Abstract
In recent times, the use of natural products has gained momentum, either as a treatment or as adjuvants for other drugs in the treatment of different conditions. Propolis is a natural substance produced by bees which has proven useful for treating periodontal disease. This systematic review and meta-analysis gather evidence of the effectiveness of propolis in this kind of condition. The MEDLINE, CENTRAL, PubMed, EMBASE and Web of Science databases were searched for scientific articles to identify the findings published up to October 2020. The MeSH phrases used in the search were: “periodontal diseases AND propolis treatment”; “gingivitis AND propolis treatment”; “periodontitis AND propolis treatment”; “propolis treatment AND oral health”; “propolis AND oxidative stress AND periodontitis”. The Boolean operator “AND” was used to combine the searches. Randomized trials where propolis was used in the treatment of different periodontal conditions were included. Non-randomized clinical studies were systematically reviewed and 224 studies were detected, eight of which met the criteria for inclusion in the meta-analysis. Only three of these were selected for quantitative synthesis. In conclusion, propolis is safe to use and can improve the results of periodontal disease treatment, reducing probing pocket depth compared with treatment with a placebo (difference in means, fixed effects −0.67 [95% CI: −0.84, −0.50]).
1. Introduction
Propolis is a non-toxic resinous substance produced by bees that has antimicrobial, antifungal, anti-inflammatory, antioxidant and antitumor properties, among others [1,2], which have attracted the attention of researchers, both in the medical and in the dental field.
It is a mixture of plant extracts mixed with the bees’ own saliva, which varies according to its place of origin, with Brazil leading the research on this product. Its composition is based on vegetable resins (50%), waxes (30%), essential and aromatic oils (10%) and pollen and other organic substances (10%). The chemical composition of propolis depends on the geographic location, the botanical origin and the species of bee [3,4]. Nevertheless, its composition is quite complex, its main components being flavonoids and phenolic esters such as caffeic acid phenethyl ester [5,6]. The flavonoids contained in propolis have been found to have antimicrobial, anti-inflammatory and immunomodulating properties, which are extremely useful to treat aphthous ulcers, candidiasis, gingivitis and periodontitis [7,8].
Periodontal disease is likely to be the most common chronic infection in adults [9]. There is evidence that suggests that nearly all forms of periodontal disease are specific chronic bacterial infections that stem from the overgrowth in dental plaque of a limited number of species, mainly anaerobic, such as Porphyromonas gingivalis, Bacteroides forsythus and Treponema denticola [10], considering the induction of inflammatory signaling pathways by pathogenic bacteria to be crucial for the development of inflammatory processes in the periodontium [11].
Periodontal diseases have traditionally been associated with the development of certain systemic conditions, systemic inflammation being one of the main reasons for this association [12].
The presence of inflammatory infiltrate and an increase in the oxidative response of peripheral polymorphonuclear cells, are constant characteristics of periodontal disease, there being greater damage as the disease advances because of the increase in the number of reactive oxygen substances (ROS). The increase in the amount of free radicals, causes oxidative damage to gingival tissue, periodontal ligament and alveolar bone. The deterioration caused by free radicals is regulated by an antioxidant defense system, in such a way that when an imbalance between the production of free radicals and antioxidant levels takes place, it triggers a condition known as oxidative stress (OS), which is defined as an imbalance between the production of free radicals and the body’s capacity to eliminate these reactive species [13,14,15].
Free radicals are extremely unstable organic and inorganic molecules that contain an unpaired electron. They have the capacity to take electrons from other atoms and molecules, causing a release of proinflammatory cytokines such as interleukin-2, 6 and 8 (IL-2, IL-6 and IL-8), interferon-β and tumor necrosis factor α (TNF-α), which play an extremely relevant role in the pathogenesis of periodontal disease [16,17].
It is believed that disturbances in the local and/or general indicators of oxidative stress are one of the mechanisms in the etiopathogenesis of periodontitis, and, in recent years, different basic clinical and experimental studies have provided evidence of a strong association between oxidative stress and periodontal disease [18].
A better understanding of this association could provide deeper insight into the pathology of this disease, shed further light on the relationship between periodontal disease and systemic inflammation, and increase knowledge of therapeutic approaches.
As systematic reviews are an essential tool to synthesize the available scientific information and identify areas of uncertainty where research is crucial, the purpose of this study was to conduct a systematic review of the literature on the effectiveness of propolis in the treatment of periodontal disease.
Meta-analysis (where possible) provides very useful information that facilitates understanding of the effect of a treatment in specific patient groups. Likewise, it enhances accuracy in the estimation of a certain effect, identifying moderate but clinically important degrees that could go unnoticed in primary studies. This meta-analysis was carried out using randomized clinical trials (RCTs) on the use of propolis in the treatment of periodontal disease.
2. Materials and Methods
The study selection process was carried out according to the Preferred Reporting Items for Systematic Review and Meta-Analyses (PRISMA) guidelines for systematic reviews and meta-analysis [19] (Table S1 Checklist).
2.1. Protocol
The search strategy was conducted using the population, intervention, comparison and outcome (PICO) framework, based on the following question:
“Is propolis effective in the treatment of periodontal disease?”
To answer this question, a sample of patients with periodontal disease, regardless of age, was selected. Treatment consisted of the use of propolis products, such as mouthwashes/gels or capsules, alone or complementary to other non-surgical therapies. Control patients were treated using conventional treatments, placebos or other types of mouthwash/gel. The results reviewed in the literature were plaque index, gingival indices, periodontal indices and microbiological parameters.
2.2. Data Sources and Search Strategy
The MEDLINE, CENTRAL, PubMed, EMBASE and Web of Science electronic databases were searched for findings published until October 2020. The MeSH phrases (Meaning of MeSH: Medical Subject Headings) used in the search were: “periodontal diseases AND propolis treatment”; “gingivitis AND propolis treatment”; “periodontitis AND propolis treatment”; “propolis treatment AND oral health”; “propolis AND oxidative stress AND periodontitis”. The Boolean operator “AND” was used to combine and narrow the searches.
2.3. Inclusion and Exclusion Criteria
Inclusion criteria:
- (a)
- Articles published in English,
- (b)
- Randomized controlled clinical trials,
- (c)
- Non-randomized studies assessing the effectiveness of propolis in the treatment of periodontal diseases.
Exclusion criteria:
- (a)
- In vitro studies,
- (b)
- Animal studies,
- (c)
- Comparative studies,
- (d)
- Systematic reviews,
- (e)
- Clinical cases,
- (f)
- Non-relevant studies (e.g., effectiveness of propolis in the treatment of other conditions, narrative reviews…), duplicate studies and those that did not meet the inclusion criteria stated above.
2.4. Data Extraction and Analysis
Studies that did not refer to the research question were removed, and the titles and abstracts of the selected articles were collected and entered in an Excel spreadsheet. Two reviewers (NL-V and AL-V) selected the titles and abstracts independently. Disagreements regarding study inclusion were resolved through discussions between the two reviewers mentioned. Subsequently, the full texts of the selected studies were obtained for review and inclusion. The bibliographical references of each study were reviewed as possible sources for finding additional studies.
2.5. Quality of the Reports of the Included Randomized Trials
This was assessed using the Jadad scale [20], which defines the methodological quality of the studies based on their description of randomization, blinding and withdrawals (dropouts). The scale ranges from 0 to 5, a score ≤ 2 meaning low report quality and a score ≥ 3 meaning high report quality (Table 1).

Table 1.
Jadad quality score of randomized controlled trials included in the meta-analysis.
2.6. Statistical Analysis
The meta-analysis was performed using RevMan 5 software (Review Manager (RevMan) [computer program], version 5.3; Copenhagen, The Nordic Cochrane Centre, The Cochrane Collaboration, 2014). Difference in means (DM) and standard deviation (SD) were used to assess continuous variables (probing depth) with a 95% confidence interval (CI). The threshold for statistical significance was p < 0.05. While the possibility of performing a quantitative analysis of the different parameters measured in the studies was considered, it was only possible to carry out the meta-analysis of probing depth assessment. Regarding the rest of the parameters assessed in the studies, the Oral Hygiene Index (OHI) was evaluated in 2 of the studies [24,25] and the Gingival Index (GI) in another 2 [25,28]. Although gingival bleeding was measured in most of the studies, it was tested using different indices depending on the study, which created a disparity of clinical criteria for assessment and precluded the unification of data [21,22,23,26,27,28]. As for plaque control, 4 studies tested it using the Plaque Index (PI) [23,24,25,26], although 2 of them [25,26] did not provide numerical data. Concerning clinical attachment level, although it was measured in 3 of the studies [21,22,28], that by Sparabombe and colleagues [22] did not explain the measurement criterion, which means that it was discordant with the measurement units used in the other 2 studies. Because of this, it was determined that the only parameter that was tested with unity of criterion among the studies selected was probing pocket depth (PPD), measured in mm and examined in 6 of the 8 studies selected for the meta-analysis [21,22,23,25,26,28].
3. Results
3.1. Characteristics of the Studies
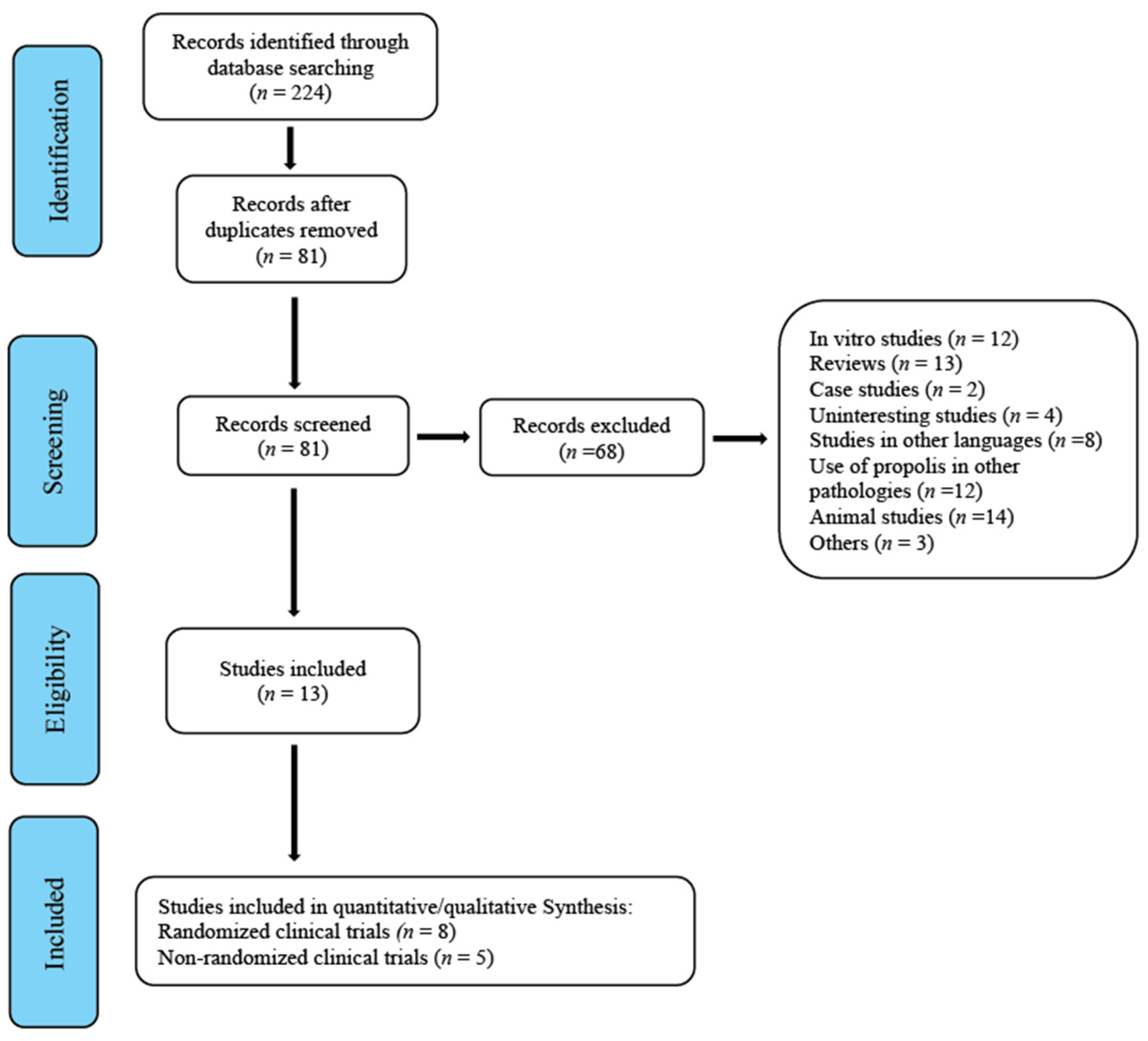
Until October 2020, 224 studies were gathered and subsequently assessed by the reviewers. The first screening led to the removal of 143 duplicates. In a second screening, 65 studies that did not clearly meet the inclusion criteria and were therefore considered inadequate were removed. After this, three more were removed for different reasons: one because it was a review/commentary [29], one that tested the efficacy of propolis in reducing chemotherapy-induced oral mucositis [30] and another [31] because it was study of equivalence conducted on induced gingivitis. This left a total of 13 studies: eight randomized clinical trials to be included in the meta-analysis [21,22,23,24,25,26,27,28] and five non-randomized studies [32,33,34,35,36] (Figure 1, flowchart).
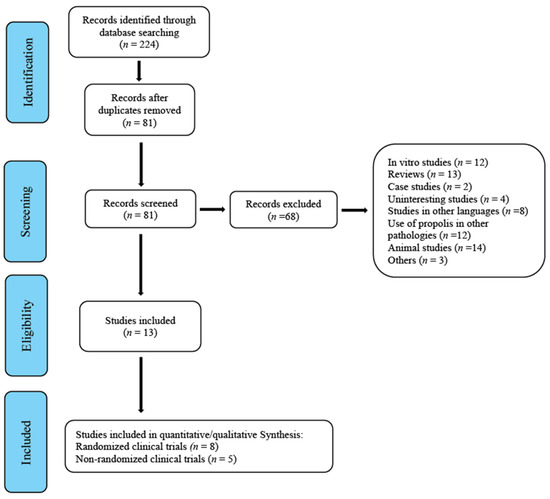
Figure 1.
Flowchart.
Table 2 provides a general description of the details of the randomized studies. The five clinical trials [32,33,34,35,36] that were not relevant to our meta-analysis were systematically reviewed.

Table 2.
Characteristics of randomized clinical studies.
3.2. Methodological Quality of the Included Randomized Studies
All the studies included in the meta-analysis reached a Jadad scale score that was compatible with high methodological quality (≥3 points), the study of Nakao and colleagues [21] achieving the highest score (Table 1).
3.3. Results of the Meta-Analysis
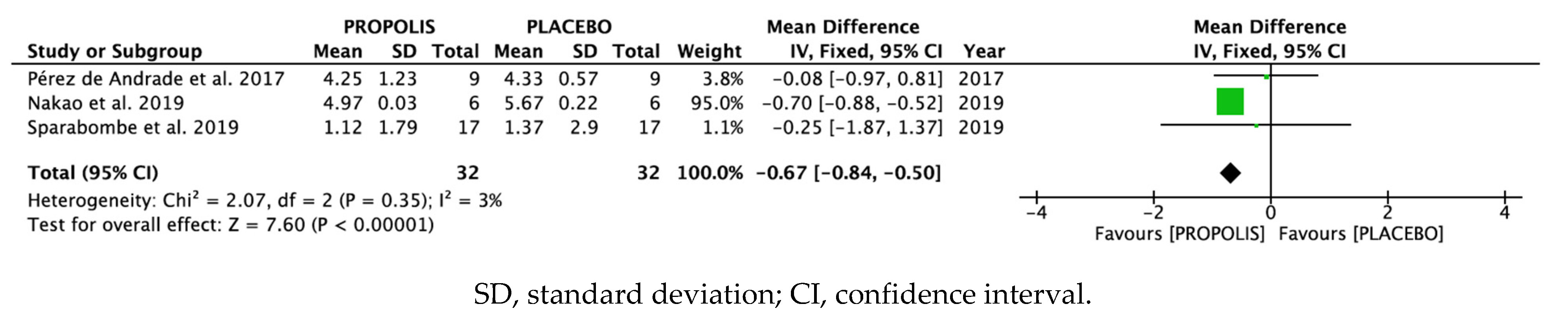
Meta-analysis was carried out to evaluate probing pocket depth at 3 months from the beginning of treatment with propolis or with a placebo, according to the tested group [21,22,23,25,26,28]. The study by Sharkawy and colleagues [26] was excluded from the meta-analysis because although it measured probing depth, it did not include numerical data. The studies by Giammarinaro and colleagues [23] and Shangani and colleagues [28] were also excluded on the grounds that they used chlorhexidine rather than a placebo with the control group to carry out the comparison and did not perform probing 3 months after the beginning of the trial. Thus, only three studies were eligible for quantitative synthesis [21,22,25].
Low heterogeneity among studies (I2 = 3%, 95% CI) led to the selection of a fixed effects model, assuming that the differences among studies were not the result of heterogeneity but of random effects.
A forest plot was used to check that the difference between both treatments in individual studies was not significant in the studies by Pérez de Andrade and colleagues [25] and Sparabombe and colleagues [22], since the 95% confidence intervals overlapped and crossed the line of no effect. In the study by Nakao and colleagues [21], the experimental group treated with propolis yielded better results than the group treated with a placebo. Moreover, this study achieved a weight of 95% in the meta-analysis.
According to the overall results of the meta-analysis, treatment with propolis reduced probing pocket depth as compared with treatment with the placebo (difference in means, fixed effects –0.67; 95% CI: −0.84, −0.50) (Figure 2).
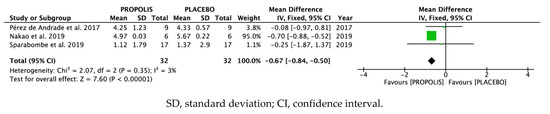
Figure 2.
Forest plot.
3.4. Publication Bias and Heterogeneity
The low number of selected studies precluded the evaluation of publication bias using a funnel plot.
3.5. Results of Systematic Review
A systematic review was conducted to summarize the clinical trials that were not eligible for meta-analysis because they were not randomized [32,33,34,35,36]. One of the studies, which tested the use of propolis-based toothpaste on a sample of 30 individuals aged 7 to 12 once a day for a 4-month period [32], reported an exponential reduction in salivary microbial load from the beginning of the treatment. Three studies [33,34,35] evaluated dental plaque and anaerobic bacteria in patients treated with propolis solutions and placebos. All the studies reported the benefits of propolis regarding the Plaque Index (PI) and Oral Hygiene Index (OHI), with a ≥25% reduction; only the study by Gebaraa and colleagues [36], based on a sample of 20 patients diagnosed with chronic periodontitis and treated with propolis or placebo, revealed no statistically significant differences between groups in terms of Plaque Index (PI), Gingival Index (GI) and Clinical Attachment Level (CAL). Nevertheless, these authors had previously reported the in vitro antimicrobial efficacy of propolis using propolis extracts in serial concentrations [37]. Table 3 provides a summary of the characteristics and results of these trials.

Table 3.
Characteristics of Non-Randomized Studies.
3.6. Assessment of Non-Randomized Clinical Trials
These were evaluated using the Newcastle–Ottawa Scale (NOS). Recommended by the Cochrane Non-Randomized Studies Methods Working Group [38], this is an instrument developed to assess the quality of non-randomized studies. Each study was assigned a score of 0–9. Studies that scored ≥7 were considered high-quality articles. All non-randomized clinical studies included in the systematic review were of high quality (Table 4).

Table 4.
The Newcastle–Ottawa Scale (NOS).
4. Discussion
Treatments based on natural products are regarded as alternative or complementary in certain oral conditions. Indeed, products from the hive such as honey and royal jelly, are used to treat mucositis and other disorders of the oral mucosa [39,40,41].
Although propolis has been traditionally used in folk medicine to treat certain diseases, its mode of action and the chemicals that are responsible for its therapeutic activity remain unknown. It is generally used as a mouthwash at different concentrations (1%, 2.5%, 5%, 10%), although there are other formulations that include oral capsules, gels or cosmetic creams [42].
The aim of this meta-analysis was to determine the effectiveness of propolis in the treatment of periodontal disease. According to the findings, propolis acts better than standardized treatments for certain therapeutic goals such as reducing dental plaque and microbial activity and stabilizing gingival and periodontal indices. It is significant that none of the results included in this review reported harmful or counterproductive effects in participants treated with propolis.
Two of the studies [22,24] addressed the effect of propolis on dental plaque control; four [23,25,26,27] explored the effects of propolis in relation to the reduction of probing pocket depth, inflammation and gingival bleeding; and two [21,28] analyzed the decrease in Porphyromonas gingivalis through the use of propolis mouthwashes.
The antimicrobial effect of propolis against periodontal pathogens has been studied in vivo and in vitro [33,43,44]. Nakao and colleagues [21] reported significant improvements in CAL and PPD alongside a trend towards a reduction of Porphyromonas gingivalis (a pathogen that plays a key role in periodontal disease) in gingival crevicular fluid (GCF) in patients treated with propolis solutions. Similarly, Yoshimasu and colleagues [45] proved the effectiveness of isolated propolis products such as artepillin C, baccharin and ursolic acid as antimicrobial compounds against Porphyromonas gingivalis; artepillin C and bacchatin are bacteriostatics and ursolic acid is a powerful destructor of the bacterial membrane, probably because of its highly lipophilic nature. In a study based on a sample of 20 patients diagnosed with chronic periodontitis, Sanghani and colleagues [28] reported a statistically significant reduction (p < 0.05) of Porphyromonas gingivalis, Prevotella intermedia and Fusobacterium nucleatum in periodontal pockets in the group of patients under propolis treatment.
Two of the selected studies analyzed the effect of propolis on dental plaque. Sparabombe and colleagues [22] evaluated its anti-inflammatory effect using a sample of patients with moderate/severe periodontitis who underwent a 3-month treatment with propolis-based mouthwashes, finding a significant improvement in the reduction of plaque buildup and gingival bleeding. Likewise, Piekarz and colleagues [24] reported a significant reduction during the first week of treatment with toothpaste containing ethanolic extract of propolis (p < 0.006), and a very significant reduction after 4 weeks of using it (p < 0.0002). These results are consistent with those of other studies on the antiplaque and antigingivitic effects of mouthwashes containing other flavonoids and essential oils in individuals with and without periodontal disease [46,47]. Nonetheless, certain studies have shown that certain forms of periodontitis are not associated with plaque and depend exclusively on the individual’s systemic condition [48].
Gingival and periodontal indices and salivary markers of oxidative stress were measured in four of the included studies [23,25,26,27]. Giammarinaro and colleagues [23] studied the efficacy of propolis as compared with chlorhexidine in a sample of 40 patients suffering from gingivitis, finding no significant differences between the control and the experimental group in probing pocket depth (PPD), bleeding on probing (BoP) and plaque index (PI); however, the patients treated with propolis achieved better results in terms of oxidative stress markers in the saliva, with considerable improvement in their periodontal health. Different studies have related oxidative stress in the saliva and the progression of periodontal disease. The main enzymatic antioxidants which have been widely studied in the gingival fluid, saliva and blood serum of patients with periodontitis are superoxide dismutase, glutathione peroxidase and catalase. The activity of these enzymes in gum tissue, gum fluid, saliva and blood serum during the different types of periodontitis (chronic or aggressive) is quite uneven. Despite the fact that Toczewska and colleagues reported that activity in gingival tissue is usually high [49], Tartaglia and colleagues [50], in a preliminary study, found that antioxidant levels in the saliva are reduced in patients with periodontal disease.
Likewise, Miricescu and colleagues [51] found high levels of oxidative stress associated with alveolar bone loss in the saliva of patients with periodontal disease. Likewise, other authors have suggested that patients suffering from this condition are more likely to experience oxidative stress imbalance and have reported that such a situation would be a consequence of periodontitis [52,53,54]. A study on test animals conducted by Aghel and colleagues [55] also proved the beneficial effect of propolis on saliva antioxidants.
The study by Sharkawy and colleagues [26] was the only one where propolis was used as a dietary supplement. It provided a comparison of the use of a placebo and the ingestion of 400 mg of propolis in patients with long-standing diabetes mellitus associated with periodontitis, reporting a significant reduction in periodontal parameters. The group treated with propolis showed a greater reduction in pocket depth (PD) and an increase in CAL as compared with the control group, probably because of the anti-inflammatory, antimicrobial and antioxidant activity of propolis. Interestingly, the use of propolis as complementary to oral hygiene revealed similar results in other studies [56,57].
Pérez de Andrade and colleagues, and Anauate-Netto and colleagues [25,27] reported a reduction in gingival inflammation and probing depth in patients treated with propolis solution mouthwashes as compared with those using 0.12% chlorhexidine or saline mouthwashes.
In general, the anti-inflammatory and antimicrobial properties of propolis have been well documented [56] and it will eventually be possible to explain any type of clinical result, either in healthy or diseased individuals, through the analysis of the oral microbiome [58,59,60].
Nevertheless, this study has a series of limitations, especially because of the heterogeneity found in the measurement of periodontal disease parameters such as plaque or bleeding, loss of clinical attachment, patient hygiene or the oxidative stress of the saliva. We only found unanimity among studies in the measurement in millimeters of probing depth, although two of the studies included did not measure this parameter [24,27]. Bleeding was measured using a variety of indices and assessment criteria: two studies did not evaluate gingival bleeding [24,25], two used the Bleeding on Probing (BoP) index [21,23], another used the Papillary Bleeding Score (PBS) [27], another the Eastman Interdental Bleeding Index (EIBI) [26] and others used unspecified bleeding indices [22,28].
In relation to plaque index, four studies used the Plaque Index (PI) [23,24,25,26], one used the Plaque Control Record (PCR) [21] and another [22] used the Plaque Score (PS); another two studies did not measure plaque [27,28]. An additional factor that is relevant to the assessment of periodontal disease is the Gingival Index (GI), which was only assessed in two studies [25,28]. It should also be noted that certain studies used different indices to measure the parameters and did not specify the criteria used for measurement with each of the indices, which hinders the interpretation of results. Others provided the data concerning the assessment of these indices at the beginning of the study but did not provide numerical data in reassessments, only showing graphs or mentioning that the parameters had been measured but providing no data. There were also discrepancies regarding assessment timing, some studies testing after 1 month and others after 3. Regarding oxidative stress, despite this being considered a predictable and measurable value related to a severe inflammatory state and a marker of the risk of periodontal disease [49,50], only the study by Giammarinaro and colleagues [23] dealt with this situation. All these factors hindered data analysis and precluded their inclusion for quantitative analysis.
For all these reasons, the authors recommend that the results be interpreted with caution, mainly due to the small number of selected studies and the small sample sizes used in each of them.
It would be advisable to perform a larger number of randomized clinical trials comparing the use of propolis with a placebo or chlorhexidine, using unified criteria, to assess periodontal parameters (plaque, bleeding, hygiene, dental mobility…) unified research indices and test timing for such parameters and structured, and standardized guidelines and measures in product administration, which could guarantee reliable and predictable results. Likewise, it would be advisable to reduce the bias of selective disclosure of results and for studies to provide the data related to all the assessed parameters, even if as annexed material to the published study.
5. Conclusions
Bearing in mind the limitations mentioned above, it can be concluded that propolis is safe to use and can enhance the results of periodontal disease treatment. Propolis-based therapies are likely to become an alternative treatment option in periodontal diseases and during supportive periodontal therapy. Nonetheless, for these conclusions to be definitely confirmed, further well-designed research, with broader samples, standardized protocols and long-term follow-up to ensure reliable results, is required.
Supplementary Materials
The following are available online at https://www.mdpi.com/2076-3921/10/2/269/s1, Table S1: PRISMA checklist.
Author Contributions
Study concept and design, N.L.-V., S.H.-H. and A.L.-V.; data collection (literature search and study selection), N.L.-V., J.F.-F. and J.M.R.; data analysis and interpretation (literature), B.P.-P.; J.F.-F., B.M.-d.-S. and J.H.-P.; drafting of the manuscript, N.L.-V.; A.L.-V. and B.P.-P.; critical revision of the manuscript for important intellectual content, A.L.-V. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| ROS | Reactive oxygen substances |
| OS | Oxidative Stress |
| SAT | Salivary antioxidant test |
| IL | Interleukin |
| TNF-α | Tumor Necrosis Factor α |
| PP | Periodontal pockets |
| PPD | Probing pocket depth |
| PI | Plaque Index |
| PS | Plaque Score |
| PCR | Plaque Control Record |
| OHI | Oral Hygiene Index |
| API | Approximal Plaque Index |
| SBI | Sulcus Bleeding Index |
| PD | Pocket depth |
| GI | Gingival Index |
| BI | Bleeding Index |
| BoP | Bleeding on Probing |
| CAL | Clinical attachment level |
| SRP | Scaling and root planing |
| PBS | Papillary Bleeding Score |
| NOS | Newcastle–Ottawa Scale |
| GCF | Gingival crevicular fluid |
| EIBI | Eastman Interdental Bleeding Index |
References
- Przybyłek, I.; Karpiński, T.M. Antibacterial properties of propolis. Molecules 2019, 24, 2047. [Google Scholar] [CrossRef]
- Martinello, M.; Mutinelli, F. Antioxidant activity in bee products: A review. Antioxidants 2021, 10, 71. [Google Scholar] [CrossRef]
- Huang, S.; Zhang, C.P.; Wang, K.; Li, G.Q.; Hu, F.L. Recent advances in the chemical composition of propolis. Molecules 2014, 19, 19610–19632. [Google Scholar] [CrossRef] [PubMed]
- Şenel, E.; Demir, E. Bibliometric analysis of apitherapy in complementary medicine literature between 1980 and 2016. Compl. Ther. Clin. Pract. 2018, 31, 47–52. [Google Scholar] [CrossRef]
- Bankova, V.; De Castro, S.; Marcucci, M. Propolis: Recent advances in chemistry and plant origin. Apidologie 2000, 31, 3–15. [Google Scholar] [CrossRef]
- Marcucci, M.C.; Ferreres, F.; Garcıa-Viguera, C.; Bankova, V.; De Castro, S.; Dantas, A.; Valente, P.; Paulino, N. Phenolic compounds from Brazilian propolis with pharmacological activities. J. Ethnopharmacol. 2001, 74, 105–112. [Google Scholar] [CrossRef]
- Almuhayawi, M.S. Propolis as a novel antibacterial agent. Saudi J. Biol. Sci. 2020, 27, 3079–3086. [Google Scholar] [CrossRef] [PubMed]
- Pasupuleti, V.R.; Sammugam, L.; Ramesh, N.; Gan, S.H. Honey, propolis, and royal jelly: A comprehensive review of their biological actions and health benefits. Oxid. Med. Cell Longev. 2017, 2017, 1259510. [Google Scholar] [CrossRef] [PubMed]
- Richards, D. Review finds that severe periodontitis affects 11% of the world population. Evid. Based Dent. 2014, 15, 70–71. [Google Scholar] [CrossRef] [PubMed]
- Loesche, W.J. Grossman NS. Periodontal disease as a specific, albeit chronic, infection: Diagnosis and treatment. Clin. Microbiol. Rev. 2001, 14, 727–752. [Google Scholar] [CrossRef] [PubMed]
- Armitage, G.C.; Robertson, P.B. The biology, prevention, diagnosis and treatment of periodontal diseases: Scientific advances in the United States. J. Am. Dent. Assoc. 2009, 140, 36S–43S. [Google Scholar] [CrossRef]
- Nazir, M.A. Prevalence of periodontal disease, its association with systemic diseases and prevention. Int. J. Health Sci. 2017, 11, 72–80. [Google Scholar]
- Hasturk, H.; Kantarci, A. Activation and resolution of periodontal inflammation and its systemic impact. Periodontol. 2000 2015, 69, 255–273. [Google Scholar] [CrossRef] [PubMed]
- Kesarwala, A.H.; Krishna, M.C.; Mitchell, J.B. Oxidative stress in oral diseases. Oral Dis. 2016, 22, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Sculley, D.; Langley-Evans, S. Periodontal disease is associated with lower antioxidant capacity in whole saliva and evidence of increased protein oxidation. Clin. Sci. 2003, 105, 167–172. [Google Scholar] [CrossRef] [PubMed]
- Pendyala, G.; Thomas, B.; Kumari, S. The challenge of antioxidants to free radicals in periodontitis. J. Ind. Soc. Periodontol. 2008, 12, 79–83. [Google Scholar] [CrossRef]
- Borges, I., Jr.; Moreira, E.A.; Filho, D.W.; de Oliveira, T.B.; da Silva, M.B.; Fröde, T.S. Proinflammatory and oxidative stress markers in patients with periodontal disease. Mediators Inflamm. 2007, 2007, 45794. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Andrukhov, O.; Rausch-Fan, X. Oxidative stress and antioxidant system in periodontitis. Front. Physiol. 2017, 8, 910. [Google Scholar] [CrossRef] [PubMed]
- Hutton, B.; Ferrán Catalá-López, F.; Moher, D. The PRISMA statement extension for systematic reviews incorporating network meta-analysis: PRISMA-NMA. Med. Clin. 2016, 16, 262–266. [Google Scholar] [CrossRef]
- Jadad, A.R.; Moore, R.A.; Carroll, D.; Jenkinson, C.; Reynolds, D.J.; Gavaghan, D.J.; McQuay, H.J. Assessing the quality of reports of randomized clinical trials: Is blinding necessary? Control. Clin. Trials 1996, 17, 1–12. [Google Scholar] [CrossRef]
- Nakao, R.; Senpuku, H.; Ohnishi, M.; Takai, H.; Ogata, Y. Effect of topical administration of propolis in chronic periodontitis. Odontology 2020, 108, 704–714. [Google Scholar] [CrossRef]
- Sparabombe, S.; Monterubbianesi, R.; Tosco, V.; Orilisi, G.; Hosein, A.; Ferrante, L.; Putignano, A.; Orsini, G. Efficacy of an all-natural polyherbal mouthwash in patients with periodontitis: A single-blind randomized controlled trial. Front. Physiol. 2019, 22, 10632. [Google Scholar] [CrossRef]
- Giammarinaro, E.; Marconcini, S.; Genovesi, A.; Poli, G.; Lorenzi, C.; Covani, U. Propolis as an adjuvant to non-surgical periodontal treatment: A clinical study with salivary antioxidant capacity assessment. Minerva Stomatol. 2018, 67, 183–188. [Google Scholar]
- Piekarz, T.; Mertas, A.; Wiatrak, K.; Rój, R.; Kownacki, P.; Śmieszek-Wilczewska, J.; Kopczyńska, E.; Wrzoł, M.; Cisowska, M.; Szliszka, E.; et al. The influence of toothpaste containing Australian melaleuca alternifolia oil and ethanolic extract of polish propolis on oral hygiene and microbiome in patients requiring conservative procedures. Molecules 2017, 22, 1957. [Google Scholar] [CrossRef] [PubMed]
- Andrade, D.P.; Carvalho, I.C.S.; Gadoi, B.H.; Rosa, L.C.L.; Barreto, L.M.R.C.; Pallos, D. Subgingival irrigation with a solution of 20% propolis extract as an adjunct to non-surgical periodontal treatment: A preliminary study. J. Int. Acad. Periodontol. 2017, 19, 145–151. [Google Scholar] [PubMed]
- El-Sharkawy, H.M.; Anees, M.M.; Van Dyke, T.E. Propolis Improves periodontal status and glycemic control in patients with type 2 diabetes mellitus and chronic periodontitis: A randomized clinical trial. J. Periodontol. 2016, 87, 1418–1426. [Google Scholar] [CrossRef] [PubMed]
- Anauate-Netto, C.; Anido-Anido, A.; Leegoy, H.R.; Matsumoto, R.; Alonso, R.C.; Marcucci, M.C.; Paulino, N.; Bretz, W.A. Randomized, double-blind, placebo-controlled clinical trial on the effects of propolis and chlorhexidine mouthrinses on gingivitis. Braz. Dent. Sci. 2014, 17, 11–15. [Google Scholar] [CrossRef]
- Sanghani, N.N.; Bm, S.S.S. Health from the hive: Propolis as an adjuvant in the treatment of chronic periodontitis—A clinicomicrobiologic study. J. Clin. Diagn. Res. 2014, 8, ZC41–ZC44. [Google Scholar] [PubMed]
- Borgnakke, W.S. Systemic propolis (adjuvant to nonsurgical periodontal treatment) may aid in glycemic control and periodontal health in type 2 diabetes of long duration. J. Evid Based Dent. Pract. 2017, 17, 132–134. [Google Scholar] [CrossRef] [PubMed]
- Akhavan Karbassi, M.H.; Yazdi, M.F.; Ahadian, H.; SadrAbad, M.J. Randomized double blind placebo controlled trial of propolis for oral mucositis in patients receiving chemotherapy for head and neck cancer. Asian Pac. J. Cancer Prev. 2016, 17, 3611–3614. [Google Scholar]
- Bretz, W.A.; Paulino, N.; Nör, J.E.; Moreira, A. The effectiveness of propolis on gingivitis: A randomized controlled trial. J. Altern. Compl. Med. 2014, 20, 943–948. [Google Scholar] [CrossRef] [PubMed]
- Mohsin, S.; Manohar, B.; Rajesh, S.; Asif, Y. The effects of a dentifrice containing propolis on Mutans Streptococci: A clinico-microbiological study. Ethiop. J. Health Sci. 2015, 25, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Coutinho, A. Honeybee propolis extract in periodontal treatment: A clinical and microbiological study of propolis in periodontal treatment. Ind. J. Dent. Res. 2012, 23, 294. [Google Scholar] [CrossRef]
- Tanasiewicz, M.; Skucha-Nowak, M.; Dawiec, M.; Król, W.; Skaba, D.; Twardawa, H. Influence of hygienic preparations with a 3% content of ethanol extract of Brazilian propolis on the state of the oral cavity. Adv. Clin. Exp. Med. 2012, 21, 81–92. [Google Scholar] [PubMed]
- Pereira, E.M.; da Silva, J.L.; Silva, F.F.; De Luca, M.P.; Ferreira, E.F.; Lorentz, T.C.; Santos, V.R. Clinical evidence of the efficacy of a mouthwash containing propolis for the control of plaque and gingivitis: A phase II study. Evid. Based Compl. Alter. Med. 2011, 2011, 750249. [Google Scholar] [CrossRef] [PubMed]
- Gebaraa, E.C.; Pustiglioni, A.N.; de Lima, L.A.; Mayer, M.P. Propolis extract as an adjuvant to periodontal treatment. Oral Health Prev. Dent. 2003, 1, 29–35. [Google Scholar]
- Gebaraa, E.C.; Lima, A.; Mayer, P.A. Propolis antimicrobial activity against periodontopathic bacteria. Braz. J. Microbiol. 2002, 33, 365–369. [Google Scholar] [CrossRef]
- Stang, A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur. J. Epidemiol. 2010, 5, 603–605. [Google Scholar] [CrossRef]
- Jayalekshmi, J.L.; Lakshmi, R.; Mukerji, A. Honey on oral mucositis: A randomized controlled trial. Gulf J. Oncol. 2016, 1, 30–37. [Google Scholar]
- Münstedt, K.; Männle, H. Using bee products for the prevention and treatment of oral mucositis induced by cancer treatment. Molecules 2019, 24, 3023. [Google Scholar] [CrossRef]
- Cherniack, E.P. Bugs as drugs, Part 1: Insects: The “new” alternative medicine for the 21st century? Altern. Med. Rev. 2010, 15, 124–135. [Google Scholar]
- Ozan, F.; Sümer, Z.; Polat, Z.A.; Er, K.; Ozan, U.; Deger, O. Effect of mouthrinse containing propolis on oral microorganisms and human gingival fibroblasts. Eur. J. Dent. 2007, 1, 195–201. [Google Scholar]
- Shabbir, A.; Rashid, M.; Tipu, H.N. Propolis—A hope for the future in treating resistant periodontal pathogens. Cureus 2016, 8, e682. [Google Scholar] [CrossRef] [PubMed]
- Eick, S.; Schäfer, G.; Kwieciński, J.; Atrott, J.; Henle, T.; Pfister, W. Honey—A potential agent against Porphyromonas gingivalis: An in vitro study. BMC Oral Health 2014, 14, 24. [Google Scholar] [CrossRef] [PubMed]
- Yoshimasu, Y.; Ikeda, T.; Sakai, N.; Yagi, A.; Hirayama, S.; Morinaga, Y.; Furukawa, S.; Nakao, R. Rapid bactericidal action of propolis against Porphyromonas gingivalis. J. Dent. Res. 2018, 97, 928–936. [Google Scholar] [CrossRef]
- Sharma, N.; Charles, C.H.; Lynch, M.C.; Qaqish, J.; Mcguire, J.A.; Galustians, J.G.; Kumar, L.D. Adjunctive benefit of an essential oil–containing mouthrinse in reducing plaque and gingivitis in patients who brush and floss regularly. J. Am. Dent. Assoc. 2004, 135, 496–504. [Google Scholar] [CrossRef]
- Cortelli, S.C.; Cortelli, J.R.; Holzhausen, M.; Franco, G.C.N.; Rebelo, R.Z.; Sonagere, A.S.; Queiroz Cda, S.; Costa, F.O. Essential oils in one-stage full-mouth disinfection: Double-blind, randomized clinical trial of long-term clinical, microbial and salivary effects. J. Clin. Periodontol. 2009, 36, 333–342. [Google Scholar] [CrossRef]
- Varela-López, A.; Quiles, J.L.; Cordero, M.; Giampieri, F.; Bullón, P. Oxidative stress and dietary fat type in relation to periodontal disease. Antioxidants 2015, 4, 322–344. [Google Scholar] [CrossRef] [PubMed]
- Toczewska, J.; Konopka, T. Activity of enzymatic antioxidants in periodontitis: A systematic overview of the literature. Dent. Med. Probl. 2019, 56, 419–426. [Google Scholar] [CrossRef]
- Tartaglia, G.M.; Gagliano, N.; Zarbin, L.; Tolomeo, G.; Sforza, C. Antioxidant capacity of human saliva and periodontal screening assessment in healthy adults. Arch. Oral Biol. 2017, 78, 34–38. [Google Scholar] [CrossRef]
- Miricescu, D.; Totan, A.; Calenic, B.; Mocanu, B.; Didilescu, A.; Mohora, M.; Spinu, T.; Greabu, M. Salivary biomarkers: Relationship between oxidative stress and alveolar bone loss in chronic periodontitis. Acta Odontol. Scand. 2014, 72, 47. [Google Scholar] [CrossRef] [PubMed]
- Wei, D.; Zhang, X.L.; Wang, Y.Z.; Yang, C.X.; Chen, G. Lipid peroxidation levels, total oxidant status and superoxide dismutase in serum, saliva and gingival crevicular fluid in chronic periodontitis patients before and after periodontal therapy. Aust. Dent. J. 2010, 55, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Brock, G.R.; Butterworth, C.J.; Matthews, J.B.; Chapple, I.L. Local and systemic total antioxidant capacity in periodontitis and health. J. Clin. Periodontol. 2004, 31, 515–521. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.; Chen, H.S.; Chen, S.L.; Ho, Y.P.; Ho, K.Y.; Wu, Y.M.; Hung, C.C. Lipid peroxidation: A possible role in the induction and progression of chronic periodontitis. J. Periodontal. Res. 2005, 40, 378–384. [Google Scholar] [CrossRef] [PubMed]
- Aghel, S.; Pouramir, M.; Moghadamnia, A.A.; Moslemi, D.; Molania, T.; Ghassemi, L.; Motallebnejad, M. Effect of Iranian propolis on salivary total antioxidant capacity in gamma-irradiated rats. J. Dent. Res. Dent. Clin. Dent. Prosp. 2014, 8, 235–239. [Google Scholar]
- Murray, M.C.; Worthington, H.V.; Blinkhorn, A.S. A study to investigate the effect of a propolis-containing mouthrinse on the inhibition of de novo plaque formation. J. Clin. Periodontol. 1997, 24, 796–798. [Google Scholar] [CrossRef] [PubMed]
- Vanni, R.; Waldner-Tomic, N.M.; Belibasakis, G.N.; Attin, T.; Schmidlin, P.R.; Thurnheer, T. Antibacterial efficacy of a propolis toothpaste and mouthrinse against a supragingival multispecies biofilm. Oral Health Prev. Dent. 2015, 3, 531–535. [Google Scholar]
- Paulino, N.; Abreu, S.R.; Uto, Y.; Koyama, D.; Nagasawa, H.; Hori, H.; Dirsch, V.M.; Vollmar, A.M.; Scremin, A.; Bretz, W.A. Anti-inflammatory effects of a bioavailable compound, Artepillin C, in Brazilian propolis. Eur. J. Pharmacol. 2008, 10, 296–301. [Google Scholar] [CrossRef] [PubMed]
- Peterson, S.N.; Snesrud, E.; Schork, N.J.; Bretz, W.A. Dental caries pathogenicity: A genomic and metagenomic perspective. Int. Dent. J. 2011, 61, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Matsha, T.E.; Prince, Y.; Davids, S.; Chikte, U.; Erasmus, R.T.; Kengne, A.P.; Davison, G.M. Oral microbiome signatures in diabetes mellitus and periodontal disease. J. Dent. Res. 2020, 99, 658–665. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).