Self-Paced Online vs. Cue-Based Offline Brain–Computer Interfaces for Inducing Neural Plasticity
Abstract
1. Introduction
2. Methods
2.1. Participants
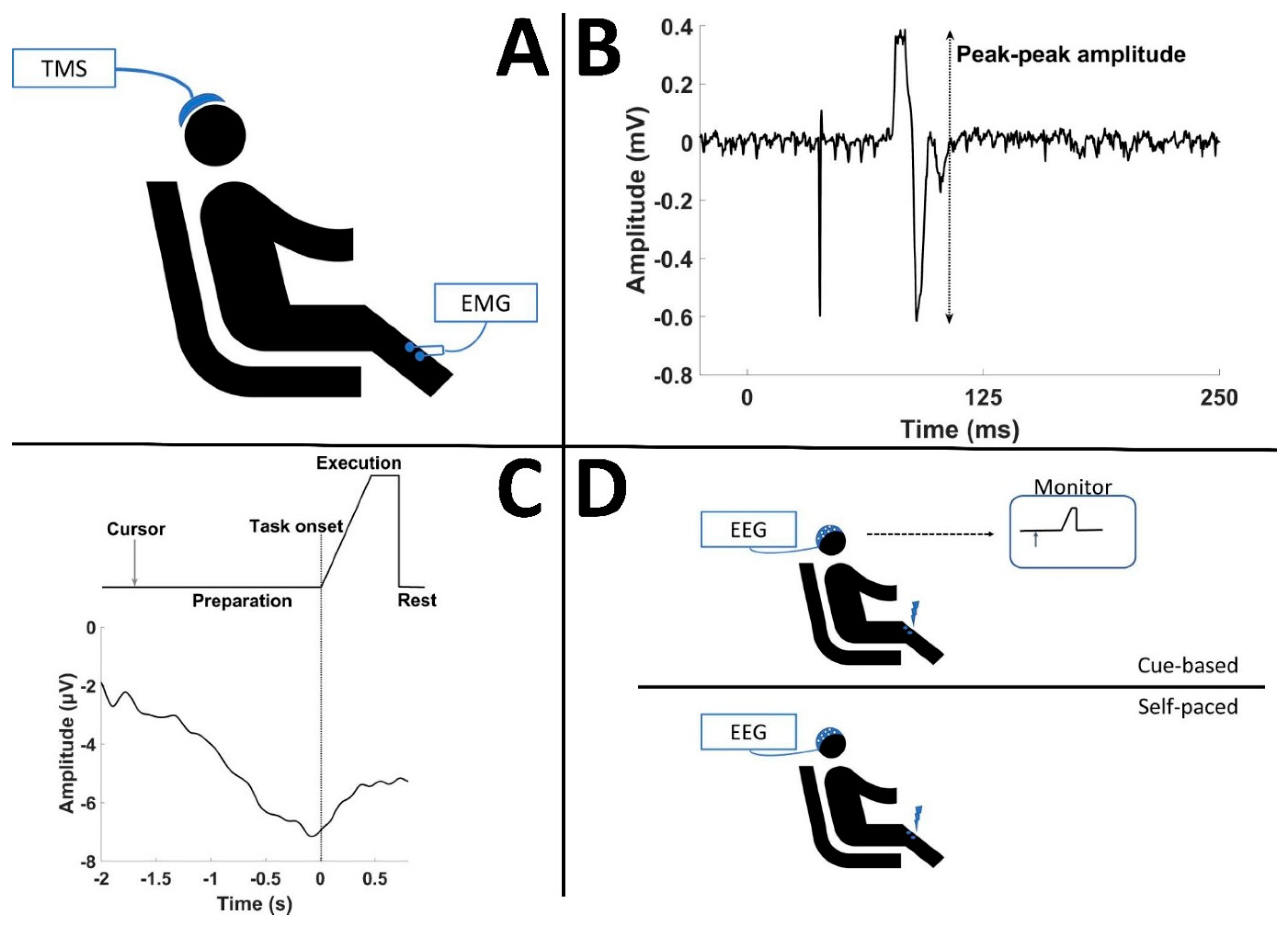
2.2. Measurements and Stimulation
2.2.1. EEG
2.2.2. Motor-Evoked Potentials
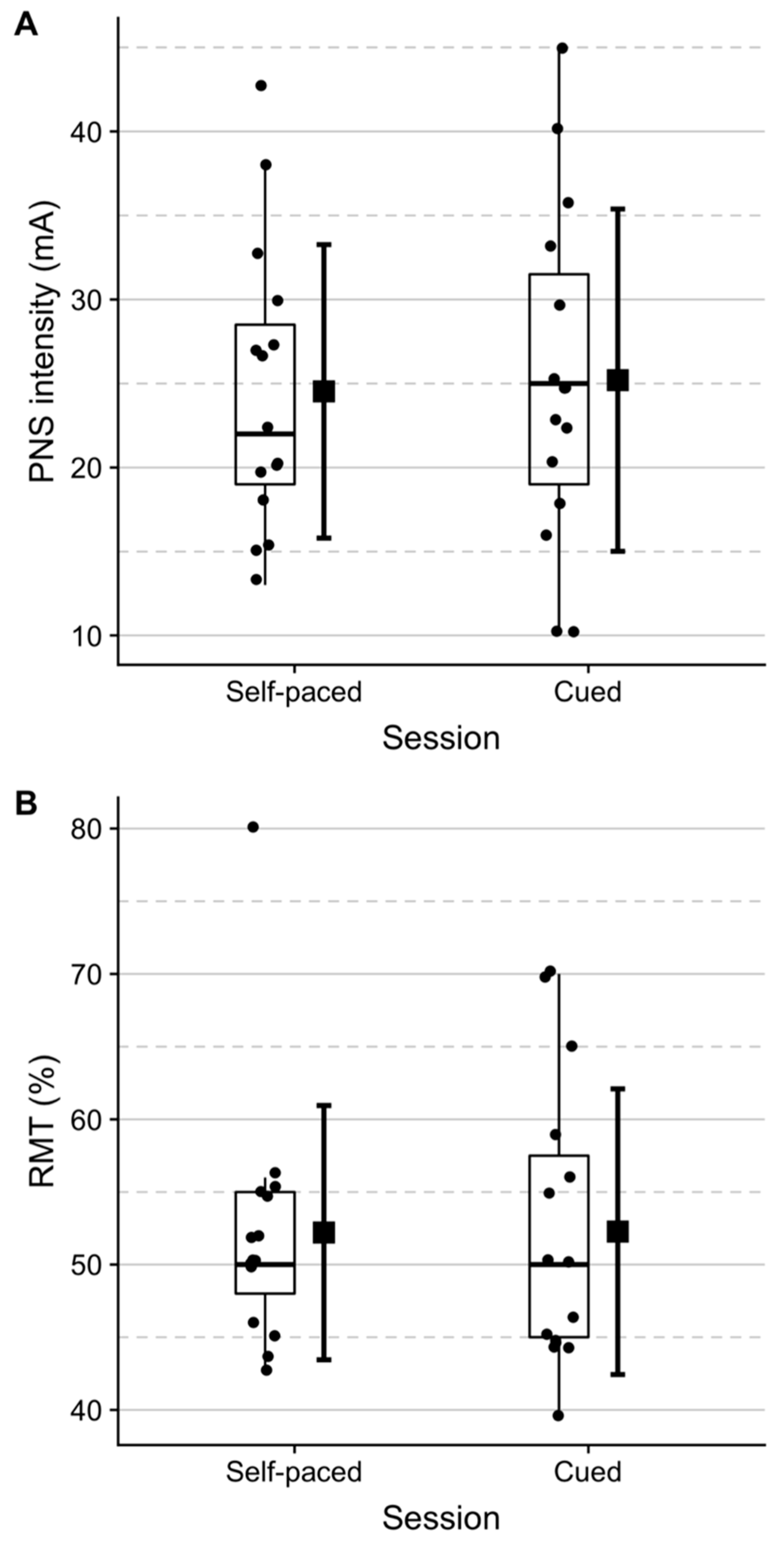
2.2.3. Transcranial Magnetic Stimulation
2.2.4. Peripheral Nerve Stimulation
2.3. Experimental Setup
2.4. Brain–Computer Interface Systems
2.4.1. Cue-Based BCI (Offline)
2.4.2. Self-Paced BCI (Online)
2.5. Statistics
3. Results
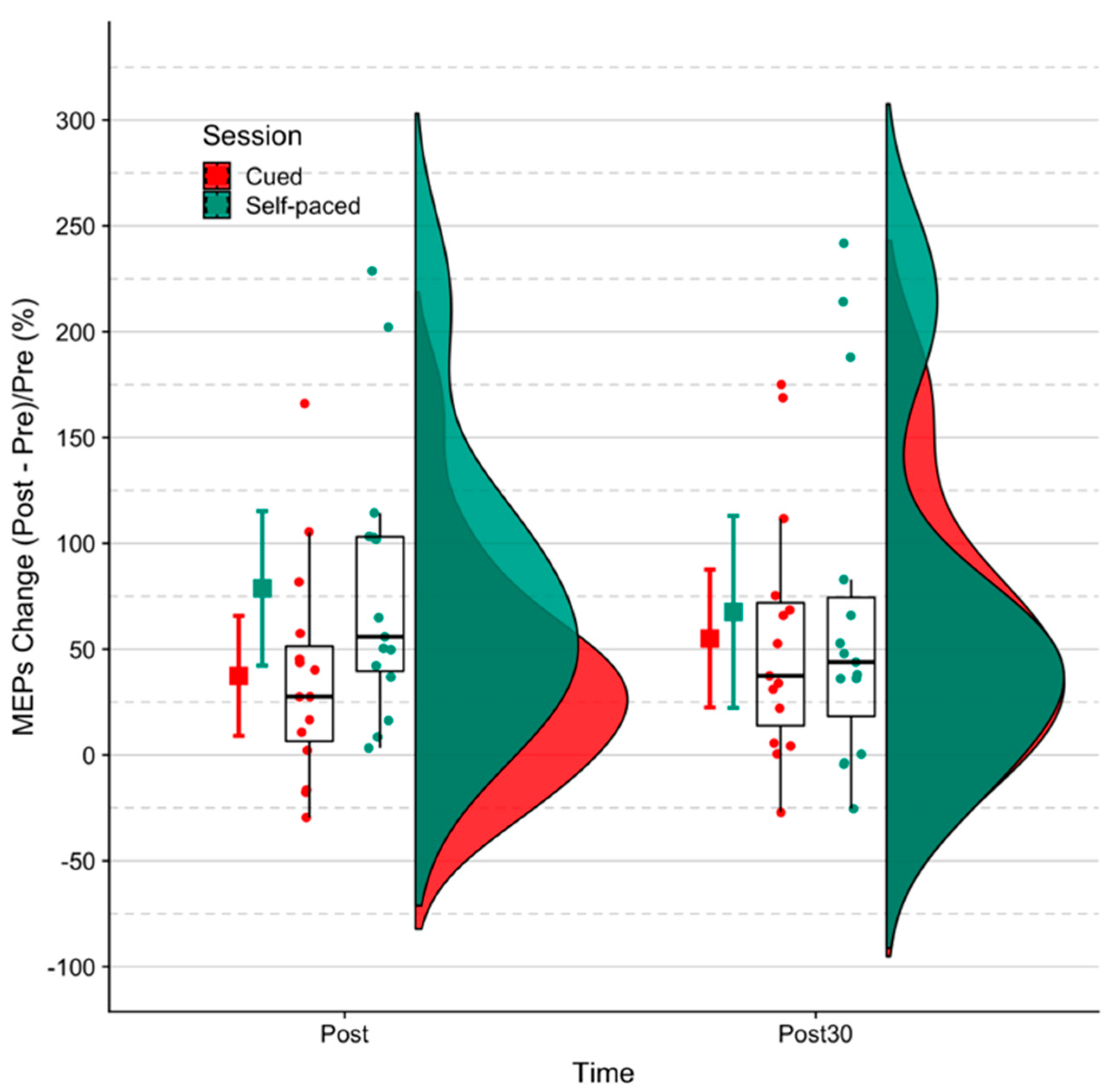
3.1. MEP Size
3.2. BCI Performance
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wolpaw, J.R.; Birbaumer, N.; McFarland, D.J.; Pfurtscheller, G.; Vaughan, T.M. Brain-computer interfaces for communication and control. Clin. Neurophys. 2002, 113, 767–791. [Google Scholar] [CrossRef]
- Daly, J.J.; Wolpaw, J.R. Brain–computer interfaces in neurological rehabilitation. Lancet Neurol. 2008, 7, 1032–1043. [Google Scholar] [CrossRef]
- Grosse-Wentrup, M.; Mattia, D.; Oweiss, K. Using brain–computer interfaces to induce neural plasticity and restore function. J. Neural Eng. 2011, 8, 025004. [Google Scholar] [CrossRef]
- Mrachacz-Kersting, N.; Kristensen, S.R.; Niazi, I.K.; Farina, D. Precise temporal association between cortical potentials evoked by motor imagination and afference induces cortical plasticity. J. Physiol. 2012, 590, 1669–1682. [Google Scholar] [CrossRef]
- Nascimento, O.F.; Dremstrup Nielsen, K.; Voigt, M. Movement-related parameters modulate cortical activity during imaginary isometric plantar-flexions. Exp. Brain Res. 2006, 171, 78–90. [Google Scholar] [CrossRef] [PubMed]
- Niazi, I.K.; Jiang, N.; Tiberghien, O.; Nielsen, J.F.; Dremstrup, K.; Farina, D. Detection of movement intention from single-trial movement-related cortical potentials. J. Neural Eng. 2011, 8, 66009. [Google Scholar] [CrossRef] [PubMed]
- Jochumsen, M.; Niazi, I.K.; Mrachacz-Kersting, N.; Farina, D.; Dremstrup, K. Detection and classification of movement-related cortical potentials associated with task force and speed. J. Neural Eng. 2013, 10, 56015. [Google Scholar] [CrossRef] [PubMed]
- Jochumsen, M.; Niazi, I.K.; Navid, M.S.; Anwar, M.N.; Farina, D.; Dremstrup, K. Online multi-class brain-computer interface for detection and classification of lower limb movement intentions and kinetics for stroke rehabilitation. Brain Comp. Interfaces 2015, 2, 202–210. [Google Scholar] [CrossRef]
- Yilmaz, Özge; Birbaumer, N.; Ramos-Murguialday, A. Movement related slow cortical potentials in severely paralyzed chronic stroke patients. Front. Hum. Neurosci. 2015, 8. [Google Scholar] [CrossRef] [PubMed]
- Shibasaki, H.; Hallett, M. What is the Bereitschaftspotential? Clin. Neurophysiol. 2006, 117, 2341–2356. [Google Scholar] [CrossRef] [PubMed]
- Pfurtscheller, G.; Da Silva, F.L. Event-related EEG/MEG synchronization and desynchronization: Basic principles. Clin. Neurophysiol. 1999, 110, 1842–1857. [Google Scholar] [CrossRef]
- Lew, E.; Chavarriaga, R.; Silvoni, S.; Millán, J.D.R. Detection of self-paced reaching movement intention from EEG signals. Front. Neuroeng. 2012, 5. [Google Scholar] [CrossRef]
- Xu, R.; Jiang, N.; Lin, C.; Mrachacz-Kersting, N.; Dremstrup, K.; Farina, D. Enhanced Low-latency Detection of Motor Intention from EEG for Closed-loop Brain-Computer Interface Applications. IEEE Trans. Biomed. Eng. 2013, 61, 288–296. [Google Scholar] [CrossRef]
- Niazi, I.K.; Mrachacz-Kersting, N.; Jiang, N.; Dremstrup, K.; Farina, D. Peripheral Electrical Stimulation Triggered by Self-Paced Detection of Motor Intention Enhances Motor Evoked Potentials. IEEE Trans. Neural Syst. Rehabil. Eng. 2012, 20, 595–604. [Google Scholar] [CrossRef]
- Xu, R.; Jiang, N.; Mrachacz-Kersting, N.; Lin, C.; Prieto, G.A.; Moreno, J.C.; Pons, J.L.; Dremstrup, K.; Farina, D. A Closed-Loop Brain–Computer Interface Triggering an Active Ankle–Foot Orthosis for Inducing Cortical Neural Plasticity. IEEE Trans. Biomed. Eng. 2014, 61, 2092–2101. [Google Scholar] [PubMed]
- Mrachacz-Kersting, N.; Voigt, M.; Stevenson, A.; Aliakbaryhosseinabadi, S.; Jiang, N.; Dremstrup, K.; Farina, D. The effect of type of afferent feedback timed with motor imagery on the induction of cortical plasticity. Brain Res. 2017, 1674, 91–100. [Google Scholar] [CrossRef]
- Mrachacz-Kersting, M.; Jiang, N.; Stevenson, A.J.T.; Niazi, I.K.; Kostic, V.; Pavlovic, A.; Radovanovic, S.; Djuric-Jovicic, M.; Agosta, F.; Dremstrup, K.; et al. Efficient neuroplasticity induction in chronic stroke patients by an associative brain-computer interface. J. Neurophysiol. 2016, 115, 1410–1421. [Google Scholar] [CrossRef]
- Olsen, S.; Signal, N.; Niazi, I.K.; Christensen, T.; Jochumsen, M.; Taylor, D. Paired Associative Stimulation Delivered by Pairing Movement-Related Cortical Potentials With Peripheral Electrical Stimulation: An Investigation of the Duration of Neuromodulatory Effects. Neuromodulation 2018, 21, 362–367. [Google Scholar] [CrossRef]
- Ang, K.K.; Chua, K.S.G.; Phua, K.S.; Wang, C.; Chin, Z.Y.; Kuah, C.W.K.; Guan, C. A Randomized Controlled Trial of EEG-Based Motor Imagery Brain-Computer Interface Robotic Rehabilitation for Stroke. Clin. EEG Neurosci. 2015, 46, 310–320. [Google Scholar] [CrossRef]
- Frolov, A.A.; Mokienko, O.; Lyukmanov, R.; Biryukova, E.; Kotov, S.; Turbina, L.; Nadareyshvily, G.; Bushkova, Y. Post-stroke Rehabilitation Training with a Motor-Imagery-Based Brain-Computer Interface (BCI)-Controlled Hand Exoskeleton: A Randomized Controlled Multicenter Trial. Front. Neurosci. 2017, 11, 400. [Google Scholar] [CrossRef] [PubMed]
- Ang, K.K.; Guan, C.; Phua, K.S.; Wang, C.; Zhou, L.; Tang, K.Y.; Joseph, G.J.E.; Kuah, C.W.K.; Chua, K.S.G. Brain-computer interface-based robotic end effector system for wrist and hand rehabilitation: Results of a three-armed randomized controlled trial for chronic stroke. Front. Neuroeng. 2014, 7. [Google Scholar] [CrossRef]
- Nijboer, F.; Bos, D.P.-O.; Blokland, Y.; Van Wijk, R.; Farquhar, J. Design requirements and potential target users for brain-computer interfaces – recommendations from rehabilitation professionals. Brain Comput. Interfaces 2014, 1, 50–61. [Google Scholar] [CrossRef]
- Leeb, R.; Perdikis, S.; Tonin, L.; Biasiucci, A.; Tavella, M.; Creatura, M.; Molina, A.; Al-Khodairy, A.; Carlson, T.; Millán, J.D. Transferring brain–computer interfaces beyond the laboratory: Successful application control for motor-disabled users. Artif. Intell. Med. 2013, 59, 121–132. [Google Scholar] [CrossRef] [PubMed]
- Morone, G.; Pisotta, I.; Pichiorri, F.; Kleih, S.; Paolucci, S.; Molinari, M.; Cincotti, F.; Kübler, A.; Mattia, D. Proof of Principle of a Brain-Computer Interface Approach to Support Poststroke Arm Rehabilitation in Hospitalized Patients: Design, Acceptability, and Usability. Arch. Phys. Med. Rehabil. 2015, 96, S71–S78. [Google Scholar] [CrossRef]
- Rossi, S.; Hallett, M.; Rossini, P.M.; Pascual-Leone, A. Safety, ethical considerations, and application guidelines for the use of transcranial magnetic stimulation in clinical practice and research. Clin. Neurophysiol. 2009, 120, 2008–2039. [Google Scholar] [CrossRef]
- Kumpulainen, S.; Mrachacz-Kersting, N.; Peltonen, J.; Voigt, M.; Avela, J. The optimal interstimulus interval and repeatability of paired associative stimulation when the soleus muscle is targeted. Exp. Brain 2012, 221, 241–249. [Google Scholar] [CrossRef]
- Krueger, C. A Comparison of the General Linear Mixed Model and Repeated Measures ANOVA Using a Dataset with Multiple Missing Data Points. Boil. Nurs. 2004, 6, 151–157. [Google Scholar] [CrossRef]
- Boisgontier, M.P.; Cheval, B. The anova to mixed model transition. Neurosci. Biobehav. Rev. 2016, 68, 1004–1005. [Google Scholar] [CrossRef]
- Frömer, R.; Maier, M.; Rahman, R.A. Group-Level EEG-Processing Pipeline for Flexible Single Trial-Based Analyses Including Linear Mixed Models. Front. Neurosci. 2018, 12. [Google Scholar] [CrossRef]
- Jochumsen, M.; Cremoux, S.; Robinault, L.; Lauber, J.; Arceo, J.C.; Navid, M.S.; Nedergaard, R.W.; Rashid, U.; Haavik, H.; Niazi, I.K. Investigation of Optimal Afferent Feedback Modality for Inducing Neural Plasticity with A Self-Paced Brain-Computer Interface. Sensors 2018, 18, 3761. [Google Scholar] [CrossRef]
- Bates, D.; Mächler, M.; Bolker, B.; Walker, S. Fitting Linear Mixed-Effects Models using lme4. Available online: cran.uvigo.es/web/packages/lme4/vignettes/lmer.pdf (accessed on 20 May 2019).
- Twisk, J.; Bosman, L.; Hoekstra, T.; Rijnhart, J.; Welten, M.; Heymans, M. Different ways to estimate treatment effects in randomised controlled trials. Contemp. Clin. Trials Commun. 2018, 10, 80–85. [Google Scholar]
- Emmeans: Estimated Marginal Means, aka Least-Squares Means. Available online: https://CRAN.R-project.org/package=emmeans (accessed on 20 May 2019).
- Zar, J.H. Biostatistical Analysis; Prentice Hall: Upper Saddle River, NJ, USA, 2010; ISBN 013081542X. [Google Scholar]
- Jochumsen, M.; Niazi, I.K.; Nedergaard, R.W.; Navid, M.S.; Dremstrup, K. Effect of subject training on a movement-related cortical potential-based brain-computer interface. Biomed. Signal Process. 2018, 41, 63–68. [Google Scholar] [CrossRef]
- Mrachacz-Kersting, N.; Aliakbaryhosseinabadi, S. Comparison of the Efficacy of a Real-Time and Offline Associative Brain-Computer-Interface. Front. Neurosci. 2018, 12. [Google Scholar] [CrossRef] [PubMed]
- Ridding, M.C.; Ziemann, U. Determinants of the induction of cortical plasticity by non-invasive brain stimulation in healthy subjects. J. Physiol. 2010, 588, 2291–2304. [Google Scholar] [CrossRef]
- Müller-Dahlhaus, J.F.M.; Orekhov, Y.; Liu, Y.; Ziemann, U. Interindividual variability and age-dependency of motor cortical plasticity induced by paired associative stimulation. Exp. Brain 2008, 187, 467–475. [Google Scholar] [CrossRef] [PubMed]
- Ziemann, U.; Paulus, W.; Nitsche, M.A.; Pascual-Leone, A.; Byblow, W.D.; Berardelli, A.; Siebner, H.R.; Classen, J.; Cohen, L.G.; Rothwell, J.C. Consensus: Motor cortex plasticity protocols. Brain Stimul. 2008, 1, 164–182. [Google Scholar] [CrossRef] [PubMed]
- Chipchase, L.; Schabrun, S.; Hodges, P.; Schabrun, S. Peripheral electrical stimulation to induce cortical plasticity: A systematic review of stimulus parameters. Clin. Neurophysiol. 2011, 122, 456–463. [Google Scholar] [CrossRef]

| Session | Time | Standard Error (mV) | ||
| Self-paced | post- | 0.25 | 0.05 | z = −7.44, p < 0.001 |
| Cue-based | post- | 0.18 | 0.04 | z = −8.53, p < 0.001 |
| Self-paced | post 30- | 0.22 | 0.04 | z = −8.05, p < 0.001 |
| Cue-based | post 30- | 0.21 | 0.04 | z = −7.95, p < 0.001 |
| Session | Time | (%) | Standard Error (%) | |
| Self-paced | post- | 93.26 | 19.82 | t[31.91] = 4.71, p < 0.001 |
| Cue-based | post- | 44.66 | 20.46 | t[31.58] = 2.18, p = 0.04 |
| Self-paced | post30- | 80.72 | 19.82 | t[31.91] = 4.07, p < 0.001 |
| Cue-based | post30- | 62.51 | 20.46 | t[31.58] = 3.05, p < 0.01 |
| Time | Self-Paced/Cue-Based (Ratio) | Standard Error (Ratio) | |
| Post-intervention | 1.34 | 0.27 | z = 1.46, p = 0.15 |
| 30-min post-intervention | 1.06 | 0.21 | z = 0.29, p = 0.77 |
| Time | Self-Paced − Cue-Based (%) | Standard Error (%) | |
| Post-intervention | 48.59 | 21.04 | t[36.2] = 2.31, p = 0.03 |
| 30-min post-intervention | 18.21 | 21.04 | t[36.2] = 0.87, p = 0.39 |
| Participant | True Positive Rate (%) | Number of False Positive Detections per Minute | Duration of the BCI Intervention (min) | Total Number of Movements Performed |
|---|---|---|---|---|
| 1 | 74 | 0.8 | 12 | 68 |
| 2 | 79 | 0.2 | 14 | 63 |
| 3 | 77 | 1.0 | 7 | 65 |
| 4 | 77 | 0.4 | 19 | 65 |
| 5 | 72 | 2.0 | 15 | 69 |
| 6 | 74 | 0.8 | 13 | 68 |
| 7 | 78 | 1.0 | 14 | 64 |
| 8 | 81 | 2.0 | 11 | 62 |
| 9 | 72 | 0.4 | 19 | 69 |
| 10 | 78 | 1.7 | 16 | 64 |
| 11 | 72 | 1.5 | 11 | 69 |
| 12 | 69 | 1.3 | 12 | 72 |
| 13 | 70 | 0.3 | 21 | 71 |
| 14 | 74 | 1.2 | 9 | 68 |
| 15 | 79 | 2.7 | 13 | 63 |
| Mean ± Std | 75 ± 3 | 1.2 ± 0.7 | 14 ± 4 | 67 ± 3 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jochumsen, M.; Navid, M.S.; Nedergaard, R.W.; Signal, N.; Rashid, U.; Hassan, A.; Haavik, H.; Taylor, D.; Niazi, I.K. Self-Paced Online vs. Cue-Based Offline Brain–Computer Interfaces for Inducing Neural Plasticity. Brain Sci. 2019, 9, 127. https://doi.org/10.3390/brainsci9060127
Jochumsen M, Navid MS, Nedergaard RW, Signal N, Rashid U, Hassan A, Haavik H, Taylor D, Niazi IK. Self-Paced Online vs. Cue-Based Offline Brain–Computer Interfaces for Inducing Neural Plasticity. Brain Sciences. 2019; 9(6):127. https://doi.org/10.3390/brainsci9060127
Chicago/Turabian StyleJochumsen, Mads, Muhammad Samran Navid, Rasmus Wiberg Nedergaard, Nada Signal, Usman Rashid, Ali Hassan, Heidi Haavik, Denise Taylor, and Imran Khan Niazi. 2019. "Self-Paced Online vs. Cue-Based Offline Brain–Computer Interfaces for Inducing Neural Plasticity" Brain Sciences 9, no. 6: 127. https://doi.org/10.3390/brainsci9060127
APA StyleJochumsen, M., Navid, M. S., Nedergaard, R. W., Signal, N., Rashid, U., Hassan, A., Haavik, H., Taylor, D., & Niazi, I. K. (2019). Self-Paced Online vs. Cue-Based Offline Brain–Computer Interfaces for Inducing Neural Plasticity. Brain Sciences, 9(6), 127. https://doi.org/10.3390/brainsci9060127








