Cognitive Function in Primary Sjögren’s Syndrome: A Systematic Review
Abstract
1. Introduction
1.1. Rationale
1.2. Objectives
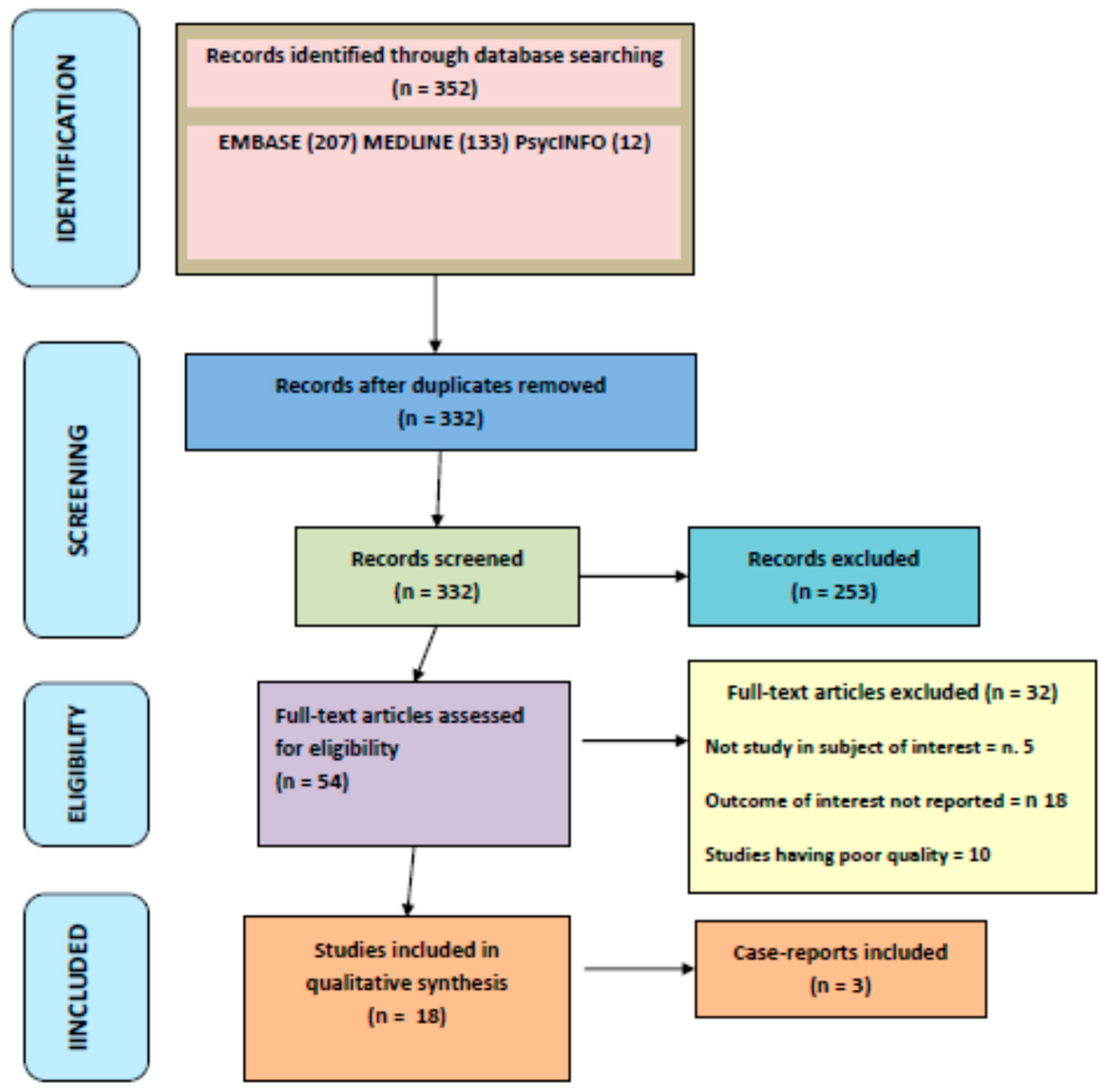
2. Materials and Methods
2.1. Search Strategy
2.2. Inclusion Criteria
2.3. Exclusion Criteria
2.4. Data Extraction
2.5. Assessment of Bias Risk
3. Results
3.1. Description of Included Studies
3.2. pSS, Mild Cognitive Impairment (MCI), and Dementia
3.3. Fatigue and Cognitive Function in pSS
3.4. Laboratory Data and Clinical Manifestations
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Patel, R.; Shahane, A. The epidemiology of Sjögren’s syndrome. Clin. Epidemiol. 2014, 6, 247–255. [Google Scholar]
- Qin, B.; Wang, J.; Yang, Z.; Yang, M.; Ma, N.; Huang, F.; Zhong, R. Epidemiology of primary Sjögren’s syndrome: A systematic review and meta-analysis. Ann. Rheum. Dis. 2015, 74, 1983–1989. [Google Scholar] [CrossRef] [PubMed]
- Mariette, X.; Criswell, L.A. Primary Sjögren’s Syndrome. N. Engl. J. Med. 2018, 378, 931–939. [Google Scholar] [CrossRef] [PubMed]
- McCoy, S.S.; Baer, A.N. Neurological Complications of Sjögren’s Syndrome: Diagnosis and Management. Curr. Treat. Options Rheumatol. 2017, 3, 275–288. [Google Scholar] [CrossRef]
- Perzyńska-Mazan, J.; Maślińska, M.; Gasik, R. Neurological manifestations of primary Sjögren’s syndrome. Reumatologia 2018, 56, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Gono, T.; Kawaguchi, Y.; Katsumata, Y.; Takagi, K.; Tochimoto, A.; Baba, S.; Okamoto, Y.; Ota, Y.; Yamanaka, H. Clinical manifestations of neurological involvement in primary Sjögren’s syndrome. Clin. Rheumatol. 2011, 30, 485–490. [Google Scholar] [CrossRef]
- Ye, W.; Chen, S.; Huang, X.; Qin, W.; Zhang, T.; Zhu, X.; Lin, C.; Wang, X. Clinical features and risk factors of neurological involvement in Sjögren’s syndrome. BMC Neurosci. 2018, 19, 26. [Google Scholar] [CrossRef] [PubMed]
- Moreira, I.; Teixeira, F.; Martins Silva, A.; Vasconcelos, C.; Farinha, F.; Santos, E. Frequent involvement of central nervous system in primary Sjögren syndrome. Rheumatol. Int. 2015, 35, 289–294. [Google Scholar] [CrossRef]
- Posso-Osorio, I.; Naranjo-Escobar, J.; Loaiza, D.M.; Polo, M.; Echeverri, A.; Tobon, G.J. Neurological involvement in primary Sjogren’s syndrome. Curr. Rheumatol. Rev. 2018. [Google Scholar] [CrossRef]
- Manzo, C.; Serra-Mestres, J. Considerations on fibrofog and fibromyalgic discognition in older patients. Giornale Italiano di Reumatologia Clinica 2016, 3, 59–75. [Google Scholar]
- Serra-Mestres, J. Problemas de memoria no debidos a demencia: Conceptos y mecanismos. Inf. Psiquiatr. 2016, 224, 13–20. [Google Scholar]
- Helme, C.; Hegarty, R.S.M.; Stebbings, S.; Treharne, G.J. “I actually just really need to stop work sometimes”: Exploring fatigue-related barriers to employment among people with rheumatic diseases. Musculoskelet. Care 2018. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. The PRISMA Group. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Vitali, C.; Bombardieri, S.; Jonsson, R.; Moutsopoulos, H.M.; Alexander, E.L.; Carsons, S.E.; Daniels, T.E.; Fox, P.C.; Fox, R.I.; Kassan, S.S.; et al. Classification criteria for Sjogren’s syndrome: A revised version of the European criteria proposed by the American-European Consensus Group. Ann. Rheum. Dis. 2002, 61, 554–558. [Google Scholar] [CrossRef]
- Shiboski, S.C.; Shiboski, C.H.; Criswell, L.A.; Baer, A.N.; Challacombe, S.; Lanfranchi, H.; Schiødt, M.; Umehara, H.; Vivino, F.; Zhao, Y.; et al. American College of Rheumatology Classification Criteria for Sjögren’s Syndrome: A Data-Driven, Expert Consensus Approach in the SICCA Cohort. Arthritis Care Res. 2012, 64, 475–487. [Google Scholar] [CrossRef]
- Shiboski, C.H.; Shiboski, S.C.; Seror, R.; Criswell, L.A.; Labetoulle, M.; Lietman, T.M.; Rasmussen, A.; Scofield, H.; Vitali, C.; Bowman, S.J.; et al. 2016 American College of Rheumatology/European League Against Rheumatism Classification Criteria for Primary Sjögren’s Syndrome: A Consensus and Data-Driven Methodology Involving Three International Patient Cohorts. Arthritis Rheumatol. 2017, 69, 35–45. [Google Scholar] [CrossRef]
- Le Goff, M.; Cornec, D.; Jousse-Joulin, S.; Costa, S.; Guellec, D.; Marhadour, T.; Le Berre, R.; Genestet, S.; Cochener, B.; Boisrame-Gastrin, S.; et al. Comparison of 2002 AECG and 2016 ACR/EULAR classification criteria and added value of salivary gland ultrasonography in a patient cohort with suspected primary Sjögren’s syndrome. Arthritis Res. Ther. 2017, 19, 269. [Google Scholar] [CrossRef]
- Franceschini, F.; Cavazzana, I.; Andreoli, L.; Tincani, A. The 2016 classification criteria for primary Sjogren’s syndrome: What’s new? BMC Med. 2017, 15, 69. [Google Scholar] [CrossRef] [PubMed]
- Stang, A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of non-randomized studies in metaanalyses. Eur. J. Epidemiol. 2010, 25, 603–605. [Google Scholar] [CrossRef]
- Segal, B.M.; Pogatchnik, B.; Holker, E.; Liu, H.; Sloan, J.; Rhodus, N.; Moser, K.L. Primary Sjogren’s syndrome: Cognitive symptoms, mood, and cognitive performance. Acta Neurol. Scand. 2012, 125, 272–278. [Google Scholar] [CrossRef] [PubMed]
- Le Guern, V.; Belin, C.; Henegar, C.; Moroni, C.; Maillet, D.; Lacau, C.; Dumas, J.L.; Vigneron, N.; Guillevin, L. Cognitive function and 99mTc-ECD brain SPECT are significantly correlated in patients with primary Sjogren syndrome: A case-control study. Ann. Rheum. Dis. 2010, 69, 132–137. [Google Scholar] [CrossRef] [PubMed]
- Tezcan, M.E.; Kocer, E.B.; Haznedaroglu, S.; Sonmez, C.; Nercan, R.; Yucel, A.A.; Irkec, C.; Bitik, B.; Goker, B. Primary Sjögren’s syndrome is associated with significant cognitive dysfunction. Int. J. Rheum. Dis. 2016, 19, 981–988. [Google Scholar] [CrossRef] [PubMed]
- Delalande, S.; de Seze, J.; Fauchais, A.-L.; Hachulla, E.; Stojkovic, T.; Ferriby, D.; Dubucquoi, S.; Pruvo, J.-P.; Vermersch, P.; Hatron, P.-Y. Neurologic manifestations in primary Sjögren syndrome: A study of 82 patients. Medicine 2004, 83, 280–291. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, D.N.; Hora, J.S.; Salgado, M.C. A short neuropsychological evaluation of patients with primary Sjögren’s syndrome. Arq. Neuropsiquiatr. 2014, 72, 38–43. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Morreale, M.; Francia, A.; Marchione, P.; Manuppella, F.; Giacomini, P. Intracranial hemodynamic changes in primary Sjögren syndrome: A transcranial Doppler case-control study. Neurol. Sci. 2015, 36, 1589–1595. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, K.; Hatate, J.; Toratani, N.; Sugiura, S.; Shimizu, Y.; Takahash, T.; Ito, T.; Fukunaga, R. Prevalence of Sjögren’s syndrome with dementia in a memory clinic. J. Neurol. Sci. 2012, 322, 217–221. [Google Scholar] [CrossRef]
- Lin, T.-M.; Chen, W.-S.; Sheu, J.-J.; Chen, Y.-H.; Chen, J.-H.; Chang, C.-C. Autoimmune rheumatic diseases increase dementia risk in middle-aged patients: A nationwide cohort study. PLoS ONE 2018, 13, e0186475. [Google Scholar] [CrossRef]
- Omma, A.; Tecer, D.; Kucuksahin, O.; Sandikci, S.C.; Yildiz, F.; Erten, S. Do the European League Against Rheumatism (EULAR) Sjögren’s syndrome outcome measures correlate with impaired quality of life, fatigue, anxiety and depression in primary Sjögren’s syndrome? Arch. Med. Sci. 2018, 14, 830–837. [Google Scholar] [CrossRef] [PubMed]
- Koçer, B.; Tezcan, M.E.; Batur, H.Z.; Haznedaroğlu, S.; Göker, B.; İrkeç, C.; Çetinkaya, R. Cognition, depression, fatigue, and quality of life in primary Sjögren’s syndrome: Correlations. Brain Behav. 2016, 6, e00586. [Google Scholar] [CrossRef]
- Wouters, E.J.M.; van Leeuwen, N.V.; Bossema, E.R.; Kruize, A.A.; Bootsma, H.; Bijlsma, J.W.J.; Geenen, R. Physical activity and physical activity cognitions are potential factors maintaining fatigue in patients with primary Sjogren’s syndrome. Ann. Rheum. Dis. 2012, 71, 668–673. [Google Scholar] [CrossRef]
- Hartkamp, A.; Geenen, R.; Bijl, M.; Kruize, A.A.; Godaert, G.L.R.; Derksen, R.H.W.M. Serum cytokine levels related to multiple dimensions of fatigue in patients with primary Sjogren’s syndrome. Ann. Rheum. Dis. 2004, 63, 1335–1337. [Google Scholar] [CrossRef]
- Blanc, F.; Longato, N.; Jung, B.; Kleitz, C.; Di Bitonto, L.; Cretin, B.; Collongues, N.; Sordet, C.; Fleury, M.; Poindron, V.; et al. Cognitive Dysfunction and Dementia in Primary Sjögren’s Syndrome. ISRN Neurol. 2013, 19, 501327. [Google Scholar] [CrossRef] [PubMed]
- Dziadkowiak, E.; Sebastian, A.; Wiland, P.; Waliszewska-Prosol, M.; Zagrajek, M.; Ejma, M. Endogenous event-related potentials in patients with primary Sjögren’s syndrome without central nervous system involvement. Scand. J. Rheumatol. 2015, 44, 487–494. [Google Scholar] [CrossRef]
- Goodchild, C.E.; Treharne, G.J.; Booth, D.A.; Bowman, S.J. Daytime patterning of fatigue and its associations with the previous night’s discomfort and poor sleep among women with primary Sjögren’s syndrome or rheumatoid arthritis. Musculoskelet. Care 2010, 8, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Lauvsnes, M.B.; Maroni, S.S.; Appenzeller, S.; Beyer, M.K.; Greve, O.J.; Kvaloy, J.T.; Harboe, E.; Goransson, L.G.; Tjensvoll, A.B.; Omdal, R. Memory dysfunction in primary Sjögren’s syndrome is associated with anti-NR2 antibodies. Arthritis Rheum. 2013, 65, 3209–3217. [Google Scholar] [CrossRef] [PubMed]
- Hackett, K.L.; Deary, V.; Deane, K.H.; Newton, J.L.; Ng, W.-F.; Rapley, T. Experience of sleep disruption in primary Sjögren’s syndrome: A focus group study. Br. J. Occup. Ther. 2018, 81, 218–226. [Google Scholar] [CrossRef] [PubMed]
- Rosado, S.N.; Silveira, V.; Reis, A.I.; Gordinho, A.; Noronha, C. Catatonia and psychosis as manifestations of primary Sjogren’s syndrome. Eur. J. Case Rep. Intern. Med. 2018, 5. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, Y.; Kimura, A.; Kato, S.; Koumura, A.; Sakurai, T.; Tanaka, Y.; Hozumi, I.; Sunden, Y.; Orba, Y.; Sawa, H.; et al. Progressive multifocal leukoencephalopathy and CD4+ T-lymphocytopenia in a patient with Sjögren syndrome. J. Neurol. Sci. 2008, 268, 195–198. [Google Scholar] [CrossRef]
- Hirohata, M.; Yasukawa, Y.; Ishida, C.; Komai, K.; Yamada, M. Reversible cortical lesions in primary Sjögren’s syndrome presenting with meningoencephalitis as an initial manifestation. J. Neurol. Sci. 2005, 232, 111–113. [Google Scholar] [CrossRef] [PubMed]
- Arends, S.; Meiners, P.M.; Moerman, R.V.; Kroese, F.G.; Brouwer, E.; Spijkervet, F.K.; Vissink, A.; Bootsma, H. Physical fatigue characterises patient experience of primary Sjögren’s syndrome. Clin. Exp. Rheumatol. 2017, 35, 255–261. [Google Scholar] [PubMed]
- Bodewes, I.L.A.; van der Spek, P.J.; Leon, L.G.; Wijkhuijs, A.J.M.; van Helden-Meeuwsen, C.G.; Tas, L.; Schreurs, M.W.J.; van Daele, P.L.A.; Katsikis, P.D.; Versnel, M.A. Fatigue in Sjögren’s Syndrome: A Search for Biomarkers and Treatment Targets. Front. Immunol. 2019, 10, 312. [Google Scholar] [CrossRef] [PubMed]
- Seror, R.; Ravaud, P.; Bowman, S.J.; Baron, G.; Tzioufas, A.; Theander, E.; Gottenberg, J.-E.; Bootsma, H.; Mariette, X.; Vitali, C. EULAR Sjogren’s syndrome disease activity index: Development of a consensus systemic disease activity index for primary Sjogren’s syndrome. Ann. Rheum. Dis. 2010, 69, 1103–1109. [Google Scholar] [CrossRef]
- Seror, R.; Bowman, S.J.; Brito-Zeron, P.; Theander, E.; Bootsma, H.; Tzioufas, A.; Gottemberg, J.E.; Roman-Casals, M.; Dorner, T.; Ravaud, P.; et al. EULAR Sjögren’s syndrome disease activity index (ESSDAI): A user guide. RMD Open 2015, 1, e000022. [Google Scholar] [CrossRef]
- Bowman, S.J.; Sutcliffe, N.; Isenberg, D.A.; Goldblatt, F.; Adler, M.; Price, E.; Canavan, A.; Hamburger, J.; Richards, A.; Regan, S.R.M.; et al. Sjögren’s Systemic Clinical Activity Index (SCAI)–A systemic disease activity measure for use in clinical trials in primary Sjögren’s syndrome. Rheumatology 2007, 46, 1845–1851. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Coates, T.; Slavotinek, J.P.; Rischmueller, M.; Schultz, D.; Anderson, C.; Dellamelva, M. Cerebral white matter lesions in primary Sjögren’s syndrome: A controlled study. J. Rheumatol. 1999, 26, 1301–1305. [Google Scholar]
- Ambrose, K.R.; Gracely, R.H.; Glass, J.M. Fibromyalgia dyscognition: Concepts and issue. Reumatismo 2012, 64, 206–215. [Google Scholar] [CrossRef]
- Torta, R.G.V.; Tesio, V.; Ieraci, V.; Castelli, L.; Zizzi, F.B. Fibro-fog. Clin. Exp. Rheumatol. 2016, 34 (Suppl. 96), S6–S8. [Google Scholar]
- Glass, J.M.; Park, D.C. Cognitive dysfunction in fibromyalgia. Curr. Rheumatol. Rep. 2001, 3, 123–127. [Google Scholar] [CrossRef]
- Choi, B.Y.; Oh, H.J.; Lee, Y.J.; Song, Y.W. Prevalence and clinical impact of fibromyalgia in patients with primary Sjögren’s syndrome. Clin. Exp. Rheumatol. 2016, 34 (Suppl. 96), S9–S13. [Google Scholar]
- Kang, J.H.; Lin, H.C. Comorbidities in patients with primary Sjogren’s syndrome: A registry-based case-control study. J. Rheumatol. 2010, 37, 1188–1194. [Google Scholar] [CrossRef]
- Manzo, C.; Maslinska, M. Primary Sjogren’s syndrome in the elderly: Does age of onset make a difference? EMJ Rheumatol. 2018, 5, 75–82. [Google Scholar]
- Liliang, P.-C.; Liang, C.-L.; Lu, K.; Yang, S.-N.; Hsieh, M.-T.; Tai, Y.-C.; Wang, K.-W. Population-based study suggests an increased risk of Alzheimer’sdisease in Sjögren’s syndrome. Clin. Rheumatol. 2018, 37, 935–941. [Google Scholar] [CrossRef] [PubMed]
- Alexander, E.; Provost, T.; Stevens, M.; Alexander, G.E. Neurologic complications of primary Sjögren’s syndrome. Medicine 1982, 61, 247–257. [Google Scholar] [CrossRef] [PubMed]
- Segal, B.M.; Pogatchnik, B.; Henn, L.; Rudser, K.; Sivils, K.M. Pain severity and neuropathic pain symptoms in primary Sjögren’s syndrome: A comparison study of seropositive and seronegative Sjögren’s syndrome patients. Arthritis Care Res. 2013, 65, 1291–1298. [Google Scholar] [CrossRef] [PubMed]
| 1st Author Year | Study Design | Patients F/M | Cognitive Impairment | Depression | Fatigue | Subset of Cognitive Impairment | Neuro Imaging | Correlation with Extraglandular Manifestations | |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Segal BM, 2012 [20] | Case-control Monocentric data | 39 39/0 | YES 30% psychomotor processing (DST) verbal reasoning (similarities) | Yes, CES_D 47% vs. 6% | Yes, FSS | Frontal and subcortical brain type | Absent | No |
| 2 | Le Guern V, 2009 [21] | Case-control Monocentric data | 10 10/0 | YES 80% visuospatial abilities executive function | Yes Hamilton depression scale | No | Frontal-lobe-related syndrome | MR and 99m Tc-ECD SPECT | Yes 7/10 patients 1 positive for antiphospholipid serology and obstetric APLS 3 still active |
| 3 | Tezcan ME, 2016 [22] | Case-control Monocentric data | 28 28/0 | YES 78.8% immediate verbal memory Clock drawing test attention and information processing speed verbal learning | Yes | Yes | Frontal and subcortical brain type 3/28 (11%) with serious cognitive dysfunction, 11 (39.3%) with MCI, 8 (28.5%) had moderate cognitive dysfunction | Absent | Yes 2/28 patients: One patient liver and one patient lung involvement Antiganglioside antibody 42.8% No active extraglandular manifestation during neurological assessment |
| 4 | Delalande S, 2004 [23] | Retrospective study Monocentric data | 82 65/17 | YES 13.04% Authors did not systematically screen their patients with pSS for cognitive impairment. | No | No | Not clear In 2 patients, subcortical dementia | Yes Brain MRI | Yes 71/86 in 47% of patients absence of sicca symptoms! |
| 5 | Rodrigues DN, 2014 [24] | Case-control Monocentric data | 18 18/0 | YES Executive functioning Long-term memory tests | Yes Beck Depression Inventory | No | Subcortical brain type | Not reported | No |
| 6 | Morreale M, 2015 [25] | Case-control Monocentric data | 87 78/9 | Yes 31.1% Executive dysfunction Visuospatial disorders Short-term memory deficits | Yes Beck Depression Inventory | Not clear | Not clear Patients with recent diagnosis (<6 months) of pSS | Yes Brain MRI Transcranial Doppler | Yes |
| 7 | Yoshikawa K, 2012 [26] | Prospective Study Cohort of patients with dementia or cognitive dysfunction Monocentric data | 20 15/5 | YES 13 had dementia (4 Alzheimer’s type; 6 vascular dementia; 1 mixed dementia) 7 had mild cognitive impairment | Yes | No | 4 Alzheimer’s type 6 vascular dementia 1 mixed dementia 1 normal pressure hydrocephalus 8 mild cognitive impairment: 5 vascular MCI | YES Brain MRI revealing subcortical lesions. Brain SPECT revealing asymmetrical focal hypoperfusion. | No |
| 8 | Lin TM, 2018 [27] | Population study Taiwan National Health Insurance Research Database | 4756 not possible | YES 238 patients had dementias No information about the type of dementia is present. | No | No | Not specified | Yes | No The NHIRD does not contain such parameters such as clinical severity and laboratory data. |
| 9 | Omma A, 2018 [28] | Case-control Multicenter data Cross-sectional study | 97/8 | YES SF-36 low score for the mental-health subdimension | YES | YES | Not clear | No | Not clear |
| 10 | Ye W, 2018 [7] | Cohort study Monocentric data Cross-sectional study | 415 10/1 ratio | YES 1.03% 4/415 had cognitive dysfunction | Yes 10 pts had depression | No | Not reported | YES Brain MRI | YES 125/415 pts |
| 11 | Koçer B, 2016 [29] | Case-control Monocentric data | 32 32/0 | YES Low performance in Clock Drawing, COWAT, PASAT, Colourless Word Reading (Stroop1) and Recognizing Colours (Stroop2) Patterns of STROOP test, AVLT immediate verbal memory (A1) and long-term verbal memory (A7) patterns, BJLOT, and in all the patterns of RCFT in PSS patients compared to the healthy control group (p < 0.05) | Yes SF (pain) 53.44 Hamilton + in 6.69 | Yes | Not reported | YES SPECT (Hypo perfusion in parietal, temporal, and frontal lobes was 56.3%) | No |
| 12 | Wouters EJM, 2012 [30] | Case-control Two-centre data | 294 273/21 | No | Not reported | Yes | Not reported | Absent | No |
| 13 | Hartkamp A, 2004 [31] | Case-control Two-centre data | 60 60/0 | No | Yes MFI; General fatigue and physical fatigue were present in 75%, reduced activity in 63%, and reduced motivation and mental fatigue in 52% | No | Not reported | Absent | Yes Cytokine level above the lower range of detection of the assay used was present in 13% of patients for IL2, 5 (8%) for IL6, 32% for IL10, and 83% for TNFa |
| 14 | Blanc F, 2013 [32] | Case-control Monocentric data Prospective data | 25 21/4 | Yes 60% Five patients had dementia in the PSS group. Ten patients had MCI, five had a non-amnestic single domain, and five an amnestic MCI multi-domain. | No, but two pSS patients took antidepressant drugs at the time of evaluation. | No | Five patients had dementia. 10 patients had MCI, five had a non-amnestic single domain, and five an amnestic MCI multi-domain | Brain MRI. A trend towards a correlation was found between WML and the severity of cognitive dysfunctions | No. All patients had sicca symptoms and in 20 of these abnormal histopathology of the minor salivary glands was found. No correlation was found between the severity of cognitive impairment and duration of pSS. |
| 15 | Dziadkowiak E, 2015 [33] | Case-control Monocentric data | 30 29/1 | No MMSE and CDT normal | Yes Subjective depressed mood >2 w 20% | Yes Subjective chronic fatigue 63% | Not Reported | Absent Endogenous cognitive event-related potentials (CERPs) | Yes Sicca symptoms 96%, lymphocytic infiltration in labial salivary gland 93%: there was a significant prolongation of the latency of P300 and N200 potentials |
| 16 | Goodchild CE, 2010 [34] | Cohorts pSS and RA Prospective Multicentric (clinics) | 14 14/0 | No | No | Yes Mental fatigue 1.74, somatic fatigue 2.83 | Not Reported | Absent | Yes Oral sicca 2.92, ocular sicca 2.36 (profile of discomfort) |
| 17 | Lauvsnes MB, 2013 [35] | Case-control Monocentric data | 66 58/8 | No | Yes BDI (30%) | No | Not reported | MRI revealed a smaller hippocampus volume | Yes Anti-NR2 antibodies (were associated with a worse performance in 8 of 10 memory and learning tests); in 20% of pSS patients they were above the cutoff value |
| 18 | Hackett K, 2015 [36] | Case-control Cross sectional Multicentric | 105 ? | Not reported | Yes Multivariate analysis (with HAD) 0.428 | Yes | Not reported | Absent | Yes Dryness VAS (multivariate analysis 0.087) |
| 19 | Rosado SN, 2018 [37] | Case report | 1 1/0 | Yes Neuropsychiatric manifestations associated with severe cognitive dysfunction | Yes | No | Not reported | MRI Normal | Yes AntiRo, antiLa +, Schimer test + Weakly positive for IgM anticardiolipin and anti-B2-glicoprotein 1 with negative IgG. |
| 20 | Hayashi Y, 2008 [38] | Case report | 1 0/1 | Yes 100% Progressive dementia and gait disturbance | No | No | Yes | MRI T1-weighted images with low-intensity lesions, and T2-weighted and FLAIR images with high-intensity lesions in the left frontal white matter, bilateral parietal and left occipital white matter, left thalamus, and right middle cerebellar peduncle, which were not enhanced by Gadolinium | No. Saxon and Shirmer tests Scintigraphy of the parotid and submandibular glands, and lip biopsy that revealed many lymphocytes and plasma cells infiltrating the small salivary glands |
| 21 | Hirohata M, 2005 [39] | Case report | 1 1/0 | Yes 100% Forgetfulness (memory disturbance) | No | No | Yes | MRI, SPECT SPECT perfusion imaging for cerebral blood flow revealed regions of decreased uptake in the parietal lobes. Brain MRI and magnetic resonance angiography (MRA) were normal |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manzo, C.; Martinez-Suarez, E.; Kechida, M.; Isetta, M.; Serra-Mestres, J. Cognitive Function in Primary Sjögren’s Syndrome: A Systematic Review. Brain Sci. 2019, 9, 85. https://doi.org/10.3390/brainsci9040085
Manzo C, Martinez-Suarez E, Kechida M, Isetta M, Serra-Mestres J. Cognitive Function in Primary Sjögren’s Syndrome: A Systematic Review. Brain Sciences. 2019; 9(4):85. https://doi.org/10.3390/brainsci9040085
Chicago/Turabian StyleManzo, Ciro, Eva Martinez-Suarez, Melek Kechida, Marco Isetta, and Jordi Serra-Mestres. 2019. "Cognitive Function in Primary Sjögren’s Syndrome: A Systematic Review" Brain Sciences 9, no. 4: 85. https://doi.org/10.3390/brainsci9040085
APA StyleManzo, C., Martinez-Suarez, E., Kechida, M., Isetta, M., & Serra-Mestres, J. (2019). Cognitive Function in Primary Sjögren’s Syndrome: A Systematic Review. Brain Sciences, 9(4), 85. https://doi.org/10.3390/brainsci9040085






