Psychomotor Performance after 30 h of Sleep Deprivation Combined with Exercise
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
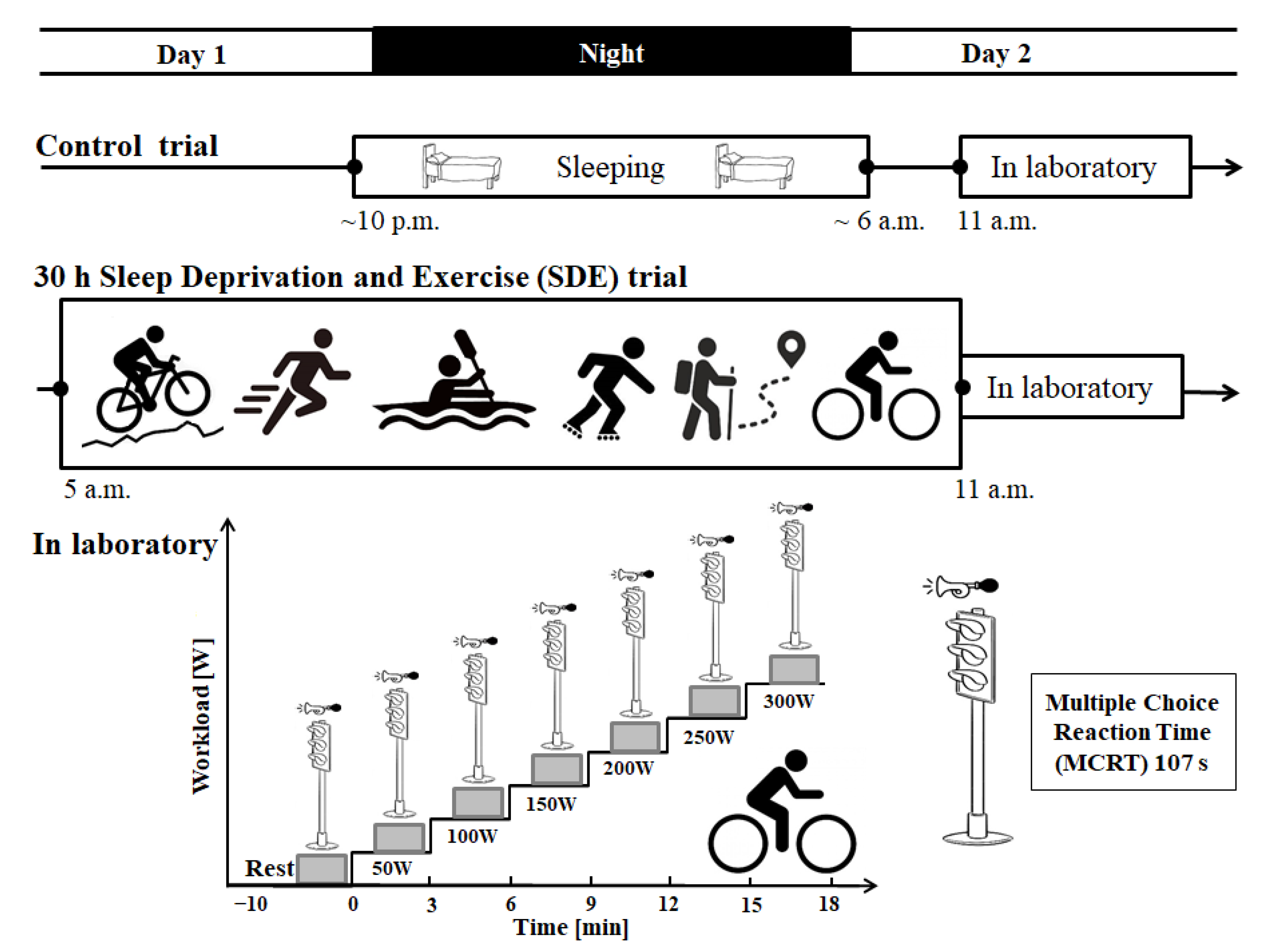
2.2. Procedure
2.3. Psychomotor Performance
2.4. Statistical Analysis
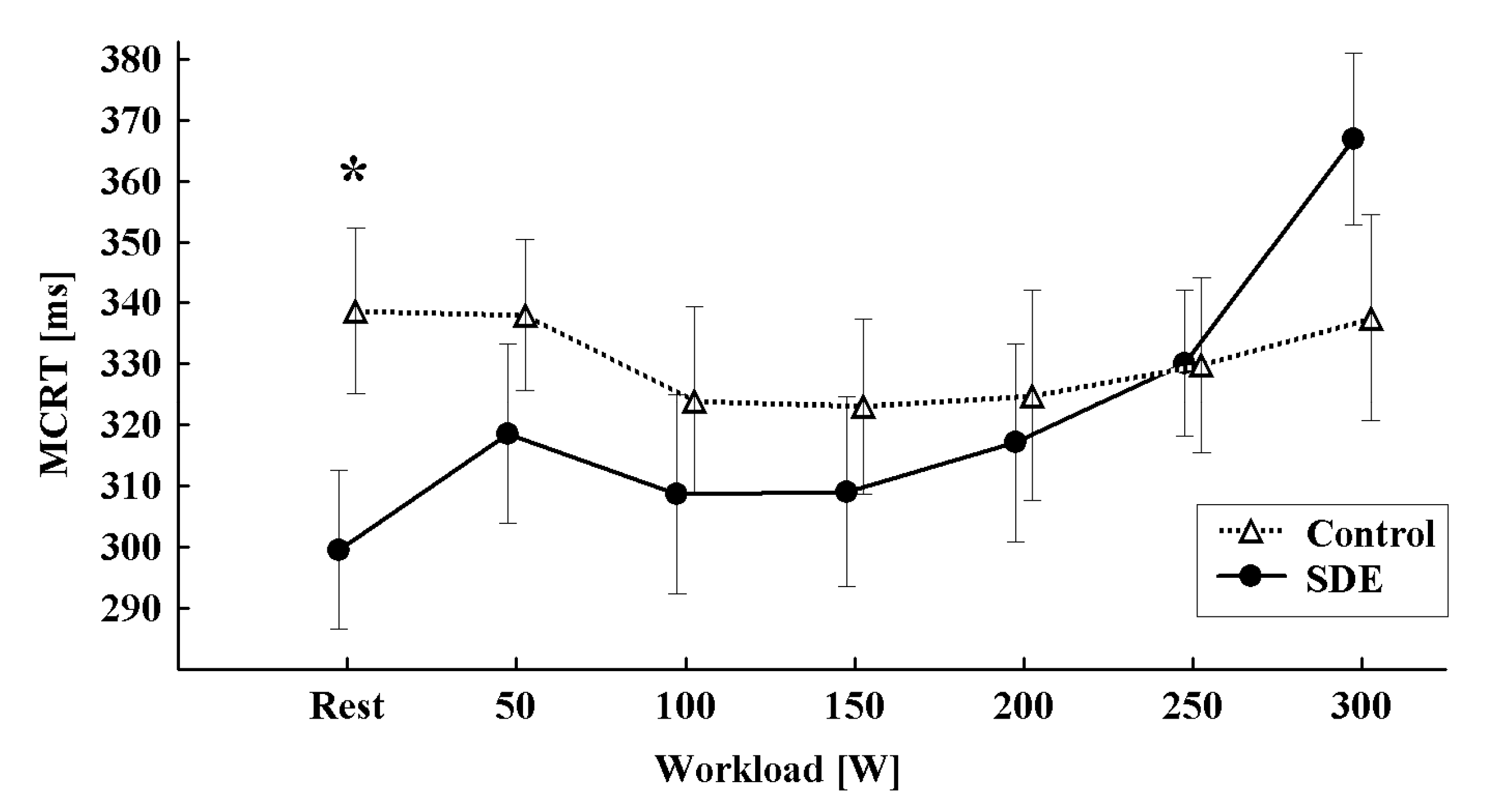
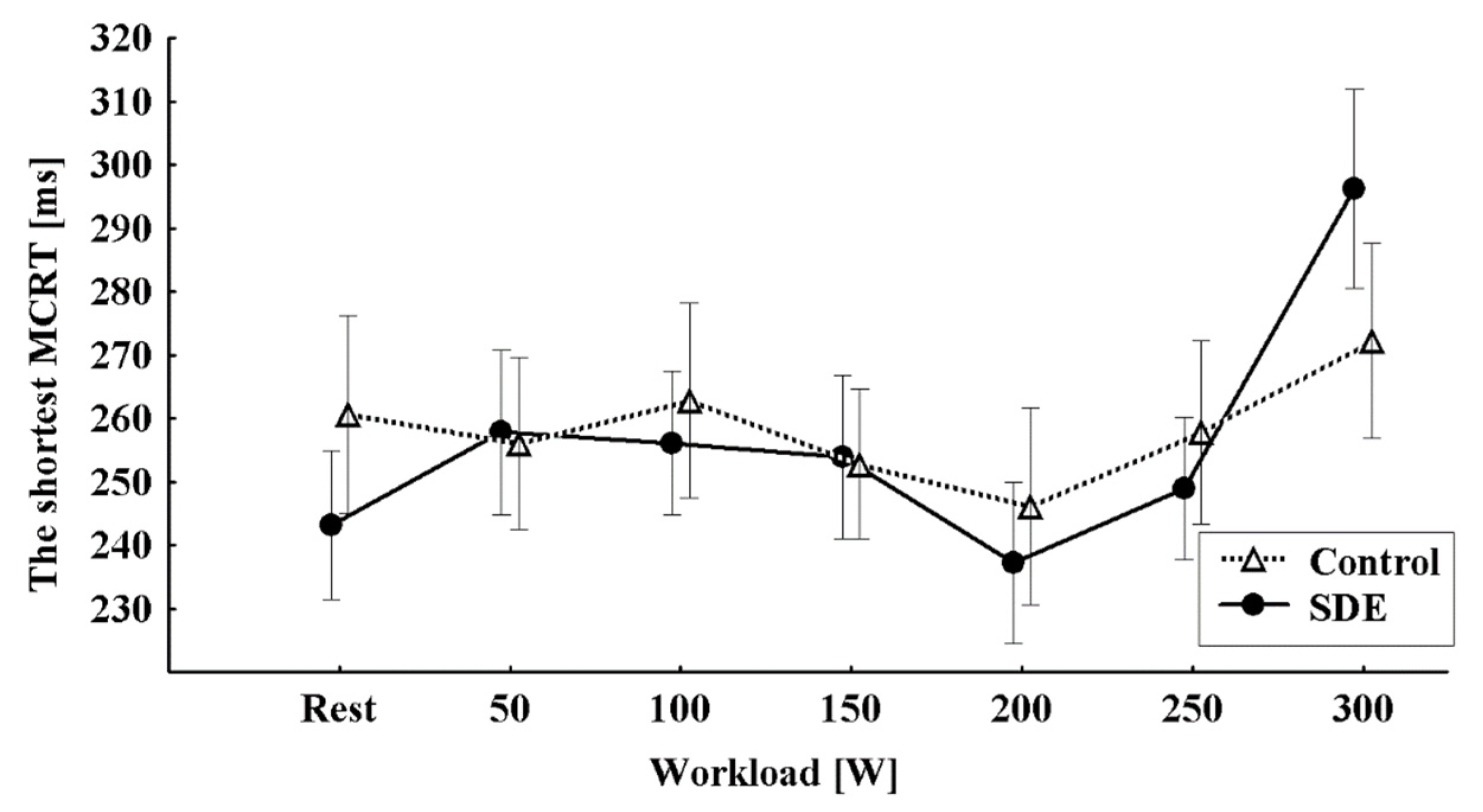
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Byun, K.; Hyodo, K.; Suwabe, K.; Ochi, G.; Sakairi, Y.; Kato, M.; Dan, I.; Soya, H. Positive effect of acute mild exercise on executive function via arousal-related prefrontal activations: An fNIRS study. Neuroimage 2014, 98, 336–345. [Google Scholar] [CrossRef] [PubMed]
- Kayser, K.C.; Puig, V.A.; Estepp, J.R. Predicting and mitigating fatigue effects due to sleep deprivation: A review. Front. Neurosci. 2022, 16, 930280. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Yu, X.; Wang, G.; Han, Y.; Wang, W. Effects of 24-h acute total sleep deprivation on physiological coupling in healthy young adults. Front. Neurosci. 2022, 16, 952329. [Google Scholar] [CrossRef] [PubMed]
- Zeng, H.R.; Xu, F.; Zhang, J.; Cao, Q.F.; Wang, Y.H.; Zhang, P.; Shao, Y.C.; Wu, S.P.; Weng, X.C. Vigilant Attention, Cerebral Blood Flow and Grey Matter Volume Change after 36 h of Acute Sleep Deprivation in Healthy Male Adults: A Pilot Study. Brain Sci. 2022, 12, 1534. [Google Scholar] [CrossRef] [PubMed]
- Tramonti Fantozzi, M.P.; Banfi, T.; Di Galante, M.; Ciuti, G.; Faraguna, U. Sleep Deprivation-Induced Changes in Baseline Brain Activity and Vigilant Attention Performance. Brain Sci. 2022, 12, 1690. [Google Scholar] [CrossRef] [PubMed]
- Skurvydas, A.; Zlibinaite, L.; Solianik, R.; Brazaitis, M.; Valanciene, D.; Baranauskiene, N.; Majauskiene, D.; Mickeviciene, D.; Venckunas, T.; Kamandulis, S. One night of sleep deprivation impairs executive function but does not affect psychomotor or motor performance. Biol. Sport. 2020, 37, 7–14. [Google Scholar] [CrossRef]
- Tomczak, A.; Dąbrowski, J.; Mikulski, T. Psychomotor performance of Polish Air Force cadets after 36 hours of survival training. Ann. Agric. Environ. Med. 2017, 24, 387–391. [Google Scholar] [CrossRef]
- Erickson, K.I.; Hillman, C.; Stillman, C.M.; Ballard, R.M.; Bloodgood, B.; Conroy, D.E.; Macko, R.; Marquez, D.X.; Petruzzello, S.J.; Powell, K.E. Physical Activity, Cognition, and Brain Outcomes: A Review of the 2018 Physical Activity Guidelines. Med. Sci. Sports Exerc. 2019, 51, 1242–1251. [Google Scholar] [CrossRef]
- Perini, R.; Bortoletto, M.; Capogrosso, M.; Fertonani, A.; Miniussi, C. Acute effects of aerobic exercise promote learning. Sci. Rep. 2016, 6, 25440. [Google Scholar] [CrossRef]
- Chang, Y.K.; Labban, J.D.; Gapin, J.I.; Etnier, J.L. The effects of acute exercise on cognitive performance: A meta-analysis. Brain Res. 2012, 1453, 87–101. [Google Scholar] [CrossRef]
- Mekari, S.; Earle, M.; Martins, R.; Drisdelle, S.; Killen, M.; Bouffard-Levasseur, V.; Dupuy, O. Effect of High Intensity Interval Training Compared to Continuous Training on Cognitive Performance in Young Healthy Adults: A Pilot Study. Brain Sci. 2020, 10, 81. [Google Scholar] [CrossRef]
- Kujach, S.; Byun, K.; Hyodo, K.; Suwabe, K.; Fukuie, T.; Laskowski, R.; Dan, I.; Soya, H. A transferable high-intensity intermittent exercise improves executive performance in association with dorsolateral prefrontal activation in young adults. Neuroimage 2018, 169, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Lucas, S.J.; Ainslie, P.N.; Murrell, C.J.; Thomas, K.N.; Franz, E.A.; Cotter, J.D. Effect of age on exercise-induced alterations in cognitive executive function: Relationship to cerebral perfusion. Exp. Gerontol. 2012, 47, 541–551. [Google Scholar] [CrossRef] [PubMed]
- Bilchak, J.N.; Caron, G.; Côté, M.P. Exercise-Induced Plasticity in Signaling Pathways Involved in Motor Recovery after Spinal Cord Injury. Int. J. Mol. Sci. 2021, 22, 4858. [Google Scholar] [CrossRef] [PubMed]
- Chmura, J.; Nazar, K.; Kaciuba-Uściłko, H. Choice reaction time during graded exercise in relation to blood lactate and plasma catecholamine thresholds. Int. J. Sports Med. 1994, 15, 172–176. [Google Scholar] [CrossRef]
- Chmura, J.; Nazar, K. Parallel changes in the onset of blood lactate accumulation (OBLA) and threshold of psychomotor performance deterioration during incremental exercise after training in athletes. Int. J. Psychophysiol. 2010, 75, 287–290. [Google Scholar] [CrossRef]
- Smith, M.; Tallis, J.; Miller, A.; Clarke, N.D.; Guimarães-Ferreira, L.; Duncan, M.J. The effect of exercise intensity on cognitive performance during short duration treadmill running. J. Hum. Kinet. 2016, 51, 27–35. [Google Scholar] [CrossRef]
- Konefał, M.; Chmura, J.; Charmas, M.; Kotowska, J.; Błażejczyk, K.; Chmura, P. Lactate Threshold and Psychomotor Fatigue Threshold in Hot Conditions: Suggestions for Soccer Players Participating in the Qatar World Cup 2022. Int. J. Environ. Res. Public Health 2022, 19, 17028. [Google Scholar] [CrossRef]
- Temesi, J.; Arnal, P.J.; Davranche, K.; Bonnefoy, R.; Levy, P.; Verges, S.; Millet, G.Y. Does central fatigue explain reduced cycling after complete sleep deprivation? Med. Sci. Sports Exerc. 2013, 45, 2243–2253. [Google Scholar] [CrossRef]
- Lambourne, K.; Tomporowski, P. The effect of exercise-induced arousal on cognitive task performance: A meta-regression analysis. Brain Res. 2010, 1341, 12–24. [Google Scholar] [CrossRef]
- McMorris, T.; Davranche, K.; Jones, G.; Hall, B.; Corbett, J.; Minter, C. Acute incremental exercise, performance of a central executive task, and sympathoadrenal system and hypothalamic-pituitary-adrenal axis activity. Int. J. Psychophysiol. 2009, 73, 334–340. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.K.; Etnier, J.L.; Barella, L.A. Exploring the relationship between exercise-induced arousal and cognition using fractionated response time. Res. Q. Exerc. Sport 2009, 80, 78–86. [Google Scholar] [CrossRef]
- Audiffren, M.; Tomporowski, P.D.; Zagrodnik, J. Acute aerobic exercise and information processing: Energizing motor processes during a choice reaction time task. Acta Psychol. 2008, 129, 410–419. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Nakagawa, S. Physical activity for cognitive health promotion: An overview of the underlying neurobiological mechanisms. Ageing Res. Rev. 2023, 86, 101868. [Google Scholar] [CrossRef] [PubMed]
- Bergersen, L.H. Lactate transport and signaling in the brain: Potential therapeutic targets and roles in body-brain interaction. J. Cereb. Blood Flow Metab. 2015, 35, 176–185. [Google Scholar] [CrossRef] [PubMed]
- Proia, P.; Di Liegro, C.M.; Schiera, G.; Fricano, A.; Di Liegro, I. Lactate as a Metabolite and a Regulator in the Central Nervous System. Int. J. Mol. Sci. 2016, 17, 1450. [Google Scholar] [CrossRef] [PubMed]
- Hertz, L.; Gibbs, M.E.; Dienel, G.A. Fluxes of lactate into, from, and among gap junction-coupled astrocytes and their interaction with noradrenaline. Front. Neurosci. 2014, 8, 261. [Google Scholar] [CrossRef]
- Lee, K.Y.; Rhodes, J.S.; Saif, M.T.A. Astrocyte-mediated Transduction of Muscle Fiber Contractions Synchronizes Hippocampal Neuronal Network Development. Neuroscience 2023, 515, 25–36. [Google Scholar] [CrossRef]
- Carvalho, C.; Moreira, P.I. Oxidative Stress: A Major Player in Cerebrovascular Alterations Associated to Neurodegenerative Events. Front. Physiol. 2018, 9, 806. [Google Scholar] [CrossRef]
- Song, K.; Li, Y.; Zhang, H.; An, N.; Wei, Y.; Wang, L.; Tian, C.; Yuan, M.; Sun, Y.; Xing, Y.; et al. Oxidative stress-mediated blood-brain barrier (BBB) disruption in neurological diseases. Oxid. Med. Cell. Longev. 2020, 2020, 4356386. [Google Scholar] [CrossRef]
- Ko, I.G.; Kim, C.J.; Kim, H. Treadmill exercise improves memory by up-regulating dopamine and down-regulating D2 dopamine receptor in traumatic brain injury rats. J. Exerc. Rehabil. 2019, 15, 504–511. [Google Scholar] [CrossRef]
- Alenina, N.; Klempin, F. The role of serotonin in adult hippocampal neurogenesis. Behav. Brain Res. 2015, 277, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Kraus, C.; Castrén, E.; Kasper, S.; Lanzenberger, R. Serotonin and neuroplasticity—Links between molecular, functional and structural pathophysiology in depression. Neurosci. Biobehav. Rev. 2017, 77, 317–326. [Google Scholar] [CrossRef] [PubMed]
- Pietrelli, A.; Matković, L.; Vacotto, M.; Lopez-Costa, J.J.; Basso, N.; Brusco, A. Aerobic exercise upregulates the BDNF-Serotonin systems and improves the cognitive function in rats. Neurobiol. Learn. Mem. 2018, 155, 528–542. [Google Scholar] [CrossRef]
- Moraine, J.J.; Lamotte, M.; Berré, J.; Niset, G.; Leduc, A.; Naeije, R. Relationship of middle cerebral artery blood flow velocity to intensity during dynamic exercise in normal subjects. Eur. J. Appl. Physiol. Occup. Physiol. 1993, 67, 35–38. [Google Scholar] [CrossRef]
- Hellström, G.; Fischer-Colbrie, W.; Wahlgren, N.G.; Jogestrand, T. Carotid artery blood flow and middle cerebral artery blood flow velocity during physical exercise. J. Appl. Physiol. 1985 1996, 81, 413–418. [Google Scholar] [CrossRef]
- Sato, K.; Ogoh, S.; Hirasawa, A.; Oue, A.; Sadamoto, T. The distribution of blood flow in the carotid and vertebral arteries during dynamic exercise in humans. J. Physiol. 2011, 589, 2847–2856. [Google Scholar] [CrossRef] [PubMed]
- Ogoh, S.; Tsukamoto, H.; Hirasawa, A.; Hasegawa, H.; Hirose, N.; Hashimoto, T. The effect of changes in cerebral blood flow on cognitive function during exercise. Physiol. Rep. 2014, 2, e12163. [Google Scholar] [CrossRef]
- Smale, B.A.; Northey, J.M.; Smee, D.J.; Versey, N.G.; Rattray, B. Compression garments and cerebral blood flow: Influence on cognitive and exercise performance. Eur. J. Sport Sci. 2018, 18, 315–322. [Google Scholar] [CrossRef]
- Hunter, G.R.; Moellering, D.R.; Carter, S.J.; Gower, B.A.; Bamman, M.M.; Hornbuckle, L.M.; Plaisance, E.P.; Fishe, R.G. Potential Causes of Elevated REE after High-Intensity Exercise. Med. Sci. Sports Exerc. 2017, 49, 2414–2421. [Google Scholar] [CrossRef]
- Moniz, S.C.; Islam, H.; Hazell, T.J. Mechanistic and methodological perspectives on the impact of intense interval training on post-exercise metabolism. Scand. J. Med. Sci. Sports 2020, 30, 638–651. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mikulski, T.; Górecka, M.; Bogdan, A.; Młynarczyk, M.; Ziemba, A.W. Psychomotor Performance after 30 h of Sleep Deprivation Combined with Exercise. Brain Sci. 2023, 13, 570. https://doi.org/10.3390/brainsci13040570
Mikulski T, Górecka M, Bogdan A, Młynarczyk M, Ziemba AW. Psychomotor Performance after 30 h of Sleep Deprivation Combined with Exercise. Brain Sciences. 2023; 13(4):570. https://doi.org/10.3390/brainsci13040570
Chicago/Turabian StyleMikulski, Tomasz, Monika Górecka, Anna Bogdan, Magdalena Młynarczyk, and Andrzej W. Ziemba. 2023. "Psychomotor Performance after 30 h of Sleep Deprivation Combined with Exercise" Brain Sciences 13, no. 4: 570. https://doi.org/10.3390/brainsci13040570
APA StyleMikulski, T., Górecka, M., Bogdan, A., Młynarczyk, M., & Ziemba, A. W. (2023). Psychomotor Performance after 30 h of Sleep Deprivation Combined with Exercise. Brain Sciences, 13(4), 570. https://doi.org/10.3390/brainsci13040570





