The Emerging Role of Tractography in Deep Brain Stimulation: Basic Principles and Current Applications
Abstract
1. Introduction
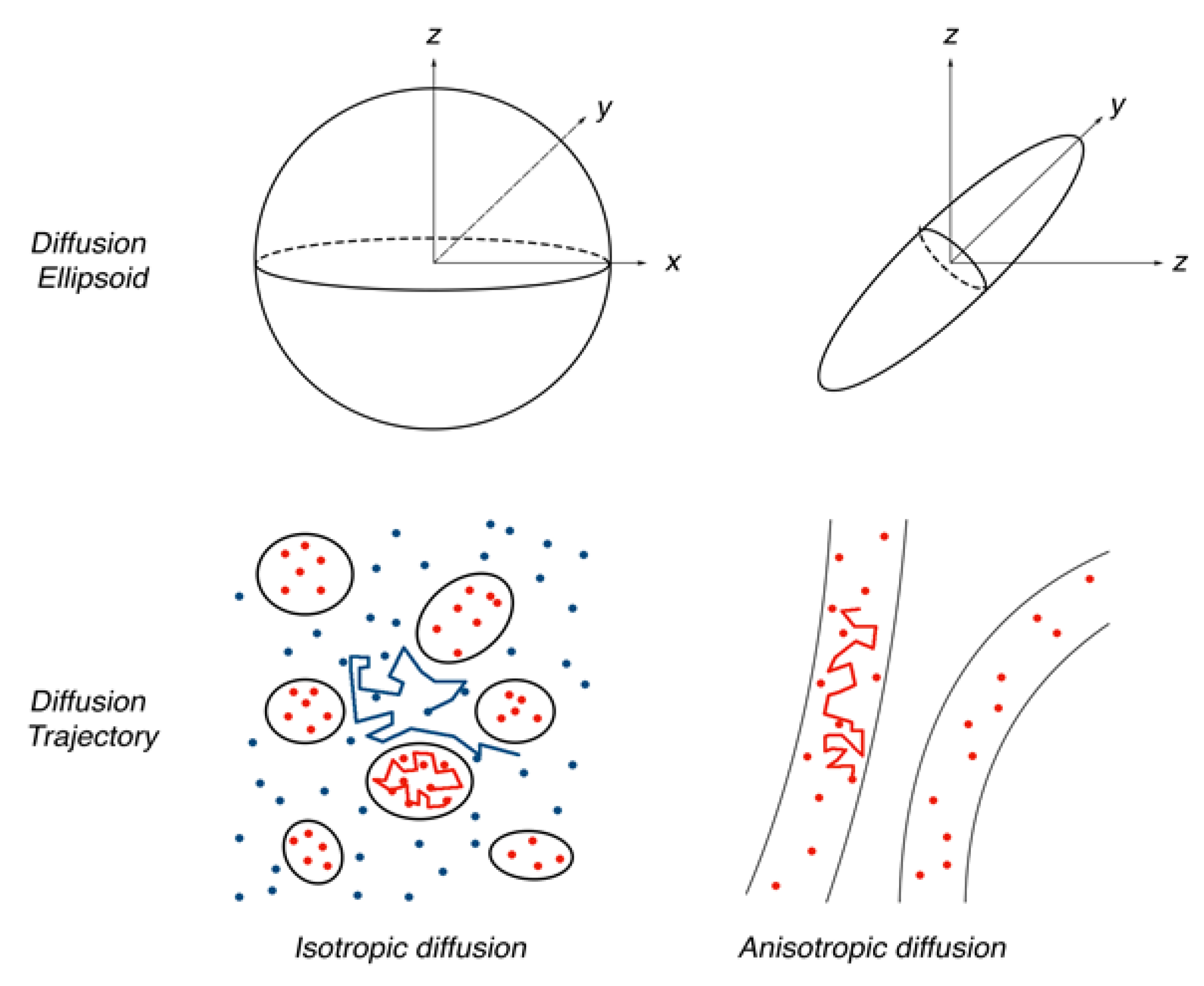
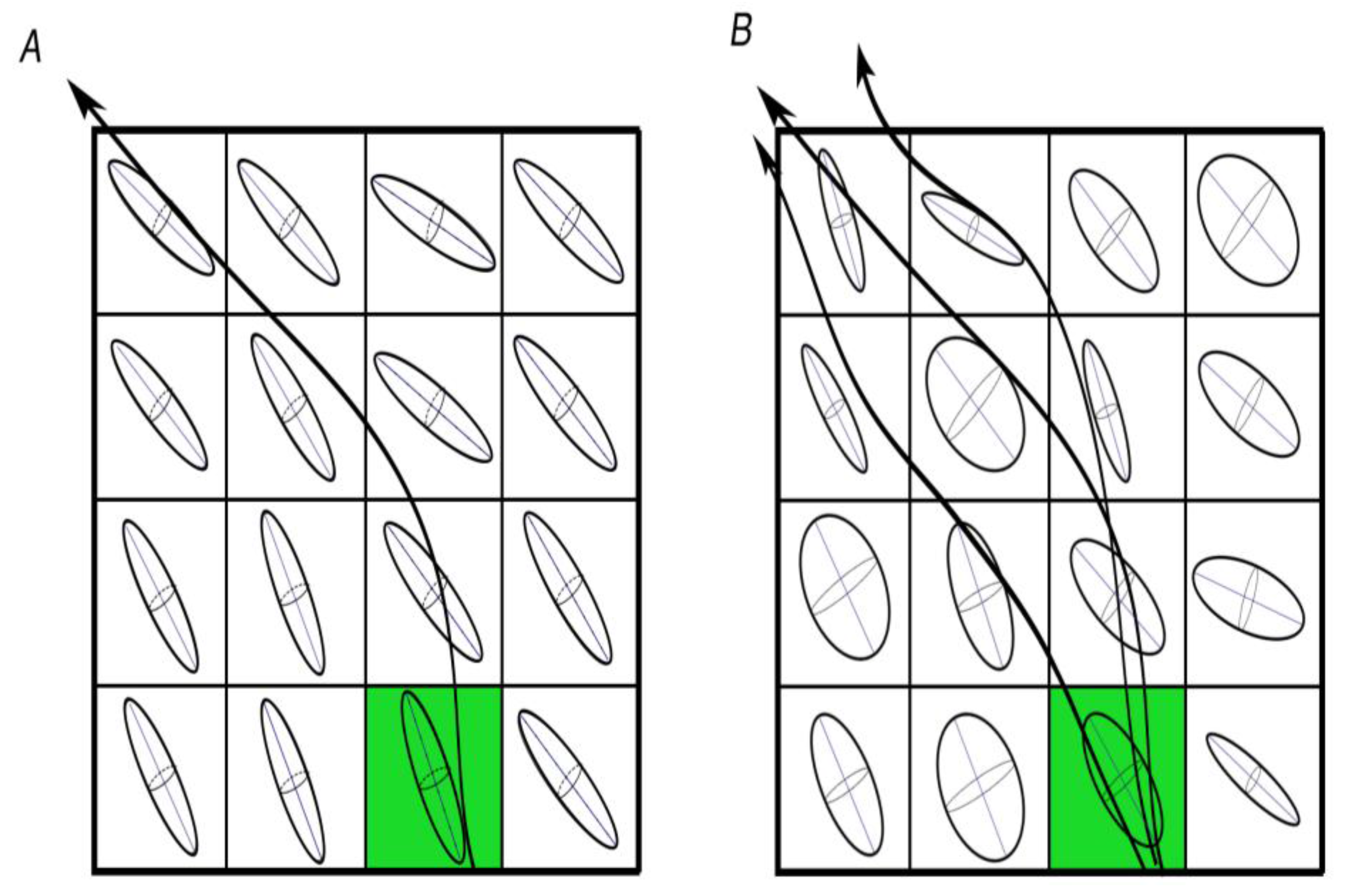
2. Tractography
3. Tractography and DBS
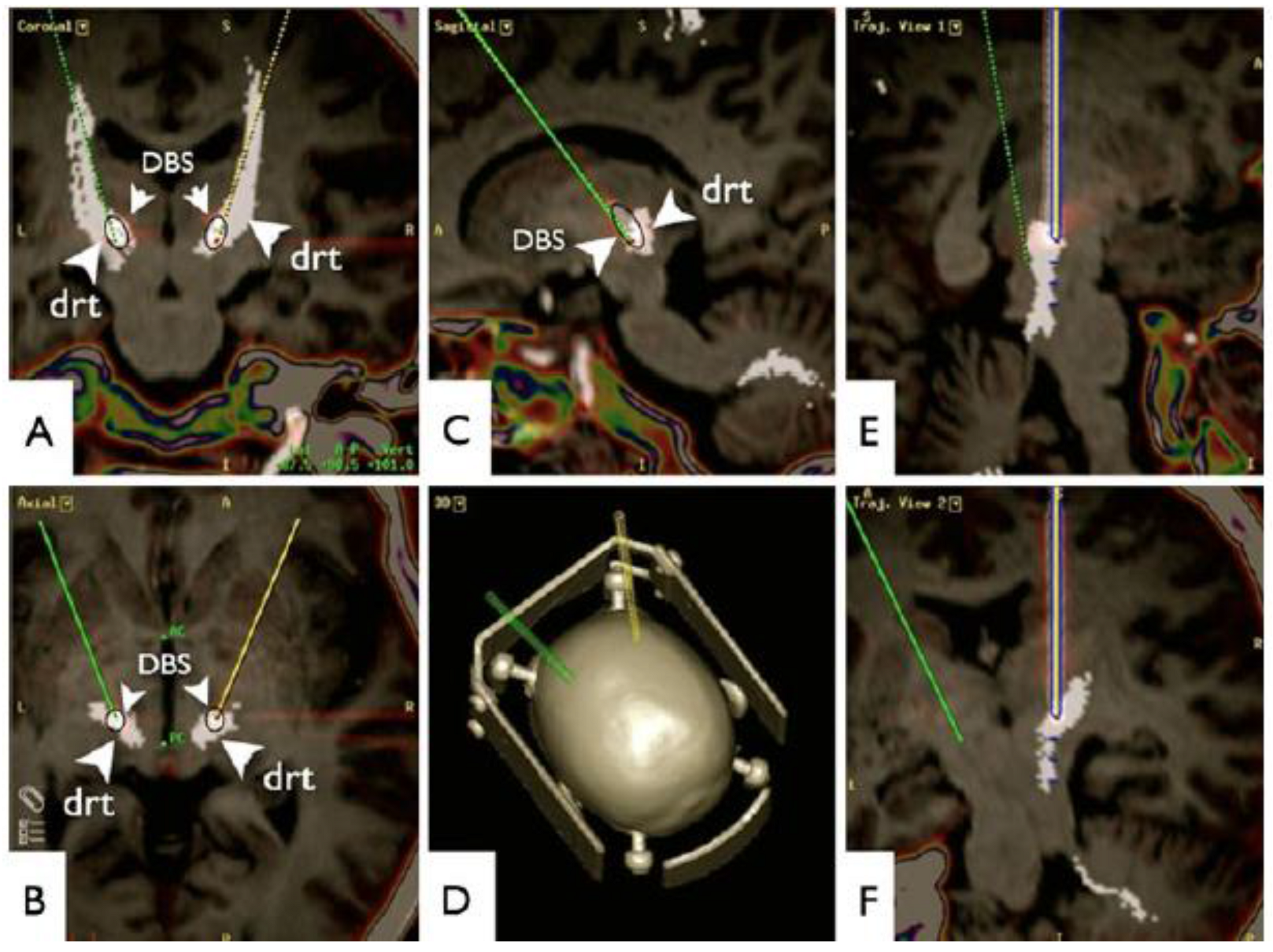
3.1. Tremor and Movement Disorders
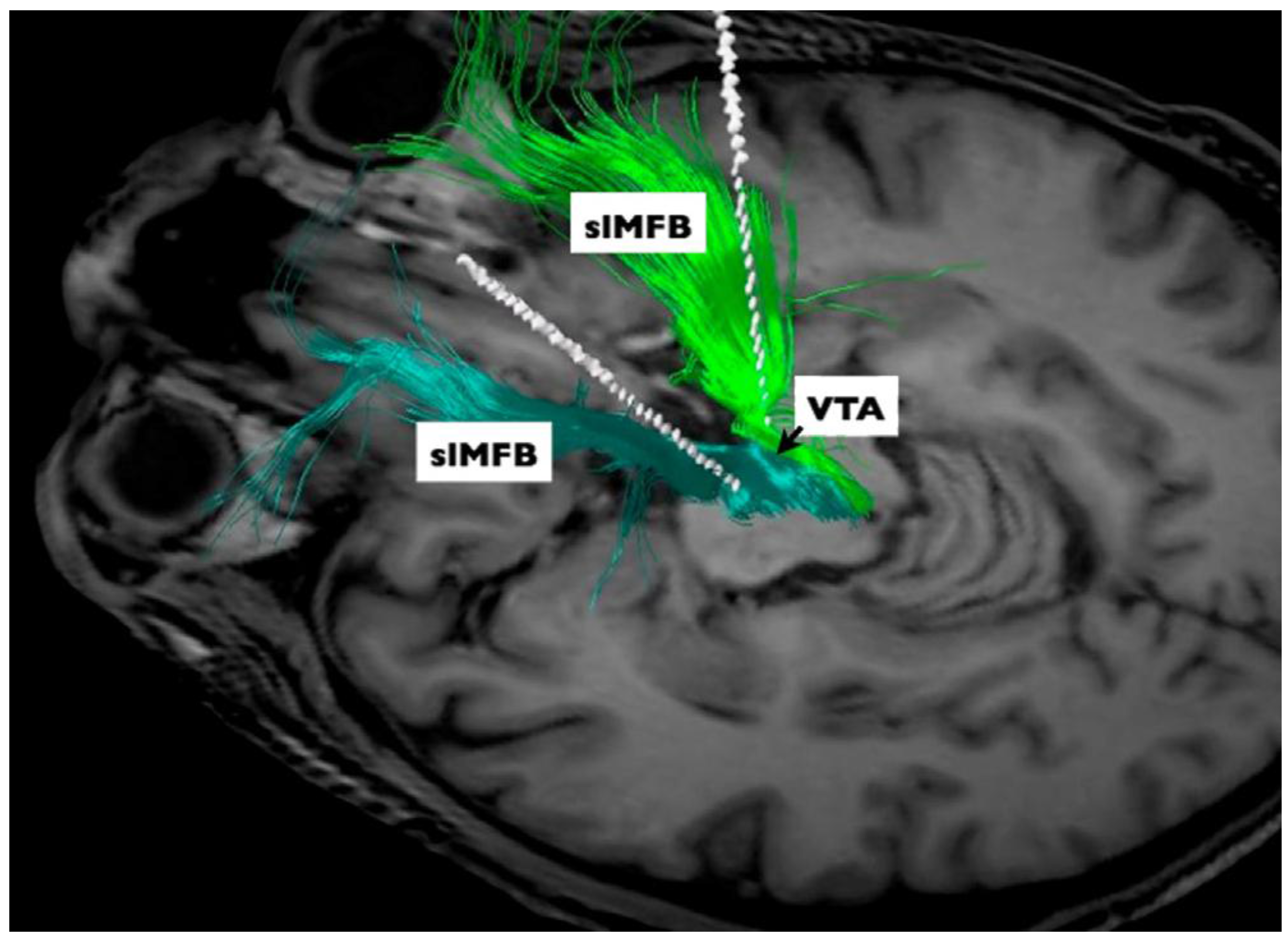
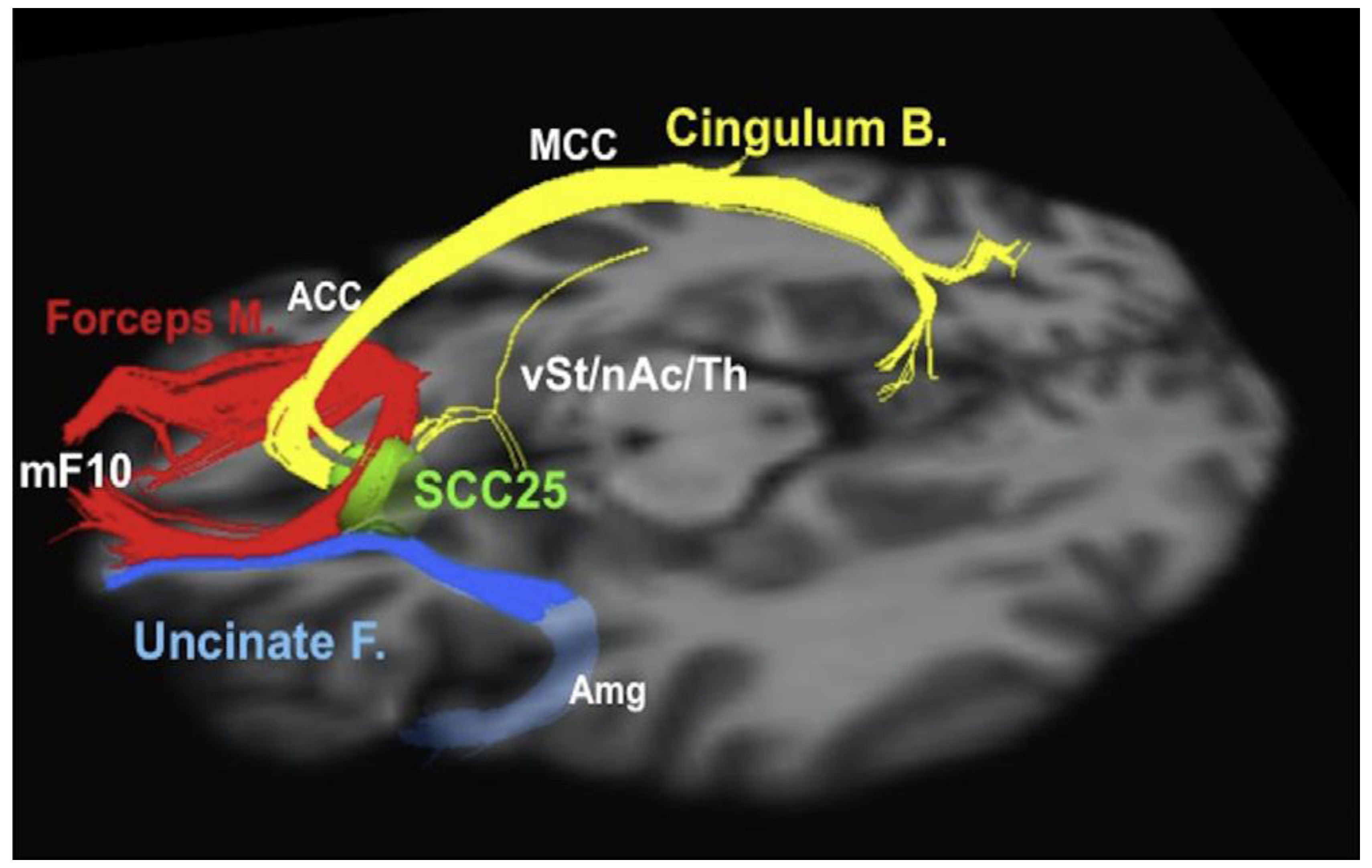
3.2. Treatment Resistant Depression
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hamani, C.; Temel, Y. Deep brain stimulation for psychiatric disease: Contributions and validity of animal models. Sci. Transl. Med. 2012, 4, 142rv8. [Google Scholar] [CrossRef] [PubMed]
- Reznikov, R.; Binko, M.; Nobrega, J.N.; Hamani, C. Deep Brain Stimulation in Animal Models of Fear, Anxiety, and Posttraumatic Stress Disorder. Neuropsychopharmacology 2016, 41, 2810–2817. [Google Scholar] [CrossRef] [PubMed]
- Basser, P.J.; Mattiello, J.; LeBihan, D. MR diffusion tensor spectroscopy and imaging. Biophys. J. 1994, 66, 259–267. [Google Scholar] [CrossRef]
- Berg, H.C. Random Walks in Biology; Princeton University Press: Princeton, NJ, USA, 1993. [Google Scholar]
- Mulkern, R.V.; Gudbjartsson, H.; Westin, C.F.; Zengingonul, H.P.; Gartner, W.; Guttmann, C.R.; Robertson, R.L.; Kyriakos, W.; Schwartz, R.; Holtzman, D.; et al. Multi-component apparent diffusion coefficients in human brain. NMR Biomed. 1999, 12, 51–62. [Google Scholar] [CrossRef]
- Tanner, J.E. Transient diffusion in a system partitioned by permeable barriers. Application to NMR measurements with a pulsed field gradient. J. Chem. Phys. 1978, 69, 1748–1754. [Google Scholar] [CrossRef]
- Aboitiz, F.; Scheibel, A.B.; Fisher, R.S.; Zaidel, E. Fiber composition of the human corpus callosum. Brain Res. 1992, 598, 143–153. [Google Scholar] [CrossRef]
- Descoteaux, M.; Deriche, R.; Knosche, T.R.; Anwander, A. Deterministic and probabilistic tractography based on complex fibre orientation distributions. IEEE Trans. Med. Imaging 2009, 28, 269–286. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.K. Precision and accuracy in diffusion tensor magnetic resonance imaging. Top. Magn. Reson. Imaging 2010, 21, 87–99. [Google Scholar] [CrossRef] [PubMed]
- Behrens, T.E.; Berg, H.J.; Jbabdi, S.; Rushworth, M.F.; Woolrich, M.W. Probabilistic diffusion tractography with multiple fibre orientations: What can we gain? Neuroimage 2007, 34, 144–155. [Google Scholar] [CrossRef] [PubMed]
- Tuch, D.S.; Reese, T.G.; Wiegell, M.R.; Makris, N.; Belliveau, J.W.; Wedeen, V.J. High angular resolution diffusion imaging reveals intravoxel white matter fiber heterogeneity. Magn. Reson. Med. 2002, 48, 577–582. [Google Scholar] [CrossRef] [PubMed]
- Tuch, D.S. Q-ball imaging. Magn. Reson. Med. 2004, 52, 1358–1372. [Google Scholar] [CrossRef] [PubMed]
- Alexander, A.L.; Wu, Y.C.; Venkat, P.C. Hybrid diffusion imaging (HYDI). Conf. Proc. IEEE Eng. Med. Biol. Soc. 2006, 1, 2245–2248. [Google Scholar] [PubMed]
- Wedeen, V.J.; Wang, R.P.; Schmahmann, J.D.; Benner, T.; Tseng, W.Y.; Dai, G.; Pandya, D.N.; Hagmann, P.; D’Arceuil, H.; de Crespigny, A.J. Diffusion spectrum magnetic resonance imaging (DSI) tractography of crossing fibers. Neuroimage 2008, 41, 1267–1277. [Google Scholar] [CrossRef] [PubMed]
- Kaden, E.; Knosche, T.R.; Anwander, A. Parametric spherical deconvolution: Inferring anatomical connectivity using diffusion MR imaging. Neuroimage 2007, 37, 474–488. [Google Scholar] [CrossRef] [PubMed]
- Assaf, Y.; Basser, P.J. Composite hindered and restricted model of diffusion (CHARMED) MR imaging of the human brain. Neuroimage 2005, 27, 48–58. [Google Scholar] [CrossRef] [PubMed]
- Jansons, K.M.; Alexander, D.C. Persistent Angular Structure: New insights from diffusion MRI data. Dummy version. Inf. Process. Med. Imaging 2003, 18, 672–683. [Google Scholar] [PubMed]
- Ozarslan, E.; Shepherd, T.M.; Vemuri, B.C.; Blackband, S.J.; Mareci, T.H. Resolution of complex tissue microarchitecture using the diffusion orientation transform (DOT). Neuroimage 2006, 31, 1086–1103. [Google Scholar] [CrossRef] [PubMed]
- Essayed, W.I.; Zhang, F.; Unadkat, P.; Cosgrove, G.R.; Golby, A.J.; O’Donnell, L.J. White matter tractography for neurosurgical planning: A topography-based review of the current state of the art. Neuroimage Clin. 2017, 15, 659–672. [Google Scholar] [CrossRef] [PubMed]
- Mollink, J.; Kleinnijenhuis, M.; Cappellen van Walsum, A.V.; Sotiropoulosa, S.N.; Cottaar, M.; Mirfin, C.; Heinrich, M.P.; Jenkinson, M.; Pallebage-Gamarallage, M.; Ansorge, O.; et al. Evaluating fibre orientation dispersion in white matter: Comparison of diffusion MRI, histology and polarized light imaging. Neuroimage 2017, 157, 561–574. [Google Scholar] [CrossRef] [PubMed]
- Seehaus, A.; Roebroeck, A.; Bastiani, M.; Fonseca, L.; Bratzke, H.; Lori, N.; Vilanova, A.; Goebel, R.; Galuske, R. Histological validation of high-resolution DTI in human post mortem tissue. Front. Neuroanat. 2015, 9, 98. [Google Scholar] [CrossRef] [PubMed]
- Gargiulo, P.; Arnadottir, I.; Gislason, M.; Edmunds, K.; Olafsson, I. New Directions in 3D Medical Modeling: 3D-Printing Anatomy and Functions in Neurosurgical Planning. J. Healthc. Eng. 2017, 2017, 1439643. [Google Scholar] [CrossRef] [PubMed]
- Mangin, J.F.; Fillard, P.; Cointepas, Y.; Le Bihan, D.; Frouin, V.; Poupon, C. Toward global tractography. Neuroimage 2013, 80, 290–296. [Google Scholar] [CrossRef] [PubMed]
- Christiaens, D.; Reisert, M.; Dhollander, T.; Sunaert, S.; Suetens, P.; Maes, F. Global tractography of multi-shell diffusion-weighted imaging data using a multi-tissue model. Neuroimage 2015, 123, 89–101. [Google Scholar] [CrossRef] [PubMed]
- Maier-Hein, K.H.; Neher, P.F.; Houde, J.C.; Côté, M.A.; Garyfallidis, E.; Zhong, J.; Chamberland, M.; Yeh, F.C.; Lin, Y.C.; Ji, Q.; et al. Reddick WE. The challenge of mapping the human connectome based on diffusion tractography. Nat. Commun. 2017, 8, 1349. [Google Scholar] [CrossRef] [PubMed]
- Pujol, S.; Wells, W.; Pierpaoli, C.; Brun, C.; Gee, J.; Cheng, G.; Vemuri, B.; Commowick, O.; Prima, S.; Stamm, A.; et al. The DTI Challenge: Toward Standardized Evaluation of Diffusion Tensor Imaging Tractography for Neurosurgery. J. Neuroimaging 2015, 25, 875–882. [Google Scholar] [CrossRef] [PubMed]
- Koller, W.C.; Lyons, K.E.; Wilkinson, S.B.; Troster, A.I.; Pahwa, R. Long-term safety and efficacy of unilateral deep brain stimulation of the thalamus in essential tremor. Mov. Disord. 2001, 16, 464–468. [Google Scholar] [CrossRef] [PubMed]
- Blomstedt, P.; Hariz, G.M.; Hariz, M.I.; Koskinen, L.O. Thalamic deep brain stimulation in the treatment of essential tremor: A long-term follow-up. Br. J. Neurosurg. 2007, 21, 504–509. [Google Scholar] [CrossRef] [PubMed]
- Blomstedt, P.; Sandvik, U.; Linder, J.; Fredricks, A.; Forsgren, L.; Hariz, M.I. Deep brain stimulation of the subthalamic nucleus versus the zona incerta in the treatment of essential tremor. Acta Neurochir. (Wien) 2011, 153, 2329–2335. [Google Scholar] [CrossRef] [PubMed]
- Sandvik, U.; Koskinen, L.O.; Lundquist, A.; Blomstedt, P. Thalamic and subthalamic deep brain stimulation for essential tremor: Where is the optimal target? Neurosurgery 2012, 70, 840–845; discussion 845–846. [Google Scholar] [CrossRef] [PubMed]
- Murata, J.; Kitagawa, M.; Uesugi, H.; Saito, H.; Iwasaki, Y.; Kikuchi, S.; Tashiro, K.; Sawamura, Y. Electrical stimulation of the posterior subthalamic area for the treatment of intractable proximal tremor. J. Neurosurg. 2003, 99, 708–715. [Google Scholar] [CrossRef] [PubMed]
- Fytagoridis, A.; Astrom, M.; Samuelsson, J.; Blomstedt, P. Deep Brain Stimulation of the Caudal Zona Incerta: Tremor Control in Relation to the Location of Stimulation Fields. Stereotact. Funct. Neurosurg. 2016, 94, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Coenen, V.A.; Allert, N.; Madler, B. A role of diffusion tensor imaging fiber tracking in deep brain stimulation surgery: DBS of the dentato-rubro-thalamic tract (DRT) for the treatment of therapy-refractory tremor. Acta Neurochir. (Wien) 2011, 153, 1579–1585; discussion 1585. [Google Scholar] [CrossRef] [PubMed]
- King, N.K.K.; Krishna, V.; Sammartino, F.; Bari, A.; Reddy, G.D.; Hodaie, M.; Kalia, S.K.; Fasano, A.; Munhoz, R.P.; Lozano, A.M.; et al. Anatomic Targeting of the Optimal Location for Thalamic Deep Brain Stimulation in Patients with Essential Tremor. World Neurosurg. 2017, 107, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Coenen, V.A.; Allert, N.; Paus, S.; Kronenburger, M.; Urbach, H.; Madler, B. Modulation of the cerebello-thalamo-cortical network in thalamic deep brain stimulation for tremor: A diffusion tensor imaging study. Neurosurgery 2014, 75, 657–669; discussion 669–670. [Google Scholar] [CrossRef] [PubMed]
- O’Halloran, R.L.; Chartrain, A.G.; Rasouli, J.J.; Ramdhani, R.A.; Kopell, B.H. Case Study of Image-Guided Deep Brain Stimulation: Magnetic Resonance Imaging-Based White Matter Tractography Shows Differences in Responders and Nonresponders. World Neurosurg. 2016, 96, 613.e9–613.e16. [Google Scholar] [CrossRef] [PubMed]
- Sajonz, B.E.; Amtage, F.; Reinacher, P.C.; Jenkner, C.; Piroth, T.; Kätzler, J.; Urbach, H.; Coenen, V.A. Deep Brain Stimulation for Tremor Tractographic Versus Traditional (DISTINCT): Study Protocol of a Randomized Controlled Feasibility Trial. JMIR Res. Protoc. 2016, 5, e244. [Google Scholar] [CrossRef] [PubMed]
- Hamani, C.; Florence, G.; Heinsen, H.; Plantinga, B.R.; Temel, Y.; Uludag, K.; Alho, E.; Teixeira, M.J.; Amaro, E.; Fonoff, E.T. Subthalamic Nucleus Deep Brain Stimulation: Basic Concepts and Novel Perspectives. eNeuro 2017, 4. [Google Scholar] [CrossRef] [PubMed]
- Petersen, M.V.; Lund, T.E.; Sunde, N.; Frandsen, J.; Rosendal, F.; Juul, N.; Østergaard, K. Probabilistic versus deterministic tractography for delineation of the cortico-subthalamic hyperdirect pathway in patients with Parkinson disease selected for deep brain stimulation. J. Neurosurg. 2017, 126, 1657–1668. [Google Scholar] [CrossRef] [PubMed]
- Akram, H.; Sotiropoulos, S.N.; Jbabdi, S.; Georgiev, D.; Mahlknecht, P.; Hyam, J.; Foltynie, T.; Limousin, P.; De Vita, E.; Jahanshahi, M.; et al. Subthalamic deep brain stimulation sweet spots and hyperdirect cortical connectivity in Parkinson’s disease. Neuroimage 2017, 158, 332–345. [Google Scholar] [CrossRef] [PubMed]
- Horn, A.; Reich, M.; Vorwerk, J.; Li, N.; Wenzel, G.; Fang, Q.; Schmitz-Hübsch, T.; Nickl, R.; Kupsch, A.; Volkmann, J.; et al. Connectivity Predicts deep brain stimulation outcome in Parkinson disease. Ann. Neurol. 2017, 82, 67–78. [Google Scholar] [CrossRef] [PubMed]
- Aravamuthan, B.R.; Stein, J.F.; Aziz, T.Z. The anatomy and localization of the pedunculopontine nucleus determined using probabilistic diffusion tractography [corrected]. Br. J. Neurosurg. 2008, 22 (Suppl. 1), S25–S32. [Google Scholar] [CrossRef] [PubMed]
- Alho, A.; Hamani, C.; Alho, E.J.L.; da Silva, R.E.; Santos, G.A.B.; Neves, R.C.; Carreira, L.L.; Araújo, C.M.M.; Magalhães, G.; Coelho, D.B.; et al. Magnetic resonance diffusion tensor imaging for the pedunculopontine nucleus: Proof of concept and histological correlation. Brain Struct. Funct. 2017, 222, 2547–2558. [Google Scholar] [CrossRef] [PubMed]
- Holtzheimer, P.E.; Kelley, M.E.; Gross, R.E.; Filkowski, M.M.; Garlow, S.J.; Barrocas, A.; Wint, D.; Craighead, M.C.; Kozarsky, J.; Chismar, R.; et al. Subcallosal cingulate deep brain stimulation for treatment-resistant unipolar and bipolar depression. Arch. Gen. Psychiatry 2012, 69, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Mayberg, H.S.; Lozano, A.M.; Voon, V.; McNeely, H.E.; Seminowicz, D.; Hamani, C.; Schwalb, J.M.; Kennedy, S.H. Deep brain stimulation for treatment-resistant depression. Neuron 2005, 45, 651–660. [Google Scholar] [CrossRef] [PubMed]
- Malone, D.A., Jr.; Dougherty, D.D.; Rezai, A.R.; Carpenter, L.L.; Friehs, G.M.; Eskandar, E.N.; Rauch, S.L.; Rasmussen, S.A.; Machado, A.G.; Kubu, C.S.; et al. Deep Brain Stimulation of the Ventral Capsule/Ventral Striatum for Treatment-Resistant Depression. Biol. Psychiatry 2009, 65, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Schlaepfer, T.E.; Cohen, M.X.; Frick, C.; Kosel, M.; Brodesser, D.; Axmacher, N.; Joe, A.Y.; Kreft, M.; Lenartz, D.; Sturm, V. Deep brain stimulation to reward circuitry alleviates anhedonia in refractory major depression. Neuropsychopharmacology 2008, 33, 368–377. [Google Scholar] [CrossRef] [PubMed]
- Dougherty, D.D.; Rezai, A.R.; Carpenter, L.L.; Howland, R.H.; Bhati, M.T.; O’Reardon, J.P.; Eskandar, E.N.; Baltuch, G.H.; Machado, A.D.; Kondziolka, D.; et al. A Randomized Sham-Controlled Trial of Deep Brain Stimulation of the Ventral Capsule/Ventral Striatum for Chronic Treatment-Resistant Depression. Biol. Psychiatry 2015, 78, 240–248. [Google Scholar] [CrossRef] [PubMed]
- Holtzheimer, P.E.; Husain, M.M.; Lisanby, S.H.; Taylor, S.F.; Whitworth, L.A.; McClintock, S.; Slavin, K.V.; Berman, J.; McKhann, G.M.; Patil, P.G.; et al. Subcallosal cingulate deep brain stimulation for treatment-resistant depression: A multisite, randomised, sham-controlled trial. Lancet Psychiatry 2017, 4, 839–849. [Google Scholar] [CrossRef]
- Schlaepfer, T.E.; Bewernick, B.H.; Kayser, S.; Madler, B.; Coenen, V.A. Rapid effects of deep brain stimulation for treatment-resistant major depression. Biol. Psychiatry 2013, 73, 1204–1212. [Google Scholar] [CrossRef] [PubMed]
- Coenen, V.A.; Honey, C.R.; Hurwitz, T.; Rahman, A.A.; McMaster, J.; Bürgel, U.; Mädler, B. Medial forebrain bundle stimulation as a pathophysiological mechanism for hypomania in subthalamic nucleus deep brain stimulation for Parkinson’s disease. Neurosurgery 2009, 64, 1106–1114; discussion 1114–1105. [Google Scholar] [CrossRef] [PubMed]
- Castrioto, A.; Lhommee, E.; Moro, E.; Krack, P. Mood and behavioural effects of subthalamic stimulation in Parkinson’s disease. Lancet Neurol. 2014, 13, 287–305. [Google Scholar] [CrossRef]
- Deuschl, G.; Paschen, S.; Witt, K. Clinical outcome of deep brain stimulation for Parkinson’s disease. Handb. Clin. Neurol. 2013, 116, 107–128. [Google Scholar] [PubMed]
- Coenen, V.A.; Panksepp, J.; Hurwitz, T.A.; Urbach, H.; Madler, B. Human medial forebrain bundle (MFB) and anterior thalamic radiation (ATR): Imaging of two major subcortical pathways and the dynamic balance of opposite affects in understanding depression. J. Neuropsychiatry Clin. Neurosci. 2012, 24, 223–236. [Google Scholar] [CrossRef] [PubMed]
- Coenen, V.A.; Schlaepfer, T.E.; Maedler, B.; Panksepp, J. Cross-species affective functions of the medial forebrain bundle-implications for the treatment of affective pain and depression in humans. Neurosci. Biobehav. Rev. 2011, 35, 1971–1981. [Google Scholar] [CrossRef] [PubMed]
- Bewernick, B.H.; Kayser, S.; Gippert, S.M.; Switala, C.; Coenen, V.A.; Schlaepfer, T.E. Deep brain stimulation to the medial forebrain bundle for depression-long-term outcomes and a novel data analysis strategy. Brain Stimul. 2017, 10, 664–671. [Google Scholar] [CrossRef] [PubMed]
- Riva-Posse, P.; Choi, K.S.; Holtzheimer, P.E.; McIntyre, C.C.; Gross, R.E.; Chaturvedi, A.; Crowell, A.L.; Garlow, S.J.; Rajendra, J.K.; Mayberg, H.S. Defining critical white matter pathways mediating successful subcallosal cingulate deep brain stimulation for treatment-resistant depression. Biol. Psychiatry 2014, 76, 963–969. [Google Scholar] [CrossRef] [PubMed]
- Riva-Posse, P.; Choi, K.S.; Holtzheimer, P.E.; Crowell, A.L.; Garlow, S.J.; Rajendra, J.K.; McIntyre, C.C.; Gross, R.E.; Mayberg, H.S. A connectomic approach for subcallosal cingulate deep brain stimulation surgery: Prospective targeting in treatment-resistant depression. Mol. Psychiatry 2017. [Google Scholar] [CrossRef] [PubMed]
- Fenoy, A.J.; Schulz, P.; Selvaraj, S.; Burrows, C.; Spiker, D.; Cao, B.; Zunta-Soares, G.; Gajwani, P.; Quevedo, J.; Soares, J. Deep brain stimulation of the medial forebrain bundle: Distinctive responses in resistant depression. J. Affect. Disord. 2016, 203, 143–151. [Google Scholar] [CrossRef] [PubMed]
- Jbabdi, S.; Johansen-Berg, H. Tractography: Where do we go from here? Brain Connect. 2011, 1, 169–183. [Google Scholar] [CrossRef] [PubMed]
- Yamada, K.; Sakai, K.; Akazawa, K.; Yuen, S.; Nishimura, T. MR tractography: A review of its clinical applications. Magn. Reson. Med. Sci. 2009, 8, 165–174. [Google Scholar] [CrossRef] [PubMed]
- Price, J.L.; Drevets, W.C. Neurocircuitry of mood disorders. Neuropsychopharmacology 2010, 35, 192–216. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodrigues, N.B.; Mithani, K.; Meng, Y.; Lipsman, N.; Hamani, C. The Emerging Role of Tractography in Deep Brain Stimulation: Basic Principles and Current Applications. Brain Sci. 2018, 8, 23. https://doi.org/10.3390/brainsci8020023
Rodrigues NB, Mithani K, Meng Y, Lipsman N, Hamani C. The Emerging Role of Tractography in Deep Brain Stimulation: Basic Principles and Current Applications. Brain Sciences. 2018; 8(2):23. https://doi.org/10.3390/brainsci8020023
Chicago/Turabian StyleRodrigues, Nelson B., Karim Mithani, Ying Meng, Nir Lipsman, and Clement Hamani. 2018. "The Emerging Role of Tractography in Deep Brain Stimulation: Basic Principles and Current Applications" Brain Sciences 8, no. 2: 23. https://doi.org/10.3390/brainsci8020023
APA StyleRodrigues, N. B., Mithani, K., Meng, Y., Lipsman, N., & Hamani, C. (2018). The Emerging Role of Tractography in Deep Brain Stimulation: Basic Principles and Current Applications. Brain Sciences, 8(2), 23. https://doi.org/10.3390/brainsci8020023




