod. Toxicol. 2013, 40, 60–68. [Google Scholar] [CrossRef]
- Qiu, L.-L.; Wang, X.; Zhang, X.-H.; Zhang, Z.; Gu, J.; Liu, L.; Wang, Y.; Wang, X.; Wang, S.-L. Decreased androgen receptor expression may contribute to spermatogenesis failure in rats exposed to low concentration of bisphenol A. Toxicol. Lett. 2013, 219, 116–124. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Luo, C.; Li, Q.; Chen, S.; Hu, Y. Mitochondrion-mediated apoptosis is involved in reproductive damage caused by BPA in male rats. Environ. Toxicol. Pharmacol. 2014, 38, 1025–1033. [Google Scholar] [CrossRef] [PubMed]
- Li, D.-K.; Zhou, Z.; Miao, M.; He, Y.; Wang, J.; Ferber, J.; Herrinton, L.J.; Gao, E.; Yuan, W. Urine bisphenol-A (BPA) level in relation to semen quality. Fertil. Steril. 2011, 95, 625–630.e4. [Google Scholar] [CrossRef] [PubMed]
- Minamiyama, Y.; Ichikawa, H.; Takemura, S.; Kusunoki, H.; Naito, Y.; Yoshikawa, T. Generation of reactive oxygen species in sperms of rats as an earlier marker for evaluating the toxicity of endocrine-disrupting chemicals. Free Radic. Res. 2010, 44, 1398–1406. [Google Scholar] [CrossRef]
- D’Cruz, S.C.; Jubendradass, R.; Jayakanthan, M.; Rani, S.J.A.; Mathur, P.P. Bisphenol A impairs insulin signaling and glucose homeostasis and decreases steroidogenesis in rat testis: An in vivo and in silico study. Food Chem. Toxicol. 2012, 50, 1124–1133. [Google Scholar] [CrossRef]
- Liu, C.; Duan, W.; Li, R.; Xu, S.; Zhang, L.; Chen, C.; He, M.; Lu, Y.; Wu, H.; Pi, H.; et al. Exposure to bisphenol A disrupts meiotic progression during spermatogenesis in adult rats through estrogen-like activity. Cell Death Dis. 2013, 4, e676. [Google Scholar] [CrossRef]
- Pan, D.; Feng, D.; Ding, H.; Zheng, X.; Ma, Z.; Yang, B.; Xie, M. Effects of bisphenol A exposure on DNA integrity and protamination of mouse spermatozoa. Andrology 2020, 8, 486–496. [Google Scholar] [CrossRef]
- Chitra, K.C.; Latchoumycandane, C.; Mathur, P.P. Induction of oxidative stress by bisphenol A in the epididymal sperm of rats. Toxicology 2003, 185, 119–127. [Google Scholar] [CrossRef]
- Barbonetti, A.; Castellini, C.; Di Giammarco, N.; Santilli, G.; Francavilla, S.; Francavilla, F. In vitro exposure of human spermatozoa to bisphenol A induces pro-oxidative/apoptotic mitochondrial dysfunction. Reprod. Toxicol. 2016, 66, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Eladak, S.; Moison, D.; Guerquin, M.-J.; Matilionyte, G.; Kilcoyne, K.; N’Tumba-Byn, T.; Messiaen, S.; Deceuninck, Y.; Pozzi-Gaudin, S.; Benachi, A.; et al. Effects of environmental Bisphenol A exposures on germ cell development and Leydig cell function in the human fetal testis. PLoS ONE 2018, 13, e0191934. [Google Scholar] [CrossRef]
- Meeker, J.D.; Ehrlich, S.; Toth, T.L.; Wright, D.L.; Calafat, A.M.; Trisini, A.T.; Ye, X.; Hauser, R. Semen quality and sperm DNA damage in relation to urinary bisphenol A among men from an infertility clinic. Reprod. Toxicol. 2010, 30, 532–539. [Google Scholar] [CrossRef] [PubMed]
- Jensen, T.K.; Andersson, A.M.; Jørgensen, N.; Andersen, A.G.; Carlsen, E.; Skakkebæk, N.E. Body mass index in relation to semen quality and reproductive hormonesamong 1,558 Danish men. Fertil. Steril. 2004, 82, 863–870. [Google Scholar] [CrossRef]
- Simioni, C.; Zauli, G.; Martelli, A.M.; Vitale, M.; Sacchetti, G.; Gonelli, A.; Neri, L.M. Oxidative stress: Role of physical exercise and antioxidant nutraceuticals in adulthood and aging. Oncotarget 2018, 9, 17181–17198. [Google Scholar] [CrossRef] [PubMed]
- Powers, S.K.; Jackson, M.J. Exercise-Induced Oxidative Stress: Cellular Mechanisms and Impact on Muscle Force Production. Physiol. Rev. 2008, 88, 1243–1276. [Google Scholar] [CrossRef]
- Brigelius-Flohé, R. Glutathione peroxidases and redox-regulated transcription factors. Biol. Chem. 2006, 387, 1329–1335. [Google Scholar] [CrossRef]
- Valko, M.; Leibfritz, D.; Moncol, J.; Cronin, M.T.D.; Mazur, M.; Telser, J. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol. 2007, 39, 44–84. [Google Scholar] [CrossRef]
- Powell, S.R. The Antioxidant Properties of Zinc. J. Nutr. 2000, 130, 1447S–1454S. [Google Scholar] [CrossRef]
- Prasad, A.S. Zinc is an Antioxidant and Anti-Inflammatory Agent: Its Role in Human Health. Front. Nutr. 2014, 1, 14. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.A.; Fischer, J.G.; Kays, S.E. Is copper an antioxidant nutrient? Crit. Rev. Food Sci. Nutr. 1992, 32, 1–31. [Google Scholar] [CrossRef]
- Sahu, C.; Charaya, A.; Singla, S.; Dwivedi, D.K.; Jena, G. Zinc deficient diet increases the toxicity of bisphenol A in rat testis. J. Biochem. Mol. Toxicol. 2020, 34. [Google Scholar] [CrossRef]
- Rhee, S.G. Overview on Peroxiredoxin. Mol. Cells 2016, 39, 1–5. [Google Scholar] [CrossRef]
- Masutani, H.; Ueda, S.; Yodoi, J. The thioredoxin system in retroviral infection and apoptosis. Cell Death Differ. 2005, 12, 991–998. [Google Scholar] [CrossRef]
- Sabeti, P.; Pourmasumi, S.; Rahiminia, T.; Akyash, F.; Talebi, A.R. Etiologies of sperm oxidative stress. Int. J. Reprod. Biomed. 2016, 14, 231–240. [Google Scholar] [CrossRef]
- De Lamirande, E.; Tsai, C.; Harakat, A.; Gagnon, C. Involvement of reactive oxygen species in human sperm arcosome reaction induced by A23187, lysophosphatidylcholine, and biological fluid ultrafiltrates. J. Androl. 1998, 19, 585–594. [Google Scholar]
- De Lamirande, E.; Gagnon, C. A positive role for the superoxide anion in triggering hyperactivation and capacitation of human spermatozoa. Int. J. Androl. 1993, 16, 21–25. [Google Scholar] [CrossRef] [PubMed]
- Paoli, D.; Pecora, G.; Pallotti, F.; Faja, F.; Pelloni, M.; Lenzi, A.; Lombardo, F. Cytological and molecular aspects of the ageing sperm. Hum. Reprod. 2019, 34, 218–227. [Google Scholar] [CrossRef] [PubMed]
- Sakkas, D.; Alvarez, J.G. Sperm DNA fragmentation: Mechanisms of origin, impact on reproductive outcome, and analysis. Fertil. Steril. 2010, 93, 1027–1036. [Google Scholar] [CrossRef]
- Wright, C.; Milne, S.; Leeson, H. Sperm DNA damage caused by oxidative stress: Modifiable clinical, lifestyle and nutritional factors in male infertility. Reprod. Biomed. Online 2014, 28, 684–703. [Google Scholar] [CrossRef]
- Aitken, R.J.; De Iuliis, G.N.; Finnie, J.M.; Hedges, A.; McLachlan, R.I. Analysis of the relationships between oxidative stress, DNA damage and sperm vitality in a patient population: Development of diagnostic criteria. Hum. Reprod. 2010, 25, 2415–2426. [Google Scholar] [CrossRef]
- Rahman, S.; Kwon, W.-S.; Lee, J.-S.; Yoon, S.-J.; Ryu, B.Y.; Pang, M.-G. Bisphenol-A Affects Male Fertility via Fertility-related Proteins in Spermatozoa. Sci. Rep. 2015, 5, srep09169. [Google Scholar] [CrossRef] [PubMed]
- Ooe, H.; Taira, T.; Iguchi-Ariga, S.M.M.; Ariga, H. Induction of Reactive Oxygen Species by Bisphenol A and Abrogation of Bisphenol A-Induced Cell Injury by DJ-1. Toxicol. Sci. 2005, 88, 114–126. [Google Scholar] [CrossRef]
- Rahman, S.; Kang, K.-H.; Arifuzzaman, S.; Pang, W.-K.; Ryu, D.-Y.; Song, W.-H.; Park, Y.-J.; Pang, M.-G. Effect of antioxidants on BPA-induced stress on sperm function in a mouse model. Sci. Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Rezaee-Tazangi, F.; Zeidooni, L.; Rafiee, Z.; Fakhredini, F.; Kalantari, H.; Alidadi, H.; Khorsandi, L. Taurine effects on Bisphenol A-induced oxidative stress in the mouse testicular mitochondria and sperm motility. JBRA Assist. Reprod. 2020, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Park, B.; Kwon, J.E.; Cho, S.M.; Kim, C.W.; Lee, D.E.; Koo, Y.T.; Lee, S.H.; Lee, H.M.; Kang, S.C. Protective effect of Lespedeza cuneata ethanol extract on Bisphenol A-induced testicular dysfunction in vivo and in vitro. Biomed. Pharmacother. 2018, 102, 76–85. [Google Scholar] [CrossRef]
- Tamilselvan, P.; Bharathiraja, K.; Vijayaprakash, S.; Balasubramanian, M.P. Protective role of lycopene on bisphenol A induced changes in sperm characteristics, testicular damage and oxidative stress in rats. Int. J. Pharm. Biol. Sci. 2013, 4, 131–143. [Google Scholar]
- Yin, L.; Dai, Y.; Cui, Z.; Jiang, X.; Liu, W.; Han, F.; Lin, A.; Cao, J.; Liu, J. The regulation of cellular apoptosis by the ROS-triggered PERK/EIF2α/chop pathway plays a vital role in bisphenol A-induced male reproductive toxicity. Toxicol. Appl. Pharmacol. 2017, 314, 98–108. [Google Scholar] [CrossRef]
- Gules, O.; Yildiz, M.; Naseer, Z.; Tatar, M. Effects of folic acid on testicular toxicity induced by bisphenol-A in male Wistar rats. Biotech. Histochem. 2018, 94, 26–35. [Google Scholar] [CrossRef]
- Aikawa, H.; Koyama, S.; Matsuda, M.; Nakahashi, K.; Akazome, Y.; Mori, T. Relief effect of vitamin A on the decreased motility of sperm and the increased incidence of malformed sperm in mice exposed neonatally to bisphenol A. Cell Tissue Res. 2004, 315, 119–124. [Google Scholar] [CrossRef]
- Huang, F.; Ning, H.; Xin, Q.-Q.; Huang, Y.; Wang, H.; Zhang, Z.-H.; Xu, D.-X.; Ichihara, G.; Ye, D.-Q. Melatonin pretreatment attenuates 2-bromopropane-induced testicular toxicity in rats. Toxicology 2009, 256, 75–82. [Google Scholar] [CrossRef]
- Kotler, M.; Rodríguez, C.; Sáinz, R.M.; Antolin, I.; Menéndez-Peláez, A. Melatonin increases gene expression for antioxidant enzymes in rat brain cortex. J. Pineal Res. 1998, 24, 83–89. [Google Scholar] [CrossRef]
- Reiter, R.J. Oxidative damage in the central nervous system: Protection by melatonin. Prog. Neurobiol. 1998, 56, 359–384. [Google Scholar] [CrossRef]
- Cui, J.D. Biotechnological production and applications of Cordyceps militaris, a valued traditional Chinese medicine. Crit. Rev. Biotechnol. 2014, 35, 475–484. [Google Scholar] [CrossRef]
- Hu, C.; Kitts, D.D. Studies on the antioxidant activity of Echinacea root extract. J. Agric. Food Chem. 2000, 48, 1466–1472. [Google Scholar] [CrossRef]
- Rivoira, M.A.; Rodriguez, V.; Talamoni, G.; De Talamoni, N.T. New perspectives in the pharmacological potential of naringin in medicine. Curr. Med. Chem. 2020, 27, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Apaydin, F.G.; Aslanturk, A.; Uzunhisarcikli, M.; Bas, H.; Kalender, S.; Kalender, Y. Histopathological and biochemical studies on the effect of curcumin and taurine against bisphenol A toxicity in male rats. Environ. Sci. Pollut. Res. 2019, 26, 12302–12310. [Google Scholar] [CrossRef] [PubMed]
- Khalaf, A.; Ahmed, W.; Moselhy, W.A.; Abdel-Halim, B.R.; Ibrahim, M.A. Protective effects of selenium and nano-selenium on bisphenol-induced reproductive toxicity in male rats. Hum. Exp. Toxicol. 2018, 38, 398–408. [Google Scholar] [CrossRef] [PubMed]

| Reference | Animal | Antioxidant | Experimental Groups (G) | Effects of BPA + Antioxidant Administration |
|---|---|---|---|---|
| [21] | Swiss albino mice (in vivo) | Melatonin (hormone) | G1: 0.2 mL olive oil (control); G2: 10 mg/kg BPA suspended in olive oil; G3: 10 mg/kg melatonin; G4: BPA 10 mg/kg + 10 mg/kg of melatonin—dose/day for 14 days | ↑ Mitochondrial marker enzymes SDH, MDH, IDH, NDH, MAO, GSH, antioxidant enzymes GPx, SOD, GR ↓ LPO |
| [25] | Sprague Dawley rats (in vivo) | Melatonin (hormone) | G1: 0.5% ethanol in normal saline (control); G2: 200 mg/kg BPA suspended in olive oil; G3: 10 mg/kg melatonin intraperitoneally 30 min before BPA administration; G4: 10 mg/kg melatonin intraperitoneally + 200 mg/kg BPA suspended in olive oil—dose/day for 10 days | ↔ body weight, reproductive organs weight, testes/body and epididymis/body weight ratios, sperm counts and apoptosis ↑ SOD activity and 4C-cells number ↓ TBARS accumulation and DNA damage in spermatocytes, number of γH2AX-positive foci |
| [26] | Sprague Dawley rats (in vivo) | Melatonin (hormone) | G1: no treatment (normal control); G2: 0.2 mL corn oil (experimental control); G3: normal saline (experimental control); G4: 50 mg/kg BPA suspended in corn oil; G5: 10 mg/kg melatonin in normal saline; G6: 10 mg/kg melatonin + 50 mg/kg BPA—3 days/week for 6 weeks | ↑ sperm count and motility, testosterone levels, GSH, viable cells ↓ mortality and abnormal sperm, % diploid sperm and spermatid; levels of H2O2 and MDA, necrotic and apoptotic cells Other alterations: seminiferous tubules showed increase in the germinal cell population with active spermatogenesis and normal arrangement of spermatogenic cell, Leydig cells population normal |
| [85] | Sprague Dawley rats (in vivo) | Melatonin (hormone) | G1: 25 mg/kg sesame oil + 25 mg/kg 0.1% ethanol (control); G2: 25 mg/kg BPA; G3: 25 mg/kg BPA + 20 mg/kg melatonin—dose/day for 60 days | ↔ total sperm counts ↑ Cldn-1, Occ and ZO-1 immunostaining, sperm motility Other alterations: Fewer vacuolations, irregular tubules and degenerative cells containing a heterochromatic nucleus in epididymis |
| [86] | Wistar albino rats (in vivo) | Melatonin (hormone) | G1: 0.2 mL 1% dimethyl sulfoxide (DMSO)/99% canola oil (control); G2: 0.025 mg/kg BPA; G3: 0.25 mg/kg BPA; G4: 0.025 mg/kg BPA + melatonin 1 mg/kg; G5: 0.25 mg/kg BPA + melatonin 1 mg/kg—dose/day; exposure in utero from gestational day 10–21 | ↑ body weight; gonosomatic index; sperm motility; viability and count; serum T levels and LH; activity of SOD, GSH, GPx, and GST; tubular and luminal diameter ↓ FSH and E2; testicular MDA and H2O2 levels, interstitial necrosis, and germinal cell degeneration |
| [134] | Wistar albino rats (in vivo) | Folic acid (vitamin B9) | G1: 0.5 mL 0.9% NaCl (control); G2: 50 mg/kg BPA in 0.5 mL corn oil; G3: 20 mg/kg/day folic acid in 0.5 mL 0.9% NaCl; G4: 20 mg/kg folic acid in 0.5 mL 0.9% NaCl + 50 mg/kg BPA in 0.5 mL corn oil—dose/day for 14 days | ↔ body weight, testes/body weight ratios, number of UTF-1 positive cells/tubule and UTF-1 positive tubules ↑ serum testosterone levels, viable sperm ↓ TUNEL positive cells and tubules, head, midpiece and total sperm abnormalities |
| [27] | Wistar albino rats (in vivo) | Vitamin C | G1: olive oil (control); G2: 25 mg/kg/day BPA; G3: 25 mg/kg/day BPA + 60 mg/kg/day of vitamin C three times a week—50 days | ↑ right epididymal weight, congestion areas, atrophy, germinal cell debris ↓ GSH |
| [129] | CD-1 (ICR) mice (in vitro) | Vitamin C, Vitamin E and GSH | Condition I: DMSO (control); Cond II: 100 µM BPA; Cond III: 100 µM BPA + 5 mM GSH; Cond IV: 100 µM BPA + 100 µM Vitamin C; Cond III: 100 µM BPA + 2 mM of Vitamin E—for 6 h | ↑ sperm motility, ATP levels ↓ acrosome-reacted spermatozoa, PKA activity, protein tyrosine phosphorylation and nitration, ROS levels |
| [135] | SHN mice (in vivo) | Vitamin A | G1: 16 mL of sesame oil and 4 mL of dimethyl sulfoxide (control); G2: 0.5 mg BPA; G3: 50 mg BPA; G4: 50 mg BPA + 100 IU Retinoic Acid—for 5 days from the date of birth | ↑ sperm motility ↓ abnormal sperm |
| [31] | Human (in vitro) | Eruca Sativa aqueous extract | Condition I: untreated (control); Cond II: 10 µM BPA; Cond III: 10 µM BPA + 15.5 µg/mL ESAE; Cond IV: 10 µM BPA + 62.55 µg/mL ESAE; Cond V: 10 µM BPA + 250 µg/mL ESAE; Cond VI: 10 µM BPA + 1000 µg/mL ESAE—ESAE incubation for 1 h followed by BPA incubation for 4 h | ↑ sperm progressive motility and viability, mitochondrial function ↓ immotile sperm |
| [32] | Wistar albino rats (in vivo) | Eruca Sativa aqueous extract | G1: 0.4 mL/kg/day of tocopherol-stripped corn oil (control); G2: 100 mg/kg BPA; G3: 200 mg/kg ESAE; G4: 100 mg/kg BPA + 50 mg/kg ESAE; G5: 100 mg/kg BPA + 100 mg/kg ESAE; G6: 100 mg/kg BPA + 200 mg/kg ESAE—dose/day for 30 days | ↑ body weight, reproductive organs weight, testosterone, and LH levels, sperm counts, motility, viability, SH group content ↓ morphologically abnormal sperm; MDA levels; SOD, CAT and GPx activities |
| [30] | Sprague Dawley rats (in vivo) | Cordyceps militaris | G1: no intervention (normal control); G2: 200 mg/kg BPA; G3: 800 mg/kg C.militaris; G4: 200 mg/kg BPA + 200 mg/kg C. militaris; G5: 200 mg/kg BPA + 400 mg/kg C. militaris; G6: 200 mg/kg BPA + 800 mg/kg C. militaris; G7: 200 mg/kg BPA + 300 mg/kg Vitamin E—28 days | ↑ body weight; SOD, GPx, GSH, testosterone, and LH serum levels; sperm counts and motility; mRNA levels of Star; CYP11A1; 3β-HSD; and CYP17A1 ↓ MDA levels |
| [87] | Sprague Dawley rats (in vivo) | Cistanche tubulosa and Echinacoside (ECH) | G1: corn oil 10 mL/kg (normal control); G2: 200 mg/kg BPA; G3: 200 mg/kg BPA + 300 mg/kg Vitamin E; G4: 200 mg/kg BPA + 6 mg/kg ECH; G5: 200 mg/kg BPA + 200 mg/kg CT; G6: 6 mg/kg EC; G7: 200 mg/kg CT—6 weeks | ↑ sperm motility; LDH-x activity; FSH, LH, and testosterone serum levels; mRNA levels of StAR, CYP17A1, 3β-HSD, and 17β-HSD; protein levels of CYP11A1 and CYP17A1 ↓ abnormal sperm Other alterations: normal histological pattern, normal spermatogenic series |
| [88] | Wistar albino rats (in vivo) | Naringin (flavonoid) | G1: Control; G2: 50 mg/kg BPA; G3: 50 mg/kg BPA + 40 mg/kg naringin; G4: 50 mg/kg BPA + 80 mg/kg naringin; G5: 50 mg/kg BPA + 160 mg/kg naringin; G6: 160 mg/kg Naringin—for 30 days | ↔ body weight ↑ testicular weight and volume; total testicular protein; epididymal sperm count; testicular enzymes (ALP, LDH); serum FSH; LH; testosterone and E2; activities of GPx, SOD, and CAT; GSH ↓ MDA, ROS Other: less testicular tissue damage |
| [89] | Sprague Dawley rats (in vivo) | Quercetin (flavonoid) | G1: normal saline (control); G2: 50 mg/kg BPA; G3: 50 mg/kg quercetin; G4: 50 mg/kg BPA + 50 mg/kg quercetin—for 52 days | ↔ body weight, reproductive organ weight ↑ plasma testosterone, LDL and HDL levels, tunica albuginea thickness, seminiferous tubule area, number of spermatogonia, primary spermatocytes, secondary spermatocytes, and spermatids ↓ oestrogen levels, blood urea nitrogen levels, creatinine, cholesterol, triglyceride levels |
| [90] | Balb/c mice (in vivo) | Trigonella foenum-graecum | G1: normal pellet diet (control); G2: 200 mg/kg fenugreek seeds aqueous extract; G3: 1 mg/kg BPA; G4: 1 mg/kg BPA + 200 mg/kg fenugreek seeds aqueous extract—2 months | ↑ testis weight, sperm concentration, sperm motility, GSH, GPx activity, Bcl-2 mRNA levels ↓ ROS and LPO, Caspase-9 and -3 mRNA level Other alterations: improved histoarchitecture, basement membrane preservation with less vacuolization and increased number of elongated, round spermatids |
| [131] | CD-1 (ICR) mice (in vivo) | Lespedeza cuneata ethanol extract (LCE) | G1: normal saline (solvent control); G2:10 mg/kg BPA; G3: 10 mg/kg BPA + 100mg/kg Saw Palmetto extract (SPE); G4: 10 mg/kg BPA + 25 mg/kg LCE; G5: 10 mg/kg BPA + 50 mg/kg LCE; G6: 10 mg/kg BPA + 100 mg/kg LCE—for 12 weeks | ↑ testis weight; sperm counts and motility; testosterone levels; GSH, CAT, and SOD1 levels; HDL-cholesterol ↓ sperm abnormalities; TBARS levels; glucose; TC, TG, and LDL- cholesterol |
| [132] | Sprague Dawley rats (in vivo) | Lycopene (carotenoid) | G1: saline following treatment with 0.5 mL corn oi (control); G2: 200 mg/kg BPA; G3: 200 mg/kg BPA + 10 mg/kg lycopene; G4: 10 mg/kg lycopene—for 30 days | ↑ body and organ weight, sperm count, sperm motility, antioxidants enzymes level (SOD, CAT, GPx, GR) ↓ LPO and H2O2 |
| [130] | NMRI mice (in vitro) | Taurine (amino acid) | Condition I: untreated (control); Cond II: 0.8 mmol/L BPA for 2 h; Cond III: 50 µmol/ L TAU for 4 h; Cond IV: pre-treated with 5 µmol/L of TAU for 2 h before BPA treatment (2 h); Cond V: pre-treated with 10 µmol/L of TAU for 2 h before BPA treatment (2 h); Cond VI: pre-treated with 30 µmol/L of TAU for 2 h before BPA treatment (2 h); Cond VII: pre-treated with 50 µmol/L of TAU for 2 h before BPA treatment (2 h) | ↑ Sperm and testicular mitochondria viability, MMP, GSH, SOD, sperm motility ↓ testicular mitochondrial ROS, MDA |
| [22] | BALB/c mice (in vivo) | Selenium | G1: diet adequate in selenium (0.2 ppm/kg diet) as sodium selenite for 12 weeks (control); G2: 0.5 ppm sodium selenite/kg for 12 weeks; G3: 0.2 ppm sodium selenite/kg for 8 weeks followed by 1 mg/kg BPA for 4 weeks; G4: 0.5 ppm sodium selenite/kg for 8 weeks followed by 1 mg/kg BPA for 4 weeks | ↑ sperm concentration and motility, GPx activity ↓ ROS and LPO levels, number of TUNEL- positive germ cells Other alterations: preserved basement membrane with less vacuolization, increased germ cell count |
| [28] | Gobiocypris rarus (in vivo) | NAC | G1: 0.001%DMSO (control); G2: 10 mg/kg NAC; G3: 100 mg/kg NAC; G4: 225 μg/L BPA; G5: 10 mg/kg NAC + 225 μg/L BPA; G6: 100 mg/kg NAC + 225 μg/L BPA — for 7 days | ↑ GPx activity ↓ levels of 5-methylcytosine (5mC), GSH, γ-glutamyl cysteine synthetase (GCS), DNA methyltransferase proteins (DNMTs), H2O2 concentration, S-adenosylhomocysteine (SAH), homocysteine (HCY), nicotinamide adenine dinucleotide phosphate (NADPH) levels, SOD, CAT activities |
| [101] | Wistar albino rats (in vivo) | NAC | 0, 1.0 or 10 mg/L BPA for 8 weeks and BPA + 0.45% NAC for 2 days prior to the administration of BPA | ↑ sperm motility ↓ HNE-modified protein at 30 kDa, ROS levels |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santiago, J.; Silva, J.V.; Santos, M.A.S.; Fardilha, M. Fighting Bisphenol A-Induced Male Infertility: The Power of Antioxidants. Antioxidants 2021, 10, 289. https://doi.org/10.3390/antiox10020289
Santiago J, Silva JV, Santos MAS, Fardilha M. Fighting Bisphenol A-Induced Male Infertility: The Power of Antioxidants. Antioxidants. 2021; 10(2):289. https://doi.org/10.3390/antiox10020289
Chicago/Turabian StyleSantiago, Joana, Joana V. Silva, Manuel A. S. Santos, and Margarida Fardilha. 2021. "Fighting Bisphenol A-Induced Male Infertility: The Power of Antioxidants" Antioxidants 10, no. 2: 289. https://doi.org/10.3390/antiox10020289
APA StyleSantiago, J., Silva, J. V., Santos, M. A. S., & Fardilha, M. (2021). Fighting Bisphenol A-Induced Male Infertility: The Power of Antioxidants. Antioxidants, 10(2), 289. https://doi.org/10.3390/antiox10020289








