Dual Mode Gait Sonification for Rehabilitation After Unilateral Hip Arthroplasty
Abstract
1. Introduction
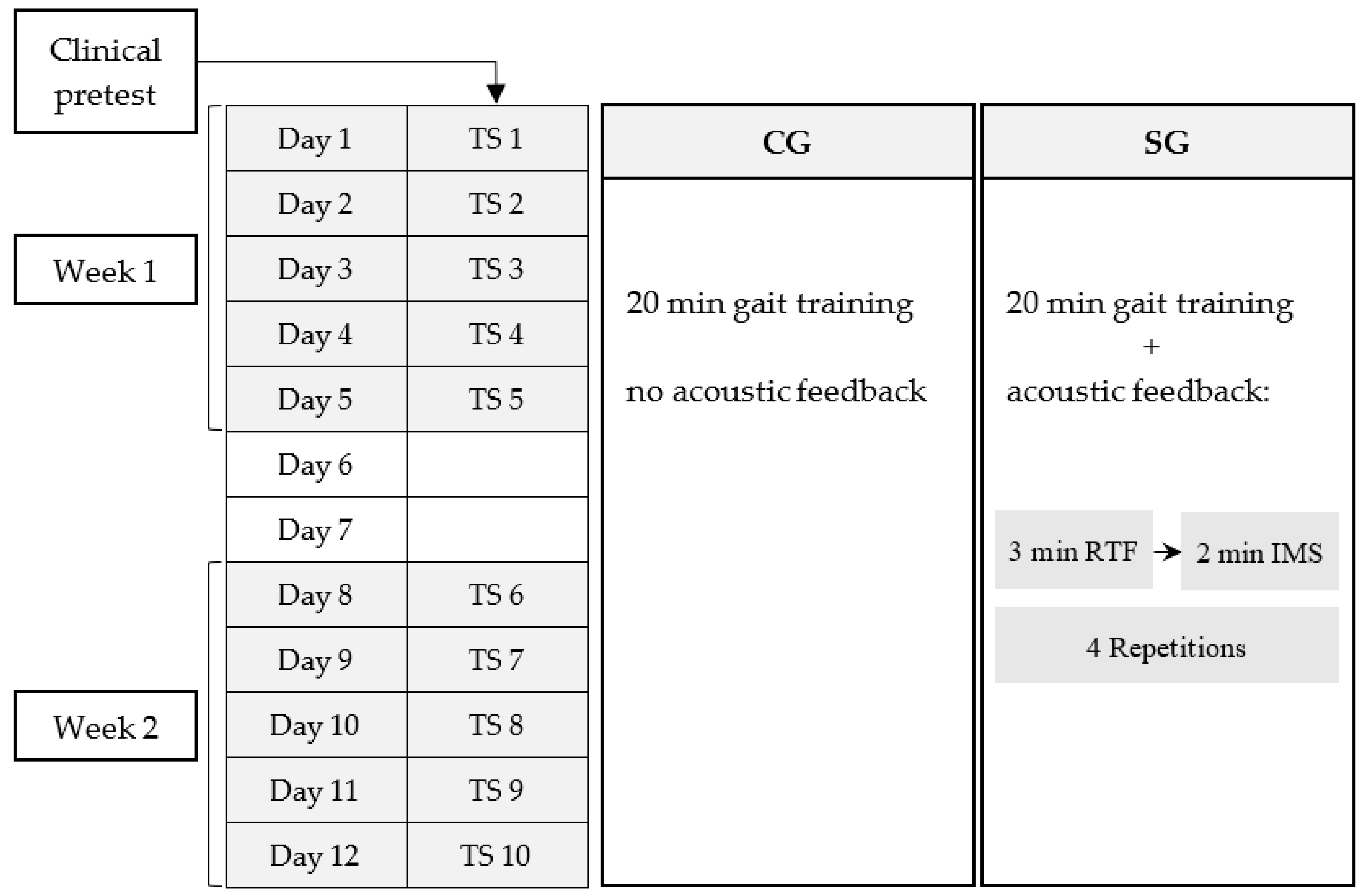
2. Materials and Methods
3. Results
3.1. Symmetry
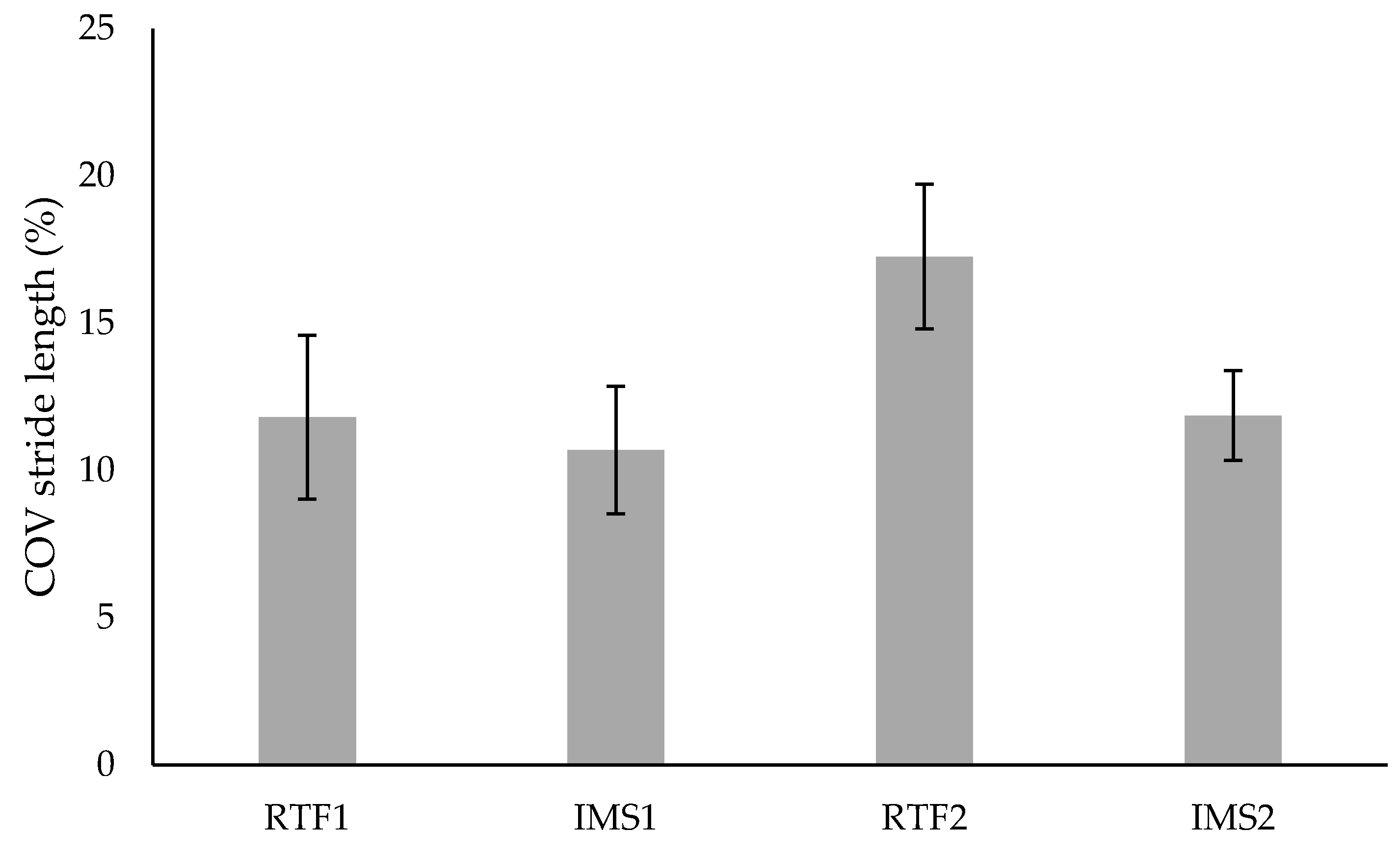
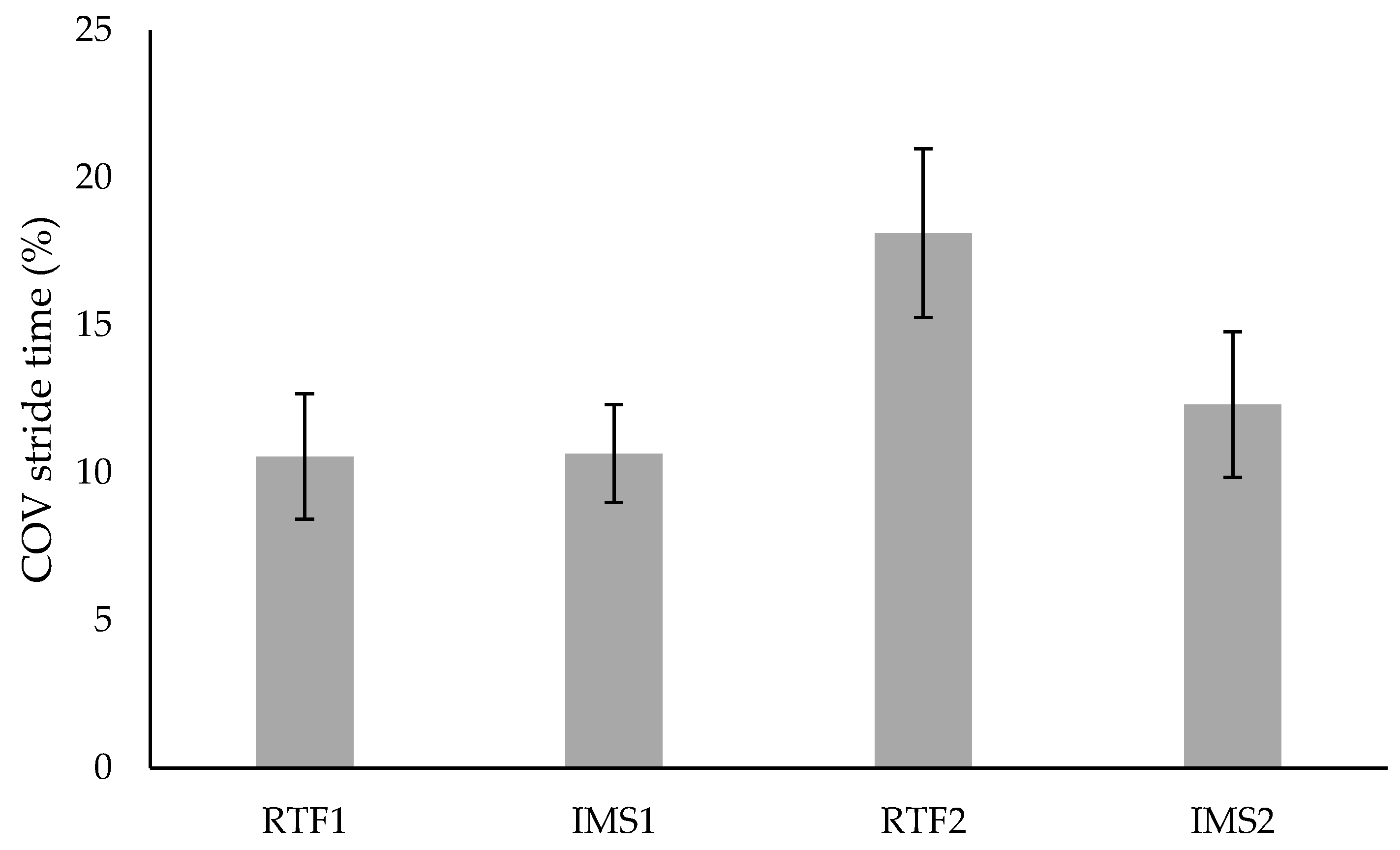
3.2. Variability
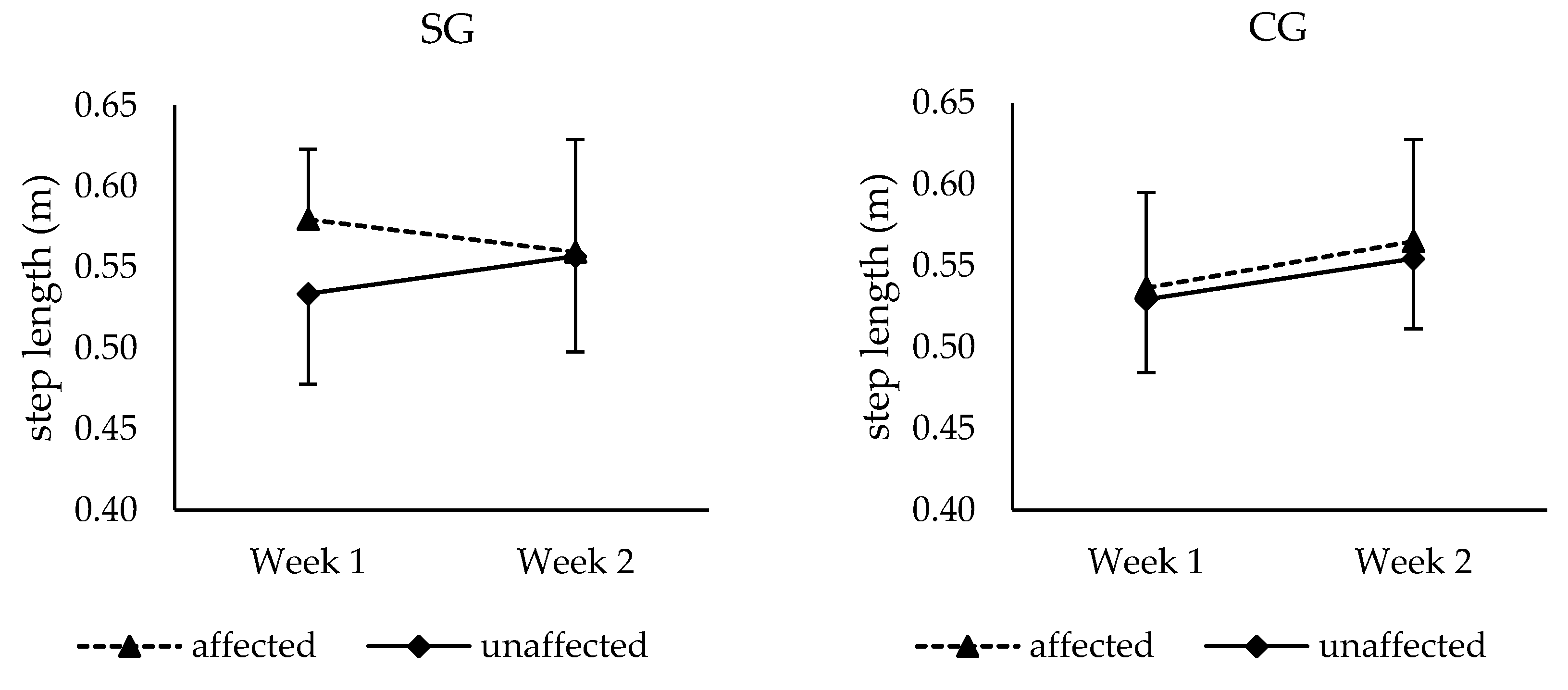
3.3. Temporo-Spatial Parameters
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Henriques, D.Y.P.; Cressman, E.K. Visuomotor adaptation and proprioceptive recalibration. J. Mot. Behav. 2012, 44, 435–444. [Google Scholar] [CrossRef] [PubMed]
- Molier, B.I.; van Asseldonk, E.H.F.; Hermens, H.J.; Jannink, M.J.A. Nature, timing, frequency and type of augmented feedback; does it influence motor relearning of the hemiparetic arm after stroke? A systematic review. Disabil. Rehabil. 2010, 32, 1799–1809. [Google Scholar] [CrossRef] [PubMed]
- Yen, S.-C.; Landry, J.M.; Wu, M. Augmented multisensory feedback enhances locomotor adaptation in humans with incomplete spinal cord injury. Hum. Mov. Sci. 2014, 35, 80–93. [Google Scholar] [CrossRef] [PubMed]
- Lauber, B.; Keller, M. Improving motor performance: Selected aspects of augmented feedback in exercise and health. Eur. J. Sport Sci. 2014, 14, 36–43. [Google Scholar] [CrossRef]
- Hunt, M.A. Movement retraining using real-time feedback of performance. J. Vis. Exp. 2013, e50182. [Google Scholar] [CrossRef] [PubMed]
- Merians, A.S.; Jack, D.; Boian, R.; Tremaine, M.; Burdea, G.C.; Adamovich, S.V.; Recce, M.; Poizner, H. Virtual Reality–Augmented Rehabilitation for Patients Following Stroke. Phys. Ther. 2002, 82, 898–915. [Google Scholar] [CrossRef]
- Zimmerli, L.; Duschau-Wicke, A.; Mayr, A.; Riener, R.; Lunenburger, L. Virtual reality and gait rehabilitation Augmented feedback for the Lokomat. In Proceedings of the Virtual Rehabilitation International Conference, Haifa, Israel, 29 June–2 July 2009; pp. 150–153. [Google Scholar]
- Lünenburger, L.; Colombo, G.; Riener, R. Biofeedback for robotic gait rehabilitation. J. Neuroeng. Rehabil. 2007, 4, 1. [Google Scholar] [CrossRef] [PubMed]
- Thaut, M.H.; Leins, A.K.; Rice, R.R.; Argstatter, H.; Kenyon, G.P.; McIntosh, G.C.; Bolay, H.V.; Fetter, M. Rhythmic auditory stimulation improves gait more than NDT/Bobath training in near-ambulatory patients early poststroke: A single-blind, randomized trial. Neurorehabilit. Neural Repair 2007, 21, 455–459. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-J.; Krebs, H.I. Effects of implicit visual feedback distortion on human gait. Exp. Brain Res. 2012, 218, 495–502. [Google Scholar] [CrossRef]
- van den Noort, J.C.; Steenbrink, F.; Roeles, S.; Harlaar, J. Real-time visual feedback for gait retraining: Toward application in knee osteoarthritis. Med. Biol. Eng. Comput. 2015, 53, 275–286. [Google Scholar] [CrossRef]
- Uzor, S.; Baillie, L.; Skelton, D.A.; Rowe, P.J. Falls prevention advice and visual feedback to those at risk of falling: Study protocol for a pilot randomized controlled trial. Trials 2013, 14, 79. [Google Scholar] [CrossRef]
- Graci, V.; Elliott, D.B.; Buckley, J.G. Peripheral visual cues affect minimum-foot-clearance during overground locomotion. Gait Posture 2009, 30, 370–374. [Google Scholar] [CrossRef]
- Goodworth, A.; Perrone, K.; Pillsbury, M.; Yargeau, M. Effects of visual focus and gait speed on walking balance in the frontal plane. Hum. Mov. Sci. 2015, 42, 15–26. [Google Scholar] [CrossRef]
- Berard, J.; Fung, J.; Lamontagne, A. Impact of aging on visual reweighting during locomotion. Clin. Neurophysiol. 2012, 123, 1422–1428. [Google Scholar] [CrossRef]
- Bock, O. Dual-task costs while walking increase in old age for some, but not for other tasks: An experimental study of healthy young and elderly persons. J. Neuroeng. Rehabil. 2008, 5, 27. [Google Scholar] [CrossRef]
- Iversen, J.R.; Patel, A.D.; Nicodemus, B.; Emmorey, K. Synchronization to auditory and visual rhythms in hearing and deaf individuals. Cognition 2015, 134, 232–244. [Google Scholar] [CrossRef]
- Grahn, J.A. See what I hear? Beat perception in auditory and visual rhythms. Exp. Brain Res. 2012, 220, 51–61. [Google Scholar] [CrossRef]
- Waterhouse, J.; Hudson, P.; Edwards, B. Effects of music tempo upon submaximal cycling performance. Scand. J. Med. Sci. Sports 2010, 20, 662–669. [Google Scholar] [CrossRef]
- Effenberg, A.O.; Fehse, U.; Schmitz, G.; Krueger, B.; Mechling, H. Movement Sonification: Effects on Motor Learning beyond Rhythmic Adjustments. Front. Neurosci. 2016, 10, 219. [Google Scholar] [CrossRef]
- Schaffert, N.; Mattes, K. Effects of acoustic feedback training in elite-standard Para-Rowing. J. Sports Sci. 2015, 33, 411–418. [Google Scholar] [CrossRef]
- Cesarini, D.; Hermann, T.; Ungerechts, B. A real-time auditory biofeedback system for sports swimming. In Proceedings of the 20th International Conference on Auditory Display (ICAD 2014), New York, NY, USA, 22 June 2014; Stockmann, T., Metatla, O., MacDonald, D., Eds.; Workshop on Sonification for Sports and Performance. International Community for Auditory Display: New York, NY, USA, 2014. [Google Scholar]
- Eriksson, M.; Bresin, R. Improving running mechanics by use of interactive sonification. In Proceedings of the Interaction Sonification Workshop (ISon); Bresin, R., Hermann, T., Hunt, A., Eds.; KTH Royal Institute of Technology: Stockholm, Sweden, 2010; pp. 95–98. [Google Scholar]
- Schauer, M.; Mauritz, K.-H. Musical motor feedback (MMF) in walking hemiparetic stroke patients: Randomized trials of gait improvement. Clin. Rehabil. 2003, 17, 713–722. [Google Scholar] [CrossRef]
- Krause, D.; Agethen, M.; Zobe, C. Error Feedback Frequency Affects Automaticity But Not Accuracy and Consistency after Extensive Motor Skill Practice. J. Mot. Behav. 2018, 50, 144–154. [Google Scholar] [CrossRef]
- Sigrist, R.; Rauter, G.; Riener, R.; Wolf, P. Augmented visual, auditory, haptic, and multimodal feedback in motor learning: A review. Psychon. Bull. Rev. 2013, 20, 21–53. [Google Scholar] [CrossRef]
- Baram, Y.; Aharon-Peretz, J.; Badarny, S.; Susel, Z.; Schlesinger, I. Closed-loop auditory feedback for the improvement of gait in patients with Parkinson’s disease. J. Neurol. Sci. 2016, 363, 104–106. [Google Scholar] [CrossRef]
- Wittwer, J.E.; Webster, K.E.; Hill, K. Music and metronome cues produce different effects on gait spatiotemporal measures but not gait variability in healthy older adults. Gait Posture 2013, 37, 219–222. [Google Scholar] [CrossRef]
- Maulucci, R.A.; Eckhouse, R.H. Retraining reaching in chronic stroke with real-time auditory feedback. NeuroRehabilitation 2001, 16, 171–182. [Google Scholar]
- Effenberg, A.O. Movement Sonification: Effects on Perception and Action. IEEE Multimed. 2005, 12, 53–59. [Google Scholar] [CrossRef]
- Ghai, S.; Schmitz, G.; Hwang, T.-H.; Effenberg, A.O. Auditory Proprioceptive Integration: Effects of Real-Time Kinematic Auditory Feedback on Knee Proprioception. Front. Neurosci. 2018, 12, 142. [Google Scholar] [CrossRef]
- Schmitz, G.; Bergmann, J.; Effenberg, A.O.; Krewer, C.; Hwang, T.-H.; Müller, F. Movement Sonification in Stroke Rehabilitation. Front. Neurol. 2018, 9, 389. [Google Scholar] [CrossRef]
- Stienstra, J.; Overbeeke, K.; Wensveen, S. Embodying complexity through movement sonification. In Proceedings of the 9th ACM SIGCHI Italian Chapter International Conference on Computer-Human Interaction Facing Complexity, Alghero, Italy, 13–16 September 2011; Marti, P., Ed.; ACM: New York, NY, USA, 2011; p. 39, ISBN 9781450308762. [Google Scholar]
- Rochester, L.; Baker, K.; Hetherington, V.; Jones, D.; Willems, A.-M.; Kwakkel, G.; van Wegen, E.; Lim, I.; Nieuwboer, A. Evidence for motor learning in Parkinson’s disease: Acquisition, automaticity and retention of cued gait performance after training with external rhythmical cues. Brain Res. 2010, 1319, 103–111. [Google Scholar] [CrossRef]
- Ford, M.P.; Malone, L.A.; Nyikos, I.; Yelisetty, R.; Bickel, C.S. Gait training with progressive external auditory cueing in persons with Parkinson’s disease. Arch. Phys. Med. Rehabil. 2010, 91, 1255–1261. [Google Scholar] [CrossRef]
- Harrison, E.C.; Horin, A.P.; Earhart, G.M. Internal cueing improves gait more than external cueing in healthy adults and people with Parkinson disease. Sci. Rep. 2018, 8, 15525. [Google Scholar] [CrossRef]
- Young, W.; Rodger, M.; Craig, C.M. Perceiving and reenacting spatiotemporal characteristics of walking sounds. J. Exp. Psychol. Hum. Percept. Perform. 2013, 39, 464–476. [Google Scholar] [CrossRef]
- Ghai, S.; Ghai, I.; Effenberg, A.O. Effect of Rhythmic Auditory Cueing on Aging Gait: A Systematic Review and Meta-Analysis. Aging Dis. 2018, 9, 901–923. [Google Scholar] [CrossRef]
- Fischer, T.; Kiselka, A.; Dlapka, R.; Doppler, J.; Iber, M.; Gradl, C.; Gorgas, A.-M.; Siragy, T.; Horsak, B. An Auditory Feedback System in Use with People Aged +50 Years: Compliance and Modifications in Gait Pattern. In Converging Clinical and Engineering Research on Neurorehabilitation II: Proceedings of the 3rd International Conference on NeuroRehabilitation (ICNR2016), Segovia, Spain, 18–21 October 2016; Ibáñez, J., González, J., Azorín, J.M., Akay, M., Pons, J.L., Eds.; Springer: Cham, Switzerland, 2016; pp. 881–885. ISBN 978-3-319-46668-2. [Google Scholar]
- Rodger, M.W.M.; Young, W.R.; Craig, C.M. Synthesis of walking sounds for alleviating gait disturbances in Parkinson’s disease. IEEE Trans. Neural Syst. Rehabil. Eng. 2014, 22, 543–548. [Google Scholar] [CrossRef]
- Horsak, B.; Dlapka, R.; Iber, M.; Gorgas, A.-M.; Kiselka, A.; Gradl, C.; Siragy, T.; Doppler, J. SONIGait: A wireless instrumented insole device for real-time sonification of gait. J. Multimodal User Interfaces 2016, 10, 195–206. [Google Scholar] [CrossRef]
- Hajinejad, N.; Vatterrott, H.-R.; Grüter, B.; Bogutzky, S. GangKlang: Designing walking experiences. In Proceedings of the 8th Audio Mostly Conference, Piteå, Sweden, 18–20 September 2013; ACM: New York, NY, USA, 2013; pp. 1–6. [Google Scholar]
- Zanotto, D.; Rosati, G.; Avanzini, F.; Stegall, P.; Agrawal, S.K. Robot-assisted gait training with complementary auditory feedback: Results on short-term motor adaptation. In Proceedings of the 4th IEEE RAS & EMBS International Conference on Biomedical Robotics and Biomechatronics (BioRob), Rome, Italy, 24–27 June 2012; IEEE: Piscataway, NJ, USA, 2012; pp. 1388–1393, ISBN 978-1-4577-1200-5. [Google Scholar]
- Vogt, K.; Pirrò, D.; Kobenz, I.; Höldrich, R.; Eckel, G. PhysioSonic—Evaluated Movement Sonification as Auditory Feedback in Physiotherapy. In Auditory Display, Proceeding of the 6th International Symposium on CMMR/ICAD 2009, Copenhagen, Denmark, 18–22 May 2009; Ystad, S., Aramaki, M., Kronland-Martinet, R., Jensen, K., Eds.; Revised Papers; Springer: Berlin, Germany, 2010; pp. 103–120. ISBN 978-3-642-12438-9. [Google Scholar]
- Brodie, M.A.D.; Dean, R.T.; Beijer, T.R.; Canning, C.G.; Smith, S.T.; Menant, J.C.; Lord, S.R. Symmetry matched auditory cues improve gait steadiness in most people with Parkinson’s disease but not in healthy older people. J. Parkinson’s Dis. 2015, 5, 105–116. [Google Scholar] [CrossRef]
- Tajadura-Jiménez, A.; Basia, M.; Deroy, O.; Fairhurst, M.; Marquardt, N.; Bianchi-Berthouze, N. As Light as your Footsteps. In Proceedings of the 33rd Annual ACM Conference on Human Factors in Computing Systems, Seoul, Republic of Korea, 18–23 April 2015; pp. 2943–2952. [Google Scholar]
- Constantinou, M.; Barrett, R.; Brown, M.; Mills, P. Spatial-temporal gait characteristics in individuals with hip osteoarthritis: A systematic literature review and meta-analysis. J. Orthop. Sports Phys. Ther. 2014, 44, 291–297. [Google Scholar] [CrossRef]
- Hodt-Billington, C.; Helbostad, J.L.; Vervaat, W.; Rognsvåg, T.; Moe-Nilssen, R. Changes in gait symmetry, gait velocity and self-reported function following total hip replacement. J. Rehabil. Med. 2011, 43, 787–793. [Google Scholar] [CrossRef]
- Sabeh, K.G.; Rosas, S.; Buller, L.T.; Roche, M.W.; Hernandez, V.H. The Impact of Discharge Disposition on Episode-of-Care Reimbursement after Primary Total Hip Arthroplasty. J. Arthroplast. 2017, 32, 2969–2973. [Google Scholar] [CrossRef]
- Ong, K.L.; Lotke, P.A.; Lau, E.; Manley, M.T.; Kurtz, S.M. Prevalence and Costs of Rehabilitation and Physical Therapy after Primary TJA. J. Arthroplast. 2015, 30, 1121–1126. [Google Scholar] [CrossRef]
- Neogi, T.; Zhang, Y. Epidemiology of osteoarthritis. Rheum. Dis. Clin. North. Am. 2013, 39, 1–19. [Google Scholar] [CrossRef]
- Allen, K.D.; Golightly, Y.M. Epidemiology of osteoarthritis: State of the evidence. Curr. Opin. Rheumatol. 2015, 27, 276–283. [Google Scholar] [CrossRef]
- Reh, J.; Hwang, T.H.; Michalke, V.; Effenberg, A.O. Instruction and real-time sonification for gait rehabilitation after unilateral hip arthroplasty. In Human Movement and Technology: Book of Abstracts, Proceedings of the 11th Joint Conference on Motor Control & Learning, Biomechanics & Training; Wiemeyer, J., Seyfarth, A., Kollegger, G., Tokur, D., Schumacher, C., Hoffmann, K., Schöberl, D., Eds.; Shaker Verlag GmbH: Aachen, Germany, 2016; ISBN 9783844047073. [Google Scholar]
- Bangert, M.; Altenmüller, E.O. Mapping perception to action in piano practice: A longitudinal DC-EEG study. BMC Neurosci. 2003, 4, 26. [Google Scholar] [CrossRef]
- Effenberg, A.O.; Schmitz, G. Acceleration and deceleration at constant speed: Systematic modulation of motion perception by kinematic sonification. Ann. N. Y. Acad. Sci. 2018. [Google Scholar] [CrossRef]
- Tagliabue, M.; McIntyre, J. A modular theory of multisensory integration for motor control. Front. Comput. Neurosci. 2014, 8, 1. [Google Scholar] [CrossRef]
- Podsiadlo, D.; Richardson, S. The Timed “Up & Go”: A Test of Basic Functional Mobility for Frail Elderly Persons. J. Am. Geriatr. Soc. 1991, 39, 142–148. [Google Scholar] [CrossRef]
- Queen, R.M.; Watters, T.S.; Abbey, A.N.; Sabesan, V.J.; Vail, T.P.; Bolognesi, M.P. Gait symmetry: A comparison of hip resurfacing and jumbo head total hip arthroplasty patients. J. Arthroplast. 2011, 26, 680–685. [Google Scholar] [CrossRef]
- Miki, H.; Sugano, N.; Hagio, K.; Nishii, T.; Kawakami, H.; Kakimoto, A.; Nakamura, N.; Yoshikawa, H. Recovery of walking speed and symmetrical movement of the pelvis and lower extremity joints after unilateral THA. J. Biomech. 2004, 37, 443–455. [Google Scholar] [CrossRef]
- Hausdorff, J.M.; Lowenthal, J.; Herman, T.; Gruendlinger, L.; Peretz, C.; Giladi, N. Rhythmic auditory stimulation modulates gait variability in Parkinson’s disease. Eur. J. Neurosci. 2007, 26, 2369–2375. [Google Scholar] [CrossRef]
- del Olmo, M.F.; Cudeiro, J. Temporal variability of gait in Parkinson disease: Effects of a rehabilitation programme based on rhythmic sound cues. Parkinsonism Relat. Disord. 2005, 11, 25–33. [Google Scholar] [CrossRef]
- Thaut, M.H.; McIntosh, G.C.; Rice, R.R. Rhythmic facilitation of gait training in hemiparetic stroke rehabilitation. J. Neurol. Sci. 1997, 151, 207–212. [Google Scholar] [CrossRef]
- Hamacher, D.; Hamacher, D.; Herold, F.; Schega, L. Effect of dual tasks on gait variability in walking to auditory cues in older and young individuals. Exp. Brain Res. 2016, 234, 3555–3563. [Google Scholar] [CrossRef]
- Thaut, M.H.; McIntosh, G.C.; Hoemberg, V. Neurobiological foundations of neurologic music therapy: Rhythmic entrainment and the motor system. Front. Psychol. 2014, 5, 1185. [Google Scholar] [CrossRef]
- Terrier, P.; Dériaz, O. Persistent and anti-persistent pattern in stride-to-stride variability of treadmill walking: Influence of rhythmic auditory cueing. Hum. Mov. Sci. 2012, 31, 1585–1597. [Google Scholar] [CrossRef]
- Dingwell, J.B.; John, J.; Cusumano, J.P. Do humans optimally exploit redundancy to control step variability in walking? PLoS Comput. Biol. 2010, 6, e1000856. [Google Scholar] [CrossRef]
- Dhawale, A.K.; Smith, M.A.; Ölveczky, B.P. The Role of Variability in Motor Learning. Annu. Rev. Neurosci. 2017, 40, 479–498. [Google Scholar] [CrossRef]
- Holm, B.; Thorborg, K.; Husted, H.; Kehlet, H.; Bandholm, T. Surgery-induced changes and early recovery of hip-muscle strength, leg-press power, and functional performance after fast-track total hip arthroplasty: A prospective cohort study. PLoS ONE 2013, 8, e62109. [Google Scholar] [CrossRef]
- Winther, S.B.; Husby, V.S.; Foss, O.A.; Wik, T.S.; Svenningsen, S.; Engdal, M.; Haugan, K.; Husby, O.S. Muscular strength after total hip arthroplasty. A prospective comparison of 3 surgical approaches. Acta Orthop. 2016, 87, 22–28. [Google Scholar] [CrossRef]

| SG (n = 10) | CG (n = 10) | |
|---|---|---|
| Age (years) | 64.0 ± 8.8 | 61.9 ± 8.4 |
| Sex | 9 male, 1 female | 7 male, 3 female |
| Duration post-surgery (days) | 11.5 ± 1.6 | 12.0 ± 2.7 |
| Height (m) | 175.1 ± 5.2 | 176.1 ± 4.1 |
| Weight (kg) | 84.4 ± 10.8 | 85.3 ± 12.4 |
| Timed-up and go (s) | 11.58 ± 3.00 | 13.82 ± 6.24 |
| Functional strength (reps) | 12.9 ± 2.2 | 9.7 ± 5.5 |
| Gait Speed (m × s−1) | Cadence (steps × minute−1) | Cadence (steps × minute−1) | Cadence (steps × minute−1) |
|---|---|---|---|
| 155–165 cm | 165–175 cm | ≥175 cm | |
| 0.4 | 78 | 69 | 69 |
| 0.6 | 91 | 80 | 79 |
| 0.8 | 102 | 91 | 88 |
| 1.0 | 113 | 101 | 98 |
| 1.2 | 124 | 111 | 106 |
| 1.4 | 135 | 121 | 115 |
| COV Stride Length (%) | COV Stride Time (%) | ||||
|---|---|---|---|---|---|
| RTF | IMS | RTF | IMS | ||
| SG (n = 10) | Week 1 | 11.81 ± 8.81 | 10.69 ± 6.84 | 10.55 ± 6.73 | 10.65 ± 5.25 |
| Week 2 | 17.26 ± 7.75 | 11.86 ± 4.81 | 18.12 ± 9.02 | 12.32 ±7.81 | |
| CG (n = 10) | Week 1 | 8.27 ± 5.55 | 8.04 ± 6.01 | 7.90 ± 4.80 | 7.86 ± 4.69 |
| Week 2 | 9.40 ± 5.94 | 9.17 ± 7.64 | 9.30 ± 5.87 | 8.94 ± 6.86 | |
| SG + CG (n = 20) | Week 1 | 10.04 ± 7.39 | 9.36 ± 6.42 | 9.22 ± 5.85 | 9.26 ± 5.05 |
| Week 2 | 13.33 ± 7.83 | 10.52 ± 6.36 | 13.71 ± 8.68 | 10.63 ± 7.36 | |
| Gait Speed | Cadence | Stride Length | Stride Time | ||
|---|---|---|---|---|---|
| (m × s−1) | (steps × min−1) | (m) | (ms) | ||
| SG (n = 10) | Week 1 | 0.93 ± 0.14 | 99.32 ± 10.76 | 1.13 ± 0.10 | 1244.5 ± 136.4 |
| Week 2 | 0.97 ± 0.18 | 103.34 ± 14.56 | 1.16 ± 0.13 | 1218.2 ± 161.6 | |
| CG (n = 10) | Week 1 | 0.85 ± 0.14 | 94.88 ± 10.86 | 1.08 ± 0.10 | 1290.6 ± 145.1 |
| Week 2 | 0.95 ± 0.15 | 101.80 ± 12.64 | 1.14 ± 0.09 | 1218.2 ± 158.7 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reh, J.; Hwang, T.-H.; Schmitz, G.; Effenberg, A.O. Dual Mode Gait Sonification for Rehabilitation After Unilateral Hip Arthroplasty. Brain Sci. 2019, 9, 66. https://doi.org/10.3390/brainsci9030066
Reh J, Hwang T-H, Schmitz G, Effenberg AO. Dual Mode Gait Sonification for Rehabilitation After Unilateral Hip Arthroplasty. Brain Sciences. 2019; 9(3):66. https://doi.org/10.3390/brainsci9030066
Chicago/Turabian StyleReh, Julia, Tong-Hun Hwang, Gerd Schmitz, and Alfred O. Effenberg. 2019. "Dual Mode Gait Sonification for Rehabilitation After Unilateral Hip Arthroplasty" Brain Sciences 9, no. 3: 66. https://doi.org/10.3390/brainsci9030066
APA StyleReh, J., Hwang, T.-H., Schmitz, G., & Effenberg, A. O. (2019). Dual Mode Gait Sonification for Rehabilitation After Unilateral Hip Arthroplasty. Brain Sciences, 9(3), 66. https://doi.org/10.3390/brainsci9030066





