Effects of Folinic Acid Administration on Lower Limb Ischemia/Reperfusion Injury in Rats
Abstract
:1. Introduction
2. Materials and Methods
2.1. Induction and Maintenance of Lower Limb Ischemia
2.2. Biochemical Determinations
2.3. Histopathological Study
2.4. Functional Analysis
2.5. Statistical Analysis
3. Results
3.1. Ischemia Induction
3.2. Effect of FA on Biochemical Parameters Analyzed in Serum Samples
3.3. Effect of FA on Functional Recovery
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Blaisdell, F.W. The pathophysiology of skeletal muscle ischemia and the reperfusion syndrome: A review. Cardiovasc. Surg. 2002, 10, 620–630. [Google Scholar] [CrossRef]
- Gillani, S.; Cao, J.; Suzuki, T.; Hak, D.J. The effect of ischemia reperfusion injury on skeletal muscle. Injury 2012, 43, 670–675. [Google Scholar] [CrossRef]
- Saied, A.; Zyaei, A. Tourniquet use during plating of acute extra-articular tibial fractures: Effects on final results of the operation. J. Trauma 2010, 69, E94–E97. [Google Scholar] [CrossRef]
- Konrad, G.; Markmiller, M.; Lenich, A.; Mayr, E.; Rüter, A. Tourniquets may increase postoperative swelling and pain after internal fixation of ankle fractures. Clin. Orthop. Relat. Res. 2005, 433, 189–194. [Google Scholar] [CrossRef]
- Kirkley, A.; Rampersaud, R.; Griffin, S.; Amendola, A.; Litchfield, R.; Fowler, P. Tourniquet versus no tourniquet use in routine knee arthroscopy: A prospective, double-blind, randomized clinical trial. Arthroscopy 2000, 16, 121–126. [Google Scholar] [CrossRef]
- Ejaz, A.; Laursen, A.C.; Kappel, A.; Laursen, M.B.; Jakobsen, T.; Rasmussen, S.; Nielsen, P.T. Faster recovery without the use of a tourniquet in total knee arthroplasty. Acta Orthop. 2014, 85, 422–426. [Google Scholar] [CrossRef]
- Liu, D.; Graham, D.; Gillies, K.; Gillies, R.M. Effects of tourniquet use on quadriceps function and pain in total knee arthroplasty. Knee Surg. Relat. Res. 2014, 26, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Dennis, D.A.; Kittelson, A.J.; Yang, C.C.; Miner, T.M.; Kim, R.H.; Stevens-Lapsley, J.E. Does Tourniquet Use in TKA Affect Recovery of Lower Extremity Strength and Function? A Randomized Trial. Clin. Orthop. Relat. Res. 2016, 474, 69–77. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nishiguchi, M.; Takamura, N.; Abe, Y.; Kono, M.; Shindo, H.; Aoyagi, K. Pilot study on the use of tourniquet: A risk factor for pulmonary thromboembolism after total knee arthroplasty? Thromb. Res. 2005, 115, 271–276. [Google Scholar] [CrossRef]
- Pedowitz, R.A.; Gershuni, D.H.; Botte, M.J.; Kuiper, S.; Rydevik, B.L.; Hargens, A.R. The Use of Lower Tourniquet Inflation Pressures in Extremity Surgery Facilitated by Curved and Wide Tourniquets and an Integrated Cuff Inflation System. Clin. Orthop. Relat. Res. 1993, 287, 237–244. [Google Scholar] [CrossRef]
- Estebe, J.P.; Davies, J.M.; Richebe, P. The pneumatic tourniquet: Mechanical, ischaemia-reperfusion and systemic effects. Eur. J. Anaesthesiol. 2011, 28, 404–411. [Google Scholar] [CrossRef]
- Wang, W.Z.; Baynosa, R.C.; Zamboni, W.A. Therapeutic interventions against reperfusion injury in skeletal muscle. J. Surg. Res. 2011, 171, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Novelli, G.P.; Adembri, C.; Gandini, E.; Orlandini, S.Z.; Papucci, L.; Formigli, L.; Manneschi, L.I.; Quattrone, A.; Pratesi, C.; Capaccioli, S. Vitamin E protects human skeletal muscle from damage during surgical ischemia-reperfusion. Am. J. Surg. 1997, 173, 206–209. [Google Scholar] [CrossRef]
- Mowafi, H.A.; Ismail, S.A. Melatonin Improves Tourniquet Tolerance and Enhances Postoperative Analgesia in Patients Receiving Intravenous Regional Anesthesia. Anesth. Analg. 2008, 107, 1422–1426. [Google Scholar] [CrossRef]
- Saricaoglu, F.; Dal, D.; Salman, A.E.; Atay, O.A.; Doral, M.N.; Salman, M.A.; Kilinç, K.; Aypar, U. Effect of low-dose N-acetyl-cysteine infusion on tourniquet-induced ischaemia-reperfusion injury in arthroscopic knee surgery. Acta Anaesthesiol. Scand. 2005, 49, 847–851. [Google Scholar] [CrossRef] [PubMed]
- Cowled, P.A.; Khanna, A.; Laws, P.E.; Field, J.B.F.; Varelias, A.; Fitridge, R.A. Statins Inhibit Neutrophil Infiltration in Skeletal Muscle Reperfusion Injury. J. Surg. Res. 2007, 141, 267–276. [Google Scholar] [CrossRef]
- Köksoy, C.; Oziş, E.; Cakmak, A.; Yazgan, U.; Okcu-Heper, A.; Köksoy, A.; Demirpençe, E.; Deniz Dinçer, U. Simvastatin pretreatment reduces the severity of limb ischemia in an experimental diabetes model. J. Vasc. Surg. 2007, 45, 590–596. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arnaoutoglou, H.; Vretzakis, G.; Souliotis, D.; Cambili, M.; Galaris, D.; Papadopoulos, G. The effects of propofol or sevoflurane on free radical production after tourniquet induced ischaemia-reperfusion injury during knee arthroplasty. Acta Anaesthesiol. Belg. 2007, 58, 3–6. [Google Scholar] [PubMed]
- Cheng, W.; Wang, M.; Liu, P.; Zhao, S.; Liu, X.; Wang, X. Protective Effects of Dexmedetomidine and Oxycodone in Patients Undergoing Limb Ischemia-Reperfusion. Med. Sci. Monit. 2019, 25, 9073–9084. [Google Scholar] [CrossRef]
- Murry, C.E.; Jennings, R.B.; Reimer, K.A. Preconditioning with ischemia: A delay of lethal cell injury in ischemic myocardium. Circulation 1986, 74, 1124–1136. [Google Scholar] [CrossRef] [Green Version]
- Kocman, E.A.; Ozatik, O.; Sahin, A.; Guney, T.; Kose, A.A.; Dag, I.; Alatas, O.; Cetin, C. Effects of ischemic preconditioning protocols on skeletal muscle ischemia-reperfusion injury. J. Surg. Res. 2015, 193, 942–952. [Google Scholar] [CrossRef]
- Mansour, Z.; Bouitbir, J.; Charles, A.L.; Talha, S.; Kindo, M.; Pottecher, J.; Zoll, J.; Geny, B. Remote and local ischemic preconditioning equivalently protects rat skeletal muscle mitochondrial function during experimental aortic cross-clamping. J. Vasc. Surg. 2012, 55, 497–505. [Google Scholar] [CrossRef] [Green Version]
- Westman, B.; Weidenhielm, L.; Rooyackers, O.; Fredriksson, K.; Wernerman, J.; Hammarqvist, F. Knee replacement surgery as a human clinical model of the effects of ischaemia/reperfusion upon skeletal muscle. Clin. Sci. 2007, 113, 313–318. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cearra, I.; de la Parte, B.H.; Moreno-Franco, D.I.; García-Alonso, I. A reproducible method for biochemical, histological and functional assessment of the effects of ischaemia-reperfusion syndrome in the lower limbs. Sci. Rep. 2021, 11, 19325. [Google Scholar] [CrossRef]
- Atteia, B.M.R.; El-Kak, A.E.-A.A.; Lucchesi, P.A.; Delafontane, P. Antioxidant activity of folic acid: From mechanism of action to clinical application. FASEB J. 2009, 23, 7–103. [Google Scholar] [CrossRef]
- Joshi, R.; Adhikari, S.; Patro, B.S.; Chattopadhyay, S.; Mukherjee, T. Free radical scavenging behavior of folic acid: Evidence for possible antioxidant activity. Free Radic. Biol. Med. 2001, 30, 1390–1399. [Google Scholar] [CrossRef]
- Asbaghi, O.; Ghanavati, M.; Ashtary-Larky, D.; Bagheri, R.; Rezaei Kelishadi, M.; Nazarian, B.; Nordvall, M.; Wong, A.; Dutheil, F.; Suzuki, K.; et al. Effects of Folic Acid Supplementation on Oxidative Stress Markers: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Antioxidants 2021, 10, 871. [Google Scholar] [CrossRef] [PubMed]
- Merrell, B.J.; McMurry, J.P. Folic Acid; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Moat, S.J.; Madhavan, A.; Taylor, S.Y.; Payne, N.; Allen, R.H.; Stabler, S.P.; Goodfellow, J.; McDowell, I.F.W.; Lewis, M.J.; Lang, D. High- but not low-dose folic acid improves endothelial function in coronary artery disease. Eur. J. Clin. Investig. 2006, 36, 850–859. [Google Scholar] [CrossRef]
- Bilbao, J.E.; Garcia-Alonso, I.; Portugal, V.; Barceló, P.; Ortiz Lacorzana, J.; Méndez, J. Therapeutic usefulness of antioxidant drugs in experimental intestinal reperfusion syndrome. Rev. Esp. Enferm. Dig. 1991, 80, 237–241. [Google Scholar]
- San Cristóbal, J.; Cearra, I.; Otero, B.; Martínez-Astorquiza, T.; Marín, H.; García-Alonso, I. Capillary blood flown as an index of the therapeutic effect of folinic acid in ischemia-reperfusion syndrome. Rev. Esp. Enferm. Dig. 2007, 99, 25–32. [Google Scholar]
- McCormack, M.C.; Kwon, E.; Eberlin, K.R.; Randolph, M.; Friend, D.S.; Thomas, A.C.; Watkins, M.T.; Austen, W.G. Development of reproducible histologic injury severity scores: Skeletal muscle reperfusion injury. Surgery 2008, 143, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Gangadharan, R.; Roslee, C.; Kelsall, N.; Taylor, H. Retrospective review of complications following long tourniquet time in foot and ankle surgery. J. Clin. Orthop. Trauma 2021, 16, 189–194. [Google Scholar] [CrossRef]
- Masri, B.A.; Eisen, A.; Duncan, C.P.; McEwen, J.A. Tourniquet-induced nerve compression injuries are caused by high pressure levels and gradients—A review of the evidence to guide safe surgical, pre-hospital and blood flow restriction usage. BMC Biomed. Eng. 2020, 2, 7. [Google Scholar] [CrossRef] [PubMed]
- Gregory, M.A.; Mars, M. Serial morphological changes in primate skeletal myofibres after 3 hours of ischaemia and 24 hours of reperfusion. S. Afr. Med. J. 1992, 81, 473–478. [Google Scholar]
- Day, R.L.; Zale, B.W. The effect of tourniquets on muscle enzymes during foot and ankle surgery. J. Foot Ankle Surg. 1993, 32, 280–285. [Google Scholar] [PubMed]
- Daniel, D.M.; Lumkong, G.; Stone, M.L.; Pedowitz, R.A. Effects of tourniquet use in anterior cruciate ligament reconstruction. Arthroscopy 1995, 11, 307–311. [Google Scholar] [CrossRef]
- Murata, I.; Ooi, K.; Sasaki, H.; Kimura, S.; Ohtake, K.; Ueda, H.; Uchida, H.; Yasui, N.; Tsutsui, Y.; Yoshizawa, N.; et al. Characterization of Systemic and Histologic Injury After Crush Syndrome and Intervals of Reperfusion in a Small Animal Model. J. Trauma Acute Care Surg. 2011, 70, 1453–1463. [Google Scholar] [CrossRef] [PubMed]
- Duarte, J.A.; Glöser, S.; Remião, F.; Carvalho, F.; Bastos, M.L.; Soares, J.M.; Appell, H.J. Administration of tourniquet. I. Are edema and oxidative stress related to each other and to the duration of ischemia in reperfused skeletal muscle? Arch. Orthop. Trauma Surg. 1997, 116, 97–100. [Google Scholar] [CrossRef] [PubMed]
- Crawford, R.S.; Hashmi, F.F.; Jones, J.E.; Albadawi, H.; McCormack, M.; Eberlin, K.; Entabi, F.; Atkins, M.D.; Conrad, M.F.; Austen, W.G.; et al. A novel model of acute murine hindlimb ischemia. Am. J. Physiol. Circ. Physiol. 2007, 292, H830–H837. [Google Scholar] [CrossRef]
- Bonheur, J.A.; Albadawi, H.; Patton, G.M.; Watkins, M.T. A noninvasive murine model of hind limb ischemia-reperfusion injury. J. Surg. Res. 2004, 116, 55–63. [Google Scholar] [CrossRef]
- Sagara, G.; Ogikubo, O.; Takeyama, H.; Tada, T.; Suzuki, N.; Okamoto, H.; Otsuka, T. Comparison of effects of ischemia period in rat limb ischemia/reperfusion injury. Nagoya Med. J. 2009, 50, 83–91. [Google Scholar]
- Goto, H.; Benson, K.T.; Katayama, H.; Tonooka, M.; Tilzer, L.L.; Arakawa, K. Effect of high-dose of methylprednisolone on tourniquet ischaemia. Can. J. Anaesth. 1988, 35, 484–488. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Woodruff, T.M.; Arumugam, T.V.; Shiels, I.A.; Reid, R.C.; Fairlie, D.P.; Taylor, S.M. Protective effects of a potent C5a receptor antagonist on experimental acute limb ischemia-reperfusion in rats. J. Surg. Res. 2004, 116, 81–90. [Google Scholar] [CrossRef]
- Garbaisz, D.; Turoczi, Z.; Aranyi, P.; Fulop, A.; Rosero, O.; Hermesz, E.; Ferencz, A.; Lotz, G.; Harsanyi, L.; Szijarto, A. Attenuation of Skeletal Muscle and Renal Injury to the Lower Limb following Ischemia-Reperfusion Using mPTP Inhibitor NIM-811. PLoS ONE 2014, 9, e101067. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, S.; Zhan, L.; Yang, Z.; Shi, R.; Li, H.; Xia, Z.; Yuan, S.; Wu, Q.-P.; Wang, T.; Yao, S. Remote Limb Ischaemic Postconditioning Protects Against Myocardial Ischaemia/Reperfusion Injury in Mice: Activation of JAK/STAT3-Mediated Nrf2-Antioxidant Signalling. Cell. Physiol. Biochem. 2017, 43, 1140–1151. [Google Scholar] [CrossRef]
- Tong, Z.; Yu, F.; Liu, Z.; Liang, H. Influence of ShuJinHuoXue tablets on ischemia reperfusion injury of animals’ skeletal muscle. Molecules 2012, 17, 8494–8505. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Holoman, J.; Glasa, J.; Galbavy, S.; Danis, D.; Molnarova, A.; Kazar, J.; Bednarova, A.; Misianik, J. Serum markers of liver fibrogenesis, and liver histology findings in patients with chronic liver diseases. Bratisl. Lek. Listy 2002, 103, 70–75. [Google Scholar]
- Frijhoff, J.; Winyard, P.G.; Zarkovic, N.; Davies, S.S.; Stocker, R.; Cheng, D.; Knight, A.R.; Taylor, E.L.; Oettrich, J.; Ruskovska, T.; et al. Clinical Relevance of Biomarkers of Oxidative Stress. Antioxid. Redox Signal. 2015, 23, 1144–1170. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.Y.; Kim, C.J.; Chung, M.Y. Effect of high-dose vitamin C on oxygen free radical production and myocardial enzyme after tourniquet ischaemia-reperfusion injury during bilateral total knee replacement. J. Int. Med. Res. 2010, 38, 1519–1529. [Google Scholar] [CrossRef] [Green Version]
- Yagmurdur, H.; Ozcan, N.; Dokumaci, F.; Kilinc, K.; Yilmaz, F.; Basar, H. Dexmedetomidine Reduces the Ischemia-Reperfusion Injury Markers During Upper Extremity Surgery with Tourniquet. J. Hand Surg. Am. 2008, 33, 941–947. [Google Scholar] [CrossRef]
- Çandar, T.; Uzunlar, Ö.; Kiseli, M.; Özcan, S. The effect of folate on ischemia/reperfusion injury in a rat adnexal torsion model. Arch. Gynecol. Obstet. 2021, 303, 1495–1500. [Google Scholar] [CrossRef]
- Knight, S.F.; Kundu, K.; Joseph, G.; Dikalov, S.; Weiss, D.; Murthy, N.; Taylor, W.R. Folate receptor-targeted antioxidant therapy ameliorates renal ischemia-reperfusion injury. J. Am. Soc. Nephrol. 2012, 23, 793–800. [Google Scholar] [CrossRef] [Green Version]
- Naito, H.; Nojima, T.; Fujisaki, N.; Tsukahara, K.; Yamamoto, H.; Yamada, T.; Aokage, T.; Yumoto, T.; Osako, T.; Nakao, A. Therapeutic strategies for ischemia reperfusion injury in emergency medicine. Acute Med. Surg. 2020, 7, e501. [Google Scholar] [CrossRef] [Green Version]
- San Cristóbal Epalza, J.; Palomares, T.; García-Alonso, I.; de la Parte, B.H. Histological Assessment of Rat Retinas with Ischemia-Reperfusion Injury. Eur. Surg. Res. 2021, 62, 144–150. [Google Scholar] [CrossRef] [PubMed]
- Portugal, V.; García-Alonso, I.; Barceló, P.; Méndez, J. Effect of Allopurinol, Folinic Acid, SOD and Cyclosporine A on Ischemic Liver Regeneration. Eur. Surg. Res. 1995, 27, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Tay, S.K.; Ong, H.T.; Low, P.S. Transaminitis in Duchenne’s muscular dystrophy. Ann. Acad. Med. Singap. 2000, 29, 719–722. [Google Scholar] [PubMed]
- Orban, J.-C.; Levraut, J.; Gindre, S.; Deroche, D.; Schlatterer, B.; Ichai, C.; Grimaud, D. Effects of acetylcysteine and ischaemic preconditioning on muscular function and postoperative pain after orthopaedic surgery using a pneumatic tourniquet. Eur. J. Anaesthesiol. 2006, 23, 1025–1030. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alexandersson, M.; Wang, E.Y.; Eriksson, S. A small difference in recovery between total knee arthroplasty with and without tourniquet use the first 3 months after surgery: A randomized controlled study. Knee Surg. Sport. Traumatol. Arthrosc. 2019, 27, 1035–1042. [Google Scholar] [CrossRef] [Green Version]
- Chen, G.; Kamat, P.K.; Ahmad, A.S.; Doré, S. Distinctive effect of anesthetics on the effect of limb remote ischemic postconditioning following ischemic stroke. PLoS ONE 2020, 15, e0227624. [Google Scholar] [CrossRef]
- Deacon, R.M.J. Measuring motor coordination in mice. J. Vis. Exp. 2013, 75, e2609. [Google Scholar] [CrossRef]
- Díaz, D.; Piquer-Gil, M.; Recio, J.S.; Martínez-Losa, M.M.; Alonso, J.R.; Weruaga, E.; Álvarez-Dolado, M. Bone marrow transplantation improves motor activity in a mouse model of ataxia. J. Tissue Eng. Regen. Med. 2018, 12, e1950–e1961. [Google Scholar] [CrossRef] [PubMed]
- Graber, T.G.; Ferguson-Stegall, L.; Kim, J.-H.; Thompson, L. V C57BL/6 neuromuscular healthspan scoring system. J. Gerontol. A Biol. Sci. Med. Sci. 2013, 68, 1326–1336. [Google Scholar] [CrossRef] [PubMed] [Green Version]




| Parameter | Control Group | Saline 3 h Group | FA 3 h Group | Saline 14 D Group | FA 14 D Group |
|---|---|---|---|---|---|
| Na+ | 142 ± 1 | 143 ± 2 | 143 ± 2 | 142 ± 1 | 142 ± 1 |
| K+ | 3.87 ± 0.29 | 3.95 ± 0.29 | 3.97 ± 0.13 | 4.05 ± 0.3 | 4.00 ± 0.21 |
| Cl− | 104 ± 2 | 100 ± 2 | 100 ± 3 | 101 ± 3 | 100 ± 3 |
| Urea | 29.0 ± 3.1 | 66.3 ± 5.1 | 59.6 ± 6.1 | 29 ± 2.8 | 28.5 ± 2.1 |
| Creatinine | 0.36 ± 0.06 | 0.80 ± 0.13 | 0.51 ± 0.13 | 0.41 ± 0.06 | 0.38 ± 0.04 |
| ALP | 123 ± 4 | 115 ± 5 | 99 ± 4 | 85 ± 5 | 82 ± 13 |
| AST | 51.4 ± 2.6 | 393.2 + 63.6 | 250.7 ± 20.7 | 87.0 ± 14.6 | 59.0 ± 10.5 |
| ALT | 41.7 ± 1.6 | 90.6 ± 4.4 | 62.0 ± 6.0 | 31.0 ± 5.2 | 28.8 ± 4.2 |
| CK | 92 ± 15 | 7792 ± 1187 | 6123 ± 1000 | 552 ± 116 | 298 ± 137 |
| LDH | 80 ± 9 | 1413 ± 227 | 848 ± 109 | 1281 ± 339 | 603 ± 255 |
| Parameter | Control Group | Saline 24 h Group | FA 24 h Group | |
|---|---|---|---|---|
| Perimeter (mm) | left limb | 54.5 ± 2.7 | 53.8 ± 3.4 | 54.5 ± 14.9 |
| right limb | 52.8 ± 3.0 | 61.7 ± 3.4 | 62.8 ± 2.2 | |
| Increase of limb section (ischemic vs. non ischemic) (%) | 0.1 ± 7.1 | 29.9 ± 10.0 | 17.4 ± 9.2 | |
| Weight (g) | left limb | 1.82 ± 0.04 | 1.81 ± 0.14 | 1.78 ± 0.10 |
| right limb | 1.84 ± 0.09 | 2.06 ± 0.15 | 1.86 ± 0.10 | |
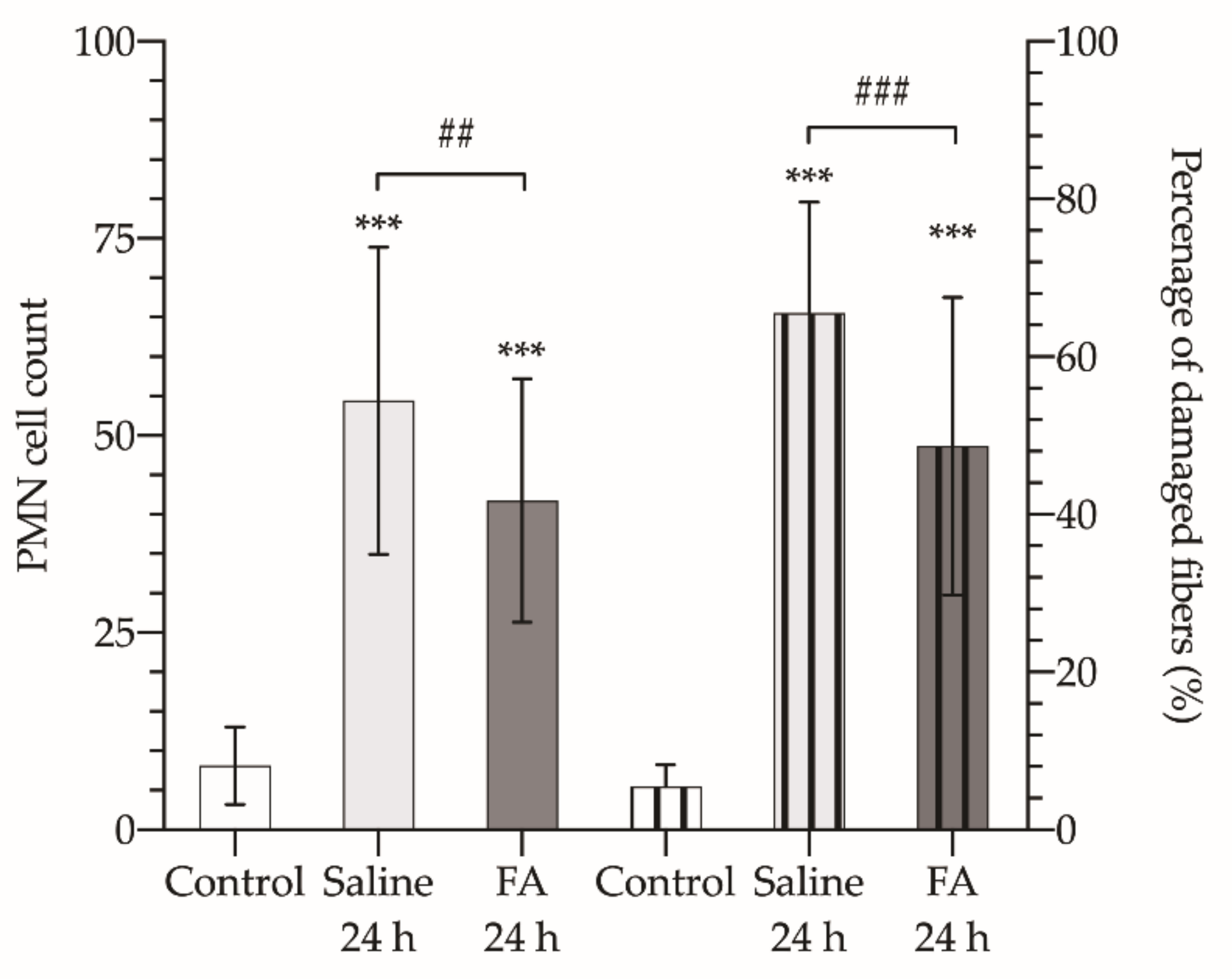
| PMN (right limb) | 8.13 ± 4.93 | 54.44 ± 19.47 | 41.80 ± 15.40 | |
| Damaged fibers (%) | 5.44 ± 2.76 | 65.53 ± 14.08 | 48.68 ± 18.86 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cearra, I.; Herrero de la Parte, B.; Ruiz Montesinos, I.; Alonso-Varona, A.; Moreno-Franco, D.I.; García-Alonso, I. Effects of Folinic Acid Administration on Lower Limb Ischemia/Reperfusion Injury in Rats. Antioxidants 2021, 10, 1887. https://doi.org/10.3390/antiox10121887
Cearra I, Herrero de la Parte B, Ruiz Montesinos I, Alonso-Varona A, Moreno-Franco DI, García-Alonso I. Effects of Folinic Acid Administration on Lower Limb Ischemia/Reperfusion Injury in Rats. Antioxidants. 2021; 10(12):1887. https://doi.org/10.3390/antiox10121887
Chicago/Turabian StyleCearra, Iñigo, Borja Herrero de la Parte, Inmaculada Ruiz Montesinos, Ana Alonso-Varona, Diana Isabel Moreno-Franco, and Ignacio García-Alonso. 2021. "Effects of Folinic Acid Administration on Lower Limb Ischemia/Reperfusion Injury in Rats" Antioxidants 10, no. 12: 1887. https://doi.org/10.3390/antiox10121887
APA StyleCearra, I., Herrero de la Parte, B., Ruiz Montesinos, I., Alonso-Varona, A., Moreno-Franco, D. I., & García-Alonso, I. (2021). Effects of Folinic Acid Administration on Lower Limb Ischemia/Reperfusion Injury in Rats. Antioxidants, 10(12), 1887. https://doi.org/10.3390/antiox10121887









