Protective Effect of Polymethoxyflavones Isolated from Kaempferia parviflora against TNF-α-Induced Human Dermal Fibroblast Damage
Abstract
:1. Introduction
2. Materials and Methods
2.1. Extraction Methods
2.2. High-Performance Liquid Chromatography (HPLC) Analysis
2.3. Cell Culture and Drug Preparation
2.4. Cell Viability
2.5. Intracellular ROS Assay
2.6. Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR)
2.7. Enzyme-Linked Immunosorbent Assay (ELISA)
2.8. Western Blotting
2.9. Immunofluorescence Staining
2.10. Statistical Methods
3. Results
3.1. Polymethoxyflavones in KP
3.2. Effect of KP-Derived Polymethoxyflavones on the Viability of HDFs
3.3. Effect of DMF, TMF, and PMF on MMP-1 Expression in TNF-α-Stimulated HDFs
3.4. Effect of DMF, TMF, and PMF on COLIA1 Expression in TNF-α-Stimulated HDFs
3.5. Effect of TMF on TNF-α-Induced Pro-Inflammatory Mediators in HDFs
3.6. Effect of TMF on TNF-α-Induced ROS Production in HDFs
3.7. Effect of TMF on TNF-α-Induced Phosphorylation of Mitogen-Activated Protein Kinase (MAPKs) in HDFs
3.8. Effect of TMF on the Expression and Phosphorylation of c-Fos and c-Jun in TNF-α-Stimulated HDFs
3.9. Effect of TMF on TNF-α-Induced Phosphorylation and Translocation of NF-κB in HDFs
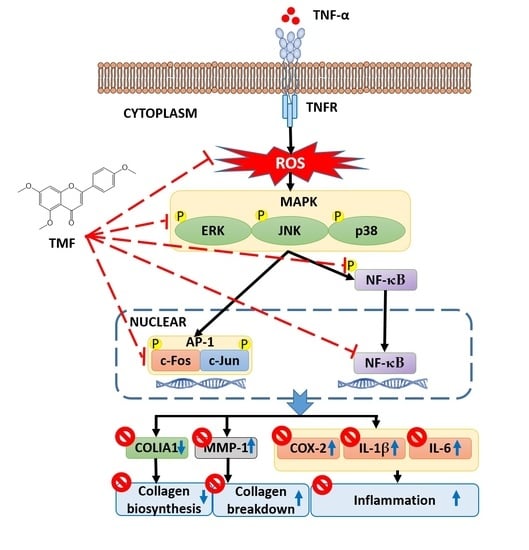
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sjerobabski-Masnec, I.; Šitum, M. Skin aging. Acta Clin. Croat. 2010, 49, 515–518. [Google Scholar] [PubMed]
- Quan, T.; Fisher, G.J. Role of age-associated alterations of the dermal extracellular matrix microenvironment in human skin aging: A mini-review. Gerontology 2015, 61, 427–434. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Imokawa, G. Mechanism of UVB-induced wrinkling of the skin: Paracrine cytokine linkage between keratinocytes and fibroblasts leading to the stimulation of elastase. J. Investig. Dermatol. Symp. Proc. 2009, 14, 36–43. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Borg, M.; Brincat, S.; Camilleri, G.; Schembri-Wismayer, P.; Brincat, M.; Calleja-Agius, J. The role of cytokines in skin aging. Climacteric 2013, 16, 514–521. [Google Scholar] [CrossRef]
- Meephansan, J.; Subpayasarn, U.; Komine, M.; Ohtsuki, M. Pathogenic Role of Cytokines and Effect of Their Inhibition in Psoriasis. In An Interdisciplinary Approach to Psoriasis; Chiriac, A., Ed.; IntechOpen: Rijeka, Croatia, 2017; p. 41. [Google Scholar]
- Brotas, A.M.; Cunha, J.M.T.; Lago, E.H.J.; Machado, C.C.N.; Carneiro, S.C.d.S. Tumor necrosis factor-alpha and the cytokine network in psoriasis. An. Bras. Dermatol. 2012, 87, 673–683. [Google Scholar] [CrossRef] [Green Version]
- Goldminz, A.; Au, S.; Kim, N.; Gottlieb, A.; Lizzul, P. NF-κB: An essential transcription factor in psoriasis. J. Dermatol. Sci. 2013, 69, 89–94. [Google Scholar] [CrossRef]
- Zhao, Y.; Yang, Z.; Lang, B.; Meng, M.S.W.; Xue, D.; Gao, L.; Yang, L. Skincare plants of the Naxi of NW Yunnan, China. Plant Divers. 2020, 42, 473–478. [Google Scholar] [CrossRef]
- Saokaew, S.; Wilairat, P.; Raktanyakan, P.; Dilokthornsakul, P.; Dhippayom, T.; Kongkaew, C.; Sruamsiri, R.; Chuthaputti, A.; Chaiyakunapruk, N. Clinical effects of Krachaidum (Kaempferia parviflora): A systematic review. J. Evid. Based Complement. Alternat. Med. 2017, 22, 413–428. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nicoll, R.; Henein, M.Y. Ginger (Zingiber officinale Roscoe): A hot remedy for cardiovascular disease? Int. J. Cardiol. 2009, 131, 408–409. [Google Scholar] [CrossRef] [PubMed]
- Akase, T.; Shimada, T.; Terabayashi, S.; Ikeya, Y.; Sanada, H.; Aburada, M. Antiobesity effects of Kaempferia parviflora in spontaneously obese type II diabetic mice. Int. J. Cardiol. 2011, 65, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Yenjai, C.; Prasanphen, K.; Daodee, S.; Wongpanich, V.; Kittakoop, P. Bioactive flavonoids from Kaempferia parviflora. Fitoterapia 2004, 75, 89–92. [Google Scholar] [CrossRef] [PubMed]
- Asamenew, G.; Kim, H.-W.; Lee, M.-K.; Lee, S.-H.; Kim, Y.J.; Cha, Y.-S.; Yoo, S.M.; Kim, J.-B. Characterization of phenolic compounds from normal ginger (Zingiber officinale Rosc.) and black ginger (Kaempferia parviflora Wall.) using UPLC–DAD–QToF–MS. Eur. Food Res. Technol. 2019, 245, 653–665. [Google Scholar] [CrossRef] [Green Version]
- Pham, N.K.; Nguyen, H.T.; Nguyen, Q.B. A review on the ethnomedicinal uses, phytochemistry and pharmacology of plant species belonging to Kaempferia L. genus (Zingiberaceae). Pharm. Sci. Asia 2021, 48, 1–24. [Google Scholar] [CrossRef]
- Azuma, T.; Tanaka, Y.; Kikuzaki, H. Phenolic glycosides from Kaempferia parviflora. Phytochemistry 2008, 69, 2743–2748. [Google Scholar] [CrossRef]
- Chaipech, S.; Morikawa, T.; Ninomiya, K.; Yoshikawa, M.; Pongpiriyadacha, Y.; Hayakawa, T.; Muraoka, O. New flav-3-en-3-ol glycosides, kaempferiaosides C and D, and acetophenone glycosides, kaempferiaosides E and F, from the rhizomes of Kaempferia parviflora. J. Nat. Med. 2012, 66, 486–492. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Li, H.; Li, W.; Feng, S.; Deng, D. Kaempferia parviflora and its methoxyflavones: Chemistry and biological activities. Evid. Based Complement. Alternat. Med. 2018, 2018, 4057456. [Google Scholar] [CrossRef] [Green Version]
- Ninomiya, K.; Matsumoto, T.; Chaipech, S.; Miyake, S.; Katsuyama, Y.; Tsuboyama, A.; Pongpiriyadacha, Y.; Hayakawa, T.; Muraoka, O.; Morikawa, T. Simultaneous quantitative analysis of 12 methoxyflavones with melanogenesis inhibitory activity from the rhizomes of Kaempferia parviflora. J. Nat. Med. 2016, 70, 179–189. [Google Scholar] [CrossRef] [PubMed]
- Toda, K.; Hitoe, S.; Takeda, S.; Shimoda, H. Black ginger extract increases physical fitness performance and muscular endurance by improving inflammation and energy metabolism. Heliyon 2016, 2, e00115. [Google Scholar] [CrossRef] [Green Version]
- Youn, K.; Lee, J.; Ho, C.-T.; Jun, M. Discovery of polymethoxyflavones from black ginger (Kaempferia parviflora) as potential β-secretase (BACE1) inhibitors. J. Funct. Foods 2016, 20, 567–574. [Google Scholar] [CrossRef]
- Horigome, S.; Yoshida, I.; Tsuda, A.; Harada, T.; Yamaguchi, A.; Yamazaki, K.; Inohana, S.; Isagawa, S.; Kibune, N.; Satoyama, T. Identification and evaluation of anti-inflammatory compounds from Kaempferia parviflora. Biosci. Biotechnol. Biochem. 2014, 78, 851–860. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Kim, M.-B.; Kim, C.; Kim, J.; Hwang, J.-K. 5, 7-Dimethoxyflavone attenuates obesity by inhibiting adipogenesis in 3T3-L1 adipocytes and high-fat diet-induced obese C57BL/6J mice. J. Med. Food. 2016, 19, 1111–1119. [Google Scholar] [CrossRef] [PubMed]
- Park, J.E.; Pyun, H.B.; Woo, S.W.; Jeong, J.H.; Hwang, J.K. The protective effect of Kaempferia parviflora extract on UVB-induced skin photoaging in hairless mice. Photodermatol. Photoimmunol. Photomed. 2014, 30, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.K.; Mun, S.; Kim, M.S.; Kim, M.B.; Sa, B.K.; Hwang, J.K. 5, 7-Dimethoxyflavone, an activator of PPARα/γ, inhibits UVB-induced MMP expression in human skin fibroblast cells. Exp. Dermatol. 2012, 21, 211–216. [Google Scholar] [CrossRef] [PubMed]
- Lim, H.; Kim, H.P. Inhibition of mammalian collagenase, matrix metalloproteinase-1, by naturally-occurring flavonoids. Planta Med. 2007, 73, 1267–1274. [Google Scholar] [CrossRef] [PubMed]
- Sim, G.-S.; Lee, B.-C.; Cho, H.S.; Lee, J.W.; Kim, J.-H.; Lee, D.-H.; Kim, J.-H.; Pyo, H.-B.; Moon, D.C.; Oh, K.-W. Structure activity relationship of antioxidative property of flavonoids and inhibitory effect on matrix metalloproteinase activity in UVA-irradiated human dermal fibroblast. Arch. Pharm. Res. 2007, 30, 290–298. [Google Scholar] [CrossRef]
- Dal Belo, S.; Gaspar, L.; Campos, P.M.; Marty, J.-P. Skin penetration of epigallocatechin-3-gallate and quercetin from green tea and Ginkgo biloba extracts vehiculated in cosmetic formulations. Skin Pharmacol. Physiol. 2009, 22, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.W.; Lee, J.; Park, Y.I. 7, 8-Dihydroxyflavone attenuates TNF-α-induced skin aging in Hs68 human dermal fibroblast cells via down-regulation of the MAPKs/Akt signaling pathways. Biomed. Pharmacother. 2017, 95, 1580–1587. [Google Scholar] [CrossRef]
- Na, J.; Bak, D.H.; Im, S.I.; Choi, H.; Hwang, J.H.; Kong, S.Y.; No, Y.A.; Lee, Y.; Kim, B.J. Anti-apoptotic effects of glycosaminoglycans via inhibition of ERK/AP-1 signaling in TNF-α-stimulated human dermal fibroblasts. Int. J. Mol. Med. 2018, 41, 3090–3098. [Google Scholar] [CrossRef]
- Shim, J.H. Prostaglandin E2 induces skin aging via E-prostanoid 1 in normal human dermal fibroblasts. Int. J. Mol. Sci. 2019, 20, 5555. [Google Scholar] [CrossRef] [Green Version]
- Gendrisch, F.; Esser, P.R.; Schempp, C.M.; Wölfle, U. Luteolin as a modulator of skin aging and inflammation. BioFactors 2021, 47, 170–180. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.-W.; Kwon, S.-H.; Choi, J.-Y.; Na, J.-I.; Huh, C.-H.; Choi, H.-R.; Park, K.-C. Molecular mechanisms of dermal aging and antiaging approaches. Int. J. Mol. Sci. 2019, 20, 2126. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ganceviciene, R.; Liakou, A.I.; Theodoridis, A.; Makrantonaki, E.; Zouboulis, C.C. Skin anti-aging strategies. Dermato-Endocrinology 2012, 4, 308–319. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, G.; Tan, F.; Zhang, Q.; Tan, A.; Cheng, Y.; Zhou, Q.; Liu, M.; Tan, X.; Huang, L.; Rouseff, R. Protective effects of polymethoxyflavone-rich cold-pressed orange peel oil against ultraviolet B-induced photoaging on mouse skin. J. Funct. Foods 2020, 67, 103834. [Google Scholar] [CrossRef]
- Van den Worm, E.; Beukelman, C.; Van den Berg, A.J.; Kroes, B.H.; Labadie, R.P.; Van Dijk, H. Effects of methoxylation of apocynin and analogs on the inhibition of reactive oxygen species production by stimulated human neutrophils. Eur. J. Pharmacol 2001, 433, 225–230. [Google Scholar] [CrossRef]
- Hwang, E.; Lin, P.; Ngo, H.T.; Gao, W.; Wang, Y.-S.; Yu, H.-S.; Yi, T.-H. Icariin and icaritin recover UVB-induced photoaging by stimulating Nrf2/ARE and reducing AP-1 and NF-κB signaling pathways: A comparative study on UVB-irradiated human keratinocytes. Photochem. Photobiol. Sci. 2018, 17, 1396–1408. [Google Scholar] [CrossRef] [PubMed]
- Fisher, G.J.; Voorhees, J.J. Molecular mechanisms of photoaging and its prevention by retinoic acid: Ultraviolet irradiation induces MAP kinase signal transduction cascades that induce Ap-1-regulated matrix metalloproteinases that degrade human skin in vivo. J. Investig. Dermatol. Symp. Proc. 1998, 3, 61–68. [Google Scholar]
- Rabe, J.H.; Mamelak, A.J.; McElgunn, P.J.; Morison, W.L.; Sauder, D.N. Photoaging: Mechanisms and repair. J. Am. Acad. Dermatol. 2006, 55, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K.; Asamitsu, K.; Uranishi, H.; Iddamalgoda, A.; Ito, K.; Kojima, H.; Okamoto, T. Protecting skin photoaging by NF-κB inhibitor. Curr. Drug Metab. 2010, 11, 431–435. [Google Scholar] [CrossRef] [PubMed]
- Kandhaya-Pillai, R.; Miro-Mur, F.; Alijotas-Reig, J.; Tchkonia, T.; Kirkland, J.L.; Schwartz Jr, S. TNFα-senescence initiates a STAT-dependent positive feedback loop, leading to a sustained interferon signature, DNA damage, and cytokine secretion. Aging (Albany NY) 2017, 9, 2411. [Google Scholar] [CrossRef] [Green Version]
- Narendhirakannan, R.; Hannah, M.A.C. Oxidative stress and skin cancer: An overview. Indian J. Clin. Biochem. 2013, 28, 110–115. [Google Scholar] [CrossRef] [PubMed] [Green Version]











| Gene | Sense Primer Sequence (5′–3′) | Antisense Primer Sequence (5′–3′) |
|---|---|---|
| MMP-1 | ATTCTACTGATATCGGGGCTTT | ATGTCCTTGGGGTATCCGTGTA |
| COLIA1 | CTCGAGGTGGACACCACCCT | CAGCTGGATGGCCACATCGG |
| IL-1β | CTGTCCTGCGTGTTGAAAGA | TTCTGCTTGAGAGGTGCTGA-3 |
| IL-6 | CTCCTTCTCCACAAGCGCC | GCCGAAGAGCCCTCAGGC |
| β-actin | AGAGATGGCCACGGCTGCTT | ATTTGCGGTGGACGATGGAG |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Phung, H.M.; Lee, S.; Hong, S.; Lee, S.; Jung, K.; Kang, K.S. Protective Effect of Polymethoxyflavones Isolated from Kaempferia parviflora against TNF-α-Induced Human Dermal Fibroblast Damage. Antioxidants 2021, 10, 1609. https://doi.org/10.3390/antiox10101609
Phung HM, Lee S, Hong S, Lee S, Jung K, Kang KS. Protective Effect of Polymethoxyflavones Isolated from Kaempferia parviflora against TNF-α-Induced Human Dermal Fibroblast Damage. Antioxidants. 2021; 10(10):1609. https://doi.org/10.3390/antiox10101609
Chicago/Turabian StylePhung, Hung Manh, Sullim Lee, Sukyung Hong, Sojung Lee, Kiwon Jung, and Ki Sung Kang. 2021. "Protective Effect of Polymethoxyflavones Isolated from Kaempferia parviflora against TNF-α-Induced Human Dermal Fibroblast Damage" Antioxidants 10, no. 10: 1609. https://doi.org/10.3390/antiox10101609
APA StylePhung, H. M., Lee, S., Hong, S., Lee, S., Jung, K., & Kang, K. S. (2021). Protective Effect of Polymethoxyflavones Isolated from Kaempferia parviflora against TNF-α-Induced Human Dermal Fibroblast Damage. Antioxidants, 10(10), 1609. https://doi.org/10.3390/antiox10101609