The Effectiveness of an Exercise Program Based on Motor Learning Principles for the Correction of the Forward Head Posture: A Randomized Controlled Trial
Abstract
1. Introduction
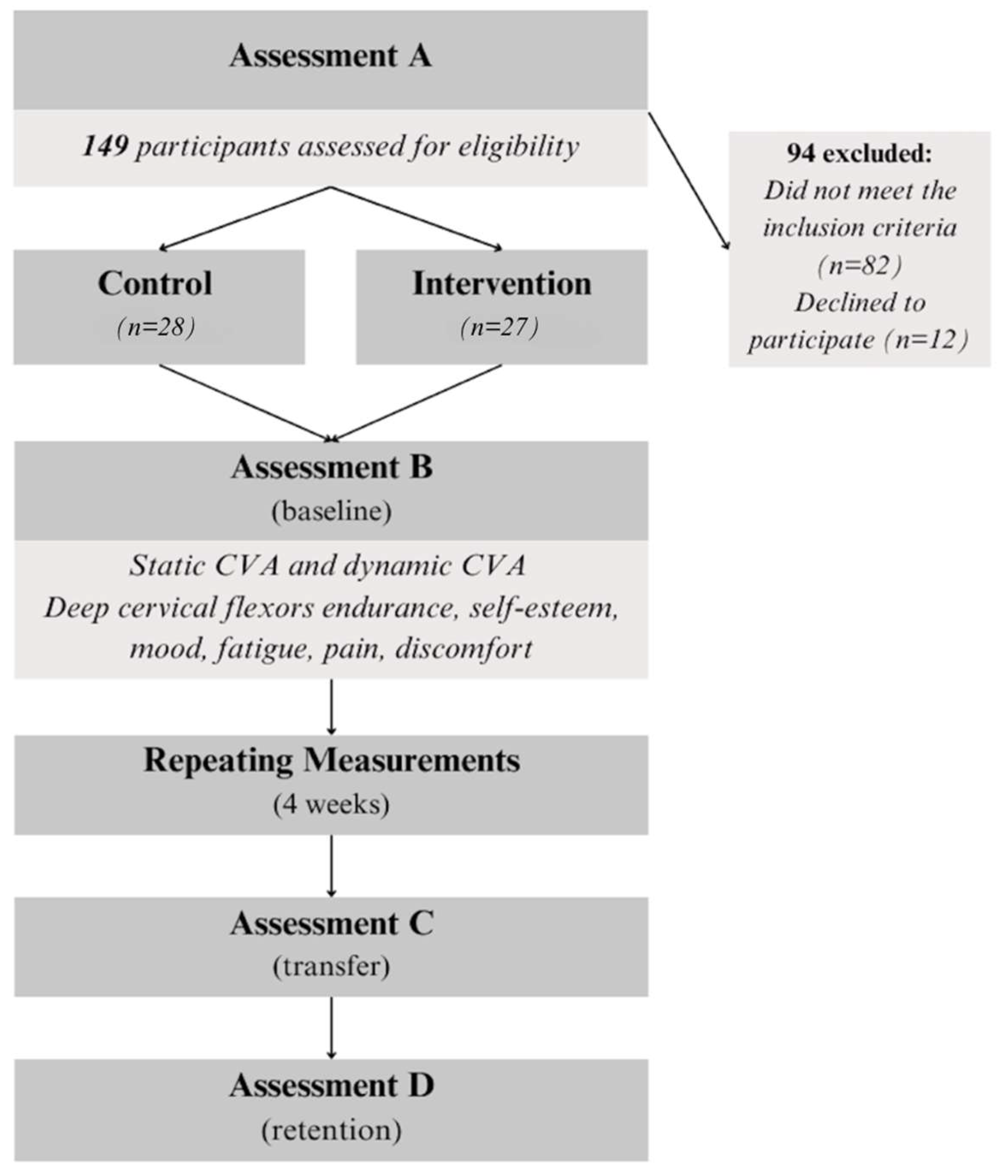
2. Materials and Methods
2.1. Study Design
2.2. Participants
2.3. Interventions
2.4. Outcome Measures
2.4.1. Static Forward Head Posture
2.4.2. Dynamic Forward Head Posture
2.4.3. Endurance of Deep Neck Flexors
2.4.4. Self-Reported Questionnaires
2.5. Procedure
2.6. Data Analysis
3. Results
3.1. Static and Dynamic FHP
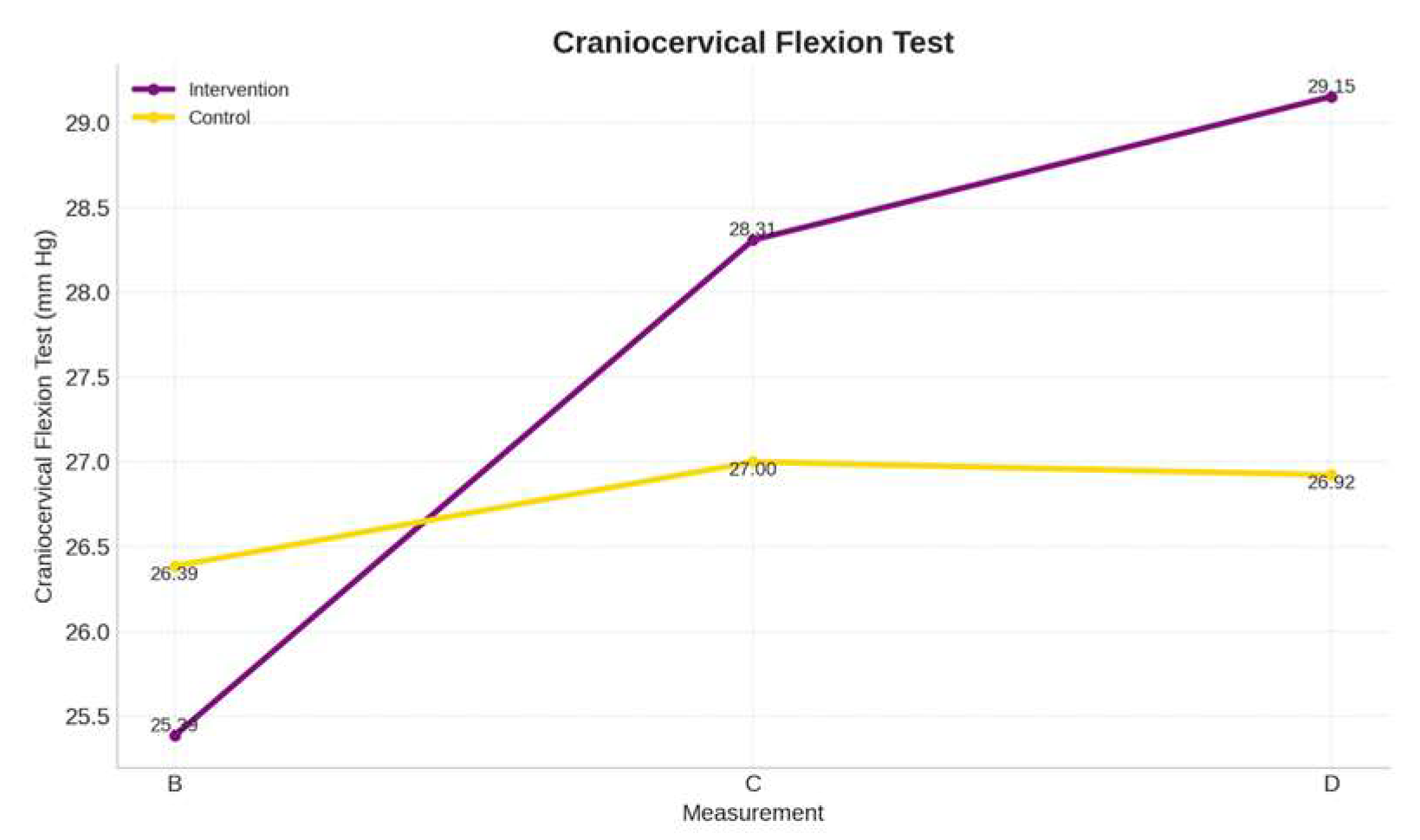
3.2. Deep Neck Flexors Endurance
3.3. Self-Reported Outcomes
4. Discussion
4.1. Limitations
4.2. Clinical Applicability
4.3. Recommendations for Future Research
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CFT | Craniocervical Flexion Test |
| CG | Control Group |
| CI | Confidence Intervals |
| CMDQ | Cornell Musculoskeletal Discomfort Questionnaire |
| CVA | Craniovertebral Angle |
| FHP | Forward Head Posture |
| IG | Intervention Group |
| MD | Mean Difference |
| MCTE | Motor Control Therapeutic Exercise |
| RSES | Rosenberg Sorensen Self-Esteem Scale |
| SD | Standard Deviation |
| SDD% | Smallest Detectable Difference (Percentage) |
| SEM | Standard Error of Measurement |
| VR | Virtual Reality |
Appendix A
References
- Elsayed, W.H.; Alowa, Z.A. Impact of Forward Head Posture Correction on Craniovertebral Angle, Neck Disability, and Spinal Electromyography: A Randomized Controlled Trial. J. Back. Musculoskelet. Rehabil. 2025, 38, 83–92. [Google Scholar] [CrossRef]
- de Miranda, A.P.O.C.; Penha, P.J.; Pereira, L.G.; Pessoa, W.C.; João, S.M.A. Posture of Adolescent and the Relationship with Body Mass Index, Sex, and Physical Activity. Child. Obes. 2022, 18, 342–349. [Google Scholar] [CrossRef]
- Sheikhhoseini, R.; Shahrbanian, S.; Sayyadi, P.; O’Sullivan, K. Effectiveness of Therapeutic Exercise on Forward Head Posture: A Systematic Review and Meta-Analysis. J. Manip. Physiol. Ther. 2018, 41, 530–539. [Google Scholar] [CrossRef]
- Korakakis, V.; O’Sullivan, K.; O’Sullivan, P.B.; Evagelinou, V.; Sotiralis, Y.; Sideris, A.; Sakellariou, K.; Karanasios, S.; Giakas, G. Physiotherapist Perceptions of Optimal Sitting and Standing Posture. Musculoskelet. Sci. Pract. 2019, 39, 24–31. [Google Scholar] [CrossRef]
- Pillastrini, P.; de Lima E Sá Resende, F.; Banchelli, F.; Burioli, A.; Di Ciaccio, E.; Guccione, A.A.; Villafañe, J.H.; Vanti, C. Effectiveness of Global Postural Re-Education in Patients with Chronic Nonspecific Neck Pain: Randomized Controlled Trial. Phys. Ther. 2016, 96, 1408–1416. [Google Scholar] [CrossRef]
- Lomas-Vega, R.; Garrido-Jaut, M.V.; Rus, A.; Del-Pino-Casado, R. Effectiveness of Global Postural Re-Education for Treatment of Spinal Disorders: A Meta-Analysis. Am. J. Phys. Med. Rehabil. 2017, 96, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Abadiyan, F.; Hadadnezhad, M.; Khosrokiani, Z.; Letafatkar, A.; Akhshik, H. Adding a Smartphone App to Global Postural Re-Education to Improve Neck Pain, Posture, Quality of Life, and Endurance in People with Nonspecific Neck Pain: A Randomized Controlled Trial. Trials 2021, 22, 274. [Google Scholar] [CrossRef] [PubMed]
- Szczygieł, E.; Fudacz, N.; Golec, J.; Golec, E. The Impact of the Position of the Head on the Functioning of the Human Body: A Systematic Review. Int. J. Occup. Med. Environ. Health 2020, 33, 559–568. [Google Scholar] [CrossRef] [PubMed]
- Lin, G.; Zhao, X.; Wang, W.; Wilkinson, T. The Relationship between Forward Head Posture, Postural Control and Gait: A Systematic Review. Gait Posture 2022, 98, 316–329. [Google Scholar] [CrossRef]
- Youssef, H.; Aydoǧdu, O.; Yildiz, A. Altered Body Balance and Plantar Pressure Distribution in Young Adults with Forward Head Posture. J. Orthop. 2025, 59, 68–75. [Google Scholar] [CrossRef]
- Mahmoud, N.F.; Hassan, K.A.; Abdelmajeed, S.F.; Moustafa, I.M.; Silva, A.G. The Relationship Between Forward Head Posture and Neck Pain: A Systematic Review and Meta-Analysis. Curr. Rev. Musculoskelet. Med. 2019, 12, 562–577. [Google Scholar] [CrossRef]
- Yang, S.; Boudier-Revéret, M.; Yi, Y.G.; Hong, K.Y.; Chang, M.C. Treatment of Chronic Neck Pain in Patients with Forward Head Posture: A Systematic Narrative Review. Healthcare 2023, 11, 2604. [Google Scholar] [CrossRef] [PubMed]
- Koseki, T.; Kakizaki, F.; Hayashi, S.; Nishida, N.; Itoh, M. Effect of Forward Head Posture on Thoracic Shape and Respiratory Function. J. Phys. Ther. Sci. 2019, 31, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Deniz, Y.; Ertekın, D.; Cokar, D. The Effect of Forward Head Posture on Dynamic Lung Volumes in Young Adults: A Systematic Review. Bull. Fac. Phys. Ther. 2024, 29, 15. [Google Scholar] [CrossRef]
- Dimitriadis, Z.; Kapreli, E.; Strimpakos, N.; Oldham, J. Pulmonary Function of Patients with Chronic Neck Pain: A Spirometry Study. Respir. Care 2014, 59, 543–549. [Google Scholar] [CrossRef] [PubMed]
- Titcomb, D.A.; Melton, B.F.; Miyashita, T.; Bland, H.W. Evidence-Based Corrective Exercise Intervention for Forward Head Posture in Adolescents and Young Adults Without Musculoskeletal Pathology: A Critically Appraised Topic. J. Sport Rehabil. 2022, 31, 640–644. [Google Scholar] [CrossRef]
- Kawasaki, T.; Ohji, S.; Aizawa, J.; Sakai, T.; Hirohata, K.; Koseki, T.; Kuruma, H.; Okawa, A.; Jinno, T. Characteristics of Head and Neck Alignment and Function of Deep Cervical Flexor Muscles in Patients with Nonspecific Neck Pain. J. Bodyw. Mov. Ther. 2024, 39, 565–571. [Google Scholar] [CrossRef]
- Kim, D.-H.; Kim, C.-J.; Son, S.-M. Neck Pain in Adults with Forward Head Posture: Effects of Craniovertebral Angle and Cervical Range of Motion. Osong Public Health Res. Perspect. 2018, 9, 309–313. [Google Scholar] [CrossRef]
- Kang, D.Y. Deep Cervical Flexor Training with a Pressure Biofeedback Unit Is an Effective Method for Maintaining Neck Mobility and Muscular Endurance in College Students with Forward Head Posture. J. Phys. Ther. Sci. 2015, 27, 3207–3210. [Google Scholar] [CrossRef]
- Fredin, K.; Lorås, H. Manual Therapy, Exercise Therapy or Combined Treatment in the Management of Adult Neck Pain—A Systematic Review and Meta-Analysis. Musculoskelet. Sci. Pract. 2017, 31, 62–71. [Google Scholar] [CrossRef]
- Nazwar, T.A.; Bal’afif, F.; Wardhana, D.W.; Mustofa, M. Impact of Physical Exercise (Strength and Stretching) on Repairing Craniovertebral and Reducing Neck Pain: A Systematic Review and Meta-Analysis. J. Craniovertebr. Junction Spine 2024, 15, 266–279. [Google Scholar] [CrossRef]
- Leech, K.A.; Roemmich, R.T.; Gordon, J.; Reisman, D.S.; Cherry-Allen, K.M. Updates in Motor Learning: Implications for Physical Therapist Practice and Education. Phys. Ther. 2021, 102, pzab250. [Google Scholar] [CrossRef]
- Chua, L.-K.; Dimapilis, M.K.; Iwatsuki, T.; Abdollahipour, R.; Lewthwaite, R.; Wulf, G. Practice Variability Promotes an External Focus of Attention and Enhances Motor Skill Learning. Hum. Mov. Sci. 2019, 64, 307–319. [Google Scholar] [CrossRef]
- Hunt, C.; Paez, A.; Folmar, E. The Impact of Attentional Focus on The Treatment of Musculoskeletal and Movement Disorders. Int. J. Sports Phys. Ther. 2017, 12, 901–907. [Google Scholar] [CrossRef]
- Tsiringakis, G.; Dimitriadis, Z.; Triantafylloy, E.; McLean, S. Motor Control Training of Deep Neck Flexors with Pressure Biofeedback Improves Pain and Disability in Patients with Neck Pain: A Systematic Review and Meta-Analysis. Musculoskelet. Sci. Pract. 2020, 50, 102220. [Google Scholar] [CrossRef] [PubMed]
- Martin-Gomez, C.; Sestelo-Diaz, R.; Carrillo-Sanjuan, V.; Navarro-Santana, M.J.; Bardon-Romero, J.; Plaza-Manzano, G. Motor Control Using Cranio-Cervical Flexion Exercises versus Other Treatments for Non-Specific Chronic Neck Pain: A Systematic Review and Meta-Analysis. Musculoskelet. Sci. Pract. 2019, 42, 52–59. [Google Scholar] [CrossRef]
- Khosrokiani, Z.; Letafatkar, A.; Gladin, A. Lumbar Motor Control Training as a Complementary Treatment for Chronic Neck Pain: A Randomized Controlled Trial. Clin. Rehabil. 2022, 36, 99–112. [Google Scholar] [CrossRef]
- Kaczmarek, C.; Schmidt, A.; Emperle, A.S.; Schaefer, S. The Influence of Social Contexts on Motor and Cognitive Performance: Performing Alone, in Front of Others, or Coacting with Others. J. Sport Exerc. Psychol. 2022, 44, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Hall, K.J.; Van Ooteghem, K.; McIlroy, W.E. Emotional State as a Modulator of Autonomic and Somatic Nervous System Activity in Postural Control: A Review. Front. Neurol. 2023, 14, 1188799. [Google Scholar] [CrossRef] [PubMed]
- Moustafa, I.M.; Youssef, A.; Ahbouch, A.; Tamim, M.; Harrison, D.E. Is Forward Head Posture Relevant to Autonomic Nervous System Function and Cervical Sensorimotor Control? Cross Sectional Study. Gait Posture 2020, 77, 29–35. [Google Scholar] [CrossRef]
- Magill, R.A. Motor Learning and Control: Concepts and Applications. McGraw-Hill Education: New York, NY, USA, 2021; ISBN 978-1-264-68193-8. [Google Scholar]
- Chaudhuri, S.; Chawla, J.K.; Phadke, V. Physiotherapeutic Interventions for Upper Cross Syndrome: A Systematic Review and Meta-Analysis. Cureus 2023, 15, e45471. [Google Scholar] [CrossRef]
- Batool, A.; Soomro, R.R.; Baig, A.A.M. Comparing the Effects of Neck Stabilization Exercises versus Dynamic Exercises among Patients Having Nonspecific Neck Pain with Forward Head Posture: A Randomized Clinical Trial. BMC Musculoskelet. Disord. 2024, 25, 707. [Google Scholar] [CrossRef]
- Oliveira, A.C.; Silva, A.G. Neck Muscle Endurance and Head Posture: A Comparison between Adolescents with and without Neck Pain. Man. Ther. 2016, 22, 62–67. [Google Scholar] [CrossRef]
- Iqbal, Z.A.; Alghadir, A.H.; Anwer, S. Efficacy of Deep Cervical Flexor Muscle Training on Neck Pain, Functional Disability, and Muscle Endurance in School Teachers: A Clinical Trial. Biomed. Res. Int. 2021, 2021, 7190808. [Google Scholar] [CrossRef] [PubMed]
- Levin, M.F.; Demers, M. Motor Learning in Neurological Rehabilitation. Disabil. Rehabil. 2021, 43, 3445–3453. [Google Scholar] [CrossRef] [PubMed]
- Yadav, G.; Duque, J. Reflecting on What Is “Skill” in Human Motor Skill Learning. Front. Hum. Neurosci. 2023, 17, 889. [Google Scholar] [CrossRef]
- Sepehri, S.; Sheikhhoseini, R.; Piri, H.; Sayyadi, P. The Effect of Various Therapeutic Exercises on Forward Head Posture, Rounded Shoulder, and Hyperkyphosis among People with Upper Crossed Syndrome: A Systematic Review and Meta-Analysis. BMC Musculoskelet. Disord. 2024, 25, 105. [Google Scholar] [CrossRef]
- Titcomb, D.A.; Melton, B.F.; Miyashita, T.; Bland, H.W. The Effects of Postural Education or Corrective Exercise on the Craniovertebral Angle in Young Adults with Forward Head Posture: A Randomized Controlled Trial. Int. J. Exerc. Sci. 2023, 16, 954–973. [Google Scholar] [CrossRef] [PubMed]
- Elgendy, M.H.; Ghaffar, M.A.A.; Sabbahi, S.A.E.; Abutaleb, E.E.S.; Elsayed, S.E.B. Efficacy of Head Postural Correction Program on Craniovertebral Angle, Scapular Position, and Dominant Hand Grip Strength in Forward Head Posture Subjects: A Randomized Controlled Trial. Physiother. Res. Int. 2024, 29, e2093. [Google Scholar] [CrossRef]
- Abu-Taleb, W.; Yamany, A.A.; Aneis, Y.M.; Kasem, S.T.A.E. Effect of Adding Global Postural Reeducation to Kendall Exercises for Treating Asymptomatic Forward Head Posture: A Single-Blinded Randomized Controlled Trial. J. Bodyw. Mov. Ther. 2025, 42, 938–947. [Google Scholar] [CrossRef]
- Hopewell, S.; Chan, A.-W.; Collins, G.S.; Hróbjartsson, A.; Moher, D.; Schulz, K.F.; Tunn, R.; Aggarwal, R.; Berkwits, M.; Berlin, J.A.; et al. CONSORT 2025 Statement: Updated Guideline for Reporting Randomised Trials. BMJ 2025, 389, e081123. [Google Scholar] [CrossRef]
- Diab, A.A.; Moustafa, I.M. The Efficacy of Forward Head Correction on Nerve Root Function and Pain in Cervical Spondylotic Radiculopathy: A Randomized Trial. Clin. Rehabil. 2012, 26, 351–361. [Google Scholar] [CrossRef]
- Dimitriadis, Z.; Kapreli, E.; Strimpakos, N.; Oldham, J. Clinical Assessment of the Deep Neck Flexors: Which Test Is Best? Int. J. Ther. Rehabil. 2015, 22, 458–459. [Google Scholar] [CrossRef]
- Gentile, A.M. A Working Model of Skill Acquisition with Application to Teaching. Quest 1972, 17, 3–23. [Google Scholar] [CrossRef]
- Wulf, G.; Lewthwaite, R. Optimizing Performance through Intrinsic Motivation and Attention for Learning: The OPTIMAL Theory of Motor Learning. Psychon. Bull. Rev. 2016, 23, 1382–1414. [Google Scholar] [CrossRef] [PubMed]
- Rafique, D.; Heggli, U.; Bron, D.; Colameo, D.; Schweinhardt, P.; Swanenburg, J. Effects of Increasing Axial Load on Cervical Motor Control. Sci. Rep. 2021, 11, 18627. [Google Scholar] [CrossRef]
- Thoomes, E.; Cleland, J.A.; Falla, D.; de Pauw, R.; Maissan, F.; de Graaf, M. Measurement Properties of a Computer Adaptive Device, the Senscoordination 3D Cervical Trainer, to Assess Cervical Range of Motion in People with Neck Pain. Musculoskelet. Sci. Pract. 2023, 67, 102861. [Google Scholar] [CrossRef]
- An, J.; Wulf, G. Golf Skill Learning: An External Focus of Attention Enhances Performance and Motivation. Psychol. Sport Exerc. 2024, 70, 102563. [Google Scholar] [CrossRef] [PubMed]
- Christiansen, L.; Larsen, M.N.; Madsen, M.J.; Grey, M.J.; Nielsen, J.B.; Lundbye-Jensen, J. Long-Term Motor Skill Training with Individually Adjusted Progressive Difficulty Enhances Learning and Promotes Corticospinal Plasticity. Sci. Rep. 2020, 10, 15588. [Google Scholar] [CrossRef]
- Kakizawa, S. Assessment of Retention and Attenuation of Motor-Learning Memory by Repeated Rotor-Rod Analyses. Sci. Rep. 2024, 14, 31003. [Google Scholar] [CrossRef]
- Dimitriadis, Z.; Argyrou, S.; Diamantis, A.; Kostakis, K.; Kanellopoulos, A.; Strimpakos, N.; Poulis, I.; Kapreli, E. Reliability of Craniovertebral Angle Measurement During Gait Using Kinovea and AutoCAD Software. Arch. Hell. Med. 2023, 40, 772–780. [Google Scholar]
- Broisler, C.N.; Gaban, G.L.N.A.; Vivaldini, M.R.S.; Nunes, G.S.; Selistre, L.F.A. Intra- and Inter-Rater Reliability, Standard Error of Measurement, and Minimal Detectable Change of the Cranio-Cervical Flexion Test in Individuals with Non-Specific Chronic Neck Pain. Musculoskelet. Sci. Pract. 2025, 76, 103258. [Google Scholar] [CrossRef]
- Selistre, L.F.A.; Melo, C.d.S.; Noronha, M.A. de Reliability and Validity of Clinical Tests for Measuring Strength or Endurance of Cervical Muscles: A Systematic Review and Meta-Analysis. Arch. Phys. Med. Rehabil. 2021, 102, 1210–1227. [Google Scholar] [CrossRef]
- Lee, H.; Lee, Y. Effects of Postural Changes Using a Standing Desk on the Craniovertebral Angle, Muscle Fatigue, Work Performance, and Discomfort in Individuals with a Forward Head Posture. Healthcare 2024, 12, 2436. [Google Scholar] [CrossRef]
- Erdinc, O.; Hot, K.; Ozkaya, M. Turkish Version of the Cornell Musculoskeletal Discomfort Questionnaire: Cross-Cultural Adaptation and Validation. Work 2011, 39, 251–260. [Google Scholar] [CrossRef] [PubMed]
- Galanou, C.; Galanakis, M.; Alexopoulos, E.; Darviri, C. Rosenberg Self-Esteem Scale Greek Validation on Student Sample. Psychology 2014, 5, 819–827. [Google Scholar] [CrossRef]
- Christakou, A.; Zervas, Y.; Psychountaki, M.; Stavrou, N.A. Development and Validation of the Attention Questionnaire of Rehabilitated Athletes Returning to Competition. Psychol. Health Med. 2012, 17, 499–510. [Google Scholar] [CrossRef] [PubMed]
- Ruivo, R.M.; Carita, A.I.; Pezarat-Correia, P. The Effects of Training and Detraining after an 8 Month Resistance and Stretching Training Program on Forward Head and Protracted Shoulder Postures in Adolescents: Randomised Controlled Study. Man. Ther. 2016, 21, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Abd El-Azeim, A.S.; Mahmoud, A.G.; Mohamed, M.T.; El-Khateeb, Y.S. Impact of Adding Scapular Stabilization to Postural Correctional Exercises on Symptomatic Forward Head Posture: A Randomized Controlled Trial. Eur. J. Phys. Rehabil. Med. 2022, 58, 757–766. [Google Scholar] [CrossRef]
- Mylonas, K.; Angelopoulos, P.; Billis, E.; Tsepis, E.; Fousekis, K. Combining Targeted Instrument-Assisted Soft Tissue Mobilization Applications and Neuromuscular Exercises Can Correct Forward Head Posture and Improve the Functionality of Patients with Mechanical Neck Pain: A Randomized Control Study. BMC Musculoskelet. Disord. 2021, 22, 212. [Google Scholar] [CrossRef]
- Heydari, Z.; Sheikhhoseini, R.; Shahrbanian, S.; Piri, H. Establishing Minimal Clinically Important Difference for Effectiveness of Corrective Exercises on Craniovertebral and Shoulder Angles among Students with Forward Head Posture: A Clinical Trial Study. BMC Pediatr. 2022, 22, 230. [Google Scholar] [CrossRef] [PubMed]
- Mylonas, K.; Tsekoura, M.; Billis, E.; Aggelopoulos, P.; Tsepis, E.; Fousekis, K. Reliability and Validity of Non-Radiographic Methods of Forward Head Posture Measurement: A Systematic Review. Cureus 2022, 14, e27696. [Google Scholar] [CrossRef] [PubMed]
- Guadagnoli, M.A.; Lee, T.D. Challenge Point: A Framework for Conceptualizing the Effects of Various Practice Conditions in Motor Learning. J. Mot. Behav. 2004, 36, 212–224. [Google Scholar] [CrossRef] [PubMed]





| Action Function (Body Stability) | ||
|---|---|---|
| Environmental Context | No Object Manipulation | Object Manipulation |
| Stationary Regulatory Conditions and No Intertrial Variability | 1A Sitting position with laser, viewing a video/reading/playing 3D games. Progressively ↓ 1a Standing position, lateral and up-down steps /playing 3D games. | 1Β Sitting position with laser: writing a text with PC. Standing position: playing with a toy or a ball/holding weights and playing 3D games. Progressively ↓ 1b Standing position: holding weights (different kilograms) and lateral and up-down steps. |
| Stationary Regulatory Conditions and Intertrial Variability | 2A Standing on different surfaces (mattress, flywheel, wood, carpet, etc.). Sitting in different chairs (hard, anatomic, high, low, stool). Progressively ↓ 2a Steps in place, sideways and backwards on different surfaces, with shoes and without. Sitting on different chairs and medicine ball (unstable) with laser viewing a video/reading/playing 3D games. | 2Β Standing, pitching, or sitting on different unstable surfaces (platforms, BOSU ball, medicine ball) and holding a weight/throwing or beating a basketball/THERABAND exercise. Progressively ↓ 2b Sitting on different unstable surfaces, balance disturbance and ball bounce, or shooting basketball free throws. |
| In-Motion Regulatory Conditions and No Intertrial Variability | 3A Steady steps in a seated position with a metronome (fixed speed and weight). Progressively ↓ 3a Sitting on a medicine ball, field steps with a metronome, or standing-position steps in place with a metronome, increasing the height of the step. | 3Β Holding a ball or weight and sitting on a medicine ball, field steps with a metronome, or standing position steps in place with a metronome and increasing the height of the step. Progressively ↓ 3b Sitting on a medicine ball/Step and shooting a basketball with constant speed and distance/ flying darts at a target. |
| In-Motion Regulatory Conditions and Intertrial Variability | 4A Steady steps in a seated position with a metronome (different speeds and weights on the legs). Progressively ↓ 4a Sitting on a medicine ball and balance disturbance, shooting a basketball on command and from different distances. | 4Β Standing on a platform (BOSU ball), balance disturbance, shooting a basketball on command. Progressively ↓ 4b Sitting on a medicine ball, balance disturbance, handling a football with the legs or basketball with the hands, and the same exercises on a standing position on a platform. |
| Control Group (n=26) | Experimental Group (n = 26) | p-Value | |||
|---|---|---|---|---|---|
| Mean | SD | Mean | SD | ||
| Age (years) | 21 | 1.72 | 20.96 | 1.68 | 0.935 |
| High (cm) | 167 | 0.08 | 167 | 0.07 | 0.944 |
| Weight (Kg) | 70.6 | 11.47 | 67.8 | 12.31 | 0.397 |
| CVA static (°) | 46.99 | 2.38 | 45.92 | 3.11 | 0.168 |
| CVA dynamic (°) | 45.89 | 2.69 | 44.84 | 3.94 | 0.269 |
| CFT (mmHg) | 26.38 | 3.21 | 25.38 | 2.45 | 0.212 |
| Current Pain (cm) | 0.72 | 1.16 | 0.47 | 0.99 | 0.402 |
| Usual pain (cm) | 1.7 | 2.40 | 1.03 | 1.90 | 0.220 |
| Discomfort Head (cm) | 1.39 | 2.34 | 0.71 | 1.58 | 0.226 |
| Discomfort Neck (cm) | 1.86 | 2.20 | 1.70 | 1.93 | 0.785 |
| Discomfort Shoulders and upper arms (cm) | 1.04 | 1.25 | 1.38 | 2.04 | 0.474 |
| Discomfort Middle back (cm) | 0.67 | 1.26 | 0.93 | 1.75 | 0.547 |
| Discomfort Lower back (cm) | 1.23 | 1.69 | 1.14 | 2.07 | 0.867 |
| Discomfort Forearms (cm) | 0.81 | 2.41 | 0.49 | 1.63 | 0.570 |
| Discomfort Wrists/hands (cm) | 0.92 | 2.29 | 0.26 | 2.01 | 0.508 |
| Outcome Measures | Change B–C | Change C–D | Change B–D | |||
|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | |
| CVAstatic (°)-CG | 0.3238 | 2.32 | −0.0551 | 0.43 | 0.2687 | 2.08 |
| CVAstatic (°)-IG | 7.5431 | 2.13 | 0.5595 | 1.63 | 8.1026 | 2.58 |
| p-value | <0.01 | 0.070 | <0.01 | |||
| Effect size (r) | 0.73 | 0.06 | 0.74 | |||
| CVAdynamic (°)-CG | −0.4497 | 2.37 | 0.1591 | 0.48 | −0.2906 | 2.29 |
| CVAdynamic (°)-IG | 5.1349 | 4.15 | 0.0282 | 1.86 | 5.1631 | 4.88 |
| p-value | <0.01 | 0.732 | <0.01 | |||
| Effect size (r) | 0.41 | 0.002 | 0.34 | |||
| CFT (mmHg)-CG | 0.6154 | 2.57 | −0.0769 | 0.39 | 0.5385 | 0.21 |
| CFT (mmHg)-IG | 2.9231 | 2.13 | 0.8462 | 1.89 | 3.7692 | 2.57 |
| p-value | <0.01 | 0.022 | <0.01 | |||
| Effect size (r) | 0.19 | 0.10 | 0.32 | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Argyrou, S.; Kitixis, P.; Dimitriadis, Z.; Christakou, A.; Strimpakos, N.; Paras, G.; Tsioutsoumaka, M.; Kapreli, E. The Effectiveness of an Exercise Program Based on Motor Learning Principles for the Correction of the Forward Head Posture: A Randomized Controlled Trial. Brain Sci. 2025, 15, 873. https://doi.org/10.3390/brainsci15080873
Argyrou S, Kitixis P, Dimitriadis Z, Christakou A, Strimpakos N, Paras G, Tsioutsoumaka M, Kapreli E. The Effectiveness of an Exercise Program Based on Motor Learning Principles for the Correction of the Forward Head Posture: A Randomized Controlled Trial. Brain Sciences. 2025; 15(8):873. https://doi.org/10.3390/brainsci15080873
Chicago/Turabian StyleArgyrou, Stephani, Pavlos Kitixis, Zacharias Dimitriadis, Anna Christakou, Nikolaos Strimpakos, George Paras, Maria Tsioutsoumaka, and Eleni Kapreli. 2025. "The Effectiveness of an Exercise Program Based on Motor Learning Principles for the Correction of the Forward Head Posture: A Randomized Controlled Trial" Brain Sciences 15, no. 8: 873. https://doi.org/10.3390/brainsci15080873
APA StyleArgyrou, S., Kitixis, P., Dimitriadis, Z., Christakou, A., Strimpakos, N., Paras, G., Tsioutsoumaka, M., & Kapreli, E. (2025). The Effectiveness of an Exercise Program Based on Motor Learning Principles for the Correction of the Forward Head Posture: A Randomized Controlled Trial. Brain Sciences, 15(8), 873. https://doi.org/10.3390/brainsci15080873









