Effects of Sub-Chronic Exposure to Imidacloprid on Reproductive Organs of Adult Male Rats: Antioxidant State, DNA Damage, and Levels of Essential Elements
Abstract
1. Introduction
2. Materials and Methods
2.1. Tested Compound
2.2. Animals and Animal Husbandry
2.3. Experimental Design
2.4. Collection of Testes and Epididymis
2.5. Determination of the Antioxidant Status
2.6. Multi-Element Analysis
2.7. Evaluation of DNA Damage
2.8. Statistical Analysis
3. Results
3.1. Changes in Weight of Testes and Epididymis
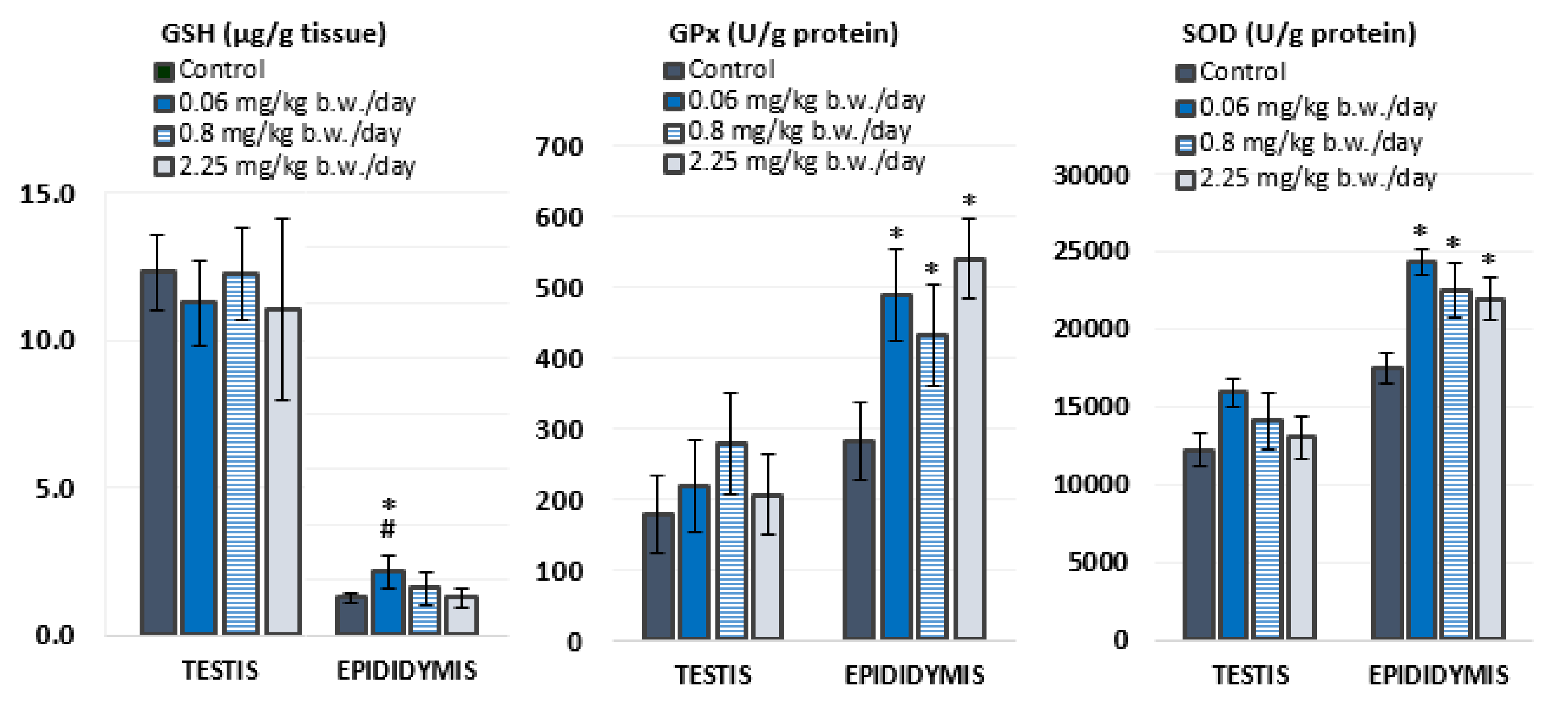
3.2. Antioxidant Status
3.3. Concentration of Essential Elements in Testis and Epididymis
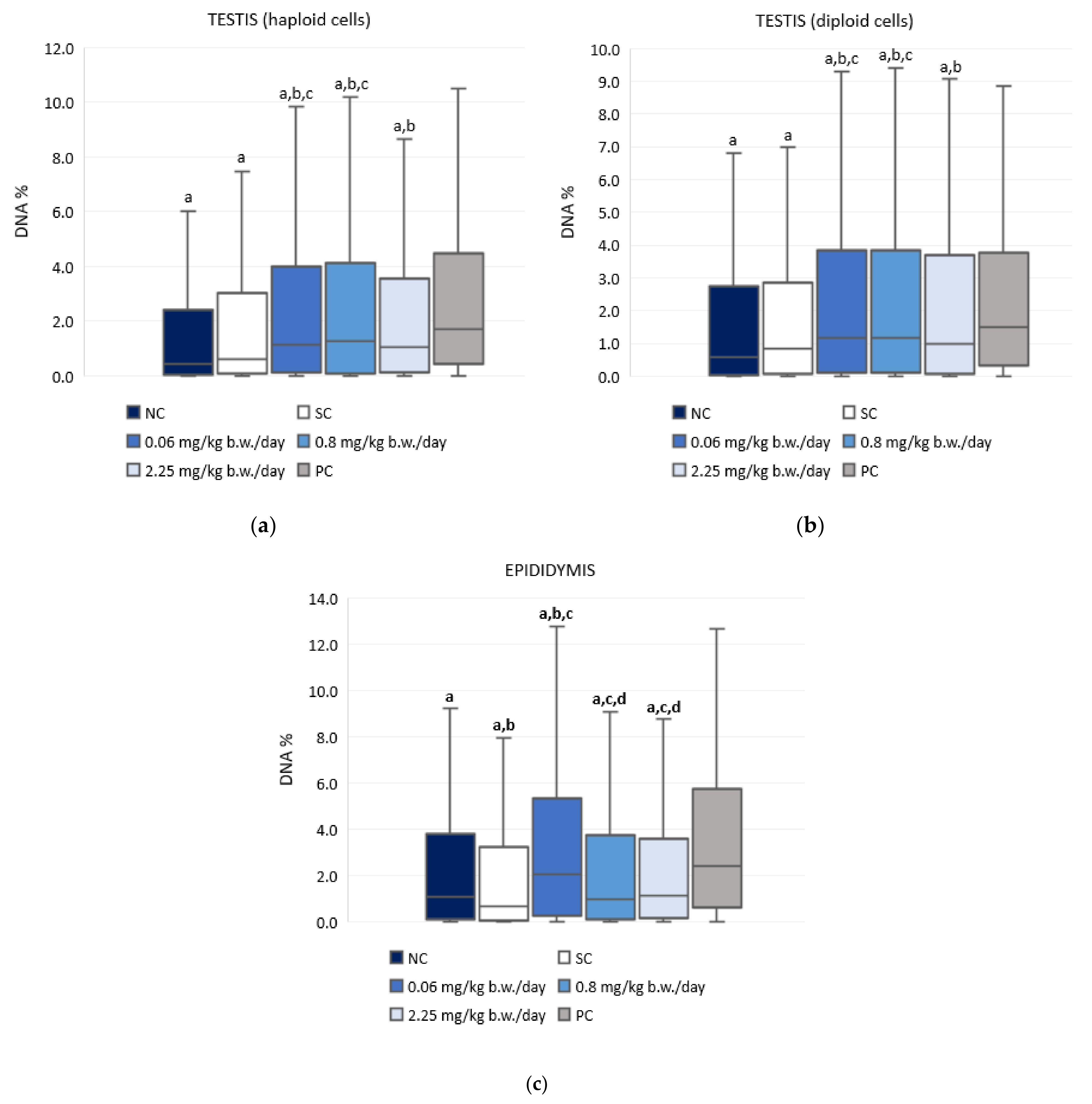
3.4. The Alkaline Comet Assay
4. Discussion
4.1. Effect on Weight of Testis and Epididymis
4.2. Oxidative Stress Response
4.3. Effect on Levels of Essential Elements
4.4. Genotoxic Effects of Exposure to Imidacloprid
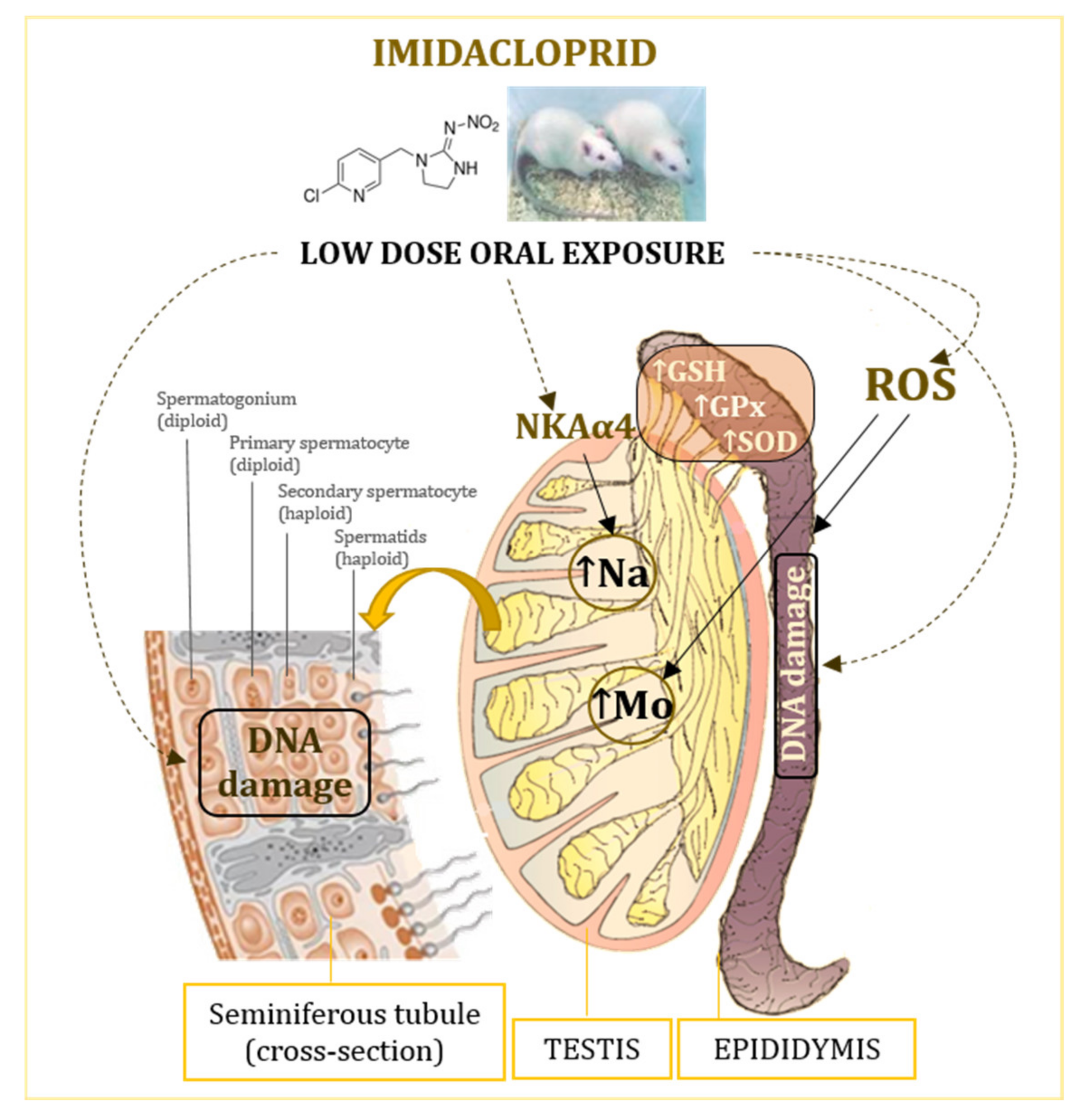
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Craddock, H.A.; Huang, D.; Turner, P.C.; Quirós-Alcalá, L.; Payne-Sturges, D.C. Trends in neonicotinoid pesticide residues in food and water in the United States, 1999–2015. Environ. Health 2019, 18, 7. [Google Scholar] [CrossRef] [PubMed]
- Thompson, D.A.; Lehmler, H.-J.; Kolpin, D.W.; Hladik, M.L.; Vargo, J.D.; Schilling, K.E.; LeFevre, G.H.; Peeples, T.L.; Poch, M.C.; LaDuca, L.E.; et al. A critical review on the potential impacts of neonicotinoid insecticide use: Current knowledge of environmental fate, toxicity, and implications for human health. Environ. Sci. Processes Impacts 2020, 22, 1315–1346. [Google Scholar] [CrossRef]
- US EPA. Imidacloprid—Proposed Interim Registration Review Decision Case Number 7605. 2020. Available online: https://www.epa.gov/sites/default/files/2020-01/documents/imidacloprid_pid_signed_1.22.2020.pdf (accessed on 20 August 2021).
- Tomizawa, M.; Casida, J.E. Neonicotinoid insecticide toxicology: Mechanisms of selective action. Annu Rev. Pharmacol. Toxicol. 2005, 45, 247–268. [Google Scholar] [CrossRef] [PubMed]
- Abu Zeid, E.H.; Alam, R.T.M.; Ali, S.A.; Hendawi, M.Y. Dose-related impacts of imidacloprid oral intoxication on brain and liver of rock pigeon (Columba livia domestica), residues analysis in different organs. Ecotoxicol. Environ. Saf. 2019, 167, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Bal, R.; Naziroğlu, M.; Türk, G.; Yilmaz, Ö.; Kuloğlu, T.; Etem, E.; Baydas, G. Insecticide imidacloprid induces morphological and DNA damage through oxidative toxicity on the reproductive organs of developing male rats. Cell Biochem. Funct. 2012, 30, 492–499. [Google Scholar] [CrossRef]
- Bal, R.; Türk, G.; Tuzcu, M.; Yilmaz, Ö.; Kuloğlu, T.; Gundogdu, R.; Gür, S.; Agca, A.; Ulas, M.; Çambay, Z.; et al. Assessment of imidacloprid toxicity on reproductive organ system of adult male rats. J. Environ. Sci. Health B 2012, 47, 434–444. [Google Scholar] [CrossRef]
- Wang, X.; Anadón, A.; Wu, Q.; Qiao, F.; Ares, I.; Martínez-Larrañaga, M.R.; Yuan, Z.; Martínez, M.A. Mechanism of Neonicotinoid Toxicity: Impact on Oxidative Stress and Metabolism. Annu. Rev. Pharmacol. Toxicol. 2018, 58, 471–507. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Shen, Q.; Zeng, T.; Li, J.; Li, W.; Wang, Y. Enrichment of imidacloprid and its metabolites in lizards and its toxic effects on gonads. Environ. Pollut. 2020, 258, 1137. [Google Scholar] [CrossRef]
- Mikolić, A.; Brčić Karačonji, I. Imidacloprid as reproductive toxicant and endocrine disruptor: Investigations in laboratory animals. Arh Hig Rada Toksikol 2018, 69, 103–108. [Google Scholar] [CrossRef]
- Hafez, E.M.; Issa, S.Y.; Al-Mazroua, M.K.; Ibrahim, K.T.; Rahman, S.M.A. The neonicotinoid insecticide imidacloprid: A male reproductive system toxicity inducer-human and experimental study. Toxicol. Open Access 2016, 2, 1000109. [Google Scholar] [CrossRef]
- Lonare, M.; Kumar, M.; Raut, S.; More, A.; Doltade, S.; Badgujar, P.; Telang, A. Evaluation of ameliorative effect of curcumin on imidacloprid-induced male reproductive toxicity in Wistar rats. Environ. Toxicol. 2016, 10, 1250–1263. [Google Scholar] [CrossRef]
- Najafi, G.; Razi, M.; Hoshyar, A.; Shahmohamadloo, S.; Feyzi, S. The effect of chronic exposure with imidacloprid insecticide on fertility in mature male rats. Int. J. Fertil. Steril 2010, 4, 9–16. [Google Scholar] [CrossRef]
- Saber, T.M.; Arisha, A.H.; Abo-Elmaaty, A.M.A.; Abdelgawad, F.E.; Metwally, M.; Saber, T.; Mansour, M.F. Thymol alleviates imidacloprid-induced testicular toxicity by modulating oxidative stress and expression of steroidogenesis and apoptosis-related genes in adult male rats. Ecotoxicol. Environ. Saf. 2021, 221, 112435. [Google Scholar] [CrossRef]
- Aitken, R.J.; Smith, T.B.; Jobling, M.S.; Baker, M.A.; De Iuliis, G.N. Oxidative stress and male reproductive health. Asian J. Androl. 2014, 16, 31–38. [Google Scholar] [CrossRef] [PubMed]
- O’Flaherty, C. Orchestrating the antioxidant defenses in the epididymis. Andrology 2019, 7, 662–668. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Sharma, B. Pesticides induced oxidative stress in mammalian systems: A review. Int. J. Biol. Med. Res. 2010, 1, 90–104. Available online: https://www.researchgate.net/publication/202037053. (accessed on 25 August 2021).
- Aitken, R.J.; Roman, S.D. Antioxidant systems and oxidative stress in the testes. Oxid. Med. Cell Longev. 2008, 1, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Baskaran, S.; Finelli, R.; Agarwal, A.; Henkel, R. Reactive oxygen species in male reproduction: A boon or a bane? Andrologia 2021, 53, e13577. [Google Scholar] [CrossRef] [PubMed]
- Valko, M.; Leibfritz, D.; Moncol, J.; Cronin, M.T.D.; Mazur, M.; Telser, J. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell B 2007, 39, 44–84. [Google Scholar] [CrossRef] [PubMed]
- Pizzorno, J. Glutathione! Integr. Med. 2014, 13, 8–12. [Google Scholar]
- Pasqualotto, F.F.; Sharma, R.K.; Kobayashi, H.; Nelson, D.R.; Thomas, A.J., Jr.; Agarwal, A. Oxidative stress in normospermic men undergoing infertility evaluation. J. Androl. 2001, 22, 316–322. [Google Scholar] [CrossRef] [PubMed]
- Vernet, P.; Aitken, R.J.; Drevet, J.R. Antioxidant strategies in the epididymis. Mol. Cell Endocrinol. 2004, 216, 31–39. [Google Scholar] [CrossRef]
- Mahajan, L.; Verma, P.K.; Raina, R.; Sood, S. Potentiating effect of imidacloprid on arsenic-induced testicular toxicity in Wistar rats. BMC Pharmacol. Toxicol. 2018, 19, 48. [Google Scholar] [CrossRef] [PubMed]
- Tetsatsi, A.C.M.; Nkeng-Effouet, P.A.; Alumeti, D.M.; Bonsou, G.R.F.; Bonsou, F.; Kamanyi, A.; Watcho, P. Colibri® insecticide induces male reproductive toxicity: Alleviating effects of Lannea acida (Anacardiaceae) in rats. Basic Clin. Androl. 2019, 29, 16. [Google Scholar] [CrossRef]
- Bianchi, J.; Cabral-de-Mello, D.C.; Marin-Morales, M.A. Toxicogenetic effects of low concentrations of the pesticides imidacloprid and sulfentrazone individually and in combination in in vitro tests with HepG2 cells and Salmonella typhimurium. Ecotoxicol. Environ. Saf. 2015, 120, 174–183. [Google Scholar] [CrossRef] [PubMed]
- Calderón-Segura, M.E.; Gómez-Arroyo, S.; Villalobos-Pietrini, R.; Martínez-Valenzuela, C.; Carbajal-López, Y.; Calderón-Ezquerro, M.C.; Cortés-Eslava, J.; García-Martínez, R.; Flores-Ramírez, D.; Rodríguez-Romero, M.I.; et al. Evaluation of genotoxic and cytotoxic effects in human peripheral blood lymphocytes exposed in vitro to neonicotinoid insecticides news. J. Toxicol. 2012, 2012, 612647. [Google Scholar] [CrossRef]
- Costa, C.; Silvari, V.; Melchini, A.; Catania, S.; Heffron, J.J.; Trovato, A.; De Pasquale, R. Genotoxicity of imidacloprid in relation to metabolic activation and composition of the commercial product. Mutat. Res. 2009, 672, 40–44. [Google Scholar] [CrossRef]
- Feng, S.; Kong, Z.; Wang, X.; Peng, P.; Zeng, E.Y. Assessing the genotoxicity of imidacloprid and RH-5849 in human peripheral blood lymphocytes in vitro with comet assay and cytogenetic tests. Ecotoxicol. Environ. Saf. 2005, 61, 239–246. [Google Scholar] [CrossRef]
- Katić, A.; Kašuba, V.; Kopjar, N.; Tariba Lovaković, B.; Marjanović Čermak, A.M.; Mendaš, G.; Micek, V.; Milić, M.; Pavičić, I.; Pizent, A.; et al. Effects of low-level imidacloprid oral exposure on cholinesterase activity, oxidative stress responses, and primary DNA damage in the blood and brain of male Wistar rats. Chem. Biol. Interact. 2021, 338, 109287. [Google Scholar] [CrossRef] [PubMed]
- Ge, W.; Yan, S.; Wang, J.; Zhu, L.; Chen, A.; Wang, J. Oxidative stress and DNA damage induced by imidacloprid in Zebrafish (Danio rerio). J. Agric. Food Chem. 2015, 63, 1856–1862. [Google Scholar] [CrossRef]
- Fenech, M. Micronutrients and genomic stability: A new paradigm for recommended dietary allowances (RDAs). Food Chem. Toxicol. 2002, 40, 1113–1117. [Google Scholar] [CrossRef]
- EFSA Scientific Report. Conclusion regarding the peer review of the pesticide risk assessment of the active substance imidacloprid. EFSA J. 2008, 148, 1–120. [Google Scholar]
- World Health Organisation (WHO), International Programme on Chemical Safety (IPCS). Toxicological Evaluations: Imidacloprid. 2001. Available online: http://www.inchem.org/jmpr/jmprmono/2001pr07.htm (accessed on 5 October 2018).
- European Comission. EU Pesticides Database, 2016. Imidacloprid. Available online: https://ec.europa.eu/food/plant/pesticides/eu-pesticidesdatabase/public/?event=activesubstance.detail&language=EN&selectedID=1473 (accessed on 5 October 2018).
- Pant, K.; Springer, S.; Bruce, S.; Lawlor, T.; Hewitt, N.; Aardema, M.J. Vehicle and positive control values from the in vivo rodent comet assay and biomonitoring studies using human lymphocytes: Historical database and influence of technical aspects. Environ. Mol. Mutagen. 2014, 55, 633–642. [Google Scholar] [CrossRef] [PubMed]
- Belsten, J.L.; Wright, A.J. European Community-FLAIR common assay for whole-blood glutathione peroxidase (GSH-Px); Results of an inter-laboratory trial. Eur. J. Clin. Nutr. 1995, 49, 921–927. [Google Scholar]
- Belsten, J.L.; Wright, A.J. European community: FLAIR common assay for erythrocyte superoxide dismutase (SOD); Results of an interlaboratory trial. Eur. J. Clin. Nutr. 1995, 49, 928–931. [Google Scholar] [PubMed]
- Ellman, G.L. A colorimetric method for determining low concentrations of mercaptans. Arch. Biochem. Biophys. 1958, 74, 443–450. [Google Scholar] [CrossRef]
- Angelis, K.J.; Dusinska, M.; Collins, A.R. Single cell gel electrophoresis: Detection of DNA damage at different levels of sensitivity. Electrophoresis 1999, 20, 2133–2138. [Google Scholar] [CrossRef]
- Kusakabe, H.; Tateno, H. Characterization of chromosomal damage accumulated in freeze-dried mouse spermatozoa preserved under ambient and heat stress conditions. Mutagenesis 2011, 26, 447–453. [Google Scholar] [CrossRef] [PubMed]
- Tateno, H.; Kamiguchi, Y. Abnormal chromosome migration and chromosome aberrations in mouse oocytes during meiosis II in the presence of topoisomerase II inhibitor ICRF-193. Mutat. Res. 2002, 502, 1–9. [Google Scholar] [CrossRef]
- Singh, N.P.; McCoy, M.T.; Tice, R.R.; Schneider, E.L. A simple technique for quantization of low levels of DNA damage in individual cells. Exp. Cell Res. 1988, 175, 184–191. [Google Scholar] [CrossRef]
- Anderson, D.; Dhawan, A.; Yu, T.-W.; Plewa, M.J. An investigation of bone marrow and testicular cells in vivo using the comet assay. Mutat. Res. 1996, 370, 159–174. [Google Scholar] [CrossRef]
- Kasson, B.G.; Hsueh, A.J. Nicotinic cholinergic agonists inhibit androgen biosynthesis by cultured rat testicular cells. Endocrinology 1985, 117, 1874–1880. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.-Y.; Lee, T-H. Antioxidant enzymes as redox-based biomarkers: A brief review. BMB Rep. 2015, 48, 200–208. [Google Scholar] [CrossRef]
- Wu, P.Y.; Scarlata, E.; O’Flaherty, C. Long-term adverse effects of oxidative stress on rat epididymis and spermatozoa. Antioxidants 2020, 9, 170. [Google Scholar] [CrossRef]
- Dacheux, J.-L.; Gatti, J.L.; Dacheux, F. Contribution of epididymal secretory proteins for spermatozoa maturation. Microsc. Res. Tech. 2003, 61, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Chabory, E.; Damon, E.C.; Lenoir, A.; Henry-Berger, J.; Vernet, P.; Cadet, R.; Saez, F.; Drevet, J.R. Mammalian glutathione peroxidases control acquisition and maintenance of spermatozoa integrity. J. Anim. Sci. 2010, 88, 1321–1331. [Google Scholar] [CrossRef] [PubMed]
- Jervis, K.M.; Robaire, B. Dynamic changes in gene expression along the rat epididymis. Biol. Reprod. 2001, 65, 696–703. [Google Scholar] [CrossRef] [PubMed]
- Hedger, M.P. Immunophysiology and pathology of inflammation in the testis and epididymis. J. Androl. 2011, 32, 625–640. [Google Scholar] [CrossRef]
- Valko, M.; Rhodes, C.J.; Moncol, J.; Izakovic, M.; Mazur, M. Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chem. Biol. Interact. 2006, 160, 1–40. [Google Scholar] [CrossRef]
- Mirnamniha, M.; Faroughi, F.; Tahmasbpoura, E.; Ebrahimi, P.; Harchegani, A.B. An overview on role of some trace elements in human reproductive health, sperm function and fertilization process. Rev. Environ. Health 2019, 34, 339–348. [Google Scholar] [CrossRef]
- Syeda, S.S.; Sánchez, G.; McDermott, J.P.; Hong, K.H.; Blanco, G.; Georg, G.I. The Na+ and K+ transport system of sperm (ATP1A4) is essential for male fertility and an attractive target for male contraception. Biol. Reprod. 2020, 103, 343–356. [Google Scholar] [CrossRef] [PubMed]
- Jimenez, T.; McDermott, J.P.; Sánchez, G.; Blanco, G. Na,K-ATPase α4 isoform is essential for sperm fertility. Proc. Natl. Acad. Sci. USA 2011, 108, 644–649. [Google Scholar] [CrossRef]
- Schwarz, G.; Mendel, R.R.; Ribbe, M.W. Molybdenum cofactors, enzymes and pathways. Nature 2009, 460, 839–847. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.-L.; Liu, F.-J.; Chen, X.-L.; Zhang, Z.-Q.; Shu, R.-Z.; Yu, X.-L.; Zhai, X.-W.; Jin, L.-J.; Ma, X.-G.; Qi, Q.; et al. Dual effects of molybdenum on mouse oocyte quality and ovarian oxidative stress. Syst. Biol. Reprod. Med. 2013, 59, 312–318. [Google Scholar] [CrossRef]
- Lewis, R.C.; Meeker, J.D. Biomarkers of exposure to molybdenum and other metals in relation to testosterone among men from the United States National Health and Nutrition Examination Survey 2011–2012. Fertil. Steril. 2015, 103, 172–178. [Google Scholar] [CrossRef] [PubMed]
- Meeker, J.D.; Rossano, M.G.; Protas, B.; Padmanahban, V.; Diamond, M.P.; Puscheck, E.; Daly, D.; Paneth, N.; Wirth, J.J. Environmental exposure to metals and male reproductive hormones: Circulating testosterone is inversely associated with blood molybdenum. Fertil. Steril. 2010, 93, 130–140. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wang, J.; Wang, G.; Zhu, L.; Wang, J. DNA damage and oxidative stress induced by imidacloprid exposure in the earthworm Eisenia fetida. Chemosphere 2016, 144, 510–517. [Google Scholar] [CrossRef] [PubMed]
- Perez-Iglesias, J.M.; Ruiz de Arcaute, C.; Nikolof, N.; Dury, L.; Soloneski, S.; Natale, G.S.; Larramendy, M.L. The genotoxic efects of the imidacloprid-based insecticide formulation Glacoxan Imida on Montevideo tree frog Hypsiboas pulchellus tadpoles (Anura, Hylidae). Ecotoxicol. Environ. Saf. 2014, 104, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Hassan, A.M.S.; Abo El-Ela, F.I.; Abdel-Aziz, A.M. Investigating the potential protective effects of natural product quercetin against imidacloprid-induced biochemical toxicity and DNA damage in adult rats. Toxicol. Rep. 2019, 6, 727–735. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.M.; Nasr, S.A. Protective effect of broccoli and ferulic acid on imidacloprid-induced neurotoxicity in rats. J. Biomed. Pharm. Res. 2015, 4, 82–89. [Google Scholar] [CrossRef]
- Senyildiz, M.; Kilinc, A.; Ozden, S. Investigation of the genotoxic and cytotoxic effects of widely used neonicotinoid insecticides in HepG2 and SH-SY5Y cells. Toxicol. Ind. Health 2018, 34, 375–383. [Google Scholar] [CrossRef] [PubMed]
- Želježić, D.; Mladinić, M.; Žunec, S.; Lucić Vrdoljak, A.; Kasuba, V.; Tariba, B.; Živković, T.; Marjanović, A.M.; Pavičić, I.; Milic, M.; et al. Cytotoxic, genotoxic and biochemical markers of insecticide toxicity evaluated in human peripheral blood lymphocytes and an HepG2 cell line. Food Chem. Toxicol. 2016, 96, 90–106. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Sharma, R.K.; Sikka, S.C.; Thomas, A.J., Jr.; Falcone, T.; Agarwal, A. Oxidative stress is associated with increased apoptosis leading to spermatozoa DNA damage in patients with male factor infertility. Fertil. Steril. 2003, 80, 531–535. [Google Scholar] [CrossRef]
- Shaposhnikov, S.; Frengen, E.; Collins, A.R. Increasing the resolution of the comet assay using fluorescent in situ hybridization—A review. Mutagenesis 2009, 24, 383–389. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Želježić, D.; Vinković, B.; Kašuba, V.; Kopjar, N.; Milić, M.; Mladinić, M. The effect of insecticides chlorpyrifos, alpha-cypermethrin and imidacloprid on primary DNA damage, TP 53 and c-Myc structural integrity by comet-FISH assay. Chemosphere 2017, 182, 332–338. [Google Scholar] [CrossRef]
- Jia, G.F.; Wang, P.; Qiu, J.; Sun, Y.; Xiao, Y.M.; Zhou, Z.Q. Determination of DNA with imidacloprid by a resonance light scattering technique at nanogram levels and its application. Anal. Lett. 2004, 37, 1339–1354. [Google Scholar] [CrossRef]
- Choucroun, P.; Gillet, D.; Dorange, G.; Sawicki, B.; Dewitte, J.D. Comet assay and early apoptosis. Mutat. Res. Mol. Mech. Mutagen. 2001, 478, 89–96. [Google Scholar] [CrossRef]
- Roser, S.; Pool-Zobel, B.-L.; Rechkemmer, G. Contribution of apoptosis to responses in the comet assay. Mutat. Res. Toxicol. Environ. Mutagen. 2001, 497, 169–175. [Google Scholar] [CrossRef]
- Nair, S.; Engelbrecht, M.; Miles, X.; Ndimba, R.; Fisher, R.; du Plessis, P.; Bolcaen, J.; Niento-Camero, J.; de Kock, E.; Vandervoorde, C. The impact of dose rate on DNA double-strand break formation and repair in human lymphocytes exposed to fast neutron irradiation. Int. J. Mol. Sci. 2019, 20, 5350. [Google Scholar] [CrossRef] [PubMed]
- Commission Implementing Regulation (EU) 2018/783 of 29 May 2018 Amending Implementing Regulation (EU) No 540/2011 as Regards the Conditions of Approval of the Active Substance Imidacloprid. Off. J. Eur. Union 2018, 61, 31–34. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX%3A32018R0783&from=DA (accessed on 7 September 2021).

| Negative Control | 0.06 mg/kg b. w./day | 0.8 mg/kg b. w./day | 2.25 mg/kg b. w./day | ||
|---|---|---|---|---|---|
| TESTIS | |||||
| Na | mg/kg | 1555 (1503–1653) | 1541 (1516–1588) | 1646 (1540–1715) | 1695 (1545–1767) a,b |
| Mg | 198 (171–213) | 195 (176–212) | 201 (179–209) | 201 (168–225) | |
| Ca | 52.3 (49.3–74.8) | 50.3 (44.2–56.7) | 51.9 (48.7–60.5) | 54.0 (49.7–85.2) | |
| K | 4282 (3805–4420) | 4272 (3798–4597) | 4378 (3819–4447) | 4311 (3661–4557) | |
| Fe | 24.2 (21.1–26.8) | 24.9 (22.9–26.8) | 25.0 (24.6–26.2) | 24.4 (21.5–30.6) | |
| Cu | 2.14 (1.85–2.25) | 2.01 (1.88–2.23) | 2.04 (2.00–2.11) | 2.08 (1.67–2.30) | |
| Zn | 27.7 (25.9–28.5) | 27.3 (24.9–29.8) | 27.8 (25.1–28.9) | 27.9 (18.6–32.1) | |
| Se | μg/kg | 966 (940–996) | 925 (859–1021) | 965 (852–997) | 965 (671–1162) |
| Mo | 60.6 (51.3–72.4) | 59.0 (55.8–65.0) | 76.5 (58.8–76.9) a,b | 72.5 (63.2–88.2) a,b | |
| Mn | 331 (295–376) | 321 (300–381) | 327 (317–341) | 337 (272–477) | |
| EPIDIDYMIS | |||||
| Na | mg/kg | 1038 (985–1292) | 1416 (971–1472) | 1331 (1210–2182) | 1338 (1209–1463) |
| Mg | 110 (76.3–128) | 129 (83.7–174) | 128 (90.8–234) | 114 (98.3–135) | |
| Ca | 72.7 (51.4–99.2) | 65.4 (61.6–72.6) | 79.8 (64.5–90.5) | 66.5 (60.1–102) | |
| K | 1469 (1172–1794) | 1875 (1237–2154) | 1724 (1396–3291) | 1620 (1544–1911) | |
| Fe | 18.2 (17.6–21.0) | 19.1 (16.9–21.0) | 18.3 (16.3–29.8) | 19.9 (17.1–28.9) | |
| Cu | 1.37 (1.16–1.85) | 1.70 (1.08–1.91) | 1.46 (1.32–2.95) | 1.58 (1.09–1.61) | |
| Zn | 24.1 (22.4–32.3) | 30.5 (17.8–36.5) | 27.0 (18.6–55.4) | 27.3 (24.1–28.8) | |
| Se | μg/kg | 851 (791–1131) | 1003 (712–1255) | 916 (652–1877) | 968 (828–1015) |
| Mo | 56.7 (53.1–67.5) | 72.7 (51.4–82.4) | 71.8 (58.0–76.8) | 68.6 (65.2–82.3) | |
| Mn | 203 (178–258) | 260 (164–286) | 237 (172–441) | 219 (207–298) | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tariba Lovaković, B.; Kašuba, V.; Sekovanić, A.; Orct, T.; Jančec, A.; Pizent, A. Effects of Sub-Chronic Exposure to Imidacloprid on Reproductive Organs of Adult Male Rats: Antioxidant State, DNA Damage, and Levels of Essential Elements. Antioxidants 2021, 10, 1965. https://doi.org/10.3390/antiox10121965
Tariba Lovaković B, Kašuba V, Sekovanić A, Orct T, Jančec A, Pizent A. Effects of Sub-Chronic Exposure to Imidacloprid on Reproductive Organs of Adult Male Rats: Antioxidant State, DNA Damage, and Levels of Essential Elements. Antioxidants. 2021; 10(12):1965. https://doi.org/10.3390/antiox10121965
Chicago/Turabian StyleTariba Lovaković, Blanka, Vilena Kašuba, Ankica Sekovanić, Tatjana Orct, Antonija Jančec, and Alica Pizent. 2021. "Effects of Sub-Chronic Exposure to Imidacloprid on Reproductive Organs of Adult Male Rats: Antioxidant State, DNA Damage, and Levels of Essential Elements" Antioxidants 10, no. 12: 1965. https://doi.org/10.3390/antiox10121965
APA StyleTariba Lovaković, B., Kašuba, V., Sekovanić, A., Orct, T., Jančec, A., & Pizent, A. (2021). Effects of Sub-Chronic Exposure to Imidacloprid on Reproductive Organs of Adult Male Rats: Antioxidant State, DNA Damage, and Levels of Essential Elements. Antioxidants, 10(12), 1965. https://doi.org/10.3390/antiox10121965








