Neuroprotective Mechanisms of Taurine against Ischemic Stroke
Abstract
:1. Introduction
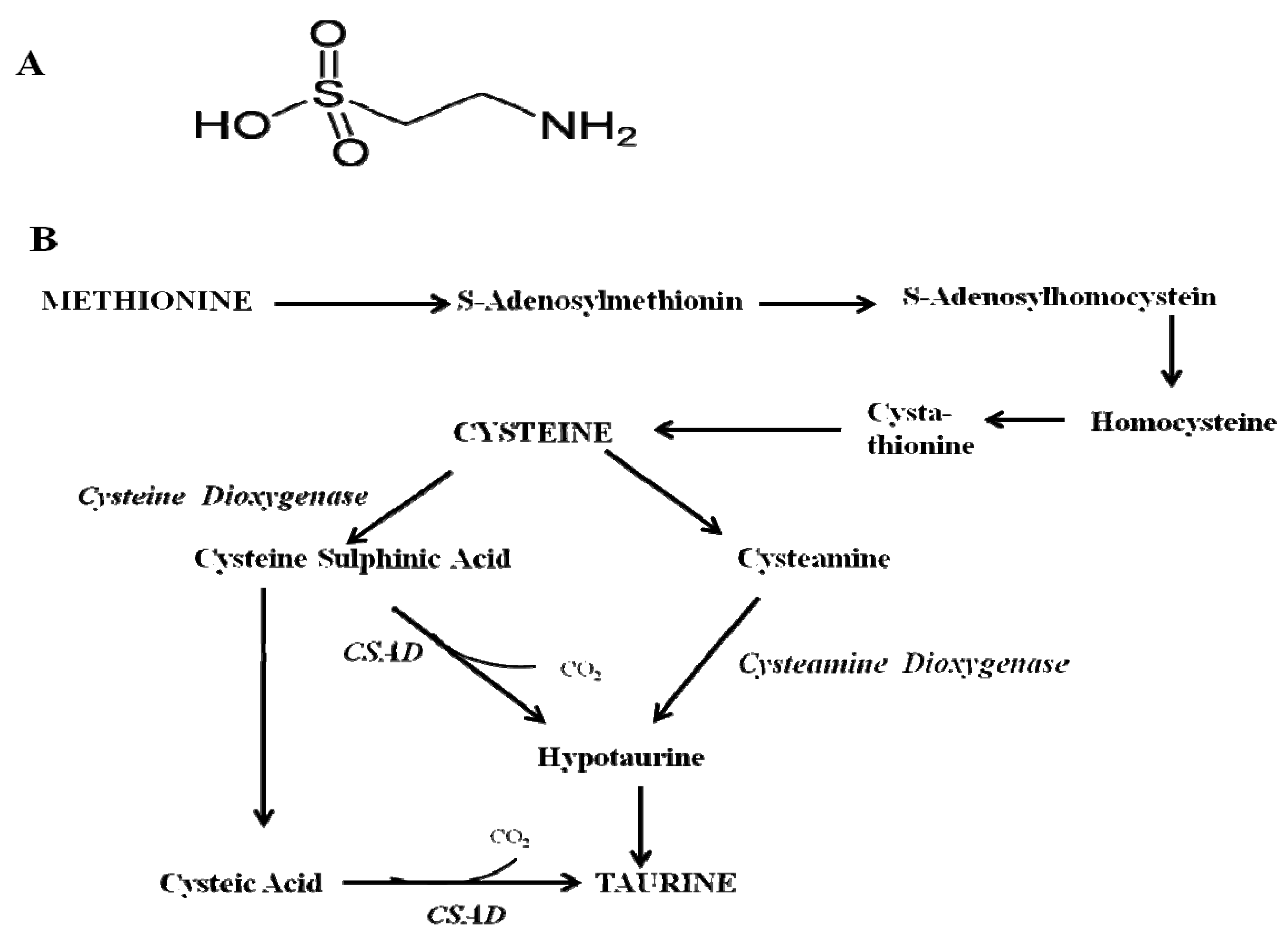
Physiological Function of Taurine in Central Nervous System
2. Neurochemical Mechanisms of Ischemic Stroke
3. Anti-Ischemic Stroke Mechanisms of Taurine
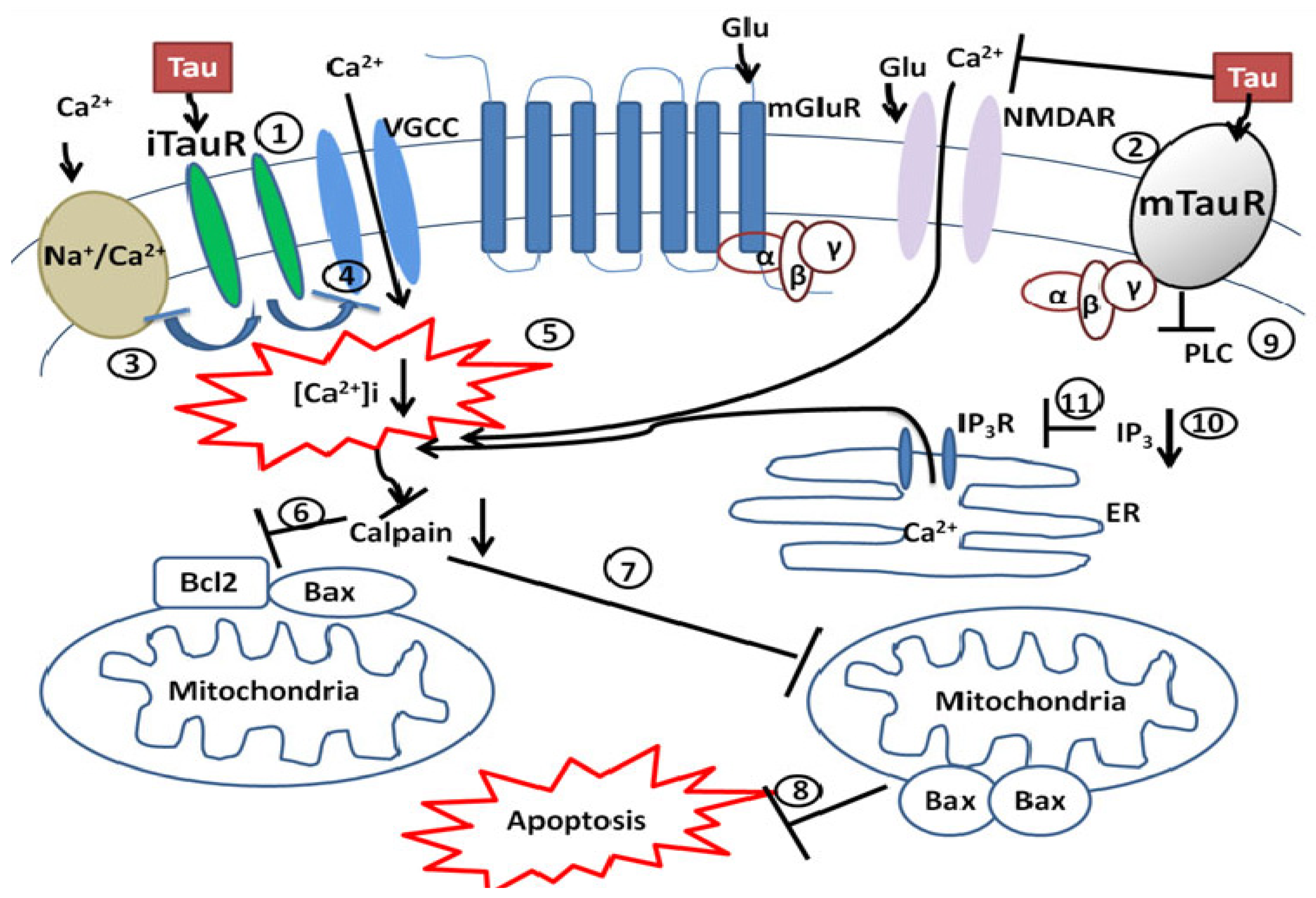
3.1. Taurine’s Neuroprotective Effect against Glutamate Excitotoxicity
3.2. Taurine’s Neuroprotective Effect on Mitochondrial Dysfunction, Calpain and Caspase Activities
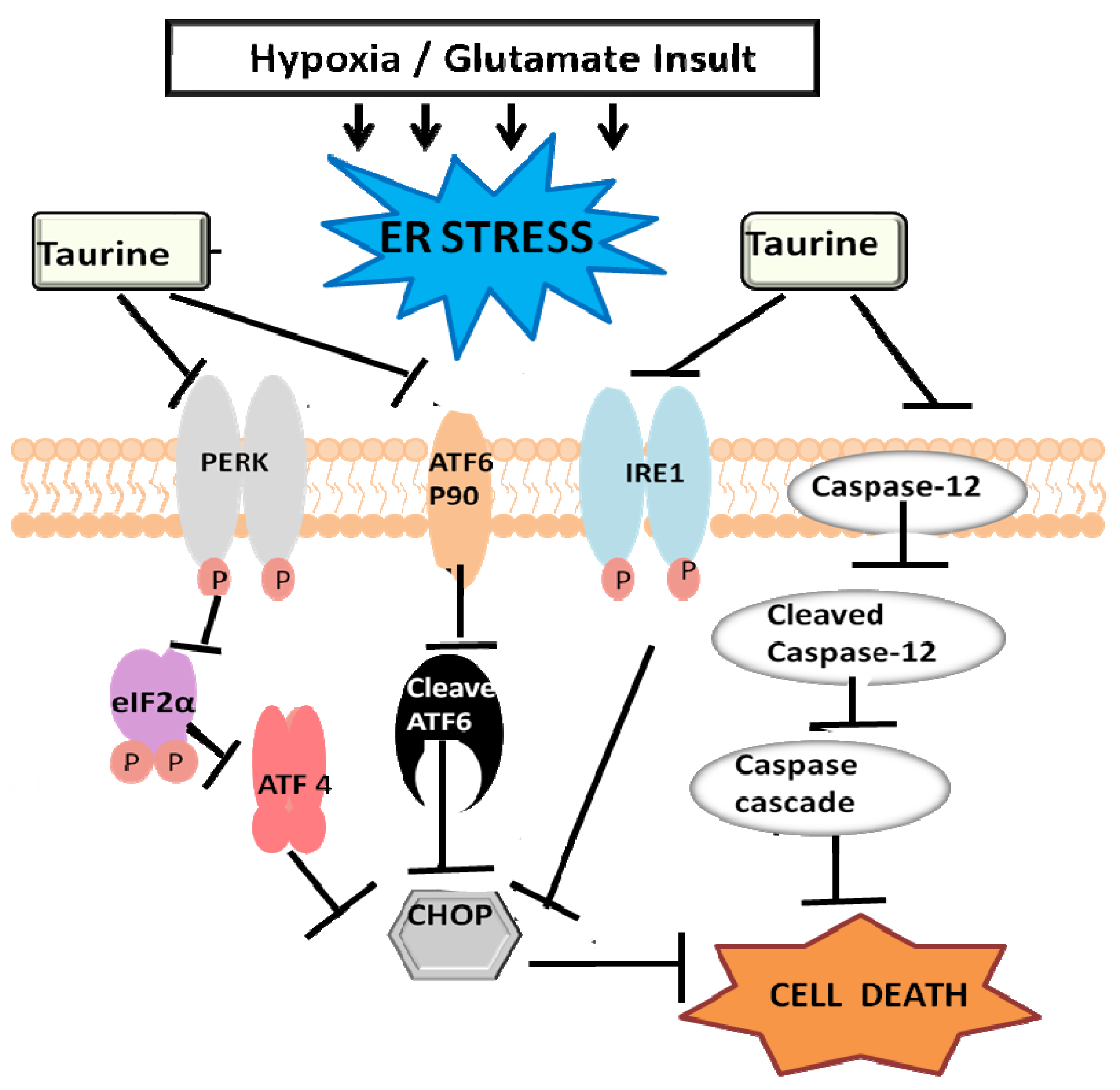
3.3. Taurine’s Neuroprotective Effect against Endoplasmic Reticulum Stress
3.4. Taurine’s Neuroprotective Effect against Oxidative Stress, Inflammation and Edema
4. Clinical Trial
5. Concluding Remarks
Acknowledgments
Conflict of Interest
References
- Feigin, V.L.; Lawes, C.M.M.; Bennett, D.A.; Barker-Collo, S.L.; Parag, V. Worldwide stroke incidence and early case fatality reported in 56 population-based studies: A systematic review. Lancet Neurol. 2009, 8, 355–369. [Google Scholar] [CrossRef]
- Strong, K.; Mathers, C.; Bonita, R. Preventing stroke: Saving lives around the world. Lancet Neurol. 2007, 6, 182–187. [Google Scholar] [CrossRef]
- MacKay, J.; Mensah, G. The Atlast of Heart Disease and Stroke; World Health Organization: Geneva, Switzerland, 2004; pp. 50–53. [Google Scholar]
- Tackling the global burden of stroke. Lancet Neurol. 2005, 4, 689. [CrossRef]
- Rosamond, W.; Flegal, K.; Friday, G.; Furie, K.; Go, A.; Greenlund, K.; Haase, N.; Ho, M.; Howard, V.; Kissela, B.; et al. Heart disease and stroke statistics—2007 update: A report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation 2007, 115, 169–171. [Google Scholar]
- Kirino, Y.; Yasukawa, T.; Ohta, S.; Akira, S.; Ishihara, K.; Watanabe, K.; Suzuki, T. Codon-specific translational defect caused by a wobble modification deficiency in mutant tRNA from a human mitochondrial disease. Proc. Natl. Acad. Sci. USA 2004, 101, 15070–15075. [Google Scholar] [CrossRef]
- Busl, K.M.; Greer, D.M. Hypoxic-ischemic brain injury: Pathophysiology, neuropathology and mechanims. NeuroRehabilitation 2010, 26, 5–13. [Google Scholar]
- Hossmann, K.A. Pathophysiology and therapy of experimental stroke. Cell. Mol. Neurobiol. 2006, 26, 1057–1083. [Google Scholar] [CrossRef]
- Hossmann, K.A. Viability thresholds and the penumbra of focal ischemia. Ann. Neurol. 1994, 36, 557–565. [Google Scholar] [CrossRef]
- Heiss, W.D.; Grond, M.; Thiel, A.; Von Stockhausen, H.M.; Rudolf, J.; Ghaemi, M.; Löttgen, J.; Stenzel, C.; Pawlik, G. Tissue at risk of infarction rescued by early reperfusion: A positron emission tomography study in systemic recombinant tissue plasminogen activator thrombolysis of acute stroke. J. Cereb. Blood Flow Metab. 1998, 18, 1298–1307. [Google Scholar]
- Obrenovitch, T.P. The ischaemic penumbra: Twenty years on. Cerebrovasc. Brain Metab. Rev. 1995, 7, 297–323. [Google Scholar]
- Fisher, M.; Garcia, J.H. Evolving stroke and the ischemic penumbra. Neurology 1996, 47, 884–888. [Google Scholar] [CrossRef]
- The National Institute of Neurological Disorders and Stroke (NINDS) rt-PA Stroke Study Group. Tissue Plasminogen Activator for Acute Ischemic Stroke. N. Engl. J. Med. 1995, 333, 1581–1588. [CrossRef]
- Hacke, W.; Kaste, M.; Bluhmki, E.; Brozman, M.; Dávalos, A.; Guidetti, D.; Larrue, V.; Lees, K.R.; Medeghri, Z.; Machnig, T.; et al. Thrombolysis with alteplase 3 to 4.5 hours after acute ischemic stroke. N. Engl. J. Med. 2008, 359, 1317–1329. [Google Scholar] [CrossRef]
- Jacobsen, J.G.; Smith, L.H. Biochemistry and physiology of taurine and taurine derivatives. Physiol. Rev. 1968, 48, 424–511. [Google Scholar]
- Reichelt, K.L.; Edminson, P.D. Biogenic amine specificity of cortical peptide synthesis in monkey brain. FEBS Lett. 1974, 47, 185–189. [Google Scholar] [CrossRef]
- Macaione, S.; Ruggeri, P.; De Luca, F.; Tucci, G. Free Amino Acids in Developing Rat Retina. J. Neurochem. 1974, 22, 887–891. [Google Scholar] [CrossRef]
- Huxtable, R.J. Metabolism and function of taurine in the heart. In Taurine; Raven Press: New York, NY, USA, 1976; pp. 99–119. [Google Scholar]
- Huxtable, R.J. Physiological actions of taurine. Physiol. Rev. 1992, 72, 101–163. [Google Scholar]
- Saransaari, P.; Oja, S.S. Taurine and neural cell damage. Amino Acids 2000, 19, 509–526. [Google Scholar] [CrossRef]
- Warskulat, U.; Flögel, U.; Jacoby, C.; Hartwig, H.-G.; Thewissen, M.; Merx, M.W.; Molojavyi, A.; Heller-Stilb, B.; Schrader, J.; Häussinger, D. Taurine transporter knockout depletes muscle taurine levels and results in severe skeletal muscle impairment but leaves cardiac function uncompromised. FASEB J. 2004, 18, 577–579. [Google Scholar]
- Oja, S.S.; Saransaari, P. Pharmacology of Taurine. Proc. West. Pharmacol. Soc. 2007, 50, 8–15. [Google Scholar]
- Wu, J.Y. Purification and characterization of cysteic acid and cysteine sulfinic acid decarboxylase and l-glutamate decarboxylase from bovine brain. Proc. Natl. Acad. Sci. USA 1982, 79, 4270–4274. [Google Scholar] [CrossRef]
- Hayes, K.C. Taurine requirement in primates. Nutr. Rev. 1985, 43, 65–70. [Google Scholar] [CrossRef]
- Stapleton, P.P.; Charles, R.P.; Redmond, H.P.; Bouchier-Hayes, D.J. Taurine and human nutrition. Clin. Nutr. 1997, 16, 103–108. [Google Scholar] [CrossRef]
- Yamori, Y.; Taguchi, T.; Hamada, A.; Kunimasa, K.; Mori, H.; Mori, M. Taurine in health and diseases: Consistent evidence from experimental and epidemiological studies. J. Biomed. Sci. 2010, 17, S6. [Google Scholar] [CrossRef]
- Curtis, D.R.; Watkins, J.C. The pharmacology of amino acids related to gamma-aminobutyric acid. Pharmacol. Rev. 1965, 17, 347–391. [Google Scholar]
- Davison, A.N.; Kaczmarek, L.K. Taurine—a possible neurotransmitter? Nature 1971, 234, 107–108. [Google Scholar] [CrossRef]
- Hayes, K.C.; Carey, R.E.; Schmidt, S.Y. Retinal degeneration associated with taurine deficiency in the cat. Science 1975, 188, 949–951. [Google Scholar]
- Imaki, H.; Jacobson, S.G.; Kemp, C.M.; Knighton, R.W.; Neuringer, M.; Sturman, J. Retinal morphology and visual pigment levels in 6- and 12-month-old rhesus monkeys fed a taurine-free human infant formula. J. Neurosci. Res. 1993, 36, 290–304. [Google Scholar] [CrossRef]
- Militante, J.D.; Lombardini, J.B. Taurine: Evidence of physiological function in the retina. Nutr. Neurosci. 2002, 5, 75–90. [Google Scholar] [CrossRef]
- Bulley, S.; Liu, Y.; Ripps, H.; Shen, W. Taurine activates delayed rectifier KV channels via a metabotropic pathway in retinal neurons. J. Physiol. 2013, 591, 123–132. [Google Scholar] [CrossRef]
- Ito, T.; Kimura, Y.; Uozumi, Y.; Takai, M.; Muraoka, S.; Matsuda, T.; Ueki, K.; Yoshiyama, M.; Ikawa, M.; Okabe, M.; Schaffer, S.W.; Fujio, Y.; Azuma, J. Taurine depletion caused by knocking out the taurine transporter gene leads to cardiomyopathy with cardiac atrophy. J. Mol. Cell. Cardiol. 2008, 44, 927–937. [Google Scholar] [CrossRef]
- Schaffer, S.W.; Jong, C.J.; Ramila, K.C.; Azuma, J. Physiological roles of taurine in heart and muscle. J. Biomed. Sci. 2010, 17, S2. [Google Scholar] [CrossRef]
- Sturman, J.A. Taurine in Development. Physiol. Rev. 1993, 73, 119–147. [Google Scholar]
- L’Amoreaux, W.J.; Cuttitta, C.; Santora, A.; Blaize, J.F.; Tachjadi, J.; El Idrissi, A. Taurine regulates insulin release from pancreatic beta cell lines. J. Biomed. Sci. 2010, 17, S11. [Google Scholar] [CrossRef]
- Sturman, J.A.; Moretz, R.C.; French, J.H.; Wisniewski, H.M. Taurine deficiency in the developing cat: Persistence of the cerebellar external granule cell layer. J. Neurosci. Res. 1985, 13, 405–416. [Google Scholar] [CrossRef]
- Sturman, J.A.; Moretz, R.C.; French, J.H.; Wisniewski, H.M. Postnatal taurine deficiency in the kitten results in a persistence of the cerebellar external granule cell layer: Correction by taurine feeding. J. Neurosci. Res. 1985, 13, 521–528. [Google Scholar] [CrossRef]
- Neuringer, M.; Palackal, T.; Kujawa, M.; Moretz, R.C.; Sturman, J.A. Visual cortex development in rhesus monkeys deprived of dietary taurine. Prog. Clin. Biol. Res. 1990, 351, 415–422. [Google Scholar]
- Hernández-Benítez, R.; Pasantes-Morales, H.; Saldaña, I.T.; Ramos-Mandujano, G. Taurine stimulates proliferation of mice embryonic cultured neural progenitor cells. J. Neurosci. Res. 2010, 88, 1673–1681. [Google Scholar]
- Hernández-Benítez, R.; Ramos-Mandujano, G.; Pasantes-Morales, H. Taurine stimulates proliferation and promotes neurogenesis of mouse adult cultured neural stem/progenitor cells. Stem Cell Res. 2012, 9, 24–34. [Google Scholar] [CrossRef]
- Bianchi, L.; Colivicchi, M.A.; Ballini, C.; Fattori, M.; Venturi, C.; Giovannini, M.G.; Healy, J.; Tipton, K.F.; Della Corte, L. Taurine, taurine analogues, and taurine functions: Overview. Adv. Exp. Med. Biol. 2006, 583, 443–448. [Google Scholar] [CrossRef]
- Ripps, H.; Shen, W. Review: Taurine: A “very essential” amino acid. Mol. Vis. 2012, 18, 2673–2686. [Google Scholar]
- Anuradha, C.V.; Balakrishnan, S.D. Taurine attenuates hypertension and improves insulin sensitivity in the fructose-fed rat, an animal model of insulin resistance. Can. J. Physiol. Pharmacol. 1999, 77, 749–754. [Google Scholar] [CrossRef]
- Nakaya, Y.; Minami, A.; Harada, N.; Sakamoto, S.; Niwa, Y.; Ohnaka, M. Taurine improves insulin sensitivity in the Otsuka Long-Evans Tokushima Fatty rat, a model of spontaneous type 2 diabetes. Am. J. Clin. Nutr. 2000, 71, 54–58. [Google Scholar]
- Ito, T.; Schaffer, S.W.; Azuma, J. The potential usefulness of taurine on diabetes mellitus and its complications. Amino Acids 2012, 42, 1529–1539. [Google Scholar] [CrossRef]
- Murakami, S. Taurine and atherosclerosis. Amino Acids 2012. [Google Scholar] [CrossRef]
- Menzie, J.; Pan, C.; Prentice, H.; Wu, J.-Y. Taurine and central nervous system disorders. Amino Acids 2012. [Google Scholar] [CrossRef]
- Pion, P.D.; Kittleson, M.D.; Rogers, Q.R.; Morris, J.G. Myocardial failure in cats associated with low plasma taurine: A reversible cardiomyopathy. Science 1987, 237, 764–768. [Google Scholar]
- Zulli, A. Taurine in cardiovascular disease. Curr. Opin. Clin. Nutr. Metab. Care 2011, 14, 57–60. [Google Scholar] [CrossRef]
- Tsuji, A.; Tamai, I. Sodium- and chloride-dependent transport of taurine at the blood-brain barrier. Adv. Exp. Med. Biol. 1996, 403, 385–391. [Google Scholar]
- Salimäki, J.; Scriba, G.; Piepponen, T.P.; Rautolahti, N.; Ahtee, L. The effects of systemically administered taurine and N-pivaloyltaurine on striatal extracellular dopamine and taurine in freely moving rats. Naunyn Schmiedebergs Arch. Pharmacol. 2003, 368, 134–141. [Google Scholar] [CrossRef]
- Huxtable, R.J. Taurine in the central nervous system and the mammalian actions of taurine. Prog. Neurobiol. 1989, 32, 471–533. [Google Scholar] [CrossRef]
- Wu, J.-Y.; Prentice, H. Role of taurine in the central nervous system. J. Biomed. Sci. 2010, 17, S1. [Google Scholar] [CrossRef]
- Kuriyama, K. Taurine as a neuromodulator. Fed. Proc. 1980, 39, 2680–2684. [Google Scholar]
- El Idrissi, A.; Trenkner, E. Taurine as a modulator of excitatory and inhibitory neurotransmission. Neurochem. Res. 2004, 29, 189–197. [Google Scholar] [CrossRef]
- Banerjee, R.; Vitvitsky, V.; Garg, S.K. The undertow of sulfur metabolism on glutamatergic neurotransmission. Trends Biochem. Sci. 2008, 33, 413–419. [Google Scholar] [CrossRef]
- Saransaari, P.; Oja, S.S. Taurine release in mouse brain stem slices under cell-damaging conditions. Amino Acids 2007, 32, 439–446. [Google Scholar] [CrossRef]
- Chen, W.Q.; Jin, H.; Nguyen, M.; Carr, J.; Lee, Y.J.; Hsu, C.C.; Faiman, M.D.; Schloss, J.V.; Wu, J.Y. Role of taurine in regulation of intracellular calcium level and neuroprotective function in cultured neurons. J. Neurosci. Res. 2001, 66, 612–619. [Google Scholar] [CrossRef]
- Foos, T.M.; Wu, J.-Y. The role of taurine in the central nervous system and the modulation of intracellular calcium homeostasis. Neurochem. Res. 2002, 27, 21–26. [Google Scholar] [CrossRef]
- El Idrissi, A.; Trenkner, E. Growth factors and taurine protect against excitotoxicity by stabilizing calcium homeostasis and energy metabolism. J. Neurosci. 1999, 19, 9459–9468. [Google Scholar]
- El Idrissi, A.; Trenkner, E. Taurine regulates mitochondrial calcium homeostasis. Adv. Exp. Med. Biol. 2003, 526, 527–536. [Google Scholar]
- El Idrissi, A. Taurine increases mitochondrial buffering of calcium: Role in neuroprotection. Amino Acids 2008, 34, 321–328. [Google Scholar] [CrossRef]
- Moran, J.; Salazar, P.; Pasantes-Morales, H. Effect of tocopherol and taurine on membrane fluidity of retinal rod outer segments. Exp. Eye Res. 1987, 45, 769–776. [Google Scholar] [CrossRef]
- Hagar, H.H. The protective effect of taurine against cyclosporine A-induced oxidative stress and hepatotoxicity in rats. Toxicol. Lett. 2004, 151, 335–343. [Google Scholar] [CrossRef]
- Schaffer, S.W.; Azuma, J.; Mozaffari, M. Role of antioxidant activity of taurine in diabetes. Can. J. Physiol. Pharmacol. 2009, 87, 91–99. [Google Scholar] [CrossRef]
- Miao, J.; Zhang, J.; Zheng, L.; Yu, X.; Zhu, W.; Zou, S. Taurine attenuates Streptococcus uberis-induced mastitis in rats by increasing T regulatory cells. Amino Acids 2012, 42, 2417–2428. [Google Scholar] [CrossRef]
- Sun, M.; Zhao, Y.; Gu, Y.; Xu, C. Anti-inflammatory mechanism of taurine against ischemic stroke is related to down-regulation of PARP and NF-κB. Amino Acids 2012, 42, 1735–1747. [Google Scholar] [CrossRef]
- Wu, H.; Jin, Y.; Wei, J.; Jin, H.; Sha, D.; Wu, J.-Y. Mode of action of taurine as a neuroprotector. Brain Res. 2005, 1038, 123–131. [Google Scholar] [CrossRef]
- Leon, R.; Wu, H.; Jin, Y.; Wei, J.; Buddhala, C.; Prentice, H.; Wu, J.-Y. Protective function of taurine in glutamate-induced apoptosis in cultured neurons. J. Neurosci. Res. 2009, 87, 1185–1194. [Google Scholar] [CrossRef]
- Junyent, F.; Romero, R.; de Lemos, L.; Utrera, J.; Camins, A.; Pallàs, M.; Auladell, C. Taurine treatment inhibits CaMKII activity and modulates the presence of calbindin D28k, calretinin, and parvalbumin in the brain. J. Neurosci. Res. 2010, 88, 136–142. [Google Scholar] [CrossRef]
- Pan, C.; Giraldo, G.S.; Prentice, H.; Wu, J.-Y. Taurine protection of PC12 cells against endoplasmic reticulum stress induced by oxidative stress. J. Biomed. Sci. 2010, 17, S17. [Google Scholar] [CrossRef]
- Pan, C.; Prentice, H.; Price, A.L.; Wu, J.-Y. Beneficial effect of taurine on hypoxia- and glutamate-induced endoplasmic reticulum stress pathways in primary neuronal culture. Amino Acids 2012, 43, 845–855. [Google Scholar]
- Lima, L.; Cubillos, S. Taurine might be acting as a trophic factor in the retina by modulating phosphorylation of cellular proteins. J. Neurosci. Res. 1998, 53, 377–384. [Google Scholar] [CrossRef]
- Kontro, P.; Oja, S.S. Sodium-independent taurine binding to brain synaptic membranes. Cell. Mol. Neurobiol. 1983, 3, 183–187. [Google Scholar]
- Wu, J.Y.; Moss, L.G.; Chen, M.S. Tissue and regional distribution of cysteic acid decarboxylase. A new assay method. Neurochem. Res. 1979, 4, 201–212. [Google Scholar] [CrossRef]
- Magnusson, K.R.; Clements, J.R.; Wu, J.Y.; Beitz, A.J. Colocalization of taurine- and cysteine sulfinic acid decarboxylase-like immunoreactivity in the hippocampus of the rat. Synapse 1989, 4, 55–69. [Google Scholar] [CrossRef]
- Philibert, R.A.; Rogers, K.L.; Dutton, G.R. Stimulus-coupled taurine efflux from cerebellar neuronal cultures: On the roles of Ca2+ and Na+. J. Neurosci. Res. 1989, 22, 167–171. [Google Scholar] [CrossRef]
- Martin, D.L. Synthesis and release of neuroactive substances by glial cells. Glia 1992, 5, 81–94. [Google Scholar] [CrossRef]
- Kozlowski, D.J.; Chen, Z.; Zhuang, L.; Fei, Y.J.; Navarre, S.; Ganapathy, V. Molecular characterization and expression pattern of taurine transporter in zebrafish during embryogenesis. Life Sci. 2008, 82, 1004–1011. [Google Scholar] [CrossRef]
- Kang, Y.-S. Downregulation of taurine transport by calcium blockers in osteoblast cells. Adv. Exp. Med. Biol. 2009, 643, 513–521. [Google Scholar] [CrossRef]
- Okamoto, K.; Kimura, H.; Sakai, Y. Taurine-induced increase of the Cl-conductance of cerebellar Purkinje cell dendrites in vitro. Brain Res. 1983, 259, 319–323. [Google Scholar] [CrossRef]
- Del Olmo, N.; Bustamante, J.; Del Río, R.M.; Solís, J.M. Taurine activates GABA(A) but not GABA(B) receptors in rat hippocampal CA1 area. Brain Res. 2000, 864, 298–307. [Google Scholar] [CrossRef]
- Albrecht, J.; Schousboe, A. Taurine interaction with neurotransmitter receptors in the CNS: An update. Neurochem. Res. 2005, 30, 1615–1621. [Google Scholar] [CrossRef]
- Wu, J.; Kohno, T.; Georgiev, S.K.; Ikoma, M.; Ishii, H.; Petrenko, A.B.; Baba, H. Taurine activates glycine and gamma-aminobutyric acid A receptors in rat substantia gelatinosa neurons. Neuroreport 2008, 19, 333–337. [Google Scholar] [CrossRef]
- Wu, J.Y.; Tang, X.W.; Tsai, W.H. Taurine receptor: Kinetic analysis and pharmacological studies. Adv. Exp. Med. Biol. 1992, 315, 263–268. [Google Scholar]
- Frosini, M.; Sesti, C.; Saponara, S.; Ricci, L.; Valoti, M.; Palmi, M.; Machetti, F.; Sgaragli, G. A specific taurine recognition site in the rabbit brain is responsible for taurine effects on thermoregulation. Br. J. Pharmacol. 2003, 139, 487–494. [Google Scholar] [CrossRef]
- Astrup, J.; Siesjö, B.K.; Symon, L. Thresholds in cerebral ischemia—the ischemic penumbra. Stroke 1981, 12, 723–725. [Google Scholar] [CrossRef]
- Kristian, T.; Siesjo, B.K. Calcium in ischemic cell death. Stroke 1998, 29, 705–718. [Google Scholar]
- Martin, R.L.; Lloyd, H.G.; Cowan, A.I. The early events of oxygen and glucose deprivation: Setting the scene for neuronal death? Trends Neurosci. 1994, 17, 251–257. [Google Scholar] [CrossRef]
- Katsura, K.; Kristian, T.; Siesjo, B.K. Energy metabolism, ion homeostasis, and cell damage in the brain. Biochem. Soc. Trans. 1994, 22, 991–996. [Google Scholar]
- Seki, Y.; Feustel, P.J.; Keller, R.W.; Tranmer, B.I.; Kimelberg, H.K.; Robinson, S.E. Inhibition of Ischemia-Induced Glutamate Release in Rat Striatum by Dihydrokinate and an Anion Channel Blocker Editorial Comment. Stroke 1999, 30, 433–440. [Google Scholar] [CrossRef]
- Globus, M.Y.-T.; Busto, R.; Dietrich, W.D.; Martinez, E.; Valdes, I.; Ginsberg, M.D. Effect of Ischemia on the in Vivo Release of Striatal Dopamine, Glutamate, and γ-Aminobutyric Acid Studied by Intracerebral Microdialysis. J. Neurochem. 1988, 51, 1455–1464. [Google Scholar] [CrossRef]
- Hillered, L.; Hallström, A.; Segersvärd, S.; Persson, L.; Ungerstedt, U. Dynamics of extracellular metabolites in the striatum after middle cerebral artery occlusion in the rat monitored by intracerebral microdialysis. J. Cereb. Blood Flow Metab. 1989, 9, 607–616. [Google Scholar] [CrossRef]
- Butcher, S.P.; Bullock, R.; Graham, D.I.; McCulloch, J. Correlation between amino acid release and neuropathologic outcome in rat brain following middle cerebral artery occlusion. Stroke 1990, 21, 1727–1733. [Google Scholar] [CrossRef]
- Yang, G.Y.; Betz, A.L. Reperfusion-induced injury to the blood-brain barrier after middle cerebral artery occlusion in rats. Stroke 1994, 25, 1658–1664. [Google Scholar] [CrossRef]
- O’Regan, M.H.; Smith-Barbour, M.; Perkins, L.M.; Phillis, J.W. A possible role for phospholipases in the release of neurotransmitter amino acids from ischemic rat cerebral cortex. Neurosci. Lett. 1995, 185, 191–194. [Google Scholar] [CrossRef]
- O’Regan, M.H.; Perkins, L.M.; Phillis, J.W. Arachidonic acid and lysophosphatidylcholine modulate excitatory transmitter amino acid release from the rat cerebral cortex. Neurosci. Lett. 1995, 193, 85–88. [Google Scholar] [CrossRef]
- Phillis, J.W.; Song, D.; O’Regan, M.H. Inhibition by anion channel blockers of ischemia-evoked release of excitotoxic and other amino acids from rat cerebral cortex. Brain Res. 1997, 758, 9–16. [Google Scholar] [CrossRef]
- Rutledge, E.M.; Aschner, M.; Kimelberg, H.K. Pharmacological characterization of swelling-induced d-[3H]aspartate release from primary astrocyte cultures. Am. J. Physiol. Cell Physiol. 1998, 274, 1511–1520. [Google Scholar]
- Feustel, P.J.; Jin, Y.; Kimelberg, H.K. Volume-regulated anion channels are the predominant contributors to release of excitatory amino acids in the ischemic cortical penumbra. Stroke 2004, 35, 1164–1168. [Google Scholar] [CrossRef]
- Szatkowski, M.; Barbour, B.; Attwell, D. Non-vesicular release of glutamate from glial cells by reversed electrogenic glutamate uptake. Nature 1990, 348, 443–446. [Google Scholar] [CrossRef]
- Rossi, D.J.; Oshima, T.; Attwell, D. Glutamate release in severe brain ischaemia is mainly by reversed uptake. Nature 2000, 403, 316–321. [Google Scholar] [CrossRef]
- Reuter, H.; Philipson, K.D. Sodium-calcium exchanger overexpression in the heart—insights from a transgenic mouse model. Basic Res. Cardiol. 2002, 97, 131–135. [Google Scholar]
- Meldrum, B.S. Glutamate as a Neurotransmitter in the Brain: Review of Physiology and Pathology. J. Nutr. 2000, 130, 1007S–1015S. [Google Scholar]
- Kwak, S.; Weiss, J.H. Calcium-permeable AMPA channels in neurodegenerative disease and ischemia. Curr. Opin. Neurobiol. 2006, 16, 281–287. [Google Scholar] [CrossRef]
- Berridge, M.J. Inositol Trisphosphate and Calcium Signaling. Ann. N. Y. Acad. Sci. 1995, 766, 31–43. [Google Scholar] [CrossRef]
- Sukhareva, M.; Smith, S.V.; Maric, D.; Barker, J.L. Functional Properties of Ryanodine Receptors in Hippocampal Neurons Change during Early Differentiation in Culture. J. Neurophysiol. 2002, 88, 1077–1087. [Google Scholar]
- Paschen, W.; Mengesdorf, T. Endoplasmic reticulum stress response and neurodegeneration. Cell Calcium 2005, 38, 409–415. [Google Scholar] [CrossRef]
- Ding, D.; Moskowitz, S.I.; Li, R.; Lee, S.B.; Esteban, M.; Tomaselli, K.; Chan, J.; Bergold, P.J. Acidosis induces necrosis and apoptosis of cultured hippocampal neurons. Exp. Neurol. 2000, 162, 1–12. [Google Scholar]
- Yermolaieva, O.; Leonard, A.S.; Schnizler, M.K.; Abboud, F.M.; Welsh, M.J. Extracellular acidosis increases neuronal cell calcium by activating acid-sensing ion channel 1a. Proc. Natl. Acad. Sci. USA 2004, 101, 6752–6757. [Google Scholar]
- Araújo, I.M.; Verdasca, M.J.; Leal, E.C.; Bahr, B.A.; Ambrósio, A.F.; Carvalho, A.P.; Carvalho, C.M. Early calpain-mediated proteolysis following AMPA receptor activation compromises neuronal survival in cultured hippocampal neurons. J. Neurochem. 2004, 91, 1322–1331. [Google Scholar] [CrossRef]
- Montague, J.; Gaido, M.; Frye, C.; Cidlowski, J. A calcium-dependent nuclease from apoptotic rat thymocytes is homologous with cyclophilin. Recombinant cyclophilins A, B, and C have nuclease activity. J. Biol. Chem. 1994, 269, 18877–18880. [Google Scholar]
- Farooqui, A.A.; Yang, H.-C.; Rosenberger, T.A.; Horrocks, L.A. Phospholipase A2 and Its Role in Brain Tissue. J. Neurochem. 2002, 69, 889–901. [Google Scholar] [CrossRef]
- Dykens, J.A. Isolated Cerebral and Cerebellar Mitochondria Produce Free Radicals when Exposed to Elevated Ca2+ and Na+: Implications for Neurodegeneration. J. Neurochem. 2002, 63, 584–591. [Google Scholar] [CrossRef]
- Lièvre, V.; Becuwe, P.; Bianchi, A.; Bossenmeyer-Pourié, C.; Koziel, V.; Franck, P.; Nicolas, M.; Dauça, M.; Vert, P.; Daval, J. Intracellular generation of free radicals and modifications of detoxifying enzymes in cultured neurons from the developing rat forebrain in response to transient hypoxia. Neuroscience 2001, 105, 287–297. [Google Scholar] [CrossRef]
- Duan, Y.; Gross, R.A.; Sheu, S.-S. Ca2+-dependent generation of mitochondrial reactive oxygen species serves as a signal for poly(ADP-ribose) polymerase-1 activation during glutamate excitotoxicity. J. Physiol. 2007, 585, 741–758. [Google Scholar] [CrossRef]
- Lipton, P. Ischemic Cell Death in Brain Neurons. Physiol. Rev. 1999, 79, 1431–1568. [Google Scholar]
- Nakka, V.P.; Gusain, A.; Mehta, S.L.; Raghubir, R. Molecular mechanisms of apoptosis in cerebral ischemia: Multiple neuroprotective opportunities. Mol. Neurobiol. 2008, 37, 7–38. [Google Scholar] [CrossRef]
- Pellegrini-Giampietro, D.E. The distinct role of mGlu1 receptors in post-ischemic neuronal death. Trends Pharmacol Sci. 2003, 24, 461–470. [Google Scholar] [CrossRef]
- Yamori, Y.; Liu, L.; Mori, M.; Sagara, M.; Murakami, S.; Nara, Y.; Mizushima, S. Taurine as the nutritional factor for the longevity of the Japanese revealed by a world-wide epidemiological survey. Adv. Exp. Med. Biol. 2009, 643, 13–25. [Google Scholar] [CrossRef]
- Wu, J.Y.; Lin, C.T.; Johansen, F.F.; Liu, J.W. Taurine neurons in rat hippocampal formation are relatively inert to cerebral ischemia. Adv. Exp. Med. Biol. 1994, 359, 289–298. [Google Scholar]
- Schurr, A.; Rigor, B.M. The mechanism of neuronal resistance and adaptation to hypoxia. FEBS Lett. 1987, 224, 4–8. [Google Scholar] [CrossRef]
- Matsumoto, K.; Lo, E.H.; Pierce, A.R.; Halpern, E.F.; Newcomb, R. Secondary elevation of extracellular neurotransmitter amino acids in the reperfusion phase following focal cerebral ischemia. J. Cereb. Blood Flow Metab. 1996, 16, 114–124. [Google Scholar]
- Torp, R.; Andiné, P.; Hagberg, H.; Karagülle, T.; Blackstad, T.W.; Ottersen, O.P. Cellular and subcellular redistribution of glutamate-, glutamine- and taurine-like immunoreactivities during forebrain ischemia: A semiquantitative electron microscopic study in rat hippocampus. Neuroscience 1991, 41, 433–447. [Google Scholar] [CrossRef]
- Uchiyama-Tsuyuki, Y.; Araki, H.; Yae, T.; Otomo, S. Changes in the Extracellular Concentrations of Amino Acids in the Rat Striatum During Transient Focal Cerebral Ischemia. J. Neurochem. 2008, 62, 1074–1078. [Google Scholar] [CrossRef]
- Lo, E.; Pierce, A.; Matsumoto, K.; Kano, T.; Evans, C.; Newcomb, R. Alterations in K+ evoked profiles of neurotransmitter and neuromodulator amino acids after focal ischemia-reperfusion. Neuroscience 1997, 83, 449–458. [Google Scholar] [CrossRef]
- Oja, S.S.; Saransaari, P. Modulation of taurine release by glutamate receptors and nitric oxide. Prog. Neurobiol. 2000, 62, 407–425. [Google Scholar] [CrossRef]
- Barakat, L.; Wang, D.; Bordey, A. Carrier-mediated uptake and release of taurine from Bergmann glia in rat cerebellar slices. J. Physiol. 2002, 541, 753–767. [Google Scholar] [CrossRef]
- Büyükuysal, R.L. Ischemia and reoxygenation induced amino acid release and tissue damage in the slices of rat corpus striatum. Amino Acids 2004, 27, 57–67. [Google Scholar]
- Saransaari, P.; Oja, S.S. Modulation of taurine release in ischemia by glutamate receptors in mouse brain stem slices. Amino Acids 2010, 38, 739–746. [Google Scholar] [CrossRef]
- Fariello, R.G.; Golden, G.T.; Pisa, M. Homotaurine (3 aminopropanesulfonic acid; 3APS) protects from the convulsant and cytotoxic effect of systemically administered kainic acid. Neurology 1982, 32, 241–245. [Google Scholar] [CrossRef]
- French, E.D.; Vezzani, A.; Whetsell, W.O.; Schwarcz, R. Anti-excitotoxic actions of taurine in the rat hippocampus studied in vivo and in vitro. Adv. Exp. Med. Biol. 1986, 203, 349–362. [Google Scholar] [CrossRef]
- Wu, J.Y.; Johansen, F.F.; Lin, C.T.; Liu, J.W. Taurine system in the normal and ischemic rat hippocampus. Adv. Exp. Med. Biol. 1987, 217, 265–274. [Google Scholar]
- Trenkner, E. The role of taurine and glutamate during early postnatal cerebellar development of normal and Weaver mutant mice. Adv. Exp. Med. Biol. 1990, 268, 239–244. [Google Scholar]
- Tang, X.W.; Deupree, D.L.; Sun, Y.; Wu, J.Y. Biphasic effect of taurine on excitatory amino acid-induced neurotoxicity. Adv. Exp. Med. Biol. 1996, 403, 499–505. [Google Scholar]
- Wu, J.; Chen, W.; Tang, X.W.; Jin, H.; Foos, T.; Schloss, J.V.; Davis, K.; Faiman, M.D.; Hsu, C. Mode of Action of Taurine and regulation Dynamics of Its Synthesis in the CNS. Adv. Exp. Med. Biol. 2000, 483, 35–44. [Google Scholar]
- Wu, J.-Y.; Wu, H.; Jin, Y.; Wei, J.; Sha, D.; Prentice, H.; Lee, H.-H.; Lin, C.-H.; Lee, Y.-H.; Yang, L.-L. Mechanism of neuroprotective function of taurine. Adv. Exp. Med. Biol. 2009, 643, 169–179. [Google Scholar] [CrossRef]
- Chen, W. Mode of Action of Taurine. Ph.D Thesis, University of Kansas, Lawrence, KS, USA, 2000. [Google Scholar]
- Schurr, A.; Tseng, M.T.; West, C.A.; Rigor, B.M. Taurine improves the recovery of neuronal function following cerebral hypoxia: An in vitro study. Life Sci. 1987, 40, 2059–2066. [Google Scholar] [CrossRef]
- Ricci, L.; Valoti, M.; Sgaragli, G.; Frosini, M. Protection by taurine of rat brain cortical slices against oxygen glucose deprivation- and reoxygenation-induced damage. Eur. J. Pharmacol. 2009, 621, 26–32. [Google Scholar] [CrossRef]
- Zhao, P.; Huang, Y.L.; Cheng, J.S. Taurine antagonizes calcium overload induced by glutamate or chemical hypoxia in cultured rat hippocampal neurons. Neurosci. Lett. 1999, 268, 25–28. [Google Scholar] [CrossRef]
- Wang, G.-H.; Jiang, Z.-L.; Fan, X.-J.; Zhang, L.; Li, X.; Ke, K.-F. Neuroprotective effect of taurine against focal cerebral ischemia in rats possibly mediated by activation of both GABAA and glycine receptors. Neuropharmacology 2007, 52, 1199–1209. [Google Scholar] [CrossRef]
- Molchanova, S.M.; Oja, S.S.; Saransaari, P. Taurine attenuates d-[3H]aspartate release evoked by depolarization in ischemic corticostriatal slices. Brain Res. 2006, 1099, 64–72. [Google Scholar] [CrossRef]
- Nicholls, D.G. Mitochondrial calcium function and dysfunction in the central nervous system. Biochim. Biophys. Acta 2009, 1787, 1416–1424. [Google Scholar] [CrossRef]
- Brustovetsky, N.; Brustovetsky, T.; Jemmerson, R.; Dubinsky, J.M. Calcium-induced cytochrome c release from CNS mitochondria is associated with the permeability transition and rupture of the outer membrane. J. Neurochem. 2002, 80, 207–218. [Google Scholar] [CrossRef]
- Bernardi, P.; Krauskopf, A.; Basso, E.; Petronilli, V.; Blachly-Dyson, E.; Blalchy-Dyson, E.; Di Lisa, F.; Forte, M.A. The mitochondrial permeability transition from in vitro artifact to disease target. FEBS J. 2006, 273, 2077–2099. [Google Scholar] [CrossRef]
- Jemmerson, R.; Dubinsky, J.M.; Brustovetsky, N. Cytochrome C release from CNS mitochondria and potential for clinical intervention in apoptosis-mediated CNS diseases. Antioxid. Redox Signal. 2005, 7, 1158–1172. [Google Scholar] [CrossRef]
- Dedkova, E.N.; Ji, X.; Lipsius, S.L.; Blatter, L.A. Mitochondrial calcium uptake stimulates nitric oxide production in mitochondria of bovine vascular endothelial cells. Am J. Physiol. Cell Physiol. 2004, 286, 406–415. [Google Scholar] [CrossRef]
- Ottersen, O.P. Quantitative assessment of taurine-like immunoreactivity in different cell types and processes in rat cerebellum: An electronmicroscopic study based on a postembedding immunogold labelling procedure. Anat. Embryol. 1988, 178, 407–421. [Google Scholar] [CrossRef]
- Lobo, M.V.; Alonso, F.J.; Martin del Rio, R. Immunocytochemical localization of taurine in different muscle cell types of the dog and rat. Histochem. J. 2000, 32, 53–61. [Google Scholar] [CrossRef]
- Bollard, M.E.; Murray, A.J.; Clarke, K.; Nicholson, J.K.; Griffin, J.L. A study of metabolic compartmentation in the rat heart and cardiac mitochondria using high-resolution magic angle spinning 1H NMR spectroscopy. FEBS Lett. 2003, 553, 73–78. [Google Scholar] [CrossRef]
- Palmi, M.; Youmbi, G.T.; Fusi, F.; Sgaragli, G.P.; Dixon, H.B.; Frosini, M.; Tipton, K.F. Potentiation of mitochondrial Ca2+ sequestration by taurine. Biochem. Pharmacol. 1999, 58, 1123–1131. [Google Scholar] [CrossRef]
- Hansen, S.H.; Andersen, M.L.; Birkedal, H.; Cornett, C.; Wibrand, F. The important role of taurine in oxidative metabolism. Adv. Exp. Med. Biol. 2006, 583, 129–135. [Google Scholar] [CrossRef]
- Chen, K.; Zhang, Q.; Wang, J.; Liu, F.; Mi, M.; Xu, H.; Chen, F.; Zeng, K. Taurine protects transformed rat retinal ganglion cells from hypoxia-induced apoptosis by preventing mitochondrial dysfunction. Brain Res. 2009, 1279, 131–138. [Google Scholar] [CrossRef]
- Hansen, S.H.; Andersen, M.L.; Cornett, C.; Gradinaru, R.; Grunnet, N. A role for taurine in mitochondrial function. J. Biomed. Sci. 2010, 17, S23. [Google Scholar] [CrossRef]
- Suzuki, T.; Suzuki, T.; Wada, T.; Saigo, K.; Watanabe, K. Taurine as a constituent of mitochondrial tRNAs: New insights into the functions of taurine and human mitochondrial diseases. EMBO J. 2002, 21, 6581–6589. [Google Scholar] [CrossRef]
- Yasukawa, T.; Suzuki, T.; Ueda, T.; Ohta, S.; Watanabe, K. Modification defect at anticodon wobble nucleotide of mitochondrial tRNAs(Leu)(UUR) with pathogenic mutations of mitochondrial myopathy, encephalopathy, lactic acidosis, and stroke-like episodes. J. Biol. Chem. 2000, 275, 4251–4257. [Google Scholar]
- Suzuki, T.; Nagao, A.; Suzuki, T. Human mitochondrial diseases caused by lack of taurine modification in mitochondrial tRNAs. Wiley Interdiscip. Rev. RNA 2011, 2, 376–386. [Google Scholar] [CrossRef]
- Rikimaru, M.; Ohsawa, Y.; Wolf, A.M.; Nishimaki, K.; Ichimiya, H.; Kamimura, N.; Nishimatsu, S. Taurine Ameliorates Impaired the Mitochondrial Function and Prevents Stroke-like Episodes in Patients with MELAS. Intern. Med. 2012, 51, 3351–3357. [Google Scholar]
- Thornberry, N.A. Caspases: Enemies Within. Science 1998, 281, 1312–1316. [Google Scholar] [CrossRef]
- Endres, M.; Namura, S.; Shimizu-Sasamata, M.; Waeber, C.; Zhang, L.; Gómez-Isla, T.; Hyman, B.T.; Moskowitz, M.A. Attenuation of delayed neuronal death after mild focal ischemia in mice by inhibition of the caspase family. J. Cereb. Blood Flow Metab. 1998, 18, 238–247. [Google Scholar]
- Rami, A.; Agarwal, R.; Botez, G.; Winckler, J. μ-Calpain activation, DNA fragmentation, and synergistic effects of caspase and calpain inhibitors in protecting hippocampal neurons from ischemic damage. Brain Res. 2000, 866, 299–312. [Google Scholar] [CrossRef]
- Love, S. Apoptosis and brain ischaemia. Prog. Neuropsychopharmacol. Biol. Psychiatry 2003, 27, 267–282. [Google Scholar] [CrossRef]
- Kambe, A.; Yokota, M.; Saido, T.C.; Satokata, I.; Fujikawa, H.; Tabuchi, S.; Kamitani, H.; Watanabe, T. Spatial resolution of calpain-catalyzed proteolysis in focal cerebral ischemia. Brain Res. 2005, 1040, 36–43. [Google Scholar] [CrossRef]
- Han, F.; Shirasaki, Y.; Fukunaga, K. 3-[2-[4-(3-Chloro-2-methylphenylmethyl)-1-piperazinyl]ethyl]-5,6-dimethoxy-1-(4-imidazolylmethyl)-1H-indazole dihydro-chloride 3.5 hydrate (DY-9760e) is neuroprotective in rat microsphere embolism: Role of the cross-talk between calpain and caspase-3 through calpastatin. J. Pharmacol. Exp. Ther. 2006, 317, 529–536. [Google Scholar] [CrossRef]
- Goll, D.E.; Thompson, V.F.; Li, H.; Wei, W.; Cong, J. The Calpain System. Physiol. Rev. 1990, 731–801. [Google Scholar]
- Wang, K.K. Calpain and caspase: Can you tell the difference? Trends Neurosci. 2000, 23, 20–26. [Google Scholar] [CrossRef]
- Ling, Y.-H.; Liebes, L.; Ng, B.; Buckley, M.; Elliott, P.J.; Adams, J.; Jiang, J.-D.; Muggia, F.M.; Perez-Soler, R. PS-341, a Novel Proteasome Inhibitor, Induces Bcl-2 Phosphorylation and Cleavage in Association with G2-M Phase Arrest and Apoptosis. Mol. Cancer Ther. 2002, 1, 841–849. [Google Scholar]
- Nakagawa, T.; Yuan, J. Cross-Talk between Two Cysteine Protease Families: Activation of Caspase-12 by Calpain in Apoptosis. J. Cell Biol. 2000, 150, 887–894. [Google Scholar] [CrossRef]
- Gao, G.; Dou, Q.P. N-terminal cleavage of bax by calpain generates a potent proapoptotic 18-kDa fragment that promotes bcl-2-independent cytochrome c release and apoptotic cell death. J. Cell Biochem. 2000, 80, 53–72. [Google Scholar] [CrossRef]
- Castro, R.E.; Solá, S.; Ramalho, R.M.; Steer, C.J.; Rodrigues, C.M.P. The bile acid tauroursodeoxycholic acid modulates phosphorylation and translocation of bad via phosphatidylinositol 3-kinase in glutamate-induced apoptosis of rat cortical neurons. J. Pharmacol. Exp. Ther. 2004, 311, 845–852. [Google Scholar] [CrossRef]
- Gil-Parrado, S.; Fernández-Montalván, A.; Assfalg-Machleidt, I.; Popp, O.; Bestvater, F.; Holloschi, A.; Knoch, T.A.; Auerswald, E.A.; Welsh, K.; Reed, J.C.; et al. Ionomycin-activated calpain triggers apoptosis. A probable role for Bcl-2 family members. J. Biol. Chem. 2002, 277, 27217–27226. [Google Scholar] [CrossRef]
- Salvesen, G.S.; Dixit, V.M. Caspases: Intracellular Signaling by Proteolysis. Cell 1997, 91, 443–446. [Google Scholar] [CrossRef]
- Li, P.; Nijhawan, D.; Budihardjo, I.; Srinivasula, S.M.; Ahmad, M.; Alnemri, E.S.; Wang, X. Cytochrome c and dATP-dependent formation of Apaf-1/caspase-9 complex initiates an apoptotic protease cascade. Cell 1997, 91, 479–489. [Google Scholar] [CrossRef]
- Takatani, T.; Takahashi, K.; Uozumi, Y.; Shikata, E.; Yamamoto, Y.; Ito, T.; Matsuda, T.; Schaffer, S.W.; Fujio, Y.; Azuma, J. Taurine inhibits apoptosis by preventing formation of the Apaf-1/caspase-9 apoptosome. Am. J. Physiol. Cell Physiol. 2004, 287, 949–953. [Google Scholar] [CrossRef]
- Sun, M.; Xu, C. Neuroprotective mechanism of taurine due to up-regulating calpastatin and down-regulating calpain and caspase-3 during focal cerebral ischemia. Cell Mol. Neurobiol. 2008, 28, 593–611. [Google Scholar] [CrossRef]
- Sun, M.; Gu, Y.; Zhao, Y.; Xu, C. Protective functions of taurine against experimental stroke through depressing mitochondria-mediated cell death in rats. Amino Acids 2011, 40, 1419–1429. [Google Scholar] [CrossRef]
- Taranukhin, A.G.; Taranukhina, E.Y.; Saransaari, P.; Djatchkova, I.M.; Pelto-Huikko, M.; Oja, S.S. Taurine reduces caspase-8 and caspase-9 expression induced by ischemia in the mouse hypothalamic nuclei. Amino Acids 2008, 34, 169–174. [Google Scholar] [CrossRef]
- Kumar, R.; Azam, S.; Sullivan, J.M.; Owen, C.; Cavener, D.R.; Zhang, P.; Ron, D.; Harding, H.P.; Chen, J.-J.; Han, A.; et al. Brain ischemia and reperfusion activates the eukaryotic initiation factor 2α kinase, PERK. J. Neurochem. 2001, 77, 1418–1421. [Google Scholar] [CrossRef]
- Azfer, A.; Niu, J.; Rogers, L.M.; Adamski, F.M.; Kolattukudy, P.E. Activation of endoplasmic reticulum stress response during the development of ischemic heart disease. Am. J. Physiol. Heart Circ. Physiol. 2006, 291, 1411–1420. [Google Scholar] [CrossRef]
- Kuznetsov, G.; Brostrom, M.; Brostrom, C. Demonstration of a calcium requirement for secretory protein processing and export. Differential effects of calcium and dithiothreitol. J. Biol. Chem. 1992, 267, 3932–3939. [Google Scholar]
- Verkhratsky, A.; Toescu, E.C. Endoplasmic reticulum Ca(2+) homeostasis and neuronal death. J. Cell. Mol. Med. 2003, 7, 351–361. [Google Scholar] [CrossRef]
- Pizzo, P.; Pozzan, T. Mitochondria-endoplasmic reticulum choreography: Structure and signaling dynamics. Trends Cell Biol. 2007, 17, 511–517. [Google Scholar] [CrossRef]
- Morimoto, N.; Oida, Y.; Shimazawa, M.; Miura, M.; Kudo, T.; Imaizumi, K.; Hara, H. Involvement of endoplasmic reticulum stress after middle cerebral artery occlusion in mice. Neuroscience 2007, 14, 957–967. [Google Scholar]
- Malhotra, J.D.; Kaufman, R.J. The endoplasmic reticulum and the unfolded proteinresponse. Semin. Cell Dev. Biol. 2007, 18, 716–731. [Google Scholar] [CrossRef]
- Rao, R.V.; Bredesen, D.E. Misfolded proteins, endoplasmic reticulum stress and neurodegeneration. Curr. Opin. Cell Biol. 2004, 16, 653–662. [Google Scholar] [CrossRef]
- Ron, D.; Walter, P. Signal integration in the endoplasmic reticulum unfolded protein response. Nat. Rev. Mol. Cell Biol. 2007, 8, 519–529. [Google Scholar] [CrossRef]
- Chakrabarti, A.; Chen, A.W.; Varner, J.D. A review of the mammalian unfolded protein response. Biotechnol. Bioeng. 2011, 108, 2777–2793. [Google Scholar] [CrossRef]
- Gharibani, P.M.; Modi, J.; Pan, C.; Menzie, J.; Ma, Z.; Chen, P.C.; Tao, R.; Prentice, H.; Wu, J.Y. The Mechanism of Taurine Protection Against Endoplasmic recticulum Stress in an Animal Stroke Model of Cerebral Artery Occlusion and Stroke-Related Conditions in Primary Neuronal Cell Culture. Adv. Exp. Med. Biol. 2013, 776, 241–258. [Google Scholar] [CrossRef]
- Ghosh, A.P.; Klocke, B.J.; Ballestas, M.E.; Roth, K.A. CHOP potentially co-operates with FOXO3a in neuronal cells to regulate PUMA and BIM expression in response to ER stress. PLoS One 2012, 7, e39586. [Google Scholar]
- McCullough, K.D.; Martindale, J.L.; Klotz, L.O.; Aw, T.Y.; Holbrook, N.J. Gadd153 sensitizes cells to endoplasmic reticulum stress by down-regulating Bcl2 and perturbing the cellular redox state. Mol. Cell. Biol. 2001, 21, 1249–1259. [Google Scholar] [CrossRef]
- Schuller-Levis, G.B.; Park, E. Taurine and Its Chloramine: Modulators of Immunity. Neurochem Res. 2004, 29, 117–126. [Google Scholar] [CrossRef]
- Halliwell, B. Reactive oxygen species and the central nervous system. J. Neurochem. 1992, 59, 1609–1623. [Google Scholar] [CrossRef]
- Zini, I.; Tomasi, A.; Grimaldi, R.; Vannini, V.; Agnati, L.F. Detection of free radicals during brain ischemia and reperfusion by spin trapping and microdialysis. Neurosci. Lett. 1992, 138, 279–282. [Google Scholar] [CrossRef]
- Sohal, R.S.; Orr, W.C. Relationship between antioxidants, prooxidants, and the aging proces. Ann. N. Y. Acad. Sci. 1992, 663, 74–84. [Google Scholar] [CrossRef]
- Turrens, J.F. Mitochondrial formation of reactive oxygen species. J. Physiol. 2003, 552, 335–344. [Google Scholar] [CrossRef]
- Brookes, P.S.; Darley-Usmar, V.M. Role of calcium and superoxide dismutase in sensitizing mitochondria to peroxynitrite-induced permeability transition. Am. J. Physiol. Heart Circ. Physiol. 2004, 286, 39–46. [Google Scholar] [CrossRef]
- Wikström, M.; Saari, H. A spectral shift in cytochrome a induced by calcium ions. Biochim. Biophys. Acta 1975, 408, 170–179. [Google Scholar] [CrossRef]
- Jekabsone, A.; Ivanoviene, L.; Brown, G.C.; Borutaite, V. Nitric oxide and calcium together inactivate mitochondrial complex I and induce cytochrome c release. J. Mol. Cell. Cardiol. 2003, 35, 803–809. [Google Scholar] [CrossRef]
- Grijalba, M.T.; Vercesi, A.E.; Schreier, S. Ca2+-induced increased lipid packing and domain formation in submitochondrial particles. A possible early step in the mechanism of Ca2+-stimulated generation of reactive oxygen species by the respiratory chain. Biochemistry 1999, 38, 13279–13287. [Google Scholar] [CrossRef]
- Cleeter, M.W.; Cooper, J.M.; Darley-Usmar, V.M.; Moncada, S.; Schapira, A.H. Reversible inhibition of cytochrome c oxidase, the terminal enzyme of the mitochondrial respiratory chain, by nitric oxide. Implications for neurodegenerative diseases. FEBS Lett. 1994, 345, 50–54. [Google Scholar] [CrossRef]
- Zoccarato, F.; Cavallini, L.; Alexandre, A. Respiration-dependent removal of exogenous H2O2 in brain mitochondria: Inhibition by Ca2+. J. Biol. Chem. 2004, 279, 4166–4174. [Google Scholar] [CrossRef]
- Patterson, S.D.; Spahr, C.S.; Daugas, E.; Susin, S.A.; Irinopoulou, T.; Koehler, C.; Kroemer, G. Mass spectrometric identification of proteins released from mitochondria undergoing permeability transition. Cell Death Differ. 2000, 7, 137–144. [Google Scholar] [CrossRef]
- Ricci, C.; Pastukh, V.; Leonard, J.; Turrens, J.; Wilson, G.; Schaffer, D.; Schaffer, S.W. Mitochondrial DNA damage triggers mitochondrial-superoxide generation and apoptosis. Am. J. Physiol. 2008, 294, 413–422. [Google Scholar]
- Mochizuki, H.; Oda, H.; Yokogoshi, H. Dietary taurine alters ascorbic acid metabolism in rats fed diets containing polychlorinated biphenyls. J. Nutr. 2000, 130, 873–876. [Google Scholar]
- Vohra, B.P.; Hui, X. Taurine protects against carbon tetrachloride toxicity in the cultured neurons and in vivo. Arch. Physiol. Biochem. 2001, 109, 90–94. [Google Scholar] [CrossRef]
- Balkan, J.; Kanbağli, O.; Aykaç-Toker, G.; Uysal, M. Taurine treatment reduces hepatic lipids and oxidative stress in chronically ethanol-treated rats. Biol. Pharm. Bull. 2002, 25, 1231–1233. [Google Scholar] [CrossRef]
- Mahalakshmi, K.; Pushpakiran, G.; Anuradha, C.V. Taurine prevents acrylonitrile-induced oxidative stress in rat brain. Pol. J. Pharmacol. 2003, 55, 1037–1043. [Google Scholar]
- Pushpakiran, G.; Mahalakshmi, K.; Anuradha, C.V. Taurine restores ethanol-induced depletion of antioxidants and attenuates oxidative stress in rat tissues. Amino Acids 2004, 27, 91–96. [Google Scholar]
- Aruoma, O.I.; Halliwell, B.; Hoey, B.M.; Butler, J. The antioxidant action of taurine, hypotaurine and their metabolic precursors. Biochem. J. 1988, 256, 251–255. [Google Scholar]
- Jong, C.J.; Azuma, J.; Schaffer, S. Mechanism underlying the antioxidant activity of taurine: Prevention of mitochondrial oxidant production. Amino Acids 2012, 42, 2223–2232. [Google Scholar] [CrossRef]
- Jong, C.J.; Ito, T.; Mozaffari, M.; Azuma, J.; Schaffer, S. Effect of beta-alanine treatment on mitochondrial taurine level and 5-taurinomethyluridine content. J. Biomed. Sci. 2010, 17, S25. [Google Scholar] [CrossRef]
- Amantea, D.; Nappi, G.; Bernardi, G.; Bagetta, G.; Corasaniti, M.T. Post-ischemic brain damage: Pathophysiology and role of inflammatory mediators. FEBS J. 2009, 276, 13–26. [Google Scholar] [CrossRef]
- Koh, S.-H.; Chang, D.-I.; Kim, H.-T.; Kim, J.; Kim, M.-H.; Kim, K. S.; Bae, I.; Kim, H.; Kim, D.W.; Kim, S.H. Effect of 3-aminobenzamide, PARP inhibitor, on matrix metalloproteinase-9 level in plasma and brain of ischemic stroke model. Toxicology 2005, 214, 131–139. [Google Scholar] [CrossRef]
- Haddad, M.; Rhinn, H.; Bloquel, C.; Coqueran, B.; Szabó, C.; Plotkine, M.; Scherman, D.; Margaill, I. Anti-inflammatory effects of PJ34, a poly(ADP-ribose) polymerase inhibitor, in transient focal cerebral ischemia in mice. Br. J. Pharmacol. 2006, 149, 23–30. [Google Scholar] [CrossRef]
- Sun, M.; Zhao, Y.-M.; Gu, Y.; Xu, C. Therapeutic window of taurine against experimental stroke in rats. Transl. Res. 2012, 160, 223–229. [Google Scholar] [CrossRef]
- Walz, W.; Klimaszewski, A.; Paterson, I.A. Glial swelling in ischemia: A hypothesis. Dev. Neurosci. 1993, 15, 216–225. [Google Scholar] [CrossRef]
- Haugstad, T.S.; Langmoen, I.A. Release of brain amino acids during hyposmolar stress and energy deprivation. J. Neurosurg. Anesthesiol. 1996, 8, 159–168. [Google Scholar] [CrossRef]
- Saransaari, P.; Oja, S.S. Taurine release is enhanced in cell-damaging conditions in cultured cerebral cortical astrocytes. Neurochem. Res. 1999, 24, 1523–1529. [Google Scholar] [CrossRef]
- Estevez, A.Y.; O’Regan, M.H.; Song, D.; Phillis, J.W. Hyposmotically induced amino acid release from the rat cerebral cortex: Role of phospholipases and protein kinases. Brain Res. 1999, 844, 1–9. [Google Scholar] [CrossRef]
- Inoue, H.; Mori, S.-I.; Morishima, S.; Okada, Y. Volume-sensitive chloride channels in mouse cortical neurons: Characterization and role in volume regulation. Eur. J. Neurosci. 2005, 21, 1648–1658. [Google Scholar] [CrossRef]
- Molchanova, S.M.; Oja, S.S.; Saransaari, P. Properties of basal taurine release in the rat striatum in vivo. Adv. Exp. Med. Biol. 2006, 583, 365–375. [Google Scholar] [CrossRef]
- Inoue, H.; Okada, Y. Roles of volume-sensitive chloride channel in excitotoxic neuronal injury. J. Neurosci. 2007, 27, 1445–1455. [Google Scholar] [CrossRef]
- Shuaib, A. The role of taurine in cerebral ischemia: Studies in transient forebrain ischemia and embolic focal ischemia in rodents. Adv. Exp. Med. Biol. 2003, 526, 421–431. [Google Scholar] [CrossRef]
- Kudo, Y.; Akiyoshi, E.; Akagi, H. Identification of two taurine receptor subtypes on the primary afferent terminal of frog spinal cord. Br. J. Pharmacol. 1988, 94, 1051–1056. [Google Scholar] [CrossRef]
- Sanberg, P.R.; Willow, M. Dose-dependent effects of taurine on convulsions induced by hypoxia in the rat. Neurosci Lett. 1980, 16, 297–300. [Google Scholar] [CrossRef]
- Stummer, W.; Betz, A.L.; Shakui, P.; Keep, R.F. Blood-brain barrier taurine transport during osmotic stress and in focal cerebral ischemia. J. Cereb. Blood Flow. Metab. 1995, 15, 852–859. [Google Scholar] [CrossRef]
- Azuma, J.; Sawamura, A.; Awata, N.; Ohta, H.; Hamaguchi, T.; Haradam, H.; Takiharma, K.; Hasegawa, H.; Yamagami, T.; Ishiyama, T.; et al. Therapeutic Effect of Taurine in Congestive Heart Failure: A Double-Blind Crossover Trial. Clin. Cardiol. 1985, 8, 276–282. [Google Scholar] [CrossRef]
- Azuma, J.; Sawamura, A.; Awata, N. Usefulness of taurine in chronic congestive heart failure and its prospective application. Jpn. Circ. J. 1992, 56, 95–99. [Google Scholar] [CrossRef]
- Yamori, Y.; Nara, Y.; Mizushima, S.; Sawamura, M.; Horie, R. Nutritional factors for stroke and major cardiovascular diseases: International epidemiological comparison of dietary prevention. Health Rep. 1994, 6, 22–27. [Google Scholar]
- Jeejeebhoy, F.; Keith, M.; Freeman, M.; Barr, A.; McCall, M.; Kudan, R.; Mazer, D.; Errett, L. Nutritional supplementation with MyoVive repletes essential cardiac myocyte nutrients and reduces left ventricular dysfunction. Am. Heart J. 2002, 143, 10092–10100. [Google Scholar]
- Kingston, R.; Kelly, C.J.; Murray, P. The therapeutic role of taurine in ischaemia-reperfusion injury. Curr. Pharm. Des. 2004, 10, 2401–2410. [Google Scholar] [CrossRef]
- Fisher, M.; Feuerstein, G.; Howells, D.W.; Hurn, P.D.; Kent, T.A.; Savitz, S.I.; Lo, E.H.; STAIR Group. Update of the stroke therapy academic industry roundtable preclinical recommendations. Stroke 2009, 40, 2244–2250. [Google Scholar] [CrossRef]
- Kanthan, R.; Shuaib, A.; Griebel, R.; Miyashita, H. Intracerebral human microdialysis. In vivo study of an acute focal ischemic model of the human brain. Stroke 1995, 26, 870–873. [Google Scholar] [CrossRef]
- Zhang, M.; Bi, L.F.; Fang, J.H.; Su, X.L.; Da, G.L.; Kuwamori, T.; Kagamimori, S. Beneficial effects of taurine on serum lipids in overweight or obese non-diabetic subjects. Amino Acids 2004, 26, 267–271. [Google Scholar]
- Takahashi, H.; Mori, T.; Fujihira, E.; Nakazawa, M. Long-term feeding of taurine in rats. Pharmacometrics 1972, 6, 529–534. [Google Scholar]
- Takahashi, H.; Kaneda, S.; Fukuda, K.; Fujihira, E.; Nakazawa, M. Studies on the teratology and three generation reproduction of taurine in mice. Pharmacometrics 1972, 6, 535–540. [Google Scholar]
- Sturman, J.A.; Messing, J.M. High dietary taurine effects on feline tissue taurine concentrations and reproductive performance. J. Nutr. 1992, 122, 82–88. [Google Scholar]
- Furukawa, S.; Katto, M.; Kouyama, H.; Nishida, I.; Kikumori, M.; Taniguchi, Y.; Toda, T.; Araki, H. Repeated dose toxicity study of intravenous treatment with taurine for 13 weeks and recovery test for 5 weeks in rat. Jpn. Pharmacol. Ther. 1991, 19, 275–306. [Google Scholar]
- Cantafora, A.; Mantovani, A.; Masella, R.; Mechelli, L.; Alvaro, D. Effect of taurine administration on liver lipids in guinea pig. Experientia 1986, 42, 407–408. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA). EFSA adopts opinion on two ingredients commonly used in some energy drinks. 2009. Available online: http://www.efsa.europa.eu/en/press/news/ans090212.htm (accessed on 15 February 2013).
- Schaffer, S.; Takahashi, K.; Azuma, J. Role of osmoregulation in the actions of taurine. Amino Acids 2000, 19, 527–546. [Google Scholar] [CrossRef]
- Morales, I.; Dopico, J.G.; Sabate, M.; Gonzalez-Hernandez, T.; Rodriguez, M. Substantia nigra osmoregulation: Taurine and ATP involvement. Am. J. Physiol. Cell Physiol. 2007, 292, 1934–1941. [Google Scholar]
- O’Byrne, M.B.; Tipton, K.F. Taurine-induced attenuation of MPP+ neurotoxicity in vitro: A possible role for the GABA(A) subclass of GABA receptors. J. Neurochem. 2000, 74, 2087–2093. [Google Scholar]
- Paula-Lima, A.C.; de Felice, F.G.; Brito-Moreira, J.; Ferreira, S.T. Activation of GABA(A) receptors by taurine and muscimol blocks the neurotoxicity of beta-amyloid in rat hippocampal and cortical neurons. Neuropharmacology 2005, 49, 1140–1148. [Google Scholar] [CrossRef]
- Chen, P.C.; Pan, C.; Gharibani, P.M.; Prentice, H.; Wu, J.Y. Taurine exerts robust protection against hypoxia and oxygen/glucose deprivation in human neuroblastoma cell culture. Adv. Exp. Med. Biol. 2013, 775, 167–175. [Google Scholar] [CrossRef]
- Das, J.; Ghosh, J.; Manna, P.; Sil, P.C. Taurine suppresses doxorubicin-triggered oxidative stress and cardiac apoptosis in rat via up-regulation of PI3-K/Akt and inhibition of p53, p38-JNK. Biochem. Pharmacol. 2011, 81, 891–909. [Google Scholar] [CrossRef]
- Smith, K.E.; Borden, L.A.; Wang, C.H.; Hartig, P.R.; Branchek, T.A.; Weinshank, R.L. Cloning and expression of a high affinity taurine transporter from rat brain. Mol. Pharmacol. 1992, 42, 563–569. [Google Scholar]
- Pow, D.V.; Sullivan, R.; Reye, P.; Hermanussen, S. Localization of taurine transporters, taurine, and (3)H taurine accumulation in the rat retina, pituitary, and brain. Glia 2002, 37, 153–168. [Google Scholar] [CrossRef]
- Wu, J.Y.; Liao, C.C.; Lin, C.J.; Lee, Y.H.; Ho, J.Y.; Tsai, W.H. Taurine receptor in the mammalian brain. Prog. Clin. Biol. Res. 1990, 351, 147–156. [Google Scholar]
- López-Colomé, A.M.; Fragoso, G.; Salceda, R. Taurine receptors in membranes from retinal pigment epithelium cells in culture. Neuroscience 1991, 41, 791–796. [Google Scholar] [CrossRef]
- Sung, D.-Y.; Walthall, W.W.; Derby, C.D. Identification and partial characterization of putative taurine receptor proteins from the olfactory organ of the spiny lobster. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 1996, 115, 19–26. [Google Scholar] [CrossRef]
- Anderson, P.A.; Trapido-Rosenthal, H.G. Physiological and chemical analysis of neurotransmitter candidates at a fast excitatory synapse in the jellyfish Cyanea capillata (Cnidaria, Scyphozoa). Invert. Neurosci. 2009, 9, 167–173. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Menzie, J.; Prentice, H.; Wu, J.-Y. Neuroprotective Mechanisms of Taurine against Ischemic Stroke. Brain Sci. 2013, 3, 877-907. https://doi.org/10.3390/brainsci3020877
Menzie J, Prentice H, Wu J-Y. Neuroprotective Mechanisms of Taurine against Ischemic Stroke. Brain Sciences. 2013; 3(2):877-907. https://doi.org/10.3390/brainsci3020877
Chicago/Turabian StyleMenzie, Janet, Howard Prentice, and Jang-Yen Wu. 2013. "Neuroprotective Mechanisms of Taurine against Ischemic Stroke" Brain Sciences 3, no. 2: 877-907. https://doi.org/10.3390/brainsci3020877
APA StyleMenzie, J., Prentice, H., & Wu, J.-Y. (2013). Neuroprotective Mechanisms of Taurine against Ischemic Stroke. Brain Sciences, 3(2), 877-907. https://doi.org/10.3390/brainsci3020877



