Evaluation of Visual-Evoked Cerebral Metabolic Rate of Oxygen as a Diagnostic Marker in Multiple Sclerosis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Study Procedures
2.3. cfMRI Parameters and Theory
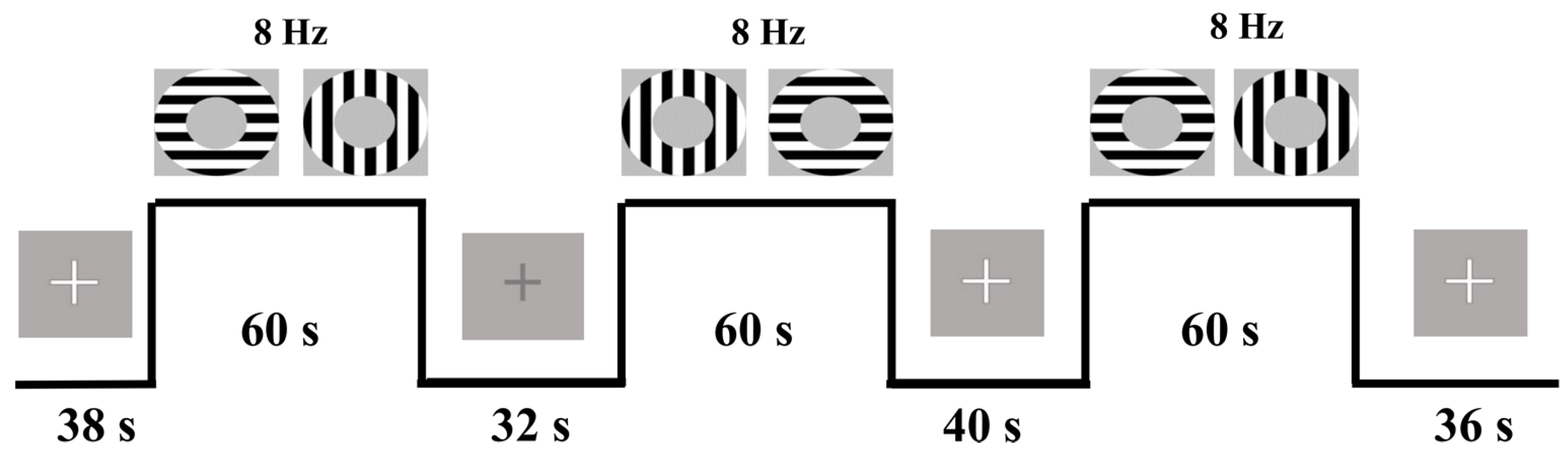
2.4. cfMRI Task and Gas Challenge
2.5. cfMRI Processing
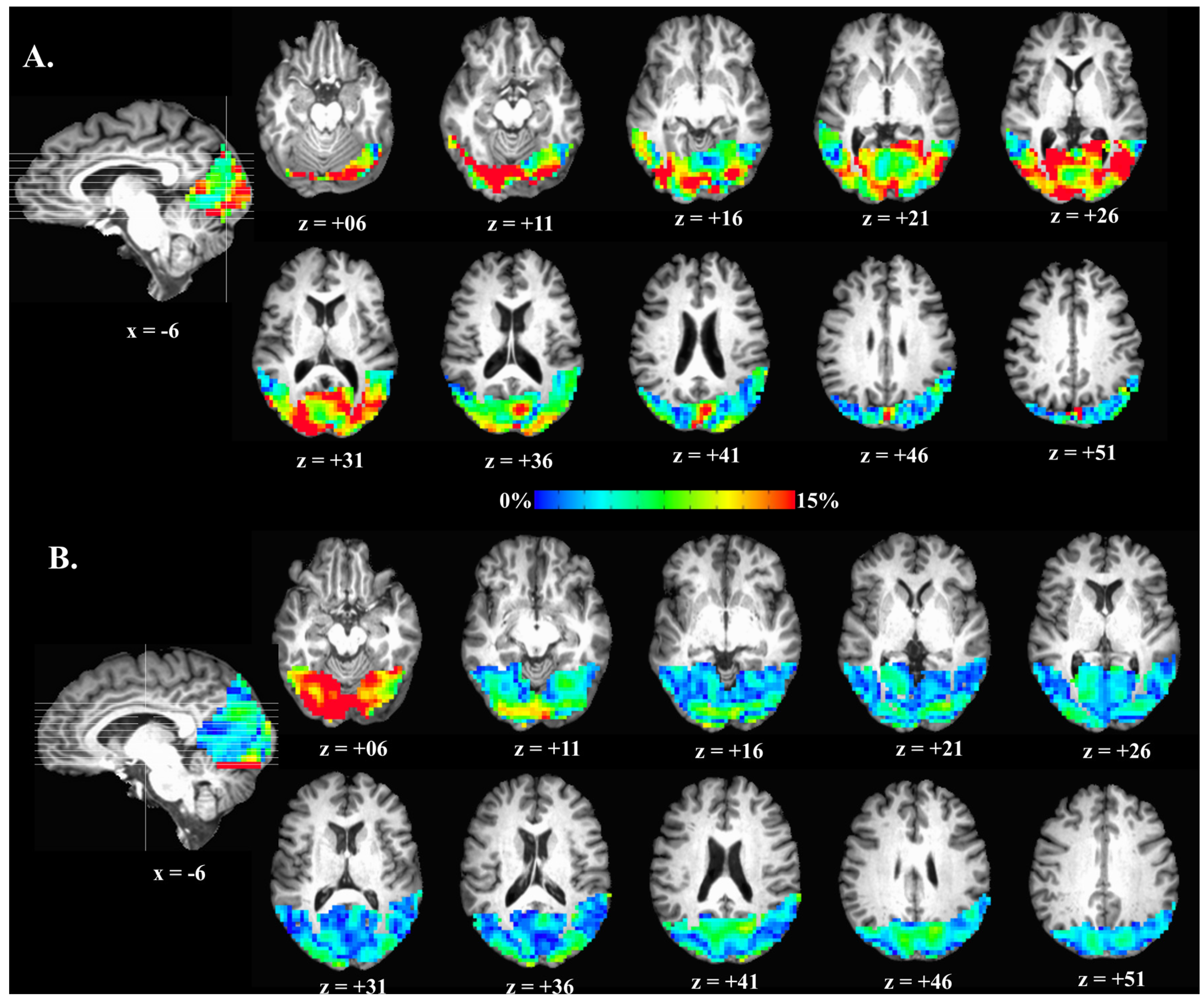
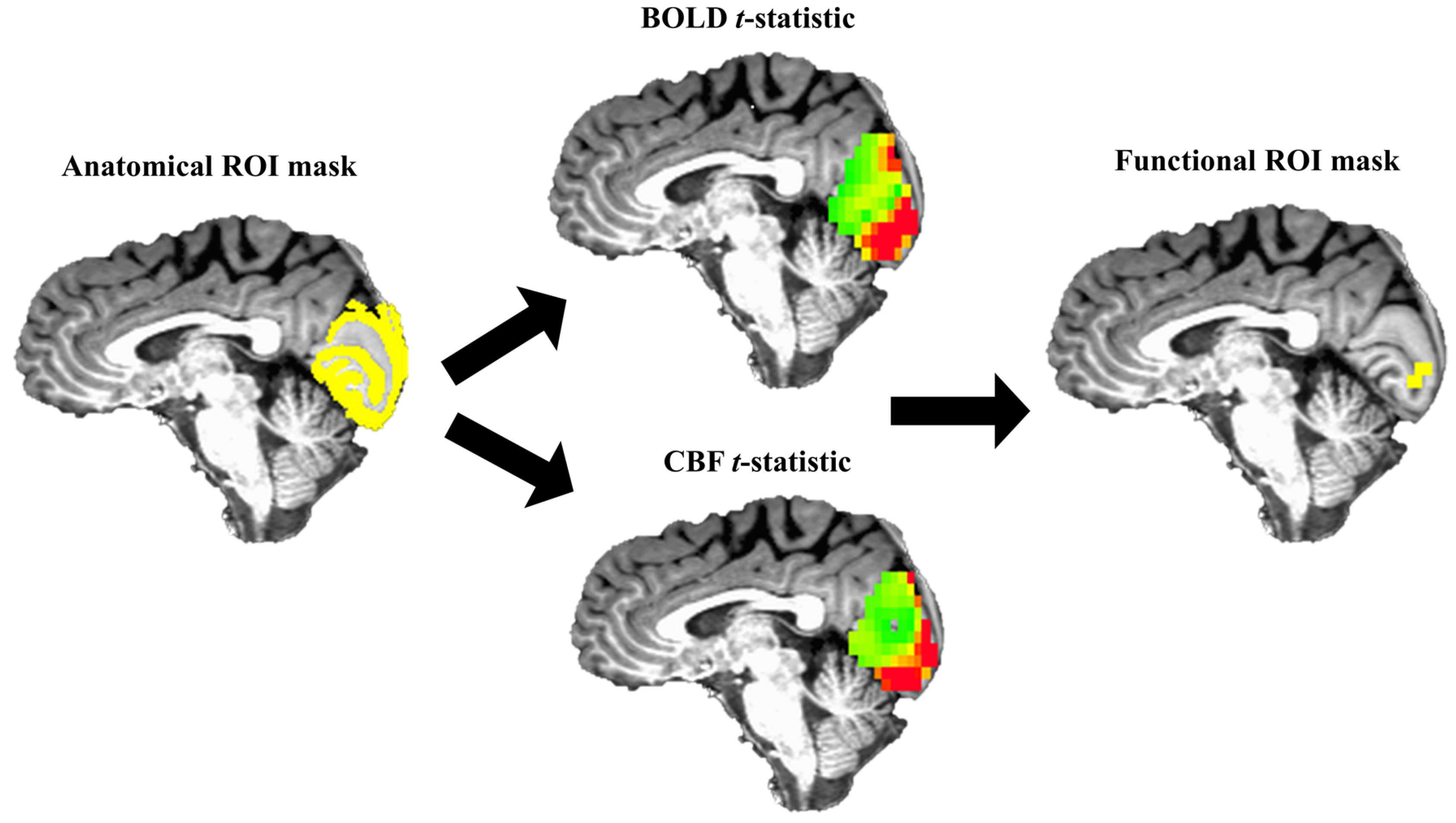
2.6. Structural and Functional ROIs
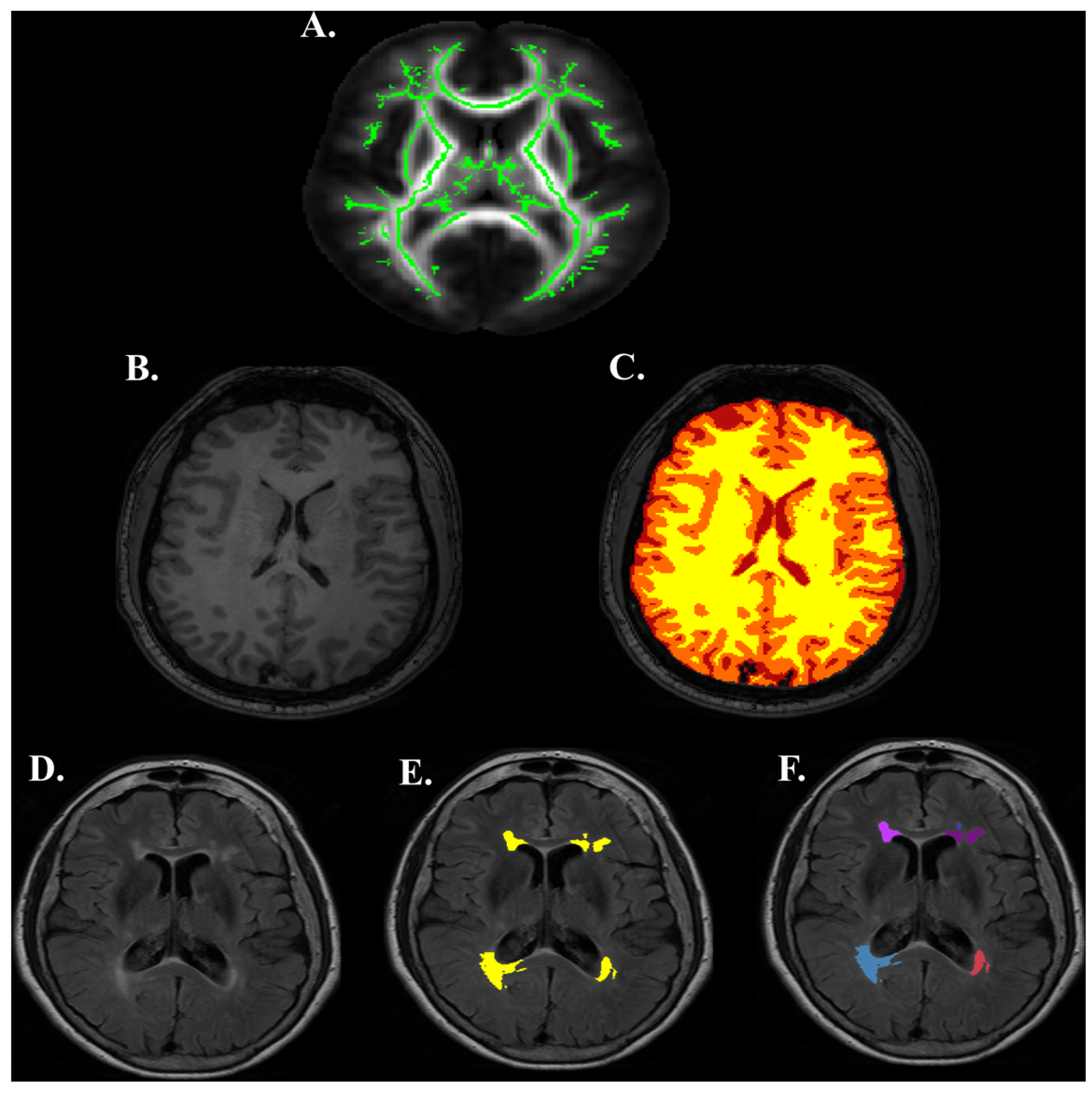
2.7. Structural Images
2.8. Diffusion Images
2.9. Statistical Analyses
3. Results
3.1. Visual Task Performance
3.2. Group Physiology, Cerebrovascular Response to Gas Challenge, and M
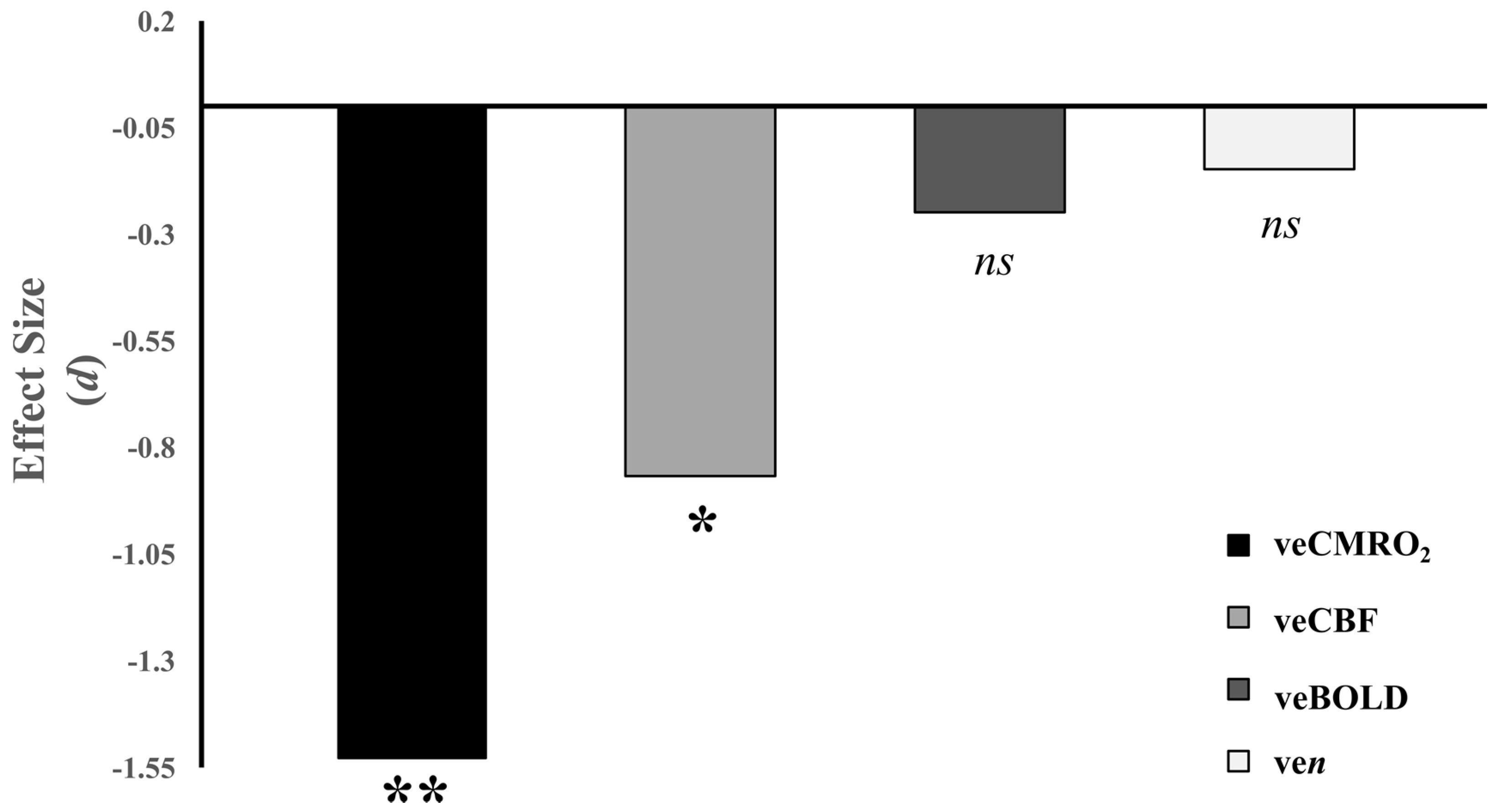
3.3. Group Comparisons on Visual Task cfMRI Measures
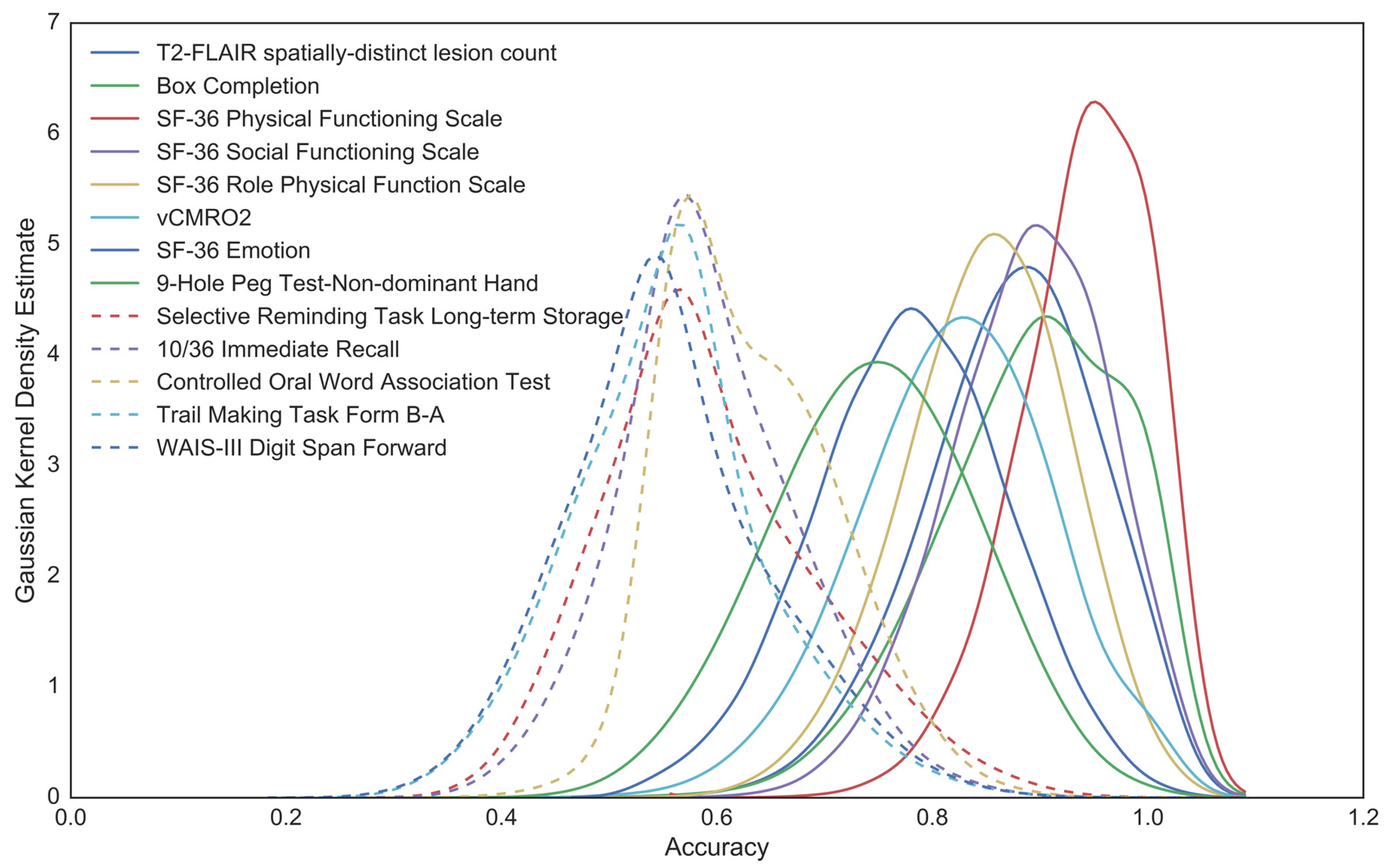
3.4. Within-Sample Classification Analyses
3.5. Out-of-Sample Classification Analyses
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Polman, C.H.; Reingold, S.C.; Banwell, B.; Clanet, M.; Cohen, J.A.; Filippi, M.; Fujihara, K.; Havrdova, E.; Hutchison, M.; Kappos, L.; et al. Diagnostic Criteria for Multiple Sclerosis: 2010 Revisions to the McDonald Criteria. Ann. Neurol. 2011, 69, 292–302. [Google Scholar] [CrossRef] [PubMed]
- George, I.C.; Sati, P.; Absinta, M.; Cortese, I.C.M.; Sweeney, E.M.; Shea, C.D.; Reich, D.S. Clinical 3-Tesla FLAIR* MRI Improves Diagnostic Accuracy in Multiple Sclerosis. Mult. Scler. 2016, 22, 1578–1586. [Google Scholar] [CrossRef] [PubMed]
- Wattjes, M.P.; Barkhof, F. High Field MRI in the Diagnosis of Multiple Sclerosis: High Field-High Yield? Neuroradiology 2009, 51, 279–292. [Google Scholar] [CrossRef] [PubMed]
- Metcalf, M.; Xu, D.; Okuda, D.T.; Carvajal, L.; Srinivasan, R.; Kelley, D.A.C.; Mukherjee, P.; Nelson, S.J.; Vigneron, D.B.; Pelletier, D. High-Resolution Phased-Array MRI of the Human Brain and 7 Tesla: Initial Experience in Multiple Sclerosis Patients. J. Neuroimaging 2010, 20, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Oberwahrenbrok, T.; Ringelstein, M.; Jentschke, S.; Deuschle, K.; Klumbies, K.; Bellmann-Strobl, J.; Harmel, J.; Ruprecht, K.; Schippling, S.; Hartung, H.P.; et al. Retinal Ganglion Cell and Inner Plexiform Layer Thinning in Clinically Isolated Syndrome. Mult. Scler. 2013, 19, 1887–1895. [Google Scholar] [CrossRef] [PubMed]
- Davis, T.L.; Kwong, K.K.; Weiskoff, R.M.; Rosen, B.R. Calibrated Functional MRI: Mapping the Dynamics of Oxidative Metabolism. Proc. Natl. Acad. Sci. USA 1998, 95, 1834–1839. [Google Scholar] [CrossRef] [PubMed]
- Hoge, S.A.; Atkinson, J.; Gill, B.; Crelier, G.R.; Marrett, S.; Pike, G.B. Linear Coupling Between Cerebral Blood Flow and Oxygen Consumption in Activated Human Cortex. Proc. Natl. Acad. Sci. USA 1999, 96, 9403–9408. [Google Scholar] [CrossRef] [PubMed]
- Hoge, R.D. Calibrated fMRI. NeuroImage 2012, 62, 930–937. [Google Scholar] [CrossRef] [PubMed]
- Herman, P.; Sanganahalli, B.G.; Blumenfeld, H.; Hyder, F. Cerebral Oxygen Demand for Short-Lived and Steady-State Events. J. Neurochem. 2009, 109, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Herman, P.; Sanganahalli, B.G.; Blumenfeld, H.; Rothman, D.L.; Hyder, F. Quantitative Basis for Neuroimaging of Cortical Laminae with Calibrated Functional MRI. Proc. Natl. Acad. Sci. USA 2013, 110, 15115–15120. [Google Scholar] [CrossRef] [PubMed]
- Hyder, F.; Kida, I.; Behar, K.L.; Kennan, R.P.; Maciejewski, P.K.; Rothman, D.L. Quantitative Functional Imaging of the Brain: Towards Mapping Neuronal Activity by BOLD fMRI. NMR Biomed. 2001, 14, 413–431. [Google Scholar] [CrossRef] [PubMed]
- Hyder, F.; Rothman, D.L.; Shulman, R.G. Total Neuroenergetics Support Localized Brain Activity: Implications for the Interpretation of fMRI. Proc. Natl. Acad. Sci. USA 2002, 99, 10771–10776. [Google Scholar] [CrossRef] [PubMed]
- Hyder, F. Neuroimaging with Calibrated FMRI. Stroke 2004, 35, 2635–2641. [Google Scholar] [CrossRef] [PubMed]
- Lin, A.-L.; Fox, P.T.; Hardies, J.; Duong, T.Q.; Gao, J.H. Nonlinear Coupling Between Cerebral Blood Flow, Oxygen Consumption, and ATP Production in Human Visual Cortex. Proc. Natl. Acad. Sci. USA 2010, 107, 8446–8451. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.J.; Blumenfeld, H.; Behar, K.J.; Rothman, D.L.; Shulman, R.G.; Hyder, F. Cerebral Energetics and Spiking Frequency: The Neurophysiological Basis of fMRI. Proc. Natl. Acad. Sci. USA 2002, 99, 10765–10770. [Google Scholar] [CrossRef] [PubMed]
- He, B.J.; Snyder, A.Z.; Zempel, J.M.; Smyth, M.D.; Raichle, M.E. Electrophysiological Correlates of the Brain’s Intrinsic Large-Scale Functional Architecture. Proc. Natl. Acad. Sci. USA 2008, 105, 16039–16044. [Google Scholar] [CrossRef] [PubMed]
- Leopold, D.A.; Maier, A. Ongoing Physiological Processes in the Cerebral Cortex. NeuroImage 2012, 62, 2190–2200. [Google Scholar] [CrossRef] [PubMed]
- Logothetis, N.K.; Pauls, J.; Augath, M.; Trinath, T.; Oeltermann, A. Neurophysiological Investigation of the Basis of the fMRI Signal. Nature 2001, 412, 150–157. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; Zuo, Y.; Gu, H.; Waltz, J.A.; Zhan, W.; Scholl, C.A.; Rea, W.; Yang, W.; Stein, E.A. Synchronized Delta Oscillations Correlate with the Resting-State Functional MRI Signal. Proc. Natl. Acad. Sci. USA 2007, 104, 18265–18269. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z.; Johnson, N.F.; Kim, C.; Gold, B.T. Reduced Frontal Cortex Efficiency is Associated with Lower White Matter Integrity in Aging. Cereb. Cortex 2015, 25, 138–146. [Google Scholar] [CrossRef] [PubMed]
- Mark, C.I.; Mazerolle, E.L.; Chen, J.J. Metabolic and Vascular Origins of the BOLD Effect: Implications for Imaging Pathology and Resting-State Brain Function. J. Magn. Reson. Imaging 2015, 42, 231–246. [Google Scholar] [CrossRef] [PubMed]
- Hutchison, J.L.; Lu, H.; Rypma, B. Neural Mechanisms of Age-Related Slowing: The ΔCBF/ΔCMRO2 Ratio Mediates Age-Differences in BOLD Signal and Human Performance. Cereb. Cortex 2013, 23, 2337–2346. [Google Scholar] [CrossRef] [PubMed]
- Hutchison, J.L.; Shokri-Kojori, E.; Lu, H.; Rypma, B. A BOLD Perspective on Age-Related Neurometabolic-Flow Coupling and Neural Efficiency Changes in Human Visual Cortex. Front. Psychol. 2013, 4, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Iannetti, G.D.; Wise, R.G. BOLD Functional MRI in Disease and Pharmacological Studies: Room for Improvement? Magn. Reson. Imaging 2007, 25, 978–988. [Google Scholar] [CrossRef] [PubMed]
- Cader, S.; Johansen-Berg, H.; Wylezinska, M.; Palace, J.; Behrens, T.E.; Smith, S.; Matthews, P.M. Discordant White Matter N-acetylasparate and Diffusion MRI Measure Suggest that Chronic Metabolic Dysfunction Contributes to Axonal Pathology in Multiple Sclerosis. NeuroImage 2007, 36, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Pfueller, C.F.; Brandt, A.U.; Schubert, F.; Bock, M.; Walaszek, B.; Waiszies, H.; Schwenteck, T.; Dörr, J.; Bellmann-Strobl, J.; Mohr, C.; et al. Metabolic Changes in the Visual Cortex are Linked to Retinal Nerve Fiber Layer Thinning in Multiple Sclerosis. PLoS ONE 2011, 6, e18019. [Google Scholar] [CrossRef] [PubMed]
- Hannoun, S.; Bagory, M.; Durand-Dubief, F.; Ibarrola, D.; Comte, J.C.; Confavreux, C.; Cotton, F.; Sappey-Marinier, D. Correlation of diffusion and Metabolic Alterations in Different Clinical Forms of Multiple Sclerosis. PLoS ONE 2012, 7, e32525. [Google Scholar] [CrossRef]
- Sijens, P.E.; Irwan, R.; Potze, J.H.; Mostert, J.P.; De Keyser, J.; Ouderk, M. Analysis of the Human Brain in Primary Progressive Multiple Sclerosis with Mapping of the Spatial Distributions Using 1H MR Spectroscopy and Diffusion Tensor Imaging. Eur. Radiol. 2005, 15, 1686–1693. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Tanaka, M.; Kondo, S. Clinical Significance of Reduced Cerebral Metabolism in Multiple Sclerosis: A Combined PET and MRI Study. Ann. Nucl. Med. 1998, 12, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Ge, Y.; Zhang, Z.; Lu, H.; Tang, L.; Jaggi, H.; Herbert, J.; Babb, J.S.; Rusinek, H.; Grossman, R.I. Characterizing Brain Oxygen Metabolism in Patients with Multiple Sclerosis with T2-Relaxation-Under-Spin-Tagging MRI. J. Cereb. Blood Flow Metab. 2012, 32, 403–412. [Google Scholar] [CrossRef] [PubMed]
- Dutta, R.; McDonough, J.; Yin, X.; Peterson, J.; Chang, A.; Torres, T.; Gudz, T.; Macklin, W.B.; Lewis, D.A.; Fox, R.J.; et al. Mitochondrial Dysfunction as a Cause of Axonal Degeneration in Multiple Sclerosis Patients. Ann. Neurol. 2006, 59, 478–489. [Google Scholar] [CrossRef] [PubMed]
- Mahad, D.J.; Ziabreva, I.; Campbell, G.; Lax, N.; White, K.; Hanson, P.S.; Lassmann, H.; Turnbull, D.M. Mitochondrial Changes Within Axons in Multiple Sclerosis. Brain 2009, 132, 1161–1174. [Google Scholar] [CrossRef] [PubMed]
- Singhal, N.K.; Li, S.; Arning, E.; Alkhayer, K.; Clements, R.; Sarcyk, Z.; Dassanayake, R.S.; Brasch, N.E.; Freeman, E.J.; Bottiglieri, T.; et al. Changes in Methionine Metabolism and Histone H3 Trimethylation are Linked to Mitochondrial Defects in Multiple Sclerosis. J. Neurosci. 2015, 35, 15170–15186. [Google Scholar] [CrossRef] [PubMed]
- Cambron, M.; D’haeseleer, M.; Laureys, G.; Clinckers, R.; Debruyne, J.; De Keyser, J. White-Matter Astrocytes, Axonal Energy Metabolism, and Axonal Degeneration in Multiple Sclerosis. J. Cereb. Blood Flow Metab. 2012, 32, 413–424. [Google Scholar] [CrossRef] [PubMed]
- Paling, D.; Golay, X.; Wheeler-Kingshott, C.; Kapoor, R.; Miller, D. Energy Failure in Multiple Sclerosis and its Investigation Using MR Techniques. J. Neurol. 2011, 258, 2113–2127. [Google Scholar] [CrossRef] [PubMed]
- Su, K.; Bourdette, D.; Forte, M. Mitochondrial Dysfunction and Neurodegeneration in Multiple Sclerosis. Front. Physiol. 2013, 4, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Frohman, E.M.; Frohman, T.C.; Zee, D.S.; McColl, R.; Galetta, S. The Neuro-Ophthalmology of Multiple Sclerosis. Lancet Neurol. 2005, 4, 111–121. [Google Scholar] [CrossRef]
- Frohman, E.M.; Fujimoto, J.G.; Frohman, T.C.; Calabresi, P.A.; Cutter, G.; Balcer, L.J. Optical Coherence Tomography: A Window Into the Mechanisms of Multiple Sclerosis. Nat. Clin. Pract. Neurol. 2008, 4, 664–675. [Google Scholar] [CrossRef] [PubMed]
- Graham, S.L.; Klistorner, A. Afferent Visual Pathways in Multiple Sclerosis: A Review. Clin. Exp. Ophthalmol. 2017, 45, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Kolappan, M.; Henderson, A.P.D.; Jenkins, T.M.; Wheeler-Kingshott, C.A.; Plant, G.T.; Miller, D.H. Assessing Structure and Function of the Afferent Visual Pathway in Multiple Sclerosis and Associated Optic Neuritis. J. Neurol. 2009, 256, 305–319. [Google Scholar] [CrossRef] [PubMed]
- Alshowaeir, D.; Yiannikas, C.; Garrick, R.; Paratt, J.; Barnett, M.H.; Graham, S.L.; Klistorner, A. Latency of Multifocal Visual Evoked Potentials in Nonoptic Neuritis Eyes of Multiple Sclerosis Patients Associated with Optic Radiation Lesions. Investig. Ophthalmol. Vis. Sci. 2014, 55, 3758–3764. [Google Scholar] [CrossRef] [PubMed]
- Hubbard, N.A.; Turner, M.; Hutchison, J.L.; Ouyang, A.; Strain, J.; Oasay, L.; Sundaram, S.; Davis, S.; Remington, G.; Brigante, R.; et al. Multiple Sclerosis-Related White Matter Microstructural Change Alters the BOLD Hemodynamic Response. J. Cereb. Blood Flow Metab. 2016, 36, 1872–1884. [Google Scholar] [CrossRef] [PubMed]
- Hubbard, N.A.; Hutchison, J.L.; Turner, M.P.; Sundaram, S.; Oasay, L.; Robinson, D.; Strain, J.; Weaver, T.; Davis, S.L.; Remington, G.M.; et al. Asynchrony in Executive Networks Predicts Cognitive Slowing in Multiple Sclerosis. Neuropsychology 2016, 30, 75. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.M. Cognitive Function Study Group of the National Multiple Sclerosis Society. In A Manual for the Brief Repeatable Battery of Neuropsychological Tests in Multiple Sclerosis; Medical College of Wisconsin: Milwaukee, WI, USA, 1990. [Google Scholar]
- Brandt, J.; Spencer, M.; Folstein, M. The Telephone Interview for Cognitive Status. Neuropsychiatry Neuropsychol. Behav. Neurol. 1988, 1, 111–117. [Google Scholar]
- Verdier-Taillefer, M.H.; Roullet, E.; Cesaro, P.; Alpérovitch, A. Validation of Self-Reported Neurological Disability in Multiple Sclerosis. Int. J. Epidemiol. 1994, 23, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Perthen, J.E.; Lansing, A.E.; Liau, J.; Liu, T.T.; Buxton, R.B. Caffeine-Induced Uncoupling of Cerebral Blood Flow and Oxygen Metabolism: A Calibrated BOLD fMRI Study. NeuroImage 2008, 40, 237–247. [Google Scholar] [CrossRef] [PubMed]
- Ware, J.E.; Kosinski, M.; Keller, S.D. SF-36 Physical and Mental Health Summary Scales: A Users’ Manual; The Health Institute: Scarborough, ON, Canada; New England Medical Center: Boston, MA, USA, 1994. [Google Scholar]
- Fisk, J.D.; Pontefract, A.; Ritvo, P.G.; Archibald, C.J.; Muarray, T.J. The impact of fatigue on patients with multiple sclerosis. Can. J. Neurol. Sci. 1994, 21, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Boringa, J.B.; Lazeron, R.H.C.; Reuling, I.E.W.; Adèr, H.J.; Pfennings, L.E.M.A.; Lindeboom, J.; de Sonneville, L.M.J.; Kalkers, N.F.; Polman, C.H. The Brief Repeatable Battery of Neuropsychological Tests: Normative Values Allow Application in Multiple Sclerosis Clinical Practice. Mult. Scler. 2001, 7, 263–267. [Google Scholar] [CrossRef] [PubMed]
- Strauss, E.; Sherman, E.M.S.; Spreen, O. A Compendium of Neuropsychological Tests: Administration, Norms, and Commentary; American Chemical Society: Washington, DC, USA, 2006. [Google Scholar]
- Lu, H.; van Zijl, P. Experimental Measurement of Extravascular Parenchymal BOLD Effects and Tissue Oxygen Extraction Fractions Using Multi-Echo VASO fMRI at 1.5 and 3.0 T. Magn. Reson. Med. 2005, 53, 808–816. [Google Scholar] [CrossRef] [PubMed]
- Ances, B.M.; Liang, C.L.; Leontiev, O.; Perthen, J.E.; Fleisher, A.S.; Lansing, A.E.; Buxton, R.B. Effects of Aging on Cerebral Blood Flow, Oxygen Metabolism, and Blood Oxygen Level Dependent Responses to Visual Stimulation. Hum. Brain Mapp. 2009, 30, 1120–1132. [Google Scholar] [CrossRef] [PubMed]
- Buxton, R.B. Interpreting Oxygenation-Based Neuroimaging Signals: The Importance and the Challenge of Understanding Brain Oxygen Metabolism. Front. Neuroenerg. 2010. [Google Scholar] [CrossRef] [PubMed]
- Leontiev, O.; Buxton, R.B. Reproducibility of BOLD, Perfusion, and CMRO2 Measurements with Calibrated-BOLD fMRI. NeuroImage 2007, 35, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Grubb, R.L.; Raichle, M.E.; Eichling, J.O.; Ter-Pogossian, M.M. The Effects of Changes in PaCO2 Cerebral Blood Volume, Blood Flow, and Vascular Mean Transit Time. Stroke 1974, 5, 630–639. [Google Scholar] [CrossRef] [PubMed]
- Hubbard, N.A.; Turner, M.P.; Robinson, D.M.; Sundaram, S.; Oasay, L.; Hutchison, J.L.; Ouyang, A.; Huang, H.; Rypma, B. Attenuated BOLD Hemodynamic Response Predicted by Degree of White Matter Insult, Slows Cognition in Multiple Sclerosis. Mult. Scler. J. 2014, 20, 267. [Google Scholar]
- Pasley, B.N.; Inglis, B.A.; Freeman, R.D. Analysis of Oxygen Metabolism Implies a Neural Origin for the Negative BOLD Response in Human Visual Cortex. NeuroImage 2007, 36, 269–276. [Google Scholar] [CrossRef] [PubMed]
- Lin, A.; Fox, P.T.; Yang, Y.; Lu, J.; Tan, L.H.; Gao, J.H. Evaluation of MRI Models in the Measurement of CMRO2 and Its Relationship with CBF. Magn. Reson. Med. 2008, 60, 380–389. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.; Kim, S.; Kim, T. Correlation Between BOLD-fMRI and EEG Signal Changes in Response to Visual Stimulus Frequency in Humans. Magn. Reson. Med. 2003, 49, 108–114. [Google Scholar] [CrossRef] [PubMed]
- Peng, S.L.; Ravi, H.; Sheng, M.; Thomas, B.P.; Lu, H. Searching for a Truly “Iso-Metabolic” Gas Challenge in Physiological MRI. J. Cereb. Blood Flow Metab. 2017, 37, 715–725. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Uh, J.; Brier, M.R.; Hart, J., Jr.; Yezhuvath, U.S.; Gu, H.; Yang, Y.; Lu, H. The Influence of Carbon Dioxide on Brain Activity and Metabolism in Conscious Humans. J. Cereb. Blood Flow Metab. 2011, 31, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Zappe, A.C.; Uludağ, K.; Oeltermann, A.; Uğurbil, K.; Logothetis, N.L. The Influence of Moderate Hypercapnia on Neural Activity in the Anesthetized Nonhuman Primate. Cereb. Cortex 2008, 18, 2666–2673. [Google Scholar] [CrossRef] [PubMed]
- Yucel, M.A.; Evans, K.C.; Selb, J.; Huppert, T.J.; Boas, D.A.; Gagnon, L. Validation of the Hypercapnic Calibrated fMRI Method Using DOT-fMRI Fusion Imaging. NeuroImage 2014, 102, 729–735. [Google Scholar] [CrossRef] [PubMed]
- Cox, R.W. AFNI: Software for Analysis and Visualization of Functional Magnetic Resonance Neuroimages. Comput. Biomed. Res. 1996, 29, 162–173. [Google Scholar] [CrossRef] [PubMed]
- FMRIB Analysis Group. FMRIB Software Library v5.0. Available online: https://fsl.fmrib.ox.ac.uk/fsl/fslwiki (accessed on 3 June 2017).
- Liu, T.T.; Wong, E.C. A Signal Processing Model for Arterial Spin Labeling Functional MRI. NeuroImage 2005, 24, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Hutchison, J.L.; Hubbard, N.A.; Brigante, R.M.; Turner, M.; Sandoval, T.I.; Hillis, G.A.; Weaver, T.; Rypma, B. The Efficiency of fMRI Region of Interest Analysis Methods for Detecting Group Differences. J. Neurosci. Methods 2014, 226, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.M.; De Stefano, N.; Jenkinson, M.; Matthews, P.M. Normalised Accurate Measurement of Longitudinal Brain Change. J. Comput. Assist. Tomogr. 2001, 25, 466–475. [Google Scholar] [CrossRef] [PubMed]
- Hart, J., Jr.; Kraut, M.A.; Womack, K.B.; Strain, J.; Didehbani, N.; Bartz, E.; Conover, H.; Mansinghani, S.; Lu, H.; Cullum, C.M. Neuroimaging of Cognitive Dysfunction and Depression in Aging Retired National Football League Players. JAMA Neurol. 2013, 70, 326–335. [Google Scholar] [CrossRef] [PubMed]
- Dice, L.R. Measures of the Amount of Ecologic Association between Species. Ecology 1945, 26, 297–302. [Google Scholar] [CrossRef]
- Zhang, L.; Dean, D.; Liu, J.Z.; Sahgal, V.; Wang, X.; Yue, G.H. Quantifying Degeneration of White Matter in Normal Aging Using Fractal Dimension. Neurobiol. Aging 2007, 28, 1543–1555. [Google Scholar] [CrossRef] [PubMed]
- Ghassemi, R.; Narayana, S.; Banwell, B.; Sled, J.G.; Shroff, M.; Arnold, D.L. Quantitative Determination of Regional Lesion Volume and distribution in Children and Adults with Relapsing-Remitting Multiple Sclerosis. PLoS ONE 2014, 9, e85741. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.K.; Simmons, A.; Williams, S.C.; Horsfield, M.A. Non-invasive Assessment of Axonal Fiber Connectivity in the Human Brain via Diffusion Tensor MRI. Magn. Reson. Med. 1999, 42, 37–41. [Google Scholar] [CrossRef]
- Woods, R.P.; Grafton, S.T.; Holmes, C.J.; Cherry, S.R.; Mazziotta, J.C. Automated Image Registration: I. General Methods and Intrasubject, Intramodality Validation. J. Comput. Assist. Tomogr. 1998, 22, 139–152. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; van Zijl, P.C.J.K.; Pearlson, G.D.; Mori, S. DtiStudio: Resource Program for Diffusion Tensor Computation and Fiber Bundle Tracking. Comput. Methods Programs Biomed. 2006, 81, 106–116. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.M.; Jenkinson, M.; Johansen-Berg, H.; Rueckert, D.; Nichols, T.E.; Mackay, C.E.; Watkins, K.E.; Ciccarelli, O.; Cader, M.Z.; Matthews, P.M.; et al. Tract-Based Spatial Statistics: Voxelwise Analysis of Multi-Subject Diffusion Data. NeuroImage 2006, 3, 1487–1505. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Gundapuneedi, T.; Rao, U. White Matter Disruptions in Adolescents Exposed to Childhood Maltreatment and Vulnerability to Psychopathology. Neuropsychopharmacology 2012, 37, 2693–2701. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Fan, X.; Weiner, M.; Martin-Cook, K.; Xiao, G.; Davis, J.; Devous, M.; Rosenberg, R. Distinctive Disruption Patterns of White Matter Tracts in Alzheimer’s Disease with Full Diffusion Tensor Characterization. Neurobiol. Aging 2012, 33, 2029–2045. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, M.; Cheng, H.; Mishra, V.; Gong, G.; Mosconi, M.; Sweeney, J.; Peng, Y.; Huang, H. Atypical age-dependent effects of autism on white matter microstructure in children of 2–7 years. Hum. Brain Mapp. 2016, 37, 819–832. [Google Scholar] [CrossRef] [PubMed]
- Mori, S.; Oishi, K.; Jiang, H.; Jiang, L.; Li, X.; Akhter, K.; Hua, K.; Faria, A.V.; Mahmood, A.; Woods, R.; et al. Stereotaxic White Matter Atlas Based on Diffusion Tensor Imaging in an ICBM Template. NeuroImage 2008, 40, 570–582. [Google Scholar] [CrossRef] [PubMed]
- Iglewicz, B.; Hoaglin, D. Volume 16: How to Detect and Handle Outliers. In The ASQC Basic References in Quality Control: Statistical Techniques; Mykytka, E.F., Ed.; American Society for Quality Control, Statistics Division: Milwaukee, WI, USA, 1993. [Google Scholar]
- Gabrieli, J.D.E.; Ghosh, S.S.; Whitfield-Gabrieli, S. Prediction as a Humanitarian and Pragmatic Contribution from Human Cognitive Neuroscience. Neuron 2015, 85, 11–26. [Google Scholar] [CrossRef] [PubMed]
- Efron, B. Better Bootstrap Confidence Intervals. J. Am. Stat. Assoc. 1987, 82, 171–185. [Google Scholar] [CrossRef]
- Kohavi, R. A Study of Cross-Validation and Bootstrap for Accuracy Estimation and Model Selection. In Proceedings of the Fourteenth International Joint Conference on Artificial Intelligence, Montreal, QC, Canada, 20–25 August 1995; Morgan Kaufmann Publishers: San Mateo, CA, USA, 1995; Volume 2, pp. 1137–1143. [Google Scholar]
- Ojala, M.; Garriga, G.C. Permutation Tests for Studying Classifier Performance. J. Mach. Learn. Res. 2010, 11, 1833–1863. [Google Scholar]
- Marshall, O.; Lu, H.; Brisset, J.C.; Xu, F.; Liu, P.; Herbert, J.; Grossman, R.I.; Ge, Y. Impaired Cerebrovascular Reactivity in Multiple Sclerosis. JAMA Neurol. 2014, 71, 1275–1281. [Google Scholar] [CrossRef] [PubMed]
- Laron, M.; Cheng, H.; Zhang, B.; Schiffman, J.S.; Tang, R.A.; Frishman, L.J. Comparison of Multifocal Visual Evoked Potential, Standard Automated Perimetry and Optical Coherence Tomography in Assessing Visual Pathways in Multiple Sclerosis Patients. Mult. Scler. 2010, 16, 412–426. [Google Scholar] [CrossRef] [PubMed]
- Fraser, C.; Klistorner, A.; Graham, S.; Garrick, R.; Billson, F.; Grigg, J. Multifocal Visual Evoked Potential Latency Analysis: Predicting Progression to Multiple Sclerosis. Arch. Neurol. 2006, 63, 847–850. [Google Scholar] [CrossRef] [PubMed]
- Ruseckaite, R.; Maddess, T.; Danta, G.; Lueck, C.J.; James, A.C. Sparse Multifocal Stimuli for the Detection of Multiple Sclerosis. Ann. Neurol. 2005, 57, 904–913. [Google Scholar] [CrossRef] [PubMed]
- Pantano, P.; Mainero, C.; Caramia, F. Functional Brain Reorganization in Multiple Sclerosis: Evidence from fMRI Studies. J. Neuroimaging 2006, 16, 104–114. [Google Scholar] [CrossRef] [PubMed]
- White, A.T.; Lee, J.N.; Light, A.R.; Light, K.C. Brain Activation in Multiple Sclerosis: A BOLD fMRI Study of the Effects of Fatiguing Hand Exercise. Mult. Scler. 2009, 15, 580–586. [Google Scholar] [CrossRef] [PubMed]
- Chiaravalloti, N.D.; Hillary, F.G.; Ricker, J.H.; Christodoulou, C.; Kalnin, A.J.; Liu, W.C.; Steffener, J.; DeLuca, J. Cerebral Activation Patterns During Working Memory Performance in Multiple Sclerosis using fMRI. J. Clin. Exp. Neuropsychol. 2005, 27, 33–54. [Google Scholar] [CrossRef] [PubMed]
- Genova, H.M.; Sumowski, J.F.; Chiaravalloti, N.; Voelbel, G.T.; DeLuca, J. Cognition in Multiple Sclerosis: A Review of Neuropsychological and fMRI Research. Front. Biosci. 2009, 14, 1730–1744. [Google Scholar] [CrossRef]
- Sweet, L.H.; Rao, S.M.; Primeau, M.; Durgerian, S.; Cohen, R.A. Functional Magnetic Resonance Imaging Response to Increased Verbal Working Memory Demands Among Patients with Multiple Sclerosis. Hum. Brain. Mapp. 2006, 27, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Genova, H.M.; Hillary, F.G.; Wylie, G.; Rypma, B.; DeLuca, J. Examination of Processing Speed Deficits in Multiple Sclerosis Using Functional Magnetic Resonance Imaging. J. Int. Neuropsychol. Soc. 2009, 15, 383–393. [Google Scholar] [CrossRef] [PubMed]
- Comabella, M.; Sastre-Garriga, J.; Montalban, X. Precision Medicine in Multiple Sclerosis: Biomarkers for Diagnosis, Prognosis, and Treatment Response. Curr. Opin. Neurol. 2016, 29, 254–262. [Google Scholar] [CrossRef] [PubMed]
- Pulicken, M.; Gordon-Lipkin, E.; Balcer, L.J.; Frohman, E.; Cutter, G.; Calabresi, P.A. Optical Coherence Tomography and Disease Subtype in Multiple Sclerosis. Neurology 2007, 69, 2085–2092. [Google Scholar] [CrossRef] [PubMed]
- Fisniku, L.K.; Brex, P.A.; Altmann, D.R.; Miszkiel, K.A.; Benton, C.E.; Lanyon, R.; Thompson, A.J.; Miller, D.H. Disability and T2 MRI Lesions: A 20-Year Follow-Up of Patients with Relapse Onset of Multiple Sclerosis. Brain 2008, 131, 808–817. [Google Scholar] [CrossRef] [PubMed]
- Lebrun, C.; Bensa, C.; Debouverie, M.; Wiertlevski, S.; Brassat, D.; de Seze, J.; Rumbach, L.; Pelletier, J.; Labauge, P.; Brochet, B.; et al. Association Between Clinical Conversion to Multiple Sclerosis in Radiologically Isolated Syndrome and Magnetic Resonance Imaging, Cerebrospinal Fluid, and Visual Evoked Potential. Arch. Neurol. 2009, 66, 841–846. [Google Scholar] [CrossRef] [PubMed]
- Leocanti, L.; Rocca, M.A.; Comi, G. MRI and Neurophysiological Measures to Predict Course, Disability and Treatment Response in Multiple Sclerosis. Curr. Opin. Neurol. 2016, 29, 243–253. [Google Scholar] [CrossRef] [PubMed]
- Okuda, D.T.; Mowry, E.M.; Cree, B.A.C.; Crabtree, E.C.; Goodin, D.S.; Waubant, E.; Pelletier, D. Asymptomatic Spinal Cord Lesions Predict Disease Progression in Radiologically Isolated Syndrome. Neurology 2011, 76, 686–692. [Google Scholar] [CrossRef] [PubMed]
- Stromillo, M.L.; Giorgio, A.; Rossi, F.; Battaglini, M.; Hakiki, B.; Malentacchi, G.; Santangelo, M.; Gasperini, C.; Bartolozzi, M.L.; Portaccio, E.; et al. Brain metabolic changes suggestive of axonal damage in radiologically isolated syndrome. Neurology 2013, 80, 2090–2094. [Google Scholar] [CrossRef] [PubMed]
- Campbell, G.R.; Worrall, J.T.; Mahad, D.J. The Central Role of Mitochondrial in Axonal Degeneration in Multiple Sclerosis. Mult. Scler. 2014, 20, 1806–1813. [Google Scholar] [CrossRef] [PubMed]
- Balcer, L.J.; Raynowska, J.; Nolan, R.; Galetta, S.L.; Kapoor, R.; Benedict, R.; Phillips, G.; LaRocca, N.; Hudson, L.; Rudick, R.; et al. Validity of Low-Contrast Letter Acuity as a Visual Performance Outcome Measure for Multiple Sclerosis. Mult. Scler. 2017, 23, 734–747. [Google Scholar] [CrossRef] [PubMed]
- Link, H.; Huang, Y.-M. Oligoclonal Bands in Multiple Sclerosis Cerebrospinal Fluid: An Update on Methodology and Clinical Usefulness. J. Immunol. 2006, 180, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Shaffer, J.L.; Petrella, J.R.; Sheldon, F.C.; Choudhury, K.R.; Calhoun, V.D.; Coleman, R.E.; Doraiswamy, P.M. Predicting Cognitive Decline in Subjects at Risk for Alzheimer Disease by Using Combined Cerebrospinal Fluid, MR Imaging, and PET Biomarkers. Radiology 2013, 266, 583–591. [Google Scholar] [CrossRef] [PubMed]
- Whitfield-Gabrieli, S.; Ghosh, S.S.; Nieto-Castanon, A.; Saygin, Z.; Doehrmann, O.; Chai, X.J.; Reynolds, G.O.; Hofmann, S.G.; Pollack, M.H.; Gabrieli, J.D.E. Brain Connectomics Predict Response to Treatment in Social Anxiety Disorder. Mol. Psychiatry 2015, 21, 680–685. [Google Scholar] [CrossRef] [PubMed]

| MS | HC | p | |
|---|---|---|---|
| Age | 50.10 (3.35) | 50.77 (3.35) | 0.885 a |
| MFIS | 39.10 (7.62) | 20.54 (4.57) | 0.046 a |
| Sex (% female) | 90.00% | 84.62% | 0.704 b |
| TICS Score | 27.00 (0.82) | 28.08 (1.43) | 0.520 a |
| Age of MS Onset | 38.67 (2.42) | - | - |
| Disease Duration | 118.80 (19.32) | - | - |
| Last Flare-up | 28.60 (11.32) | - | - |
| Neurological Disability Score | 15.70 (3.71) | - | - |
| Disease Modifying Therapies | |||
| Dalfampridine | 50% | - | - |
| Dimethyl fumarate | 10% | - | - |
| Fingolimod | 20% | - | - |
| Glatiramer acetate | 10% | - | - |
| Predictor (Units if Available) | Predictor Category | What Predictor Measures |
|---|---|---|
| Normalized Grey Matter Volume (mm3) | MR Image | Total grey matter volume normalized to skull |
| Normalized White Matter Volume (mm3) | MR Image | Total white matter volume normalized to skull |
| Normalized Whole Brain Volume (mm3) | MR Image | Total brain volume normalized to skull |
| Skeleton AD (mm2/s) | MR Image | Diffusion along primary diffusion axis |
| Skeleton FA (proportion) | MR Image | Proportion of anisotropic diffusion |
| Skeleton MD (mm2/s) | MR Image | Average Diffusion in primary diffusion axes |
| Skeleton RD (mm2/s) | MR Image | Diffusion orthogonal to primary diffusion axis |
| T2-FLAIR Lesion Burden-absolute lesion volume (mm3) | MR Image | Total volume of lesioned brain tissue |
| T2-FLAIR Lesion Burden-relative lesion volume (%) | MR Image | Total lesioned brain tissue relative to total white matter volume |
| T2-FLAIR spatially distinct lesion count | MR Image | Total number of spatially distinct lesions |
| veBOLD (% signal change) | MR Image | Visual cortex BOLD response to visual stimulation task |
| veCBF (% signal change) | MR Image | Visual cortex CBF response to visual stimulation task |
| veCMRO2 (% signal change) | MR Image | Visual cortex CMRO2 response to visual stimulation task |
| ven (proportion) | MR Image | Visual cortex neural-vascular coupling |
| 10/36 Delayed Recall (total correct after 15 min) | Neuropsych | Visuospatial memory/learning and delayed recall |
| 10/36 Immediate Recall (total correct) | Neuropsych | Visuospatial memory/learning |
| 25 Foot Walk (s) | Neuropsych | Walking ability and gait speed |
| 9-Hole Peg Test-Dominant Hand (s) | Neuropsych | Finger and hand dexterity |
| 9-Hole Peg Test-Non-dominant Hand (s) | Neuropsych | Finger and hand dexterity |
| Box Completion (items completed) | Neuropsych | Motor control |
| Controlled Oral Word Association Test (total correct) | Neuropsych | Verbal association fluency |
| Number Comparison (items completed) | Neuropsych | Processing speed |
| Paced Auditory Serial Addition Test 2 (% correct) | Neuropsych | Processing speed and selective/sustained attention |
| Paced Auditory Serial Addition Test 3 (% correct) | Neuropsych | Processing speed and selective/sustained attention |
| Selective Reminding Task Delayed (items recalled) | Neuropsych | Verbal learning and memory |
| Selective Reminding Task Long-term Storage (items recalled) | Neuropsych | Verbal learning and long-term memory |
| Symbol-digit Modalities Test (items completed) | Neuropsych | Sustained attention and concentration |
| Trail Making Task Form A (s) | Neuropsych | Visual search, attention, mental flexibility, and motor function |
| Trail Making Task Form B (s) | Neuropsych | Visual search, attention, mental flexibility, and motor function |
| Trail Making Task Form B-A (s) | Neuropsych | Visual search, attention, mental flexibility, and motor function |
| WAIS-III Digit Span Backward (items completed) | Neuropsych | Short-term, working memory |
| WAIS-III Digit Span Forward (items completed) | Neuropsych | Short-term, working memory |
| WAIS-III Digit Span Total (items completed) | Neuropsych | Short-term, working memory |
| WAIS-III Digit symbol coding (items completed) | Neuropsych | Performance subtest of WAIS |
| Modified Fatigue Impact Score | Symptoms | Fatigue symptomology |
| SF-36 Bodily Pain Scale | Symptoms | General measure of bodily pain |
| SF-36 Emotion | Symptoms | Role limitations due to emotional problems |
| SF-36 General Health Scale | Symptoms | General measure of health wellbeing |
| SF-36 Mental Health Scale | Symptoms | General measure of mental health |
| SF-36 Physical Functioning Scale | Symptoms | General measure of physical functioning |
| SF-36 Role Physical Function Scale | Symptoms | Role limitations due to physical problems |
| SF-36 Social Functioning Scale | Symptoms | General measure of social functioning |
| SF-36 Vitality Scale | Symptoms | General measure of energy/fatigue |
| MS | HC | p | |
|---|---|---|---|
| Baseline | |||
| Breath Rate | 11.20 (1.00) | 10.25 (0.79) | 0.747 a |
| EtCO2 | 42.70 (1.81) | 39.23 (0.74) | 0.101 b |
| Heart Rate | 66.90 (2.38) | 72.08 (3.18) | 0.207 b |
| SpO2 | 98.10% (0.35%) | 97.85% (0.32%) | 0.596 b |
| 5% CO2 | |||
| Breath Rate | 13.35 (1.28) | 15.42 (1.07) | 0.236 c |
| EtCO2 | 48.95 (1.45) | 49.06 (0.64) | 0.950 c |
| Heart Rate | 69.67 (2.38) | 75.04 (2.60) | 0.147 d |
| SpO2 | 97.58% (0.39%) | 98.20% (0.20%) | 0.139 d |
| Predictor | Predictor Accuracy | 95% LCL | 95% UCL | Significant |
|---|---|---|---|---|
| SF-36 Physical Functioning Scale | 0.94 | 0.65 | 1.00 | Yes † |
| SF-36 Social Functioning Scale | 0.89 | 0.61 | 0.94 | Yes † |
| T2-FLAIR spatially distinct lesion count | 0.86 | 0.57 | 0.95 | Yes † |
| Box Completion | 0.86 | 0.52 | 0.95 | Yes † |
| SF-36 Role Physical Function Scale | 0.85 | 0.60 | 0.95 | Yes † |
| veCMRO2 | 0.82 | 0.55 | 0.91 | Yes ‡ |
| Normalized Grey Matter Volume | 0.81 | 0.43 | 0.95 | No ‡ |
| T2-FLAIR Lesion Burden-absolute lesion volume | 0.80 | 0.50 | 0.90 | No ‡ |
| T2-FLAIR Lesion Burden-relative lesion volume | 0.80 | 0.50 | 0.90 | No ‡ |
| SF-36 Emotion | 0.78 | 0.56 | 0.89 | Yes |
| 9-Hole Peg Test-Non-dominant Hand | 0.77 | 0.55 | 0.91 | Yes ‡ |
| SF-36 General Health Scale | 0.77 | 0.50 | 0.86 | No ‡ |
| veCBF | 0.75 | 0.45 | 0.85 | No ‡ |
| Normalized Whole Brain Volume | 0.73 | 0.45 | 0.86 | No |
| 9-Hole Peg Test-Dominant Hand | 0.73 | 0.50 | 0.82 | No |
| SF-36 Bodily Pain Scale | 0.73 | 0.45 | 0.86 | No |
| Skeleton AD | 0.71 | 0.43 | 0.81 | No |
| Skeleton MD | 0.71 | 0.48 | 0.86 | No |
| Paced Auditory Serial Addition Test 2 s | 0.71 | 0.48 | 0.86 | No |
| Modified Fatigue Impact Score Total | 0.70 | 0.43 | 0.78 | No ‡ |
| Normalized White Matter Volume | 0.68 | 0.45 | 0.82 | No |
| Paced Auditory Serial Addition Test 3 s | 0.68 | 0.45 | 0.82 | No |
| Skeleton RD | 0.67 | 0.48 | 0.76 | No |
| Trail Making Task Form A | 0.65 | 0.43 | 0.78 | No |
| SF-36 Vitality Scale | 0.65 | 0.43 | 0.74 | No |
| 25 Foot Walk | 0.64 | 0.50 | 0.77 | No |
| WAIS-III Digit Span Backward | 0.64 | 0.41 | 0.77 | No |
| WAIS-III Digit Span Total | 0.64 | 0.41 | 0.82 | No |
| 10/36 Delayed Recall | 0.63 | 0.42 | 0.74 | No |
| Trail Making Task Form B | 0.62 | 0.33 | 0.76 | No |
| SF-36 Mental Health Scale | 0.62 | 0.38 | 0.62 | No |
| veBOLD | 0.61 | 0.48 | 0.78 | No |
| Selective Reminding Task Delayed | 0.60 | 0.35 | 0.60 | No |
| Symbol-digit Modalities Test | 0.60 | 0.30 | 0.70 | No |
| Number Comparison | 0.59 | 0.36 | 0.68 | No |
| WAIS-III Digit symbol coding | 0.58 | 0.37 | 0.58 | No |
| Skeleton FA | 0.57 | 0.37 | 0.67 | No |
| ven | 0.57 | 0.39 | 0.52 | No |
| Selective Reminding Task Long-term Storage | 0.57 | 0.35 | 0.70 | No |
| Controlled Oral Word Association Test | 0.57 | 0.35 | 0.57 | No |
| 10/36 Immediate Recall | 0.52 | 0.30 | 0.57 | No |
| WAIS-III Digit Span Forward | 0.50 | 0.27 | 0.55 | No |
| Trail Making Task Form B-A | 0.48 | 0.29 | 0.52 | No |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hubbard, N.A.; Sanchez Araujo, Y.; Caballero, C.; Ouyang, M.; Turner, M.P.; Himes, L.; Faghihahmadabadi, S.; Thomas, B.P.; Hart, J.; Huang, H.; et al. Evaluation of Visual-Evoked Cerebral Metabolic Rate of Oxygen as a Diagnostic Marker in Multiple Sclerosis. Brain Sci. 2017, 7, 64. https://doi.org/10.3390/brainsci7060064
Hubbard NA, Sanchez Araujo Y, Caballero C, Ouyang M, Turner MP, Himes L, Faghihahmadabadi S, Thomas BP, Hart J, Huang H, et al. Evaluation of Visual-Evoked Cerebral Metabolic Rate of Oxygen as a Diagnostic Marker in Multiple Sclerosis. Brain Sciences. 2017; 7(6):64. https://doi.org/10.3390/brainsci7060064
Chicago/Turabian StyleHubbard, Nicholas A., Yoel Sanchez Araujo, Camila Caballero, Minhui Ouyang, Monroe P. Turner, Lyndahl Himes, Shawheen Faghihahmadabadi, Binu P. Thomas, John Hart, Hao Huang, and et al. 2017. "Evaluation of Visual-Evoked Cerebral Metabolic Rate of Oxygen as a Diagnostic Marker in Multiple Sclerosis" Brain Sciences 7, no. 6: 64. https://doi.org/10.3390/brainsci7060064
APA StyleHubbard, N. A., Sanchez Araujo, Y., Caballero, C., Ouyang, M., Turner, M. P., Himes, L., Faghihahmadabadi, S., Thomas, B. P., Hart, J., Huang, H., Okuda, D. T., & Rypma, B. (2017). Evaluation of Visual-Evoked Cerebral Metabolic Rate of Oxygen as a Diagnostic Marker in Multiple Sclerosis. Brain Sciences, 7(6), 64. https://doi.org/10.3390/brainsci7060064





