EEG Dynamics of a Go/Nogo Task in Children with ADHD
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
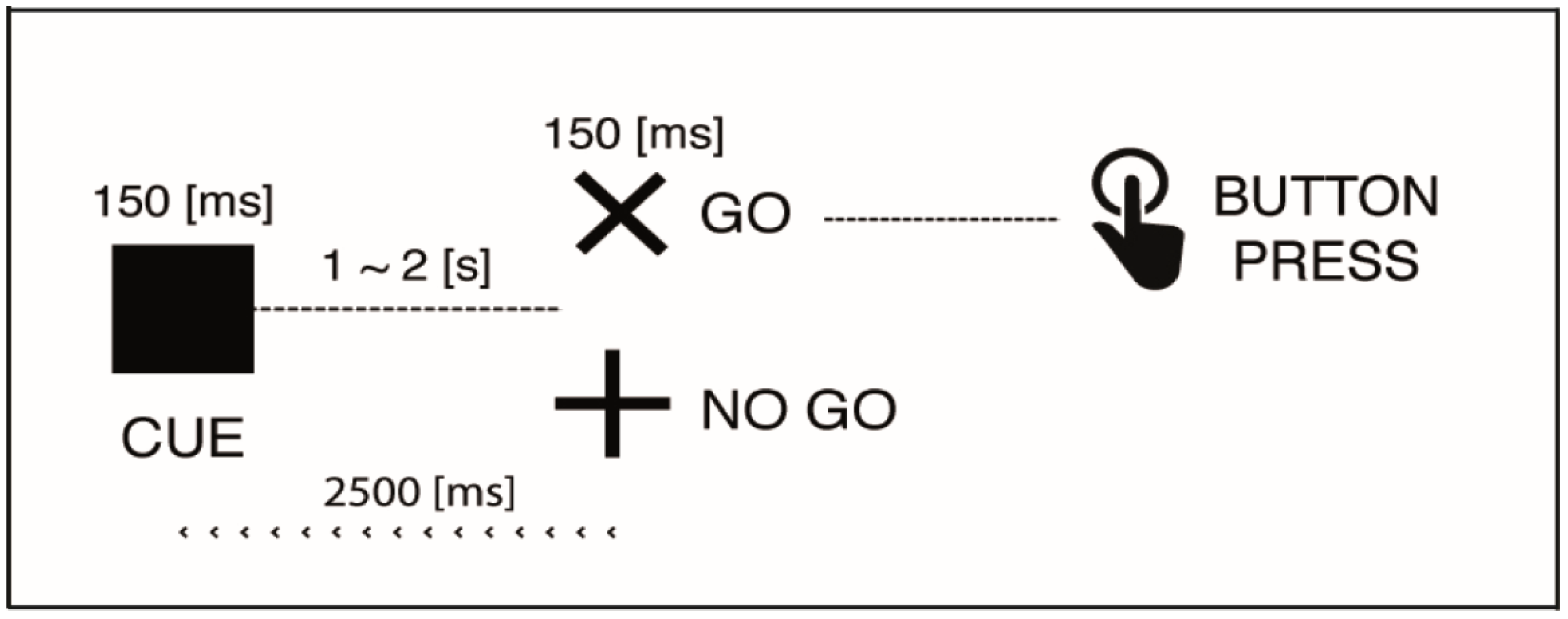
2.2. Stimuli
2.3. Behavioral Analysis
2.4. EEG Recording
2.5. EEG Data Processing
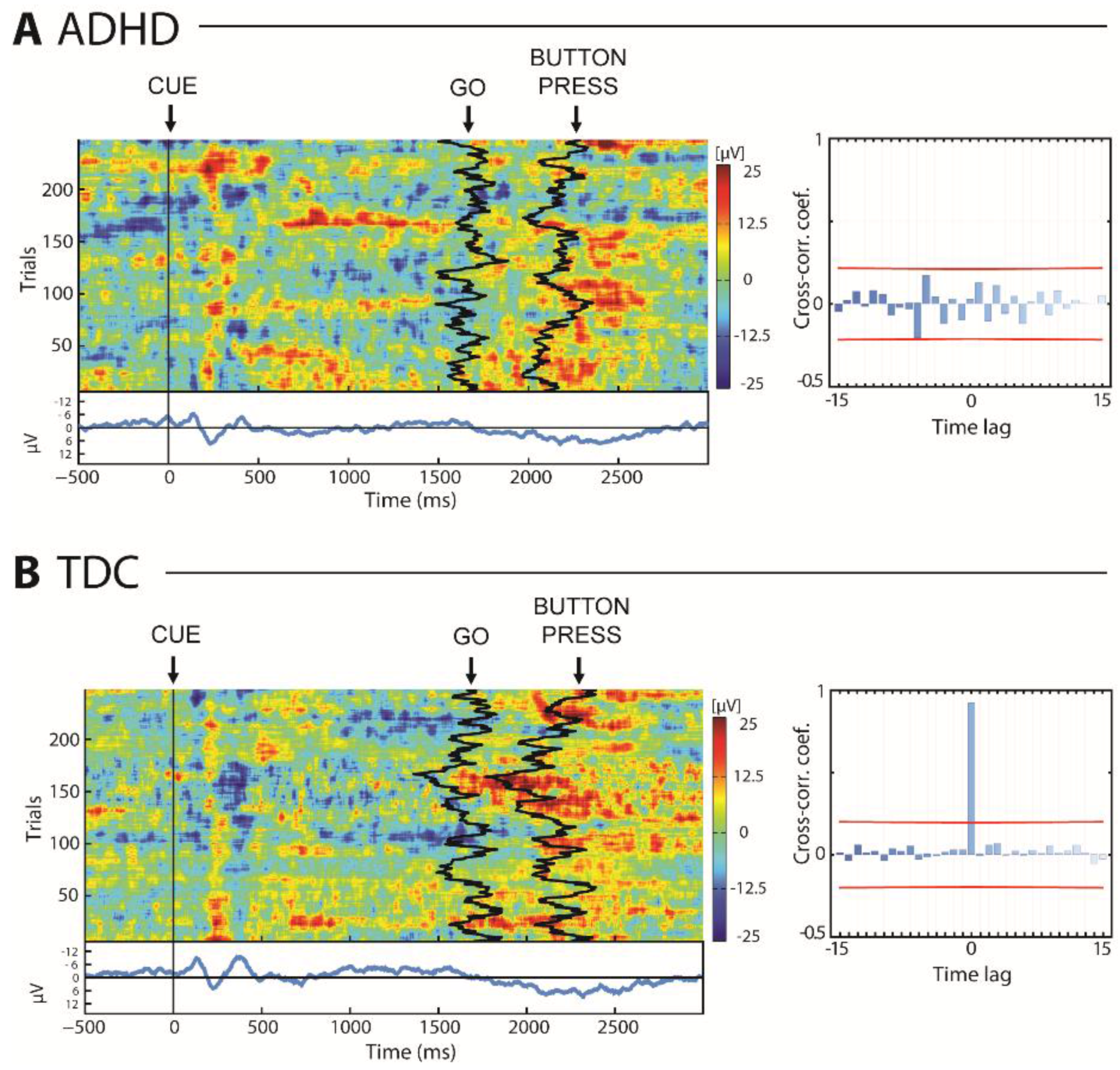
2.6. EEG Data Analysis
3. Results
3.1. Omission, Commission & Reaction Time
3.2. ERP, ERSP & ITC Summarized in Table 2
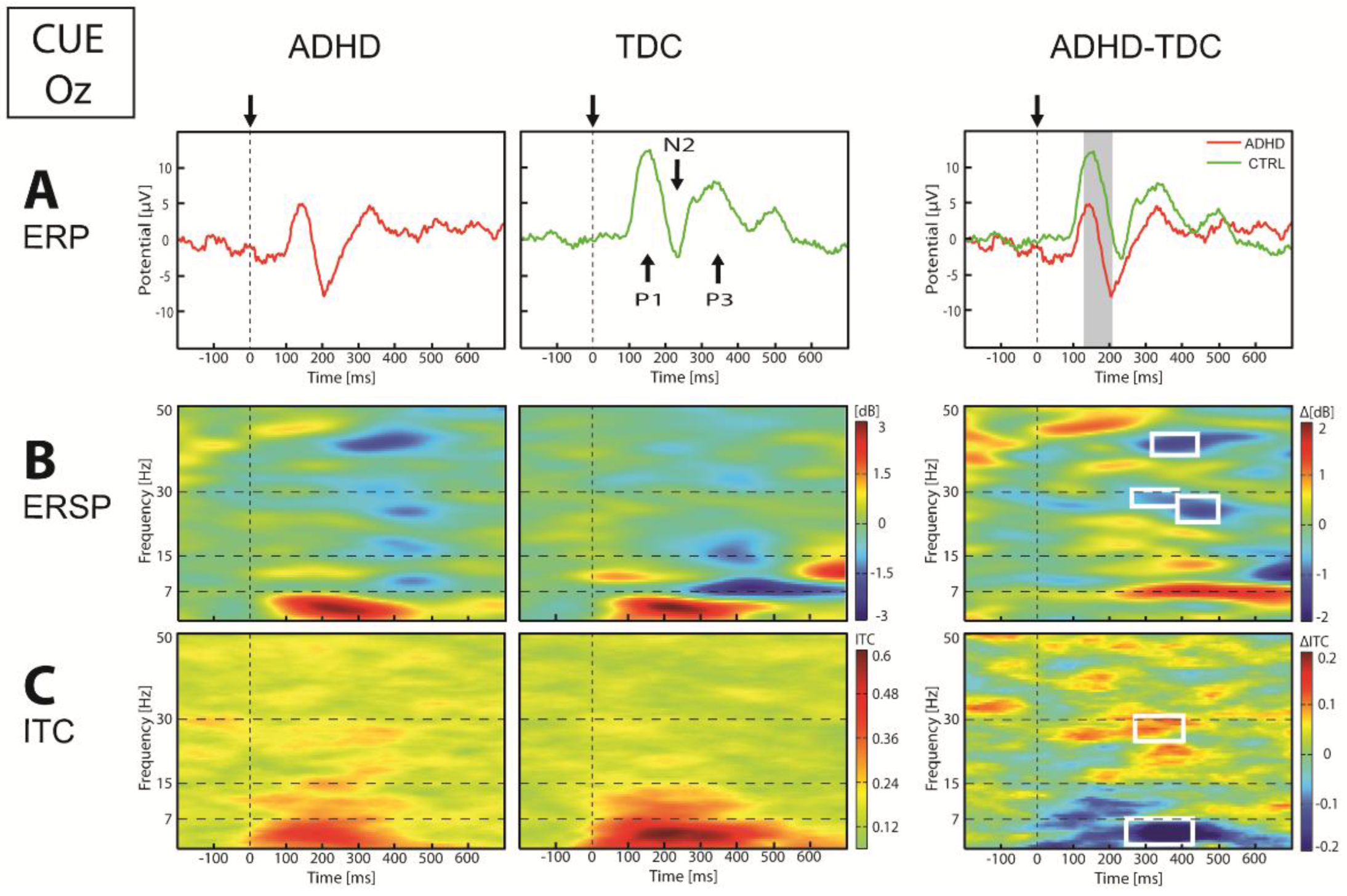
3.2.1. EEG Dynamics Responses to Cue Stimuli
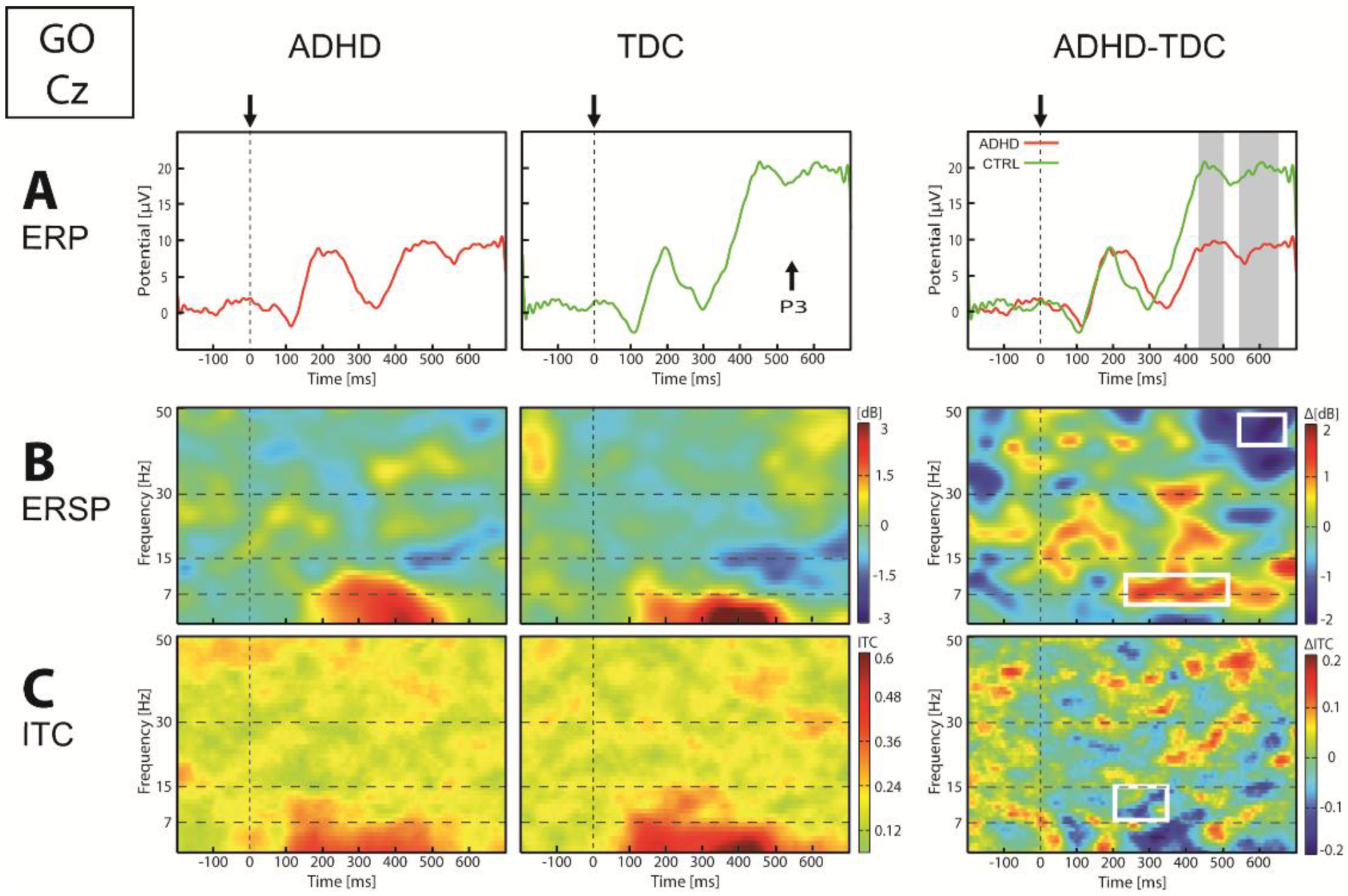
3.2.2. EEG Dynamics Responses to Go Stimuli
3.2.3. EEG Dynamics Responses to Nogo Stimuli
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Polanczyk, G.V.; Willcutt, E.G.; Salum, G.A.; Kieling, C.; Rohde, L.A. ADHD prevalence estimates across three decades: An updated systematic review and meta-regression analysis. Int. J. Epidemiol. 2014, 43, 434–442. [Google Scholar] [CrossRef] [PubMed]
- Matthews, M.; Nigg, J.T.; Fair, D.A. Attention deficit hyperactivity disorder. Curr. Top. Behav. Neurosci. 2014, 16, 235–266. [Google Scholar] [CrossRef] [PubMed]
- Castellanos, F.X.; Sonuga-Barke, E.J.S.; Scheres, A.; di Martino, A.; Hyde, C.; Walters, J.R. Varieties of Attention-Deficit/Hyperactivity Disorder-Related Intra-Individual Variability. Biol. Psychiatry 2005, 57, 1416–1423. [Google Scholar] [CrossRef] [PubMed]
- Nigg, J.T. Is ADHD a disinhibitory disorder? Psychol. Bull. 2001, 127, 571–598. [Google Scholar] [CrossRef] [PubMed]
- Willcutt, E.G.; Doyle, A.E.; Nigg, J.T.; Faraone, S.V.; Pennington, B.F. Validity of the Executive Function Theory of Attention-Deficit/Hyperactivity Disorder: A Meta-Analytic Review. Biol. Psychiatry 2005, 57, 1336–1346. [Google Scholar] [CrossRef] [PubMed]
- Gilger, J.W.; Pennington, B.F.; DeFries, J.C. A twin study of the etiology of comorbidity: Attention-deficit hyperactivity disorder and dyslexia. J. Am. Acad. Child Adolesc. Psychiatry 1992, 31, 343–348. [Google Scholar] [CrossRef] [PubMed]
- Faraone, S.V.; Perlis, R.H.; Doyle, A.E.; Smoller, J.W.; Goralnick, J.J.; Holmgren, M.A.; Sklar, P. Molecular genetics of attention-deficit/hyperactivity disorder. Biol. Psychiatry 2005, 57, 1313–1323. [Google Scholar] [CrossRef] [PubMed]
- Castellanos, F.X.; Sonuga-Barke, E.J.S.; Milham, M.P.; Tannock, R. Characterizing cognition in ADHD: Beyond executive dysfunction. Trends Cogn. Sci. 2006, 10, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Millichap, J.G. Etiologic classification of attention-deficit/hyperactivity disorder. Pediatrics 2008, 121, e358–e365. [Google Scholar] [CrossRef] [PubMed]
- Rubia, K. “Cool” inferior frontostriatal dysfunction in attention-deficit/hyperactivity disorder versus “hot” ventromedial orbitofrontal-limbic dysfunction in conduct disorder: A review. Biol. Psychiatry 2011, 69, e69–e87. [Google Scholar] [CrossRef] [PubMed]
- Seidman, L.J.; Valera, E.M.; Makris, N.; Monuteaux, M.C.; Boriel, D.L.; Kelkar, K.; Kennedy, D.N.; Caviness, V.S.; Bush, G.; Aleardi, M.; et al. Dorsolateral prefrontal and anterior cingulate cortex volumetric abnormalities in adults with attention-deficit/hyperactivity disorder identified by magnetic resonance imaging. Biol. Psychiatry 2006, 60, 1071–1080. [Google Scholar] [CrossRef] [PubMed]
- Sonuga-Barke, E.J.S.; Castellanos, F.X. Spontaneous attentional fluctuations in impaired states and pathological conditions: A neurobiological hypothesis. Neurosci. Biobehav. Rev. 2007, 31, 977–986. [Google Scholar] [CrossRef] [PubMed]
- Bush, G. Attention-Deficit/Hyperactivity Disorder and Attention Networks. Neuropsychopharmacology 2010, 35, 278–300. [Google Scholar] [CrossRef] [PubMed]
- Konrad, K.; Eickhoff, S.B. Is the ADHD brain wired differently? A review on structural and functional connectivity in attention deficit hyperactivity disorder. Hum. Brain Mapp. 2010, 31, 904–916. [Google Scholar] [CrossRef] [PubMed]
- Karch, S.; Thalmeier, T.; Lutz, J.; Cerovecki, A.; Opgen-Rhein, M.; Hock, B.; Leicht, G.; Hennig-Fast, K.; Meindl, T.; Riedel, M.; et al. Neural correlates (ERP/fMRI) of voluntary selection in adult ADHD patients. Eur. Arch. Psychiatry Clin. Neurosci. 2010, 260, 427–440. [Google Scholar] [CrossRef] [PubMed]
- De la Fuente, A.; Xia, S.; Branch, C.; Li, X. A review of attention-deficit/hyperactivity disorder from the perspective of brain networks. Front. Hum. Neurosci. 2013, 7, 192. [Google Scholar] [CrossRef] [PubMed]
- Barry, R.J.; Johnstone, S.J.; Clarke, A.R. A review of electrophysiology in attention-deficit/hyperactivity disorder: II. Event-related potentials. Clin. Neurophysiol. 2003, 114, 184–198. [Google Scholar] [CrossRef]
- Johnstone, S.J.; Barry, R.J.; Clarke, A.R. Ten years on: A follow-up review of ERP research in attention-deficit/hyperactivity disorder. Clin. Neurophysiol. 2013, 124, 644–657. [Google Scholar] [CrossRef] [PubMed]
- Brown, C.R.; Clarke, A.R.; Barry, R.J.; McCarthy, R.; Selikowitz, M.; Magee, C. Event-related potentials in attention-deficit/hyperactivity disorder of the predominantly inattentive type: An investigation of EEG-defined subtypes. Int. J. Psychophysiol. 2005, 58, 94–107. [Google Scholar] [CrossRef] [PubMed]
- Johnstone, S.J.; Barry, R.J.; Markovska, V.; Dimoska, A.; Clarke, A.R. Response inhibition and interference control in children with AD/HD: A visual ERP investigation. Int. J. Psychophysiol. 2009, 72, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, C.A.; Barry, R.J.; Clarke, A.R.; Johnstone, S.J.; McCarthy, R.; Selikowitz, M.; Broyd, S.J. Methylphenidate effects in attention deficit/hyperactivity disorder: Electrodermal and ERP measures during a continuous performance task. Psychopharmacology 2005, 183, 81–91. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.L.; Johnstone, S.J.; Barry, R.J. Inhibitory processing during the Go/NoGo task: An ERP analysis of children with attention-deficit/hyperactivity disorder. Clin. Neurophysiol. 2004, 115, 1320–1331. [Google Scholar] [CrossRef] [PubMed]
- Delorme, A.; Makeig, S. EEGLAB: An open source toolbox for analysis of single-trial EEG dynamics including independent component analysis. J. Neurosci. Methods 2004, 134, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Delorme, A.; Miyakoshi, M.; Jung, T.-P.; Makeig, S. Grand average ERP-image plotting and statistics: A method for comparing variability in event-related single-trial EEG activities across subjects and conditions. J. Neurosci. Methods 2015, 250, 3–6. [Google Scholar] [CrossRef] [PubMed]
- Loo, S.K.; Makeig, S. Clinical utility of EEG in attention-deficit/hyperactivity disorder: A research update. Neurotherapeutics 2012, 9, 569–587. [Google Scholar] [CrossRef] [PubMed]
- Yordanova, J.; Banaschewski, T.; Kolev, V.; Woerner, W.; Rothenberger, A. Abnormal early stages of task stimulus processing in children with attention-deficit hyperactivity disorder—Evidence from event-related gamma oscillations. Clin. Neurophysiol. 2001, 112, 1096–1108. [Google Scholar] [CrossRef]
- Johnstone, S.J.; Barry, R.J.; Dimoska, A. Event-related slow-wave activity in two subtypes of attention-deficit/hyperactivity disorder. Clin. Neurophysiol. 2003, 114, 504–514. [Google Scholar] [CrossRef]
- Yordanova, J.; Heinrich, H.; Kolev, V.; Rothenberger, A. Increased event-related theta activity as a psychophysiological marker of comorbidity in children with tics and attention-deficit/hyperactivity disorders. NeuroImage 2006, 32, 940–955. [Google Scholar] [CrossRef] [PubMed]
- Yordanova, J.; Kolev, V.; Rothenberger, A. Event-related oscillations reflect functional asymmetry in children with attention deficit/hyperactivity disorder. Suppl. Clin. Neurophysiol. 2013, 62, 289–301. [Google Scholar] [PubMed]
- Alexander, D.M.; Hermens, D.F.; Keage, H.A.D.; Clark, C.R.; Williams, L.M.; Kohn, M.R.; Clarke, S.D.; Lamb, C.; Gordon, E. Event-related wave activity in the EEG provides new marker of ADHD. Clin. Neurophysiol. 2008, 119, 163–179. [Google Scholar] [CrossRef] [PubMed]
- Loo, S.K.; Smalley, S.L. Preliminary report of familial clustering of EEG measures in ADHD. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2008, 147B, 107–109. [Google Scholar] [CrossRef] [PubMed]
- Lenz, D.; Krauel, K.; Schadow, J.; Baving, L.; Duzel, E.; Herrmann, C.S. Enhanced gamma-band activity in ADHD patients lacks correlation with memory performance found in healthy children. Brain Res. 2008, 1235, 117–132. [Google Scholar] [CrossRef] [PubMed]
- Lenz, D.; Krauel, K.; Flechtner, H.-H.; Schadow, J.; Hinrichs, H.; Herrmann, C.S. Altered evoked gamma-band responses reveal impaired early visual processing in ADHD children. Neuropsychologia 2010, 48, 1985–1993. [Google Scholar] [CrossRef] [PubMed]
- Nazari, M.A.; Wallois, F.; Aarabi, A.; Berquin, P. Dynamic changes in quantitative electroencephalogram during continuous performance test in children with attention-deficit/hyperactivity disorder. Int. J. Psychophysiol. 2011, 81, 230–236. [Google Scholar] [CrossRef] [PubMed]
- Lenartowicz, A.; Delorme, A.; Walshaw, P.D.; Cho, A.L.; Bilder, R.M.; McGough, J.J.; McCracken, J.T.; Makeig, S.; Loo, S.K. Electroencephalography correlates of spatial working memory deficits in attention-deficit/hyperactivity disorder: Vigilance, encoding, and maintenance. J. Neurosci. 2014, 34, 1171–1182. [Google Scholar] [CrossRef] [PubMed]
- Sridhar, C.; Bhat, S.; Acharya, U.R.; Adeli, H.; Bairy, G.M. Diagnosis of attention deficit hyperactivity disorder using imaging and signal processing techniques. Comput. Biol. Med. 2017, 88, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Rommel, A.-S.; James, S.-N.; McLoughlin, G.; Brandeis, D.; Banaschewski, T.; Asherson, P.; Kuntsi, J. Altered EEG spectral power during rest and cognitive performance: A comparison of preterm-born adolescents to adolescents with ADHD. Eur. Child Adolesc. Psychiatry 2017, 26, 1511–1522. [Google Scholar] [CrossRef] [PubMed]
- Öztoprak, H.; Toycan, M.; Alp, Y.K.; Arıkan, O.; Doğutepe, E.; Karakaş, S. Machine-based classification of ADHD and nonADHD participants using time/frequency features of event-related neuroelectric activity. Clin. Neurophysiol. 2017, 128, 2400–2410. [Google Scholar] [CrossRef] [PubMed]
- Auerbach, J.G.; Lerner, Y. Syndromes derived from the Child Behavior Checklist for clinically referred Israeli boys aged 6-11: A research note. J. Child Psychol. Psychiatry 1991, 32, 1017–1024. [Google Scholar] [CrossRef] [PubMed]
- World Medical Association. World medical association declaration of Helsinki: Ethical principles for medical research involving human subjects. JAMA 2013, 310, 2191–2194. [Google Scholar] [CrossRef]
- Baijot, S.; Slama, H.; Söderlund, G.; Dan, B.; Deltenre, P.; Colin, C.; Deconinck, N. Neuropsychological and neurophysiological benefits from white noise in children with and without ADHD. Behav. Brain Funct. 2016, 12, 11. [Google Scholar] [CrossRef] [PubMed]
- Drechsler, R.; Brandeis, D.; Földényi, M.; Imhof, K.; Steinhausen, H.-C. The course of neuropsychological functions in children with attention deficit hyperactivity disorder from late childhood to early adolescence. J. Child Psychol. Psychiatry 2005, 46, 824–836. [Google Scholar] [CrossRef] [PubMed]
- Hendricks, W.A.; Robey, K.W. The Sampling Distribution of the Coefficient of Variation. Ann. Math. Stat. 1936, 7, 129–132. [Google Scholar] [CrossRef]
- Brunner, C.; Delorme, A.; Makeig, S. Eeglab—An Open Source Matlab Toolbox for Electrophysiological Research. Biomed. Technol. 2013. [Google Scholar] [CrossRef] [PubMed]
- Cheron, G.; Leroy, A.; Palmero-Soler, E.; de Saedeleer, C.; Bengoetxea, A.; Cebolla, A.-M.; Vidal, M.; Dan, B.; Berthoz, A.; McIntyre, J. Gravity influences top-down signals in visual processing. PLoS ONE 2014, 9, e82371. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, S.; Jadhav, P.; Adapa, B.; Acharyya, A.; Naik, G.R. Online and automated reliable system design to remove blink and muscle artefact in EEG. Conf. Proc. Annu. Int. Conf. IEEE Eng. Med. Biol. Soc. 2015, 2015, 6784–6787. [Google Scholar] [CrossRef]
- Feis, R.A.; Smith, S.M.; Filippini, N.; Douaud, G.; Dopper, E.G.P.; Heise, V.; Trachtenberg, A.J.; van Swieten, J.C.; van Buchem, M.A.; Rombouts, S.A.R.B.; et al. ICA-based artifact removal diminishes scan site differences in multi-center resting-state Fmri. Front. Neurosci. 2015, 9, 395. [Google Scholar] [CrossRef] [PubMed]
- Jadhav, P.N.; Shanamugan, D.; Chourasia, A.; Ghole, A.R.; Acharyya, A.; Naik, G. Automated detection and correction of eye blink and muscular artefacts in EEG signal for analysis of Autism Spectrum Disorder. Conf. Proc. Annu. Int. Conf. IEEE Eng. Med. Biol. Soc. 2014, 2014, 1881–1884. [Google Scholar] [CrossRef]
- Makeig, S. Auditory event-related dynamics of the EEG spectrum and effects of exposure to tones. Electroencephalogr. Clin. Neurophysiol. 1993, 86, 283–293. [Google Scholar] [CrossRef]
- Pfurtscheller, G.; Neuper, C. Event-related synchronization of mu rhythm in the EEG over the cortical hand area in man. Neurosci. Lett. 1994, 174, 93–96. [Google Scholar] [CrossRef]
- Tallon-Baudry, C.; Bertrand, O.; Delpuech, C.; Pernier, J. Stimulus specificity of phase-locked and non-phase-locked 40 Hz visual responses in human. J. Neurosci. 1996, 16, 4240–4249. [Google Scholar] [PubMed]
- Benjamini, Y.; Hochberg, Y. Controlling the False Discovery Rate: A Practical and Powerful Approach to Multiple Testing. J. R. Stat. Soc. Ser. B Methodol. 1995, 57, 289–300. [Google Scholar]
- Banaschewski, T.; Brandeis, D.; Heinrich, H.; Albrecht, B.; Brunner, E.; Rothenberger, A. Association of ADHD and conduct disorder–brain electrical evidence for the existence of a distinct subtype. J. Child Psychol. Psychiatry 2003, 44, 356–376. [Google Scholar] [CrossRef] [PubMed]
- Perchet, C.; Revol, O.; Fourneret, P.; Mauguière, F.; Garcia-Larrea, L. Attention shifts and anticipatory mechanisms in hyperactive children: An ERP study using the Posner paradigm. Biol. Psychiatry 2001, 50, 44–57. [Google Scholar] [CrossRef]
- Barry, R.J.; Clarke, A.R.; Johnstone, S.J.; McCarthy, R.; Selikowitz, M. Electroencephalogram theta/beta ratio and arousal in attention-deficit/hyperactivity disorder: Evidence of independent processes. Biol. Psychiatry 2009, 66, 398–401. [Google Scholar] [CrossRef] [PubMed]
- Arns, M.; Conners, C.K.; Kraemer, H.C. A decade of EEG Theta/Beta Ratio Research in ADHD: A meta-analysis. J. Atten. Disord. 2013, 17, 374–383. [Google Scholar] [CrossRef] [PubMed]
- Sergeant, J. The cognitive-energetic model: An empirical approach to attention-deficit hyperactivity disorder. Neurosci. Biobehav. Rev. 2000, 24, 7–12. [Google Scholar] [CrossRef]
- Fries, P. A mechanism for cognitive dynamics: Neuronal communication through neuronal coherence. Trends Cogn. Sci. 2005, 9, 474–480. [Google Scholar] [CrossRef] [PubMed]
- Salti, M.; Monto, S.; Charles, L.; King, J.-R.; Parkkonen, L.; Dehaene, S. Distinct cortical codes and temporal dynamics for conscious and unconscious percepts. eLife 2015, 4. [Google Scholar] [CrossRef] [PubMed]
- Casey, B.J.; Castellanos, F.X.; Giedd, J.N.; Marsh, W.L.; Hamburger, S.D.; Schubert, A.B.; Vauss, Y.C.; Vaituzis, A.C.; Dickstein, D.P.; Sarfatti, S.E.; et al. Implication of right frontostriatal circuitry in response inhibition and attention-deficit/hyperactivity disorder. J. Am. Acad. Child Adolesc. Psychiatry 1997, 36, 374–383. [Google Scholar] [CrossRef] [PubMed]
- Banaschewski, T.; Brandeis, D.; Heinrich, H.; Albrecht, B.; Brunner, E.; Rothenberger, A. Questioning inhibitory control as the specific deficit of ADHD—Evidence from brain electrical activity. J. Neural Transm. 2004, 111, 841–864. [Google Scholar] [CrossRef] [PubMed]
- Casey, B.J.; Trainor, R.J.; Orendi, J.L.; Schubert, A.B.; Nystrom, L.E.; Giedd, J.N.; Castellanos, F.X.; Haxby, J.V.; Noll, D.C.; Cohen, J.D.; et al. A Developmental Functional MRI Study of Prefrontal Activation during Performance of a Go-No-Go Task. J. Cogn. Neurosci. 1997, 9, 835–847. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.B.; Taylor, E.; Brammer, M.; Toone, B.; Rubia, K. Task-specific hypoactivation in prefrontal and temporoparietal brain regions during motor inhibition and task switching in medication-naive children and adolescents with attention deficit hyperactivity disorder. Am. J. Psychiatry 2006, 163, 1044–1051. [Google Scholar] [CrossRef] [PubMed]
- Fries, P.; Neuenschwander, S.; Engel, A.K.; Goebel, R.; Singer, W. Rapid feature selective neuronal synchronization through correlated latency shifting. Nat. Neurosci. 2001, 4, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Gregoriou, G.G.; Gotts, S.J.; Zhou, H.; Desimone, R. Long-range neural coupling through synchronization with attention. Prog. Brain Res. 2009, 176, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Dipoppa, M.; Gutkin, B.S. Correlations in background activity control persistent state stability and allow execution of working memory tasks. Front. Comput. Neurosci. 2013, 7, 139. [Google Scholar] [CrossRef] [PubMed]
- Harris, K.D.; Csicsvari, J.; Hirase, H.; Dragoi, G.; Buzsáki, G. Organization of cell assemblies in the hippocampus. Nature 2003, 424, 552–556. [Google Scholar] [CrossRef] [PubMed]
- Demiralp, T.; Bayraktaroglu, Z.; Lenz, D.; Junge, S.; Busch, N.A.; Maess, B.; Ergen, M.; Herrmann, C.S. Gamma amplitudes are coupled to theta phase in human EEG during visual perception. Int. J. Psychophysiol. 2007, 64, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Park, J.Y.; Lee, Y.-R.; Lee, J. The relationship between theta-gamma coupling and spatial memory ability in older adults. Neurosci. Lett. 2011, 498, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Tort, A.B.L.; Komorowski, R.W.; Manns, J.R.; Kopell, N.J.; Eichenbaum, H. Theta-gamma coupling increases during the learning of item-context associations. Proc. Natl. Acad. Sci. USA 2009, 106, 20942–20947. [Google Scholar] [CrossRef] [PubMed]
- Nakatani, C.; Raffone, A.; van Leeuwen, C. Efficiency of conscious access improves with coupling of slow and fast neural oscillations. J. Cogn. Neurosci. 2014, 26, 1168–1179. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.W.; Lee, J.; Kim, B.-N.; Kang, T.; Min, K.J.; Han, D.H.; Lee, Y.S. Theta-phase gamma-amplitude coupling as a neurophysiological marker of attention deficit/hyperactivity disorder in children. Neurosci. Lett. 2015, 603, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.W.; Lee, J.; Kim, H.-J.; Lee, Y.S.; Min, K.J. Relationship between theta-phase gamma-amplitude coupling and attention-deficit/hyperactivity behavior in children. Neurosci. Lett. 2015, 590, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Silberstein, R.B.; Pipingas, A.; Farrow, M.; Levy, F.; Stough, C.K.; Camfield, D.A. Brain functional connectivity abnormalities in attention-deficit hyperactivity disorder. Brain Behav. 2016, 6, e00583. [Google Scholar] [CrossRef] [PubMed]
- Silberstein, R.B.; Pipingas, A.; Farrow, M.; Levy, F.; Stough, C.K. Dopaminergic modulation of default mode network brain functional connectivity in attention deficit hyperactivity disorder. Brain Behav. 2016, 6, e00582. [Google Scholar] [CrossRef] [PubMed]
- Silberstein, R.B.; Levy, F.; Pipingas, A.; Farrow, M. First-Dose Methylphenidate-Induced Changes in Brain Functional Connectivity Are Correlated with 3-Month Attention-Deficit/Hyperactivity Disorder Symptom Response. Biol. Psychiatry 2017, 82, 679–686. [Google Scholar] [CrossRef] [PubMed]
- Klimesch, W. EEG alpha and theta oscillations reflect cognitive and memory performance: A review and analysis. Brain Res. Rev. 1999, 29, 169–195. [Google Scholar] [CrossRef]
- Klimesch, W. α-band oscillations, attention, and controlled access to stored information. Trends Cogn. Sci. 2012, 16, 606–617. [Google Scholar] [CrossRef] [PubMed]
- Klimesch, W.; Doppelmayr, M.; Schimke, H.; Pachinger, T. Alpha frequency, reaction time, and the speed of processing information. J. Clin. Neurophysiol. 1996, 13, 511–518. [Google Scholar] [CrossRef] [PubMed]
- Klimesch, W.; Sauseng, P.; Gerloff, C. Enhancing cognitive performance with repetitive transcranial magnetic stimulation at human individual alpha frequency. Eur. J. Neurosci. 2003, 17, 1129–1133. [Google Scholar] [CrossRef] [PubMed]
- Klimesch, W.; Hanslmayr, S.; Sauseng, P.; Gruber, W.R.; Doppelmayr, M. P1 and traveling alpha waves: Evidence for evoked oscillations. J. Neurophysiol. 2007, 97, 1311–1318. [Google Scholar] [CrossRef] [PubMed]
- Cheron, G.; Leroy, A.; de Saedeleer, C.; Bengoetxea, A.; Lipshits, M.; Cebolla, A.; Servais, L.; Dan, B.; Berthoz, A.; McIntyre, J. Effect of gravity on human spontaneous 10-Hz electroencephalographic oscillations during the arrest reaction. Brain Res. 2006, 1121, 104–116. [Google Scholar] [CrossRef] [PubMed]
- Haegens, S.; Osipova, D.; Oostenveld, R.; Jensen, O. Somatosensory working memory performance in humans depends on both engagement and disengagement of regions in a distributed network. Hum. Brain Mapp. 2010, 31, 26–35. [Google Scholar] [CrossRef] [PubMed]
- Sadaghiani, S.; Scheeringa, R.; Lehongre, K.; Morillon, B.; Giraud, A.-L.; D’Esposito, M.; Kleinschmidt, A. α-band phase synchrony is related to activity in the fronto-parietal adaptive control network. J. Neurosci. 2012, 32, 14305–14310. [Google Scholar] [CrossRef] [PubMed]
- Gruber, W.R.; Zauner, A.; Lechinger, J.; Schabus, M.; Kutil, R.; Klimesch, W. Alpha phase, temporal attention, and the generation of early event related potentials. NeuroImage 2014, 103, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Börger, N.; van der Meere, J. Visual behaviour of ADHD children during an attention test: An almost forgotten variable. Attention-Deficit Hyperactivity Disorder. J. Child Psychol. Psychiatry 2000, 41, 525–532. [Google Scholar] [CrossRef] [PubMed]
- Kofler, M.J.; Rapport, M.D.; Sarver, D.E.; Raiker, J.S.; Orban, S.A.; Friedman, L.M.; Kolomeyer, E.G. Reaction time variability in ADHD: A meta-analytic review of 319 studies. Clin. Psychol. Rev. 2013, 33, 795–811. [Google Scholar] [CrossRef] [PubMed]
- Wiersema, R.; van der Meere, J.; Roeyers, H.; van Coster, R.; Baeyens, D. Event rate and event-related potentials in ADHD. J. Child Psychol. Psychiatry 2006, 47, 560–567. [Google Scholar] [CrossRef] [PubMed]
| W | p | p a | |
|---|---|---|---|
| Full-scale QI | 21.50 | 0.747 | |
| Affective Problems | 30.00 | 0.007 | 0.04 |
| Anxiety Problems | 20.00 | 0.407 | 1 |
| Somatic Problems | 14.50 | 1.000 | 1 |
| ADHD Problems | 30.00 | 0.005 | 0.03 |
| Oppositional Defiant Problems | 28.50 | 0.017 | 0.11 |
| Conduct Problems | 28.50 | 0.09 | 0.11 |
| ADHD–TDC | ||||||
|---|---|---|---|---|---|---|
| Stim. | Regions | Measure | Comp./Freq. | Time (ms) | Electrodes | Sig. 1 |
| Cue | Posterior Figure 3 | ERP | ↓P1-N2 | 115–205 | Oz, O1, O2 | p < 0.009 |
| ↑N2 | 170–191 | Pz, P3 | p < 0.045 | |||
| ERSP | ↓β (27–32) | 238–485 | Oz, O1, O2, P4 | p < 0.044 | ||
| ↓γ (40–43) | 332–427 | Oz, O2, O1 | p < 0.049 | |||
| ITC | ↓θ (4–8) | 261–425 | Oz, O1, O2, Pz, P3, P4 | p < 0.039 | ||
| ↑β (25–30) | 273–422 | Oz, O1, P4, P3 | p < 0.044 | |||
| Anterior | ERP | ↓N1 | 121–161 | Fz, F3, F4, Fpz | p < 0.018 | |
| ↑P2 | 223–273 | Fz, F3, F4, Fpz | p < 0.019 | |||
| ERSP | ↓α (9–12) | 150–312 | Fz, F3, F4 | p < 0.035 | ||
| ITC | ↓α (10–13) | 101–263 | Fz, F4 | p < 0.049 | ||
| Go | Posterior | ERP | ↓P1-N2 | 146–189 | Oz, O1, O2, Pz, P3 | p < 0.022 |
| ↓P3 | 345–439 | Oz, O1, O2, Pz, P3 | p < 0.044 | |||
| ERSP | ↓θ (4–7) | 302–390 | Oz, O1, O2, P3, P4 | p < 0.034 | ||
| ↓γ (42–47) | 570–679 | Oz, O1, O2, P3, P4 | p < 0.032 | |||
| ITC | ↓θ-α (5–12) | 320–453 | Oz, O1, O2, Pz, P3 | p < 0.041 | ||
| Central Figure 4 | ERP | ↓P3 | 464–656 | Cz, C3 | p < 0.012 | |
| ERSP | ↑α (7–13) | 252–541 | Cz, C3, C4 | p < 0.031 | ||
| ↓γ (42–47) | 566–593 | Cz, C4 | p < 0.032 | |||
| ITC | ↓α (8–14) | 220–347 | Cz, C3, C4 | p < 0.018 | ||
| Anterior | ERP | ↓N1 | 117–128 | Fz, F3, F4 | p < 0.044 | |
| ↑P2 | 207–260 | Fz, F3, F4 | p < 0.011 | |||
| ERSP | ↓α (10–18) | 109–234 | Fz, F3, F4 | p < 0.003 | ||
| ↓γ (38–47) | 517–654 | Fz, F4 | p < 0.032 | |||
| Nogo | Posterior | ERP | ↓P1, ↑N2 | 136–209 | Oz, O1, O2, Pz, P3 | p < 0.005 |
| ERSP | ↑α (12–16) | 126–273 | Oz, O1, O2, Pz, P3, P4 | p < 0.003 | ||
| ↓γ (31–36) | 296–380 | Oz, O1, O2, Pz, P3, P4 | p < 0.018 | |||
| ITC | ↓θ-α (5–14) | 308–414 | Oz, O1, O2, P3 | p < 0.005 | ||
| ↑γ (31–36) | 302–410 | Oz, O2, Pz, P3, P4 | p < 0.007 | |||
| Central | ERSP | ↓θ (4–8) | 255–423 | Cz, C3 | p < 0.002 | |
| ↓γ (38–43) | 428–509 | Cz, C3, C4 | p < 0.048 | |||
| ITC | ↓θ (4–8) | 291–369 | Cz, C3 | p < 0.016 | ||
| ↑γ (31–37) | 275–345 | Cz, C3, C4 | p < 0.041 | |||
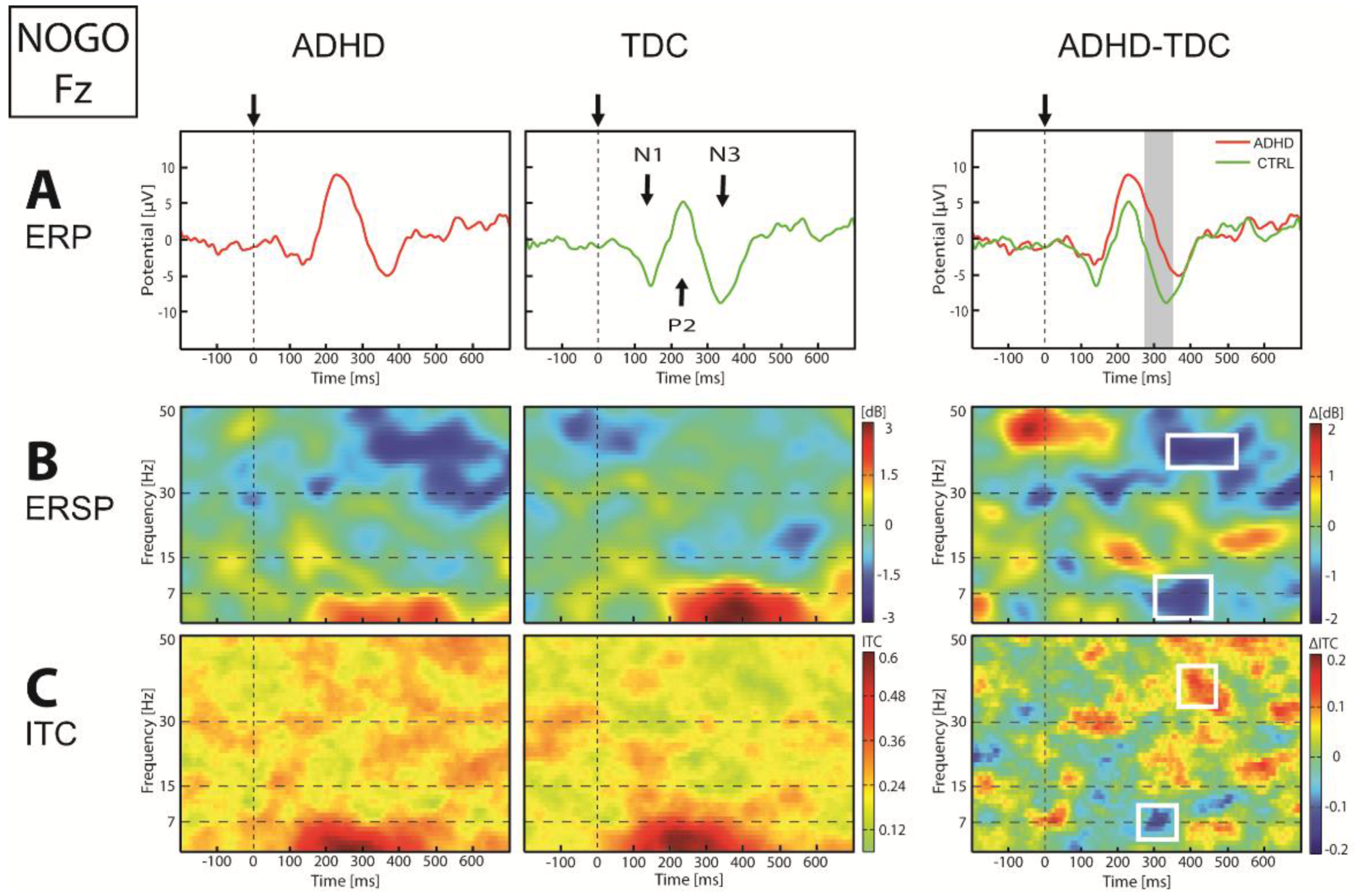
| Anterior Figure 5 | ERP | ↓N1-P2 | 136–211 | F3, F4 | p < 0.049 | |
| ↓N3 | 279–344 | Fz, F3, F4, Fpz | p < 0.018 | |||
| ERSP | ↓θ-α (4–11) | 277–396 | Fz, F3, F4 | p < 0.014 | ||
| ↓γ (38–45) | 316–517 | Fz, F3, F4, Fpz | p < 0.022 | |||
| ITC | ↓θ-α (5–11) | 256–390 | Fz, F3 | p < 0.016 | ||
| ↑γ (34–44) | 382–511 | Fz, F3, F4 | p < 0.003 | |||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baijot, S.; Cevallos, C.; Zarka, D.; Leroy, A.; Slama, H.; Colin, C.; Deconinck, N.; Dan, B.; Cheron, G. EEG Dynamics of a Go/Nogo Task in Children with ADHD. Brain Sci. 2017, 7, 167. https://doi.org/10.3390/brainsci7120167
Baijot S, Cevallos C, Zarka D, Leroy A, Slama H, Colin C, Deconinck N, Dan B, Cheron G. EEG Dynamics of a Go/Nogo Task in Children with ADHD. Brain Sciences. 2017; 7(12):167. https://doi.org/10.3390/brainsci7120167
Chicago/Turabian StyleBaijot, Simon, Carlos Cevallos, David Zarka, Axelle Leroy, Hichem Slama, Cecile Colin, Nicolas Deconinck, Bernard Dan, and Guy Cheron. 2017. "EEG Dynamics of a Go/Nogo Task in Children with ADHD" Brain Sciences 7, no. 12: 167. https://doi.org/10.3390/brainsci7120167
APA StyleBaijot, S., Cevallos, C., Zarka, D., Leroy, A., Slama, H., Colin, C., Deconinck, N., Dan, B., & Cheron, G. (2017). EEG Dynamics of a Go/Nogo Task in Children with ADHD. Brain Sciences, 7(12), 167. https://doi.org/10.3390/brainsci7120167






