Neuroplasticity beyond Sounds: Neural Adaptations Following Long-Term Musical Aesthetic Experiences
Abstract
:1. Introduction
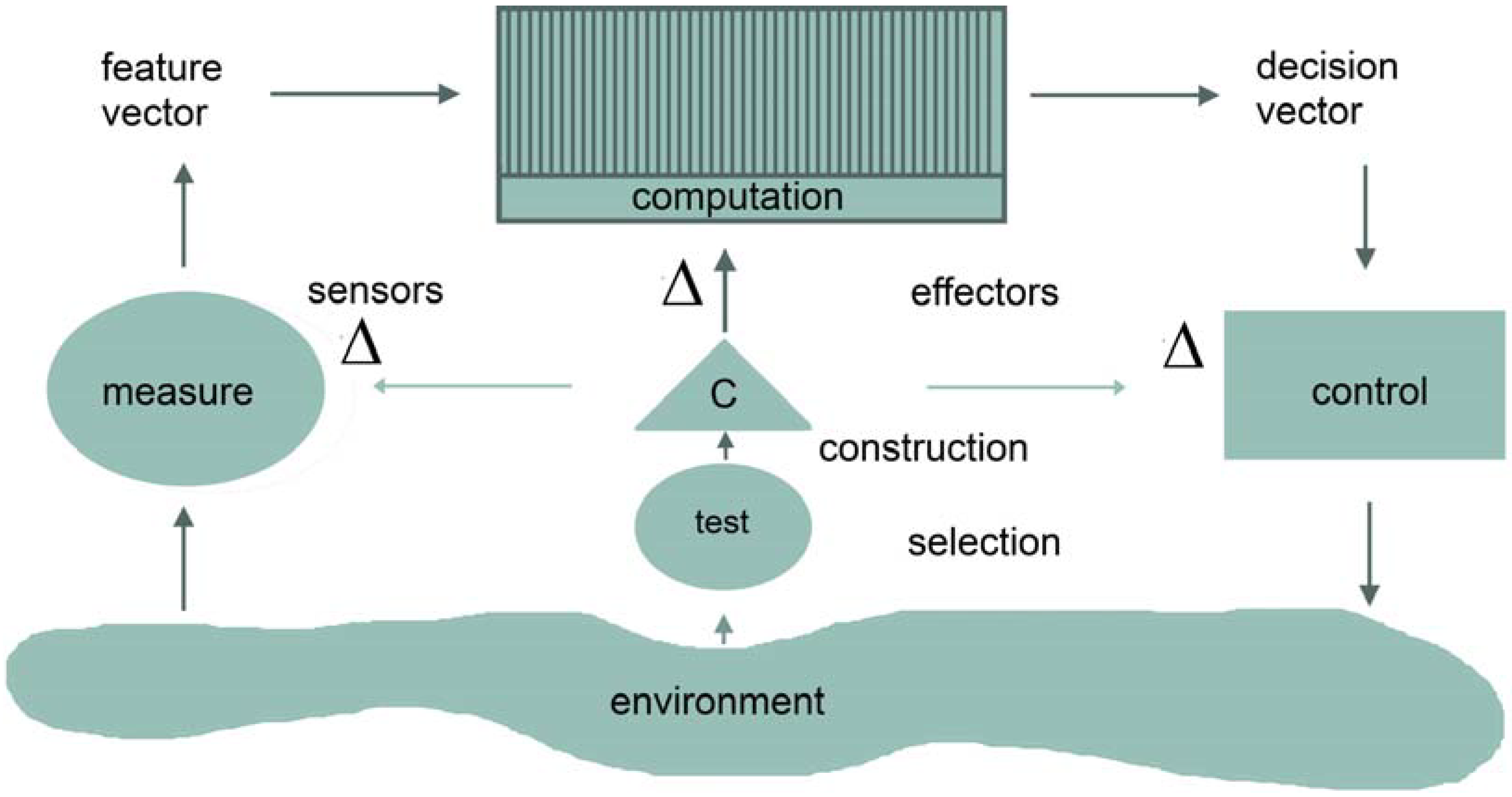

2. Feature Encoding and Integration
3. Early Emotional Reactions and Motor Tendencies/Actions
3.1. Early Emotional Reactions and Involuntary Motor Tendencies
3.2. Voluntary Motor Tendencies/Actions
4. Style Mastering and Conceptualization
5. Emotion and Proprioception
6. Evaluation and Preference
7. Concluding Remarks
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Peretz, I.; Zatorre, R.J. (Eds.) The Cognitive Neuroscience of Music; Oxford University Press: Oxford, UK, 2003.
- Peretz, I.; Coltheart, M. Modularity of music processing. Nat. Neurosci. 2003, 6, 688–691. [Google Scholar] [CrossRef] [PubMed]
- Patel, A. Music, Language, and the Brain; Oxford University Press: New York, NY, USA, 2008. [Google Scholar]
- Koelsch, S. Toward a neural basis of music perception—A review and updated model. Front. Psychol. 2011, 2. [Google Scholar] [CrossRef]
- Zatorre, R.; Krumhansl, C.L. Neuroscience. Mental models and musical minds. Science 2002, 298, 2138–2139. [Google Scholar] [CrossRef] [PubMed]
- Salimpoor, V.N.; Zatorre, R.J. Complex cognitive functions underlie aesthetic emotions: Comment on “From everyday emotions to aesthetic emotions: Towards a unified theory of musical emotions”. Phys. Life Rev. 2013, 10, 279–280. [Google Scholar] [CrossRef] [PubMed]
- Vuust, P.; Kringelbach, M.L. The pleasure of making sense of music. Interdiscip. Sci. Rev. 2010, 35, 166–182. [Google Scholar] [CrossRef]
- Salimpoor, V.N.; Zald, D.H.; Zatorre, R.; Dagher, A.; McIntosh, A.R. Predictions and the brain: How musical sounds become rewarding. Trends Cogn. Sci. 2015, 19, 86–91. [Google Scholar] [CrossRef] [PubMed]
- Brattico, E.; Bogert, B.; Jacobsen, T. Toward a neural chronometry for the aesthetic experience of music. Front. Psychol. 2013, 4. [Google Scholar] [CrossRef]
- Brincker, M. The Aesthetic Stance—On the Conditions and Consequences of Becoming a Beholder. In Aesthetics and the Embodied Mind: Beyond Art Theory and the Cartesian Mind-Body Dichotomy; Springer Netherlands: Berlin, Germany, 2015; pp. 117–138. [Google Scholar]
- Scherer, K.; Zentner, M. Music evoked emotions are different-more often aesthetic than utilitarian. Behav. Brain Sci. 2008, 5, 595–596. [Google Scholar]
- Ellsworth, P.C.; Scherer, K.R. Appraisal processes in emotion. In Handbook of Affective Sciences; Davidson, R., Sherer, K., Hill, H., Eds.; Oxford University Press: New York, NY, USA, 2003; pp. 572–595. [Google Scholar]
- Juslin, P.N. From everyday emotions to aesthetic emotions: Toward a unified theory of musical emotions. Phys. Life Rev. 2013, 10, 253–266. [Google Scholar]
- Hargreaves, D.J.; North, A.C. Experimental aesthetics and liking for music. In Handbook of Music and Emotion: Theory, Research, Applications; Patrik, N.J., Ed.; Oxford University Press: Oxford, UK, 2010; pp. 515–546. [Google Scholar]
- Pask, G. An Approach to Cybernetics. Science Today Series; Harper & Brothers: New York, NY, USA, 1961. [Google Scholar]
- Pask, G. Different kinds of Cybernetics. In New Perspectives on Cybernetics: Self-Organization, Autonomy and Connectionism; van de Vijver, G., Ed.; Kluwer Academic: Dordrecht, The Netherlands, 1992; pp. 11–31. [Google Scholar]
- Von Foerster, H. (Ed.) Cybernetics of Cybernetics; University of Illinois: Urbana, IL, USA, 1974.
- Von Foerster, H. (Ed.) Observing Systems; Intersystems Press: Seaside, CA, USA, 1984.
- Maturana, H.; Varela, F. Autopoiesis and Cognition: The Realization of the Living; Reidel Publishing: Boston, MA, USA, 1980. [Google Scholar]
- Luhmann, N. Essays on Self-Reference; Columbia University Press: New York, NY, USA, 1990. [Google Scholar]
- Luhmann, N. Social Systems; Stanford University Press: Stanford, CA, USA, 1995. [Google Scholar]
- Reybrouck, M. Biological roots of musical epistemology: Functional cycles, umwelt, and enactive listening. Semiotica 2001, 134, 599–633. [Google Scholar]
- Reybrouck, M. A Biosemiotic and ecological approach to music cognition: Event perception between auditory listening and cognitive economy. Axiomathes 2005, 15, 229–266. [Google Scholar] [CrossRef]
- Reybrouck, M. Musical Creativity between Symbolic Modelling and Perceptual Constraints: The Role of Adaptive Behaviour and Epistemic Autonomy. In Musical Creativity: Multidisciplinary Research in Theory and Practice; Deliège, I., Wiggins, G., Eds.; Psychology Press: Oxford, UK, 2006; pp. 42–59. [Google Scholar]
- Reybrouck, M. Musical sense-making and the concept of affordance: An ecosemiotic and experiential approach. Biosemiotics 2012, 5, 391–409. [Google Scholar] [CrossRef]
- Cariani, P. Some epistemological implications of devices which construct their own sensors and effectors. In Towards a Practice of Autonomous Systems, Proceedings of the First European Workshop on Artificial Life; Varela, F., Bourgine, P., Eds.; MIT Press: Cambridge, MA, USA, 1991; pp. 484–493. [Google Scholar]
- Cariani, P. Life’s journey through the semiosphere. Semiotica 1998, 120, 243–257. [Google Scholar]
- Laske, O.-E. Music, Memory and Thought. Explorations in Cognitive Musicology; University of Pittsburgh, University Microfilms International: Ann Arbor, MI, USA, 1977. [Google Scholar]
- Cariani, P.A. Cybernetic systems and the semiotics of translation. In Translation, Translation; Petrilli, S., Ed.; Rodopi: Amsterdam, The Netherlands, 2003; pp. 349–367. [Google Scholar]
- Cariani, P. Symbols and dynamics in the brain. BioSystems 2001, 60, 59–83. [Google Scholar] [CrossRef] [PubMed]
- Damasio, A. Descartes’ Error: Emotion, Reason, and the Human Brain; Harper Collins: New York, NY, USA, 1994. [Google Scholar]
- Pantev, C.; Engelien, A.; Candia, V.; Elbert, T. Representational cortex in musicians. In The Cognitive Neuroscience of Music; Peretz, I., Zatorre, R., Eds.; Oxford University Press: Oxford, NY, USA, 2003; pp. 381–395. [Google Scholar]
- Gaser, C.; Schlaug, G. Brain structures differ between musicians and non-musicians. J. Neurosci. 2003, 23, 9240–9245. [Google Scholar] [PubMed]
- Schlaug, G.; Forgeard, M.; Zhu, L.; Norton, A.; Norton, A.; Winner, E. Training-induced neuroplasticity in young children. Ann. N. Y. Acad. Sci. 2005, 1169, 205–208. [Google Scholar] [CrossRef]
- Herholz, S.; Zatorre, R. Musical training as a framework for brain plasticity: Behavior, function, and structure. Neuron 2012, 76, 486–502. [Google Scholar] [CrossRef] [PubMed]
- Rauschecker, J.P. Mechanisms of visual plasticity: Hebb synapses, NMDA receptors, and beyond. Physiol. Rev. 1991, 71, 587–615. [Google Scholar] [PubMed]
- Rauschecker, J.P. Functional organization and plasticity of auditory cortex. In The Cognitive Neuroscience of Music; Peretz, I., Zatorre, R., Eds.; Oxford University Press: Oxford, NY, USA, 2003; pp. 357–365. [Google Scholar]
- Merrett, D.L.; Peretz, I.; Wilson, S.J. Moderating variables of music training-induced neuroplasticity: A review and discussion. Front. Psychol. 2013, 4, 606–608. [Google Scholar] [CrossRef] [PubMed]
- Leroy, J.-L. (Ed.) Actualités des Universaux en Musique/Topics in Universals in Music; Edition des Archives Contemporaines: Paris, France, 2012.
- Miereanu, C.; Hascher, X. (Eds.) Les Universaux en Musique, Actes du 4e Congrès International sur la Signification Musicale; Publications de la Sorbonne: Paris, France, 1998; pp. 637–648.
- Reybrouck, M. The musical code between nature and nurture. In The Codes of Life: The Rules of Macroevolution; Barbieri, M., Ed.; Springer: Dordrecht, The Netherlands, 2008; pp. 395–434. [Google Scholar]
- Brattico, E.; Olcese, C.; Tervaniemi, M. Automatic processing of musical sounds in the brain. In Handbook of Systematic Musicology; Koelsch, S., Ed.; Springer: Berlin, Germany.
- Morrison, S.J.; Demorest, S.M.; Aylward, E.H.; Cramer, S.C.; Maravilla, K.R. FMRI investigation of cross-cultural music comprehension. Neuroimage 2013, 20, 378–384. [Google Scholar] [CrossRef]
- Schlaug, G. The brain of musicians: A model for functional and structural adaptation. Ann. N. Y. Acad. Sci. 2001, 930, 281–299. [Google Scholar] [CrossRef] [PubMed]
- Münte, T.; Altenmüller, E.; Jäncke, L. The musician’s brain as a model of neuroplasticity. Nat. Rev. Neurosci. 2002, 3, 473–478. [Google Scholar] [PubMed]
- Johansson, B. Music and brain plasticity. Eur. Rev. 2006, 14, 49–64. [Google Scholar] [CrossRef]
- Altenmüller, E. Neurology of musical performance. Clin. Med. 2008, 8, 410–413. [Google Scholar] [CrossRef] [PubMed]
- Stewart, L. Do musicians have different brains? Clin. Med. 2008, 8, 304–308. [Google Scholar] [CrossRef] [PubMed]
- Habib, M.; Besson, M. What do music training and musical experience teach us about brain plasticity? Music Percept. 2009, 26, 279–285. [Google Scholar] [CrossRef]
- Jäncke, L. Music drives brain plasticity. Biol. Rep. 2009, 1. [Google Scholar] [CrossRef]
- Jäncke, L. The plastic human brain. Restor. Neurol. Neurosci. 2009, 27, 521–538. [Google Scholar] [PubMed]
- Tervaniemi, M. Musicians—Same or different? Ann. N. Y. Acad. Sci. 2009, 1169, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Kraus, N.; Chandrasekaran, B. Music training for the development of auditory skills. Nat. Rev. Neurosci. 2010, 11, 599–605. [Google Scholar] [CrossRef] [PubMed]
- Schlaug, G.; Marchina, S.; Norton, A. Evidence for plasticity in white-matter tracts of patients with chronic Broca’s aphasia undergoing intense intonation-based speech therapy. Ann. N. Y. Acad. Sci. 2009, 1169, 385–394. [Google Scholar] [CrossRef] [PubMed]
- Wan, C.; Schlaug, G. Music making as a tool for promoting brain plasticity across the lifespan. Neuroscientist 2010, 16, 566–577. [Google Scholar] [CrossRef] [PubMed]
- Merrett, D.; Wilson, S. Music and neural plasticity. In Lifelong Engagement with Music: Benefits for Mental Health and Well-being; Rickard, N., McFerran, K., Eds.; Nova Science Publishers: Hauppauge, NY, USA, 2012; pp. 123–162. [Google Scholar]
- Gärtner, H.; Minnerop, M.; Pieperhoff, P.; Zilles, K.; Altenmüller, E.; Amunts, K. Brain morphometry shows effects of long-term musical practice in middle-aged keyboard players. Front. Psychol. 2013, 4. [Google Scholar] [CrossRef] [PubMed]
- Koelsch, S.; Schröger, E.; Tervaniemi, M. Superior pre-attentive auditory processing in musicians. Neuroreport 1999, 10, 1309–1313. [Google Scholar] [CrossRef] [PubMed]
- Brattico, E.; Brattico, P.; Jacobsen, T. The origins of the aesthetic enjoyment of music—A review of the literature. Music Sci. Spec. Issue 2009, 13, 15–39. [Google Scholar]
- Tervaniemi, M.; Rytkönen, M.; Schröger, E.; Ilmoniemi, R.J.; Näätänen, R. Superior formation of cortical memory traces for melodic patterns in musicians. Learn. Mem. 2011, 8, 295–300. [Google Scholar] [CrossRef]
- Rodrigues, A.C.; Loureiro, M.A.; Caramelli, P. Long-term musical training may improve different forms of visual attention ability. Brain Cogn. 2013, 82, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Schneider, P.; Scherg, M.; Dosch, H.G.; Specht, H.J.; Gutschalk, A.; Rupp, A. Morphology of Heschl’s gyrus reflects enhanced activation in the auditory cortex of musicians. Nat. Neurosci. 2002, 5, 688–694. [Google Scholar] [CrossRef] [PubMed]
- Hyde, K.; Lerch, J.; Norton, A.; Forgeard, M.; Winner, E.; Evans, A.; Schlaug, G. Musical training shapes structural brain development. J. Neurosci. 2009, 29, 3019–3025. [Google Scholar] [CrossRef] [PubMed]
- Pantev, C.; Herholtz, S.C. Plasticity of the human auditory cortex related to musical training. Neurosci. Biobehav. Rev. 2011, 35, 2140–2154. [Google Scholar] [CrossRef] [PubMed]
- Putkinen, V.; Tervaniemi, M.; Huotilainen, M. Informal musical activities are linked to auditory discrimination and attention in 2–3-year-old children: An event-related potential study. Eur. J. Neurosci. 2013, 37, 654–661. [Google Scholar] [CrossRef] [PubMed]
- Moreno, S.; Bialystok, E.; Barac, R.; Schellenberg, E.G.; Cepeda, N.J.; Chau, T. Short-term music training enhances verbal intelligence and executive function. Psychol. Sci. 2011, 22, 1425–1433. [Google Scholar] [CrossRef] [PubMed]
- Moreno, S.; Bidelman, G.M. Examining neural plasticity and cognitive benefit through the unique lens of musical training. Hear. Res. 2014, 308, 84–97. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, A.H.; Hulse, S.H. Absolute pitch. Psychol. Bull. 1993, 113, 345–361. [Google Scholar] [CrossRef] [PubMed]
- Wengenroth, M.; Blatow, M.; Heinecke, A.; Reinhardt, J.; Stippich, C.; Hofmann, E.; Schneider, P. Increased volume and function of right auditory cortex as a marker for absolute pitch. Cereb. Cortex 2014, 24, 1127–1137. [Google Scholar] [CrossRef] [PubMed]
- Zatorre, R.J. Absolute pitch: A model for understanding the influence of genes and development on neural and cognitive function. Nat. Neurosci. 2003, 6, 692–695. [Google Scholar] [CrossRef] [PubMed]
- Elmer, S.; Sollberger, S.; Meyer, M.; Jäncke, L. An empirical re-evaluation of Absolute Pitch: Behavioral and electrophysiological measurements. J. Cogn. Neurosci. 2013, 25, 1736–1753. [Google Scholar] [CrossRef] [PubMed]
- Hall, D.A.; Hart, H.C.; Johnsrude, I.S. Relationships between human auditory cortical structure and function. Audiol. Neurootol. 2003, 8, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Schlaug, G. Increased corpus callosum size in musicians. Neuropsychologia 1995, 33, 1047–1055. [Google Scholar] [CrossRef] [PubMed]
- Keenan, J.P.; Thangaraj, V.; Halpern, A.R.; Schlaug, G. Absolute pitch and planum temporale. Neuroimage 2001, 14, 1402–1408. [Google Scholar] [CrossRef] [PubMed]
- Oechslin, M.S.; Meyer, M.; Jäncke, L. Absolute pitch—Functional evidence of speech-relevant auditory acuity. Cereb. Cortex 2010, 20, 447–455. [Google Scholar] [CrossRef] [PubMed]
- Luders, E.; Gaser, C.; Jancke, L.; Schlaug, G. A voxel-based approach to gray matter asymmetries. Neuroimage 2004, 22, 656–664. [Google Scholar] [CrossRef] [PubMed]
- Wilson, S.J.; Lusher, D.; Wan, C.Y.; Dudgeon, P.; Reutens, D.C. The neurocognitive components of pitch processing: Insights from absolute pitch. Cereb. Cortex 2009, 19, 724–732. [Google Scholar] [CrossRef] [PubMed]
- Loui, P.; Zamm, A.; Schlaug, G. Enhanced functional networks in absolute pitch. Neuroimage 2012, 63, 632–640. [Google Scholar] [CrossRef] [PubMed]
- Juslin, P.; Västfjäll, D. Emotional responses to music: The need to consider underlying mechanisms. Behav. Brain Sci. 2008, 31, 559–575. [Google Scholar] [CrossRef] [PubMed]
- Panksepp, J.; Bernatzky, G. Emotional sounds and the brain: The neuro-affective foundations of musical appreciation. Behav. Processes 2002, 60, 133–155. [Google Scholar] [CrossRef] [PubMed]
- Schneck, D.; Berger, D. The Music Effect. Music Physiology and Clinical Applications; Kingsley Publishers: London, UK and Philadelphia, PA, USA, 2010. [Google Scholar]
- Peretz, I. Listen to the brain: A biological perspective on musical emotions. In Music and Emotion: Theory and Research; Juslin, P.N., Sloboda, J., Eds.; Oxford University Press: Oxford, UK, 2001; pp. 105–134. [Google Scholar]
- Balkwill, L.L.; Thompson, W.F. A cross-cultural investigation of the perception of emotion in music: Psychophysical and cultural cues. Music Percept. 1999, 17, 43–64. [Google Scholar] [CrossRef]
- Schubert, E. Measuring emotion continuously: Validity and reliability of the two dimensional emotion space. Aust. J. Psychol. 1999, 51, 154–165. [Google Scholar] [CrossRef]
- Schubert, E. Continuous measurement of self-report emotional response to music. In Music and Emotion: Theory and Research; Juslin, P.N., Sloboda, J., Eds.; Oxford University Press: Oxford, UK, 2001; pp. 393–414. [Google Scholar]
- Schubert, E. Modeling perceived emotion with continuous musical features. Music Percept. 2004, 21, 561–585. [Google Scholar] [CrossRef]
- Juslin, P. Communicating emotion in music performance: A review of and theoretical framework. In Music and Emotion: Theory and Research; Juslin, P.N., Sloboda, J., Eds.; Oxford University Press: Oxford, UK, 2001; pp. 309–337. [Google Scholar]
- Juslin, P.N.; Sloboda, J. Music and Emotion: Theory and Research; Oxford University Press: Oxford, UK, 2001. [Google Scholar]
- Bigand, E.; Vieillard, S.; Madurell, F.; Marozeau, J.; Dacquet, A. Multidimensional scaling of emotional responses to music: The effect of musical expertise and of the duration of the excerpts. Cognit. Emot. 2005, 1, 1113–1139. [Google Scholar] [CrossRef]
- Zentner, M.; Grandjean, D.; Scherer, K. Emotions evoked by the sound of music: Characterization, classification, and measurement. Emotion 2008, 8, 494–521. [Google Scholar] [CrossRef] [PubMed]
- Fritz, T.; Jentschke, S.; Gosselin, N.; Sammler, D.; Peretz, I.; Turner, R.; Friederici, A.D.; Koelsch, S. Universal recognition of three basic emotions in music. Curr. Biol. 2009, 19, 573–576. [Google Scholar] [CrossRef] [PubMed]
- Fritz, T.H.; Renders, W.; Müller, K.; Schmude, P.; Leman, M.; Turner, R.; Villringer, A. Anatomical differences in the human inferior colliculus relate to the perceived valence of musical consonance and dissonance. Eur. J. Neurosci. 2013, 38, 3099–3105. [Google Scholar]
- Davidson, R.J.; Scherer, K.R.; Goldsmith, H. Handbook of Affective Sciences; Davidson, R.J., Scherer, K.R., Goldsmith, H.H., Eds.; Oxford University Press: Oxford, UK, 2003. [Google Scholar]
- Panksepp, J. The emotional sources of “chills” induced by music. Music Percep. 1995, 13, 171–207. [Google Scholar]
- Craig, D. An exploratory study of physiological changes during “chills” induced by music. Music. Sci. 2005, 9, 273–285. [Google Scholar]
- Blood, A.; Zatorre, R. Intensely pleasurable responses to music correlate with activity in brain regions implicated in reward and emotion. Proc. Nat. Acad. Sci. 2001, 98, 11818–11823. [Google Scholar] [CrossRef] [PubMed]
- Hodges, D. Psychophysiological measures. In Handbook of Music and Emotion: Theory, Research, Applications; Juslin, P.N., Sloboda, J.A., Eds.; Oxford University Press: Oxford, UK, 2010; pp. 279–311. [Google Scholar]
- Grewe, O.; Kopiez, R.; Altenmuller, E. Chills as an indicator of individual emotional peaks. Ann. N. Y. Acad. Sci. 2009, 1169, 351–354. [Google Scholar] [CrossRef] [PubMed]
- Fishman, Y.I.; Volkov, I.O.; Noh, M.D.; Garell, P.C.; Bakken, H.; Arezzo, J.C.; Howard, M.A.; Steinschneider, M. Consonance and dissonance of musical chords: Neural correlates in auditory cortex of monkeys and humans. J. Neurophysiol. 2001, 86, 2761–2788. [Google Scholar] [PubMed]
- Dellacherie, D.; Roy, M.; Hugueville, L.; Peretz, I.; Samson, S. The effect of musical experience on emotionals self-reports and psychophysiological responses to dissonance. Psychophysiology 2011, 48, 337–349. [Google Scholar] [CrossRef] [PubMed]
- Amunts, K.; Schlaug, G.; Jäncke, L.; Steinmetz, H.; Schleicher, A.; Dabringhaus, A.; Zilles, K. Motor cortex and hand motor skills: Structural compliance in the human brain. Hum. Brain Mapp. 1997, 5, 206–215. [Google Scholar] [CrossRef] [PubMed]
- Hutchinson, S.; Hui-Lin Lee, L.; Gaab, N.; Schlaug, G. Cerebellar volume in musicians. Cereb. Cortex 2003, 13, 943–949. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.; Chen, Y.; Schlaug, G. Corpus callosum: Musician and gender effect. Neuroreport 2003, 14, 205–209. [Google Scholar] [CrossRef] [PubMed]
- Moore, E.; Schaefer, R.S.; Bastin, M.E; Roberts, N.; Overy, K. Can musical training influence brain connectivity? Evidence from Diffusion Tensor MRI. Brain Sci. 2014, 4, 405–427. [Google Scholar] [CrossRef] [PubMed]
- Rüber, T.; Lindenberg, R.; Schlaug, G. Differential adaptation of descending motor tracts in musicians. Cereb. Cortex 2013. [Google Scholar] [CrossRef]
- Imfeld, A.; Oechslin, M.S.; Meyer, M.; Loenneker, T.; Jancke, L. White matter plasticity in the corticospinal tract of musicians: A diffusion tensor imaging study. Neuroimage 2009, 46, 600–607. [Google Scholar] [CrossRef] [PubMed]
- Bengtsson, S.; Nagy, Z.; Skare, S.; Forsman, L.; Forssberg, H.; Ulle, F. Extensive piano practicing has regionally specific effects on white matter development. Nat. Neurosci. 2005, 8, 1148–1150. [Google Scholar] [CrossRef] [PubMed]
- Zatorre, R.; Chen, L.; Penhune, V.B. When the brain plays music auditory-motor interactions in music reception and production. Nat. Rev. Neurosci. 2007, 8, 547–558. [Google Scholar] [CrossRef] [PubMed]
- Burunat, I.; Brattico, E.; Puolivali, T.; Ristaniemi, T.; Sams, M.; Toivainen, P. Prominent interhemispheric functional symmetry in musicians during free listening. In Proceedings of the Neurosciences and Music V: Cognitive Stimulation and Rehabilitation, Dijon, France, 29 May–1 June 2014; Fondazione Mariani: Milano, Italy, 2014. [Google Scholar]
- Liberman, A.; Mattingly, I. The motor theory of perception of speech revisited. Cognition 1985, 21, 1–36. [Google Scholar] [CrossRef] [PubMed]
- Viviani, P. Motor-perceptual interactions: The evolution of an idea. In Cognitive Science in Europe: Issues and Trends; Viviani, P., Ed.; Golem: Ivrea, Italy, 1990; pp. 11–39. [Google Scholar]
- Scheerer, E. Motor theories of cognitive structure: A historical review. In Cognition and Motor Processes; Prinz, W., Sanders, A., Eds.; Springer: Berlin, Germany, 1984. [Google Scholar]
- Berthoz, A. Le sens du movement; Odile Jacob: Paris, France, 1997. [Google Scholar]
- Jeannerod, M. The representing brain: Neural correlates of motor intention and imagery. Behav. Brain Sci. 1994, 17, 187–202. [Google Scholar] [CrossRef]
- Jeannerod, M. Neural simulation of action: A unifying mechanism for motor cognition. Neuroimage 2011, 14, 103–109. [Google Scholar] [CrossRef]
- MacKay, D. Constraints on theories of inner speech. In Auditory Imagery; Reisberg, D., Ed.; Lawrence Erlbaum: Hillsdale, NJ, USA and London, UK, 1992; pp. 121–149. [Google Scholar]
- Viviani, P.; Stucchi, N. Biological movements look uniform: Evidence of motor-perceptual interactions. J. Exp. Psychol. Hum. Percept. Perform. 1992, 18, 603–623. [Google Scholar] [CrossRef] [PubMed]
- Reybrouck, M. Musical imagery between sensory processing and ideomotor simulation. In Musical Imagery; Godøy, R.I., Jörgensen, H., Eds.; Swets & Zeitlinger: Lisse, The Netherlands, 2001; pp. 117–136. [Google Scholar]
- Annett, J. On knowing how to do things: A theory of motor imagery. Cogn. Brain Res. 1996, 3, 65–69. [Google Scholar] [CrossRef]
- Decety, J. Do imagined actions share the same neural substrate? Cogn. Brain Res. 1996, 3, 87–93. [Google Scholar] [CrossRef]
- Deecke, L. Planning, preparation, execution, and imagery of volitional action. Cogn. Brain Res. 1996, 3, 59–64. [Google Scholar] [CrossRef]
- Deecke, L. Voluntary action and movement disorders. Biomed. Eng. 2001, 5, 367–469. [Google Scholar]
- Paillard, J. La conscience. In Traité de Psychologie Expérimentale; Richelle, M., Requin, J., Robert, M., Eds.; Presses Universitaires de France: Paris, France, 1994; pp. 639–684. [Google Scholar]
- Gallese, V.; Rochat, M.; Cossu, G.; Sinigaglia, C. Motor cognition and its role in the phylogeny and ontogeny of action understanding. Dev. Psychol. 2009, 45, 103–113. [Google Scholar] [CrossRef] [PubMed]
- Gallese, V.; Fadiga, L.; Fogassi, L.; Rizzolatti, G. Action recognition in the premotor cortex. Brain 1996, 119, 593–609. [Google Scholar] [CrossRef] [PubMed]
- Rizzolatti, G.; Sinigaglia, C. How Our Minds Share Actions, Emotions, and Experience; Oxford University Press: Oxford, UK, 2008. [Google Scholar]
- Leman, M. Embodied Music Cognition and Mediation Technology; The MIT Press: Cambridge, MA, USA and London, UK, 2007. [Google Scholar]
- Godøy, R.I.; Leman, M. (Eds.) Musical Gestures: Sound, Movement, and Meaning; Routledge: New York, USA and London, UK, 2010.
- Godøy, R.I. Sound-action awareness in music. In Music and Consciousness: Philosophical, Psychological, and Cultural Perspectives; Clarke, D., Clarke, E., Eds.; Oxford University Press: Oxford, UK, 2012; pp. 215–230. [Google Scholar]
- Mahoney, M.; Avener, M. Psychology of the elite athlete. Cogn. Ther. Res. 1987, 1, 135–141. [Google Scholar] [CrossRef]
- Calvo-Merino, B.; Glaser, D.E.; Grèzes, J.; Passingham, R.E.; Haggard, P. Action observation and acquired motor skills: An fMRI study with expert dancers. Cereb. Cort. 2005, 15, 1243–1249. [Google Scholar] [CrossRef]
- Di Pellegrino, G.; Fadiga, L.; Fogassi, L.; Gallese, V.; Rizzolatti, G. Understanding motor events: A neurophysiological study. Exp. Brain Res. 1992, 19, 176–180. [Google Scholar] [CrossRef]
- Kohler, E.; Keysers, C.; Umilta’, M.A.; Fogassi, L.; Gallese, V.; Rizzolatti, G. Hearing sounds, understanding actions: Action representation in mirror neurons. Science 2002, 297, 846–848. [Google Scholar] [CrossRef] [PubMed]
- Molnar-Szakacs, I.; Overy, K. Music and Mirror Neurons: From Motion to Emotion. Soc. Cogn. Affect. Neurosci. 2006, 119, 593–609. [Google Scholar]
- Overy, K.; Molnar-Szakacs, I. Being together in time: Musical experience and the mirror neuron system. Music Percept. 2009, 26, 489–504. [Google Scholar] [CrossRef]
- Peretz, I.; Brattico, E.; Tervaniemi, M. Abnormal electrical brain responses to pitch in congenital amusia. Ann. Neurol. 2005, 58, 478–482. [Google Scholar] [CrossRef] [PubMed]
- Loui, P.; Alsop, D.; Schlaug, G. Tone-deafness: A new disconnection syndrome? J. Neurosci. 2009, 29, 10215–10220. [Google Scholar] [CrossRef] [PubMed]
- Hyde, K.L.; Zatorre, R.J.; Peretz, I. Functional MRI evidence of an abnormal neural network for pitch processing in congenital amusia. Cereb. Cortex 2011, 21, 292–299. [Google Scholar] [CrossRef] [PubMed]
- Large, E.; Jones, M.R. The dynamics of attending: How people track time-varying events. Psychol. Rev. 1999, 106, 119–159. [Google Scholar] [CrossRef]
- Koelsch, S. Music-syntactic processing and auditory memory: Similarities and differences between ERAN and MMN. Psychophysiology 2009, 46, 179–190. [Google Scholar] [CrossRef] [PubMed]
- Garza Villarreal, E.A.; Brattico, E.; Leino, S.; Ostergaard, L.; Vuust, P. Distinct neural responses to chord violations: A multiple source analysis study. Brain Res. 2011, 1389, 103–114. [Google Scholar] [CrossRef] [PubMed]
- Koelsch, S.; Schmidt, B.-H.; Kansok, J. Effects of musical expertise on the early right anterior negativity: An event-related brain potential study. Psychophysiology 2002, 39, 657–663. [Google Scholar] [CrossRef] [PubMed]
- Brattico, E.; Tupala, T.; Glerean, E.; Tervaniemi, M. Modulated neural processing of Western harmony in folk musicians. Psychophysiology 2013, 50, 653–663. [Google Scholar] [CrossRef] [PubMed]
- Platel, H.; Baron, J.C.; Desgranges, B.; Bernard, F.; Eustache, F. Semantic and episodic memory of music are subserved by distinct neural networks. Neuroimage 2003, 20, 244–256. [Google Scholar] [CrossRef] [PubMed]
- Janata, P. Brain networks that track musical structure. Ann. N. Y. Acad. Sci. 2005, 1060, 111–124. [Google Scholar] [CrossRef] [PubMed]
- Alluri, V.; Toiviainen, P.; Jääskeläinen, I.P.; Glerean, E.; Sams, M.; Brattico, E. Large-scale brain networks emerge from dynamic processing of musical timbre, key and rhythm. Neuroimage 2012, 59, 3677–3689. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, S.J.; Spengler, S.; Simons, J.S.; Steele, J.D.; Lawrie, S.M.; Frith, C.D.; Burgess, P.W. Functional specialization within rostral prefrontal cortex (area10): A meta-analysis. J. Cogn. Neurosci. 2006, 18, 932–948. [Google Scholar] [CrossRef] [PubMed]
- Strait, D.; Parbery-Clark, A.; Hittner, E.; Kraus, N. Musical training during early childhood enhances the neural encoding of speech in noise. Brain Lang. 2012, 123, 191–201. [Google Scholar] [CrossRef] [PubMed]
- Schellenberg, E.G. Examining the association between music lessons and intelligence. Br. J. Psychol. 2011, 102, 283–302. [Google Scholar] [CrossRef] [PubMed]
- Seinfeld, S.; Figueroa, H.; Ortiz-Gil, J.; Sanchez-Vives, M.V. Effects of music learning and piano practice on cognitive function, mood and quality of life in older adults. Front. Psychol. 2013, 4, 1–13. [Google Scholar] [CrossRef]
- Oechslin, M.S.; Descloux, C.; Croquelois, A.; Chanal, J.; van de Ville, D.; Lazeyras, F.; James, C.E. Hippocampal Volume Predicts Fluid Intelligence in Musically Trained People. Hippocampus 2013, 23, 552–558. [Google Scholar] [CrossRef] [PubMed]
- James, C.E.; Cereghetti, D.M.; Tribes, E.R.; Oechslin, M.S. Electrophysiological evidence for a specific neural correlate of musical violation expectation in primary-school children. NeuroImage 2015, 104, 386–397. [Google Scholar] [CrossRef] [PubMed]
- Bogert, B.; Numminen-Kontti, T.; Numminen, J.; Gold, B.; Lampinen, J.; Sams, M.; Brattico, E. The neural substrates underlying explicit and implicit processing of emotions in music. In Proceedings of the OHBM Annual Meeting, Seattle, USA, 16–20 June 2013; Organization or Human Brain Mapping: Minneapolis, MN, USA, 2013. [Google Scholar]
- Koelsch, S. Brain correlates of music-evoked emotions. Nat. Rev. Neurosci. 2014, 15, 170–183. [Google Scholar] [CrossRef] [PubMed]
- Chapin, H.; Jantzen, K.; Kelso, J.S.; Steinberg, F.; Large, E. Dynamic emotional and neural responses to music depend on performance expression and listener experience. PLoS One 2010, 5, e13812. [Google Scholar] [CrossRef] [PubMed]
- Bigand, E.; Poulin-Charronnat, B. Are we “experienced listeners”? A review of the musical capacities that do not depend on formal musical training. Cognition 2006, 100, 100–130. [Google Scholar] [CrossRef] [PubMed]
- Lima, C.F.; Castro, S.L. Speaking to the trained ear: Musical expertise enhances the recognition of emotions in speech prosody. Emotion 2011, 11, 21–31. [Google Scholar] [CrossRef]
- Schön, D.; Magne, C.; Besson, M. The music of speech: Music training facilitates pitch processing in both music and language. Psychophysiology 2004, 41, 341–349. [Google Scholar] [CrossRef] [PubMed]
- Vieillard, S.; Roy, M.; Peretz, I. Expressiveness in musical emotions. Psychol. Res. 2012, 76, 641–653. [Google Scholar] [CrossRef] [PubMed]
- Park, M.; Gutyrchik, E.; Bao, Y.; Zavtseva, Y.; Carl, P.; Welker, L.; Pöppel, E.; Reiser, M.; Blautzik, J.; Meindl, T. Differences between musicians and non-musicians in neuro-affective processing of sadness and fear expressed in music. Neurosci. Lett. 2014, 566, 120–124. [Google Scholar] [CrossRef] [PubMed]
- Alluri, V.; Toiviainen, P.; Burunat, I.; Bogert, B.; Brattico, E. Musical training modulates liking-dependent connectivity during continuous listening. In Proceedings of the Neurosciences and Music V: Cognitive Stimulation and Rehabilitation, Dijon, France, 29 May–1 June 2014; Fondazione Mariani: Milano, Italy, 2014. [Google Scholar]
- Salimpoor, V.; Zald, D.H.; Zatorre, R.J.; Dagher, A.; McIntosh, A.R. Interactions between the nucleus accumbens and auditory cortices predicts music reward value. Science 2013, 340, 216–219. [Google Scholar] [CrossRef] [PubMed]
- Bever, T.G.; Chiarello, R.J. Cerebral dominance in musicians and nonmusicians. Science 1974, 185, 537–539. [Google Scholar] [CrossRef] [PubMed]
- Ohnishi, T.; Matsuda, H.; Asada, T.; Aruga, M.; Hirakata, M.; Nishikawa, M.; Katoh, A.; Imabayashi, E. Functional anatomy of musical perception in musicians. Cereb. Cortex 2001, 11, 754–760. [Google Scholar] [CrossRef] [PubMed]
- James, C.E.; Britz, J.; Vuilleumier, P.; Hauert, C.A.; Michel, C.M. Early neuronal responses in right limbic structures mediate harmony incongruity processing in musical experts. Neuroimage 2008, 42, 1597–1608. [Google Scholar] [CrossRef] [PubMed]
- Craig, A.D. The sentient self. Brain Struct. Funct. 2010, 214, 563–577. [Google Scholar] [CrossRef] [PubMed]
- Acitores, A.P. Towards a theory of proprioception as a bodily basis for consciousness in music. In Music and Consciousness. Philosophical, Psychological, and Cultural Perspectives; Clarke, D., Clarke, E., Eds.; Oxford University Press: Oxford, UK, 2011; pp. 215–230. [Google Scholar]
- Gallagher, S. How the Body Shapes the Mind; Clarendon Press: Oxford, UK, 2005. [Google Scholar]
- Zamorano, A.M.; Riquelme, I.; Kleber, B.; Altenmüller, E.; Hatem, S.M.; Montoya, P. Pain sensitivity and tactile spatial acuity are altered in healthy musicians as in chronic pain patients. Front. Hum. Neurosci. 2015, 6. [Google Scholar] [CrossRef]
- Steinmetz, A.; Scheffer, I.; Esmer, E.; Delank, K.S.; Peroz, I. Frequency, severity and predictors of playing-related musculoskeletal pain in professional orchestral musicians in Germany. Clin. Rheumatol. 2014. [Google Scholar] [CrossRef]
- Edelman, G. Bright Air, Brilliant Fire: On the Matter of Mind; Basis Books: New York, NY, USA, 1992. [Google Scholar]
- Müller, M.; Höfel, L.; Brattico, E.; Jacobsen, T. Aesthetic judgments of music in experts and laypersons—An ERP study. Int. J. Psychophysiol. 2010, 76, 40–51. [Google Scholar] [CrossRef] [PubMed]
- Berns, G.S.; Capra, C.M.; Moore, S.; Noussair, C. Neural mechanisms of the influence of popularity on adolescent ratings of music. Neuroimage 2010, 49, 2687–2696. [Google Scholar] [CrossRef] [PubMed]
- Salimpoor, V.N.; Benovoy, M.; Larcher, K.; Dagher, A.; Zatorre, R.J. Anatomically distinct dopamine releaseduring anticipation and experience of peak emotion to music. Nat. Neurosci. 2011, 14, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Menon, V.; Levitin, D. The rewards of music listening: Responses and physiological connectivity of the mesolimbic system. Neuroimage 2005, 28, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Koelsch, S.; Fritz, T.; Müller, K.; Friederici, A.D. Investigating emotion with music: An fMRI study. Hum. Brain Mapp. 2006, 27, 239–250. [Google Scholar] [CrossRef] [PubMed]
- Brattico, E.; Alluri, V.; Jacobsen, T.; Vartiainen, N.; Nieminen, S.; Tervaniemi, M. A functional MRI study of happy and sad emotions in music with and without lyrics. Front. Psychol. 2011, 2. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reybrouck, M.; Brattico, E. Neuroplasticity beyond Sounds: Neural Adaptations Following Long-Term Musical Aesthetic Experiences. Brain Sci. 2015, 5, 69-91. https://doi.org/10.3390/brainsci5010069
Reybrouck M, Brattico E. Neuroplasticity beyond Sounds: Neural Adaptations Following Long-Term Musical Aesthetic Experiences. Brain Sciences. 2015; 5(1):69-91. https://doi.org/10.3390/brainsci5010069
Chicago/Turabian StyleReybrouck, Mark, and Elvira Brattico. 2015. "Neuroplasticity beyond Sounds: Neural Adaptations Following Long-Term Musical Aesthetic Experiences" Brain Sciences 5, no. 1: 69-91. https://doi.org/10.3390/brainsci5010069
APA StyleReybrouck, M., & Brattico, E. (2015). Neuroplasticity beyond Sounds: Neural Adaptations Following Long-Term Musical Aesthetic Experiences. Brain Sciences, 5(1), 69-91. https://doi.org/10.3390/brainsci5010069





