Neuromodulation Strategies in Post-Traumatic Stress Disorder: From Preclinical Models to Clinical Applications
Abstract
1. Introduction
2. Preclinical Models
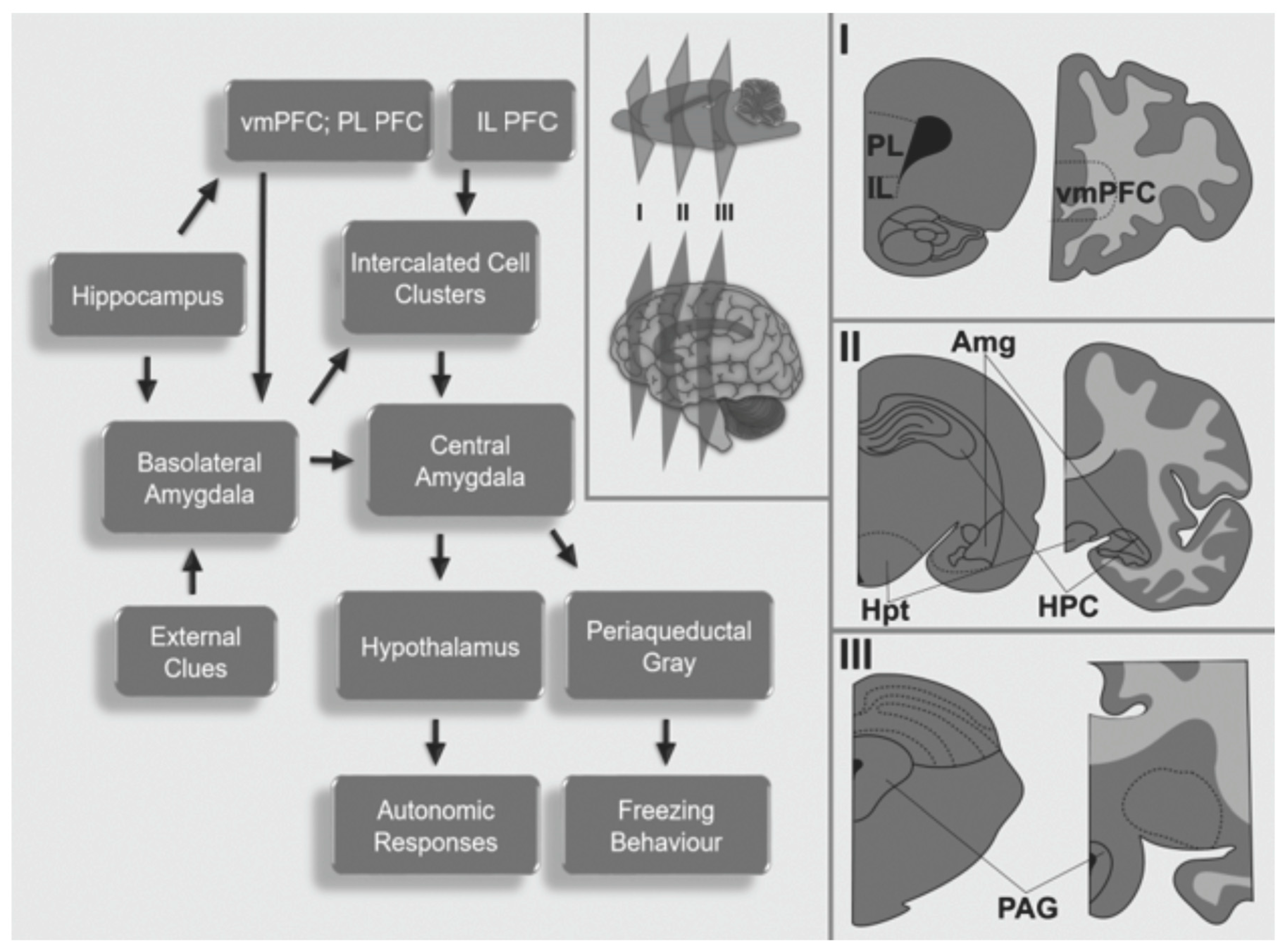
3. Neurocircuitry
4. Stimulation in Preclinical Models
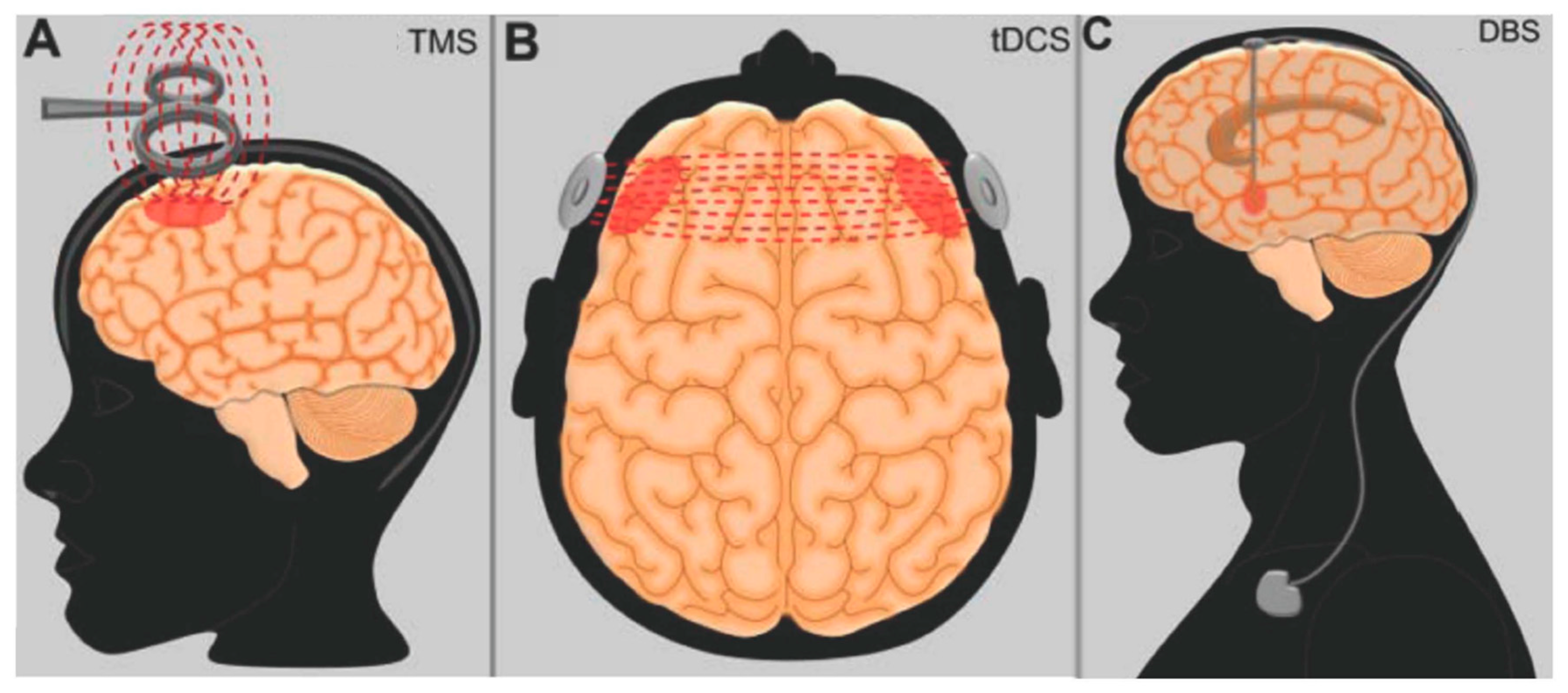
5. Neuromodulation Studies in Clinical Practice
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Breslau, N.; Davis, G.C.; Andreski, P.; Peterson, E. Traumatic events and posttraumatic stress disorder in an urban population of young adults. Arch. Gen. Psychiatry 1991, 48, 216–222. [Google Scholar] [CrossRef] [PubMed]
- Breslau, N.; Kessler, R.C.; Chilcoat, H.D.; Schultz, L.R.; Davis, G.C.; Andreski, P. Trauma and posttraumatic stress disorder in the community: The 1996 Detroit Area Survey of Trauma. Arch. Gen. Psychiatry 1998, 55, 626–632. [Google Scholar] [CrossRef]
- Fairbank, J.A.; Ebert, L.; Costello, E.J. Epidemiology of traumatic events and post-traumatic stress disorder. In Post Traumatic Stress Disorder: Diagnosis, Management and Treatment; Nutt, D., Davidson, J.R.T., Zohar, J., Eds.; Martin Dunitz Ltd.: London, UK, 2000. [Google Scholar]
- Kessler, R.C.; Sonnega, A.; Bromet, E.; Hughes, M.; Nelson, C.B. Posttraumatic stress disorder in the National Comorbidity Survey. Arch. Gen. Psychiatry 1995, 52, 1048–1060. [Google Scholar] [CrossRef]
- Trivedi, R.B.; Post, E.P.; Sun, H.; Pomerantz, A.; Saxon, A.J.; Piette, J.D.; Maynard, C.; Arnow, B.; Curtis, I.; Fihn, S.D.; et al. Prevalence, comorbidity, and prognosis of mental health among US veterans. Am. J. Public Health 2015, 105, 2564–2569. [Google Scholar] [CrossRef]
- Fulton, J.J.; Calhoun, P.S.; Wagner, H.R.; Schry, A.R.; Hair, L.P.; Feeling, N.; Elbogen, E.; Beckham, J.C. The prevalence of posttraumatic stress disorder in Operation Enduring Freedom/Operation Iraqi Freedom (OEF/OIF) Veterans: A meta-analysis. J. Anxiety Disord 2015, 31, 98–107. [Google Scholar] [CrossRef] [PubMed]
- Stewart, C.L.; Wrobel, T.A. Evaluation of the efficacy of pharmacotherapy and psychotherapy in treatment of combat-related post-traumatic stress disorder: A meta-analytic review of outcome studies. Mil. Med. 2009, 174, 460–469. [Google Scholar] [CrossRef] [PubMed]
- Powers, M.B.; Halpern, J.M.; Ferenschak, M.P.; Gillihan, S.J.; Foa, E.B. A meta-analytic review of prolonged exposure for posttraumatic stress disorder. Clin. Psychol Rev. 2010, 30, 635–641. [Google Scholar] [CrossRef]
- Bradley, R.; Greene, J.; Russ, E.; Dutra, L.; Westen, D. A multidimensional meta-analysis of psychotherapy for PTSD. Am. J. Psychiatry 2005, 162, 214–227. [Google Scholar] [CrossRef]
- Bisson, J.I.; Ehlers, A.; Matthews, R.; Pilling, S.; Richards, D.; Turner, S. Psychological treatments for chronic post-traumatic stress disorder. Systematic review and meta-analysis. Br. J. Psychiatry 2007, 190, 97–104. [Google Scholar] [CrossRef]
- Katzman, M.A.; Bleau, P.; Blier, P.; Chokka, P.; Kjernisted, K.; Van Ameringen, M.; Canadian Anxiety Guidelines Initiative Group on behalf of the Anxiety Disorders Association of Canada/Association Canadienne des troubles anxieux and McGill University; Antony, M.M.; Bouchard, S.; Brunet, A.; et al. Canadian clinical practice guidelines for the management of anxiety, posttraumatic stress and obsessive-compulsive disorders. BMC Psychiatry 2014, 14 (Suppl. 1), S1. [Google Scholar] [CrossRef]
- Davidson, J.R.; Connor, K.M.; Hertzberg, M.A.; Weisler, R.H.; Wilson, W.H.; Payne, V.M. Maintenance therapy with fluoxetine in posttraumatic stress disorder: A placebo-controlled discontinuation study. J. Clin. Psychopharmacol. 2005, 25, 166–169. [Google Scholar] [CrossRef] [PubMed]
- Murphy, D.; Smith, K.V. Treatment efficacy for veterans with posttraumatic stress disorder: Latent class trajectories of treatment response and their predictors. J. Trauma Stress 2018, 31, 753–763. [Google Scholar] [CrossRef] [PubMed]
- VanElzakker, M.B.; Dahlgren, M.K.; Davis, F.C.; Dubois, S.; Shin, L.M. From Pavlov to PTSD: The extinction of conditioned fear in rodents, humans, and anxiety disorders. Neurobiol. Learn. Mem. 2014, 113, 3–18. [Google Scholar] [CrossRef] [PubMed]
- Bonanno, G.A. Loss, trauma, and human resilience: Have we underestimated the human capacity to thrive after extremely aversive events? Am. Psychol. 2004, 59, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Van Minnen, A.; Wessel, I.; Dijkstra, T.; Roelofs, K. Changes in PTSD patients’ narratives during prolonged exposure therapy: A replication and extension. J. Trauma Stress 2002, 15, 255–258. [Google Scholar] [CrossRef]
- Foa, E.B. Psychosocial treatment of posttraumatic stress disorder. J. Clin. Psychiatry 2000, 61, 43–48; discussion 49–51. [Google Scholar] [PubMed]
- Eysenck, H.J. The conditioning model of neurosis. Behav. Brain Sci. 1979, 2, 155–199. [Google Scholar] [CrossRef]
- Blechert, J.; Michael, T.; Vriends, N.; Margraf, J.; Wilhelm, F.H. Fear conditioning in posttraumatic stress disorder: Evidence for delayed extinction of autonomic, experiential, and behavioural responses. Behav. Res. Ther. 2007, 45, 2019–2033. [Google Scholar] [CrossRef]
- Orr, S.P.; Metzger, L.J.; Lasko, N.B.; Macklin, M.L.; Peri, T.; Pitman, R.K. De novo conditioning in trauma-exposed individuals with and without posttraumatic stress disorder. J. Abnorm. Psychol. 2000, 109, 290–298. [Google Scholar] [CrossRef]
- Pitman, R.K.; Orr, S.P. Test of the conditioning model of neurosis: Differential aversive conditioning of angry and neutral facial expressions in anxiety disorder patients. J. Abnorm. Psychol. 1986, 95, 208–213. [Google Scholar] [CrossRef]
- Reznikov, R.; Binko, M.; Nobrega, J.N.; Hamani, C. Deep brain stimulation in animal models of fear, anxiety, and posttraumatic stress disorder. Neuropsychopharmacology 2016, 41, 2810–2817. [Google Scholar] [CrossRef] [PubMed]
- Reznikov, R.; Diwan, M.; Nobrega, J.N.; Hamani, C. Towards a better preclinical model of PTSD: Characterizing animals with weak extinction, maladaptive stress responses and low plasma corticosterone. J. Psychiatr. Res. 2015, 61, 158–165. [Google Scholar] [CrossRef] [PubMed]
- Blanchard, R.J.; Blanchard, D.C. Passive and active reactions to fear-eliciting stimuli. J. Comp. Physiol. Psychol. 1969, 68, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Lissek, S.; Powers, A.S.; McClure, E.B.; McClure, E.B.; Phelps, E.A.; Woldehawariat, G.; Grillon, C.; Pine, D.S. Classical fear conditioning in the anxiety disorders: A meta-analysis. Behav. Res. Ther. 2005, 43, 1391–1424. [Google Scholar] [CrossRef] [PubMed]
- Myers, K.M.; Davis, M. Mechanisms of fear extinction. Mol. Psychiatry 2007, 12, 120–150. [Google Scholar] [CrossRef] [PubMed]
- Rodgers, R.J.; Dalvi, A. Anxiety, defence and the elevated plus-maze. Neurosci. Biobehav. Rev. 1997, 21, 801–810. [Google Scholar] [CrossRef]
- Besnard, A.; Sahay, A. Adult hippocampal neurogenesis, fear generalization, and stress. Neuropsychopharmacology 2016, 41, 24–44. [Google Scholar] [CrossRef]
- Ahmadizadeh, M.J.; Rezaei, M. Unilateral right and bilateral dorsolateral prefrontal cortex transcranial magnetic stimulation in treatment post-traumatic stress disorder: A randomized controlled study. Brain Res. Bull. 2018, 140, 334–340. [Google Scholar] [CrossRef]
- LeDoux, J. The amygdala. Curr. Biol. 2007, 17, 868–874. [Google Scholar] [CrossRef]
- Garrido, M.I.; Barnes, G.R.; Sahani, M.; Dolan, R.J. Functional evidence for a dual route to amygdala. Curr. Biol. 2012, 22, 129–134. [Google Scholar] [CrossRef]
- Romanski, L.M.; Clugnet, M.C.; Bordi, F.; LeDoux, J.E. Somatosensory and auditory convergence in the lateral nucleus of the amygdala. Behav. Neurosci. 1993, 107, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Johnson, L.R.; McGuire, J.; Lazarus, R.; Palmer, A.A. Pavlovian fear memory circuits and phenotype models of PTSD. Neuropharmacology 2012, 62, 638–646. [Google Scholar] [CrossRef] [PubMed]
- LeDoux, J.E.; Iwata, J.; Cicchetti, P.; Reis, D.J. Different projections of the central amygdaloid nucleus mediate autonomic and behavioral correlates of conditioned fear. J. Neurosci. 1988, 8, 2517–2529. [Google Scholar] [CrossRef] [PubMed]
- Assareh, N.; Sarrami, M.; Carrive, P.; McNally, G.P. The organization of defensive behavior elicited by optogenetic excitation of rat lateral or ventrolateral periaqueductal gray. Behav. Neurosci. 2016, 130, 406–414. [Google Scholar] [CrossRef] [PubMed]
- Hsu, D.T.; Chen, F.L.; Takahashi, L.K.; Kalin, N.H. Rapid stress-induced elevations in corticotropin-releasing hormone mRNA in rat central amygdala nucleus and hypothalamic paraventricular nucleus: An in situ hybridization analysis. Brain Res. 1998, 788, 305–310. [Google Scholar] [CrossRef]
- Dekeyzer, S.; De Kock, I.; Nikoubashman, O.; Vanden Bossche, S.; Van Eetvelde, R.; De Groote, J.; Acou, M.; Wiesmann, M.; Deblaere, K.; Achten, E. “Unforgettable”—a pictorial essay on anatomy and pathology of the hippocampus. Insights Imaging 2017, 8, 199–212. [Google Scholar] [CrossRef] [PubMed]
- Giap, B.T.; Jong, C.N.; Ricker, J.H.; Cullen, N.K.; Zafonte, R.D. The hippocampus: Anatomy, pathophysiology, and regenerative capacity. J. Head Trauma Rehabil. 2000, 15, 875–894. [Google Scholar] [CrossRef]
- Fanselow, M.S.; Dong, H.W. Are the dorsal and ventral hippocampus functionally distinct structures? Neuron 2010, 65, 7–19. [Google Scholar] [CrossRef]
- Maren, S.; Aharonov, G.; Fanselow, M.S. Neurotoxic lesions of the dorsal hippocampus and Pavlovian fear conditioning in rats. Behav. Brain Res. 1997, 88, 261–274. [Google Scholar] [CrossRef]
- Wood, J.N.; Grafman, J. Human prefrontal cortex: Processing and representational perspectives. Nat. Rev. Neurosci. 2003, 4, 139–147. [Google Scholar] [CrossRef]
- Diekhof, E.K.; Geier, K.; Falkai, P.; Gruber, O. Fear is only as deep as the mind allows: A coordinate-based meta-analysis of neuroimaging studies on the regulation of negative affect. Neuroimage 2011, 58, 275–285. [Google Scholar] [CrossRef] [PubMed]
- Britton, J.C.; Phan, K.L.; Taylor, S.F.; Fig, L.M.; Liberzon, I. Corticolimbic blood flow in posttraumatic stress disorder during script-driven imagery. Biol. Psychiatry 2005, 57, 832–840. [Google Scholar] [CrossRef] [PubMed]
- Bremner, J.D.; Narayan, M.; Staib, L.H.; Southwick, S.M.; McGlashan, T.; Charney, D.S. Neural correlates of memories of childhood sexual abuse in women with and without posttraumatic stress disorder. Am. J. Psychiatry 1999, 156, 1787–1795. [Google Scholar] [PubMed]
- Shin, L.M.; Wright, C.I.; Cannistraro, P.A.; Wedig, M.M.; McMullin, K.; Martis, B.; Macklin, M.L.; Lasko, N.B.; Cavanagh, S.R.; Krangel, T.S.; et al. A functional magnetic resonance imaging study of amygdala and medial prefrontal cortex responses to overtly presented fearful faces in posttraumatic stress disorder. Arch. Gen. Psychiatry 2005, 62, 273–281. [Google Scholar] [CrossRef] [PubMed]
- Shin, L.M.; Orr, S.P.; Carson, M.A.; Rauch, S.L.; Macklin, M.L.; Lasko, N.B.; Peters, P.M.; Metzger, L.J.; Dougherty, D.D.; Cannistraro, P.A.; et al. Regional cerebral blood flow in the amygdala and medial prefrontal cortex during traumatic imagery in male and female Vietnam veterans with PTSD. Arch. Gen. Psychiatry 2004, 61, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Sripada, R.K.; King, A.P.; Garfinkel, S.N.; Wang, X.; Sripada, C.S.; Welsh, R.C.; Liberzon, I. Altered resting-state amygdala functional connectivity in men with posttraumatic stress disorder. J. Psychiatry Neurosci. 2012, 37, 241–249. [Google Scholar] [CrossRef] [PubMed]
- Carrion, V.G.; Haas, B.W.; Garrett, A.; Song, S.; Reiss, A.L. Reduced hippocampal activity in youth with posttraumatic stress symptoms: An FMRI study. J. Pediatr. Psychol. 2010, 35, 559–569. [Google Scholar] [CrossRef]
- Thomaes, K.; Dorrepaal, E.; Draijer, N.P.; de Ruiter, M.B.; Elzinga, B.M.; van Balkom, A.J.; Smoor, P.L.; Smit, J.; Veltman, D.J. Increased activation of the left hippocampus region in Complex PTSD during encoding and recognition of emotional words: A pilot study. Psychiatry Res. 2009, 171, 44–53. [Google Scholar] [CrossRef]
- Hamani, C.; Nobrega, J.N. Preclinical studies modeling deep brain stimulation for depression. Biol. Psychiatry 2012, 72, 916–923. [Google Scholar] [CrossRef]
- Uylings, H.B.; Groenewegen, H.J.; Kolb, B. Do rats have a prefrontal cortex? Behav. Brain Res. 2003, 146, 3–17. [Google Scholar] [CrossRef]
- Milad, M.R.; Quirk, G.J. Neurons in medial prefrontal cortex signal memory for fear extinction. Nature 2002, 420, 70–74. [Google Scholar] [CrossRef] [PubMed]
- Corcoran, K.A.; Quirk, G.J. Recalling safety: Cooperative functions of the ventromedial prefrontal cortex and the hippocampus in extinction. CNS Spectr. 2007, 12, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Amano, T.; Unal, C.T.; Pare, D. Synaptic correlates of fear extinction in the amygdala. Nat. Neurosci. 2010, 13, 489–494. [Google Scholar] [CrossRef] [PubMed]
- Royer, S.; Martina, M.; Pare, D. An inhibitory interface gates impulse traffic between the input and output stations of the amygdala. J. Neurosci. 1999, 19, 10575–10583. [Google Scholar] [CrossRef] [PubMed]
- Likhtik, E.; Popa, D.; Apergis-Schoute, J.; Fidacaro, G.A.; Pare, D. Amygdala intercalated neurons are required for expression of fear extinction. Nature 2008, 454, 642–645. [Google Scholar] [CrossRef]
- Ranck, J.B., Jr. Which elements are excited in electrical stimulation of mammalian central nervous system: A review. Brain Res. 1975, 98, 417–440. [Google Scholar] [CrossRef]
- Lozano, A.M.; Dostrovsky, J.; Chen, R.; Ashby, P. Deep brain stimulation for Parkinson’s disease: Disrupting the disruption. Lancet Neurol. 2002, 1, 225–231. [Google Scholar] [CrossRef]
- Hamani, C.; Temel, Y. Deep brain stimulation for psychiatric disease: Contributions and validity of animal models. Sci. Transl. Med. 2012, 4, 142–148. [Google Scholar] [CrossRef]
- Cleren, C.; Tallarida, I.; Guiniec, E.L.; Janin, F.; Nachon, O.; Canini, F.; Spennato, G.; Moreau, J.L.; Garcia, R. Low-frequency stimulation of the ventral hippocampus facilitates extinction of contextual fear. Neurobiol. Learn. Mem. 2013, 101, 39–45. [Google Scholar] [CrossRef]
- Deschaux, O.; Thevenet, A.; Spennato, G.; Arnaud, C.; Moreau, J.L.; Garcia, R. Low-frequency stimulation of the hippocampus following fear extinction impairs both restoration of rapid eye movement sleep and retrieval of extinction memory. Neuroscience 2010, 170, 92–98. [Google Scholar] [CrossRef]
- Garcia, R.; Spennato, G.; Nilsson-Todd, L.; Moreau, J.L.; Deschaux, O. Hippocampal low-frequency stimulation and chronic mild stress similarly disrupt fear extinction memory in rats. Neurobiol. Learn. Mem. 2008, 89, 560–566. [Google Scholar] [CrossRef] [PubMed]
- Farinelli, M.; Deschaux, O.; Hugues, S.; Thevenet, A.; Garcia, R. Hippocampal train stimulation modulates recall of fear extinction independently of prefrontal cortex synaptic plasticity and lesions. Learn. Mem. 2006, 13, 329–334. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Romaguera, J.; Do Monte, F.H.; Quirk, G.J. Deep brain stimulation of the ventral striatum enhances extinction of conditioned fear. Proc. Natl. Acad. Sci. USA 2012, 109, 8764–8769. [Google Scholar] [CrossRef] [PubMed]
- Sui, L.; Huang, S.; Peng, B.; Ren, J.; Tian, F.; Wang, Y. Deep brain stimulation of the amygdala alleviates fear conditioning-induced alterations in synaptic plasticity in the cortical-amygdala pathway and fear memory. J. Neural. Transm. 2014, 121, 773–782. [Google Scholar] [CrossRef] [PubMed]
- Saldivar-Gonzalez, J.A.; Posadas-Andrews, A.; Rodriguez, R.; Gómez, C.; Hernández-Manjarrez, M.E.; Ortiz-León, S.; Martínez-Pineda, A.; Gómez-Laguna, D.; Salgado, V.; Manjarrez, J.; et al. Effect of electrical stimulation of the baso-lateral amygdala nucleus on defensive burying shock probe test and elevated plus maze in rats. Life Sci. 2003, 72, 819–829. [Google Scholar] [CrossRef]
- Langevin, J.P.; De Salles, A.A.; Kosoyan, H.P.; Krahl, S.E. Deep brain stimulation of the amygdala alleviates post-traumatic stress disorder symptoms in a rat model. J. Psychiatr. Res. 2010, 44, 1241–1245. [Google Scholar] [CrossRef] [PubMed]
- Vidal-Gonzalez, I.; Vidal-Gonzalez, B.; Rauch, S.L.; Quirk, G.J. Microstimulation reveals opposing influences of prelimbic and infralimbic cortex on the expression of conditioned fear. Learn. Mem. 2006, 13, 728–733. [Google Scholar] [CrossRef] [PubMed]
- Maroun, M.; Kavushansky, A.; Holmes, A.; Wellman, C.; Motanis, H. Enhanced extinction of aversive memories by high-frequency stimulation of the rat infralimbic cortex. PLoS ONE 2012, 7, e35853. [Google Scholar] [CrossRef]
- Zheng, X.; Deschaux, O.; Lavigne, J.; Le Guisquet, A.M.; Belzung, C.; El-Hage, W. Prefrontal high-frequency stimulation prevents sub-conditioning procedure-provoked, but not acute stress-provoked, reemergence of extinguished fear. Neurobiol. Learn. Mem. 2013, 101, 33–38. [Google Scholar] [CrossRef]
- Deschaux, O.; Motanis, H.; Spennato, G.; Moreau, J.L.; Garcia, R. Re-emergence of extinguished auditory-cued conditioned fear following a sub-conditioning procedure: Effects of hippocampal and prefrontal tetanic stimulations. Neurobiol. Learn. Mem. 2011, 95, 510–518. [Google Scholar] [CrossRef]
- Reznikov, R.; Bambico, F.R.; Diwan, M.; Nachon, O.; Cleren, C.; Moreau, J.L.; Garcia, R. prefrontal cortex deep brain stimulation improves fear and anxiety-like behavior and reduces basolateral amygdala activity in a preclinical model of posttraumatic stress disorder. Neuropsychopharmacology 2018, 43, 1099–1106. [Google Scholar] [CrossRef] [PubMed]
- Van’t Wout, M.; Longo, S.M.; Reddy, M.K.; Philip, N.S.; Bowker, M.T.; Greenberg, B.D. Transcranial direct current stimulation may modulate extinction memory in posttraumatic stress disorder. Brain Behav. 2017, 7, e00681. [Google Scholar] [CrossRef] [PubMed]
- Zwanzger, P.; Steinberg, C.; Rehbein, M.A.; Bröckelmann, A.K.; Dobel, C.; Zavorotnyy, M.; Domschke, K.; Junghöfer, M. Inhibitory repetitive transcranial magnetic stimulation (rTMS) of the dorsolateral prefrontal cortex modulates early affective processing. Neuroimage 2014, 101, 193–203. [Google Scholar] [CrossRef] [PubMed]
- Baek, K.; Chae, J.H.; Jeong, J. The effect of repetitive transcranial magnetic stimulation on fear extinction in rats. Neuroscience 2012, 200, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.N.; Bai, Y.H.; Chen, Y.C.; Zhang, R.G.; Wang, H.H.; Zhang, Y.H.; Gan, J.L.; Peng, Z.W.; Tan, Q.R. Repetitive transcranial magnetic stimulation ameliorates anxiety-like behavior and impaired sensorimotor gating in a rat model of post-traumatic stress disorder. PLoS ONE 2015, 10, e0117189. [Google Scholar] [CrossRef] [PubMed]
- Legrand, M.; Troubat, R.; Brizard, B.; Le Guisquet, A.M.; Belzung, C.; El-Hage, W. Prefrontal cortex rTMS reverses behavioral impairments and differentially activates c-Fos in a mouse model of post-traumatic stress disorder. Brain Stimul. 2019, 12, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Feng, D.; Wang, J.; Zhang, H.; Peng, Z.; Cai, M.; Yang, J.; Zhang, R.; Wang, H.; Wu, S.; et al. rTMS ameliorates PTSD symptoms in rats by enhancing glutamate transmission and synaptic plasticity in the ACC via the PTEN/Akt signalling pathway. Mol. Neurobiol. 2018, 55, 3946–3958. [Google Scholar] [CrossRef]
- Sparta, D.R.; Jennings, J.H.; Ung, R.L.; Stuber, G.D. Optogenetic strategies to investigate neural circuitry engaged by stress. Behav. Brain Res. 2013, 255, 19–25. [Google Scholar] [CrossRef]
- Sparta, D.R.; Smithuis, J.; Stamatakis, A.M.; Jennings, J.H.; Kantak, P.A.; Ung, R.L. Stuber GDInhibition of projections from the basolateral amygdala to the entorhinal cortex disrupts the acquisition of contextual fear. Front. Behav. Neurosci. 2014, 8, 129. [Google Scholar] [CrossRef] [PubMed]
- Tovote, P.; Fadok, J.P.; Luthi, A. Neuronal circuits for fear and anxiety. Nat. Rev. Neurosci. 2015, 16, 317–331. [Google Scholar] [CrossRef]
- Johansen, J.P.; Wolff, S.B.; Luthi, A.; LeDoux, J.E. Controlling the elements: An optogenetic approach to understanding the neural circuits of fear. Biol. Psychiatry 2012, 71, 1053–1060. [Google Scholar] [CrossRef] [PubMed]
- Gafford, G.M.; Ressler, K.J. Mouse models of fear-related disorders: Cell-type-specific manipulations in amygdala. Neuroscience 2016, 321, 108–120. [Google Scholar] [CrossRef] [PubMed]
- Allsop, S.A.; Vander Weele, C.M.; Wichmann, R.; Tye, K.M. Optogenetic insights on the relationship between anxiety-related behaviors and social deficits. Front. Behav. Neurosci. 2014, 8, 241. [Google Scholar] [CrossRef] [PubMed]
- Kozel, F.A. Clinical repetitive transcranial magnetic stimulation for posttraumatic stress disorder, generalized anxiety disorder, and bipolar disorder. Psychiatr. Clin. North. Am. 2018, 41, 433–446. [Google Scholar] [CrossRef] [PubMed]
- Fryml, L.D.; Pelic, C.G.; Acierno, R.; Tuerk, P.; Yoder, M.; Borckardt, J.J.; Juneja, N.; Schmidt, M.; Beaver, K.L.; George, M.S. Exposure therapy and simultaneous repetitive transcranial magnetic stimulation: A controlled pilot trial for the treatment of posttraumatic stress disorder. J. ECT 2018. [Google Scholar] [CrossRef] [PubMed]
- Kozel, F.A.; Motes, M.A.; Didehbani, N.; DeLaRosa, B.; Bass, C.; Schraufnagel, C.D.; Jones, P.; Morgan, C.R.; Spence, J.S.; Kraut, M.A.; et al. Repetitive TMS to augment cognitive processing therapy in combat veterans of recent conflicts with PTSD: A randomized clinical trial. J. Affect. Disord. 2018, 229, 506–514. [Google Scholar] [CrossRef] [PubMed]
- Boggio, P.S.; Rocha, M.; Oliveira, M.O.; Fecteau, S.; Cohen, R.B.; Campanhã, C.; Ferreira-Santos, E.; Meleiro, A.; Corchs, F.; Zaghi, S.; et al. Noninvasive brain stimulation with high-frequency and low-intensity repetitive transcranial magnetic stimulation treatment for posttraumatic stress disorder. J. Clin. Psychiatry 2010, 71, 992–999. [Google Scholar] [CrossRef]
- Carpenter, L.L.; Conelea, C.; Tyrka, A.R.; Welch, E.S.; Greenberg, B.D.; Price, L.H.; Niedzwiecki, M.; Yip, A.G.; Barnes, J.; Philip, N.S. 5Hz Repetitive transcranial magnetic stimulation for posttraumatic stress disorder comorbid with major depressive disorder. J. Affect. Disord. 2018, 235, 414–420. [Google Scholar] [CrossRef]
- Fryml, L.D.; Sahlem, G.; Fox, J.; Short, E.B. The role of rTMS for patients with severe PTSD and depression. Evid Based Ment. Health 2018, 21, 39–40. [Google Scholar] [CrossRef]
- Nam, D.H.; Pae, C.U.; Chae, J.H. Low-frequency, repetitive transcranial magnetic stimulation for the treatment of patients with posttraumatic stress disorder: A double-blind, sham-controlled study. Clin. Psychopharmacol Neurosci. 2013, 11, 96–102. [Google Scholar] [CrossRef]
- Oznur, T.; Akarsu, S.; Celik, C.; Bolu, A.; Ozdemir, B.; Akcay, B.D.; Ozselek, S.; Bozkurt, A.; Ozmenler, K.N. Is transcranial magnetic stimulation effective in treatment-resistant combat related posttraumatic stress disorder? Neurosciences 2014, 19, 29–32. [Google Scholar] [PubMed]
- Philip, N.S.; Barredo, J.; van’t Wout-Frank, M.; Tyrka, A.R.; Price, L.H.; Carpenter, L.L. Network mechanisms of clinical response to transcranial magnetic stimulation in posttraumatic stress disorder and major depressive disorder. Biol. Psychiatry 2018, 83, 263–272. [Google Scholar] [CrossRef] [PubMed]
- Philip, N.S.; Ridout, S.J.; Albright, S.E.; Sanchez, G.; Carpenter, L.L. 5-Hz Transcranial Magnetic Stimulation for Comorbid Posttraumatic Stress Disorder and Major Depression. J. Trauma Stress 2016, 29, 93–96. [Google Scholar] [CrossRef] [PubMed]
- Yesavage, J.A.; Fairchild, J.K.; Mi, Z.; Biswas, K.; Davis-Karim, A.; Phibbs, C.S.; Forman, S.D.; Thase, M.; Williams, L.M.; Etkin, A.; et al. Effect of Repetitive Transcranial Magnetic Stimulation on Treatment-Resistant Major Depression in US Veterans: A Randomized Clinical Trial. JAMA Psychiatry 2018, 75, 884–893. [Google Scholar] [CrossRef] [PubMed]
- Saunders, N.; Downham, R.; Turman, B.; Kropotov, J.; Clark, R.; Yumash, R.; Szatmary, A. Working memory training with tDCS improves behavioral and neurophysiological symptoms in pilot group with post-traumatic stress disorder (PTSD) and with poor working memory. Neurocase 2015, 21, 271–278. [Google Scholar] [CrossRef] [PubMed]
- van’t Wout-Frank, M.; Shea, M.T.; Larson, V.C.; Greenberg, B.D.; Philip, N.S. Combined transcranial direct current stimulation with virtual reality exposure for posttraumatic stress disorder: Feasibility and pilot results. Brain Stimul. 2018. [Google Scholar] [CrossRef]
- Hamani, C.; Lozano, A.M. Hardware-related complications of deep brain stimulation: A review of the published literature. Stereotact. Funct. Neurosurg. 2006, 84, 248–251. [Google Scholar] [CrossRef]
- Awan, N.R.; Lozano, A.; Hamani, C. Deep brain stimulation: Current and future perspectives. Neurosurg. Focus 2009, 27, E2. [Google Scholar] [CrossRef]
- Langevin, J.P.; Koek, R.J.; Schwartz, H.N.; Chen, J.W.Y.; Sultzer, D.L.; Mandelkern, M.A.; Kulick, A.D.; Krahl, S.E. Deep brain stimulation of the basolateral amygdala for treatment-refractory posttraumatic stress disorder. Biol. Psychiatry 2016, 79, e82–e84. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gouveia, F.V.; Gidyk, D.C.; Giacobbe, P.; Ng, E.; Meng, Y.; Davidson, B.; Abrahao, A.; Lipsman, N.; Hamani, C. Neuromodulation Strategies in Post-Traumatic Stress Disorder: From Preclinical Models to Clinical Applications. Brain Sci. 2019, 9, 45. https://doi.org/10.3390/brainsci9020045
Gouveia FV, Gidyk DC, Giacobbe P, Ng E, Meng Y, Davidson B, Abrahao A, Lipsman N, Hamani C. Neuromodulation Strategies in Post-Traumatic Stress Disorder: From Preclinical Models to Clinical Applications. Brain Sciences. 2019; 9(2):45. https://doi.org/10.3390/brainsci9020045
Chicago/Turabian StyleGouveia, Flavia V., Darryl C. Gidyk, Peter Giacobbe, Enoch Ng, Ying Meng, Benjamin Davidson, Agessandro Abrahao, Nir Lipsman, and Clement Hamani. 2019. "Neuromodulation Strategies in Post-Traumatic Stress Disorder: From Preclinical Models to Clinical Applications" Brain Sciences 9, no. 2: 45. https://doi.org/10.3390/brainsci9020045
APA StyleGouveia, F. V., Gidyk, D. C., Giacobbe, P., Ng, E., Meng, Y., Davidson, B., Abrahao, A., Lipsman, N., & Hamani, C. (2019). Neuromodulation Strategies in Post-Traumatic Stress Disorder: From Preclinical Models to Clinical Applications. Brain Sciences, 9(2), 45. https://doi.org/10.3390/brainsci9020045




