Acute Stress Dysregulates the LPP ERP Response to Emotional Pictures and Impairs Sustained Attention: Time-Sensitive Effects
Abstract
:1. Introduction
2. Method
2.1. Participants
2.2. Procedure

2.3. Stimuli and Physiological Recording
2.3.1. SECPT and Control
2.3.2. Salivary Cortisol Collection
2.3.3. Emotional Stimuli
2.3.4. Electrophysiological Recordings
2.3.5. SART Performance
2.3.6. Statistical Analyses
3. Results
3.1. Cortisol
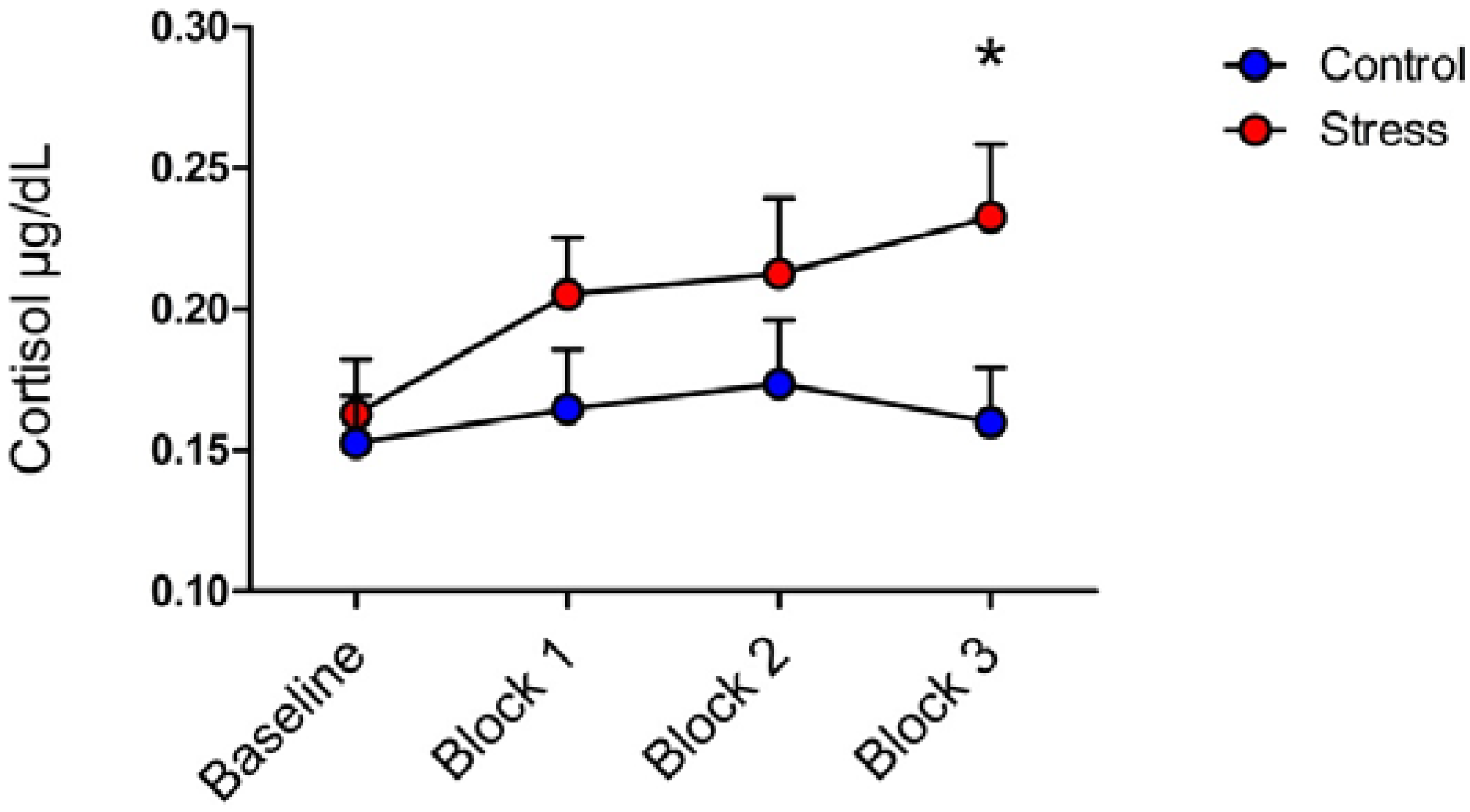
3.2. LPP and Picture Analyses
3.3. Picture Rating
3.4. SART
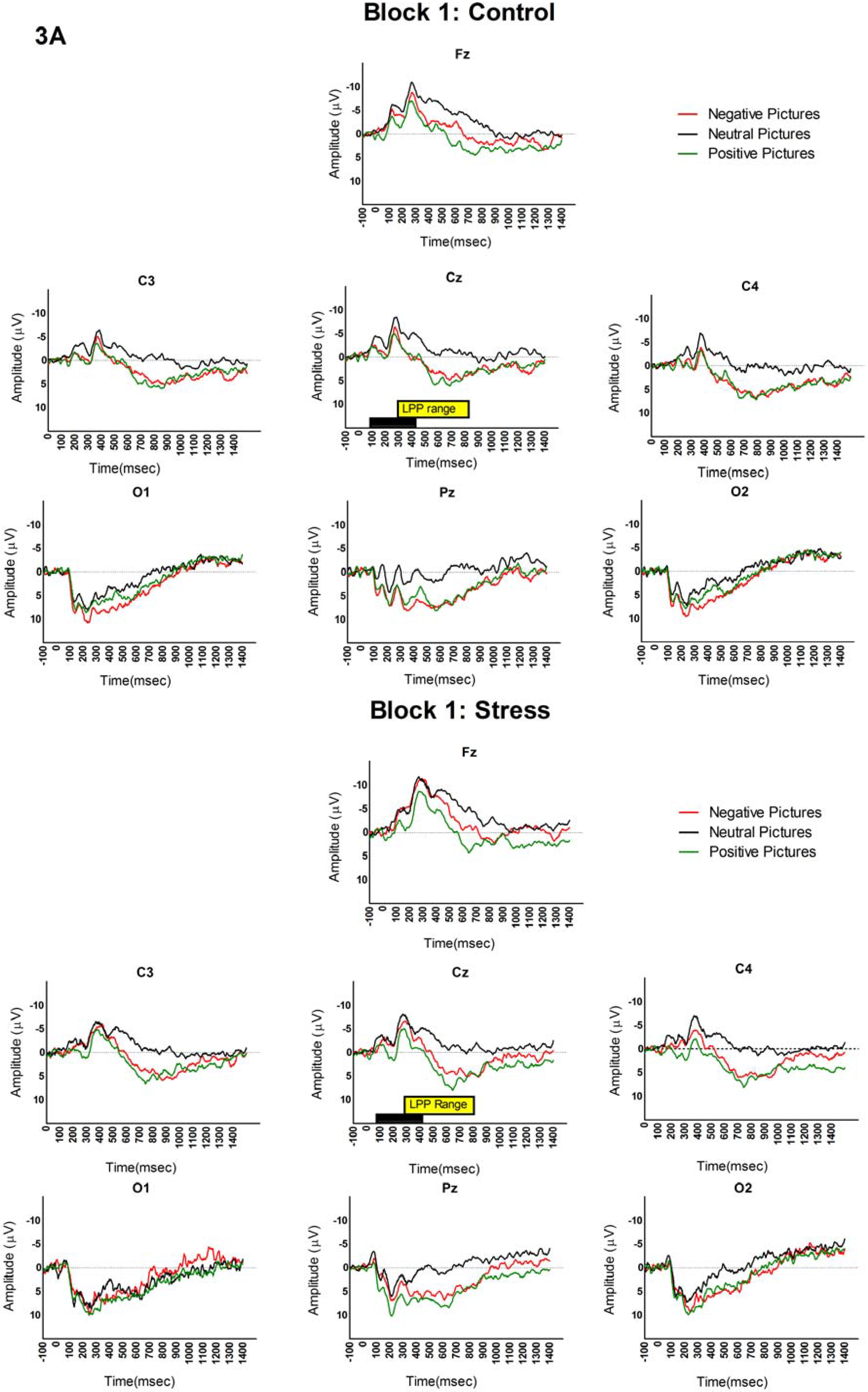
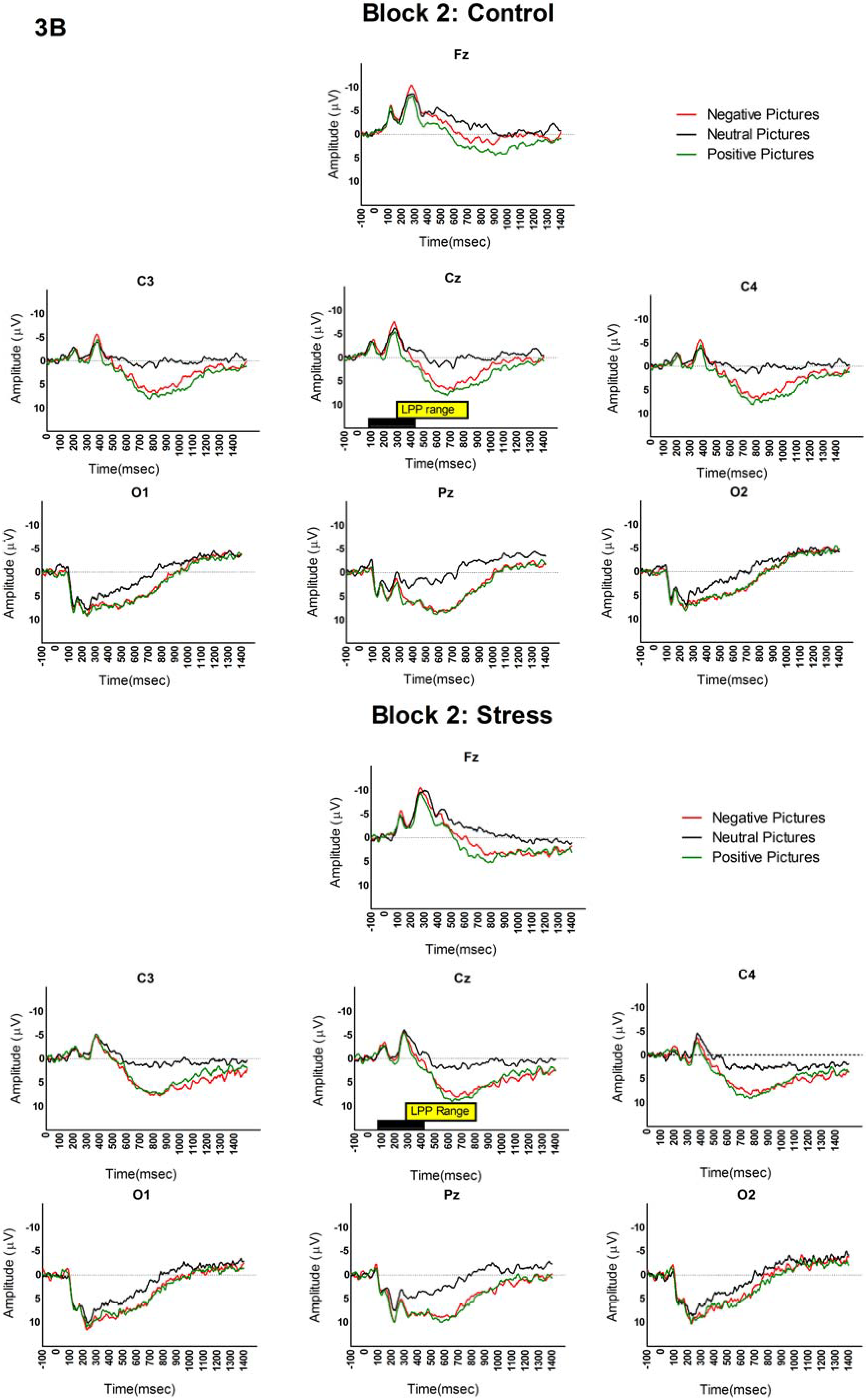
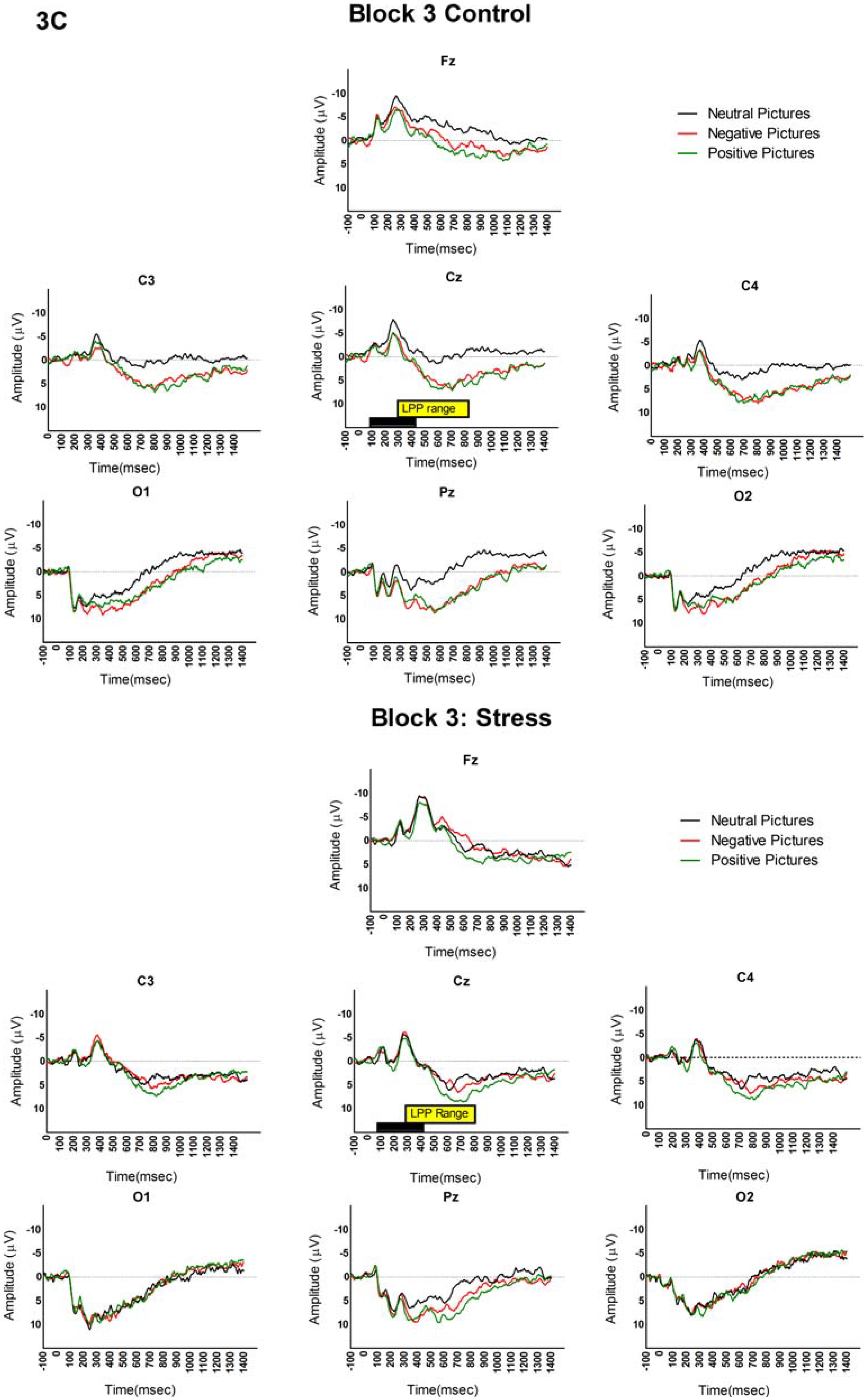

4. Discussion
5. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Desimone, R.; Duncan, J. Neural mechanisms of selective visual attention. Annu. Rev. Neurosci. 1995, 18, 193–222. [Google Scholar] [CrossRef] [PubMed]
- Mogg, K.; Bradley, B.P.; de Bono, J.; Painter, M. Time course of attentional bias for threat information in non-clinical anxiety. Behav. Res. Ther. 1997, 35, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Olofsson, J.K.; Nordin, S.; Sequeira, H.; Polich, J. Affective picture processing: An integrative review of ERP findings. Biol. Psychol. 2008, 77, 247–265. [Google Scholar] [CrossRef] [PubMed]
- Schupp, H.T.; Flaisch, T.; Stockburger, J.; Junghöfer, M. Emotion and attention: Event-related brain potential studies. Prog. Brain Res. 2006, 156, 31–51. [Google Scholar] [PubMed]
- Veer, I.M.; Oei, N.Y.; Spinhoven, P.; van Buchem, M.A.; Elzinga, B.M.; Rombouts, S.A. Beyond acute social stress: Increased functional connectivity between amygdala and cortical midline structures. Neuroimage 2011, 57, 1534–1541. [Google Scholar] [CrossRef] [PubMed]
- Van Marle, H.J.; Hermans, E.J.; Qin, S.; Fernández, G. From specificity to sensitivity: How acute stress affects amygdala processing of biologically salient stimuli. Biol. Psychiatry 2009, 66, 649–655. [Google Scholar] [CrossRef] [PubMed]
- Putman, P.; Roelofs, K. Effects of single cortisol administrations on human affect reviewed: Coping with stress through adaptive regulation of automatic cognitive processing. Psychoneuroendocrinology 2011, 36, 439–448. [Google Scholar] [CrossRef] [PubMed]
- Braunstein-bercovitz, H. Does stress enhance or impair selective attention? The effects of stress and perceptual load on negative priming. Anxiety Stress Coping 2003, 16, 345–357. [Google Scholar] [CrossRef]
- De Kloet, E.R.; Joëls, M.; Holsboer, F. Stress and the brain: From adaptation to disease. Nat. Rev. Neurosci. 2005, 6, 463–475. [Google Scholar] [CrossRef] [PubMed]
- Qin, S.; Hermans, E.J.; van Marle, H.J.; Luo, J.; Fernández, G. Acute psychological stress reduces working memory-related activity in the dorsolateral prefrontal cortex. Biol. Psychiatry 2009, 66, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Van Marle, H.J.; Hermans, E.J.; Qin, S.; Fernández, G. Enhanced resting-state connectivity of amygdala in the immediate aftermath of acute psychological stress. Neuroimage 2010, 53, 348–354. [Google Scholar] [CrossRef] [PubMed]
- Kavushansky, A.; Richter-Levin, G. Effects of stress and corticosterone on activity and plasticity in the amygdala. J. Neurosci. Res. 2006, 84, 1580–1587. [Google Scholar] [CrossRef] [PubMed]
- Karst, H.; Berger, S.; Erdmann, G.; Schütz, G.; Joëls, M. Metaplasticity of amygdalar responses to the stress hormone corticosterone. Proc. Natl. Acad. Sci. USA 2010, 107, 14449–14454. [Google Scholar] [CrossRef] [PubMed]
- Van Stegeren, A.H.; Wolf, O.T.; Everaerd, W.; Scheltens, P.; Barkhof, F.; Rombouts, S.A. Endogenous cortisol level interacts with noradrenergic activation in the human amygdala. Neurobiol. Learn. Mem. 2007, 87, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Van Stegeren, A.H.; Wolf, O.T.; Everaerd, W.; Rombouts, S.A. Interaction of endogenous cortisol and noradrenaline in the human amygdala. Prog. Brain Res. 2007, 167, 263–268. [Google Scholar]
- McReynolds, J.R.; Donowho, K.; Abdi, A.; McGaugh, J.L.; Roozendaal, B.; McIntyre, C.K. Memory-enhancing corticosterone treatment increases amygdala norepinephrine and Arc protein expression in hippocampal synaptic fractions. Neurobiol. Learn. Mem. 2010, 93, 312–321. [Google Scholar] [CrossRef] [PubMed]
- Van Stegeren, A.H.; Roozendaal, B.; Kindt, M.; Wolf, O.T.; Joëls, M. Interacting noradrenergic and corticosteroid systems shift human brain activation patterns during encoding. Neurobiol. Learn. Mem. 2010, 93, 56–65. [Google Scholar] [CrossRef] [PubMed]
- Kukolja, J.; Schläpfer, T.E.; Keysers, C.; Klingmüller, D.; Maier, W.; Fink, G.R.; Hurlemann, R. Modeling a negative response bias in the human amygdala by noradrenergic—Glucocorticoid interactions. J. Neurosci. 2008, 28, 12868–12876. [Google Scholar] [CrossRef] [PubMed]
- Ling, M.H.; Perry, P.J.; Tsuang, M.T. Side effects of corticosteroid therapy: Psychiatric aspects. Arch. Gen. Psychiatry 1981, 38, 471–477. [Google Scholar] [CrossRef] [PubMed]
- Clewett, D.; Schoeke, A.; Mather, M. Amygdala functional connectivity is reduced after the cold pressor task. Cognit. Affect. Behav. Neurosci. 2013, 13, 501–518. [Google Scholar] [CrossRef]
- Henckens, M.J.; van Wingen, G.A.; Joëls, M.; Fernández, G. Corticosteroid induced decoupling of the amygdala in men. Cereb. Cortex 2012, 22, 2336–2345. [Google Scholar] [CrossRef] [PubMed]
- Henckens, M.J.; van Wingen, G.A.; Joëls, M.; Fernández, G. Time-dependent effects of corticosteroids on human amygdala processing. J. Neurosci. 2010, 30, 12725–12732. [Google Scholar] [CrossRef] [PubMed]
- Schwabe, L.; Haddad, L.; Schachinger, H. HPA axis activation by a socially evaluated cold-pressor test. Psychoneuroendocrinology 2008, 33, 890–895. [Google Scholar] [CrossRef] [PubMed]
- Robertson, I.H.; Manly, T.; Andrade, J.; Baddeley, B.T.; Yiend, J. “Oops!”: Performance correlates of everyday attentional failures in traumatic brain injured and normal subjects. Neuropsychologia 1997, 35, 747–758. [Google Scholar] [CrossRef] [PubMed]
- Klein, K.; Boals, A. The relationship of life event stress and working memory capacity. Appl. Cognit. Psychol. 2001, 15, 565–579. [Google Scholar] [CrossRef]
- Cuthbert, B.N.; Schupp, H.T.; Bradley, M.M.; Birbaumer, N.; Lang, P.J. Brain potentials in affective picture processing: Covariation with autonomic arousal and affective report. Biol. Psychol. 2000, 52, 95–111. [Google Scholar] [CrossRef] [PubMed]
- Keil, A.; Bradley, M.M.; Hauk, O.; Rockstroh, B.; Elbert, T.; Lang, P.J. Large-scale neural correlates of affective picture processing. Psychophysiology 2002, 39, 641–649. [Google Scholar] [CrossRef] [PubMed]
- Olofsson, J.K.; Polich, J. Affective visual event-related potentials: Arousal, repetition, and time-on-task. Biol. Psychol. 2007, 75, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Schupp, H.T.; Cuthbert, B.N.; Bradley, M.M.; Cacioppo, J.T.; Ito, T.; Lang, P.J. Affective picture processing: The late positive potential is modulated by motivational relevance. Psychophysiology 2000, 37, 257–261. [Google Scholar] [CrossRef] [PubMed]
- Dolcoş, F.; Strungaru, C.; Dolcoş, S.M.; Turcu, E. The involvement of endogenous opiates in emotional-cognitive interaction in stress. Rom. J. Physiol. 1997, 35, 259–274. [Google Scholar]
- Liu, Y.; Huang, H.; McGinnis-Deweese, M.; Keil, A.; Ding, M. Neural substrate of the late positive potential in emotional processing. J. Neurosci. 2012, 32, 14563–14572. [Google Scholar] [CrossRef] [PubMed]
- Weymar, M.; Schwabe, L.; Löw, A.; Hamm, A.O. Stress sensitizes the brain: Increased processing of unpleasant pictures after exposure to acute stress. J. Cognit. Neurosci. 2012, 24, 1511–1518. [Google Scholar] [CrossRef]
- Patil, P.G.; Apfelbaum, J.L.; Zacny, J.P. Effects of a cold-water stressor on psychomotor and cognitive functioning in humans. Physiol. Behav. 1995, 58, 1281–1286. [Google Scholar] [CrossRef] [PubMed]
- Lang, P.J.; Bradley, M.M.; Cuthbert, B.N. Motivated attention: Affect, activation, and action. In Attention and Orienting: Sensory and Motivational Processes; Lawrence Erlbaum Associates: Hillsdale, NJ, USA, 1997; pp. 97–135. [Google Scholar]
- Lang, P.J.; Bradley, M.M.; Cuthbert, B.N. International Affective Picture System (IAPS): Technical Manual and Affective Ratings; The Center for Research in Psychophysiology: Gainesville, FL, USA, 1999. [Google Scholar]
- Schupp, H.T.; Junghöfer, M.; Weike, A.I.; Hamm, A.O. The selective processing of briefly presented affective pictures: An ERP analysis. Psychophysiology 2004, 41, 441–449. [Google Scholar] [CrossRef] [PubMed]
- Putnam, L.E.; Johnson, R.; Roth, W.T. Guidelines for reducing the risk of disease transmission in the psychophysiology laboratory. Psychophysiology 1992, 29, 127–141. [Google Scholar] [CrossRef] [PubMed]
- Crawford, L.E.; Cacioppo, J.T. Learning where to look for danger: Integrating affective and spatial information. Psychol. Sci. 2002, 13, 449–453. [Google Scholar] [CrossRef] [PubMed]
- Öhman, A.; Mineka, S. Fears, phobias, and preparedness: Toward an evolved module of fear and fear learning. Psychol. Rev. 2001, 108, 483–522. [Google Scholar] [CrossRef] [PubMed]
- Bradley, M.M.; Greenwald, M.K.; Petry, M.C.; Lang, P.J. Remembering pictures: Pleasure and arousal in memory. J. Exp. Psychol. Learn. Mem. Cognit. 1992, 18, 379–390. [Google Scholar] [CrossRef]
- Yiend, J. The effects of emotion on attention: A review of attentional processing of emotional information. Cognit. Emot. 2010, 24, 3–47. [Google Scholar] [CrossRef]
- Dickerson, S.S.; Kemeny, M.E. Acute stressors and cortisol responses: A theoretical integration and synthesis of laboratory research. Psychol. Bull. 2004, 130, 355–391. [Google Scholar] [CrossRef] [PubMed]
- Meiran, N. Reconfiguration of processing mode prior to task performance. J. Exp. Psychol. Learn. Mem. Cognit. 1996, 22, 1423–1442. [Google Scholar] [CrossRef]
- Rogers, R.D.; Monsell, S. Costs of a predictible switch between simple cognitive tasks. J. Exp. Psychol. 1995, 124, 207–231. [Google Scholar] [CrossRef]
- Alfarra, R.; Fins, A.I.; Chayo, I.; Tartar, J.L. Changes in attention to an emotional task after sleep deprivation: Neurophysiological and behavioral findings. Biol. Psychol. 2015, 104, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Joëls, M.; Baram, T.Z. The neuro-symphony of stress. Nat. Rev. Neurosci. 2009, 10, 459–466. [Google Scholar] [PubMed]
- Roozendaal, B.; Okuda, S.; van der Zee, E.A.; McGaugh, J.L. Glucocorticoid enhancement of memory requires arousal-induced noradrenergic activation in the basolateral amygdala. Proc. Natl. Acad. Sci. 2006, 103, 6741–6746. [Google Scholar] [CrossRef] [PubMed]
- Diamond, D.M.; Campbell, A.M.; Park, C.R.; Halonen, J.; Zoladz, P.R. The temporal dynamics model of emotional memory processing: A synthesis on the neurobiological basis of stress-induced amnesia, flashbulb and traumatic memories, and the Yerkes-Dodson law. Neural Plast. 2007, 2007. [Google Scholar] [CrossRef]
- Quax, R.A.; Manenschijn, L.; Koper, J.W.; Hazes, J.M.; Lamberts, S.W.J.; van Rossum, E.F.C.; Feelders, R.A. Glucocorticoid sensitivity in health and disease. Nat. Rev. Endocrinol. 2013, 9, 670–686. [Google Scholar] [CrossRef] [PubMed]
- DeRijk, R.; de Kloet, E.R. Corticosteroid receptor genetic polymorphisms and stress responsivity. Endocrine 2005, 28, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Zoccola, P.M.; Dickerson, S.S.; Zaldivar, F.P. Rumination and cortisol responses to laboratory stressors. Psychosom. Med. 2008, 70, 661–667. [Google Scholar] [CrossRef] [PubMed]
- Veer, I.M.; Oei, N.Y.; Spinhoven, P.; van Buchem, M.A.; Elzinga, B.M.; Rombouts, S.A. Endogenous cortisol is associated with functional connectivity between the amygdala and medial prefrontal cortex. Psychoneuroendocrinology 2012, 37, 1039–1047. [Google Scholar] [CrossRef] [PubMed]
- Ikezawa, S.; Corbera, S.; Wexler, B.E. Emotion self-regulation and empathy depend upon longer stimulus exposure. Soc. Cognit. Affect. Neurosci. 2013, 9, 1561–1568. [Google Scholar] [CrossRef]
- Blechert, J.; Sheppes, G.; di Tella, C.; Williams, H.; Gross, J.J. See what you think reappraisal modulates behavioral and neural responses to social stimuli. Psychol. Sci. 2012, 23, 346–353. [Google Scholar] [CrossRef] [PubMed]
- Tartar, J.L.; Sheehan, C.M.; Nash, A.J.; Starratt, C.; Puga, A.; Widmayer, S. ERPs differ from neurometric tests in assessing HIV-associated cognitive deficit. Neuroreport 2004, 15, 1675–1678. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, M.; Tartar, J.L.; Padron, D.; Acosta, J. Neurophysiological marker of inhibition distinguishes language groups on a non-linguistic executive function test. Brain Cognit. 2013, 83, 330–336. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alomari, R.A.; Fernandez, M.; Banks, J.B.; Acosta, J.; Tartar, J.L. Acute Stress Dysregulates the LPP ERP Response to Emotional Pictures and Impairs Sustained Attention: Time-Sensitive Effects. Brain Sci. 2015, 5, 201-219. https://doi.org/10.3390/brainsci5020201
Alomari RA, Fernandez M, Banks JB, Acosta J, Tartar JL. Acute Stress Dysregulates the LPP ERP Response to Emotional Pictures and Impairs Sustained Attention: Time-Sensitive Effects. Brain Sciences. 2015; 5(2):201-219. https://doi.org/10.3390/brainsci5020201
Chicago/Turabian StyleAlomari, Rima A., Mercedes Fernandez, Jonathan B. Banks, Juliana Acosta, and Jaime L. Tartar. 2015. "Acute Stress Dysregulates the LPP ERP Response to Emotional Pictures and Impairs Sustained Attention: Time-Sensitive Effects" Brain Sciences 5, no. 2: 201-219. https://doi.org/10.3390/brainsci5020201
APA StyleAlomari, R. A., Fernandez, M., Banks, J. B., Acosta, J., & Tartar, J. L. (2015). Acute Stress Dysregulates the LPP ERP Response to Emotional Pictures and Impairs Sustained Attention: Time-Sensitive Effects. Brain Sciences, 5(2), 201-219. https://doi.org/10.3390/brainsci5020201




