Exoscopic Microsurgery: A Change of Paradigm in Brain Tumor Surgery? Comparison with Standard Operative Microscope
Abstract
1. Introduction
2. Materials and Methods
2.1. Design and Inclusion Criteria
2.2. Exclusion Criteria
2.3. Data Collection
2.4. Radiological Data
2.5. Neuro-Oncological Treatment and Follow-Up
2.6. Statistical Analysis
3. Results
3.1. Study Population
3.2. Overall Outcome
3.3. Exoscope and Microscope Comparison
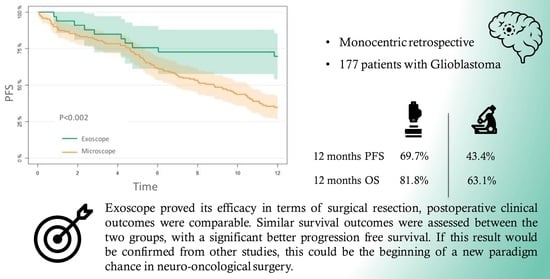
3.4. PFS
3.5. OS
3.6. Cox Model
4. Discussion
4.1. Surgical Resection of Gliomas: Implication of Introduction of a 3D Exoscope
4.2. Our Experience
4.3. Limitations of the Study
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bernardo, A. The Changing Face of Technologically Integrated Neurosurgery: Today’s High-Tech Operating Room. World Neurosurg. 2017, 106, 1001–1014. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.I.; Javed, G.; Mubeen, B.; Bareeqa, S.B.; Rasheed, H.; Rehman, A.; Phulpoto, M.M.; Samar, S.S.; Aziz, K. Robotics in neurosurgery: A literature review. J. Pak. Med. Assoc. 2018, 68, 258–263. [Google Scholar]
- Senders, J.T.; Arnaout, O.; Karhade, A.V.; Dasenbrock, H.H.; Gormley, W.B.; Broekman, M.L.; Smith, T.R. Natural and Artificial Intelligence in Neurosurgery: A Systematic Review. Neurosurgery 2018, 83, 181–192. [Google Scholar] [CrossRef] [PubMed]
- Uluç, K.; Kujoth, G.C.; Başkaya, M.K. Operating microscopes: Past, present, and future. Neurosurg. Focus 2009, 27, E4. [Google Scholar] [CrossRef] [PubMed]
- Miller, K.D.; Ostrom, Q.T.; Kruchko, C.; Patil, N.; Tihan, T.; Cioffi, G.; Fuchs, H.E.; Waite, K.A.; Jemal, A.; Siegel, R.L.; et al. Brain and other central nervous system tumor statistics, 2021. CA. Cancer J. Clin. 2021, 71, 381–406. [Google Scholar] [CrossRef]
- Ostrom, Q.T.; Cioffi, G.; Waite, K.; Kruchko, C.; Barnholtz-Sloan, J.S. CBTRUS Statistical Report: Primary Brain and Other Central Nervous System Tumors Diagnosed in the United States in 2014–2018. Neuro. Oncol. 2021, 23, iii1–iii105. [Google Scholar] [CrossRef]
- Youngblood, M.W.; Stupp, R.; Sonabend, A.M. Role of Resection in Glioblastoma Management. Neurosurg. Clin. N. Am. 2021, 32, 9–22. [Google Scholar] [CrossRef]
- Stupp, R.; Hegi, M.E.; Mason, W.P.; van den Bent, M.J.; Taphoorn, M.J.B.; Janzer, R.C.; Ludwin, S.K.; Allgeier, A.; Fisher, B.; Belanger, K.; et al. Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet. Oncol. 2009, 10, 459–466. [Google Scholar] [CrossRef]
- Stupp, R.; Mason, W.P.; van den Bent, M.J.; Weller, M.; Fisher, B.; Taphoorn, M.J.B.; Belanger, K.; Brandes, A.A.; Marosi, C.; Bogdahn, U.; et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N. Engl. J. Med. 2005, 352, 987–996. [Google Scholar] [CrossRef]
- Yordanova, Y.N.; Duffau, H. Supratotal resection of diffuse gliomas—An overview of its multifaceted implications. Neurochirurgie 2017, 63, 243–249. [Google Scholar] [CrossRef]
- Duffau, H. Is supratotal resection of glioblastoma in noneloquent areas possible? World Neurosurg. 2014, 82, e101–e103. [Google Scholar] [CrossRef] [PubMed]
- Certo, F.; Stummer, W.; Farah, J.O.; Freyschlag, C.; Visocchi, M.; Morrone, A.; Altieri, R.; Toccaceli, G.; Peschillo, S.; Thomè, C.; et al. Supramarginal resection of glioblastoma: 5-ALA fluorescence, combined intraoperative strategies and correlation with survival. J. Neurosurg. Sci. 2019, 63, 625–632. [Google Scholar] [CrossRef] [PubMed]
- Molinaro, A.M.; Hervey-Jumper, S.; Morshed, R.A.; Young, J.; Han, S.J.; Chunduru, P.; Zhang, Y.; Phillips, J.J.; Shai, A.; Lafontaine, M.; et al. Association of Maximal Extent of Resection of Contrast-Enhanced and Non-Contrast-Enhanced Tumor With Survival Within Molecular Subgroups of Patients With Newly Diagnosed Glioblastoma. JAMA Oncol. 2020, 6, 495–503. [Google Scholar] [CrossRef] [PubMed]
- Hervey-Jumper, S.L.; Berger, M.S. Maximizing safe resection of low- and high-grade glioma. J. Neurooncol. 2016, 130, 269–282. [Google Scholar] [CrossRef]
- Sanai, N.; Polley, M.-Y.; McDermott, M.W.; Parsa, A.T.; Berger, M.S. An extent of resection threshold for newly diagnosed glioblastomas. J. Neurosurg. 2011, 115, 3–8. [Google Scholar] [CrossRef]
- Giussani, C.; Carrabba, G.; Rui, C.B.; Chiarello, G.; Stefanoni, G.; Julita, C.; De Vito, A.; Cinalli, M.A.; Basso, G.; Remida, P.; et al. Perilesional resection technique of glioblastoma: Intraoperative ultrasound and histological findings of the resection borders in a single center experience. J. Neurooncol. 2023, 161, 625–632. [Google Scholar] [CrossRef]
- Di Cristofori, A.; Basso, G.; de Laurentis, C.; Mauri, I.; Sirtori, M.A.; Ferrarese, C.; Isella, V.; Giussani, C. Perspectives on (A)symmetry of Arcuate Fasciculus. A Short Review About Anatomy, Tractography and TMS for Arcuate Fasciculus Reconstruction in Planning Surgery for Gliomas in Language Areas. Front. Neurol. 2021, 12, 639822. [Google Scholar] [CrossRef]
- Ricciardi, L.; Chaichana, K.L.; Cardia, A.; Stifano, V.; Rossini, Z.; Olivi, A.; Sturiale, C.L. The Exoscope in Neurosurgery: An Innovative “Point of View”. A Systematic Review of the Technical, Surgical, and Educational Aspects. World Neurosurg. 2019, 124, 136–144. [Google Scholar] [CrossRef]
- Fiani, B.; Jarrah, R.; Griepp, D.W.; Adukuzhiyil, J. The Role of 3D Exoscope Systems in Neurosurgery: An Optical Innovation. Cureus 2021, 13, e15878. [Google Scholar] [CrossRef]
- Baron, R.B.; Lakomkin, N.; Schupper, A.J.; Nistal, D.; Nael, K.; Price, G.; Hadjipanayis, C.G. Postoperative outcomes following glioblastoma resection using a robot-assisted digital surgical exoscope: A case series. J. Neurooncol. 2020, 148, 519–527. [Google Scholar] [CrossRef]
- Calloni, T.; Antolini, L.; Roumy, L.-G.; Nicolosi, F.; Carrabba, G.G.; Di Cristofori, A.; Fontanella, M.M.; Giussani, C.G. Exoscope and operative microscope for training in microneurosurgery: A laboratory investigation on a model of cranial approach. Front. Surg. 2023, 10, 1150981. [Google Scholar] [CrossRef] [PubMed]
- O’Malley, B.W.J.; Grady, M.S.; Gabel, B.C.; Cohen, M.A.; Heuer, G.G.; Pisapia, J.; Bohman, L.-E.; Leibowitz, J.M. Comparison of endoscopic and microscopic removal of pituitary adenomas: Single-surgeon experience and the learning curve. Neurosurg. Focus 2008, 25, E10. [Google Scholar] [CrossRef]
- Lorentzen, H.; Weismann, K.; Secher, L.; Petersen, C.S.; Larsen, F.G. The dermatoscopic ABCD rule does not improve diagnostic accuracy of malignant melanoma. Acta Derm. Venereol. 1999, 79, 469–472. [Google Scholar] [CrossRef]
- Louis, D.N.; Perry, A.; Wesseling, P.; Brat, D.J.; Cree, I.A.; Figarella-Branger, D.; Hawkins, C.; Ng, H.K.; Pfister, S.M.; Reifenberger, G. The 2021 WHO classification of tumors of the central nervous system: A summary. Neuro. Oncol. 2021, 23, 1231–1251. [Google Scholar] [CrossRef] [PubMed]
- Montemurro, N.; Scerrati, A.; Ricciardi, L.; Trevisi, G. The Exoscope in Neurosurgery: An Overview of the Current Literature of Intraoperative Use in Brain and Spine Surgery. J. Clin. Med. 2021, 11, 223. [Google Scholar] [CrossRef]
- Della Pepa, G.M.; Mattogno, P.; Menna, G.; Agostini, L.; Olivi, A.; Doglietto, F. A Comparative Analysis with Exoscope and Optical Microscope for Intraoperative Visualization and Surgical Workflow in 5-Aminolevulinic Acid-Guided Resection of High-Grade Gliomas. World Neurosurg. 2023, 170, 133–137. [Google Scholar] [CrossRef] [PubMed]
- Abunimer, A.M.; Abou-Al-Shaar, H.; White, T.G.; Park, J.; Schulder, M. The Utility of High-Definition 2-Dimensional Stereotactic Exoscope in Cranial and Spinal Procedures. World Neurosurg. 2022, 158, e231–e236. [Google Scholar] [CrossRef]
- Keric, N.; Krenzlin, H.; Kurz, E.; Wesp, D.M.A.; Kalasauskas, D.; Ringel, F. Evaluation of 3D Robotic-Guided Exoscopic Visualization in Microneurosurgery. Front. Surg. 2021, 8, 791427. [Google Scholar] [CrossRef]
- Vogelbaum, M.A.; Kroll, D.; Etame, A.; Tran, N.; Liu, J.; Ford, A.; Sparr, E.; Kim, Y.; Forsyth, P.; Sahebjam, S.; et al. A Prospective Validation Study of the First 3D Digital Exoscope for Visualization of 5-ALA-Induced Fluorescence in High-Grade Gliomas. World Neurosurg. 2021, 149, e498–e503. [Google Scholar] [CrossRef]
- Mamelak, A.N.; Drazin, D.; Shirzadi, A.; Black, K.L.; Berci, G. Infratentorial supracerebellar resection of a pineal tumor using a high definition video exoscope (VITOM®). J. Clin. Neurosci. 2012, 19, 306–309. [Google Scholar] [CrossRef]
- Birch, K.; Drazin, D.; Black, K.L.; Williams, J.; Berci, G.; Mamelak, A.N. Clinical experience with a high definition exoscope system for surgery of pineal region lesions. J. Clin. Neurosci. 2014, 21, 1245–1249. [Google Scholar] [CrossRef] [PubMed]
- Huff, W.X.; Witten, A.J.; Shah, M. V Resection of a pineal region papillary tumor using robotic exoscope: Improved visualization and ergonomics for deep seeded tumor. Neurosurg. Focus Video 2021, 5, V14. [Google Scholar] [CrossRef] [PubMed]
- Schupper, A.J.; Eskandari, R.; Kosnik-Infinger, L.; Olivera, R.; Nangunoori, R.; Patel, S.; Williamson, R.; Yu, A.; Hadjipanayis, C.G. A Multicenter Study Investigating the Surgeon Experience with a Robotic-Assisted Exoscope as Part of the Neurosurgical Armamentarium. World Neurosurg. 2023, 173, e571–e577. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Iwami, K.; Kishida, Y.; Nagatani, T.; Yatsuya, H.; Miyachi, S. Combined Exoscopic and Endoscopic Two-Step Keyhole Approach for Intracranial Meningiomas. Curr. Oncol. 2022, 29, 5370–5382. [Google Scholar] [CrossRef]
- Haeren, R.; Hafez, A.; Lehecka, M. Visualization and Maneuverability Features of a Robotic Arm Three-Dimensional Exoscope and Operating Microscope for Clipping an Unruptured Intracranial Aneurysm: Video Comparison and Technical Evaluation. Oper. Neurosurg. 2022, 22, 28–34. [Google Scholar] [CrossRef]
- Calloni, T.; Roumy, L.G.; Cinalli, M.A.; Rocca, A.; Held, A.; Trezza, A.; Carrabba, G.G.; Giussani, C.G. Exoscope as a Teaching Tool: A Narrative Review of the Literature. Front. Surg. 2022, 9, 878293. [Google Scholar] [CrossRef]
- Chu, T.-S.; Chu, T.-H.; Huynh, T.-D.; Phan, V.-D.; Dang, B.N.; Tran, Q.D. Radical resection of trigeminal schwannoma at the cerebellopontine angle with support of the digital robotic exoscope Synaptive Modus V system: A case report and literature review. Medicine 2023, 102, e33492. [Google Scholar] [CrossRef]
- Schupper, A.J.; Roa, J.A.; Hadjipanayis, C.G. Contemporary intraoperative visualization for GBM with use of exoscope, 5-ALA fluorescence-guided surgery and tractography. Neurosurg. Focus Video 2022, 6, V5. [Google Scholar] [CrossRef]
- Maeda, M.; Nonaka, M.; Naito, N.; Ueno, K.; Kamei, T.; Asai, A. 5-ALA fluorescence-guided resection of pediatric low-grade glioma using the ORBEYE 3D digital exoscope: A technical report. Child’s Nerv. Syst. 2023, 39, 1061–1064. [Google Scholar] [CrossRef]
- Beez, T.; Munoz-Bendix, C.; Beseoglu, K.; Steiger, H.-J.; Ahmadi, S.A. First Clinical Applications of a High-Definition Three-Dimensional Exoscope in Pediatric Neurosurgery. Cureus 2018, 10, e2108. [Google Scholar] [CrossRef]
- Gassie, K.; Wijesekera, O.; Chaichana, K.L. Minimally invasive tubular retractor-assisted biopsy and resection of subcortical intra-axial gliomas and other neoplasms. J. Neurosurg. Sci. 2018, 62, 682–689. [Google Scholar] [CrossRef]
- Ikeda, N.; Furuse, M.; Futamura, G.; Kimura, S.; Nonoguchi, N.; Kawabata, S.; Kameda, M.; Yokoyama, K.; Takami, T.; Kawanishi, M.; et al. The Characteristic of Light Sources and Fluorescence in the 3-Dimensional Digital Exoscope “ORBEYE” for 5-Aminolevulinic Acid-Induced Fluorescence-Guided Surgery Compared with a Conventional Microscope. World Neurosurg. 2022, 167, e1268–e1274. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, S.; Toda, M.; Nishimoto, M.; Ishihara, E.; Miwa, T.; Akiyama, T.; Horiguchi, T.; Sasaki, H.; Yoshida, K. Pros and cons of using ORBEYETM for microneurosurgery. Clin. Neurol. Neurosurg. 2018, 174, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Khalessi, A.A.; Rahme, R.; Rennert, R.C.; Borgas, P.; Steinberg, J.A.; White, T.G.; Santiago-Dieppa, D.R.; Boockvar, J.A.; Hatefi, D.; Pannell, J.S.; et al. First-in-Man Clinical Experience Using a High-Definition 3-Dimensional Exoscope System for Microneurosurgery. Oper. Neurosurg. 2019, 16, 717–725. [Google Scholar] [CrossRef]
- Muscas, G.; Battista, F.; Boschi, A.; Morone, F.; Della Puppa, A. A Single-Center Experience with the Olympus ORBEYE 4K-3D Exoscope for Microsurgery of Complex Cranial Cases: Technical Nuances and Learning Curve. J. Neurol. Surg. A Cent. Eur. Neurosurg. 2021, 82, 484–489. [Google Scholar] [CrossRef]
- Haglund, M.M.; Berger, M.S.; Shamseldin, M.; Lettich, E.; Ojemann, G.A. Cortical localization of temporal lobe language sites in patients with gliomas. Neurosurgery 1994, 34, 567–576; discussion 576. [Google Scholar] [CrossRef] [PubMed]
- Prada, F.; Del Bene, M.; Moiraghi, A.; Casali, C.; Legnani, F.G.; Saladino, A.; Perin, A.; Vetrano, I.G.; Mattei, L.; Richetta, C.; et al. From Grey Scale B-Mode to Elastosonography: Multimodal Ultrasound Imaging in Meningioma Surgery-Pictorial Essay and Literature Review. Biomed Res. Int. 2015, 2015, 925729. [Google Scholar] [CrossRef]
- Strickland, B.A.; Zada, G. 5-ALA Enhanced Fluorescence-Guided Microscopic to Endoscopic Resection of Deep Frontal Subcortical Glioblastoma Multiforme. World Neurosurg. 2021, 148, 65. [Google Scholar] [CrossRef]
- Duffau, H. Intraoperative direct subcortical stimulation for identification of the internal capsule, combined with an image-guided stereotactic system during surgery for basal ganglia lesions. Surg. Neurol. 2000, 53, 250–254. [Google Scholar] [CrossRef]
- Bello, L.; Gallucci, M.; Fava, M.; Carrabba, G.; Giussani, C.; Acerbi, F.; Baratta, P.; Songa, V.; Conte, V.; Branca, V.; et al. Intraoperative subcortical language tract mapping guides surgical removal of gliomas involving speech areas. Neurosurgery 2007, 60, 62–67. [Google Scholar] [CrossRef]
- Bello, L.; Acerbi, F.; Giussani, C.; Baratta, P.; Taccone, P.; Songa, V.; Fava, M.; Stocchetti, N.; Papagno, C.; Gaini, S.M. Intraoperative language localization in multilingual patients with gliomas. Neurosurgery 2006, 59, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Orringer, D.A.; Golby, A.; Jolesz, F. Neuronavigation in the surgical management of brain tumors: Current and future trends. Expert Rev. Med. Devices 2012, 9, 491–500. [Google Scholar] [CrossRef] [PubMed]
- Rohde, V.; Spangenberg, P.; Mayfrank, L.; Reinges, M.; Gilsbach, J.M.; Coenen, V.A. Advanced neuronavigation in skull base tumors and vascular lesions. Minim. Invasive Neurosurg. 2005, 48, 13–18. [Google Scholar] [CrossRef]
- Veiceschi, P.; Locatelli, D.; Dario, A.; Agresta, G. Frameless neuronavigation-assisted brain biopsy with electromagnetic tracking: How I do it? Acta Neurochir. 2022, 164, 3317–3322. [Google Scholar] [CrossRef] [PubMed]
- Motomura, K.; Saito, R. Intraoperative Functional Monitoring in Brain Tumor Surgery. No Shinkei Geka. 2023, 51, 481–489. [Google Scholar] [CrossRef]
- Shao, K.N.; Chen, S.S.; Lee, L.S. Intraoperative neurosurgical ultrasonography. Zhonghua Yi Xue Za Zhi 1999, 62, 775–781. [Google Scholar]
- Prada, F.; Del Bene, M.; Mattei, L.; Lodigiani, L.; DeBeni, S.; Kolev, V.; Vetrano, I.; Solbiati, L.; Sakas, G.; DiMeco, F. Preoperative magnetic resonance and intraoperative ultrasound fusion imaging for real-time neuronavigation in brain tumor surgery. Ultraschall Med. 2015, 36, 174–186. [Google Scholar] [CrossRef]
- Regelsberger, J.; Fritzsche, E.; Langer, N.; Westphal, M. Intraoperative sonography of intra- and extramedullary tumors. Ultrasound Med. Biol. 2005, 31, 593–598. [Google Scholar] [CrossRef]
- Prada, F.; Del Bene, M.; Rampini, A.; Mattei, L.; Casali, C.; Vetrano, I.G.; Gennari, A.G.; Sdao, S.; Saini, M.; Sconfienza, L.M.; et al. Intraoperative Strain Elastosonography in Brain Tumor Surgery. Oper. Neurosurg. 2019, 17, 227–236. [Google Scholar] [CrossRef]
- Seifert, V. Intraoperative MRI in neurosurgery: Technical overkill or the future of brain surgery? Neurol. India 2003, 51, 329–332. [Google Scholar]
- Nabavi, A.; Dörner, L.; Stark, A.M.; Mehdorn, H.M. Intraoperative MRI with 1.5 Tesla in neurosurgery. Neurosurg. Clin. N. Am. 2009, 20, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Di Cristofori, A.; Carone, G.; Rocca, A.; Rui, C.B.; Trezza, A.; Carrabba, G.; Giussani, C. Fluorescence and Intraoperative Ultrasound as Surgical Adjuncts for Brain Metastases Resection: What Do We Know? A Systematic Review of the Literature. Cancers 2023, 15, 2047. [Google Scholar] [CrossRef] [PubMed]
- Fatemi, P.; Zhang, M.; Miller, K.J.; Robe, P.; Li, G. How Intraoperative Tools and Techniques Have Changed the Approach to Brain Tumor Surgery. Curr. Oncol. Rep. 2018, 20, 89. [Google Scholar] [CrossRef]
- Witten, A.J.; Ben-Shalom, N.; Ellis, J.A.; Boockvar, J.A.; D’Amico, R.S. Optimization of novel exoscopic blue light filter during fluorescence-guided resection of Glioblastoma. J. Neurooncol. 2023, 161, 617–623. [Google Scholar] [CrossRef] [PubMed]
- Murai, Y.; Sato, S.; Yui, K.; Morimoto, D.; Ozeki, T.; Yamaguchi, M.; Tateyama, K.; Nozaki, T.; Tahara, S.; Yamaguchi, F.; et al. Preliminary Clinical Microneurosurgical Experience With the 4K3-Dimensional Microvideoscope (ORBEYE) System for Microneurological Surgery: Observation Study. Oper. Neurosurg. 2019, 16, 707–716. [Google Scholar] [CrossRef]
- Doglietto, F.; Belotti, F.; Panciani, P.; Poliani, P.L.; Fontanella, M.M. High-Definition 3-Dimensional Exoscope for 5-ALA Glioma Surgery: 3-Dimensional Operative Video. Oper. Neurosurg. 2020, 18, E82. [Google Scholar] [CrossRef]
- Belloch, J.P.; Rovira, V.; Llácer, J.L.; Riesgo, P.A.; Cremades, A. Fluorescence-guided surgery in high grade gliomas using an exoscope system. Acta Neurochir. 2014, 156, 653–660. [Google Scholar] [CrossRef]
- Marenco-Hillembrand, L.; Suarez-Meade, P.; Chaichana, K.L. Bur Hole-Based Resections of Intrinsic Brain Tumors with Exoscopic Visualization. J. Neurol. Surg. A Cent. Eur. Neurosurg. 2021, 82, 105–111. [Google Scholar] [CrossRef]
- Piquer, J.; Llácer, J.L.; Rovira, V.; Riesgo, P.; Rodriguez, R.; Cremades, A. Fluorescence-guided surgery and biopsy in gliomas with an exoscope system. Biomed Res. Int. 2014, 2014, 207974. [Google Scholar] [CrossRef]
- Roethe, A.L.; Landgraf, P.; Schröder, T.; Misch, M.; Vajkoczy, P.; Picht, T. Monitor-based exoscopic 3D4k neurosurgical interventions: A two-phase prospective-randomized clinical evaluation of a novel hybrid device. Acta Neurochir. 2020, 162, 2949–2961. [Google Scholar] [CrossRef]



| Overall | EXOSCOPE | MICROSCOPE | p | |
|---|---|---|---|---|
| n (%) | 177 | 33 (18.6%) | 144 (81.4%) | |
| AGE (median I–III) | 65 (57,73) | 62 (51, 73) | 65 (58, 73) | 0.139 |
| SEX = M, n (%) | 111 (62.7) | 23 (69.7) | 88 (61.1) | 0.471 |
| Experience = LOW, n (%) | 47 (26.6) | 11 (33.3) | 36 (25.0) | 0.448 |
| LOBE, n (%) | 0.749 | |||
| Frontal | 53 (29.9) | 11 (33.3) | 42 (29.2) | |
| Temporal | 53 (29.9) | 9 (27.3) | 44 (30.6) | |
| Insular | 11 (6.2) | 4 (12.1) | 7 (4.9) | |
| Occipital | 24 (13.6) | 4 (12.1) | 20 (13.9) | |
| Parietal | 24 (13.6) | 4 (12.1) | 20 (13.9) | |
| Corpus callosum | 5 (2.8) | 0 (0.0) | 5 (3.5) | |
| Thalamus/pineal | 5 (2.8) | 1 (3.0) | 4 (2.8) | |
| Multifocal | 2 (1.1) | 0 (0.0) | 2 (1.4) | |
| SIDE, n (%) | 0.846 | |||
| Right | 92 (52.0) | 17 (51.5) | 75 (52.1) | |
| Midline | 3 (1.7) | 0 (0.0) | 3 (2.1) | |
| Multifocal | 4 (2.3) | 1 (3.0) | 3 (2.1) | |
| Left | 78 (44.1) | 15 (45.5) | 63 (43.8) | |
| IDH = Mutation, n (%) | 9 (5.2) | 4 (12.1) | 5 (3.6) | 0.120 |
| MGMT = Methylation, n (%) | 76 (44.4) | 10 (30.3) | 66 (47.8) | 0.104 |
| GTR, n (%) | 0.148 | |||
| GTR | 122 (68.9) | 23 (69.7) | 99 (68.8) | |
| PR | 41 (23.2) | 5 (15.2) | 36 (25.0) | |
| STR | 14 (7.9) | 5 (15.2) | 9 (6.2) | |
| KPS ≥ 70, n (%) | 116 (67.8) | 26 (83.9) | 90 (64.3) | 0.057 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Cristofori, A.; Graziano, F.; Rui, C.B.; Rebora, P.; Di Caro, D.; Chiarello, G.; Stefanoni, G.; Julita, C.; Florio, S.; Ferlito, D.; et al. Exoscopic Microsurgery: A Change of Paradigm in Brain Tumor Surgery? Comparison with Standard Operative Microscope. Brain Sci. 2023, 13, 1035. https://doi.org/10.3390/brainsci13071035
Di Cristofori A, Graziano F, Rui CB, Rebora P, Di Caro D, Chiarello G, Stefanoni G, Julita C, Florio S, Ferlito D, et al. Exoscopic Microsurgery: A Change of Paradigm in Brain Tumor Surgery? Comparison with Standard Operative Microscope. Brain Sciences. 2023; 13(7):1035. https://doi.org/10.3390/brainsci13071035
Chicago/Turabian StyleDi Cristofori, Andrea, Francesca Graziano, Chiara Benedetta Rui, Paola Rebora, Diego Di Caro, Gaia Chiarello, Giovanni Stefanoni, Chiara Julita, Santa Florio, Davide Ferlito, and et al. 2023. "Exoscopic Microsurgery: A Change of Paradigm in Brain Tumor Surgery? Comparison with Standard Operative Microscope" Brain Sciences 13, no. 7: 1035. https://doi.org/10.3390/brainsci13071035
APA StyleDi Cristofori, A., Graziano, F., Rui, C. B., Rebora, P., Di Caro, D., Chiarello, G., Stefanoni, G., Julita, C., Florio, S., Ferlito, D., Basso, G., Citerio, G., Remida, P., Carrabba, G., & Giussani, C. (2023). Exoscopic Microsurgery: A Change of Paradigm in Brain Tumor Surgery? Comparison with Standard Operative Microscope. Brain Sciences, 13(7), 1035. https://doi.org/10.3390/brainsci13071035