Comparison of Anti-Oxidative Effect of Human Adipose- and Amniotic Membrane-Derived Mesenchymal Stem Cell Conditioned Medium on Mouse Preimplantation Embryo Development
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
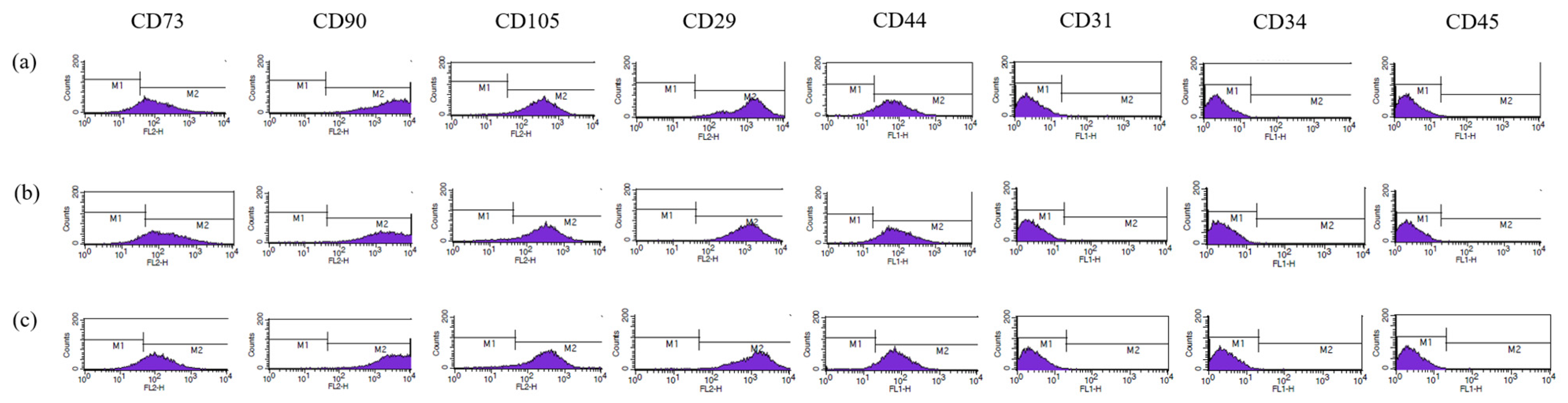
2.2. Culture and Characterization of ASCs and AMSCs
2.3. Preparation of ASC-CM and AMSC-CM
2.4. Experimental Animals
2.5. In Vitro Fertilization and Culture
2.6. Experimental Design
2.7. Quantitative Reverse Transcription-Polymerase Chain Reaction (qRT-PCR)
2.8. Intracellular ROS and Glutathione (GSH) Detection
2.9. Antioxidant Capacity and Enzyme Activity Assays
2.10. Mitochondrial Membrane Potential Assay
2.11. Statistical Analysis
3. Results
3.1. Characterization of AMSC and ASC
3.2. Effects of Various Concentrations of AMSC-CM on Embryo Development
3.3. Comparison of the Effects of ASC-CM and AMSC-CM on Embryo Development
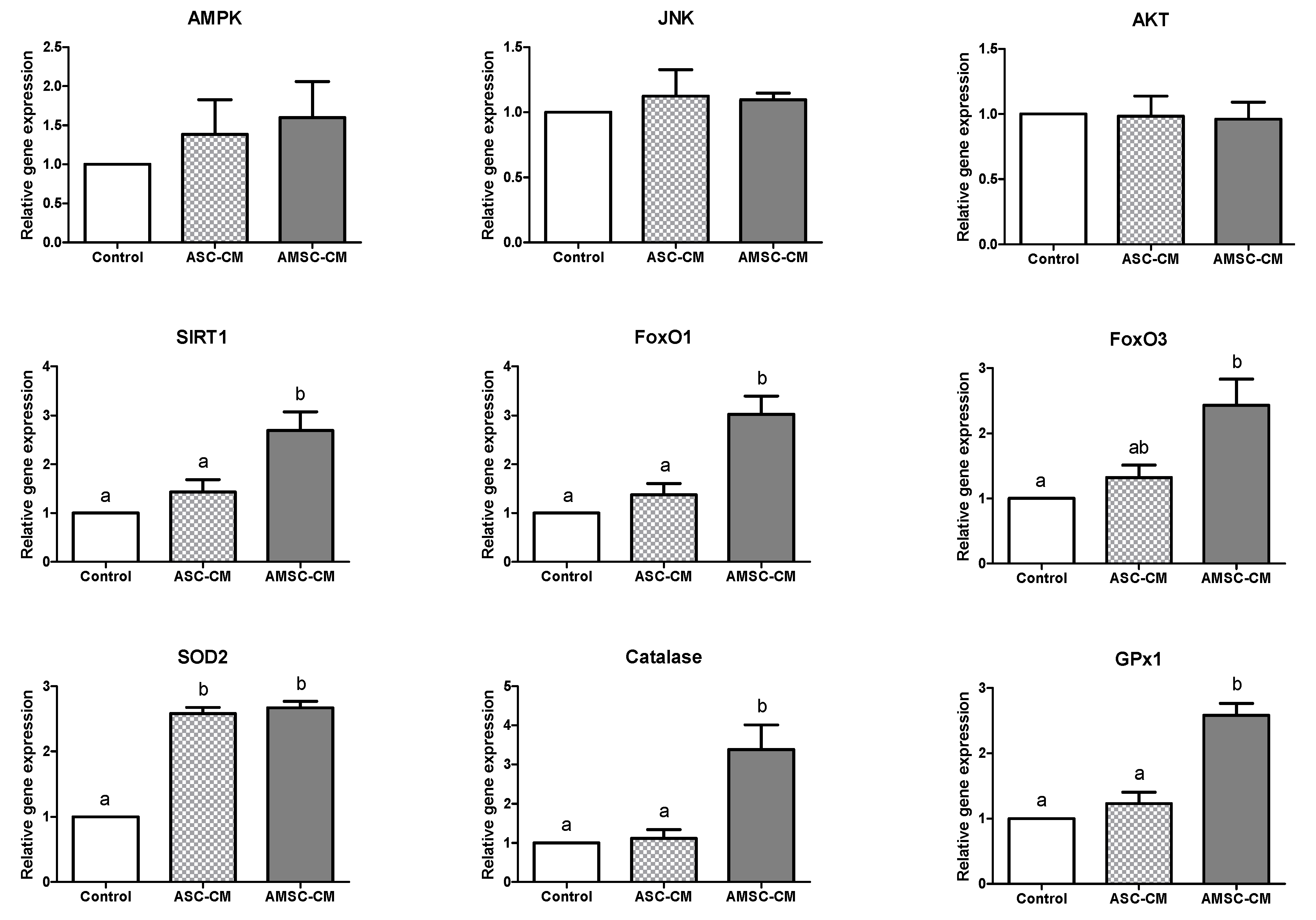
3.4. Comparative Effects of ASC-CM and AMSC-CM on Antioxidant Gene Expression in BL
3.5. Comparative Effects of ASC-CM and AMSC-CM on Intracellular Oxidative Stress in BL
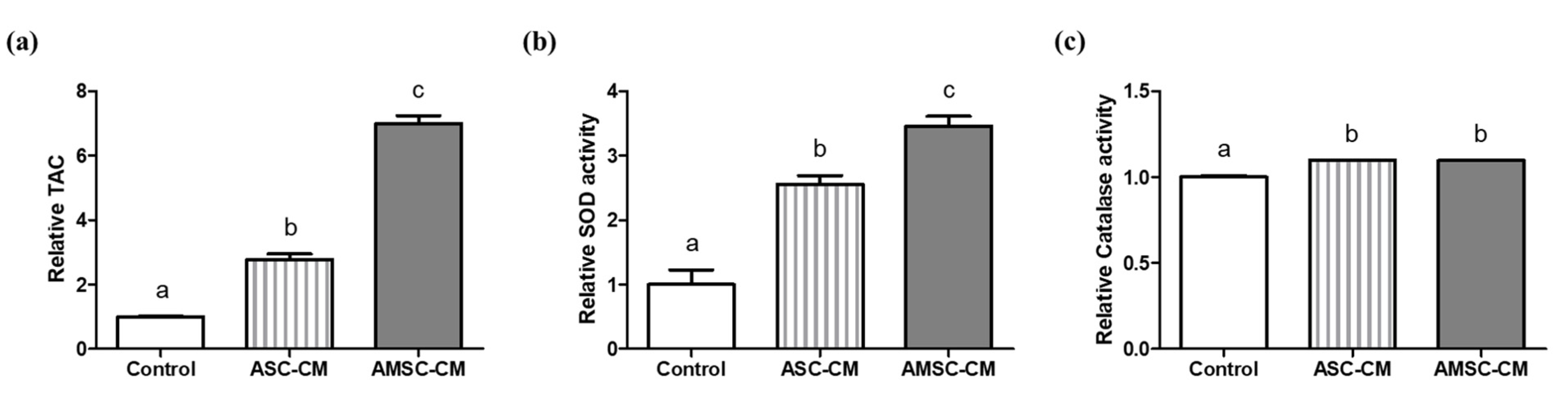
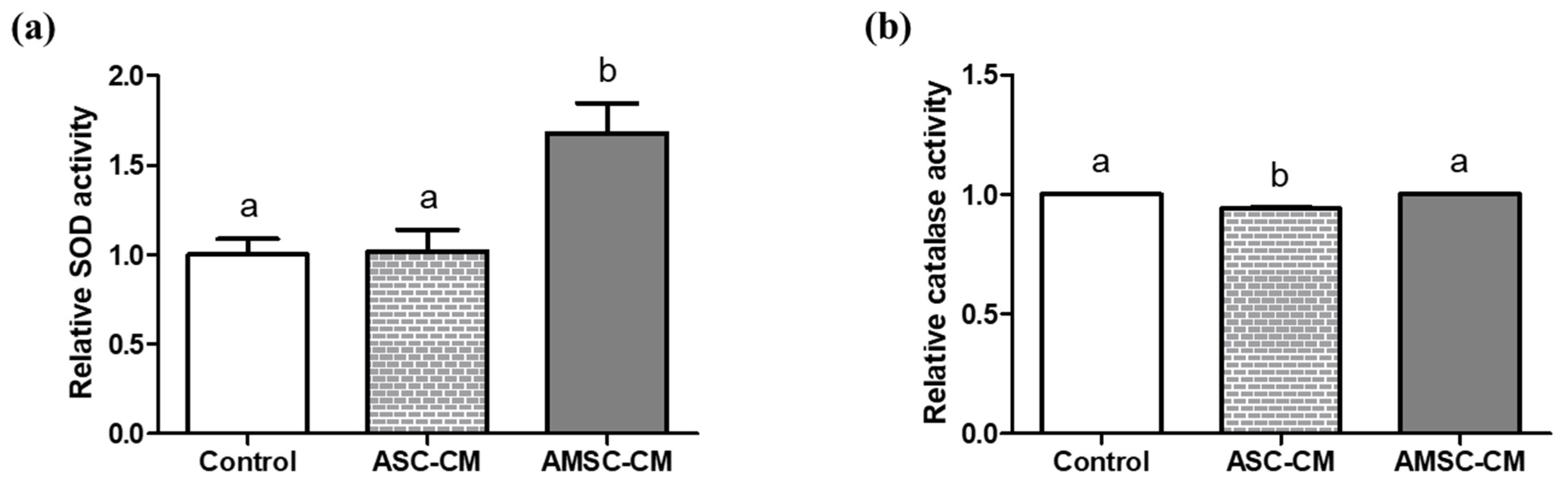
3.6. Comparison of Antioxidant Biomarkers in ASC-CM and AMSC-CM
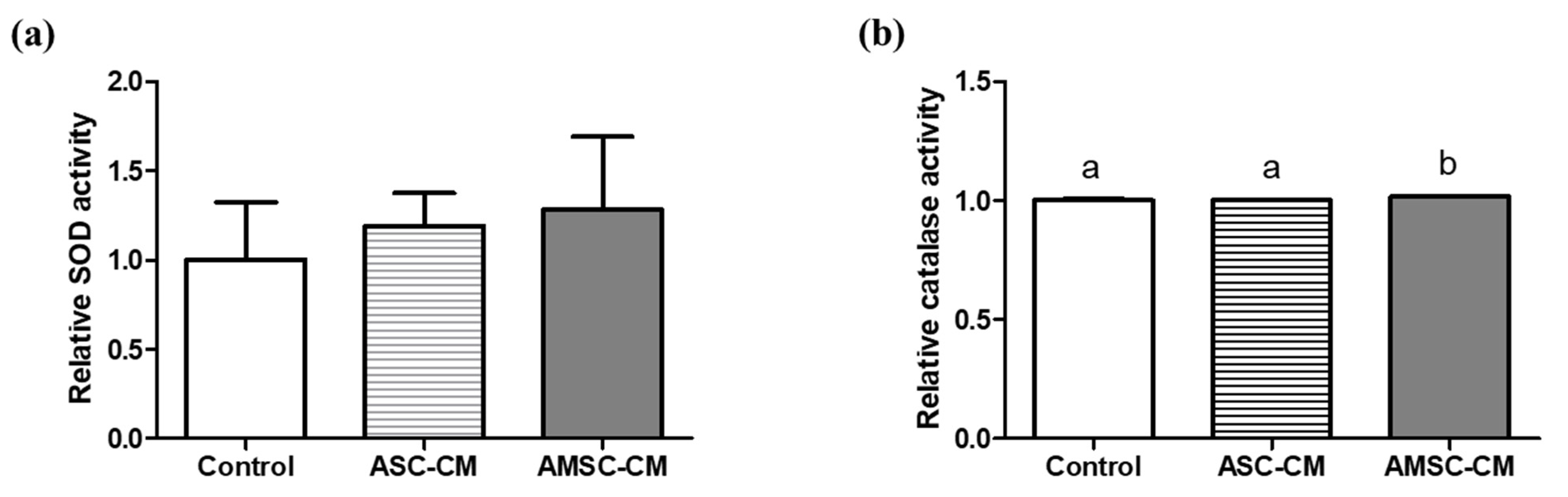
3.7. Comparison of Antioxidant Biomarkers in Culture Medium with ASC-CM and AMSC-CM
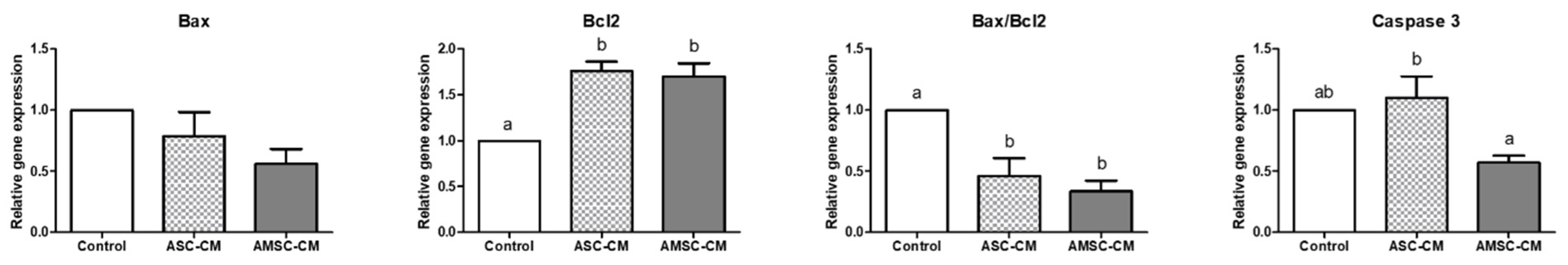
3.8. Comparative Effects of ASC-CM and AMSC-CM on Apoptosis-Related Gene Expression in BL
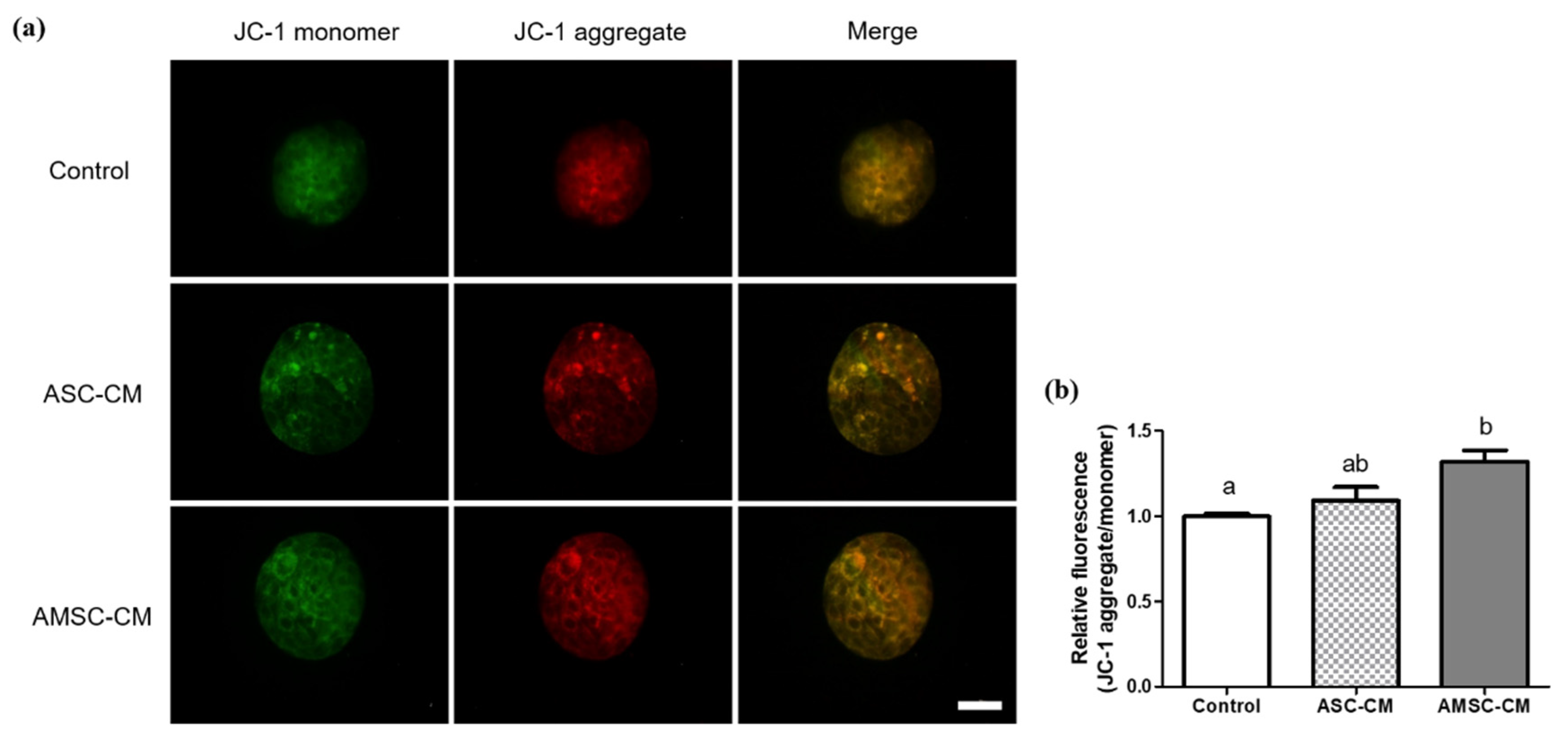
3.9. Comparative Effects of ASC-CM and AMSC-CM on Intracellular Apoptosis in BL
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Chronopoulou, E.; Harper, J.C. IVF culture media: Past, present and future. Hum. Reprod. Update 2015, 21, 39–55. [Google Scholar] [CrossRef]
- Agarwal, A.; Allamaneni, S.S. Role of free radicals in female reproductive diseases and assisted reproduction. Reprod. Biomed. Online 2004, 9, 338–347. [Google Scholar] [CrossRef]
- Du Plessis, S.S.; Makker, K.; Desai, N.R.; Agarwal, A. Impact of oxidative stress on IVF. Expert Rev. Obstet. Gynecol. 2008, 3, 539–554. [Google Scholar] [CrossRef]
- Yang, H.W.; Hwang, K.J.; Kwon, H.C.; Kim, H.S.; Choi, K.W.; Oh, K.S. Detection of reactive oxygen species (ROS) and apoptosis in human fragmented embryos. Hum. Reprod. 1998, 13, 998–1002. [Google Scholar] [CrossRef] [PubMed]
- Oyawoye, O.; Abdel Gadir, A.; Garner, A.; Constantinovici, N.; Perrett, C.; Hardiman, P. Antioxidants and reactive oxygen species in follicular fluid of women undergoing IVF: Relationship to outcome. Hum. Reprod. 2003, 18, 2270–2274. [Google Scholar] [CrossRef]
- Ra, K.; Oh, H.J.; Kim, E.Y.; Kang, S.K.; Ra, J.C.; Kim, E.H.; Lee, B.C. Anti-Oxidative Effects of Human Adipose Stem Cell Conditioned Medium with Different Basal Medium during Mouse Embryo In Vitro Culture. Animals 2020, 10, 1414. [Google Scholar] [CrossRef]
- Agarwal, A.; Said, T.M.; Bedaiwy, M.A.; Banerjee, J.; Alvarez, J.G. Oxidative stress in an assisted reproductive techniques setting. Fertil. Steril. 2006, 86, 503–512. [Google Scholar] [CrossRef]
- Cambra, J.M.; Martinez, C.A.; Rodriguez-Martinez, H.; Martinez, E.A.; Cuello, C.; Gil, M.A. N-(2-mercaptopropionyl)-glycine enhances in vitro pig embryo production and reduces oxidative stress. Sci. Rep. 2020, 10, 18632. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Falcone, T.; Attaran, M.; Goldberg, J.M.; Agarwal, A.; Sharma, R.K. Vitamin C and vitamin E supplementation reduce oxidative stress-induced embryo toxicity and improve the blastocyst development rate. Fertil. Steril. 2002, 78, 1272–1277. [Google Scholar] [CrossRef]
- Abdelrazik, H.; Sharma, R.; Mahfouz, R.; Agarwal, A. L-carnitine decreases DNA damage and improves the in vitro blastocyst development rate in mouse embryos. Fertil. Steril. 2009, 91, 589–596. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Tian, X.; Zhang, L.; He, C.; Ji, P.; Li, Y.; Tan, D.; Liu, G. Beneficial effect of resveratrol on bovine oocyte maturation and subsequent embryonic development after in vitro fertilization. Fertil. Steril. 2014, 101, 577–586. [Google Scholar] [CrossRef] [PubMed]
- Gruber, I.; Klein, M. Embryo culture media for human IVF: Which possibilities exist? J. Turk. Ger. Gynecol. Assoc. 2011, 12, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Stavely, R.; Nurgali, K. The emerging antioxidant paradigm of mesenchymal stem cell therapy. Stem Cells Transl. Med. 2020, 9, 985–1006. [Google Scholar] [CrossRef]
- Ullah, I.; Subbarao, R.B.; Rho, G.J. Human mesenchymal stem cells—Current trends and future prospective. Biosci. Rep. 2015, 35. [Google Scholar] [CrossRef]
- Pittenger, M.F.; Discher, D.E.; Peault, B.M.; Phinney, D.G.; Hare, J.M.; Caplan, A.I. Mesenchymal stem cell perspective: Cell biology to clinical progress. NPJ Regen. Med. 2019, 4, 22. [Google Scholar] [CrossRef]
- Kim, W.S.; Park, B.S.; Sung, J.H. The wound-healing and antioxidant effects of adipose-derived stem cells. Expert Opin. Biol. Ther. 2009, 9, 879–887. [Google Scholar] [CrossRef]
- Kim, W.S.; Park, B.S.; Kim, H.K.; Park, J.S.; Kim, K.J.; Choi, J.S.; Chung, S.J.; Kim, D.D.; Sung, J.H. Evidence supporting antioxidant action of adipose-derived stem cells: Protection of human dermal fibroblasts from oxidative stress. J. Dermatol. Sci. 2008, 49, 133–142. [Google Scholar] [CrossRef]
- Ra, K.; Oh, H.J.; Kim, G.A.; Kang, S.K.; Ra, J.C.; Lee, B.C. High Frequency of Intravenous Injection of Human Adipose Stem Cell Conditioned Medium Improved Embryo Development of Mice in Advanced Maternal Age through Antioxidant Effects. Animals 2020, 10, 978. [Google Scholar] [CrossRef]
- Toda, A.; Okabe, M.; Yoshida, T.; Nikaido, T. The potential of amniotic membrane/amnion-derived cells for regeneration of various tissues. J. Pharmacol. Sci. 2007, 105, 215–228. [Google Scholar] [CrossRef]
- Kim, E.Y.; Lee, K.B.; Kim, M.K. The potential of mesenchymal stem cells derived from amniotic membrane and amniotic fluid for neuronal regenerative therapy. BMB Rep. 2014, 47, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Diaz-Prado, S.; Muinos-Lopez, E.; Hermida-Gomez, T.; Rendal-Vazquez, M.E.; Fuentes-Boquete, I.; de Toro, F.J.; Blanco, F.J. Multilineage differentiation potential of cells isolated from the human amniotic membrane. J. Cell Biochem. 2010, 111, 846–857. [Google Scholar] [CrossRef]
- Abbasi-Kangevari, M.; Ghamari, S.H.; Safaeinejad, F.; Bahrami, S.; Niknejad, H. Potential Therapeutic Features of Human Amniotic Mesenchymal Stem Cells in Multiple Sclerosis: Immunomodulation, Inflammation Suppression, Angiogenesis Promotion, Oxidative Stress Inhibition, Neurogenesis Induction, MMPs Regulation, and Remyelination Stimulation. Front. Immunol. 2019, 10, 238. [Google Scholar] [CrossRef]
- Bulati, M.; Miceli, V.; Gallo, A.; Amico, G.; Carcione, C.; Pampalone, M.; Conaldi, P.G. The Immunomodulatory Properties of the Human Amnion-Derived Mesenchymal Stromal/Stem Cells Are Induced by INF-gamma Produced by Activated Lymphomonocytes and Are Mediated by Cell-To-Cell Contact and Soluble Factors. Front. Immunol. 2020, 11, 54. [Google Scholar] [CrossRef]
- Wang, Y.; Ma, J.; Du, Y.; Miao, J.; Chen, N. Human Amnion-Derived Mesenchymal Stem Cells Protect Human Bone Marrow Mesenchymal Stem Cells against Oxidative Stress-Mediated Dysfunction via ERK1/2 MAPK Signaling. Mol. Cells 2016, 39, 186–194. [Google Scholar] [CrossRef]
- He, F.; Wang, Y.; Li, Y.; Yu, L. Human amniotic mesenchymal stem cells alleviate paraquat-induced pulmonary fibrosis in rats by inhibiting the inflammatory response. Life Sci. 2020, 243, 117290. [Google Scholar] [CrossRef]
- Xie, C.; Jin, J.; Lv, X.; Tao, J.; Wang, R.; Miao, D. Anti-aging Effect of Transplanted Amniotic Membrane Mesenchymal Stem Cells in a Premature Aging Model of Bmi-1 Deficiency. Sci. Rep. 2015, 5, 13975. [Google Scholar] [CrossRef] [PubMed]
- Jiao, H.; Shi, K.; Zhang, W.; Yang, L.; Yang, L.; Guan, F.; Yang, B. Therapeutic potential of human amniotic membrane-derived mesenchymal stem cells in APP transgenic mice. Oncol. Lett. 2016, 12, 1877–1883. [Google Scholar] [CrossRef] [PubMed]
- Li, J.Y.; Ren, K.K.; Zhang, W.J.; Xiao, L.; Wu, H.Y.; Liu, Q.Y.; Ding, T.; Zhang, X.C.; Nie, W.J.; Ke, Y.; et al. Human amniotic mesenchymal stem cells and their paracrine factors promote wound healing by inhibiting heat stress-induced skin cell apoptosis and enhancing their proliferation through activating PI3K/AKT signaling pathway. Stem Cell Res. Ther. 2019, 10, 247. [Google Scholar] [CrossRef] [PubMed]
- Kusuma, G.D.; Carthew, J.; Lim, R.; Frith, J.E. Effect of the Microenvironment on Mesenchymal Stem Cell Paracrine Signaling: Opportunities to Engineer the Therapeutic Effect. Stem Cells Dev. 2017, 26, 617–631. [Google Scholar] [CrossRef]
- Pawitan, J.A. Prospect of stem cell conditioned medium in regenerative medicine. Biomed. Res. Int. 2014, 2014, 965849. [Google Scholar] [CrossRef]
- Sagaradze, G.; Grigorieva, O.; Nimiritsky, P.; Basalova, N.; Kalinina, N.; Akopyan, Z.; Efimenko, A. Conditioned Medium from Human Mesenchymal Stromal Cells: Towards the Clinical Translation. Int. J. Mol. Sci. 2019, 20, 1656. [Google Scholar] [CrossRef] [PubMed]
- Simopoulou, M.; Sfakianoudis, K.; Rapani, A.; Giannelou, P.; Anifandis, G.; Bolaris, S.; Pantou, A.; Lambropoulou, M.; Pappas, A.; Deligeoroglou, E.; et al. Considerations Regarding Embryo Culture Conditions: From Media to Epigenetics. In Vivo 2018, 32, 451–460. [Google Scholar] [CrossRef] [PubMed]
- Leese, H.J.; Baumann, C.G.; Brison, D.R.; McEvoy, T.G.; Sturmey, R.G. Metabolism of the viable mammalian embryo: Quietness revisited. Mol. Hum. Reprod. 2008, 14, 667–672. [Google Scholar] [CrossRef] [PubMed]
- Eisenbach, M.; Giojalas, L.C. Sperm guidance in mammals—An unpaved road to the egg. Nat. Rev. Mol. Cell Biol. 2006, 7, 276–285. [Google Scholar] [CrossRef]
- Baak, N.A.; Cantineau, A.E.; Farquhar, C.; Brison, D.R. Temperature of embryo culture for assisted reproduction. Cochrane Database Syst. Rev. 2019, 9, CD012192. [Google Scholar] [CrossRef]
- Higdon, H.L., 3rd; Blackhurst, D.W.; Boone, W.R. Incubator management in an assisted reproductive technology laboratory. Fertil. Steril. 2008, 89, 703–710. [Google Scholar] [CrossRef]
- Muller, W.U. Temperature dependence of combined exposure of preimplantation mouse embryos to X-rays and mercury. Radiat. Environ. Biophys. 1990, 29, 109–114. [Google Scholar] [CrossRef]
- Morbeck, D.E.; Paczkowski, M.; Fredrickson, J.R.; Krisher, R.L.; Hoff, H.S.; Baumann, N.A.; Moyer, T.; Matern, D. Composition of protein supplements used for human embryo culture. J. Assist. Reprod. Genet. 2014, 31, 1703–1711. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, M.; Yoshida, K.; Kitada, K.; Kizu, A.; Tachibana, D.; Fukui, M.; Morita, T.; Koyama, M. Low-dose irradiation of mouse embryos increases Smad-p21 pathway activity and preserves pluripotency. J. Assist. Reprod. Genet. 2018, 35, 1061–1069. [Google Scholar] [CrossRef]
- Edwards, L.J.; Kind, K.L.; Armstrong, D.T.; Thompson, J.G. Effects of recombinant human follicle-stimulating hormone on embryo development in mice. Am. J. Physiol. Endocrinol. Metab. 2005, 288, E845–E851. [Google Scholar] [CrossRef]
- Zhang, S.; Lin, H.; Kong, S.; Wang, S.; Wang, H.; Wang, H.; Armant, D.R. Physiological and molecular determinants of embryo implantation. Mol. Aspects Med. 2013, 34, 939–980. [Google Scholar] [CrossRef]
- Hammadeh, M.E.; Fischer-Hammadeh, C.; Ali, K.R. Assisted hatching in assisted reproduction: A state of the art. J. Assist. Reprod. Genet. 2011, 28, 119–128. [Google Scholar] [CrossRef]
- Hwang, I.S.; Kim, H.G.; Jo, D.H.; Lee, D.H.; Koo, Y.H.; Song, Y.J.; Na, Y.J.; Choi, O.H. Comparison with human amniotic membrane- and adipose tissue-derived mesenchymal stem cells. Korean J. Obstet. Gynecol. 2011, 54, 674–683. [Google Scholar] [CrossRef]
- Kim, D.; Kyung, J.; Park, D.; Choi, E.K.; Kim, K.S.; Shin, K.; Lee, H.; Shin, I.S.; Kang, S.K.; Ra, J.C.; et al. Health Span-Extending Activity of Human Amniotic Membrane- and Adipose Tissue-Derived Stem Cells in F344 Rats. Stem Cells Transl. Med. 2015, 4, 1144–1154. [Google Scholar] [CrossRef]
- Dizaji Asl, K.; Shafaei, H.; Soleimani Rad, J.; Nozad, H.O. Comparison of Characteristics of Human Amniotic Membrane and Human Adipose Tissue Derived Mesenchymal Stem Cells. World J. Plast. Surg. 2017, 6, 33–39. [Google Scholar]
- Kuscu, N.; Gungor-Ordueri, N.E.; Sozen, B.; Adiguzel, D.; Celik-Ozenci, C. FoxO transcription factors 1 regulate mouse preimplantation embryo development. J. Assist. Reprod. Genet. 2019, 36, 2121–2133. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Wang, Z.; Cao, J.; Chen, Y.; Dong, Y. A novel and compact review on the role of oxidative stress in female reproduction. Reprod. Biol. Endocrinol. 2018, 16, 80. [Google Scholar] [CrossRef]
- Wang, Y.; Zhou, Y.; Graves, D.T. FOXO transcription factors: Their clinical significance and regulation. Biomed. Res. Int. 2014, 2014, 925350. [Google Scholar] [CrossRef] [PubMed]
- Greer, E.L.; Oskoui, P.R.; Banko, M.R.; Maniar, J.M.; Gygi, M.P.; Gygi, S.P.; Brunet, A. The energy sensor AMP-activated protein kinase directly regulates the mammalian FOXO3 transcription factor. J. Biol. Chem. 2007, 282, 30107–30119. [Google Scholar] [CrossRef]
- Essers, M.A.; Weijzen, S.; de Vries-Smits, A.M.; Saarloos, I.; de Ruiter, N.D.; Bos, J.L.; Burgering, B.M. FOXO transcription factor activation by oxidative stress mediated by the small GTPase Ral and JNK. EMBO J. 2004, 23, 4802–4812. [Google Scholar] [CrossRef]
- Brunet, A.; Bonni, A.; Zigmond, M.J.; Lin, M.Z.; Juo, P.; Hu, L.S.; Anderson, M.J.; Arden, K.C.; Blenis, J.; Greenberg, M.E. Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell 1999, 96, 857–868. [Google Scholar] [CrossRef]
- Fasano, C.; Disciglio, V.; Bertora, S.; Lepore Signorile, M.; Simone, C. FOXO3a from the Nucleus to the Mitochondria: A Round Trip in Cellular Stress Response. Cells 2019, 8, 1110. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.K.; Webb, A.E. Regulation of FOXO Factors in Mammalian Cells. Curr. Top. Dev. Biol. 2018, 127, 165–192. [Google Scholar] [CrossRef] [PubMed]
- Tatone, C.; Di Emidio, G.; Vitti, M.; Di Carlo, M.; Santini, S., Jr.; D’Alessandro, A.M.; Falone, S.; Amicarelli, F. Sirtuin Functions in Female Fertility: Possible Role in Oxidative Stress and Aging. Oxid. Med. Cell Longev. 2015, 2015, 659687. [Google Scholar] [CrossRef]
- Salmen, J.J.; Skufca, F.; Matt, A.; Gushansky, G.; Mason, A.; Gardiner, C.S. Role of glutathione in reproductive tract secretions on mouse preimplantation embryo development. Biol. Reprod. 2005, 73, 308–314. [Google Scholar] [CrossRef]
- Khazaei, M.; Aghaz, F. Reactive Oxygen Species Generation and Use of Antioxidants during In Vitro Maturation of Oocytes. Int. J. Fertil. Steril. 2017, 11, 63–70. [Google Scholar] [CrossRef]
- Zhang, T.; Andrukhov, O.; Haririan, H.; Muller-Kern, M.; Liu, S.; Liu, Z.; Rausch-Fan, X. Total Antioxidant Capacity and Total Oxidant Status in Saliva of Periodontitis Patients in Relation to Bacterial Load. Front. Cell Infect. Microbiol. 2015, 5, 97. [Google Scholar] [CrossRef]
- Baraniak, P.R.; McDevitt, T.C. Stem cell paracrine actions and tissue regeneration. Regen. Med. 2010, 5, 121–143. [Google Scholar] [CrossRef]
- Maguire, G. Stem cell therapy without the cells. Commun. Integr. Biol. 2013, 6, e26631. [Google Scholar] [CrossRef]
- Gunawardena, T.N.A.; Rahman, M.T.; Abdullah, B.J.J.; Abu Kasim, N.H. Conditioned media derived from mesenchymal stem cell cultures: The next generation for regenerative medicine. J. Tissue Eng. Regen. Med. 2019, 13, 569–586. [Google Scholar] [CrossRef]
- Grzywocz, Z.; Pius-Sadowska, E.; Klos, P.; Gryzik, M.; Wasilewska, D.; Aleksandrowicz, B.; Dworczynska, M.; Sabalinska, S.; Hoser, G.; Machalinski, B.; et al. Growth factors and their receptors derived from human amniotic cells in vitro. Folia Histochem. Cytobiol. 2014, 52, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Fernandez, M.; Castilla-Cortazar, I.; Diaz-Sanchez, M.; Navarro, I.; Puche, J.E.; Castilla, A.; Casares, A.D.; Clavijo, E.; Gonzalez-Baron, S. Antioxidant effects of insulin-like growth factor-I (IGF-I) in rats with advanced liver cirrhosis. BMC Gastroenterol. 2005, 5, 7. [Google Scholar] [CrossRef] [PubMed]
- Goksen, S.; Balabanli, B.; Coskun-Cevher, S. Application of platelet derived growth factor-BB and diabetic wound healing: The relationship with oxidative events. Free Radic. Res. 2017, 51, 498–505. [Google Scholar] [CrossRef] [PubMed]
- Kalay, Z.; Cevher, S.C. Oxidant and antioxidant events during epidermal growth factor therapy to cutaneous wound healing in rats. Int. Wound J. 2012, 9, 362–371. [Google Scholar] [CrossRef]
- Santangelo, C.; Matarrese, P.; Masella, R.; Di Carlo, M.C.; Di Lillo, A.; Scazzocchio, B.; Vecci, E.; Malorni, W.; Perfetti, R.; Anastasi, E. Hepatocyte growth factor protects rat RINm5F cell line against free fatty acid-induced apoptosis by counteracting oxidative stress. J. Mol. Endocrinol. 2007, 38, 147–158. [Google Scholar] [CrossRef] [PubMed]
- Wei, T.; Shu, Q.; Ning, J.; Wang, S.; Li, C.; Zhao, L.; Zheng, H.; Gao, H. The Protective Effect of Basic Fibroblast Growth Factor on Diabetic Nephropathy Through Remodeling Metabolic Phenotype and Suppressing Oxidative Stress in Mice. Front. Pharmacol. 2020, 11, 66. [Google Scholar] [CrossRef]
- McIlwain, D.R.; Berger, T.; Mak, T.W. Caspase functions in cell death and disease. Cold Spring Harb. Perspect. Biol. 2013, 5, a008656. [Google Scholar] [CrossRef]
- Rigoulet, M.; Yoboue, E.D.; Devin, A. Mitochondrial ROS generation and its regulation: Mechanisms involved in H2O2 signaling. Antioxid. Redox. Signal. 2011, 14, 459–468. [Google Scholar] [CrossRef]
- Amable, P.R.; Teixeira, M.V.; Carias, R.B.; Granjeiro, J.M.; Borojevic, R. Protein synthesis and secretion in human mesenchymal cells derived from bone marrow, adipose tissue and Wharton’s jelly. Stem Cell Res. Ther. 2014, 5, 53. [Google Scholar] [CrossRef]
- Pires, A.O.; Mendes-Pinheiro, B.; Teixeira, F.G.; Anjo, S.I.; Ribeiro-Samy, S.; Gomes, E.D.; Serra, S.C.; Silva, N.A.; Manadas, B.; Sousa, N.; et al. Unveiling the Differences of Secretome of Human Bone Marrow Mesenchymal Stem Cells, Adipose Tissue-Derived Stem Cells, and Human Umbilical Cord Perivascular Cells: A Proteomic Analysis. Stem Cells Dev. 2016, 25, 1073–1083. [Google Scholar] [CrossRef]
- Pervaiz, S.; Holme, A.L. Resveratrol: Its biologic targets and functional activity. Antioxid. Redox. Signal. 2009, 11, 2851–2897. [Google Scholar] [CrossRef] [PubMed]
- Pinarli, F.A.; Turan, N.N.; Pinarli, F.G.; Okur, A.; Sonmez, D.; Ulus, T.; Oguz, A.; Karadeniz, C.; Delibasi, T. Resveratrol and adipose-derived mesenchymal stem cells are effective in the prevention and treatment of doxorubicin cardiotoxicity in rats. Pediatr. Hematol. Oncol. 2013, 30, 226–238. [Google Scholar] [CrossRef] [PubMed]
- Okay, E.; Simsek, T.; Subasi, C.; Gunes, A.; Duruksu, G.; Gurbuz, Y.; Gacar, G.; Karaoz, E. Cross effects of resveratrol and mesenchymal stem cells on liver regeneration and homing in partially hepatectomized rats. Stem Cell Rev. Rep. 2015, 11, 322–331. [Google Scholar] [CrossRef] [PubMed]
- Xian, Y.; Lin, Y.; Cao, C.; Li, L.; Wang, J.; Niu, J.; Guo, Y.; Sun, Y.; Wang, Y.; Wang, W. Protective effect of umbilical cord mesenchymal stem cells combined with resveratrol against renal podocyte damage in NOD mice. Diabetes Res. Clin. Pract. 2019, 156, 107755. [Google Scholar] [CrossRef]

| Gene | Accession No. | Primer Sequence |
|---|---|---|
| 18S rRNA | NR_003278.3 | F: ACCGCGGTTCTATTTTGTTG |
| R: CCCTCTTAATCATGGCCTCA | ||
| AMPK | NM_001013367.3 | F: GCTGTGGCTCACCCAATTAT |
| R: ATCAAAAGGGAGGGTTCCAC | ||
| JNK | NM_016700.4 | F: CGGAACACCTTGTCCTGAAT |
| R: GAGTCAGCTGGGAAAAGCAC | ||
| AKT | NM_001165894.1 | F: ACTCATTCCAGACCCACGAC |
| R: GTCCAGGGCAGACACAATCT | ||
| SIRT1 | NM_001159589.2 | F: AGTTCCAGCCGTCTCTGTGT |
| R: GATCCTTTGGATTCCTGCAA | ||
| FoxO1 | NM_019739.3 | F: ACATTTCGTCCTCGAACCAG |
| R: CAGGTCATCCTGCTCTGTCA | ||
| FoxO3 | NM_019740.3 | F: ATGGGAGCTTGGAATGTGAC |
| R: TTAAAATCCAACCCGTCAGC | ||
| SOD2 | NM_013671.3 | F: CTGTCTTCAGCCACACCAGA |
| R: CTGCTCTTCCAAAGGTCCTG | ||
| Catalase | NM_009804.2 | F: TTGACAGAGAGCGGATTCCT |
| R: TCTGGTGATATCGTGGGTGA | ||
| GPx1 | NM_008160.6 | F: CCGACCCCAAGTACATCATT |
| R: CCCACCAGGAACTTCTCAAA | ||
| Bax | NM_007527.3 | F: ACCAAGAAGCTGAGCGAGTG |
| R: TGCAGCTCCATATTGCTGTC | ||
| Bcl2 | NM_009741.5 | F: ATGATAACCGGGAGATCGTG |
| R: AGCCCCTCTGTGACAGCTTA | ||
| Caspase3 | NM_001284409.1 | F: TGTCATCTCGCTCTGGTACG |
| R: ATTTCAGGCCCATGAATGTC |
| Group | No. of Cultured Embryos | No. of Embryos Developed to (%) | |||
|---|---|---|---|---|---|
| 4-Cell | 16-Cell | Blastocyst | Hatched Blastocyst | ||
| Control | 72 | 66 (91.8 ± 1.8) b | 51 (71.1 ± 2.6) ab | 28 (38.6 ± 4.5) a | 14 (19.4 ± 4.6) a |
| 10% AMSC-CM | 74 | 69 (92.5 ± 4.8) b | 55 (74.3 ± 4.8) b | 39 (51.7 ± 4.1) b | 25 (32.6 ± 5.9) b |
| 20% AMSC-CM | 74 | 69 (93.4 ± 3.0) b | 45 (61.2 ± 3.5) a | 23 (30.7 ± 5.5) a | 15 (19.1 ± 3.5) a |
| 50% AMSC-CM | 76 | 61 (79.6 ± 4.0) a | 46 (59.5 ± 3.8) a | 24 (28.3 ± 6.8) a | 16 (18.8 ± 3.4) a |
| Group | No. of Cultured Embryos | Number of Embryos Developed to (%) | |||
|---|---|---|---|---|---|
| 4-Cell | 16-Cell | Blastocyst | Hatched Blastocyst | ||
| Control | 135 | 127 (93.1 ± 2.0) | 101 (73.7 ± 3.3) a | 62 (44.4 ± 5.2) a | 40 (27.4 ± 8.0) |
| ASC-CM | 134 | 124 (91.9 ± 1.7) | 108 (79.2 ± 4.0) ab | 76 (56.4 ± 2.8) ab | 44 (32.2 ± 4.0) |
| AMSC-CM | 130 | 125 (95.7 ± 1.4) | 117 (87.6 ± 5.1) b | 85 (65.7 ± 3.3) b | 53 (39.7 ± 2.8) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ra, K.; Oh, H.J.; Kim, E.Y.; Kang, S.K.; Ra, J.C.; Kim, E.H.; Park, S.C.; Lee, B.C. Comparison of Anti-Oxidative Effect of Human Adipose- and Amniotic Membrane-Derived Mesenchymal Stem Cell Conditioned Medium on Mouse Preimplantation Embryo Development. Antioxidants 2021, 10, 268. https://doi.org/10.3390/antiox10020268
Ra K, Oh HJ, Kim EY, Kang SK, Ra JC, Kim EH, Park SC, Lee BC. Comparison of Anti-Oxidative Effect of Human Adipose- and Amniotic Membrane-Derived Mesenchymal Stem Cell Conditioned Medium on Mouse Preimplantation Embryo Development. Antioxidants. 2021; 10(2):268. https://doi.org/10.3390/antiox10020268
Chicago/Turabian StyleRa, Kihae, Hyun Ju Oh, Eun Young Kim, Sung Keun Kang, Jeong Chan Ra, Eui Hyun Kim, Se Chang Park, and Byeong Chun Lee. 2021. "Comparison of Anti-Oxidative Effect of Human Adipose- and Amniotic Membrane-Derived Mesenchymal Stem Cell Conditioned Medium on Mouse Preimplantation Embryo Development" Antioxidants 10, no. 2: 268. https://doi.org/10.3390/antiox10020268
APA StyleRa, K., Oh, H. J., Kim, E. Y., Kang, S. K., Ra, J. C., Kim, E. H., Park, S. C., & Lee, B. C. (2021). Comparison of Anti-Oxidative Effect of Human Adipose- and Amniotic Membrane-Derived Mesenchymal Stem Cell Conditioned Medium on Mouse Preimplantation Embryo Development. Antioxidants, 10(2), 268. https://doi.org/10.3390/antiox10020268






