Parallel Emergence of a Compartmentalized Striatum with the Phylogenetic Development of the Cerebral Cortex
Abstract
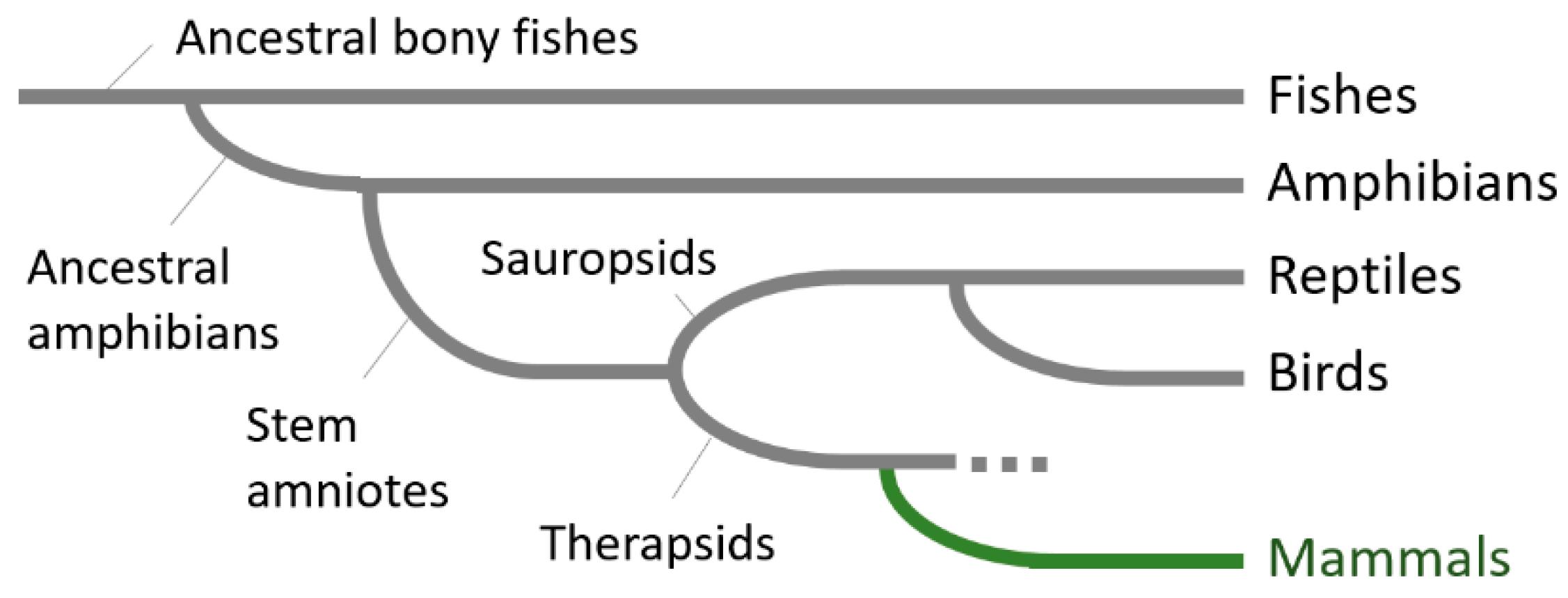
1. Introduction
2. The Striatum in Anamniotes
3. The Striatum in Non-Mammalian Amniotes
3.1. Reptiles
3.2. Birds
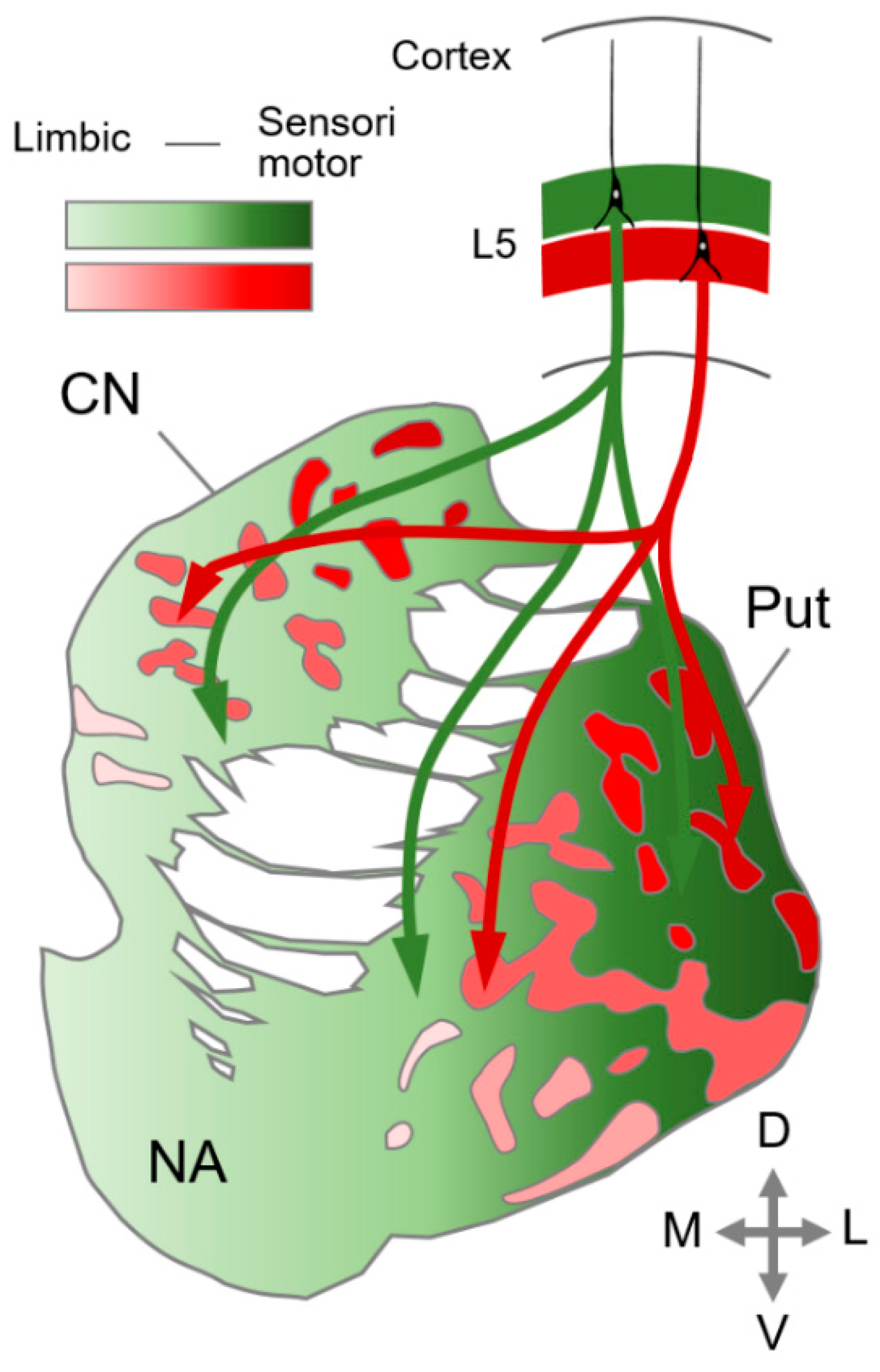
4. Striatal Compartmentalization in the Mammalian Brain
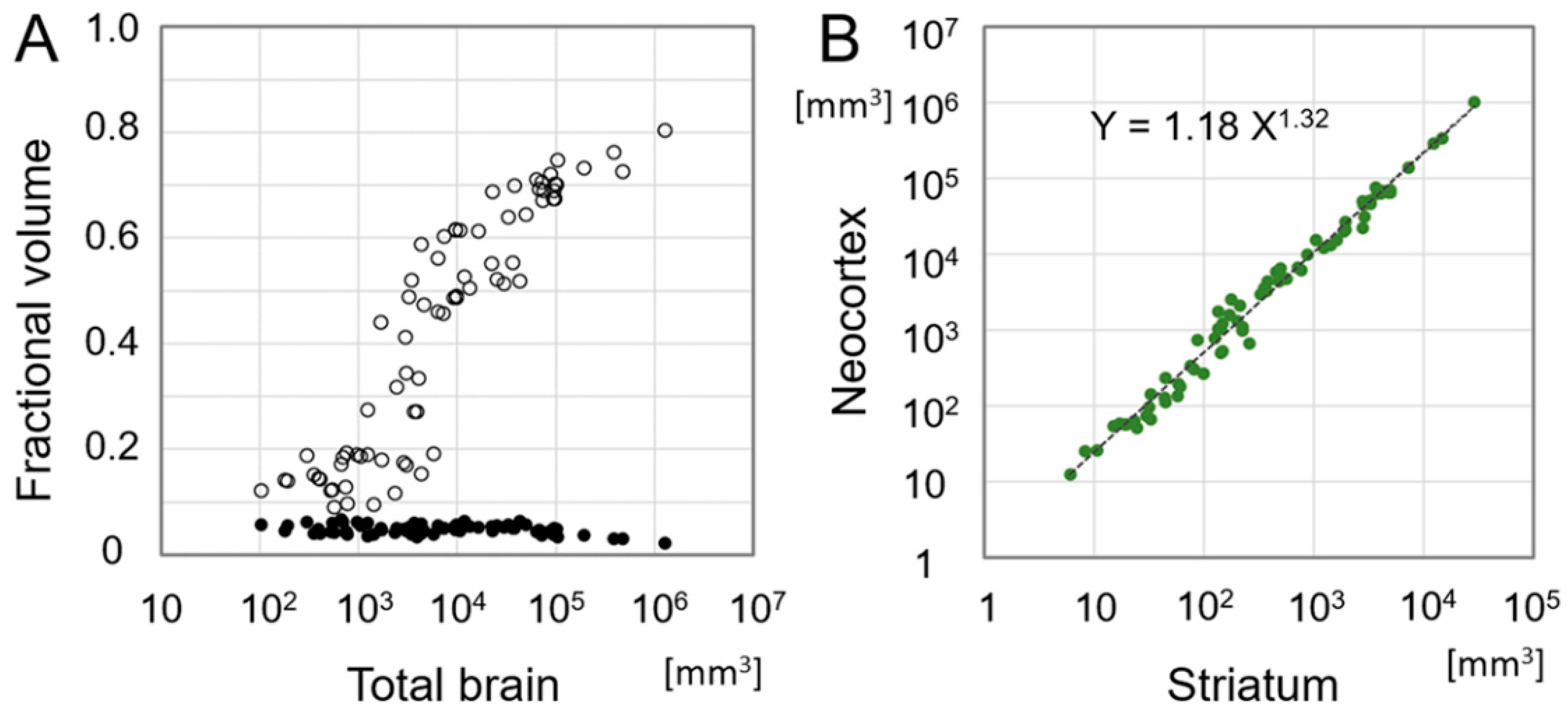
5. Emergence of the Six-Layered Cortex and the Striatal Compartment
6. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Crittenden, J.R.; Graybiel, A.M. Basal ganglia disorders associated with imbalances in the striatal striosome and matrix compartments. Front. Neuroanat. 2011, 5, 59. [Google Scholar] [CrossRef] [PubMed]
- Graybiel, A.M.; Canales, J.J.; Capper-Loup, C. Levodopa-induced dyskinesias and dopamine-dependent stereotypies: A new hypothesis. Trends. Neurosci. 2000, 23, S71–S77. [Google Scholar] [CrossRef]
- Gerfen, C.R. The neostriatal mosaic: Multiple levels of compartmental organization in the basal ganglia. Annu. Rev. Neurosci. 1992, 15, 285–320. [Google Scholar] [CrossRef]
- Crittenden, J.R.; Tillberg, P.W.; Riad, M.H.; Shima, Y.; Gerfen, C.R.; Curry, J.; Housman, D.E.; Nelson, S.B.; Boyden, E.S.; Graybiel, A.M. Striosome-dendron bouquets highlight a unique striatonigral circuit targeting dopamine-containing neurons. Proc. Natl. Acad. Sci. USA 2016, 113, 11318–11323. [Google Scholar] [CrossRef]
- Goto, S.; Kawarai, T.; Morigaki, R.; Okita, S.; Koizumi, H.; Nagahiro, S.; Munoz, E.L.; Lee, L.V.; Kaji, R. Defects in the striatal neuropeptide Y system in X-linked dystonia-parkinsonism. Brain 2013, 136, 1555–1567. [Google Scholar] [CrossRef]
- Goto, S.; Lee, L.V.; Munoz, E.L.; Tooyama, I.; Tamiya, G.; Makino, S.; Ando, S.; Dantes, M.B.; Yamada, K.; Matsumoto, S.; et al. Functional anatomy of the basal ganglia in X-linked recessive dystonia-parkinsonism. Ann. Neurol. 2005, 58, 7–17. [Google Scholar] [CrossRef]
- Beste, C.; Muckschel, M.; Rosales, R.; Domingo, A.; Lee, L.; Ng, A.; Klein, C.; Munchau, A. Striosomal dysfunction affects behavioral adaptation but not impulsivity-evidence from x-linked dystonia-parkinsonism. Mov. Disord. 2017, 32, 576–584. [Google Scholar] [CrossRef]
- Reiner, A.; Medina, L.; Veenman, C.L. Structural and functional evolution of the basal ganglia in vertebrates. Brain Res. Brain Res. Rev. 1998, 28, 235–285. [Google Scholar] [CrossRef]
- Smeets, W.J.; Marin, O.; Gonzalez, A. Evolution of the basal ganglia: New perspectives through a comparative approach. J. Anat. 2000, 196, 501–517. [Google Scholar] [CrossRef] [PubMed]
- Northcutt, R.G.; Kaas, J.H. The emergence and evolution of mammalian neocortex. Trends Neurosci. 1995, 18, 373–379. [Google Scholar] [CrossRef]
- Jarvis, E.D.; Gunturkun, O.; Bruce, L.; Csillag, A.; Karten, H.; Kuenzel, W.; Medina, L.; Paxinos, G.; Perkel, D.J.; Shimizu, T.; et al. Avian brains and a new understanding of vertebrate brain evolution. Nat. Rev. Neurosci. 2005, 6, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Ericsson, J.; Stephenson-Jones, M.; Perez-Fernandez, J.; Robertson, B.; Silberberg, G.; Grillner, S. Dopamine differentially modulates the excitability of striatal neurons of the direct and indirect pathways in lamprey. J. Neurosci. 2013, 33, 8045–8054. [Google Scholar] [CrossRef] [PubMed]
- Stephenson-Jones, M.; Kardamakis, A.A.; Robertson, B.; Grillner, S. Independent circuits in the basal ganglia for the evaluation and selection of actions. Proc. Natl. Acad. Sci. USA 2013, 110, E3670–E3679. [Google Scholar] [CrossRef]
- Perez-Fernandez, J.; Stephenson-Jones, M.; Suryanarayana, S.M.; Robertson, B.; Grillner, S. Evolutionarily conserved organization of the dopaminergic system in lamprey: SNc/VTA afferent and efferent connectivity and D2 receptor expression. J. Comp. Neurol. 2014, 522, 3775–3794. [Google Scholar] [CrossRef]
- Robertson, B.; Kardamakis, A.; Capantini, L.; Perez-Fernandez, J.; Suryanarayana, S.M.; Wallen, P.; Stephenson-Jones, M.; Grillner, S. The lamprey blueprint of the mammalian nervous system. Prog. Brain Res. 2014, 212, 337–349. [Google Scholar]
- Sharma, S.C.; Berthoud, V.M.; Breckwoldt, R. Distribution of substance P-like immunoreactivity in the goldfish brain. J. Comp. Neurol. 1989, 279, 104–116. [Google Scholar] [CrossRef]
- Martinoli, M.G.; Dubourg, P.; Geffard, M.; Calas, A.; Kah, O. Distribution of GABA-immunoreactive neurons in the forebrain of the goldfish, Carassius auratus. Cell Tissue Res. 1990, 260, 77–84. [Google Scholar] [CrossRef]
- Rink, E.; Wullimann, M.F. Connections of the ventral telencephalon (subpallium) in the zebrafish (Danio rerio). Brain Res. 2004, 1011, 206–220. [Google Scholar] [CrossRef]
- Rink, E.; Wullimann, M.F. Connections of the ventral telencephalon and tyrosine hydroxylase distribution in the zebrafish brain (Danio rerio) lead to identification of an ascending dopaminergic system in a teleost. Brain Res. Bull. 2002, 57, 385–387. [Google Scholar] [CrossRef]
- Ganz, J.; Kaslin, J.; Freudenreich, D.; Machate, A.; Geffarth, M.; Brand, M. Subdivisions of the adult zebrafish subpallium by molecular marker analysis. J. Comp. Neurol. 2012, 520, 633–655. [Google Scholar] [CrossRef]
- Reiner, A.; Northcutt, R.G. An immunohistochemical study of the telencephalon of the African lungfish, Protopterus annectens. J. Comp. Neurol. 1987, 256, 463–481. [Google Scholar] [CrossRef] [PubMed]
- Vallarino, M.; Tranchand-Bunel, D.; Thoumas, J.L.; Masini, M.A.; Conlon, J.M.; Fournier, A.; Pelletier, G.; Vaudry, H. Neuropeptide tyrosine in the brain of the African lungfish, Protopterus annectens: immunohistochemical localization and biochemical characterization. J. Comp. Neurol. 1995, 356, 537–551. [Google Scholar] [CrossRef] [PubMed]
- Nishikawa, S.; Goto, S.; Yamada, K.; Hamasaki, T.; Ushio, Y. Lack of Reelin causes malpositioning of nigral dopaminergic neurons: Evidence from comparison of normal and Reln(rl) mutant mice. J. Comp. Neurol. 2003, 461, 166–173. [Google Scholar] [CrossRef] [PubMed]
- Lopez, J.M.; Gonzalez, A. Organization of the catecholaminergic systems in the brain of lungfishes, the closest living relatives of terrestrial vertebrates. J. Comp. Neurol. 2017, 525, 3083–3109. [Google Scholar] [CrossRef]
- Brox, A.; Puelles, L.; Ferreiro, B.; Medina, L. Expression of the genes Emx1, Tbr1, and Eomes (Tbr2) in the telencephalon of Xenopus laevis confirms the existence of a ventral pallial division in all tetrapods. J. Comp. Neurol. 2004, 474, 562–577. [Google Scholar] [CrossRef]
- Papalopulu, N.; Kintner, C. Xenopus Distal-less related homeobox genes are expressed in the developing forebrain and are induced by planar signals. Development 1993, 117, 961–975. [Google Scholar] [PubMed]
- Bandin, S.; Morona, R.; Lopez, J.M.; Moreno, N.; Gonzalez, A. Immunohistochemical analysis of Pax6 and Pax7 expression in the CNS of adult Xenopus laevis. J. Chem. Neuroanat. 2014, 57–58, 24–41. [Google Scholar] [CrossRef]
- Marin, O.; Smeets, W.J.; Gonzalez, A. Basal ganglia organization in amphibians: Chemoarchitecture. J. Comp. Neurol. 1998, 392, 285–312. [Google Scholar] [CrossRef]
- Heer, T.; Pozzi, A.G.; Yovanovich, C.A.; Paz, D.A. Distribution pattern of neuropeptide Y in the brain, pituitary and olfactory system during the larval development of the toad Rhinella arenarum (Amphibia: Anura). Anat. Histol. Embryol. 2009, 38, 89–95. [Google Scholar] [CrossRef]
- Fernandez, A.S.; Pieau, C.; Reperant, J.; Boncinelli, E.; Wassef, M. Expression of the Emx-1 and Dlx-1 homeobox genes define three molecularly distinct domains in the telencephalon of mouse, chick, turtle and frog embryos: Implications for the evolution of telencephalic subdivisions in amniotes. Development 1998, 125, 2099–2111. [Google Scholar]
- Gonzalez, C.; Mendoza, J.; Avila-Costa, M.R.; Arias, J.M.; Barral, J. Golgi study of medium spiny neurons from dorsolateral striatum of the turtle Trachemys scripta elegans. Neurosci. Lett. 2013, 556, 227–231. [Google Scholar] [CrossRef]
- Bennis, M.; Ba M’hamed, S.; Rio, J.P.; Le Cren, D.; Reperant, J.; Ward, R. The distribution of NPY-like immunoreactivity in the chameleon brain. Anat. Embryol. (Berl.) 2001, 203, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Brauth, S.E.; Kitt, C.A. The paleostriatal system of Caiman crocodilus. J. Comp. Neurol. 1980, 189, 437–465. [Google Scholar] [CrossRef] [PubMed]
- Brauth, S.E.; Reiner, A.; Kitt, C.A.; Karten, H.J. The substance P-containing striatotegmental path in reptiles: An immunohistochemical study. J. Comp. Neurol. 1983, 219, 305–327. [Google Scholar] [CrossRef] [PubMed]
- Clark, E.C.; Baxter, L.R., Jr. Mammal-like striatal functions in Anolis. I. Distribution of serotonin receptor subtypes, and absence of striosome and matrix organization. Brain Behav. Evol. 2000, 56, 235–248. [Google Scholar] [CrossRef] [PubMed]
- Reiner, A.; Perkel, D.J.; Bruce, L.L.; Butler, A.B.; Csillag, A.; Kuenzel, W.; Medina, L.; Paxinos, G.; Shimizu, T.; Striedter, G.; et al. Revised nomenclature for avian telencephalon and some related brainstem nuclei. J. Comp. Neurol. 2004, 473, 377–414. [Google Scholar] [CrossRef] [PubMed]
- Bruce, L.L.; Erichsen, J.T.; Reiner, A. Neurochemical compartmentalization within the pigeon basal ganglia. J. Chem. Neuroanat. 2016, 78, 65–86. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Calero, E.; Scharff, C. Calbindin expression in developing striatum of zebra finches and its relation to the formation of area X. J. Comp. Neurol. 2013, 521, 326–341. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Calero, E.; Bahamonde, O.; Martinez, S. Differences in number and distribution of striatal calbindin medium spiny neurons between a vocal-learner (Melopsittacus undulatus) and a non-vocal learner bird (Colinus virginianus). Front. Neuroanat. 2013, 7, 46. [Google Scholar] [CrossRef]
- Morel, A.; Loup, F.; Magnin, M.; Jeanmonod, D. Neurochemical organization of the human basal ganglia: Anatomofunctional territories defined by the distributions of calcium-binding proteins and SMI-32. J. Comp. Neurol. 2002, 443, 86–103. [Google Scholar] [CrossRef]
- Balint, E.; Csillag, A. Nucleus accumbens subregions: hodological and immunohistochemical study in the domestic chick (Gallus domesticus). Cell Tissue Res. 2007, 327, 221–230. [Google Scholar] [CrossRef] [PubMed]
- Kuenzel, W.J.; Medina, L.; Csillag, A.; Perkel, D.J.; Reiner, A. The avian subpallium: New insights into structural and functional subdivisions occupying the lateral subpallial wall and their embryological origins. Brain Res. 2011, 1424, 67–101. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Reiner, A. Localization of dopamine D1A and D1B receptor mRNAs in the forebrain and midbrain of the domestic chick. J. Chem. Neuroanat. 2000, 19, 211–224. [Google Scholar] [CrossRef]
- Basham, M.E.; Nordeen, E.J.; Nordeen, K.W. Blockade of NMDA receptors in the anterior forebrain impairs sensory acquisition in the zebra finch (Poephila guttata). Neurobiol. Learn. Mem. 1996, 66, 295–304. [Google Scholar] [CrossRef]
- Wada, K.; Sakaguchi, H.; Jarvis, E.D.; Hagiwara, M. Differential expression of glutamate receptors in avian neural pathways for learned vocalization. J. Comp. Neurol. 2004, 476, 44–64. [Google Scholar] [CrossRef]
- Martin, L.J.; Blackstone, C.D.; Huganir, R.L.; Price, D.L. The striatal mosaic in primates: striosomes and matrix are differentially enriched in ionotropic glutamate receptor subunits. J. Neurosci. 1993, 13, 782–792. [Google Scholar] [CrossRef] [PubMed]
- Karten, H.J.; Dubbeldam, J.L. The organization and projections of the paleostriatal complex in the pigeon (Columba livia). J. Comp. Neurol. 1973, 148, 61–89. [Google Scholar] [CrossRef] [PubMed]
- Bottjer, S.W.; Halsema, K.A.; Brown, S.A.; Miesner, E.A. Axonal connections of a forebrain nucleus involved with vocal learning in zebra finches. J. Comp. Neurol. 1989, 279, 312–326. [Google Scholar] [CrossRef] [PubMed]
- Luo, M.; Perkel, D.J. Long-range GABAergic projection in a circuit essential for vocal learning. J. Comp. Neurol. 1999, 403, 68–84. [Google Scholar] [CrossRef]
- Graybiel, A.M.; Ragsdale, C.W. Histochemically distinct compartments in the striatum of human, monkeys, and cat demonstrated by acetylthiocholinesterase staining. Proc. Natl. Acad. Sci. USA 1978, 75, 5723–5726. [Google Scholar] [CrossRef]
- Brand, S.; Rakic, P. Genesis of the primate neostriatum: [3H] thymidine autoradiographic analysis of the time of neuron origin in the rhesus monkey. Neuroscience 1979, 4, 767–778. [Google Scholar] [CrossRef]
- Van der Kooy, D.; Fishell, G. Neuronal birthdate underlies the development of striatal compartments. Brain Res. 1987, 401, 155–161. [Google Scholar] [CrossRef]
- Mason, H.A.; Rakowiecki, S.M.; Raftopoulou, M.; Nery, S.; Huang, Y.; Gridley, T.; Fishell, G. Notch signaling coordinates the patterning of striatal compartments. Development 2005, 132, 4247–4258. [Google Scholar] [CrossRef]
- Johnston, J.G.; Gerfen, C.R.; Haber, S.N.; van der Kooy, D. Mechanisms of striatal pattern formation: Conservation of mammalian compartmentalization. Brain Res. Dev. Brain Res. 1990, 57, 93–102. [Google Scholar] [CrossRef]
- Gerfen, C.R. The neostriatal mosaic: Striatal patch-matrix organization is related to cortical lamination. Science 1989, 246, 385–388. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.B.; Klug, J.R.; Ross, D.L.; Howard, C.D.; Hollon, N.G.; Ko, V.I.; Hoffman, H.; Callaway, E.M.; Gerfen, C.R.; Jin, X. Genetic-based dissection unveils the inputs and outputs of striatal patch and matrix compartments. Neuron 2016, 91, 1069–1084. [Google Scholar] [CrossRef] [PubMed]
- Fishell, G.; van Der Kooy, D. Pattern formation in the striatum: neurons with early projections to the substantia nigra survive the cell death period. J. Comp. Neurol. 1991, 312, 33–42. [Google Scholar] [CrossRef]
- Hagimoto, K.; Takami, S.; Murakami, F.; Tanabe, Y. Distinct migratory behaviors of striosome and matrix cells underlying the mosaic formation in the developing striatum. J. Comp. Neurol. 2017, 525, 794–817. [Google Scholar] [CrossRef]
- Lanca, A.J.; Boyd, S.; Kolb, B.E.; van der Kooy, D. The development of a patchy organization of the rat striatum. Brain Res. 1986, 392, 1–10. [Google Scholar] [CrossRef]
- Fishell, G.; van der Kooy, D. Pattern formation in the striatum: developmental changes in the distribution of striatonigral projections. Brain Res Dev. Brain Res. 1989, 45, 239–255. [Google Scholar] [CrossRef]
- Song, D.D.; Harlan, R.E. Genesis and migration patterns of neurons forming the patch and matrix compartments of the rat striatum. Brain Res. Dev. Brain Res. 1994, 83, 233–245. [Google Scholar] [CrossRef]
- Krushel, L.A.; Fishell, G.; Van Der Kooy, D. Pattern formation in the mammalian forebrain: Striatal patch and matrix neurons intermix prior to compartment formation. Eur. J. Neurosci. 1995, 7, 1210–1219. [Google Scholar] [CrossRef] [PubMed]
- Banghart, M.R.; Neufeld, S.Q.; Wong, N.C.; Sabatini, B.L. Enkephalin disinhibits mu opioid receptor-rich striatal patches via delta opioid receptors. Neuron 2015, 88, 1227–1239. [Google Scholar] [CrossRef] [PubMed]
- Salinas, A.G.; Davis, M.I.; Lovinger, D.M.; Mateo, Y. Dopamine dynamics and cocaine sensitivity differ between striosome and matrix compartments of the striatum. Neuropharmacology 2016, 108, 275–283. [Google Scholar] [CrossRef]
- Deacon, T.W.; Pakzaban, P.; Isacson, O. The lateral ganglionic eminence is the origin of cells committed to striatal phenotypes: Neural transplantation and developmental evidence. Brain Res. 1994, 668, 211–219. [Google Scholar] [CrossRef]
- Anderson, S.A.; Qiu, M.; Bulfone, A.; Eisenstat, D.D.; Meneses, J.; Pedersen, R.; Rubenstein, J.L. Mutations of the homeobox genes Dlx-1 and Dlx-2 disrupt the striatal subventricular zone and differentiation of late born striatal neurons. Neuron 1997, 19, 27–37. [Google Scholar] [CrossRef]
- Xu, Q.; Tam, M.; Anderson, S.A. Fate mapping Nkx2.1-lineage cells in the mouse telencephalon. J. Comp. Neurol. 2008, 506, 16–29. [Google Scholar] [CrossRef] [PubMed]
- Marin, O.; Anderson, S.A.; Rubenstein, J.L. Origin and molecular specification of striatal interneurons. J. Neurosci. 2000, 20, 6063–6076. [Google Scholar] [CrossRef] [PubMed]
- Nobrega-Pereira, S.; Kessaris, N.; Du, T.; Kimura, S.; Anderson, S.A.; Marin, O. Postmitotic Nkx2-1 controls the migration of telencephalic interneurons by direct repression of guidance receptors. Neuron 2008, 59, 733–745. [Google Scholar] [CrossRef]
- Hamasaki, T.; Goto, S.; Nishikawa, S.; Ushio, Y. A role of netrin-1 in the formation of the subcortical structure striatum: Repulsive action on the migration of late-born striatal neurons. J. Neurosci. 2001, 21, 4272–4280. [Google Scholar] [CrossRef]
- Powell, E.M.; Mars, W.M.; Levitt, P. Hepatocyte growth factor/scatter factor is a motogen for interneurons migrating from the ventral to dorsal telencephalon. Neuron 2001, 30, 79–89. [Google Scholar] [CrossRef]
- Flames, N.; Long, J.E.; Garratt, A.N.; Fischer, T.M.; Gassmann, M.; Birchmeier, C.; Lai, C.; Rubenstein, J.L.; Marín, O. Short- and long-range attraction of cortical GABAergic interneurons by neuregulin-1. Neuron 2004, 44, 251–261. [Google Scholar] [CrossRef]
- Villar-Cervino, V.; Kappeler, C.; Nobrega-Pereira, S.; Henkemeyer, M.; Rago, L.; Nieto, M.A.; Marín, O. Molecular mechanisms controlling the migration of striatal interneurons. J. Neurosci. 2015, 35, 8718–8729. [Google Scholar] [CrossRef] [PubMed]
- Kuo, H.Y.; Liu, F.C. Valproic acid induces aberrant development of striatal compartments and corticostriatal pathways in a mouse model of autism spectrum disorder. FASEB J. 2017, 31, 4458–4471. [Google Scholar] [CrossRef] [PubMed]
- Hunnicutt, B.J.; Jongbloets, B.C.; Birdsong, W.T.; Gertz, K.J.; Zhong, H.; Mao, T. A comprehensive excitatory input map of the striatum reveals novel functional organization. Elife 2016, 5, e19103. [Google Scholar] [CrossRef] [PubMed]
- Thorn, C.A.; Atallah, H.; Howe, M.; Graybiel, A.M. Differential dynamics of activity changes in dorsolateral and dorsomedial striatal loops during learning. Neuron 2010, 66, 781–795. [Google Scholar] [CrossRef]
- Gangarossa, G.; Espallergues, J.; Mailly, P.; De Bundel, D.; De Kerchove D’exaerde, A.; Herve, D.; Girault, J.A.; Valjent, E.; Krieger, P. Spatial distribution of D1R- and D2R-expressing medium-sized spiny neurons differs along the rostro-caudal axis of the mouse dorsal striatum. Front. Neural. Circuits 2013, 7, 124. [Google Scholar] [CrossRef]
- Miyamoto, Y.; Katayama, S.; Shigematsu, N.; Nishi, A.; Fukuda, T. Striosome-based map of the mouse striatum that is conformable to both cortical afferent topography and uneven distributions of dopamine D1 and D2 receptor-expressing cells. Brain Struct. Funct. 2018, 223, 4275–4291. [Google Scholar] [CrossRef]
- Gerfen, C.R.; Herkenham, M.; Thibault, J. The neostriatal mosaic: II. Patch- and matrix-directed mesostriatal dopaminergic and non-dopaminergic systems. J. Neurosci. 1987, 7, 3915–3934. [Google Scholar] [CrossRef]
- Sato, K.; Sumi-Ichinose, C.; Kaji, R.; Ikemoto, K.; Nomura, T.; Nagatsu, I.; Ichinose, H.; Ito, M.; Sako, W.; Nagahiro, S.; et al. Differential involvement of striosome and matrix dopamine systems in a transgenic model of dopa-responsive dystonia. Proc. Natl. Acad. Sci. USA 2008, 105, 12551–12556. [Google Scholar] [CrossRef]
- Morigaki, R.; Goto, S. Putaminal Mosaic Visualized by Tyrosine Hydroxylase Immunohistochemistry in the Human Neostriatum. Front. Neuroanat. 2016, 10, 34. [Google Scholar] [CrossRef] [PubMed]
- Ragsdale, C.W., Jr.; Graybiel, A.M. Compartmental organization of the thalamostriatal connection in the cat. J. Comp. Neurol. 1991, 311, 134–167. [Google Scholar] [CrossRef] [PubMed]
- Alexander, G.E.; Crutcher, M.D.; Delong, M.R. Basal ganglia-thalamocortical circuits: Parallel substrates for motor, oculomotor, “prefrontal” and “limbic” functions. Prog. Brain Res. 1990, 85, 119–146. [Google Scholar]
- Groenewegen, H.J.; Berendse, H.W. The specificity of the ’nonspecific’ midline and intralaminar thalamic nuclei. Trends Neurosci. 1994, 17, 52–57. [Google Scholar] [CrossRef]
- Fujiyama, F.; Unzai, T.; Nakamura, K.; Nomura, S.; Kaneko, T. Difference in organization of corticostriatal and thalamostriatal synapses between patch and matrix compartments of rat neostriatum. Eur. J. Neurosci. 2006, 24, 2813–2824. [Google Scholar] [CrossRef] [PubMed]
- Fujiyama, F.; Kuramoto, E.; Okamoto, K.; Hioki, H.; Furuta, T.; Zhou, L.; Nomura, S.; Kaneko, T. Presynaptic localization of an AMPA-type glutamate receptor in corticostriatal and thalamostriatal axon terminals. Eur. J. Neurosci. 2004, 20, 3322–3330. [Google Scholar] [CrossRef] [PubMed]
- Raju, D.V.; Shah, D.J.; Wright, T.M.; Hall, R.A.; Smith, Y. Differential synaptology of vGluT2-containing thalamostriatal afferents between the patch and matrix compartments in rats. J. Comp. Neurol. 2006, 499, 231–243. [Google Scholar] [CrossRef]
- Marin, O.; Gonzalez, A.; Smeets, W.J. Basal ganglia organization in amphibians: afferent connections to the striatum and the nucleus accumbens. J. Comp. Neurol. 1997, 378, 16–49. [Google Scholar] [CrossRef]
- Rakic, P. Evolution of the neocortex: a perspective from developmental biology. Nat. Rev. Neurosci. 2009, 10, 724–735. [Google Scholar] [CrossRef]
- Lee, A.M.; Tai, L.H.; Zador, A.; Wilbrecht, L. Between the primate and ‘reptilian’ brain: Rodent models demonstrate the role of corticostriatal circuits in decision making. Neuroscience 2015, 296, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Hamasaki, T.; Leingartner, A.; Ringstedt, T.; O’leary, D.D. EMX2 regulates sizes and positioning of the primary sensory and motor areas in neocortex by direct specification of cortical progenitors. Neuron 2004, 43, 359–372. [Google Scholar] [CrossRef]
- O’leary, D.D.; Chou, S.J.; Hamasaki, T.; Sahara, S.; Takeuchi, A.; Thuret, S.; Leingärtner, A. Regulation of laminar and area patterning of mammalian neocortex and behavioural implications. Novartis Found. Symp. 2007, 288, 141–159. [Google Scholar] [PubMed]
- Hamasaki, T.; Goto, S.; Nishikawa, S.; Ushio, Y. Neuronal cell migration for the developmental formation of the mammalian striatum. Brain Res. Brain Res. Rev. 2003, 41, 1–12. [Google Scholar] [CrossRef]
- Stephan, H.; Frahm, H.; Baron, G. New and revised data on volumes of brain structures in insectivores and primates. Folia Primatol. (Basel) 1981, 35, 1–29. [Google Scholar] [CrossRef] [PubMed]
- Clark, D.A.; Mitra, P.P.; Wang, S.S. Scalable architecture in mammalian brains. Nature 2001, 411, 189–193. [Google Scholar] [CrossRef] [PubMed]
- Stevens, C.F. An evolutionary scaling law for the primate visual system and its basis in cortical function. Nature 2001, 411, 193–195. [Google Scholar] [CrossRef]
- Shepherd, G.M. Corticostriatal connectivity and its role in disease. Nat. Rev. Neurosci. 2013, 14, 278–291. [Google Scholar] [CrossRef] [PubMed]
- Hooks, B.M.; Papale, A.E.; Paletzki, R.F.; Feroze, M.W.; Eastwood, B.S.; Couey, J.J.; Winnubst, J.; Chandrashekar, J.; Gerfen, C.R. Topographic precision in sensory and motor corticostriatal projections varies across cell type and cortical area. Nat. Commun. 2018, 9, 3549. [Google Scholar] [CrossRef] [PubMed]
- Gerfen, C.R.; Baimbridge, K.G.; Miller, J.J. The neostriatal mosaic: Compartmental distribution of calcium-binding protein and parvalbumin in the basal ganglia of the rat and monkey. Proc. Natl. Acad. Sci. USA 1985, 82, 8780–8784. [Google Scholar] [CrossRef] [PubMed]
- Ragsdale, C.W., Jr.; Graybiel, A.M. Fibers from the basolateral nucleus of the amygdala selectively innervate striosomes in the caudate nucleus of the cat. J. Comp. Neurol. 1988, 269, 506–522. [Google Scholar] [CrossRef]
- Parent, A.; Hazrati, L.N. Functional anatomy of the basal ganglia. I. The cortico-basal ganglia-thalamo-cortical loop. Brain Res. Brain Res. Rev. 1995, 20, 91–127. [Google Scholar] [CrossRef]
- Kincaid, A.E.; Wilson, C.J. Corticostriatal innervation of the patch and matrix in the rat neostriatum. J. Comp. Neurol. 1996, 374, 578–592. [Google Scholar] [CrossRef]
- Eblen, F.; Graybiel, A.M. Highly restricted origin of prefrontal cortical inputs to striosomes in the macaque monkey. J. Neurosci. 1995, 15, 5999–6013. [Google Scholar] [CrossRef]
- Levesque, M.; Parent, A. Axonal arborization of corticostriatal and corticothalamic fibers arising from prelimbic cortex in the rat. Cereb.Cortex 1998, 8, 602–613. [Google Scholar] [CrossRef] [PubMed]
- Sohur, U.S.; Padmanabhan, H.K.; Kotchetkov, I.S.; Menezes, J.R.; Macklis, J.D. Anatomic and molecular development of corticostriatal projection neurons in mice. Cereb. Cortex 2014, 24, 293–303. [Google Scholar] [CrossRef] [PubMed]
- Sugrue, L.P.; Corrado, G.S.; Newsome, W.T. Choosing the greater of two goods: Neural currencies for valuation and decision making. Nat. Rev. Neurosci. 2005, 6, 363–375. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hamasaki, T.; Goto, S. Parallel Emergence of a Compartmentalized Striatum with the Phylogenetic Development of the Cerebral Cortex. Brain Sci. 2019, 9, 90. https://doi.org/10.3390/brainsci9040090
Hamasaki T, Goto S. Parallel Emergence of a Compartmentalized Striatum with the Phylogenetic Development of the Cerebral Cortex. Brain Sciences. 2019; 9(4):90. https://doi.org/10.3390/brainsci9040090
Chicago/Turabian StyleHamasaki, Tadashi, and Satoshi Goto. 2019. "Parallel Emergence of a Compartmentalized Striatum with the Phylogenetic Development of the Cerebral Cortex" Brain Sciences 9, no. 4: 90. https://doi.org/10.3390/brainsci9040090
APA StyleHamasaki, T., & Goto, S. (2019). Parallel Emergence of a Compartmentalized Striatum with the Phylogenetic Development of the Cerebral Cortex. Brain Sciences, 9(4), 90. https://doi.org/10.3390/brainsci9040090



