Omega−3 Polyunsaturated Fatty Acids (PUFAs): Emerging Plant and Microbial Sources, Oxidative Stability, Bioavailability, and Health Benefits—A Review
Abstract
:1. Introduction
2. Are All n−3 PUFAs (ALA, SDA, EPA, and DHA) Equally Beneficial for Health?
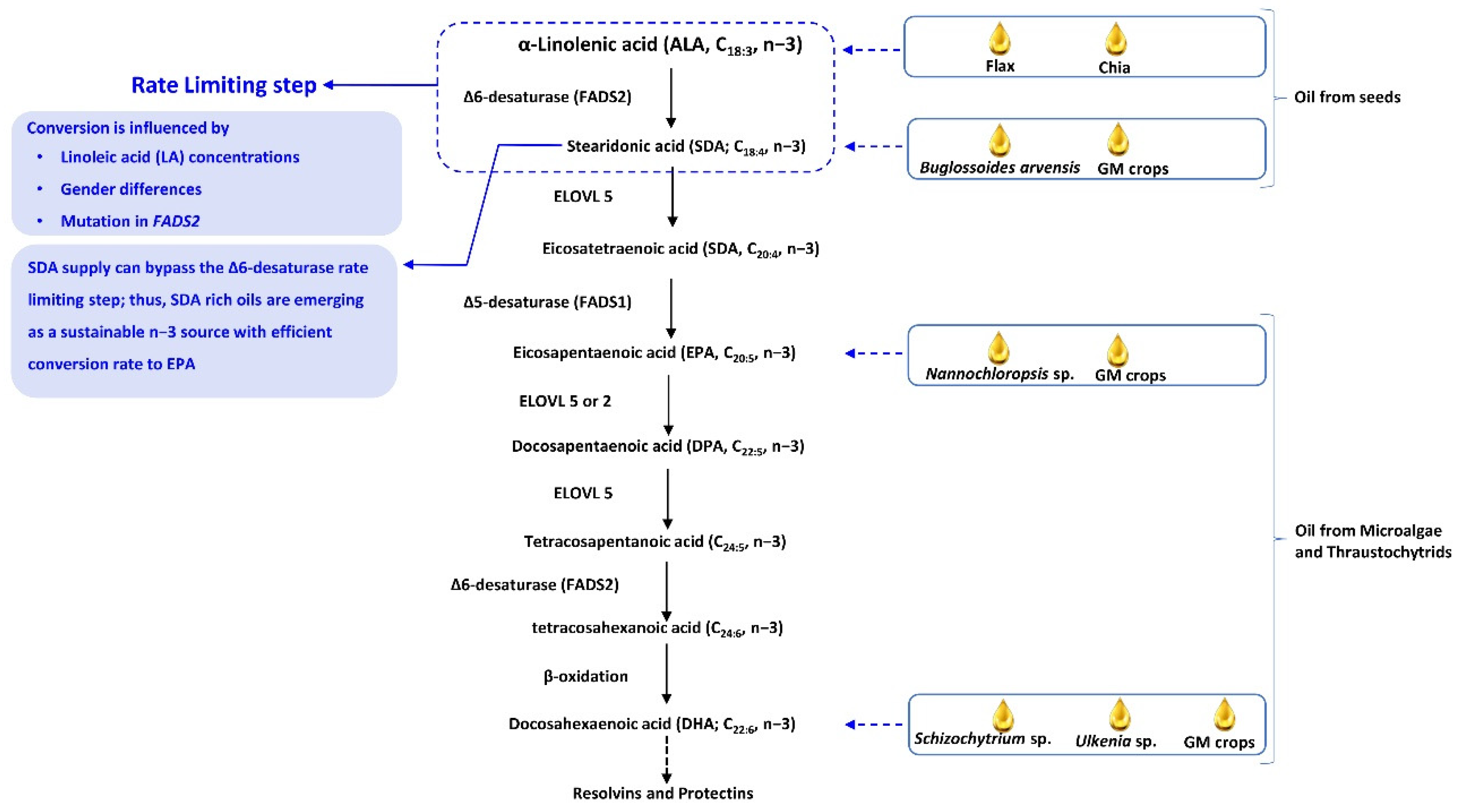
3. A Low Conversion Rate of ALA to EPA and DHA Is a Challenge
4. The Recommended Intake of n−3 PUFAs
5. Dietary Sources of Vegan n−3 PUFAs
5.1. ALA-Rich Seeds
5.2. ALA-Rich Herb: Purslane
5.3. SDA-Rich Seeds and Herbs
5.4. Thraustochytrids, Microalgae, and Macroalgae: Source of EPA and DHA
| Source | Oil Content (%) | ALA | SDA | EPA | DHA | n−6/n−3 | Reference | |
|---|---|---|---|---|---|---|---|---|
| Seeds | Brassica napus sp. oleifera L. (rapeseed/canola) | 36.9–40.5 | 8.7–9.5 | 1.9–2.5 | [56] | |||
| Buglossoides arvensis (L.) I.M. Johnst. (Corn gromwell; Ahiflower®) * | 20.0 | 49.6 | 21 | 0.18 | [92] | |||
| Camelina sativa (L.) Crtz. (Camelina) * | 29.6–49.0 | 19.1–43.1 | [71] | |||||
| Echium canatabricum | 33.6 | 14.7 | 0.55 | [91] | ||||
| Echium plantagineum (Purple viper’s bugloss) | 24.1 | 34.5 | 11.0 | 0.6 | [90] | |||
| Eucommia ulmoides Oliver | 34.63 | 61.36 | [82] | |||||
| Lappula patula | 40.0 | 13.6 | 0.40 | [91] | ||||
| Lepidium sativum (Garden cress) | 21–24 | 30.34 | 0.42 | [114] | ||||
| Linum usitatissimum (Flax) | 38.76 | 53.4 | 0.290 | [60] | ||||
| Mertensia alpine (Torr.) G.Don. | - | 12.9 | 9.3 | 1.6 | [115] | |||
| Mertensia ciliata (James ex Torr.) G. Don. | - | 11.8 | 6.4 | 1.6 | [115] | |||
| Ocimum basilicum (Basil) | 22.0 | 63.8 | 0.320 | [60] | ||||
| Paeonia section Moutan DC. (Tree peony) | 26.1–54.7 | 0.4–1.0 | [75] | |||||
| Perilla frutescens (Perilla) | 42.8 | 65.6 | 0.190 | [60] | ||||
| Plukenetia volubilis L. (Sacha inchi) | 33.4–37.6 | 37.3–44.2 | 0.83–1.09 | [77] | ||||
| Ribes nidrigolaria Bauer (Jostaberry) | 28.01 | 5.45 | - | - | 1.17 | [94] | ||
| Ribes nigrum L. (Blackcurrant) | 14.89 | 2.86 | - | - | 3.17 | [94] | ||
| Ribes rubrum L. (Redcurrant) | 24.40 | 3.35 | - | - | 1.48 | [94] | ||
| Ribes uva-crispa L. (Gooseberry) | 20.54 | 4.32 | - | - | 1.82 | [94] | ||
| Salvia hispanica L. (Chia) * | 30.17–32.16 | 54.5–64.7 | [68] | |||||
| Herb | Mertensia maritima (L.) Gray | 10.9 | 30.4 | 6 | 0.85 | [95] | ||
| Portulaca oleracea L. (Purslane) | 45.30–51.2 | 1:1–1:3 | [85] | |||||
| Microalgae | Isochrysis galbana | 3.1 | 11.8 | [116] | ||||
| Nannochloropsis salina * | 25–30 | http://www.lyxia.com/product/ [Accessed 14 Oct 2021] | ||||||
| Nannochloropsis sp. | 37–60 | 0.1–17.5 | 4.7–33.7 | [107] | ||||
| Nannochloropsis sp. CCNM 1081 | 39.8 | 27.6 | [109] | |||||
| Nannochloropsis sp. BR2 | 0.4 | 18.8 | [116] | |||||
| Pavlova lutheri | 0.1 | 21.8 | [116] | |||||
| Phaeodactylum tricornutum Bohlin | 0.38–0.40 | 0.87–1.14 | 22.8–30.7 | 0.98–1.70 | [117] | |||
| Thraustochytrid | Aurantiochytrium limacinum SR21 | 30–40 | [101] | |||||
| Crypthecodinium cohnii * | 40–45 | [102] | ||||||
| Schizochytrium sp. | 0.07 | 16.2 | 33.7 | [100] | ||||
| Schizochytrium limacinum SR21 | 52.3 | 66.7 | [118] | |||||
| Thraustochytrium sp. ONC T18 | 37.8 | [106] | ||||||
| Schizochytrium sp. FCC-3204 | 0.1 | 0.3–0.4 | 0.5-0.9 | 59.8–63.1 | GRAS Notice (GRN) No. 844 | |||
| Schizochytrium sp. ONC-Tl8 * | 0.20–0.32 | 37.10–42.47 | GRAS Notice (GRN) No. 677 | |||||
| Ulkenia sp. SAM 2179 * | 45 | [103] | ||||||
| Seaweeds | Codium fragile (Suhr) Hariot (Chlorophyta) | 2.7 | 14.2–19.9 | 3.0–4.4 | 0.3 | [119] | ||
| Laminaria digitata (Hudson) J.V. Lamouroux (Phaeophyceae) | ̴1.5 | 5.0–5.5 | 12.5–13.1 | 0.5–0.7 | [119] | |||
| Palmaria palmata | 1.39 | 0.8 | 32.1 | 0.4 | [111] | |||
| Palmaria palmata (L.) O. Kuntze (Rhodophyta) | ̴1.5 | 1.5 | 36.8–41.2 | 0.1 | [119] | |||
| Vertebrata lanosa | 1.8 | 34.3 | 0.03 | [111] |
5.5. Genetically Modified (GM) Plants
6. Low Oxidative Stability of PUFAs in Foods Is a Challenge
7. Emulsion-Based PUFAs Can Be Used in Beverages
8. Bioavailability of Vegan n−3 PUFAs: Algal-Oil Supplements Are a Viable Alternative to Fish Oil
9. Commercial Availability of Plant and Microbial-Based n−3 PUFAs
10. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ALA | α-Linolenic acid |
| AP | Ascorbyl palmitate |
| ARA | Arachidonic acid |
| BHA | Butylated hydroxyanisole |
| BHT | Butylated hydroxytoluene |
| CHD | Coronary heart disease |
| CVD | Cardiovascular diseases |
| DAG | Diacylglycerol |
| DHA | Docosahexaenoic acid |
| DPA | Docosapentaenoic acid |
| EPA | Eicosapentaenoic acid |
| GCO | Garden cress oil |
| GLA | γ-Linolenic acid |
| GM | Genetically modified |
| GRAS | Generally recognized as safe |
| LA | Linoleic acid |
| LDL | Low-density lipoprotein |
| MAG | Monoacylglycerol |
| MUFAs | Monounsaturated fatty acids |
| PBMCs | Peripheral blood mononuclear cell |
| PLs | Phospholipids |
| RBC | Red blood cells |
| SDA | Stearidonic acid |
| SPM | Pro-resolving mediators |
| TAG | Triacylglycerol |
| VLC-PUFAs | Very long chain-poly unsaturated fatty acids |
References
- Bazinet, R.P.; Layé, S. Polyunsaturated fatty acids and their metabolites in brain function and disease. Nat. Rev. Neurosci. 2014, 15, 771–785. [Google Scholar] [CrossRef]
- Saini, R.K.; Keum, Y.S. Omega-3 and omega-6 polyunsaturated fatty acids: Dietary sources, metabolism, and significance—A review. Life Sci. 2018, 203, 255–267. [Google Scholar] [CrossRef]
- Johnson, G.H.; Fritsche, K. Effect of dietary linoleic acid on markers of inflammation in healthy persons: A systematic review of randomized controlled trials. J. Acad. Nutr. Diet 2012, 112, 1029–1041.e15. [Google Scholar] [CrossRef]
- Harris, W.S. The Omega-6:Omega-3 ratio: A critical appraisal and possible successor. Prostaglandins Leukot. Essent. Fat. Acids 2018, 132, 34–40. [Google Scholar] [CrossRef]
- Appleby, P.N.; Key, T.J. The long-term health of vegetarians and vegans. Proc. Nutr. Soc. 2016, 75, 287–293. [Google Scholar] [CrossRef] [Green Version]
- Craig, W.J.; Mangels, A.R. Position of the American Dietetic Association: Vegetarian diets. J. Am. Diet. Assoc. 2009, 109, 1266. [Google Scholar] [PubMed]
- Sarter, B.; Kelsey, K.S.; Schwartz, T.A.; Harris, W.S. Blood docosahexaenoic acid and eicosapentaenoic acid in vegans: Associations with age and gender and effects of an algal-derived omega-3 fatty acid supplement. Clin. Nutr. 2015, 34, 212–218. [Google Scholar] [CrossRef] [Green Version]
- Burns-Whitmore, B.; Froyen, E.; Heskey, C.; Parker, T.; San Pablo, G. Alpha-Linolenic and Linoleic Fatty Acids in the Vegan Diet: Do They Require Dietary Reference Intake/Adequate Intake Special Consideration? Nutrients 2019, 11, 2365. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adarme-Vega, T.; Lim, D.K.Y.; Timmins, M.; Vernen, F.; Li, Y.; Schenk, P.M. Microalgal biofactories: A promising approach towards sustainable omega-3 fatty acid production. Microb. Cell Factories 2012, 11, 96. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wells, M.L.; Potin, P.; Craigie, J.S.; Raven, J.A.; Merchant, S.S.; Helliwell, K.E.; Smith, A.G.; Camire, M.E.; Brawley, S.H. Algae as nutritional and functional food sources: Revisiting our understanding. J. Appl. Phycol. 2017, 29, 949–982. [Google Scholar] [CrossRef]
- Orozco Colonia, B.S.; Vinícius de Melo Pereira, G.; Soccol, C.R. Omega-3 microbial oils from marine thraustochytrids as a sustainable and technological solution: A review and patent landscape. Trends Food Sci. Technol. 2020, 99, 244–256. [Google Scholar] [CrossRef]
- Burdge, G.C. Metabolism of α-linolenic acid in humans. Prostaglandins Leukot. Essent. Fat. Acids 2006, 75, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Botelho, P.B.; Mariano, K.D.R.; Rogero, M.M.; De Castro, I.A. Effect of Echium oil compared with marine oils on lipid profile and inhibition of hepatic steatosis in LDLr knockout mice. Lipids Health Dis. 2013, 12, 38. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schwab, U.S.; Callaway, J.C.; Erkkilä, A.T.; Gynther, J.; Uusitupa, M.I.J.; Järvinen, T. Effects of hempseed and flaxseed oils on the profile of serum lipids, serum total and lipoprotein lipid concentrations and haemostatic factors. Eur. J. Nutr. 2006, 45, 470–477. [Google Scholar] [CrossRef] [PubMed]
- Yue, H.; Qiu, B.; Jia, M.; Liu, W.; Guo, X.-F.; Li, N.; Xu, Z.-X.; Du, F.-L.; Xu, T.; Li, D. Effects of α-linolenic acid intake on blood lipid profiles: A systematic review and meta-analysis of randomized controlled trials. Crit. Rev. Food Sci. Nutr. 2020, 1–17. [Google Scholar] [CrossRef]
- Sherratt, S.C.R.; Mason, R.P. Eicosapentaenoic acid and docosahexaenoic acid have distinct membrane locations and lipid interactions as determined by X-ray diffraction. Chem. Phys. Lipids 2018, 212, 73–79. [Google Scholar] [CrossRef]
- Saenz de Viteri, M.; Hernandez, M.; Bilbao-Malavé, V.; Fernandez-Robredo, P.; González-Zamora, J.; Garcia-Garcia, L.; Ispizua, N.; Recalde, S.; Garcia-Layana, A. A Higher Proportion of Eicosapentaenoic Acid (EPA) When Combined with Docosahexaenoic Acid (DHA) in Omega-3 Dietary Supplements Provides Higher Antioxidant Effects in Human Retinal Cells. Antioxidants 2020, 9, 828. [Google Scholar] [CrossRef]
- Belayev, L.; Hong, S.H.; Menghani, H.; Marcell, S.J.; Obenaus, A.; Freitas, R.S.; Khoutorova, L.; Balaszczuk, V.; Jun, B.; Oriá, R.B.; et al. Docosanoids Promote Neurogenesis and Angiogenesis, Blood-Brain Barrier Integrity, Penumbra Protection, and Neurobehavioral Recovery After Experimental Ischemic Stroke. Mol. Neurobiol. 2018, 55, 7090–7106. [Google Scholar] [CrossRef]
- Mason, R.P.; Sherratt, S.C.R.; Jacob, R.F. Eicosapentaenoic Acid Inhibits Oxidation of ApoB-containing Lipoprotein Particles of Different Size In Vitro When Administered Alone or in Combination With Atorvastatin Active Metabolite Compared With Other Triglyceride-lowering Agents. J. Cardiovasc. Pharmacol. 2016, 68, 33. [Google Scholar] [CrossRef] [Green Version]
- Mayurasakorn, K.; Niatsetskaya, Z.V.; Sosunov, S.A.; Williams, J.J.; Zirpoli, H.; Vlasakov, I.; Deckelbaum, R.J.; Ten, V.S. DHA but Not EPA Emulsions Preserve Neurological and Mitochondrial Function after Brain Hypoxia-Ischemia in Neonatal Mice. PLoS ONE 2016, 11, e0160870. [Google Scholar] [CrossRef] [Green Version]
- Ohnishi, H.; Saito, Y. Eicosapentaenoic Acid (EPA) Reduces Cardiovascular Events: Relationship with the EPA/Arachidonic Acid Ratio. J. Atheroscler. Thromb. 2013, 20, 861–877. [Google Scholar] [CrossRef] [Green Version]
- Mason, R.P.; Libby, P.; Bhatt, D.L. Emerging Mechanisms of Cardiovascular Protection for the Omega-3 Fatty Acid Eicosapentaenoic Acid. Arterioscler. Thromb. Vasc. Biol. 2020, 40, 1135–1147. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi Fard, S.; Wang, F.; Sinclair, A.J.; Elliott, G.; Turchini, G.M. How does high DHA fish oil affect health? A systematic review of evidence. Crit. Rev. Food Sci. Nutr. 2019, 59, 1684–1727. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lafuente, M.; Rodríguez González-Herrero, M.E.; Romeo Villadóniga, S.; Domingo, J.C. Antioxidant Activity and Neuroprotective Role of Docosahexaenoic Acid (DHA) Supplementation in Eye Diseases That Can Lead to Blindness: A Narrative Review. Antioxidants 2021, 10, 386. [Google Scholar] [CrossRef]
- Del Gobbo, L.C.; Imamura, F.; Aslibekyan, S.; Marklund, M.; Virtanen, J.K.; Wennberg, M.; Yakoob, M.Y.; Chiuve, S.E.; dela Cruz, L.; Frazier-Wood, A.C.; et al. ω-3 Polyunsaturated Fatty Acid Biomarkers and Coronary Heart Disease: Pooling Project of 19 Cohort Studies. JAMA Intern. Med. 2016, 176, 1155–1166. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Delgado-Lista, J.; Perez-Martinez, P.; Lopez-Miranda, J.; Perez-Jimenez, F. Long chain omega-3 fatty acids and cardiovascular disease: A systematic review. Br. J. Nutr. 2012, 107, S201–S213. [Google Scholar] [CrossRef] [Green Version]
- Mozaffarian, D.; Lemaitre, R.N.; King, I.B.; Song, X.; Huang, H.; Sacks, F.M.; Rimm, E.B.; Wang, M.; Siscovick, D.S. Plasma Phospholipid Long-Chain ω-3 Fatty Acids and Total and Cause-Specific Mortality in Older Adults. Ann. Intern. Med. 2013, 158, 515. [Google Scholar] [CrossRef]
- Deckelbaum, R.J.; Calder, P.C. Editorial: Is it time to separate EPA from DHA when using omega-3 fatty acids to protect heart and brain? Curr. Opin. Clin. Nutr. Metab. Care 2020, 23, 65–67. [Google Scholar] [CrossRef]
- Bhatt, D.L.; Steg, P.G.; Miller, M.; Brinton, E.A.; Jacobson, T.A.; Ketchum, S.B.; Doyle, R.T.; Juliano, R.A.; Jiao, L.; Granowitz, C.; et al. Cardiovascular Risk Reduction with Icosapent Ethyl for Hypertriglyceridemia. N. Engl. J. Med. 2018, 380, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A. An Increase in the Omega-6/Omega-3 Fatty Acid Ratio Increases the Risk for Obesity. Nutrients 2016, 8, 128. [Google Scholar] [CrossRef] [Green Version]
- Grosso, G.; Pajak, A.; Marventano, S.; Castellano, S.; Galvano, F.; Bucolo, C.; Drago, F.; Caraci, F. Role of Omega-3 Fatty Acids in the Treatment of Depressive Disorders: A Comprehensive Meta-Analysis of Randomized Clinical Trials. PLoS ONE 2014, 9, e96905. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yates, C.M.; Calder, P.C.; Ed Rainger, G. Pharmacology and therapeutics of omega-3 polyunsaturated fatty acids in chronic inflammatory disease. Pharmacol. Ther. 2014, 141, 272–282. [Google Scholar] [CrossRef]
- Yan, Y.; Jiang, W.; Spinetti, T.; Tardivel, A.; Castillo, R.; Bourquin, C.; Guarda, G.; Tian, Z.; Tschopp, J.; Zhou, R. Omega-3 Fatty Acids Prevent Inflammation and Metabolic Disorder through Inhibition of NLRP3 Inflammasome Activation. Immunity 2013, 38, 1154–1163. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Swanson, D.; Block, R.; Mousa, S.A. Omega-3 Fatty Acids EPA and DHA: Health Benefits Throughout Life. Adv. Nutr. 2012, 3, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.H.Y.; Micha, R.; Imamura, F.; Pan, A.; Biggs, M.L.; Ajaz, O.; Djousse, L.; Hu, F.B.; Mozaffarian, D. Omega-3 fatty acids and incident type 2 diabetes: A systematic review and meta-analysis. Br. J. Nutr. 2012, 107, S214–S227. [Google Scholar] [CrossRef]
- Abdelhamid, A.S.; Brown, T.J.; Brainard, J.S.; Biswas, P.; Thorpe, G.C.; Moore, H.J.; Deane, K.H.; Summerbell, C.D.; Worthington, H.V.; Song, F.; et al. Omega-3 fatty acids for the primary and secondary prevention of cardiovascular disease. Cochrane Database Syst. Rev. 2020, 3, CD003177. [Google Scholar] [CrossRef] [PubMed]
- Lange, K.W.; Nakamura, Y.; Gosslau, A.M.; Li, S. Are there serious adverse effects of omega-3 polyunsaturated fatty acid supplements? J. Food Bioact. 2019, 7, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Nogueira, M.S.; Scolaro, B.; Milne, G.L.; Castro, I.A. Oxidation products from omega-3 and omega-6 fatty acids during a simulated shelf life of edible oils. LWT 2019, 101, 113–122. [Google Scholar] [CrossRef]
- Zaloga, G.P. Narrative Review of n-3 Polyunsaturated Fatty Acid Supplementation upon Immune Functions, Resolution Molecules and Lipid Peroxidation. Nutrients 2021, 13. [Google Scholar] [CrossRef]
- Burdge, G.C.; Jones, A.E.; Wootton, S.A. Eicosapentaenoic and docosapentaenoic acids are the principal products of α-linolenic acid metabolism in young men. Br. J. Nutr. 2002, 88, 355–363. [Google Scholar] [CrossRef] [Green Version]
- Childs, C.E.; Kew, S.; Finnegan, Y.E.; Minihane, A.M.; Leigh-Firbank, E.C.; Williams, C.M.; Calder, P.C. Increased dietary α-linolenic acid has sex-specific effects upon eicosapentaenoic acid status in humans: Re-examination of data from a randomised, placebo-controlled, parallel study. Nutr. J. 2014, 13, 113. [Google Scholar] [CrossRef] [Green Version]
- Harnack, K.; Andersen, G.; Somoza, V. Quantitation of alpha-linolenic acid elongation to eicosapentaenoic and docosahexaenoic acid as affected by the ratio of n6/n3 fatty acids. Nutr. Metab. 2009, 6, 8. [Google Scholar] [CrossRef] [Green Version]
- Plourde, M.; Cunnane, S.C. Extremely limited synthesis of long chain polyunsaturates in adults: Implications for their dietary essentiality and use as supplements. Appl. Physiol. Nutr. Metab. 2007, 32, 619–634. [Google Scholar] [CrossRef] [Green Version]
- Brenna, J.T.; Salem, N.; Sinclair, A.J.; Cunnane, S.C. α-Linolenic acid supplementation and conversion to n-3 long-chain polyunsaturated fatty acids in humans. Prostaglandins Leukot. Essent. Fat. Acids 2009, 80, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Cumberford, G.; Hebard, A. Ahiflower oil: A novel non-GM plant-based omega-3+6 source. Lipid Technol. 2015, 27, 207–210. [Google Scholar] [CrossRef]
- Simopoulos, A.P. Evolutionary aspects of diet, the omega-6/omega-3 ratio and genetic variation: Nutritional implications for chronic diseases. Biomed. Pharmacother. 2006, 60, 502–507. [Google Scholar] [CrossRef]
- Schulze, M.B.; Minihane, A.M.; Saleh, R.N.M.; Risérus, U. Intake and metabolism of omega-3 and omega-6 polyunsaturated fatty acids: Nutritional implications for cardiometabolic diseases. Lancet Diabetes Endocrinol. 2020, 8, 915–930. [Google Scholar] [CrossRef]
- FSA Panel on Dietetic Products, Nutrition, and Allergies (NDA). Scientific Opinion on Dietary Reference Values for fats, including saturated fatty acids, polyunsaturated fatty acids, monounsaturated fatty acids, trans fatty acids, and cholesterol. EFSA J. 2010, 8, 1461. [Google Scholar] [CrossRef] [Green Version]
- WHO. WHO Technical Report Series 916; The World Health Organisation: Geneva, Switzerland, 2003. [Google Scholar]
- Thompson, M.; Hein, N.; Hanson, C.; Smith, L.M.; Anderson-Berry, A.; Richter, C.K.; Stessy Bisselou, K.; Kusi Appiah, A.; Kris-Etherton, P.; Skulas-Ray, A.C.; et al. Omega-3 Fatty Acid Intake by Age, Gender, and Pregnancy Status in the United States: National Health and Nutrition Examination Survey 2003–2014. Nutrients 2019, 11, 177. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harris, W.S.; von Schacky, C. The Omega-3 Index: A new risk factor for death from coronary heart disease? Prev. Med. 2004, 39, 212–220. [Google Scholar] [CrossRef]
- Harris, W.S. The omega-3 index: From biomarker to risk marker to risk factor. Curr. Atheroscler. Rep. 2009, 11, 411. [Google Scholar] [CrossRef] [PubMed]
- Lane, K.E.; Wilson, M.; Hellon, T.G.; Davies, I.G. Bioavailability and conversion of plant based sources of omega-3 fatty acids—A scoping review to update supplementation options for vegetarians and vegans. Crit. Rev. Food Sci. Nutr. 2021, 1–16. [Google Scholar] [CrossRef]
- OECD/FAO. OECD-FAO Agricultural Outlook 2019-2028; OECD/FAO: Rome, Italy, 2019. [Google Scholar]
- Dorni, C.; Sharma, P.; Saikia, G.; Longvah, T. Fatty acid profile of edible oils and fats consumed in India. Food Chem. 2018, 238, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Beyzi, E.; Gunes, A.; Buyukkilic Beyzi, S.; Konca, Y. Changes in fatty acid and mineral composition of rapeseed (Brassica napus ssp. oleifera L.) oil with seed sizes. Ind. Crop. Prod. 2019, 129, 10–14. [Google Scholar] [CrossRef]
- Saini, R.K.; Rengasamy, K.R.R.; Ko, E.Y.; Kim, J.T.; Keum, Y.S. Korean Maize Hybrids Present Significant Diversity in Fatty Acid Composition: An Investigation to Identify PUFA-Rich Hybrids for a Healthy Diet. Front. Nutr. 2020, 7, 578761. [Google Scholar] [CrossRef] [PubMed]
- Zuk, M.; Richter, D.; Matuła, J.; Szopa, J. Linseed, the multipurpose plant. Ind. Crop. Prod. 2015, 75, 165–177. [Google Scholar] [CrossRef]
- Goyal, A.; Sharma, V.; Upadhyay, N.; Gill, S.; Sihag, M. Flax and flaxseed oil: An ancient medicine & modern functional food. J. Food Sci. Technol. 2014, 51, 1633–1653. [Google Scholar] [CrossRef] [Green Version]
- Zamani Ghaleshahi, A.; Ezzatpanah, H.; Rajabzadeh, G.; Ghavami, M. Comparison and analysis characteristics of flax, perilla and basil seed oils cultivated in Iran. J. Food Sci. Technol. 2020, 57, 1258–1268. [Google Scholar] [CrossRef]
- Marineli, R.D.S.; Moraes, É.A.; Lenquiste, S.A.; Godoy, A.T.; Eberlin, M.N.; Maróstica, M.R., Jr. Chemical characterization and antioxidant potential of Chilean chia seeds and oil (Salvia hispanica L.). LWT—Food Sci. Technol. 2014, 59, 1304–1310. [Google Scholar] [CrossRef]
- Knez Hrnčič, M.; Ivanovski, M.; Cör, D.; Knez, Ž. Chia Seeds (Salvia Hispanica L.): An Overview—Phytochemical Profile, Isolation Methods, and Application. Molecules 2019, 25, 11. [Google Scholar] [CrossRef] [Green Version]
- Gopalam, R.; Tumaney, A.W. Functional characterization of acyltransferases from Salvia hispanica that can selectively catalyze the formation of trilinolenin. Phytochemistry 2021, 186, 112712. [Google Scholar] [CrossRef]
- RV, S.; Kumari, P.; Rupwate, S.D.; Rajasekharan, R.; Srinivasan, M. Exploring Triacylglycerol Biosynthetic Pathway in Developing Seeds of Chia (Salvia hispanica L.): A Transcriptomic Approach. PLoS ONE 2015, 10, e0123580. [Google Scholar] [CrossRef]
- Muñoz, L.A.; Cobos, A.; Diaz, O.; Aguilera, J.M. Chia Seed (Salvia hispanica): An Ancient Grain and a New Functional Food. Food Rev. Int. 2013, 29, 394–408. [Google Scholar] [CrossRef]
- Ullah, R.; Nadeem, M.; Khalique, A.; Imran, M.; Mehmood, S.; Javid, A.; Hussain, J. Nutritional and therapeutic perspectives of Chia (Salvia hispanica L.): A review. J. Food Sci. Technol. 2016, 53, 1750–1758. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zúñiga-López, M.C.; Maturana, G.; Campmajó, G.; Saurina, J.; Núñez, O. Determination of Bioactive Compounds in Sequential Extracts of Chia Leaf (Salvia hispanica L.) Using UHPLC-HRMS (Q-Orbitrap) and a Global Evaluation of Antioxidant In Vitro Capacity. Antioxidants 2021, 10, 1151. [Google Scholar] [CrossRef] [PubMed]
- Melo, D.; Machado, T.B.; Oliveira, M.B.P.P. Chia seeds: An ancient grain trending in modern human diets. Food Funct. 2019, 10, 3068–3089. [Google Scholar] [CrossRef] [PubMed]
- Zettel, V.; Hitzmann, B. Applications of chia (Salvia hispanica L.) in food products. Trends Food Sci. Technol. 2018, 80, 43–50. [Google Scholar] [CrossRef]
- Campbell, M. Camelina–An alternative oil crop. In Biokerosene; Springer: Berlin/Heidelberg, Germany, 2018; pp. 259–275. [Google Scholar]
- Vollmann, J.; Eynck, C. Camelina as a sustainable oilseed crop: Contributions of plant breeding and genetic engineering. Biotechnol. J. 2015, 10, 525–535. [Google Scholar] [CrossRef] [PubMed]
- Diwakar, B.T.; Dutta, P.K.; Lokesh, B.R.; Naidu, K.A. Physicochemical Properties of Garden Cress (Lepidium sativum L.) Seed Oil. J. Am. Oil Chem. Soc. 2010, 87, 539–548. [Google Scholar] [CrossRef]
- Umesha, S.S.; Naidu, K.A. Vegetable oil blends with α-linolenic acid rich Garden cress oil modulate lipid metabolism in experimental rats. Food Chem. 2012, 135, 2845–2851. [Google Scholar] [CrossRef]
- Umesha, S.S.; Manohar, R.S.; Indiramma, A.R.; Akshitha, S.; Naidu, K.A. Enrichment of biscuits with microencapsulated omega-3 fatty acid (Alpha-linolenic acid) rich Garden cress (Lepidium sativum) seed oil: Physical, sensory and storage quality characteristics of biscuits. LWT—Food Sci. Technol. 2015, 62, 654–661. [Google Scholar] [CrossRef]
- Li, S.-S.; Yuan, R.-Y.; Chen, L.-G.; Wang, L.-S.; Hao, X.-H.; Wang, L.-J.; Zheng, X.-C.; Du, H. Systematic qualitative and quantitative assessment of fatty acids in the seeds of 60 tree peony (Paeonia section Moutan DC.) cultivars by GC–MS. Food Chem. 2015, 173, 133–140. [Google Scholar] [CrossRef]
- Wang, S.; Zhu, F.; Kakuda, Y. Sacha inchi (Plukenetia volubilis L.): Nutritional composition, biological activity, and uses. Food Chem. 2018, 265, 316–328. [Google Scholar] [CrossRef]
- Chirinos, R.; Zuloeta, G.; Pedreschi, R.; Mignolet, E.; Larondelle, Y.; Campos, D. Sacha inchi (Plukenetia volubilis): A seed source of polyunsaturated fatty acids, tocopherols, phytosterols, phenolic compounds and antioxidant capacity. Food Chem. 2013, 141, 1732–1739. [Google Scholar] [CrossRef]
- Saini, R.K.; Keum, Y.-S.; Rengasamy, K.R. Profiling of nutritionally important metabolites in green/red and green perilla (Perilla frutescens Britt.) cultivars: A comparative study. Ind. Crop. Prod. 2020, 151, 112441. [Google Scholar] [CrossRef]
- Prabu, S.L.; Umamaheswari, A.; Puratchikody, A. Phytopharmacological potential of the natural gift Moringa oleifera Lam and its therapeutic application: An overview. Asian Pac. J. Trop. Med. 2019, 12, 485–498. [Google Scholar] [CrossRef]
- Nazir, S.; Wani, I.A. Physicochemical characterization of basil (Ocimum basilicum L.) seeds. J. Appl. Res. Med. Aromat. Plants 2021, 22, 100295. [Google Scholar] [CrossRef]
- Dhama, K.; Sharun, K.; Gugjoo, M.B.; Tiwari, R.; Alagawany, M.; Iqbal Yatoo, M.; Thakur, P.; Iqbal, H.M.N.; Chaicumpa, W.; Michalak, I.; et al. A Comprehensive Review on Chemical Profile and Pharmacological Activities of Ocimum basilicum. Food Rev. Int. 2021, 1–29. [Google Scholar] [CrossRef]
- Zhang, Z.-S.; Liu, Y.-L.; Che, L.-M. Characterization of a New α-Linolenic Acid-Rich Oil: Eucommia ulmoides Seed Oil. J. Food Sci. 2018, 83, 617–623. [Google Scholar] [CrossRef]
- Saini, R.K.; Shetty, N.P.; Giridhar, P. GC-FID/MS Analysis of Fatty Acids in Indian Cultivars of Moringa oleifera: Potential Sources of PUFA. J. Am. Oil Chem. Soc. 2014, 91, 1029–1034. [Google Scholar] [CrossRef]
- Kim, D.E.; Shang, X.; Assefa, A.D.; Keum, Y.S.; Saini, R.K. Metabolite profiling of green, green/red, and red lettuce cultivars: Variation in health beneficial compounds and antioxidant potential. Food Res. Int. 2018, 105, 361–370. [Google Scholar] [CrossRef]
- Nemzer, B.; Al-Taher, F.; Abshiru, N. Phytochemical composition and nutritional value of different plant parts in two cultivated and wild purslane (Portulaca oleracea L.) genotypes. Food Chem. 2020, 320, 126621. [Google Scholar] [CrossRef]
- Uddin, M.K.; Juraimi, A.S.; Hossain, M.S.; Nahar, M.A.; Ali, M.E.; Rahman, M.M. Purslane weed (Portulaca oleracea): A prospective plant source of nutrition, omega-3 fatty acid, and antioxidant attributes. Sci. World J. 2014, 2014, 951019. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Petropoulos, S.A.; Karkanis, A.; Fernandes, Â.; Barros, L.; Ferreira, I.C.F.R.; Ntatsi, G.; Petrotos, K.; Lykas, C.; Khah, E. Chemical Composition and Yield of Six Genotypes of Common Purslane (Portulaca oleracea L.): An Alternative Source of Omega-3 Fatty Acids. Plant Foods Hum. Nutr. 2015, 70, 420–426. [Google Scholar] [CrossRef]
- Petropoulos, S.A.; Fernandes, Â.; Arampatzis, D.A.; Tsiropoulos, N.G.; Petrović, J.; Soković, M.; Barros, L.; Ferreira, I.C.F.R. Seed oil and seed oil byproducts of common purslane (Portulaca oleracea L.): A new insight to plant-based sources rich in omega-3 fatty acids. LWT 2020, 123, 109099. [Google Scholar] [CrossRef]
- Prasad, P.; Anjali, P.; Sreedhar, R.V. Plant-based stearidonic acid as sustainable source of omega-3 fatty acid with functional outcomes on human health. Crit. Rev. Food Sci. Nutr. 2020, 61, 1725–1737. [Google Scholar] [CrossRef] [PubMed]
- Rincón-Cervera, M.Á.; Galleguillos-Fernández, R.; González-Barriga, V.; Valenzuela, R.; Speisky, H.; Fuentes, J.; Valenzuela, A. Fatty Acid Profile and Bioactive Compound Extraction in Purple Viper's Bugloss Seed Oil Extracted with Green Solvents. J. Am. Oil Chem. Soc. 2020, 97, 319–327. [Google Scholar] [CrossRef]
- Guil-Guerrero, J.L.; Gómez-Mercado, F.; Ramos-Bueno, R.P.; Rincón-Cervera, M.Á.; Venegas-Venegas, E. Restricted-Range Boraginaceae Species Constitute Potential Sources of Valuable Fatty Acids. J. Am. Oil Chem. Soc. 2014, 91, 301–308. [Google Scholar] [CrossRef]
- Sreedhar, R.V.; Prasad, P.; Reddy, L.P.A.; Rajasekharan, R.; Srinivasan, M. Unravelling a stearidonic acid-rich triacylglycerol biosynthetic pathway in the developing seeds of Buglossoides arvensis: A transcriptomic landscape. Sci. Rep. 2017, 7, 10473. [Google Scholar] [CrossRef] [Green Version]
- Guil-Guerrero, J.L.; Gómez-Mercado, F.; Ramos-Bueno, R.P.; González-Fernández, M.J.; Urrestarazu, M.; Jiménez-Becker, S.; de Bélair, G. Fatty acid profiles and sn-2 fatty acid distribution of γ-linolenic acid-rich Borago species. J. Food Compos. Anal. 2018, 66, 74–80. [Google Scholar] [CrossRef]
- Piskernik, S.; Vidrih, R.; Demsar, L.; Koron, D.; Rogelj, M.; Zontar, T.P. Fatty acid profiles of seeds from different Ribes species. LWT—Food Sci. Technol. 2018, 98, 424–427. [Google Scholar] [CrossRef]
- Park, H.Y.; Kim, D.H.; Saini, R.K.; Gopal, J.; Keum, Y.S.; Sivanesan, I. Micropropagation and Quantification of Bioactive Compounds in Mertensia maritima (L.) Gray. Int. J. Mol. Sci. 2019, 20, 2141. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leyland, B.; Leu, S.; Boussiba, S. Are Thraustochytrids algae? Fungal Biol. 2017, 121, 835–840. [Google Scholar] [CrossRef]
- Lopes da Silva, T.; Moniz, P.; Silva, C.; Reis, A. The Dark Side of Microalgae Biotechnology: A Heterotrophic Biorefinery Platform Directed to ω-3 Rich Lipid Production. Microorganisms 2019, 7, 670. [Google Scholar] [CrossRef] [Green Version]
- Russo, G.L.; Langellotti, A.L.; Oliviero, M.; Sacchi, R.; Masi, P. Sustainable production of food grade omega-3 oil using aquatic protists: Reliability and future horizons. New Biotechnol. 2021, 62, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.; Karageorgou, D.; Katapodis, P.; Sharma, A.; Rova, U.; Christakopoulos, P.; Matsakas, L. Bioprospecting of thraustochytrids for omega-3 fatty acids: A sustainable approach to reduce dependency on animal sources. Trends Food Sci. Technol. 2021, 115, 433–444. [Google Scholar] [CrossRef]
- Gray, R.J. Application for the Authorization of DHA and EPA-Rich Algal oil from Schizochytrium sp.; Martek Biosciences Corporation: Columbia, ML, USA, 2017. [Google Scholar]
- Huang, T.Y.; Lu, W.C.; Chu, I.M. A fermentation strategy for producing docosahexaenoic acid in Aurantiochytrium limacinum SR21 and increasing C22:6 proportions in total fatty acid. Bioresour. Technol. 2012, 123, 8–14. [Google Scholar] [CrossRef]
- FSANZ. DHASCO and ARASCO Oils as Sources of Long-Chain Polyunsaturated Fatty Acids in Infant Formula; Food Standards Australia New Zealand: Canberra, BC, Australia, 2003. [Google Scholar]
- Kiy, T.; Luy, M.; Zeumer, O. Production of Omega-3 Fatty Acids in Microflora of Thraustochytriales Using Modified Media. Google Patents. 2010. Available online: https://data.epo.org/gpi/EP2084290B1 (accessed on 14 October 2021).
- Li, J.; Liu, R.; Chang, G.; Li, X.; Chang, M.; Liu, Y.; Jin, Q.; Wang, X. A strategy for the highly efficient production of docosahexaenoic acid by Aurantiochytrium limacinum SR21 using glucose and glycerol as the mixed carbon sources. Bioresour. Technol. 2015, 177, 51–57. [Google Scholar] [CrossRef]
- Patel, A.; Liefeldt, S.; Rova, U.; Christakopoulos, P.; Matsakas, L. Co-production of DHA and squalene by thraustochytrid from forest biomass. Sci. Rep. 2020, 10, 1992. [Google Scholar] [CrossRef] [PubMed]
- Scott, S.D.; Armenta, R.E.; Berryman, K.T.; Norman, A.W. Use of raw glycerol to produce oil rich in polyunsaturated fatty acids by a thraustochytrid. Enzym. Microb. Technol. 2011, 48, 267–272. [Google Scholar] [CrossRef]
- Ma, X.-N.; Chen, T.-P.; Yang, B.; Liu, J.; Chen, F. Lipid Production from Nannochloropsis. Mar. Drugs 2016, 14, 61. [Google Scholar] [CrossRef] [Green Version]
- Paliwal, C.; Mitra, M.; Bhayani, K.; Bharadwaj, S.V.V.; Ghosh, T.; Dubey, S.; Mishra, S. Abiotic stresses as tools for metabolites in microalgae. Bioresour. Technol. 2017, 244, 1216–1226. [Google Scholar] [CrossRef] [PubMed]
- Mitra, M.; Patidar, S.K.; Mishra, S. Integrated process of two stage cultivation of Nannochloropsis sp. for nutraceutically valuable eicosapentaenoic acid along with biodiesel. Bioresour. Technol. 2015, 193, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Thomas-Hall, S.R.; Schenk, P.M. Phaeodactylum tricornutum microalgae as a rich source of omega-3 oil: Progress in lipid induction techniques towards industry adoption. Food Chem. 2019, 297, 124937. [Google Scholar] [CrossRef] [PubMed]
- Maehre, H.K.; Malde, M.K.; Eilertsen, K.-E.; Elvevoll, E.O. Characterization of protein, lipid and mineral contents in common Norwegian seaweeds and evaluation of their potential as food and feed. J. Sci. Food Agric. 2014, 94, 3281–3290. [Google Scholar] [CrossRef] [PubMed]
- Pereira, H.; Barreira, L.; Figueiredo, F.; Custódio, L.; Vizetto-Duarte, C.; Polo, C.; Rešek, E.; Engelen, A.; Varela, J. Polyunsaturated Fatty Acids of Marine Macroalgae: Potential for Nutritional and Pharmaceutical Applications. Mar. Drugs 2012, 10, 1920–1935. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gosch, B.J.; Magnusson, M.; Paul, N.A.; De Nys, R. Total lipid and fatty acid composition of seaweeds for the selection of species for oil-based biofuel and bioproducts. GCB Bioenergy 2012, 4, 919–930. [Google Scholar] [CrossRef] [Green Version]
- Umesha, S.S.; Monahar, B.; Naidu, K.A. Microencapsulation of α-linolenic acid-rich garden cress seed oil: Physical characteristics and oxidative stability. Eur. J. Lipid Sci. Technol. 2013, 115, 1474–1482. [Google Scholar] [CrossRef]
- Lyashenko, S.; González-Fernández, M.J.; Borisova, S.; Belarbi, E.-H.; Guil-Guerrero, J.L. Mertensia (Boraginaceae) seeds are new sources of γ-linolenic acid and minor functional compounds. Food Chem. 2021, 350, 128635. [Google Scholar] [CrossRef]
- Lim, D.K.Y.; Garg, S.; Timmins, M.; Zhang, E.S.B.; Thomas-Hall, S.R.; Schuhmann, H.; Li, Y.; Schenk, P.M. Isolation and Evaluation of Oil-Producing Microalgae from Subtropical Coastal and Brackish Waters. PLoS ONE 2012, 7, e40751. [Google Scholar] [CrossRef] [PubMed]
- Qiao, H.; Cong, C.; Sun, C.; Li, B.; Wang, J.; Zhang, L. Effect of culture conditions on growth, fatty acid composition and DHA/EPA ratio of Phaeodactylum tricornutum. Aquaculture 2016, 452, 311–317. [Google Scholar] [CrossRef]
- Patel, A.; Mu, L.; Shi, Y.; Rova, U.; Christakopoulos, P.; Matsakas, L. Novel Biorefinery Approach Aimed at Vegetarians Reduces the Dependency on Marine Fish Stocks for Obtaining Squalene and Docosahexaenoic Acid. ACS Sustain. Chem. Eng. 2020, 8, 8803–8813. [Google Scholar] [CrossRef]
- Schmid, M.; Guihéneuf, F.; Stengel, D.B. Fatty acid contents and profiles of 16 macroalgae collected from the Irish Coast at two seasons. J. Appl. Phycol. 2014, 26, 451–463. [Google Scholar] [CrossRef]
- Ruiz-Lopez, N.; Sayanova, O.; Napier, J.A.; Haslam, R.P. Metabolic engineering of the omega-3 long chain polyunsaturated fatty acid biosynthetic pathway into transgenic plants. J. Exp. Bot. 2012, 63, 2397–2410. [Google Scholar] [CrossRef]
- Haslam, R.P.; Ruiz-Lopez, N.; Eastmond, P.; Moloney, M.; Sayanova, O.; Napier, J.A. The modification of plant oil composition via metabolic engineering-better nutrition by design. Plant Biotechnol. J. 2013, 11, 157–168. [Google Scholar] [CrossRef] [Green Version]
- Petrie, J.R.; Shrestha, P.; Belide, S.; Kennedy, Y.; Lester, G.; Liu, Q.; Divi, U.K.; Mulder, R.J.; Mansour, M.P.; Nichols, P.D.; et al. Metabolic Engineering Camelina sativa with Fish Oil-Like Levels of DHA. PLoS ONE 2014, 9, e85061. [Google Scholar] [CrossRef]
- Usher, S.; Han, L.; Haslam, R.P.; Michaelson, L.V.; Sturtevant, D.; Aziz, M.; Chapman, K.D.; Sayanova, O.; Napier, J.A. Tailoring seed oil composition in the real world: Optimising omega-3 long chain polyunsaturated fatty acid accumulation in transgenic Camelina sativa. Sci. Rep. 2017, 7, 6570. [Google Scholar] [CrossRef]
- Han, L.; Usher, S.; Sandgrind, S.; Hassall, K.; Sayanova, O.; Michaelson, L.V.; Haslam, R.P.; Napier, J.A. High level accumulation of EPA and DHA in field-grown transgenic Camelina—A multi-territory evaluation of TAG accumulation and heterogeneity. Plant Biotechnol. J. 2020, 18, 2280–2291. [Google Scholar] [CrossRef]
- Xie, D.; Jackson, E.N.; Zhu, Q. Sustainable source of omega-3 eicosapentaenoic acid from metabolically engineered Yarrowia lipolytica: From fundamental research to commercial production. Appl. Microbiol. Biotechnol. 2015, 99, 1599–1610. [Google Scholar] [CrossRef] [Green Version]
- Ganesan, B.; Brothersen, C.; McMahon, D.J. Fortification of Foods with Omega-3 Polyunsaturated Fatty Acids. Crit. Rev. Food Sci. Nutr. 2014, 54, 98–114. [Google Scholar] [CrossRef]
- Wang, Y.Z.; Fu, S.G.; Wang, S.Y.; Yang, D.J.; Wu, Y.H.S.; Chen, Y.C. Effects of a natural antioxidant, polyphenol-rich rosemary (Rosmarinus officinalis L.) extract, on lipid stability of plant-derived omega-3 fatty-acid rich oil. LWT—Food Sci. Technol. 2018, 89, 210–216. [Google Scholar] [CrossRef]
- Shen, Y.; Lu, T.; Liu, X.-Y.; Zhao, M.-T.; Yin, F.-W.; Rakariyatham, K.; Zhou, D.-Y. Improving the oxidative stability and lengthening the shelf life of DHA algae oil with composite antioxidants. Food Chem. 2020, 313, 126139. [Google Scholar] [CrossRef]
- Nain, C.W.; Berdal, G.; Thao, P.T.P.; Mignolet, E.; Buchet, M.; Page, M.; Larondelle, Y. Green Tea Extract Enhances the Oxidative Stability of DHA-Rich Oil. Antioxidants 2021, 10, 982. [Google Scholar] [CrossRef] [PubMed]
- Mikołajczak, N.; Sobiechowska, D.A.; Tańska, M. Edible flowers as a new source of natural antioxidants for oxidative protection of cold-pressed oils rich in omega-3 fatty acids. Food Res. Int. 2020, 134, 109216. [Google Scholar] [CrossRef]
- Yui, Y.; Miyazaki, S.; Ma, Y.; Ohira, M.; Fiehn, O.; Ikegami, T.; McCalley, D.V.; Tanaka, N. Distinction of synthetic dl-α-tocopherol from natural vitamin E (d-α-tocopherol) by reversed-phase liquid chromatography. Enhanced selectivity of a polymeric C18 stationary phase at low temperature and/or at high pressure. J. Chromatogr. A 2016, 1450, 45–52. [Google Scholar] [CrossRef]
- Jurić, S.; Jurić, M.; Siddique, M.A.B.; Fathi, M. Vegetable Oils Rich in Polyunsaturated Fatty Acids: Nanoencapsulation Methods and Stability Enhancement. Food Rev. Int. 2020, 1–38. [Google Scholar] [CrossRef]
- Kaushik, P.; Dowling, K.; Barrow, C.J.; Adhikari, B. Microencapsulation of omega-3 fatty acids: A review of microencapsulation and characterization methods. J. Funct. Foods 2015, 19, 868–881. [Google Scholar] [CrossRef]
- Geranpour, M.; Assadpour, E.; Jafari, S.M. Recent advances in the spray drying encapsulation of essential fatty acids and functional oils. Trends Food Sci. Technol. 2020, 102, 71–90. [Google Scholar] [CrossRef]
- Chang, C.; Nickerson, M.T. Encapsulation of omega 3-6-9 fatty acids-rich oils using protein-based emulsions with spray drying. J. Food Sci. Technol. 2018, 55, 2850–2861. [Google Scholar] [CrossRef] [PubMed]
- Feizollahi, E.; Hadian, Z.; Honarvar, Z. Food fortification with omega-3 fatty acids; microencapsulation as an addition method. Curr. Nutr. Food Sci. 2018, 14, 90–103. [Google Scholar] [CrossRef]
- Bakry, A.M.; Abbas, S.; Ali, B.; Majeed, H.; Abouelwafa, M.Y.; Mousa, A.; Liang, L. Microencapsulation of Oils: A Comprehensive Review of Benefits, Techniques, and Applications. Compr. Rev. Food Sci. Food Saf. 2016, 15, 143–182. [Google Scholar] [CrossRef]
- Rodríguez, J.; Martín, M.J.; Ruiz, M.A.; Clares, B. Current encapsulation strategies for bioactive oils: From alimentary to pharmaceutical perspectives. Food Res. Int. 2016, 83, 41–59. [Google Scholar] [CrossRef]
- Gulotta, A.; Saberi, A.H.; Nicoli, M.C.; McClements, D.J. Nanoemulsion-Based Delivery Systems for Polyunsaturated (ω-3) Oils: Formation Using a Spontaneous Emulsification Method. J. Agric. Food Chem. 2014, 62, 1720–1725. [Google Scholar] [CrossRef] [PubMed]
- Inapurapu, S.P.; Ibrahim, A.; Kona, S.R.; Pawar, S.C.; Bodiga, S.; Bodiga, V.L. Development and characterization of ω-3 fatty acid nanoemulsions with improved physicochemical stability and bioaccessibility. Colloids Surf. A Physicochem. Eng. Asp. 2020, 606, 125515. [Google Scholar] [CrossRef]
- Cholewski, M.; Tomczykowa, M.; Tomczyk, M. A Comprehensive Review of Chemistry, Sources and Bioavailability of Omega-3 Fatty Acids. Nutrients 2018, 10, 1662. [Google Scholar] [CrossRef] [Green Version]
- Schuchardt, J.P.; Hahn, A. Bioavailability of long-chain omega-3 fatty acids. Prostaglandins Leukot. Essent. Fat. Acids 2013, 89, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Lee-Chang, K.J.; Taylor, M.C.; Drummond, G.; Mulder, R.J.; Mansour, M.P.; Brock, M.; Nichols, P.D. Docosahexaenoic Acid Is Naturally Concentrated at the sn-2 Position in Triacylglycerols of the Australian Thraustochytrid Aurantiochytrium sp. Strain TC 20. Mar. Drugs 2021, 19, 382. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.; Jin, Q.; Wang, X.; Akoh, C.C. High Sn-2 Docosahexaenoic Acid Lipids for Brain Benefits, and Their Enzymatic Syntheses: A Review. Engineering 2020, 6, 424–431. [Google Scholar] [CrossRef]
- Alfieri, A.; Imperlini, E.; Nigro, E.; Vitucci, D.; Orrù, S.; Daniele, A.; Buono, P.; Mancini, A. Effects of Plant Oil Interesterified Triacylglycerols on Lipemia and Human Health. Int. J. Mol. Sci. 2018, 19, 104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuhnt, K.; Fuhrmann, C.; Köhler, M.; Kiehntopf, M.; Jahreis, G. Dietary Echium Oil Increases Long-Chain n–3 PUFAs, Including Docosapentaenoic Acid, in Blood Fractions and Alters Biochemical Markers for Cardiovascular Disease Independently of Age, Sex, and Metabolic Syndrome. J. Nutr. 2014, 144, 447–460. [Google Scholar] [CrossRef] [Green Version]
- Dittrich, M.; Jahreis, G.; Bothor, K.; Drechsel, C.; Kiehntopf, M.; Blüher, M.; Dawczynski, C. Benefits of foods supplemented with vegetable oils rich in α-linolenic, stearidonic or docosahexaenoic acid in hypertriglyceridemic subjects: A double-blind, randomized, controlled trail. Eur. J. Nutr. 2015, 54, 881–893. [Google Scholar] [CrossRef]
- Maki, K.C.; Yurko-Mauro, K.; Dicklin, M.R.; Schild, A.L.; Geohas, J.G. A new, microalgal DHA- and EPA-containing oil lowers triacylglycerols in adults with mild-to-moderate hypertriglyceridemia. Prostaglandins Leukot. Essent. Fat. Acids 2014, 91, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Ryan, L.; Symington, A.M. Algal-oil supplements are a viable alternative to fish-oil supplements in terms of docosahexaenoic acid (22:6n-3; DHA). J. Funct. Foods 2015, 19, 852–858. [Google Scholar] [CrossRef]
- Khandelwal, S.; Kondal, D.; Chaudhry, M.; Patil, K.; Swamy, M.K.; Metgud, D.; Jogalekar, S.; Kamate, M.; Divan, G.; Gupta, R.; et al. Effect of Maternal Docosahexaenoic Acid (DHA) Supplementation on Offspring Neurodevelopment at 12 Months in India: A Randomized Controlled Trial. Nutrients 2020, 12, 3041. [Google Scholar] [CrossRef]
- Dewell, A.; Marvasti, F.F.; Harris, W.S.; Tsao, P.; Gardner, C.D. Low- and High-Dose Plant and Marine (n-3) Fatty Acids Do Not Affect Plasma Inflammatory Markers in Adults with Metabolic Syndrome. J. Nutr. 2011, 141, 2166–2171. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ghobadi, S.; Hassanzadeh-Rostami, Z.; Mohammadian, F.; Zare, M.; Faghih, S. Effects of Canola Oil Consumption on Lipid Profile: A Systematic Review and Meta-Analysis of Randomized Controlled Clinical Trials. J. Am. Coll. Nutr. 2019, 38, 185–196. [Google Scholar] [CrossRef]
- Günç Ergönül, P.; Aksoylu Özbek, Z. Chapter 21—Cold pressed camelina (Camelina sativa L.) seed oil. In Cold Pressed Oils; Ramadan, M.F., Ed.; Academic Press: Cambridge, MA, USA, 2020; pp. 255–266. [Google Scholar] [CrossRef]
- Shetty, U.S.; Akhilender, N.K. Garden cress (Lepidium sativum L.) Seed Oil: Alternative Source for ALA. FASEB J. 2017, 31, 971.12–971.12. [Google Scholar] [CrossRef]
- Copetta, A.; Bazzicalupo, M.; Cassetti, A.; Marchioni, I.; Mascarello, C.; Cornara, L.; Pistelli, L.; Ruffoni, B. Plant Production and Leaf Anatomy of Mertensia maritima (L.) Gray: Comparison of In Vitro Culture Methods to Improve Acclimatization. Horticulturae 2021, 7, 111. [Google Scholar] [CrossRef]
| Company | Ingredient Brand | Major n−3 Fatty Acids |
|---|---|---|
| FrieslandCampina N.V. (Amersfoort, Netherlands) | Vana®-Sana algae DHA 11 IF | Microalgal derived DHA |
| Algarithm Ingredients, Inc. (Saskatoon, Saskatchewan) | Betamega³ | Microalgal oil powder (120 mg DHA/g) |
| Gamma³ | Microalgal DHA emulsions (400 mg DHA/g emulsion) | |
| Cubiq Foods (Granollers. Barcelona) | Go!Mega3® | Microalgal DHA+EPA (2% w/w) |
| Seanova (Finistère, Brittany) | Algal DHA powder H100 | 100 mg/g DHA from Schizochytrium sp. |
| Chia powder-125 | 60 mg/g ALA from chia seeds | |
| Chia powder-435 | 55 mg/g ALA from chia seeds |
| n−3 PUFA | Commercially Available Product | Dietary Uses | Reference |
|---|---|---|---|
| ALA | Canola seed oil | Vegetable oil | [152] |
| Flax seeds and seed oil | Baked goods, juices, dairy products, and dry pasta products | [59] | |
| Chia seeds and seed oil | Baked and dairy products as emulsifier and stabilizer | [68,69] | |
| Camelina seed oil | Edible oil, food supplement | [153] | |
| Garden cress seed oil | Salad dressings | [154] | |
| Perilla seed oil | Edible oil | [60] | |
| Purslane plant (stems and leaves) | Green leafy vegetable | [85] | |
| SDA | Echium plantagineum oil | Salad dressings/smoothies | [90] |
| Buglossoides arvensis oil (Ahiflower® oil) | Oil for salad dressing, soft-gel capsules for dietary supplement | [45] | |
| Foliage of Mertensia maritima | Culinary recipes | [155] | |
| EPA | Microaglage Nannochloropsis sp. | Dietary supplement | http://jemmaxnutraceuticals.com/ [Accessed 14 Oct 2021] |
| DHA | DHASCO-B® nutritional oil and powder, and life’sDHA® vegetarian capsules from microalgae | Dietary supplement | https://www.dsm.com/ [Accessed 14 Oct 2021] |
| Vegan Omega 3 DHA capsules from Thraustochytrid Schizochytrium sp. | Dietary supplement | https://omvits.com/ [Accessed 14 Oct 2021]; https://www.naturopathica.com.au/ [Accessed 14 Oct 2021] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saini, R.K.; Prasad, P.; Sreedhar, R.V.; Akhilender Naidu, K.; Shang, X.; Keum, Y.-S. Omega−3 Polyunsaturated Fatty Acids (PUFAs): Emerging Plant and Microbial Sources, Oxidative Stability, Bioavailability, and Health Benefits—A Review. Antioxidants 2021, 10, 1627. https://doi.org/10.3390/antiox10101627
Saini RK, Prasad P, Sreedhar RV, Akhilender Naidu K, Shang X, Keum Y-S. Omega−3 Polyunsaturated Fatty Acids (PUFAs): Emerging Plant and Microbial Sources, Oxidative Stability, Bioavailability, and Health Benefits—A Review. Antioxidants. 2021; 10(10):1627. https://doi.org/10.3390/antiox10101627
Chicago/Turabian StyleSaini, Ramesh Kumar, Parchuri Prasad, Reddampalli Venkataramareddy Sreedhar, Kamatham Akhilender Naidu, Xiaomin Shang, and Young-Soo Keum. 2021. "Omega−3 Polyunsaturated Fatty Acids (PUFAs): Emerging Plant and Microbial Sources, Oxidative Stability, Bioavailability, and Health Benefits—A Review" Antioxidants 10, no. 10: 1627. https://doi.org/10.3390/antiox10101627
APA StyleSaini, R. K., Prasad, P., Sreedhar, R. V., Akhilender Naidu, K., Shang, X., & Keum, Y.-S. (2021). Omega−3 Polyunsaturated Fatty Acids (PUFAs): Emerging Plant and Microbial Sources, Oxidative Stability, Bioavailability, and Health Benefits—A Review. Antioxidants, 10(10), 1627. https://doi.org/10.3390/antiox10101627









