Regulatory Clearance and Approval of Therapeutic Protocols of Transcranial Magnetic Stimulation for Psychiatric Disorders
Abstract
1. What Is Transcranial Magnetic Stimulation and Repetitive Transcranial Magnetic Stimulation?
2. How Is Transcranial Magnetic Stimulation Used?
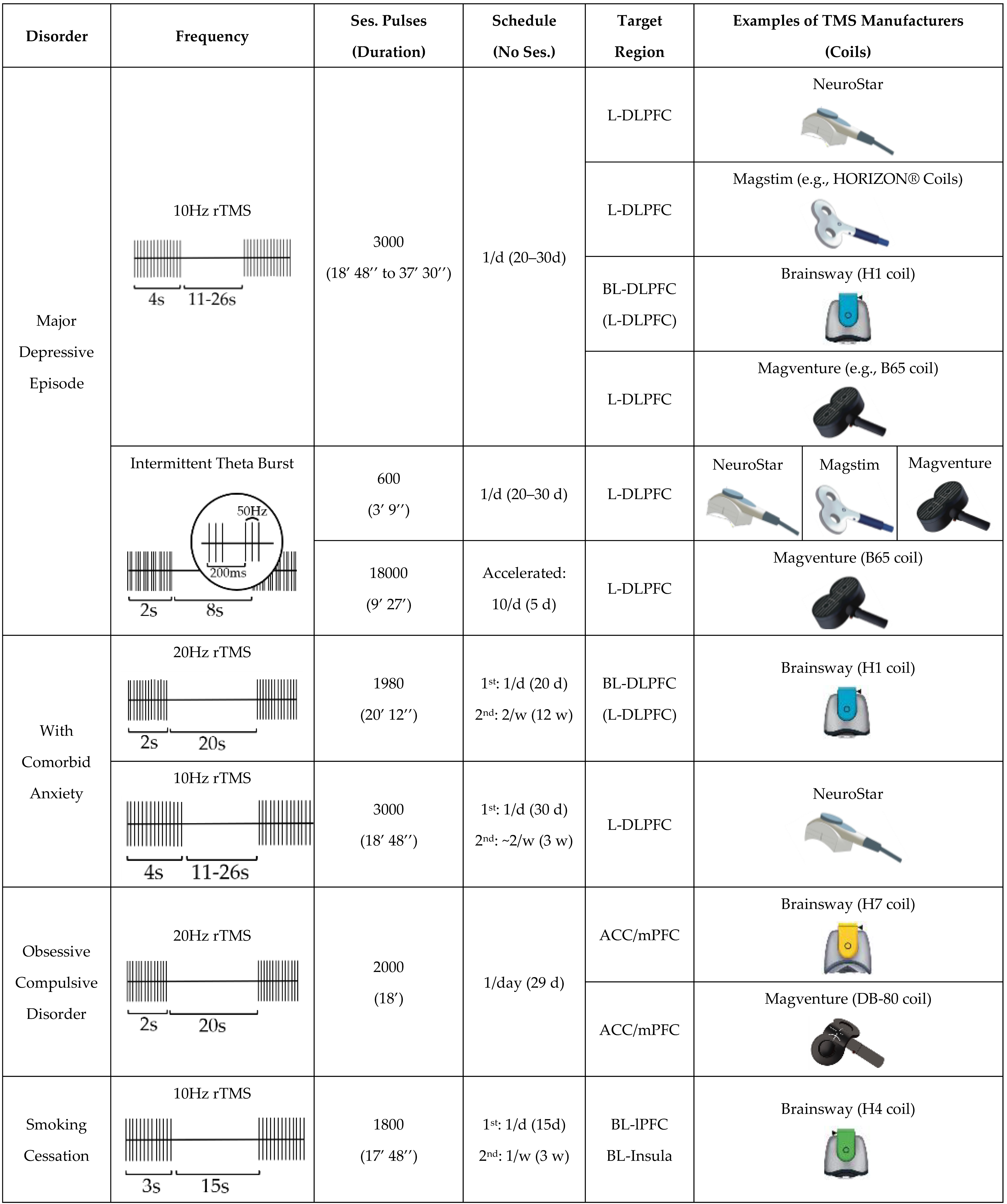
3. Transcranial Magnetic Stimulation Therapeutic Protocols for Psychiatric Disorders
3.1. TMS Protocols for Major Depressive Disorder (with/without Comorbid Anxiety)
3.2. TMS Protocols for Obsessive Compulsive Disorder
3.3. TMS Protocols for Other Psychiatric Disorders/Conditions
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Polanía, R.; Nitsche, M.A.; Ruff, C.C. Studying and modifying brain function with non-invasive brain stimulation. Nat. Neurosci. 2018, 21, 174–187. [Google Scholar] [CrossRef]
- Citri, A.; Malenka, R.C. Synaptic plasticity: Multiple forms, functions, and mechanisms. Neuropsychopharmacology 2008, 33, 18–41. [Google Scholar] [CrossRef]
- Lefaucheur, J.-P. Transcranial magnetic stimulation. Handb. Clin. Neurol. 2019, 160, 559–580. [Google Scholar]
- Turi, Z.; Normann, C.; Domschke, K.; Vlachos, A. Transcranial Magnetic Stimulation in Psychiatry: Is There a Need for Electric Field Standardization? Front. Hum. Neurosci. 2021, 15, 639640. [Google Scholar] [CrossRef] [PubMed]
- Valero-Cabré, A.; Amengual, J.L.; Stengel, C.; Pascual-Leone, A.; Coubard, O.A. Transcranial magnetic stimulation in basic and clinical neuroscience: A comprehensive review of fundamental principles and novel insights. Neurosci. Biobehav. Rev. 2017, 83, 381–404. [Google Scholar] [CrossRef] [PubMed]
- Roth, Y.; Amir, A.; Levkovitz, Y.; Zangen, A. Three-dimensional distribution of the electric field induced in the brain by transcranial magnetic stimulation using figure-8 and deep H-coils. J. Clin. Neurophysiol. 2007, 24, 31–38. [Google Scholar] [CrossRef]
- Lu, M.; Ueno, S. Comparison of the induced fields using different coil configurations during deep transcranial magnetic stimulation. PLoS ONE 2017, 12, e0178422. [Google Scholar] [CrossRef]
- Klomjai, W.; Katz, R.; Lackmy-Vallée, A. Basic principles of transcranial magnetic stimulation (TMS) and repetitive TMS (rTMS). Ann. Phys. Rehabil. Med. 2015, 58, 208–213. [Google Scholar] [CrossRef] [PubMed]
- Turi, Z.; Lenz, M.; Paulus, W.; Mittner, M.; Vlachos, A. Selecting stimulation intensity in repetitive transcranial magnetic stimulation studies: A systematic review between 1991 and 2020. Eur. J. Neurosci. 2021, 53, 3404–3415. [Google Scholar] [CrossRef]
- Cotovio, G.; Oliveira-Maia, A.J.; Paul, C.; Faro Viana, F.; Rodrigues da Silva, D.; Seybert, C.; Stern, A.P.; Pascual-Leone, A.; Press, D.Z. Day-to-day variability in motor threshold during rTMS treatment for depression: Clinical implications. Brain Stimul. 2021, 14, 1118–1125. [Google Scholar] [CrossRef]
- Badawy, R.A.; Loetscher, T.; Macdonell, R.A.; Brodtmann, A. Cortical excitability and neurology: Insights into the pathophysiology. Funct. Neurol. 2012, 27, 131–145. [Google Scholar]
- Gottmann, K.; Mittmann, T.; Lessmann, V. BDNF signaling in the formation, maturation and plasticity of glutamatergic and GABAergic synapses. Exp. Brain Res. 2009, 199, 203–234. [Google Scholar] [CrossRef] [PubMed]
- Reis, H.J.; Guatimosim, C.; Paquet, M.; Santos, M.; Ribeiro, F.M.; Kummer, A.; Schenatto, G.; Salgado, J.V.; Vieira, L.B.; Teixeira, A.L. Neuro-transmitters in the central nervous system & their implication in learning and memory processes. Curr. Med. Chem. 2009, 16, 796–840. [Google Scholar] [PubMed]
- Suppa, A.; Asci, F.; Guerra, A. Transcranial magnetic stimulation as a tool to induce and explore plasticity in humans. Handb. Clin. Neurol. 2022, 184, 73–89. [Google Scholar]
- Barker, A.T.; Jalinous, R.; Freeston, I.L. Non-Invasive Magnetic Stimulation of Human Motor Cortex. Lancet 1985, 1, 1106–1107. [Google Scholar] [CrossRef]
- Vucic, S.; Chen, K.-H.S.; Kiernan, M.C.; Hallett, M.; Benninger, D.; Di Lazzaro, V.; Rossini, P.M.; Benussi, A.; Berardelli, A.; Currà, A. Clinical diagnostic utility of transcranial magnetic stimulation in neurological disorders. Updated report of an IFCN committee. Clin. Neurophysiol. 2023, 150, 131–175. [Google Scholar] [CrossRef]
- Tanaka, M.; Szabó, Á.; Vécsei, L. Preclinical modeling in depression and anxiety: Current challenges and future research directions. Adv. Clin. Exp. Med. 2023, 32, 505–509. [Google Scholar] [CrossRef]
- Tajti, J.; Szok, D.; Csáti, A.; Szabó, Á.; Tanaka, M.; Vécsei, L. Exploring Novel Therapeutic Targets in the Common Pathogenic Factors in Migraine and Neuropathic Pain. Int. J. Mol. Sci. 2023, 24, 4114. [Google Scholar] [CrossRef] [PubMed]
- Battaglia, S.; Di Fazio, C.; Vicario, C.M.; Avenanti, A. Neuropharmacological modulation of N-methyl-D-aspartate, noradrenaline and endocannabinoid receptors in fear extinction learning: Synaptic transmission and plasticity. Int. J. Mol. Sci. 2023, 24, 5926. [Google Scholar] [CrossRef]
- Battaglia, S.; Nazzi, C.; Thayer, J. Fear-induced bradycardia in mental disorders: Foundations, current advances, future perspectives. Neurosci. Biobehav. Rev. 2023, 149, 105163. [Google Scholar] [CrossRef]
- Post, A.; Keck, M.E. Transcranial magnetic stimulation as a therapeutic tool in psychiatry: What do we know about the neurobiological mechanisms? J. Psychiatr. Res. 2001, 35, 193–215. [Google Scholar] [CrossRef]
- Marder, K.G.; Barbour, T.; Ferber, S.; Idowu, O.; Itzkoff, A. Psychiatric Applications of Repetitive Transcranial Magnetic Stimulation. Focus 2022, 20, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Wieczorek, T.; Kobyłko, A.; Stramecki, F.; Fila-Witecka, K.; Beszłej, J.A.; Jakubczyk, M.; Piotrowski, P.; Senczyszyn, A.; Siwicki, D.; Szcześniak, D. Transcranial magnetic stimulation (TMS) in treatment of psychiatric disorders-review of current studies. Psychiatr. Pol. 2021, 55, 565–583. [Google Scholar] [CrossRef] [PubMed]
- Cole, E.J.; Phillips, A.L.; Bentzley, B.S.; Stimpson, K.H.; Nejad, R.; Barmak, F.; Veerapal, C.; Khan, N.; Cherian, K.; Felber, E. Stanford Neuromodulation Therapy (SNT): A double-blind randomized controlled trial. Am. J. Psychiatry 2021, 179, 132–141. [Google Scholar] [CrossRef] [PubMed]
- Senova, S.; Cotovio, G.; Pascual-Leone, A.; Oliveira-Maia, A.J. Durability of antidepressant response to repetitive transcranial magnetic stimulation: Systematic review and meta-analysis. Brain Stimul. 2019, 12, 119–128. [Google Scholar] [CrossRef]
- Kar, S.K. Predictors of response to repetitive transcranial magnetic stimulation in depression: A review of recent updates. Clin. Psychopharmacol. Neurosci. 2019, 17, 25–33. [Google Scholar] [CrossRef]
- Valiengo, L.; Maia, A.; Cotovio, G.; Gordon, P.C.; Brunoni, A.R.; Forlenza, O.V.; Oliveira-Maia, A.J. Repetitive Transcranial Magnetic Stimulation for Major Depressive Disorder in Older Adults: Systematic Review and Meta-analysis. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2022, 77, 851–860. [Google Scholar] [CrossRef]
- Siddiqi, S.H.; Chockalingam, R.; Cloninger, C.R.; Lenze, E.J.; Cristancho, P. Use of the temperament and character inventory to predict response to repetitive transcranial magnetic stimulation for major depression. J. Psychiatr. Pract. 2016, 22, 193–202. [Google Scholar] [CrossRef][Green Version]
- Oliveira-Maia, A.J.; Press, D.; Pascual-Leone, A. Modulation of motor cortex excitability predicts antidepressant response to prefrontal cortex repetitive transcranial magnetic stimulation. Brain Stimul. 2017, 10, 787–794. [Google Scholar] [CrossRef]
- Cotovio, G.; Rodrigues da Silva, D.; Real Lage, E.; Seybert, C.; Oliveira-Maia, A.J. Hemispheric asymmetry of motor cortex excitability in mood disorders—Evidence from a systematic review and meta-analysis. Clin. Neurophysiol. 2022, 137, 25–37. [Google Scholar] [CrossRef]
- Drysdale, A.T.; Grosenick, L.; Downar, J.; Dunlop, K.; Mansouri, F.; Meng, Y.; Fetcho, R.N.; Zebley, B.; Oathes, D.J.; Etkin, A. Resting-state connectivity biomarkers define neurophysiological subtypes of depression. Nat. Med. 2017, 23, 28–38. [Google Scholar] [CrossRef]
- Acevedo, N.; Bosanac, P.; Pikoos, T.; Rossell, S.; Castle, D. Therapeutic neurostimulation in obsessive-compulsive and related disorders: A systematic review. Brain Sci. 2021, 11, 948. [Google Scholar] [CrossRef] [PubMed]
- Cash, R.F.; Weigand, A.; Zalesky, A.; Siddiqi, S.H.; Downar, J.; Fitzgerald, P.B.; Fox, M.D. Using brain imaging to improve spatial targeting of transcranial magnetic stimulation for depression. Biol. Psychiatry 2021, 90, 689–700. [Google Scholar] [CrossRef] [PubMed]
- Cash, R.F.; Cocchi, L.; Lv, J.; Wu, Y.; Fitzgerald, P.B.; Zalesky, A. Personalized connectivity-guided DLPFC-TMS for depression: Advancing computational feasibility, precision and reproducibility. Hum. Brain Mapp. 2021, 42, 4155–4172. [Google Scholar] [CrossRef] [PubMed]
- Maia, A.; Almeida, S.; Cotovio, G.; Rodrigues da Silva, D.; Viana, F.F.; Grácio, J.; Oliveira-Maia, A.J. Symptom provocation for treatment of obsessive-compulsive disorder using transcranial magnetic stimulation: A step-by-step guide for professional training. Front. Psychiatry 2022, 13, 924370. [Google Scholar] [CrossRef]
- Seybert, C.; Cotovio, G.; Grácio, J.; Oliveira-Maia, A.J. Future Perspectives from a Case Report of Transcranial Magnetic Stimulation, Cognitive Behavioral Therapy, and Psychopharmacological Treatment for Post-traumatic Stress Disorder. Front. Psychol. 2021, 12, 728130. [Google Scholar] [CrossRef]
- Ontario, H.Q. Repetitive transcranial magnetic stimulation for treatment-resistant depression: A systematic review and meta-analysis of randomized controlled trials. Ont. Health Technol. Assess. Ser. 2016, 16, 1–66. [Google Scholar]
- Rossi, S.; Antal, A.; Bestmann, S.; Bikson, M.; Brewer, C.; Brockmöller, J.; Carpenter, L.L.; Cincotta, M.; Chen, R.; Daskalakis, J.D.; et al. Safety and recommendations for TMS use in healthy subjects and patient populations, with updates on training, ethical and regulatory issues: Expert Guidelines. Clin. Neurophysiol. 2021, 132, 269–306. [Google Scholar] [CrossRef]
- Stultz, D.J.; Osburn, S.; Burns, T.; Pawlowska-Wajswol, S.; Walton, R. Transcranial Magnetic Stimulation (TMS) Safety with Respect to Seizures: A Literature Review. Neuropsychiatr. Dis. Treat. 2020, 16, 2989–3000. [Google Scholar] [CrossRef] [PubMed]
- Lefaucheur, J.P.; Aleman, A.; Baeken, C.; Benninger, D.H.; Brunelin, J.; Di Lazzaro, V.; Filipović, S.R.; Grefkes, C.; Hasan, A.; Hummel, F.C.; et al. Evidence-based guidelines on the therapeutic use of repetitive transcranial magnetic stimulation (rTMS): An update (2014–2018). Clin. Neurophysiol. 2020, 131, 474–528. [Google Scholar] [CrossRef]
- George, M.S.; Wassermann, E.M.; Williams, W.A.; Callahan, A.; Ketter, T.A.; Basser, P.; Hallett, M.; Post, R.M. Daily repetitive transcranial magnetic stimulation (rTMS) improves mood in depression. Neuroreport 1995, 6, 1853–1856. [Google Scholar] [CrossRef]
- O’Reardon, J.P.; Solvason, H.B.; Janicak, P.G.; Sampson, S.; Isenberg, K.E.; Nahas, Z.; McDonald, W.M.; Avery, D.; Fitzgerald, P.B.; Loo, C.; et al. Efficacy and safety of transcranial magnetic stimulation in the acute treatment of major depression: A multisite randomized controlled trial. Biol. Psychiatry 2007, 62, 1208–1216. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.J.; Hoy, K.E.; Daskalakis, Z.J.; Fitzgerald, P.B. Repetitive transcranial magnetic stimulation for treatment resistant depression: Re-establishing connections. Clin. Neurophysiol. 2016, 127, 3394–3405. [Google Scholar] [CrossRef]
- Salehinejad, M.A.; Ghanavai, E.; Rostami, R.; Nejati, V. Cognitive control dysfunction in emotion dysregulation and psychopathology of major depression (MD): Evidence from transcranial brain stimulation of the dorsolateral prefrontal cortex (DLPFC). J. Affect. Disord. 2017, 210, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Parazzini, M.; Fiocchi, S.; Chiaramello, E.; Roth, Y.; Zangen, A.; Ravazzani, P. Electric field estimation of deep transcranial magnetic stimulation clinically used for the treatment of neuropsychiatric disorders in anatomical head models. Med. Eng. Phys. 2017, 43, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Levkovitz, Y.; Isserles, M.; Padberg, F.; Lisanby, S.H.; Bystritsky, A.; Xia, G.; Tendler, A.; Daskalakis, Z.J.; Winston, J.L.; Dannon, P. Efficacy and safety of deep transcranial magnetic stimulation for major depression: A prospective multicenter randomized controlled trial. World Psychiatry 2015, 14, 64–73. [Google Scholar] [CrossRef]
- Kaster, T.S.; Daskalakis, Z.J.; Noda, Y.; Knyahnytska, Y.; Downar, J.; Rajji, T.K.; Levkovitz, Y.; Zangen, A.; Butters, M.A.; Mulsant, B.H. Efficacy, tolerability, and cognitive effects of deep transcranial magnetic stimulation for late-life depression: A prospective randomized controlled trial. Neuropsychopharmacology 2018, 43, 2231–2238. [Google Scholar] [CrossRef] [PubMed]
- Tavares, D.F.; Myczkowski, M.L.; Alberto, R.L.; Valiengo, L.; Rios, R.M.; Gordon, P.; de Sampaio-Junior, B.; Klein, I.; Mansur, C.G.; Marcolin, M.A. Treatment of bipolar depression with deep TMS: Results from a double-blind, randomized, parallel group, sham-controlled clinical trial. Neuropsychopharmacology 2017, 42, 2593–2601. [Google Scholar] [CrossRef]
- Blumberger, D.M.; Vila-Rodriguez, F.; Thorpe, K.E.; Feffer, K.; Noda, Y.; Giacobbe, P.; Knyahnytska, Y.; Kennedy, S.H.; Lam, R.W.; Daskalakis, Z.J.; et al. Effectiveness of theta burst versus high-frequency repetitive transcranial magnetic stimulation in patients with depression (THREE-D): A randomised non-inferiority trial. Lancet 2018, 391, 1683–1692. [Google Scholar] [CrossRef]
- Mutz, J.; Vipulananthan, V.; Carter, B.; Hurlemann, R.; Fu, C.H.; Young, A.H. Comparative efficacy and acceptability of non-surgical brain stimulation for the acute treatment of major depressive episodes in adults: Systematic review and network meta-analysis. BMJ 2019, 364, l1079. [Google Scholar] [CrossRef]
- Sehatzadeh, S.; Daskalakis, Z.J.; Yap, B.; Tu, H.-A.; Palimaka, S.; Bowen, J.M.; O’Reilly, D.J. Unilateral and bilateral repetitive transcranial magnetic stimulation for treatment-resistant depression: A meta-analysis of randomized controlled trials over 2 decades. J. Psychiatry Neurosci. 2019, 44, 151–163. [Google Scholar] [CrossRef]
- Li, H.; Cui, L.; Li, J.; Liu, Y.; Chen, Y. Comparative efficacy and acceptability of neuromodulation procedures in the treatment of treatment-resistant depression: A network meta-analysis of randomized controlled trials. J. Affect. Disord. 2021, 287, 115–124. [Google Scholar] [CrossRef]
- Thompson, L. Treating major depression and comorbid disorders with transcranial magnetic stimulation. J. Affect. Disord. 2020, 276, 453–460. [Google Scholar] [CrossRef]
- Filipčić, I.; Filipčić, I.Š.; Milovac, Ž.; Sučić, S.; Gajšak, T.; Ivezić, E.; Bašić, S.; Bajić, Ž.; Heilig, M. Efficacy of repetitive transcranial magnetic stimulation using a figure-8-coil or an H1-Coil in treatment of major depressive disorder; A randomized clinical trial. J. Psychiatr. Res. 2019, 114, 113–119. [Google Scholar] [CrossRef]
- Clarke, E.; Clarke, P.; Gill, S.; Paterson, T.; Hahn, L.; Galletly, C. Efficacy of repetitive transcranial magnetic stimulation in the treatment of depression with comorbid anxiety disorders. J. Affect. Disord. 2019, 252, 435–439. [Google Scholar] [CrossRef] [PubMed]
- Cotovio, G.; Boes, A.D.; Press, D.Z.; Oliveira-Maia, A.J.; Pascual-Leone, A. In Older Adults the Antidepressant Effect of Repetitive Transcranial Magnetic Stimulation Is Similar but Occurs Later Than in Younger Adults. Front. Aging Neurosci. 2022, 14, 919734. [Google Scholar] [CrossRef]
- Cole, J.; Bright, K.; Gagnon, L.; McGirr, A. A systematic review of the safety and effectiveness of repetitive transcranial magnetic stimulation in the treatment of peripartum depression. J. Psychiatr. Res. 2019, 115, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.D.; Hieronymus, F.; Lorentzen, R.; McGirr, A.; Ostergaard, S.D. The efficacy of repetitive transcranial magnetic stimulation (rTMS) for bipolar depression: A systematic review and meta-analysis. J. Affect. Disord. 2021, 279, 250–255. [Google Scholar] [CrossRef] [PubMed]
- Lefaucheur, J.P.; André-Obadia, N.; Antal, A.; Ayache, S.S.; Baeken, C.; Benninger, D.H.; Cantello, R.M.; Cincotta, M.; de Carvalho, M.; De Ridder, D.; et al. Evidencebased guidelines on the therapeutic use of repetitive transcranial magnetic stimulation (rTMS). Clin. Neurophysiol. 2014, 125, 2150–2206. [Google Scholar] [CrossRef] [PubMed]
- Stein, D.J.; Costa, D.L.C.; Lochner, C.; Miguel, E.C.; Reddy, Y.C.J.; Shavitt, R.G.; van den Heuvel, O.A.; Simpson, H.B. Obsessive-compulsive disorder. Nat. Rev. Dis. Prim. 2019, 5, 52. [Google Scholar] [CrossRef] [PubMed]
- Maia, T.V.; Cooney, R.E.; Peterson, B.S. The neural bases of obsessive-compulsive disorder in children and adults. Dev. Psychopathol. 2008, 20, 1251–1283. [Google Scholar] [CrossRef]
- Boedhoe, P.S.; Schmaal, L.; Abe, Y.; Ameis, S.H.; Arnold, P.D.; Batistuzzo, M.C.; Benedetti, F.; Beucke, J.C.; Bollettini, I.; Bose, A. Distinct subcortical volume alterations in pediatric and adult OCD: A worldwide meta-and mega-analysis. Am. J. Psychiatry 2017, 174, 60–69. [Google Scholar] [CrossRef]
- Rotge, J.-Y.; Guehl, D.; Dilharreguy, B.; Tignol, J.; Bioulac, B.; Allard, M.; Burbaud, P.; Aouizerate, B. Meta-analysis of brain volume changes in obsessive-compulsive disorder. Biol. Psychiatry 2009, 65, 75–83. [Google Scholar] [CrossRef]
- Menzies, L.; Chamberlain, S.R.; Laird, A.R.; Thelen, S.M.; Sahakian, B.J.; Bullmore, E.T. Integrating evidence from neuroimaging and neuropsychological studies of obsessive-compulsive disorder: The orbitofronto-striatal model revisited. Neurosci. Biobehav. Rev. 2008, 32, 525–549. [Google Scholar] [CrossRef] [PubMed]
- Friedlander, L.; Desrocher, M. Neuroimaging studies of obsessive–compulsive disorder in adults and children. Clin. Psychol. Rev. 2006, 26, 32–49. [Google Scholar] [CrossRef]
- Liang, K.; Li, H.; Bu, X.; Li, X.; Cao, L.; Liu, J.; Gao, Y.; Li, B.; Qiu, C.; Bao, W.; et al. Efficacy and tolerability of repetitive transcranial magnetic stimulation for the treatment of obsessive-compulsive disorder in adults: A systematic review and network meta-analysis. Transl. Psychiatry 2021, 11, 332. [Google Scholar] [CrossRef]
- Carmi, L.; Tendler, A.; Bystritsky, A.; Hollander, E.; Blumberger, D.M.; Daskalakis, J.; Ward, H.; Lapidus, K.; Goodman, W.; Casuto, L.; et al. Efficacy and Safety of Deep Transcranial Magnetic Stimulation for Obsessive-Compulsive Disorder: A Prospective Multicenter Randomized Double-Blind Placebo-Controlled Trial. Am. J. Psychiatry 2019, 176, 931–938. [Google Scholar] [CrossRef] [PubMed]
- Tendler, A.; Sisko, E.; Barnea-Ygael, N.; Zangen, A.; Storch, E.A. A Method to Provoke Obsessive Compulsive Symptoms for Basic Research and Clinical Interventions. Front. Psychiatry 2019, 10, 814. [Google Scholar] [CrossRef]
- Dinur-Klein, L.; Dannon, P.; Hadar, A.; Rosenberg, O.; Roth, Y.; Kotler, M.; Zangen, A. Smoking cessation induced by deep repetitive transcranial magnetic stimulation of the prefrontal and insular cortices: A prospective, randomized controlled trial. Biol. Psychiatry 2014, 76, 742–749. [Google Scholar] [CrossRef] [PubMed]
- Madeo, G.; Terraneo, A.; Cardullo, S.; Gómez Pérez, L.J.; Cellini, N.; Sarlo, M.; Bonci, A.; Gallimberti, L. Long-Term Outcome of Repetitive Transcranial Magnetic Stimulation in a Large Cohort of Patients with Cocaine-Use Disorder: An Observational Study. Front. Psychiatry 2020, 11, 158. [Google Scholar] [CrossRef]
- Terraneo, A.; Leggio, L.; Saladini, M.; Ermani, M.; Bonci, A.; Gallimberti, L. Transcranial magnetic stimulation of dorsolateral prefrontal cortex reduces cocaine use: A pilot study. Eur. Neuropsychopharmacol. 2016, 26, 37–44. [Google Scholar] [CrossRef]
- Isserles, M.; Shalev, A.Y.; Roth, Y.; Peri, T.; Kutz, I.; Zlotnick, E.; Zangen, A. Effectiveness of deep transcranial magnetic stimulation combined with a brief exposure procedure in post-traumatic stress disorder—A pilot study. Brain Stimul. 2013, 6, 377–383. [Google Scholar] [CrossRef] [PubMed]
- Ahmadizadeh, M.-J.; Rezaei, M. Unilateral right and bilateral dorsolateral prefrontal cortex transcranial magnetic stimulation in treatment post-traumatic stress disorder: A randomized controlled study. Brain Res. Bull. 2018, 140, 334–340. [Google Scholar] [CrossRef]
- Cirillo, P.; Gold, A.K.; Nardi, A.E.; Ornelas, A.C.; Nierenberg, A.A.; Camprodon, J.; Kinrys, G. Transcranial magnetic stimulation in anxiety and trauma-related disorders: A systematic review and meta-analysis. Brain Behav. 2019, 9, e01284. [Google Scholar] [CrossRef]
- Kozel, F.A.; Motes, M.A.; Didehbani, N.; DeLaRosa, B.; Bass, C.; Schraufnagel, C.D.; Jones, P.; Morgan, C.R.; Spence, J.S.; Kraut, M.A. Repetitive TMS to augment cognitive processing therapy in combat veterans of recent conflicts with PTSD: A randomized clinical trial. J. Affect. Disord. 2018, 229, 506–514. [Google Scholar] [CrossRef]
- Schiller, D.; Kanen, J.W.; LeDoux, J.E.; Monfils, M.-H.; Phelps, E.A. Extinction during reconsolidation of threat memory diminishes prefrontal cortex involvement. Proc. Natl. Acad. Sci. USA 2013, 110, 20040–20045. [Google Scholar] [CrossRef]
- Marzouk, T.; Winkelbeiner, S.; Azizi, H.; Malhotra, A.K.; Homan, P. Transcranial Magnetic Stimulation for Positive Symptoms in Schizophrenia: A Systematic Review. Neuropsychobiology 2020, 79, 384–396. [Google Scholar] [CrossRef]
- Hoffman, R.E.; Boutros, N.N.; Berman, R.M.; Roessler, E.; Belger, A.; Krystal, J.H.; Charney, D.S. Transcranial magnetic stimulation of left temporoparietal cortex in three patients reporting hallucinated “voices”. Biol. Psychiatry 1999, 46, 130–132. [Google Scholar] [CrossRef]
- Aleman, A.; Lincoln, T.M.; Bruggeman, R.; Melle, I.; Arends, J.; Arango, C.; Knegtering, H. Treatment of negative symptoms: Where do we stand, and where do we go? Schizophr. Res. 2017, 186, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Shukla, D.K.; Chiappelli, J.J.; Sampath, H.; Kochunov, P.; Hare, S.M.; Wisner, K.; Rowland, L.M.; Hong, L.E. Aberrant Frontostriatal Connectivity in Negative Symptoms of Schizophrenia. Schizophr. Bull. 2019, 45, 1051–1059. [Google Scholar] [CrossRef] [PubMed]
- Tseng, P.T.; Zeng, B.S.; Hung, C.M.; Liang, C.S.; Stubbs, B.; Carvalho, A.F.; Brunoni, A.R.; Su, K.P.; Tu, Y.K.; Wu, Y.C.; et al. Assessment of Noninvasive Brain Stimulation Interventions for Negative Symptoms of Schizophrenia: A Systematic Review and Network Meta-analysis. JAMA Psychiatry 2022, 79, 770–779. [Google Scholar] [CrossRef]
- Eldaief, M.C.; Dickerson, B.C.; Camprodon, J.A. Transcranial magnetic stimulation for the neurological patient: Scientific principles and applications. Semin. Neurol. 2022, 42, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Somaa, F.A.; de Graaf, T.A.; Sack, A.T. Transcranial magnetic stimulation in the treatment of neurological diseases. Front. Neurol. 2022, 13, 793253. [Google Scholar] [CrossRef] [PubMed]
- Solvason, H.B.; Husain, M.; Fitzgerald, P.B.; Rosenquist, P.; McCall, W.V.; Kimball, J.; Gilmer, W.; Demitrack, M.A.; Lisanby, S.H. Improvement in quality of life with left prefrontal transcranial magnetic stimulation in patients with pharmacoresistant major depression: Acute and six month outcomes. Brain Stimul. 2014, 7, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Janicak, P.G.; Dunner, D.L.; Aaronson, S.T.; Carpenter, L.L.; Boyadjis, T.A.; Brock, D.G.; Cook, I.A.; Lanocha, K.; Solvason, H.B.; Bonneh-Barkay, D. Transcranial magnetic stimulation (TMS) for major depression: A multisite, naturalistic, observational study of quality of life outcome measures in clinical practice. CNS Spectr. 2013, 18, 322–332. [Google Scholar] [CrossRef] [PubMed]
- Dumas, R.; Richieri, R.; Guedj, E.; Auquier, P.; Lancon, C.; Boyer, L. Improvement of health-related quality of life in depression after transcranial magnetic stimulation in a naturalistic trial is associated with decreased perfusion in precuneus. Health Qual. Life Outcomes 2012, 10, 87. [Google Scholar] [CrossRef] [PubMed]
- Gregory, S.T.; Goodman, W.K.; Kay, B.; Riemann, B.; Storch, E.A. Cost-effectiveness analysis of deep transcranial magnetic stimulation relative to evidence-based strategies for treatment-refractory obsessive-compulsive disorder. J. Psychiatr. Res. 2022, 146, 50–54. [Google Scholar] [CrossRef]
- Pohar, R.; Farrah, K. Repetitive Transcranial Magnetic Stimulation for Patients with Depression: A Review of Clinical Effectiveness, Cost-Effectiveness and Guidelines—An Update; Canadian Agency for Drugs and Technologies in Health: Ottawa, ON, Canada, 2019. [Google Scholar]
- Matsuda, Y.; Yamazaki, R.; Shigeta, M.; Kito, S. Transcranial magnetic stimulation modalities for psychiatric disorders: Publication trends from 1985 to 2019. Neuropsychopharmacol. Rep. 2021, 41, 538–543. [Google Scholar] [CrossRef]
- Ridding, M.C.; Rothwell, J.C. Is there a future for therapeutic use of transcranial magnetic stimulation? Nat. Rev. Neurosci. 2007, 8, 559–567. [Google Scholar] [CrossRef]
- Cotovio, G.; Oliveira-Maia, A.J. Functional neuroanatomy of mania. Transl. Psychiatry 2022, 12, 29. [Google Scholar] [CrossRef]
 |
| Major Depressive Episode | Definite antidepressant effect of HF-TMS of the left DLPFC (Level A) |
| Probable antidepressant effect of LF-TMS of the right DLPFC (Level B) and probably no differential antidepressant effect between right LF-TMS and left HF-TMS (Level B) | |
| Definite antidepressant effect of rTMS of the DLPFC in unipolar depression (Level A), but no recommendation for bipolar depression | |
| Antidepressant effect of rTMS of the DLPFC is probably additive to the efficacy of antidepressant drugs (Level B) and possibly potentiating (Level C) | |
| PTSD | Possible effect of HF-TMS of the right DLPFC (Level C) |
| Auditory hallucinations | Possible effect of LF-TMS of the left TPC (Level C) |
| Negative symptoms of schizophrenia | Probable effect of HF-TMS of the left DLPFC (Level B) |
| Addiction and craving | Possible effect of HF-rTMS of the left DLPFC on cigarette craving and consumption (Level C) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cotovio, G.; Ventura, F.; Rodrigues da Silva, D.; Pereira, P.; Oliveira-Maia, A.J. Regulatory Clearance and Approval of Therapeutic Protocols of Transcranial Magnetic Stimulation for Psychiatric Disorders. Brain Sci. 2023, 13, 1029. https://doi.org/10.3390/brainsci13071029
Cotovio G, Ventura F, Rodrigues da Silva D, Pereira P, Oliveira-Maia AJ. Regulatory Clearance and Approval of Therapeutic Protocols of Transcranial Magnetic Stimulation for Psychiatric Disorders. Brain Sciences. 2023; 13(7):1029. https://doi.org/10.3390/brainsci13071029
Chicago/Turabian StyleCotovio, Gonçalo, Fabiana Ventura, Daniel Rodrigues da Silva, Patrícia Pereira, and Albino J. Oliveira-Maia. 2023. "Regulatory Clearance and Approval of Therapeutic Protocols of Transcranial Magnetic Stimulation for Psychiatric Disorders" Brain Sciences 13, no. 7: 1029. https://doi.org/10.3390/brainsci13071029
APA StyleCotovio, G., Ventura, F., Rodrigues da Silva, D., Pereira, P., & Oliveira-Maia, A. J. (2023). Regulatory Clearance and Approval of Therapeutic Protocols of Transcranial Magnetic Stimulation for Psychiatric Disorders. Brain Sciences, 13(7), 1029. https://doi.org/10.3390/brainsci13071029







