Abstract
Objective: The comorbidity of alcohol and substance use disorders among persons with bipolar disorder is elevated, as indicated by epidemiological and clinical studies. Following alcohol use, cannabis is the most frequently used and abused illicit substance among bipolar individuals, and such use may lead to comorbid cannabis use disorders (CUD). Previous research indicated that CUDs were related to a more severe course of bipolar disorder and higher rates of other comorbid alcohol and substance use disorders. Few studies, however, have conducted longitudinal research on this comorbidity. The aim of this study is to investigate the influence of CUD on the course of bipolar I and II individuals during a 5-year follow-up. Methods: The characteristics of bipolar disorder, cannabis use disorders, and other alcohol and substance use disorders, as well as comorbid mental disorders, were assessed using a standardized semi-structured interview (SSAGA) at both baseline and the 5-year follow-up. N = 180 bipolar I and II patients were subdivided into groups of with and without comorbid cannabis use disorders (CUD). Results: Of the 77 bipolar I and 103 bipolar II patients, n = 65 (36.1%) had a comorbid diagnosis of any CUD (DSM-IV cannabis abuse or dependence). Comorbid bipolar patients with CUD had higher rates of other substance use disorders and posttraumatic stress disorders, more affective symptoms, and less psychosocial functioning at baseline and at 5-year follow-up. In contrast to previously reported findings, higher rates of anxiety disorders and bipolar disorder complications (e.g., mixed episodes, rapid cycling, and manic or hypomanic episodes) were not found. The effect of CUD on other substance use disorders was confirmed using moderation analyses. Conclusions: A 5-year prospective evaluation of bipolar patients with and without CUD confirmed previous investigations, suggesting that the risk of other substance use disorders is significantly increased in comorbid individuals. CUD has a moderation effect, while no effect was found for other mental disorders. Findings from this study and previous research may be due to the examination of different phenotypes (Cannabis use vs. CUD) and sample variation (family study vs. clinical and epidemiological populations).
1. Introduction
Bipolar disorders (BDs) are reported to have a lifetime prevalence of 0.8% for BD-I and 1.1% for BD-II [1]. BDs is a risk factor for both behavioral, (e.g., gambling) [2] and substance use disorders (SUDs) [3,4].
Among these substances, worldwide cannabis is the most widely used illicit drug [5]. Cannabis use (CU) in the United States is reported to be 11.8% in individuals older than 12 years and 10.8% in those individuals aged 26 years and older [5,6]. The CU rate in adults aged 26 and older increased from 7.9% to 10.8% from 2017 to 2020. Cannabis use disorders (CUDs) were diagnosed in 13.5% in 18–25-year-olds and 4.0% in those individuals aged 26+ years. By comparison, among European adults, past-month prevalence of CU increased by 27% from 2010–2019 (from 3.1 to 3.9%), with the most pronounced relative increases observed among 35–64-year-olds [7].
A recent review indicates that lifetime cannabis use (CU) was very frequent among persons with BD, ranging from 50–66% using cannabis over their lifetime [8]. This reported rate of lifetime cannabis use (LT-CU) is as much as sevenfold higher in individuals with BD compared to people without BD (71.3%, OR 6.8, CI, 5.41 to 8.52) [9], while cross-sectional prevalence rates of CU varied from 3.3% to approximately 18% [8].
Not surprisingly, CUD was also increased among individuals with BD when compared to the general population (7.2% vs. 1.2%, respectively), ranging from 7.2–30% across several studies [9,10,11]. The prevalence rates of CU, cannabis abuse (CA) and cannabis dependence (CD) were found to be higher in BD-I (CD: 23.6%, CA: 9.7%, CUD: 11.8%) versus BD-II (CD: 10.2%, CD: 4.9%, CUD: 5.7%) [12].
Interestingly, among individuals diagnosed with CUD, BD comorbidity is lower but still substantial. A meta-analysis found an approximately 10% prevalence rate of BD in individuals with CUD in both community and clinical samples [5,6]. BD-I (8.8–9.0%) is more prevalent than BD-II (0.8–1.5%) in individuals with CUD [13]. Although less common than in CUD, BD is also more common in cannabis users without CUD compared to the general population (2.5%) [14]. In a more recent review and meta-analysis in subjects with cannabis use, the bipolar disorder rate was 9.6 % as indicated by Wave 2 results of the National Epidemiologic Survey on Alcohol and Related Conditions [15].
There are certainly significant and complex Interactions between CU, use disorders (CUD) and mood disorders: CU may contribute to the risk for developing psychopathology, which may in turn lead to CU and eventually to CUD. In addition, underlying factors may contribute to both mood disorder psychopathology and CU and CUD [16,17].
Previous epidemiological studies [8] indicated that CU is also associated with the worsening of mood disorder symptoms in a dose-dependent manner [10], suggesting that CU and use disorders, like alcohol use disorders [16,17], have a detrimental influence on the development and course of bipolar disorders.
While there is a difference between CU and use disorders, CU in BD was associated with hypomanic and manic episodes, but not depression [8,18,19]. Individuals with BD and CU displayed increased severities of mania [20], hallucinations, delusions and overall illness [21]. Several studies reported higher rates of complications and more severe bipolar symptoms, including more time in manic and mixed episodes [9], more frequent affective episodes [10], and mixed states [22]. Psychotic symptoms [23] were also more prevalent in BD with CUD.
Further, other SUD also occur more often in individuals with either CUD and BD. Approximately two-thirds of individuals with bipolar disorder and CUD report nicotine dependence and alcohol and drug use disorders. A meta-analysis reported that among SUDs seen in patients with BD, alcohol (42%) and cannabis (20%) were most prevalent, followed by other illicit substances (17%) [5,6]. Other studies confirm that BD with comorbid CU was associated with an increased frequency of SUDs [21]. This is a particularly relevant finding, since co-occurring alcohol and SUDs among individuals with bipolar disorder are negatively associated with on the course of illness (even after adjusting for non-compliance) including a delayed onset of symptomatic recovery when treated [24,25]. The significance of these findings is underscored when considering the heavy burden of disease already associated with bipolar disorder [26,27].
BD patients have a higher prevalence of psychiatric comorbidities with anxiety disorder (24.1%) being most prevalent followed by personality disorders (17.5%) and PTSD (9.7%) [19]. Anxiety disorders, including panic disorders, are reported to occur in BD over the course of disease in high rates ranging from 39% to 55% [28] and PTSD [29]. However, no study has investigated this comorbidity in bipolar subjects with CUD. Comorbid anxiety disorders increase the risk for hospitalization for BD depression but not for BD mania [20]. A higher proportion of BD patients with borderline personality disorders are female and are at higher risk for suicidal behaviors, emphasizing the need for acute inpatient care [19]. Comorbid anxiety disorders increase the odds for hospitalization for BD depression and not for BD mania [28].
Few studies, however, used a prospective design (follow-up between 1 to 3 years) to investigate the course of both comorbid disorders. Results from previous research from retrospective and cross-sectional designs indicate a more severe course of comorbid bipolar disorder and CUD [30], more comorbidity [21], more psychopathological symptoms and poorer functional outcome [31]. Importantly, CU was associated with a higher rate of cannabis and other SUDs in an epidemiological prospective study in non-comorbid individuals [32].
The aim of the present study was to analyze the clinical course and prognosis of bipolar I and II patients with and without CUDs (DSM-IV Cannabis dependence and Cannabis abuse) using the Collaborative Study on the Genetics of Alcoholism (COGA) sample. Subgroups of bipolar individuals with and without comorbid CUDs were compared over a five-year follow-up time period. Subjects were assessed at baseline regarding their lifetime history of SUD, bipolar disorders and then re-interviewed prospectively after 5 years. In these analyses, both bipolar I and II subjects are included to investigate the lifetime characteristics of CUDs, other alcohol and substance dependence and bipolar disorders retrospectively. Secondly, we examined the course of CUDs, other alcohol and substance dependence, BD and comorbidity with mental and other SUD during the 5-year follow-up period. Thirdly, we analysed the moderating effect of CUD on the risk for developing other alcohol and SUDs or the number of comorbid psychiatric illnesses.
2. Methods
2.1. Sample
The Collaborative Study on the Genetics of Alcoholism (COGA) is a family pedigree investigation which enrolled treatment-seeking alcohol-dependent probands who initially met the DSM-IV for alcohol dependence [33]. Six medical centers in the USA recruited the initial probands plus first-degree family members. The only exclusion criteria include life-threatening medical disorders, repeated intravenous drug use, and an inability to speak English. Written informed consent to participate in the study was obtained from all subjects. Participants and their relatives were interviewed at baseline using the Semi-Structured Assessment for the Genetics of Alcoholism (SSAGA), which focuses on demography, substance use patterns, and the assessment of 17 axis I DSM-IV diagnoses, as well as characteristics of bipolar disorder [16,34].
While the SSAGA was developed prior to the publication of the DSM-IV criteria, all criteria symptoms for the DSM-IV diagnosis were assessed ages of onset and remission of symptoms [35]. Only the original probands or comparison subjects, their first-degree relatives, and offspring aged ≤20 years in the participating families were eligible for follow-up. Of all eligible subjects, the follow-up rate was 60% in probands, 65% in family members, and 78% in controls [35].
The interview also assessed past episodes of affective disorders, including depressive and manic episodes and the characteristics of the most severe episode. To receive a DSM-IV bipolar I disorder diagnosis, subjects had to report a lifetime diagnosis of both major depression and mania or any lifetime diagnosis of a manic episode. Individuals who had at least one major depression and hypomanic episode were considered to have bipolar II disorder.
N = 180 subjects with bipolar I or II disorder were identified. Of these n = 65 (36.1%) had an additional diagnosis of DSM IV CUD (cannabis dependence and cannabis abuse, CUD in 23 of 77 (29.8%) individuals, in bipolar II subjects and 40.8% (42 of 103 individuals in bipolar I subjects). Any CUDs (dependence and abuse) was found in 36.1% of bipolar I and II individuals.
Subjects with a bipolar II disorder without comorbid CUD but abstinent or with social CU were included into group 1 (n = 54) while group 2 (n = 23) consists of individuals with comorbid bipolar II and CUD diagnoses. Group 3 included subjects with a bipolar I diagnosis without CUD (n = 61) but either abstinence or social CU and group 4 were bipolar I subjects with a comorbid CUD (n = 42).
The probands and appropriate relatives were re-assessed at a mean of 5.72 years (±1.1 years) after the initial interview.
2.2. Bipolar Patients with and without Any Cannabis Use Disorders
Of the 180 bipolar subjects interviewed at baseline, n = 117 (65.0%) subjects were successfully re-evaluated at follow-up using the SSAGA (Group 1: 62.9%, n = 34; Group 2: 91.3%; n = 21; Group 3: 37.7%, n = 23; group 4: 95.1%; n = 39). There were no statistically significant differences across groups regarding the rate of being re-interviewed, nor were there differences across groups regarding age, gender and number of symptoms during the most severe depressive or manic episode when re-evaluated subjects were compared to those who could not be re-assessed at follow-up. N = 63 individuals did not agree to be re-interviewed, deceased (n = 2) or could not be located due to address change.
All individuals were interviewed in an outpatient setting usually located in a research lab interview room. All the participants were in euthymic state at time of the interview.
Additional sections of the SSAGA interview were used to determine the age at onset of SUDs (i.e., the age by which three or more criteria were met) and for additional psychiatric diagnoses conditions (e.g., DSM IV anxiety disorders, post-traumatic stress disorders, antisocial personality and conduct disorder).
Psychopathology and behavior during the interview observed by the interviewer included assessments of appearance, orientation, level of consciousness, memory, mood, and formal thought. Global level of functioning was obtained using the Global Assessment of Functioning (GAF) [33] at baseline and at the follow-up interview.
Differences across groups were evaluated by using Chi-square statistics for categorical data and one-way analysis of variance (ANOVA) for continuous variables. Scheffé post hoc tests were used to determine significant differences in specific group comparisons. To compare characteristics of continuous variables over time, repeated measurement ANOVA (MANOVA) was used. For post-hoc group comparisons, Scheffé post-hoc tests were employed, when applicable. Group 2 and 4 individuals were compared for characteristics of CUDs while Group 1 and 3 members (per definition no CUD) were skipped from these analyses.
Moderating effects of any CUD in bipolar patients on number of other substance use (SU) and comorbid mental disorders was computed using SPSS module PROCESS (Ver. 4.1 by Andrew Hayes, http://www.processmacro.org/download.html, accessed 26 July 2023).
3. Results
3.1. Sample Characteristics
The sociodemographic characteristics of the four groups are presented in Table 1. At the baseline interview, no differences were noted across groups regarding years of education, employment, ethnicity, marital status, and education. A significantly higher proportion of Group 1 to 3 members compared to Group 4 were female.

Table 1.
Baseline Demographic Characteristics of Bipolar I and II Subjects Divided by Cannabis Use Disorder Diagnosis.
3.2. Baseline Analyses
3.2.1. Characteristics of Cannabis Use Disorders
Individuals with bipolar I and II with Cannabis use disorders (Groups 2 and 4) were compared and presented in Table 2. No significant differences were found regarding age at onset of Cannabis use, number of units used per day, number of DSM-IV criteria endorsed, number of withdrawal symptoms, and other characteristics, e.g., previous treatment or Cannabis-induced affective or other symptoms.

Table 2.
Baseline Alcohol Dependence-related Characteristics and Treatment Histories of Bipolar I and II Subjects Divided by DSM-IV Cannabis Use Disorder Diagnosis.
3.2.2. Comorbidity with Other DSM-IV Mental, Alcohol, and Substance Use Disorders
As shown in Table 3, posttraumatic stress disorder (PTSD) was diagnosed most often in Group 4, while antisocial personality disorder (ASPD) had the highest rate in Group 3. At baseline, comorbid bipolar I and II individuals also had a significantly higher rate of alcohol, cocaine, and stimulant dependence compared to non-comorbid Groups 1 to 3. Group 2 (bipolar II + CUD) had a higher rate of sedative and opioid dependence compared to Groups 1, 3, and 4.

Table 3.
Baseline Cannabis-related Characteristics and Comorbidity with Mental Disorders of Bipolar I and II Subjects Divided by DSM-IV Cannabis Use Disorder Diagnosis.
3.2.3. Characteristics of Manic and Depressive Episodes, Mixed Episodes, Rapid Cycling, and Suicidal Ideation and Behaviors
As expected, bipolar I Individuals with depression and manic episodes reported receiving significantly more often professional help independent of a comorbid Cannabis use disorder. Comorbid bipolar I individuals also reported a higher number of affective symptoms. General psychosocial functioning was higher in bipolar II vs. bipolar I patients, independent of comorbid CUD. As also presented in Table 4, no differences were found across groups regarding the number of mixed episodes, rapid cycling, and suicidal ideation and behavior.

Table 4.
Baseline Characteristics of Bipolar Disorders of Bipolar I and II Subjects Divided by Cannabis Use Disorder Diagnosis.
3.3. Follow-Up Analyses
3.3.1. Characteristics of Cannabis Use Disorders at the 5-Year Follow-Up
As demonstrated in Table 5, several criteria of CUD were more often found in comorbid bipolar I and bipolar II individuals, including tolerance, withdrawal, and a long time of using and giving up activities. Comorbid bipolar I individuals more often reported a “desire to cut down on use, but could not”, compared to Group 1 to 3 members.

Table 5.
Characteristics of Cannabis Use during 5-year Follow-Up in Bipolar I and II Subjects Divided by Cannabis Use Disorder Diagnosis.
3.3.2. Comorbid Alcohol and Substance Use, Mental Disorders, and Suicidal Behaviors during a Follow-Up
Comorbid alcohol and substance dependence (including cocaine, stimulant, sedative, and opioid dependence) occur more frequently among comorbid bipolar I and II individuals (see Table 6) and resemble diagnostic rates obtained at baseline. Further, members of Groups 1, 3, and 4 developed depressive episodes more frequently, and bipolar I individuals reported more panic attacks. Non-comorbid bipolar I patients (Group 3) reported a higher number of suicide attempts. Rates of other comorbid mental disorders also mainly replicated those from the baseline assessments. Significant differences were found for higher PTSD rates in comorbid bipolar I subjects across groups. Reports of affective symptoms during the follow-up interview were highest in Group 4 (bipolar I and CUD) individuals. Comorbid bipolar I and II individuals had significantly lower GAF scores than Group 1 and 3 individuals.

Table 6.
Characteristics of Alcohol and Substance Dependence during 5-year Follow-Up in Bipolar I and II Subjects Divided by Cannabis Use Disorder Diagnosis.
3.3.3. Cannabis Use Disorders as Potential Moderators for Comorbid Alcohol and Substance Use and Mental Disorders

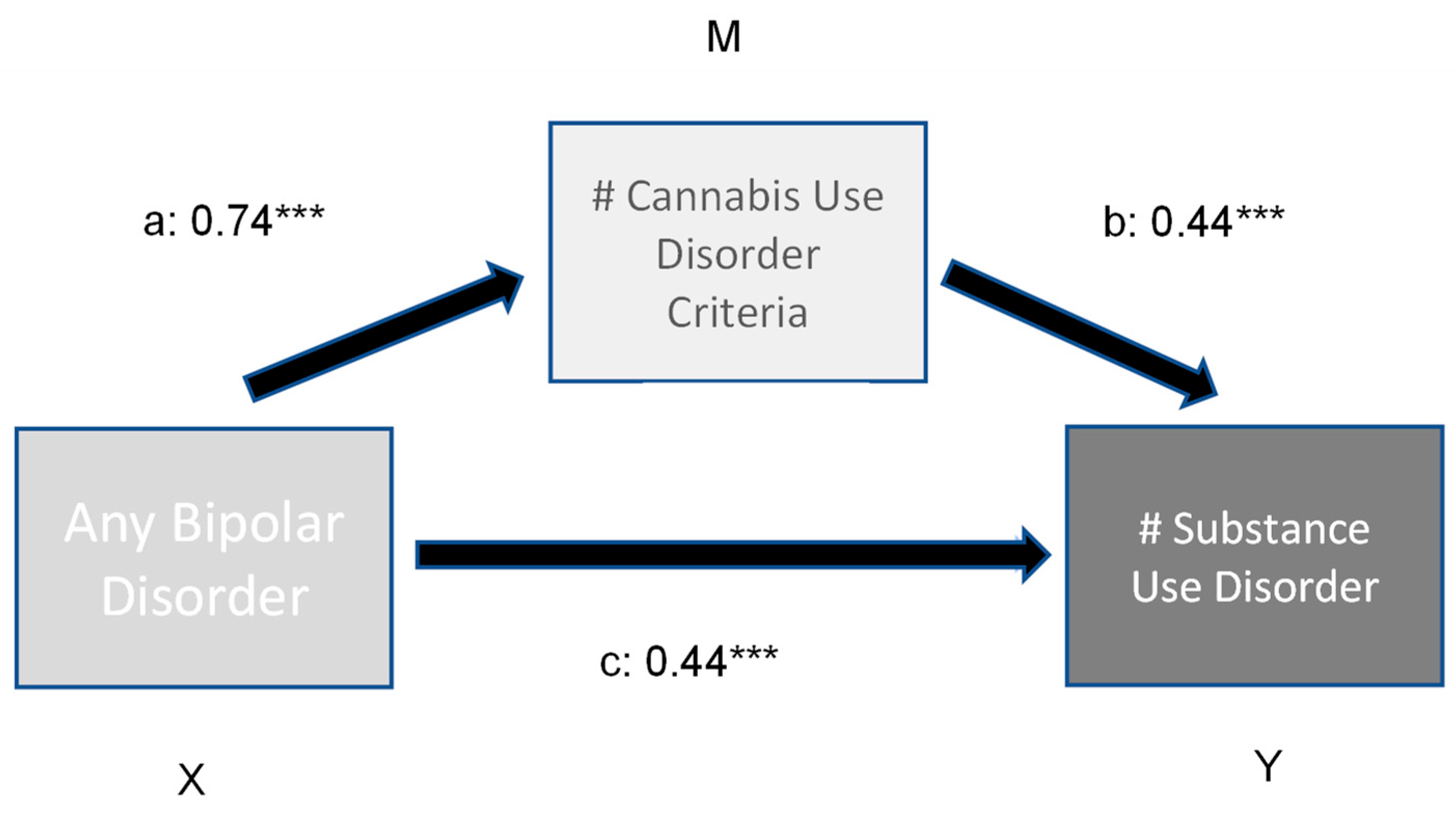
Figure 1.
Moderation effect of number of CUD criteria on number of (#) comorbid substance use disorders in bipolar subjects. Indirect effect (X -> Y) 0.24 ***; ***: p < 0.001.

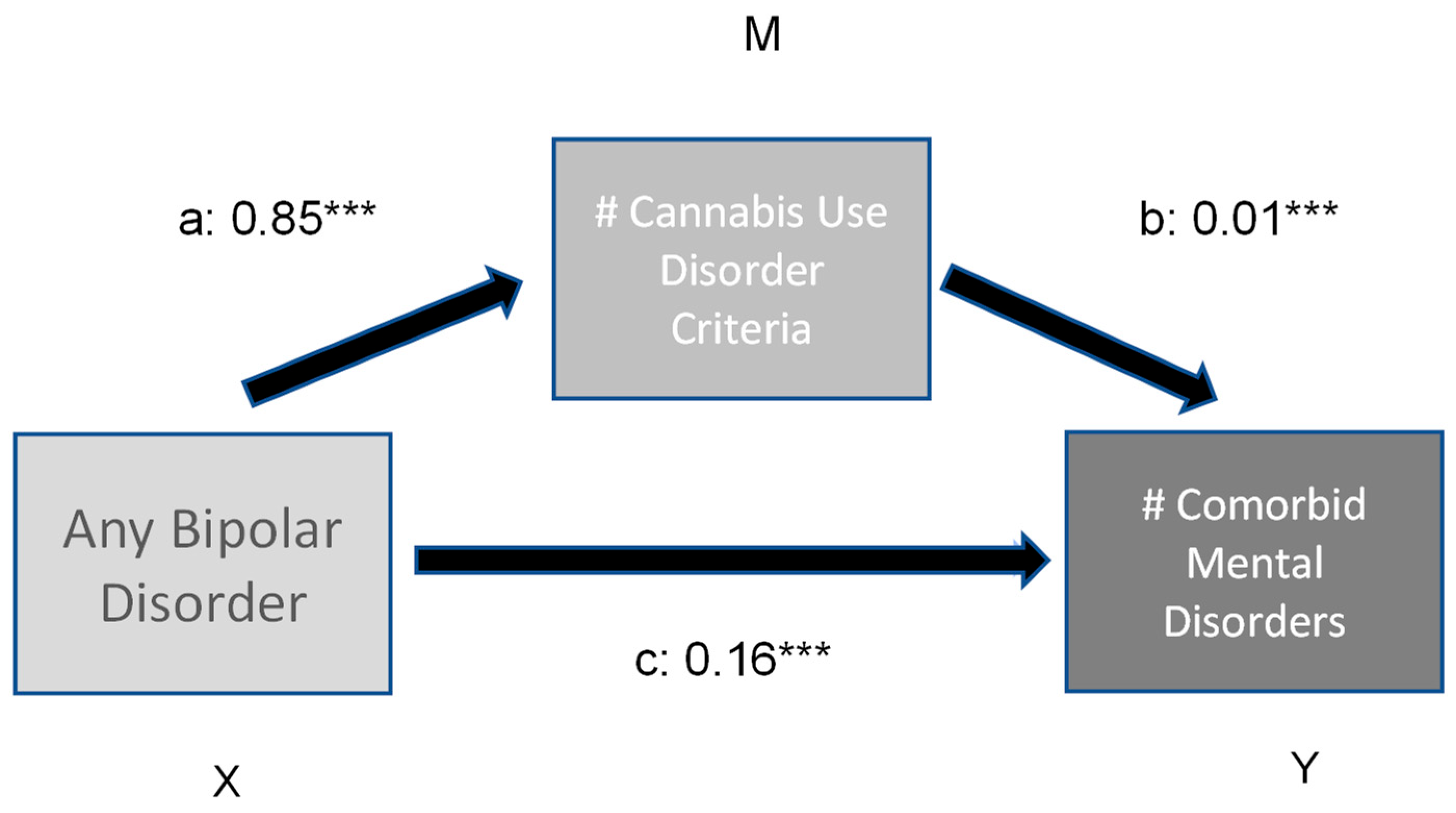
Figure 2.
Moderation effect of number of CUD criteria on number of (#) comorbid mental disorders in bipolar subjects. Indirect effect (X -> Y) 0.01 ***; ***: p < 0.001.
The first model (Figure 1) revealed a significant direct and indirect effect of bipolar disorder on a number of other comorbid substance use disorders. The number of Cannabis use disorder criteria met had a significant moderating effect (variance explained 3.7%). In comparison, the direct and indirect effects of bipolar illness and the number of Cannabis disorder criteria on the prevalence of other comorbid psychiatric disorders (model 2, Figure 2) is low (variance explained 4.8%).
4. Medication and Functional and Affective Syndrome Changes over Time of Bipolar Groups
While there was no difference across groups in the rate of subjects receiving medication during their most severe affective episode, subjects were prescribed antidepressants (35%), benzodiazepines (16%), neuroleptics (18%), lithium (9%), anticonvulsants (13%), or a combination of these medications (23%). At the follow-up interview, no differences were found across groups in type and rate of medication for the treatment of the most severe affective episode. Patients were prescribed antidepressants (31%), benzodiazepines (8%), neuroleptics (13%), lithium (35%), and anticonvulsants (22%). At follow-up, 35% of the bipolar subjects in all groups took a combination of these compounds.
Of the affective symptoms rated by trained SSAGA interviewers, no significant differences between bipolar groups were detected over time (MANOVA F-value: 0.76; df 3; p: 0.842). GAF (social functioning) scores significantly improved in Groups 1, 3, and 4 over time; while for Group 2 subjects, a significant decrease in their level of social functioning was observed (MANOVA F-value: 5.17; df 3; p < 0.02, see Table 6).
5. Discussion
The lifetime characteristics of CUD, other alcohol- and substance dependence and BDs were examined retrospectively while the course of CUD, other alcohol and substance dependence, bipolar disorder and comorbidity with mental and other SUD were examined during the 5-year follow-up period. We also analysed the possible moderating effect of CUD on the risk for other alcohol and SUD or number of comorbid mental illnesses.
The primary focus of our analysis of the COGA-data set was to investigate the course of bipolar I and II with and without CUDs (cannabis abuse and dependence). According to the National Survey on Drug use and Health (NSDUH) [6], the rate of CUD in the US general population accounts for 14.2 million individuals (a prevalence rate of approximately 6.0%). In the current sample, the rate of CUD was 36.1% for bipolar I and II individuals. This finding indicates that bipolar disorders are a considerable risk factor for comorbid CUD. Since this is a sample from a family study, the rate of CUD is higher compared to epidemiological studies (Peters et al., 2014) but lower than rates from clinical samples (47.9%) [9].
With respect to demography, more females not having a CUD were found in bipolar I and II groups. A previous analysis of this dataset in bipolar individuals with and without alcohol dependence had a similar finding [16]. Bipolar individuals without a CUD also had a higher rate of females, except group 4 (CUD and bipolar I disorder). Therefore, bipolar males may be more prone to develop a CUD compared to females.
In both the baseline and prospective analyses of characteristics of hypomania and mania, no differences between bipolar II and bipolar I subjects with and without CUD were detected. Previous studies indicated that CU in BD was associated with hypomanic and manic episodes, but not depression [8,18,20]. Also, no significant differences regarding depression episode characteristics were detected across groups with and without CUD. The statistically significant results presented in Table 4 for mania and depression, including seeking help more often from a professional, refer to differences between mania (bipolar I) and hypomania (bipolar II) or various degrees of depression in these subtypes of bipolar disorders. Further, no differences were found in several additional characteristics of bipolar I and II disorders, including rates of rapid cycling, mixed episodes and suicidal ideation and behavior. Previous studies reported higher rates of complications and more severe bipolar symptoms including more time in manic and mixed episodes [9], more frequent affective episodes [10], mixed states [22]. The differing results found in the current analysis may be due to different sample and sampling characteristics. The current sample is a high-risk family study rather than a clinical treatment sample and may therefore show different patterns of comorbidity.
5.1. Comorbidity with Other Mental Disorders Retro- and Prospectively
Comorbidity with psychiatric disorders, including anxiety disorders and DSM-IV antisocial personality disorder, except for posttraumatic stress disorders (PTSD), surprisingly, did not differ across groups. PTSD had a significantly higher rate in comorbid vs. non-comorbid individuals at baseline and comorbid CUD and bipolar I subjects at follow-up. Subjects with PTSD may, therefore, more often use and abuse cannabinoids to cope with their symptoms cf., anxiety, sleep disorders and intrusions. A recent small randomized controlled trial (RCT) using medical cannabis (nabilone) to treat PTSD found that nabilone significantly reduced the severity of PTSD-related nightmares [36]. Participants also reported significant improvements on secondary measures of general well-being and mean global improvements.
This finding, however, parallels the results from a previous analysis of this sample where no influence of alcohol dependence was reported on these characteristics of bipolar disorder [16]. Additional findings such as the number of affective symptoms at baseline interview were most severe in Group 4 and measures of social functioning (GAF) which were lowest in both bipolar I and II subjects with CUD, may reflect a more severe impairment and more mood disorder symptoms in comorbid BD and CUD individuals.
Comorbid mental disorders in the current analysis included mainly anxiety disorders (panic disorder, PTSD, generalised anxiety disorders, Agoraphobia). Previous biological and genetic studies found a rather weak biological relationship between endogenous cannabinoid system (ECS) functioning [37] or genetics of CUD and anxiety disorders [38], even after controlling for several confounding factors [37]. In comparison, a stronger genetic relationship between affective disorders and ECS has been reported [38]. Recent research also demonstrates that the genetic predisposition for CUD polygenic scores for cannabis phenotypes predicted psychotic disorders independently of other psychiatric disorders [39]. Together these results support the hypothesis that there is little biological or genetic relationship between the ECS and anxiety-related disorders.
5.2. Comorbidity of Any Other Substance Use Disorders
In comparison, rates of other comorbid substance dependence were significantly increased in comorbid vs. non-comorbid groups at both time-points, except for sedative dependence. Further, no significant difference for any SUD was found comparing comorbid bipolar I vs. bipolar II group members. Since cannabinoids also have some sedative and hypnotic characteristics, users of cannabinoids may therefore abuse other sedatives (e.g., benzodiazepines), less often than other substances such as alcohol, stimulants, or cocaine. However, these results do not support a potential protective effect of CU and abuse from sedative dependence. Individuals with multiple SUDs also include CUD along with alcohol, opioids, stimulants, and cocaine use disorders. Further, the results do not support a ‘gateway’ hypothesis of cannabinoids i.e., CU early in life subsequently increases the risk for developing other alcohol and SUDs. To test this “gateway hypothesis”, a different study design (e.g., longitudinal studies on cannabis users during adolescence and early adulthood) is needed.
Other studies confirm that BD with comorbid CU is related with increased frequency of SUDs [21,32]. This is a particularly relevant finding, as co-occurring alcohol- and SUDs among individuals with bipolar disorder are associated with negative effects on course of illness (even after adjusting for non-compliance) including a delayed onset of symptomatic recovery when treated [24,25]. The significance of these findings is underscored when considering the heavy burden of disease already associated with bipolar disorder [26,27].
Few studies hitherto used a prospective design (follow-up between 1 to 3 years) to investigate the course of both comorbid disorders [21,30,31]. In contrast to these studies, few results in this sample support the hypothesis that comorbid CUD cause more severe course of bipolar disorder [30]. However, the higher rates of psychopathology and social functioning could be replicated [31]. Higher rates of comorbidity were found for comorbid PTSD in CUD—bipolar I subjects but no more other anxiety disorders. As with a prospective study [21], higher rates of alcohol- and other substance dependence were detected in the current study while rates of manic and hypomanic syndromes did not differ across bipolar Groups 1 to 4.
Across these types of studies, there are different rates of CU in bipolar disorder. While the current study had high rates of CUDs in bipolar I and II patients (any CUDs in 29.9% of bipolar II and 40.8% in bipolar I subjects). The study of Bahorik et al. [31] reported CU in 27%, Blanco et al. [32] 3.4% in non-bipolar individuals and van Rossum et al. [21] in 12.7% of their samples. However, in this study, compared to previous investigations, data from a highly affected and comorbid sample (CUD and bipolar disorders) are analyzed compared to cannabis use (CU and bipolar disorders) in the previous studies [21,30,31,32]. Thus, individuals in the current sample are affected by multiple substance use and mental comorbidities and have a high rate of comorbid CUD. Further, two previous studies [30,32] used epidemiological samples while clinical patients were enrolled in two other prospective investigations [21,31]. Also, assessment methods differed across prospective studies, using structured interviews and the same sample [30,32], general clinical assessments, self-reports and questionnaires [21] or self-reports, clinical interviews and psychopathology questionnaires [31]. The current sample is from a high-risk genetic family pedigree study. Participants were assessed using structured interviews at two time points four to five years apart which has been reported to have high clinical reliability and validity [34] and may partly explain differing results across studies.
Thus, the comorbid individuals in the current analysis of the COGA sample may be in a different and more severe stage of their comorbid disorder than individuals with CU and bipolar disorders. In this stage, CUD may be more often co-occur with another SUDs and may be less related to comorbid anxiety disorders. The CUD diagnosis indicates that comorbid individuals have not only a frequent CU but also mental and physical consequences due to the use, withdrawal symptoms and problems to stop or control use. These characteristics may be more strongly associated with the risk of other alcohol- and SUDs rather than anxiety symptoms and disorders.
The interactions between CUDs and related disorders and mood disorders are certainly complex. CU and CUD may contribute to psychopathology, which may in turn lead to CU. In this sample, however, more potential underlying factors were found for comorbid alcohol and substance use rather than other mental disorders.
As reported by recent genetic research, there may be a common genetic “addiction” factor which influences all alcohol and SUDs, including CUD, independent of other mental disorders [40,41]. Therefore, the diagnosis of a CUD may increase the liability for developing other alcohol- and SUDs also in these comorbid CUD and bipolar individuals and prospectively not influence the risk for developing an anxiety disorder.
This significant effect of CUD on other SUDs is confirmed by the statistical moderation effects. In this model, a significant indirect effect of CUD (0.44) was found on the relationship between bipolar disorders and number of other alcohol and SUDs. No such effect was detected for CUD on the relationship between bipolar disorders and any other comorbid mental disorders. Thus, comorbid CUD influence significantly the liability for comorbid other SUDs. Certainly, beside genetic factors, other biological and psychosocial mechanisms behind this relationship need to be elucidated in subsequent research.
From the clinical perspective, comorbid CUD and bipolar patients certainly should be assessed regarding additional alcohol- and SUDs. In treatment, the increased risk for other SUDs should be addressed and specific integrated therapy programs in in- and outpatient settings should be provided [42].
There are several limitations to this analysis. First, primarily alcohol-dependent subjects in treatment, their relatives and control families were enrolled into this study. This might explain the rather high rate of alcohol and substance dependence diagnosis in this but may also indicate that a severely comorbid sample with bipolar disorders and several comorbidities in these analyses. Second, several previous investigations included first-episode manic patients to overcome potential bias caused by the number of affective episodes and chronic course of bipolar disorders. The COGA sample included inpatient subjects with and control subjects without alcohol dependence. The enrolees were not first-episode bipolar subjects, compared to other samples [43]. Thus, individuals in a different stage of their disease and more chronic bipolar disorder individuals were recruited. Chronic patients are reported to have a higher rate of previous affective episodes and other comorbidities which in turn increase the likelihood of future affective episodes. When these individuals at different stages of their disease are investigated, it may be more difficult to identify other course predictors and to evaluate the influence of a comorbid disorder on prognosis of bipolar disorder.
As mentioned above, the sample is neither from a clinical or epidemiological population but rather a family study. Comparison of the study’s results to inpatient samples is therefore limited. Further, many but not all potential comorbidities (e.g., full range of DSM-IV personality disorders) were assessed in this family study. However, the structured clinical interview employed (SSAGA) covered many relevant characteristics of comorbid mental and SUDs in bipolar I and II individuals. In previous studies, different assessment methods were employed, including self-reports, global measures of improvement and clinical questionnaires in samples with bipolar disorders and cannabis use. In this sample, comorbid CUD and bipolar disorder subjects were assessed which may in part explain different results across samples. However, the results from the COGA sample may be representative for a non-clinical but severely affected comorbid CUD and bipolar population where a thorough clinical assessment for additional alcohol- and SUDs is indicated.
Further, prospective course of comorbid CUD and bipolar disorders should be investigated in available epidemiological or in clinical samples to confirm results from the current analysis. Treatment studies using integrative therapy approaches covering both comorbid disorders should be initiated to meet the needs of these severely affected individuals.
In the prospective analysis, several additional characteristics of bipolar disorder, including rapid cycling and mixed episode were not traced during the follow-up period. Thus, a higher rate of these potential complications of bipolar disorder in comorbid subjects could not be investigated prospectively.
A significant number of subjects could not be re-interviewed after 5 years, primarily due to changes in addresses and they could not be contacted (approximately 65%). The characteristics of re-interviewed and not-re-interviewed individuals did not differ significantly across groups.
Finally, contrasting differences of baseline and follow-up analyses are often observed and may be due to a patient’s ability to recall recent versus past events. In the COGA study, subjects were assessed at baseline regarding their current and lifetime characteristics. At the follow-up interview, they were again assessed for both their current (including most recent events during the follow-up period) period as well as lifetime characteristics. Thus, individuals might recall the more recent events more completely and accurately than more remote events. This potential recall difference underscores the necessity for prospective studies which might provide more accurate data than cross-sectional data collection designs to reduce individual recall bias.
Author Contributions
V.M.H. and M.N.H. are investigators in the COGA study; U.W.P. contributed to data analysis and writing of the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
The COGA Study is supported by NIH Grant U10AA008401 from the National Institute on Alcohol Abuse and Alcoholism (NIAAA) and the National Institute on Drug Abuse (NIDA).
Institutional Review Board Statement
The COGA protocol was approved by IRBs at all six COGA centers [44].
Informed Consent Statement
Informed consent was obtained from all subjects prior to administering the protocol [44].
Data Availability Statement
COGA Collaboration.
Acknowledgments
The analyses took advantage of the COGA (Collaborative Study on Genetics in Alcoholism) Dataset. The Collaborative Study on the Genetics of Alcoholism (COGA), Principal Investigators B. Porjesz, V. Hesselbrock, T. Foroud; Scientific Director, A. Agrawal; Translational Director, D. Dick, includes ten different centers: University of Connecticut (V. Hesselbrock); Indiana University (H.J. Edenberg, T. Foroud, Y. Liu, M.H. Plawecki); University of Iowa Carver College of Medicine (S. Kuperman, J. Kramer); SUNY Downstate Health Sciences University (B. Porjesz, J. Meyers, C. Kamarajan, A. Pandey); Washington University in St. Louis (L. Bierut, J. Rice, K. Bucholz, A. Agrawal); University of California at San Diego (M. Schuckit); Rutgers University (J. Tischfield, D. Dick, R. Hart, J. Salvatore); The Children’s Hospital of Philadelphia, University of Pennsylvania (L. Almasy); Icahn School of Medicine at Mount Sinai (A. Goate, P. Slesinger); and Howard University (D. Scott). Other COGA collaborators include: L. Bauer (University of Connecticut); J. Nurnberger Jr., L. Wetherill, X., Xuei, D. Lai, S. O’Connor (Indiana University); G. Chan (University of Iowa; University of Connecticut); D.B. Chorlian, J. Zhang, P. Barr, S. Kinreich, G. Pandey (SUNY Downstate); N. Mullins (Icahn School of Medicine at Mount Sinai); A. Anokhin, S. Hartz, E. Johnson, V. McCutcheon, S. Saccone (Washington University); J. Moore, F. Aliev, Z. Pang, S. Kuo (Rutgers University); A. Merikangas (The Children’s Hospital of Philadelphia and University of Pennsylvania); H. Chin and A. Parsian are the NIAAA Staff Collaborators. We continue to be inspired by our memories of Henri Begleiter and Theodore Reich, founding PI and Co-PI of COGA, and also owe a debt of gratitude to other past organizers of COGA, including Ting- Kai Li, P. Michael Conneally, Raymond Crowe, and Wendy Reich, for their critical contributions [44].
Conflicts of Interest
Ulrich W. Preuss, Michie N. Hesselbrock and Victor M. Hesselbrock have no conflicts of interest to disclose.
Abbreviations
| ANOVA | One-way Analysis of Variance |
| ASPD | Antisocial Personality Disorder |
| BD | Bipolar Disorder |
| CA | Cannabis Abuse |
| COGA | Collaborative Study on the Genetics of Alcoholism |
| CU | Cannabis Use |
| CUD | Cannabis Use Disorder |
| DSM-IV | Diagnostic and Statistical Manual Version IV |
| ECS | Endogenous Cannabinoid System |
| GAF | General Assessment of Functioning |
| LT-CU | Life-time Cannabis Use |
| MANOVA | Multivariate Analysis of Variance |
| NSDUH | National Survey on Drug use and Health |
| PTSD | Post-Traumatic Stress Disorder |
| RCT | Randomized Controlled Trial |
| SSAGA | Semi-Structured Assessment on Genetics in Alcoholism |
| SUD | Substance Use Disorders |
| UNODOC | United Nations Office on Drugs and Crime |
References
- Kessler, R.C.; Akiskal, H.S.; Angst, J.; Guyer, M.; Hirschfeld, R.M.; Merikangas, K.R.; Stang, P.E. Validity of the assessment of bipolar spectrum disorders in the WHO CIDI 3.0. J. Affect. Disord. 2006, 96, 259–269. [Google Scholar] [CrossRef] [PubMed]
- Jones, L.; Metcalf, A.; Gordon-Smith, K.; Forty, L.; Perry, A.; Lloyd, J.; Geddes, J.R.; Goodwin, G.M.; Jones, I.; Craddock, N.; et al. Gambling problems in bipolar disorder in the UK: Prevalence and distribution. Br. J. Psychiatry 2015, 207, 328–333. [Google Scholar] [CrossRef] [PubMed]
- Hunt, G.E.; Malhi, G.S.; Cleary, M.; Lai, H.M.; Sitharthan, T. Comorbidity of bipolar and substance use disorders in national surveys of general populations, 1990–2015: Systematic review and meta-analysis. J. Affect. Disord. 2016, 206, 321–330. [Google Scholar] [CrossRef]
- Hunt, G.E.; Malhi, G.S.; Cleary, M.; Lai, H.M.X.; Sitharthan, T. Prevalence of comorbid bipolar and substance use disorders in clinical settings, 1990–2015: Systematic review and meta-analysis. J. Affect. Disord. 2016, 206, 331–349. [Google Scholar] [CrossRef] [PubMed]
- United Nations Office on Drugs Crime. World Drug Report. 2022. Available online: https://www.unodc.org/unodc/en/data-and-analysis/world-drug-report-2022.html (accessed on 12 May 2023).
- Delphin-Rittmon ME 2022 The National Survey on Drug Use and Health. 2020. Available online: https://www.samhsa.gov/data/sites/default/files/reports/slides-2020-nsduh/2020NSDUHNationalSlides072522.pdf (accessed on 12 May 2023).
- Manthey, J.; Freeman, T.P.; Kilian, C.; López-Pelayo, H.; Rehm, J. Public health monitoring of cannabis use in Europe: Prevalence of use, cannabis potency, and treatment rates. Lancet Reg. Health Eur. 2021, 10, 100227. [Google Scholar] [CrossRef] [PubMed]
- Tourjman, S.V.; Buck, G.; Jutras-Aswad, D.; Khullar, A.; McInerney, S.; Saraf, G.; Pinto, J.V.; Potvin, S.; Poulin, M.J.; Frey, B.N.; et al. Canadian Network for Mood and Anxiety Treatments (CANMAT) Task Force Report: A Systematic Review and Recommendations of Cannabis use in Bipolar Disorder and Major Depressive Disorder. Can. J. Psychiatry 2022, 68, 299–311. [Google Scholar] [CrossRef] [PubMed]
- Strakowski, S.M.; DelBello, M.P.; Fleck, D.E.; Adler, C.M.; Anthenelli, R.M.; Keck, P.E.; Arnold, L.M.; Amicone, J. Effects of co-occurring cannabis use disorders on the course of bipolar disorder after a first hospitalization for mania. Arch. Gen. Psychiatry 2007, 64, 57–64. [Google Scholar]
- Lev-Ran, S.; Le Foll, B.; McKenzie, K.; George, T.P.; Rehm, J. Bipolar disorder and co-occurring cannabis use disorders: Characteristics, co-morbidities and clinical correlates. Psychiatry Res. 2013, 209, 459–465. [Google Scholar] [CrossRef] [PubMed]
- Taub, S.; Feingold, D.; Rehm, J.; Lev-Ran, S. Patterns of cannabis use and clinical correlates among individuals with Major Depressive Disorder and Bipolar Disorder. Compr. Psychiatry 2018, 80, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Conway, K.P.; Compton, W.; Stinson, F.S.; Grant, B.F. Lifetime comorbidity of DSM-IV mood and anxiety disorders and specific drug use disorders: Results from the national epidemiologic survey on alcohol and related conditions. J. Clin. Psychiatry 2006, 67, 247–258. [Google Scholar] [CrossRef]
- Kerridge, B.T.; Pickering, R.; Chou, P.; Saha, T.D.; Hasin, D.S. DSM-5 cannabis use disorder in the National Epidemiologic Survey on Alcohol and Related Conditions-III: Gender-specific profiles. Addict. Behav. 2018, 76, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Teesson, M.; Slade, T.; Swift, W.; Mills, K.; Memedovic, S.; Mewton, L.; Grove, R.; Newton, N.; Hall, W. Prevalence, correlates and comorbidity of DSM-IV cannabis use and cannabis use disorders in Australia. Aust. N. Z. J. Psychiatry 2012, 46, 1182–1192. [Google Scholar] [CrossRef] [PubMed]
- Peters, E.N.; Schwartz, R.P.; Wang, S.; O’Grady, K.E.; Blanco, C. Psychiatric, psychosocial, and physical health correlates of co-occurring cannabis use disorders and nicotine dependence. Drug Alcohol Depend. 2014, 134, 228–234. [Google Scholar] [CrossRef]
- Preuss, U.W.; Hesselbrock, M.N.; Hesselbrock, V.M. A Prospective Comparison of Bipolar I and II Subjects With and Without Comorbid Alcohol Dependence From the COGA Dataset. Front. Psychiatry 2020, 11, 522228. [Google Scholar] [CrossRef]
- Salloum, I.M.; Brown, E.S. Management of comorbid bipolar disorder and substance use disorders. Am. J. Drug Alcohol Abus. 2017, 43, 366–376. [Google Scholar] [CrossRef]
- Baethge, C.; Hennen, J.; Khalsa, H.M.K.; Salvatore, P.; Tohen, M.; Baldessarini, R.J. Sequencing of substance use and affective morbidity in 166 first-episode bipolar I disorder patients. Bipolar Disord. 2008, 10, 738–741. [Google Scholar] [CrossRef] [PubMed]
- Patel, R.S.; Manikkara, G.; Chopra, A. Bipolar Disorder and Comorbid Borderline Personality Disorder: Patient Characteristics and Outcomes in US Hospitals. Medicina 2019, 55, 13. [Google Scholar] [CrossRef]
- Patel, R.S.; Cheema, Z.; Singla, A.; Cornejo, M.; Verma, G. Cannabis Use is an Independent Risk Factor for Manic Episode: A Report from 380,265 Bipolar Inpatients. Subst. Use Misuse 2022, 57, 344–349. [Google Scholar] [CrossRef]
- Van Rossum, I.; Boomsma, M.; Tenback, D.; Reed, C.; van Os, J. Does cannabis use affect treatment outcome in bipolar disorder? A longitudinal analysis. J. Nerv. Ment. Dis. 2009, 197, 35–40. [Google Scholar] [CrossRef]
- Agrawal, A.; Nurnberger, J.I.; Lynskey, M.T. Cannabis involvement in individuals with bipolar disorder. Psychiatry Res. 2011, 185, 459–461. [Google Scholar] [CrossRef]
- Weinstock, L.M.; Gaudiano, B.A.; Wenze, S.J.; Epstein-Lubow, G.; Miller, I.W. Demographic and clinical characteristics associated with comorbid cannabis use disorders (CUDs) in hospitalized patients with bipolar I disorder. Compr. Psychiatry 2016, 65, 57–62. [Google Scholar] [CrossRef]
- Strakowski, S.M.; DelBello, M.P.; Fleck, D.E.; Arndt, S. The impact of substance abuse on the course of bipolar disorder. Biol. Psychiatry 2000, 48, 477–485. [Google Scholar] [CrossRef]
- Fossey, M.D.; Otto, M.W.; Yates, W.R.; Wisniewski, S.R.; Gyulai, L.; Allen, M.H.; Miklowitz, D.J.; Coon, K.A.; Ostacher, M.J.; Step-BD Investigators. Validity of the distinction between primary and secondary substance use disorder in patients with bipolar disorder: Data from the first 1000 STEP-BD participants. Am. J. Addict. 2006, 15, 138–143. [Google Scholar] [CrossRef] [PubMed]
- Wyatt, R.J.; Henter, I. An economic evaluation of manic-depressive illness—1991. Soc. Psychiatry Psychiatr. Epidemiol. 1995, 30, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Begley, C.E.; Annegers, J.F.; Swann, A.C.; Lewis, C.; Coan, S.; Schnapp, W.B.; Bryant-Comstock, L. The lifetime cost of bipolar disorder in the US: An. estimate for new cases in 1998. Pharmacoeconomics 2001, 19, 483–495. [Google Scholar] [CrossRef] [PubMed]
- Vázquez, G.H.; Baldessarini, R.J.; Tondo, L. Co-occurrence of anxiety and bipolar disorders: Clinical and therapeutic overview. Depress. Anxiety 2014, 31, 196–206. [Google Scholar] [CrossRef] [PubMed]
- Patel, R.S.; Virani, S.; Saeed, H.; Nimmagadda, S.; Talukdar, J.; Youssef, N.A. Gender Differences and Comorbidities in U.S. Adults with Bipolar Disorder. Brain Sci. 2018, 8, 168. [Google Scholar] [CrossRef]
- Feingold, D.; Weiser, M.; Rehm, J.; Lev-Ran, S. The association between cannabis use and mood disorders: A longitudinal study. J. Affect. Disord. 2015, 172, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Bahorik, A.L.; Newhill, C.E.; Eack, S.M. Characterizing the longitudinal patterns of substance use among individuals diagnosed with serious mental illness after psychiatric hospitalization. Addiction 2013, 108, 1259–1269. [Google Scholar] [CrossRef] [PubMed]
- Blanco, C.; Hasin, D.S.; Wall, M.M.; Flórez-Salamanca, L.; Hoertel, N.; Wang, S.; Kerridge, B.T.; Olfson, M. Cannabis use and risk of psychiatric disorders: Prospective evidence from an US national longitudinal study. JAMA Psychiatry 2016, 73, 388–395. [Google Scholar] [CrossRef]
- APA American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 4th ed.; American Psychiatric Association: Washington, DC, USA, 1994. [Google Scholar]
- Hesselbrock, M.; Easton, C.; Bucholz, K.K.; Schuckit, M.; Hesselbrock, V. A validity study of the SSAGA—A comparison with the SCAN. Addiction 1994, 94, 1361–1370. [Google Scholar]
- Culverhouse, R.; Bucholz, K.K.; Crowe, R.R.; Hesselbrock, V.; Nurnberger, J.I., Jr.; Porjesz, B.; Schuckit, M.A.; Reich, T.; Bierut, L.J. Long-term stability of alcohol and other substance dependence diagnoses and habitual smoking: An evaluation after 5 years. Arch. Gen. Psychiatry 2005, 62, 753–760. [Google Scholar] [CrossRef] [PubMed]
- Jetly, R.; Heber, A.; Fraser, G.; Boisvert, D. The efficacy of nabilone, a synthetic cannabinoid, in the treatment of PTSD-associated nightmares: A preliminary randomized, double-blind, placebo-controlled cross-over design study. Psychoneuroendocrinology 2015, 51, 585–588. [Google Scholar] [CrossRef] [PubMed]
- Hasbi, A.; Madras, B.K.; George, S.R. Endocannabinoid System and Exogenous Cannabinoids in Depression and Anxiety: A Review. Brain Sci. 2023, 13, 325. [Google Scholar] [CrossRef] [PubMed]
- Lowe, D.J.E.; Sasiadek, J.D.; Coles, A.S.; George, T.P. Cannabis and mental illness: A review. Eur. Arch. Psychiatry Clin. Neurosci. 2019, 269, 107–120. [Google Scholar] [CrossRef] [PubMed]
- Hill, M.N.; Gorzalka, B.B. Impairments in endocannabinoid signaling and depressive illness. JAMA 2009, 301, 1165–1166. [Google Scholar] [CrossRef] [PubMed]
- Cheng, W.; Parker, N.; Karadag, N.; Koch, E.; Hindley, G.; Icick, R.; Shadrin, A.; O’Connell, K.S.; Bjella, T.; Bahrami, S.; et al. The relationship between cannabis use, schizophrenia, and bipolar disorder: A genetically informed study. Lancet Psychiatry 2023, 10, 441–451. [Google Scholar] [PubMed]
- Hatoum, A.S.; Johnson, E.C.; Colbert, S.M.C.; Polimanti, R.; Zhou, H.; Walters, R.K.; Gelernter, J.; Edenberg, H.J.; Bogdan, R.; Agrawal, A. The addiction risk factor: A unitary genetic vulnerability characterizes substance use disorders and their associations with common correlates. Neuropsychopharmacology 2022, 47, 1739–1745. [Google Scholar] [CrossRef] [PubMed]
- Preuss, U.W.; Moggi, F. Affektive Störungen und Sucht; Kohlhammer: Stuttgart, Germany, 2023. [Google Scholar]
- Strakowski, S.M.; Sax, K.W.; McElroy, S.L.; Keck, P.E., Jr.; Hawkins, J.M.; West, S.A. Course of psychiatric and substance abuse syndromes co-occurring with bipolar disorder after a first psychiatric hospitalization. J. Clin. Psychiatry 1998, 59, 465–471. [Google Scholar] [CrossRef] [PubMed]
- Bucholz, K.K.; Hesselbrock, V.M.; Heath, A.C.; Kramer, J.R.; Schuckit, M.A. A latent class analysis of antisocial personality disorder symptom data from a multi-centre family study of alcoholism. Addiction 2000, 95, 553–567. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).