A Correlation Study on In Vitro Physiological Activities of Soybean Cultivars, 19 Individual Isoflavone Derivatives, and Genetic Characteristics
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Materials
2.2. Preparation of Soybean Seed Extract
2.3. Antioxidant Activity Assay
2.3.1. ABTS Radical Scavenging Activity
2.3.2. DPPH Radical Scavenging Activity
2.4. Cell Potency Assay
2.4.1. Estrogen and ER Alpha Activity
2.4.2. Uncoupling Protein-1 (UCP-1) Activity
2.4.3. Nitric Oxide (NO) Inhibition Activity
2.5. Characterization of Isoflavone in Soybean Core Genetic Resource Seeds
2.5.1. Isoflavone Extraction and Solid Phase Extraction (SPE) Process
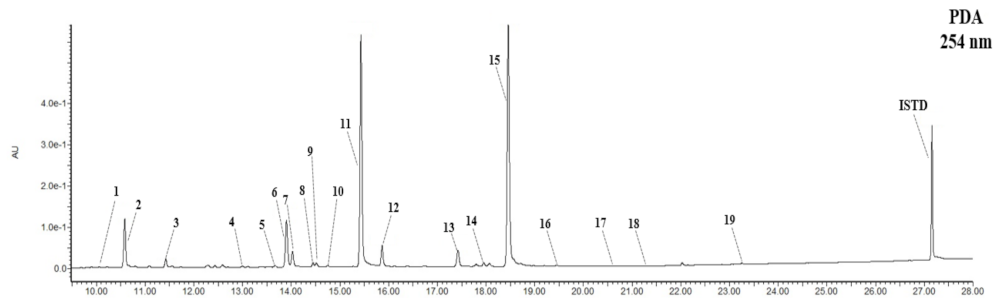
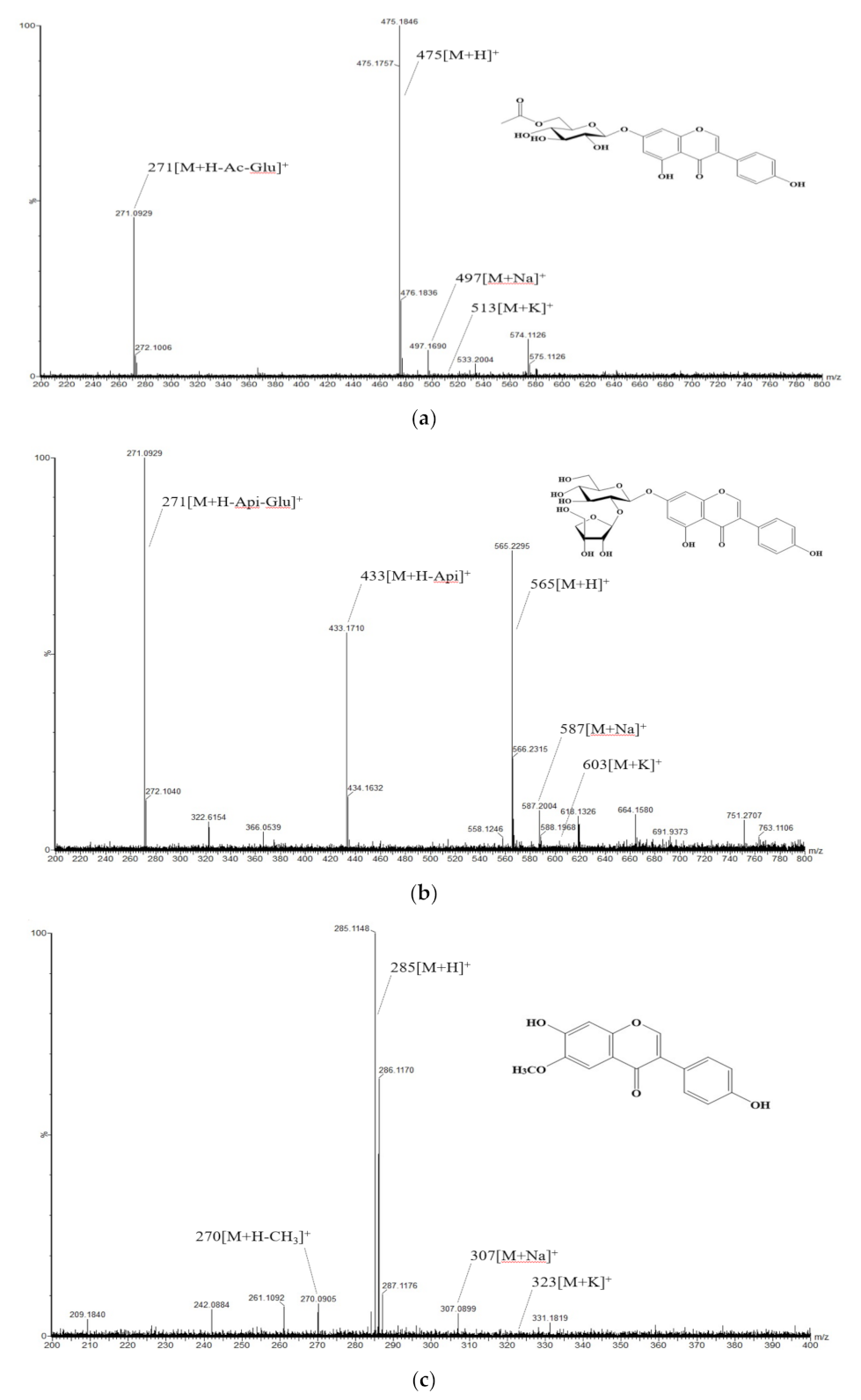
2.5.2. Isolation and Component Analysis of Individual Isoflavone Derivatives
2.6. DNA Extraction and Genotyping of Soybean Seeds
2.7. Statistical Analysis
3. Results and Discussion
3.1. Physiological Activity Analysis of Soybean Seeds
3.1.1. Antioxidant Activity of Soybean Seeds (Radical Scavenging Activity)
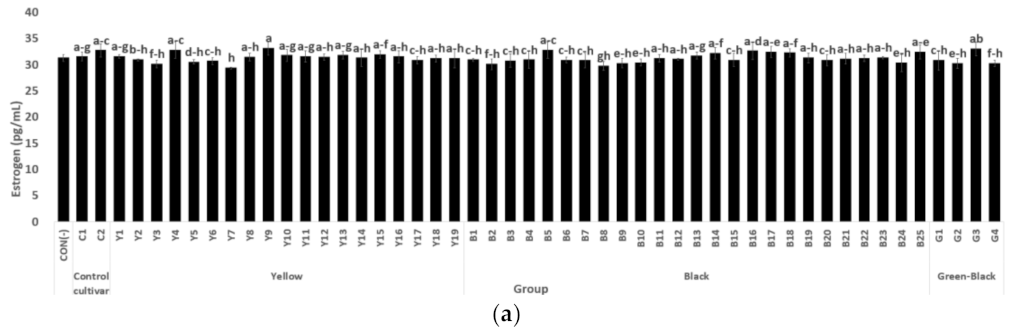
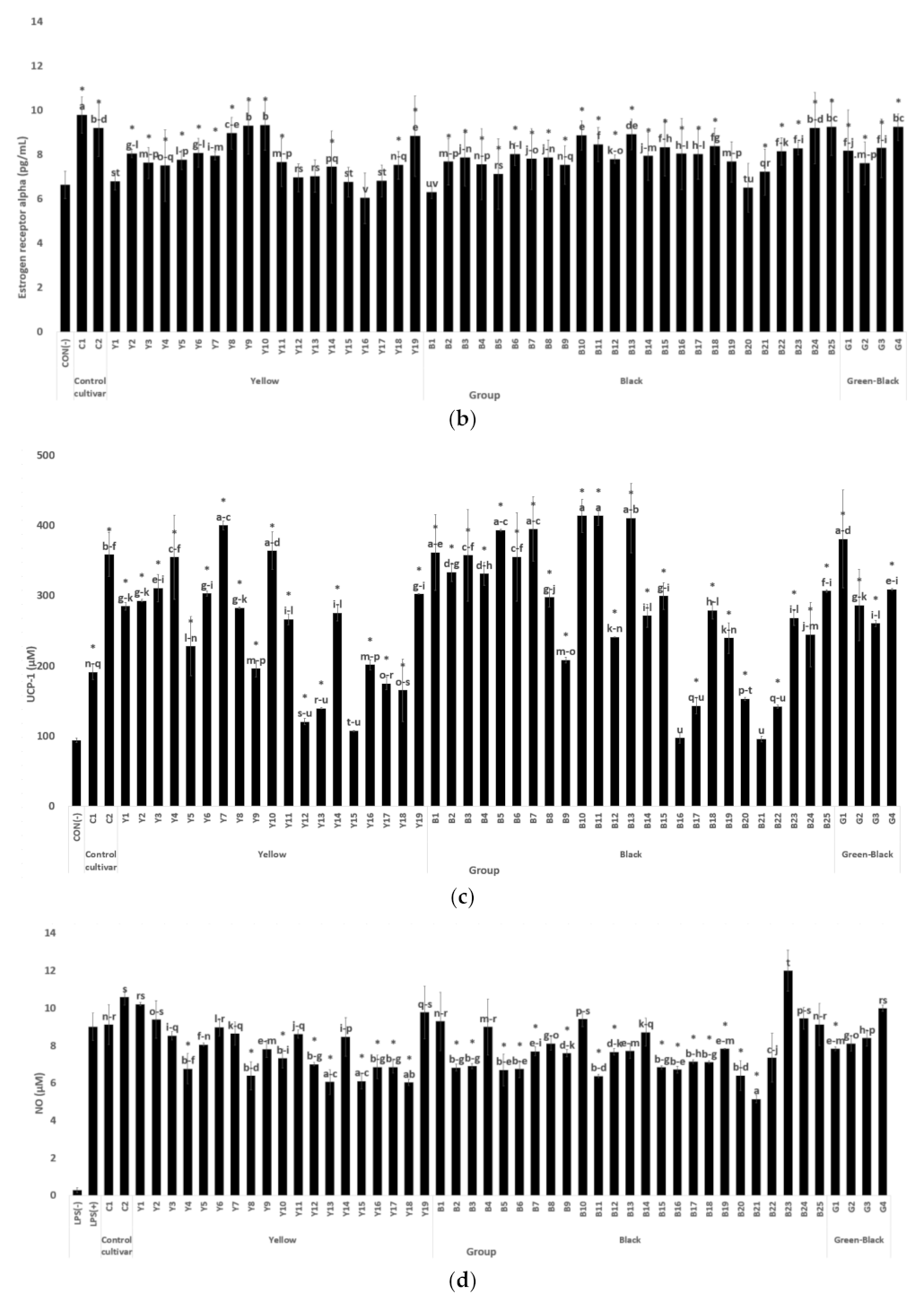
3.1.2. Estrogen and ER Alpha Activity in MCF-7 Cells
3.1.3. Antiobesity Activity Based on Brown Fat Transformation Activity (UCP-1 Activity of C3H10T1/2 Cells)
3.1.4. NO Production Inhibition Activity of RAW 264.7 Cells
3.2. Composition and Content of Isoflavone in Soybean Seed Core Genetic Resources
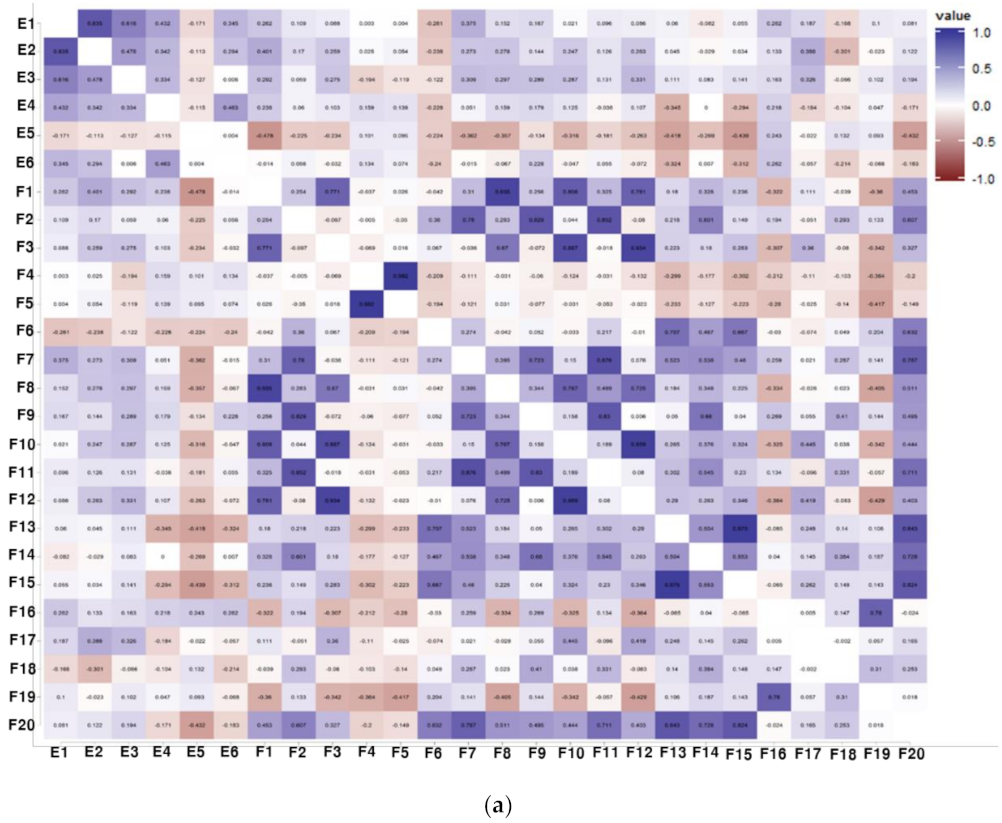
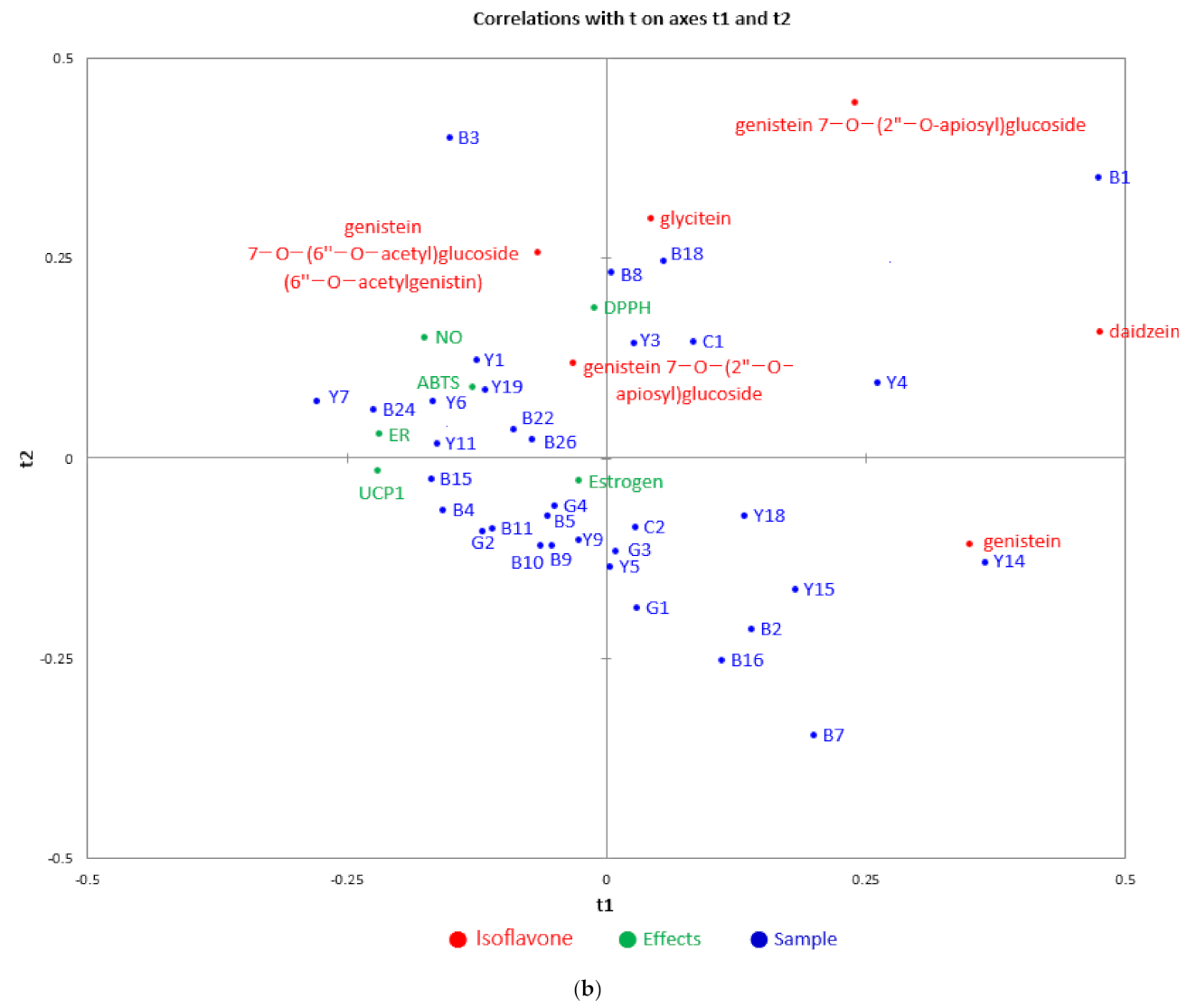
3.3. Correlation Analysis between Physiological Activities and Components of Soybean Samples
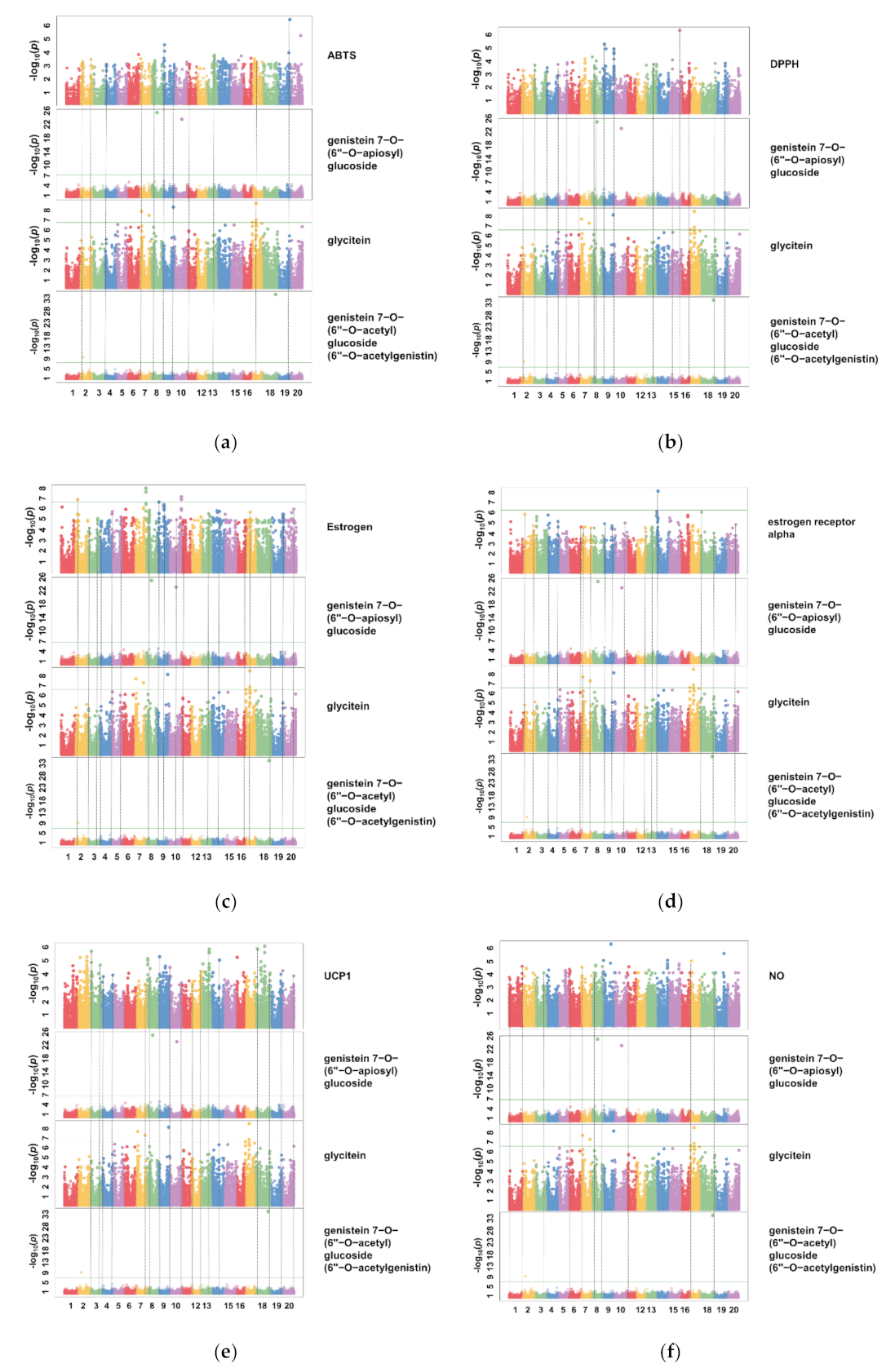
3.4. Identification of the Relationship between Individual Isoflavone Derivatives Having High Correlation with Physiological Activities of Soybean Cultivars, and Genetic Characteristics
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Choi, Y.M.; Yoon, H.M.; Shin, M.J.; Lee, Y.J.; Hur, O.S.; Lee, B.C.; Ha, B.K.; Wang, X.H.; Desta, K.T. Metabolite contents and antioxidant activities of soybean (Glycine max (L.) Merrill) seeds of different seed coat colors. Antioxidants 2021, 10, 1210. [Google Scholar] [CrossRef]
- Choi, Y.M.; Yoon, H.M.; Lee, S.K.; Ko, H.C.; Shin, M.J.; Lee, M.C.; Hur, O.S.; Ro, N.Y.; Desta, K.T. Isoflavones, anthocyanins, phenolic content, and antioxidant activities of black soybeans (Glycine max (L.) Merrill) as affected by seed weight. Sci. Rep. 2020, 10, 19960. [Google Scholar] [CrossRef]
- Lee, S.J.; Kim, J.J.; Moon, H.I.; Ahn, J.K.; Chun, S.C.; Jung, W.S.; Lee, O.K.; Chung, I.M. Analysis of isoflavones and phenolic compounds in Korean soybean [Glycine max (L.) Merrill] seeds of different seed weights. J. Agric. Food Chem. 2008, 56, 2751–2758. [Google Scholar] [CrossRef]
- Lee, S.J.; Kim, H.W.; Lee, S.J.; Na, H.M.; Kwon, R.H.; Kim, J.H.; Yoon, H.M.; Chol, Y.M.; Wee, C.D.; Yoo, S.M.; et al. Characterization of isoflavones from seed of selected soybean (Glycine max L.) resources using high-resolution mass spectrometry. Korean J. Food Nutr. 2020, 33, 655–665. [Google Scholar]
- Somekawa, Y.; Chiguchi, M.; Ishibashi, T.; Aso, T. Soy intake realated to menopause symtoms, serum lipids, and bone mineral density in postmenopausal Japanese women. Obstet. Gynecol. 2001, 97, 109–115. [Google Scholar] [PubMed]
- Taku, K.; Melby, M.K.; Fronenberg, F.; Kurzer, M.S.; Messina, M. Extracted or synthesized soybean isoflavones ruduce menopausal hot flash frequency and severity: Systematic review and meta-analysis of randomized controlled trials. Menopause 2012, 19, 776–790. [Google Scholar] [CrossRef]
- Widowati, W.; Prahastuti, S.; Ekayanti, N.L.W.; Munshy, U.Z.; Kusuma, H.S.W.; Wibowo, S.H.B.; Amalia, A. Widodo, W.S.; Rizal, R. Anti-inflammation assay of black soybean extract and its compounds in lipopolysaccharide-induced RAW 264.7 cell. J. Phys. Conf. Ser. 2019, 1374, 012052. [Google Scholar] [CrossRef]
- Kim, S.Y.; Wi, H.R.; Choi, S.J.; Ha, T.J.; Lee, B.W.; Lee, M.S. Inhibitory effect of anthocyanin-rich black soybean testa Glycine max (L.) Merr.) on the inflammation-induced adipogenesis in a DIO mouse model. J. Funct. Foods 2015, 14, 623–633. [Google Scholar] [CrossRef]
- Basharat, S.; Saeed, A.; Baig, W.; Gilani, S.A.; Ijaz, A.; Abid, F.; Ifthikar, H.; Ansari, I.K. Therapeutic effect of Glycine max (soybean) bioactive components in CVD and obesity. J. Food Nutr. 2020, 6, 1–7. [Google Scholar]
- Woumbo, C.Y.; Kuate, D.; Womeni, H.M. Cooking methods affects phytochemical composition and anti-obesity potential of soybean (Glycine max) seeds in Wistar rats. Heliyon 2017, 3, e00382. [Google Scholar] [CrossRef] [Green Version]
- Jang, H.H.; Kim, H.W.; Kim, S.Y.; Kim, S.M.; Kim, J.B.; Lee, Y.M. In vitro and in vivo hypoglycemic effects of cyanidin 3-caffeoyl-p-hydroxybenzoylsophoroside-5-glucose, an anthocyanin isolated from purple-fleshed sweet potato. Food Chem. 2019, 272, 688–693. [Google Scholar] [CrossRef]
- Zhang, E.J.; Ng, K.M.; Luo, K.Q. Extraction and purification of isoflavones from soybeans and characterization of their estrogenic activities. J. Agric. Food Chem. 2007, 55, 6940–6950. [Google Scholar] [CrossRef]
- Lee, S.H.; Jeong, Y.S.; Song, J.; Hwang, K.A.; Noh, G.M.; Hwang, I.G. Chemical composition and physiological activity of Opuntia ficus-indica depending on different cultivation regions. Korean J. Food Nutr. 2016, 29, 521–528. [Google Scholar] [CrossRef] [Green Version]
- Park, J.H.; Lee, S.R. Anti-inflammatory activities of Scolopendra subspinipes mutilans in RAW 264.7 cells. J. Nutr. Health 2018, 8, 323–329. [Google Scholar] [CrossRef]
- Lee, Y.G.; Jeong, N.H.; Kim, J.H.; Lee, K.H.; Kim, K.H.; Pirani, A.; Ha, B.K.; Kang, S.T.; Park, B.S.; Moon, J.K.; et al. Development, validation and genetic analysis of a large soybean SNP genotyping array. Plant J. 2015, 81, 625–636. [Google Scholar] [CrossRef]
- Cho, K.M.; Ha, T.J.; Lee, Y.B.; Seo, W.D.; Kim, J.Y.; Ryu, H.W.; Jeong, S.H.; Kang, Y.M.; Lee, J.H. Soluble phenolics and antioxidant properties of soybean (Glycine max L.) cultivars with varying seed coat colours. J. Funct. Foods 2013, 5, 1065–1076. [Google Scholar] [CrossRef]
- Choi, E.M.; Yoon, H.M.; Shin, M.J.; Lee, Y.J.; Lee, S.K.; Hur, O.S.; Ro, N.Y.; Ko, H.C.; Yi, J.Y.; Lee, S.H.; et al. Differences in cotyledon color and harvest period affect the contents of major isoflavones and anthocyanins in black soybeans. Plant Breed. Biotechnol. 2021, 9, 65–76. [Google Scholar] [CrossRef]
- Ji, Y.M.; Kim, M.Y.; Lee, S.H.; Jang, G.Y.; Li, M.; Yoon, N.; Kim, K.M.; Lee, J.; Jeong, H.S. Effects of acidic treatment for abthocyanin and proanthocyanidin extraction on black bean (Glycine max Merrill. ) J. Korean Soc. Food Sci. Nutr. 2015, 44, 1594–1598. [Google Scholar] [CrossRef]
- Bae, J.Y.; Kim, H.J.; Park, W.S.; Ahn, M.J. Estrogenic activity of leguminosae species in Korea using MCF-7 cells. Korean J. Pharmacogn. 2021, 52, 118–125. [Google Scholar]
- Kim, I.S. Current perspectives on the beneficial effects of soybean isoflavones and their metabolites for humans. Antioxidants 2021, 10, 1064. [Google Scholar] [CrossRef]
- Wang, D.F.; Zhang, N.Y.; Peng, Y.Z.; Qi, D.S. Interaction of zearalenone and soybean isoflavone on the development of reproductive organs, reproductive hormones and estrogen receptor expression in prepubertal gilts. Anim. Reprod. Sci. 2010, 122, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Guilherme, A.; Virbasius, J.V.; Puri, V.; Czech, M.P. Adipocyte dysfunctions linking obesity to insulin resistance and type 2 diabetes. Nat. Rev. Mol. Cell Biol. 2008, 9, 367–377. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bartelt, A.; Heeren, J. Adipose tissue browning and metabolic health. Nat. Rev. Endocrinol. 2014, 10, 24–36. [Google Scholar] [CrossRef]
- Saito, M. High incidence of metabolically active brown adipose tissue in healthy adult humans: Effects of cold exposure and adiposity. Diabetes 2009, 58, 1531–1626. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ryu, H.S.; Lee, M.J.; Ham, J.R.; Choi, R.Y.; Lee, H.I.; Lee, H.J.; Son, Y.J.; Lee, M.K. Anti-inflammatory effects soybean embryo extract in LPS-Stimulated RAW264.7 macrophages. J. Korean Soc. Food Sci. Nutr. 2019, 48, 1303–1309. [Google Scholar] [CrossRef]
- Lee, H.J.; Do, W.N.; Kim, Y.H. In vitro biological activities of anthocyanin crude extracts from black soybean. Korean J. Crop Sci. 2010, 55, 65–69. [Google Scholar]
- Choi, J.H.; Jun, S.H. AI technology analysis using partial least square regression. J. Korea Soc. Comput. Inf. 2020, 25, 109–115. [Google Scholar]
- Nordborg, M.; Weigel, D. Next-generation genetics in plants. Nature 2008, 456, 720–723. [Google Scholar] [CrossRef] [PubMed]
- Mandel, J.R.; Nambeesan, S.; Bowers, J.E.; Marek, L.F.; Ebert, D.; Rieseberg, L.H.; Knapp, S.J.; Burke, J.M. Association mapping and the genomic consequences of selection in sunflower. PLoS Genet. 2013, 9, e1003378. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khan, S.U.; Saeed, S.; Khan, M.H.U.; Fan, C.; Ahmar, S.; Arriagada, O.; Shahzad, R.; Branca, F.; Mora-Poblete, F. Advances and Challenges for QTL Analysis and GWAS in the Plant-Breeding of High-Yielding: A Focus on Rapeseed. Biomolecules 2021, 11, 1516. [Google Scholar] [CrossRef]
- Han, Z.; Zhang, J.; Cai, S.; Chen, X.; Quan, X.; Zhang, G. Association mapping for total polyphenol content, total flavonoid content and antioxidant activity in barley. BMC Genom. 2018, 19, 81. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Seed Coat Color | Code | Introduction Number | Name | Origin | Cultivar Type |
|---|---|---|---|---|---|
| Control | C1(Yellow) | IT 212859 | Daewon | Korea | Control cultivar |
| C2(Black) | IT 213192 | Cheongja 2 | Korea | Control cultivar | |
| Yellow | Y1 | IT 024099 | YJ208-1 | Korea | Landrace |
| Y2 | IT 104690 | Kongnamul Kong | Korea | Landrace | |
| Y3 | IT 113218 | Kongnamul Kong | Korea | Landrace | |
| Y4 | IT 153844 | KLS 87248 | Korea | Landrace | |
| Y5 | IT 155963 | Nongrim 51 | Japan | Variety | |
| Y6 | IT 171080 | PI 467319 | China | Variety | |
| Y7 | IT 195514 | Jang Kong | Korea | Landrace | |
| Y8 | IT 219581 | Myungjunamul Kong | Korea | Variety | |
| Y9 | IT 229418 | Danmi 2 | Korea | Variety | |
| Y10 | IT 229421 | Hoseo | Korea | Variety | |
| Y11 | IT 231360 | Kongnamul Kong | Korea | Landrace | |
| Y12 | IT 263155 | Sinpaldalkong 2 | Korea | Variety | |
| Y13 | IT 263167 | Uram | Korea | Variety | |
| Y14 | IT 263852 | Chungnamyeongi-1997-3 | Korea | Landrace | |
| Y15 | IT 269982 | Milyang 247 | Korea | Breeding line | |
| Y16 | IT 270002 | Jungmo 3008 | Korea | Variety | |
| Y17 | IT 274571 | GNU-2007-14613 | Korea | Landrace | |
| Y18 | IT 274592 | GNU-2007-14723 | Korea | Landrace | |
| Y19 | IT 324099 | CS 00728 | China | Breeding line | |
| Black | B1 | IT 021665 | PI 90763 | China | Landrace |
| B2 | IT 143347 | KLS86185 | Korea | Landrace | |
| B3 | IT 161904 | PI 84578 | Korea | Landrace | |
| B4 | IT 177271 | Geomjeong Kong-5 | Korea | Landrace | |
| B5 | IT 177573 | Geomjeong Kong-5 | Korea | Landrace | |
| B6 | IT 177709 | Geomjeong Kong-4 | Korea | Landrace | |
| B7 | IT 178054 | Geomjeong Kong-1 | Korea | Landrace | |
| B8 | IT 186183 | Kongnamul Kong | Korea | Landrace | |
| B9 | IT 189215 | 94yuja4 | Korea | Landrace | |
| B10 | IT 194560 | Geomjeong Kong | Korea | Landrace | |
| B11 | IT 212805 | Chungnamseocheon-1999-98 | Korea | Landrace | |
| B12 | IT 224192 | Jeonbukgunsansujip | Korea | Landrace | |
| B13 | IT 228822 | 409 | Korea | Landrace | |
| B14 | IT 231544 | Jwinuni Kong | Korea | Landrace | |
| B15 | IT 239896 | Jwineori Kong | Korea | Landrace | |
| B16 | IT 252252 | Neoljeokseoritae | Korea | Landrace | |
| B17 | IT 252748 | 294 | Korea | Landrace | |
| B18 | IT 252768 | 326 | Korea | Landrace | |
| B19 | IT 263853 | Geomen Kong | Korea | Landrace | |
| B20 | IT 274515 | GNU-2007-14502 | Korea | Landrace | |
| B21 | IT 275005 | 197 | Korea | Landrace | |
| B22 | IT 308619 | Junyeori Kong | Korea | Landrace | |
| B23 | IT 311261 | Jwinuni Kong | Korea | Landrace | |
| B24 | K 137773 | Heugseong | Korea | Variety | |
| B25 | IT 194558 | Geomjeong Kong | Korea | Landrace | |
| Green–Black | G1 | IT 154351 | KAS579-1 | Korea | Landrace |
| G2 | IT 154724 | KAS651-21 | Korea | Landrace | |
| G3 | IT 178160 | Geomjeong Kong | Korea | Landrace | |
| G4 | IT 186048 | Gangwonyanggu-1994-3709 | Korea | Landrace |
| Seed Coat Color | Code | ABTS (mg AA eq/g) | DPPH (mg AA eq/g) |
|---|---|---|---|
| Control cultivar | C1 | 4.84 ± 1.1 e–i | 2.43 ± 0.52 b–i |
| C2 | 5.04 ± 1.32 d–h | 1.32 ± 0.50 d–j | |
| Range | 4.84–5.04 | 1.32–2.43 | |
| Mean | 4.94 | 1.88 | |
| Yellow | Y1 | 2.25 ± 0.83 m–o | 1.20 ± 0.14 d–j |
| Y2 | 1.92 ± 0.98 n–o | 0.99 ± 0.13 f–j | |
| Y3 | 2.47 ± 1.05 l–o | 1.30 ± 0.05 d–j | |
| Y4 | 3.24 ± 0.71 h–o | 1.50 ± 0.23 c–j | |
| Y5 | 2.27 ± 0.80 m–o | 1.01 ± 0.70 f–j | |
| Y6 | 1.60 ± 0.43 o | 0.64 ± 0.26 h–j | |
| Y7 | 3.83 ± 0.56 g–n | 2.04 ± 0.39 b–j | |
| Y8 | 3.15 ± 0.57 h–o | 1.41 ± 0.29 c–j | |
| Y9 | 2.87 ± 0.87 j–o | 0.67 ± 0.75 h–j | |
| Y10 | 3.27 ± 0.76 h–o | 0.97 ± 0.85 f–j | |
| Y11 | 5.64 ± 1.38 d–g | 1.38 ± 0.86 d–j | |
| Y12 | 4.18 ± 0.64 f–m | 2.43 ± 0.59 b–i | |
| Y13 | 2.64 ± 0.63 k–o | 1.12 ± 0.27 e–j | |
| Y14 | 5.52 ± 1.71 d–g | 3.10 ± 0.40 b–d | |
| Y15 | 3.22 ± 0.59 h–o | 2.30 ± 0.27 b–i | |
| Y16 | 2.76 ± 0.42 k–o | 1.38 ± 0.21 d–j | |
| Y17 | 4.51 ± 1.16 e–k | 2.00 ± 2.19 b–j | |
| Y18 | 4.53 ± 1.08 e–k | 2.29 ± 2.52 b–i | |
| Y19 | 6.25 ± 0.93 c–e | 3.41 ± 2.16 b | |
| Range | 1.60–6.25 | 0.64–3.41 | |
| Mean | 3.48 | 1.64 | |
| Black | B1 | 4.49 ± 0.67 e–k | 2.40 ± 0.25 b–i |
| B2 | 2.01 ± 0.60 n–o | 0.88 ± 0.16 g–j | |
| B3 | 1.83 ± 0.77 o | 0.28 ± 0.39 j | |
| B4 | 2.49 ± 0.58 l–o | 0.83 ± 0.27 g–j | |
| B5 | 2.76 ± 1.27 k–o | 0.58 ± 0.62 i–j | |
| B6 | 4.11 ± 0.66 f–m | 1.80 ± 0.38 b–j | |
| B7 | 2.35 ± 0.19 l–o | 1.02 ± 0.37 f–j | |
| B8 | 4.02 ± 0.31 f–m | 2.27 ± 0.43 b–i | |
| B9 | 3.21 ± 0.82 h–o | 1.35 ± 031 d–j | |
| B10 | 4.08 ± 0.65 f–m | 1.96 ± 0.24 b–j | |
| B11 | 5.23 ± 1.06 d–g | 2.35 ± 0.40 b–i | |
| B12 | 2.93 ± 0.81 i–o | 1.66 ± 0.69 b–j | |
| B13 | 4.78 ± 0.71 e–j | 1.71 ± 0.43 b–j | |
| B14 | 4.87 ± 1.22 e–i | 1.25 ± 0.99 d–j | |
| B15 | 8.34 ± 1.30 b | 3.02 ± 1.04 b–e | |
| B16 | 4.26 ± 1.38 f–l | 0.77 ± 0.43 g–j | |
| B17 | 11.93 ± 1.08 a | 6.20 ± 0.35 a | |
| B18 | 4.82 ± 0.63 e–i | 2.84 ± 0.60 b–f | |
| B19 | 3.82 ± 0.63 g–n | 1.79 ± 0.35 b–j | |
| B20 | 7.42 ± 1.76 bc | 3.53 ± 0.72 b | |
| B21 | 5.78 ± 1.11 c–f | 2.22 ± 1.98 b–i | |
| B22 | 4.71 ± 1.24 e–j | 1.31 ± 1.96 d–j | |
| B23 | 7.98 ± 1.49 b | 3.42 ± 2.16 b | |
| B24 | 6.77 ± 1.03 b–d | 3.29 ± 1.22 bc | |
| B25 | 4.71 ± 0.96 e–j | 2.57 ± 0.72 b–h | |
| Range | 1.83–11.93 | 0.28–6.20 | |
| Mean | 4.79 | 2.05 | |
| Green–Black | G1 | 2.39 ± 1.08 l–o | 1.38 ± 0.14 d–j |
| G2 | 1.91 ± 0.92 n–o | 0.67 ± 0.96 h–j | |
| G3 | 2.71 ± 0.45 k–o | 1.48 ± 0.65 c–j | |
| G4 | 5.66 ± 1.29 d–g | 2.63 ± 1.80 b–g | |
| Range | 1.91–5.66 | 0.67–2.63 | |
| Mean | 3.17 | 1.54 |
| Traits | Count | |||||
|---|---|---|---|---|---|---|
| ABTS | DPPH | Estrogen | ER-α | UCP-1 | NO | |
| Genistein 7-O-(2″-O-apiosyl)glucoside | 67 | 22 | 33 | 73 | 66 | 42 |
| Genistein 7-O-(6″-O-acetyl)glucoside (6″-O-acetylgenistin) | 4 | 2 | 29 | 21 | 43 | 14 |
| Glycitein | 36 | 30 | 113 | 367 | 185 | 209 |
| Total | 107 | 54 | 175 | 461 | 294 | 265 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chu, H.-N.; Lee, S.-J.; Wang, X.; Lee, S.-H.; Yoon, H.-M.; Hwang, Y.-J.; Jung, E.-S.; Kwon, Y.; Wee, C.-D.; Jang, K.-A.; et al. A Correlation Study on In Vitro Physiological Activities of Soybean Cultivars, 19 Individual Isoflavone Derivatives, and Genetic Characteristics. Antioxidants 2021, 10, 2027. https://doi.org/10.3390/antiox10122027
Chu H-N, Lee S-J, Wang X, Lee S-H, Yoon H-M, Hwang Y-J, Jung E-S, Kwon Y, Wee C-D, Jang K-A, et al. A Correlation Study on In Vitro Physiological Activities of Soybean Cultivars, 19 Individual Isoflavone Derivatives, and Genetic Characteristics. Antioxidants. 2021; 10(12):2027. https://doi.org/10.3390/antiox10122027
Chicago/Turabian StyleChu, Han-Na, Su-Ji Lee, Xiaohan Wang, Sang-Hoon Lee, Hye-Myeong Yoon, Yu-Jin Hwang, Eun-Suk Jung, Yongseok Kwon, Chi-Do Wee, Kyeong-A Jang, and et al. 2021. "A Correlation Study on In Vitro Physiological Activities of Soybean Cultivars, 19 Individual Isoflavone Derivatives, and Genetic Characteristics" Antioxidants 10, no. 12: 2027. https://doi.org/10.3390/antiox10122027
APA StyleChu, H.-N., Lee, S.-J., Wang, X., Lee, S.-H., Yoon, H.-M., Hwang, Y.-J., Jung, E.-S., Kwon, Y., Wee, C.-D., Jang, K.-A., & Kim, H.-R. (2021). A Correlation Study on In Vitro Physiological Activities of Soybean Cultivars, 19 Individual Isoflavone Derivatives, and Genetic Characteristics. Antioxidants, 10(12), 2027. https://doi.org/10.3390/antiox10122027






