Two-Stage Surgical Management for Acutely Presented Large Vestibular Schwannomas: Report of Two Cases
Abstract
1. Introduction
2. Results
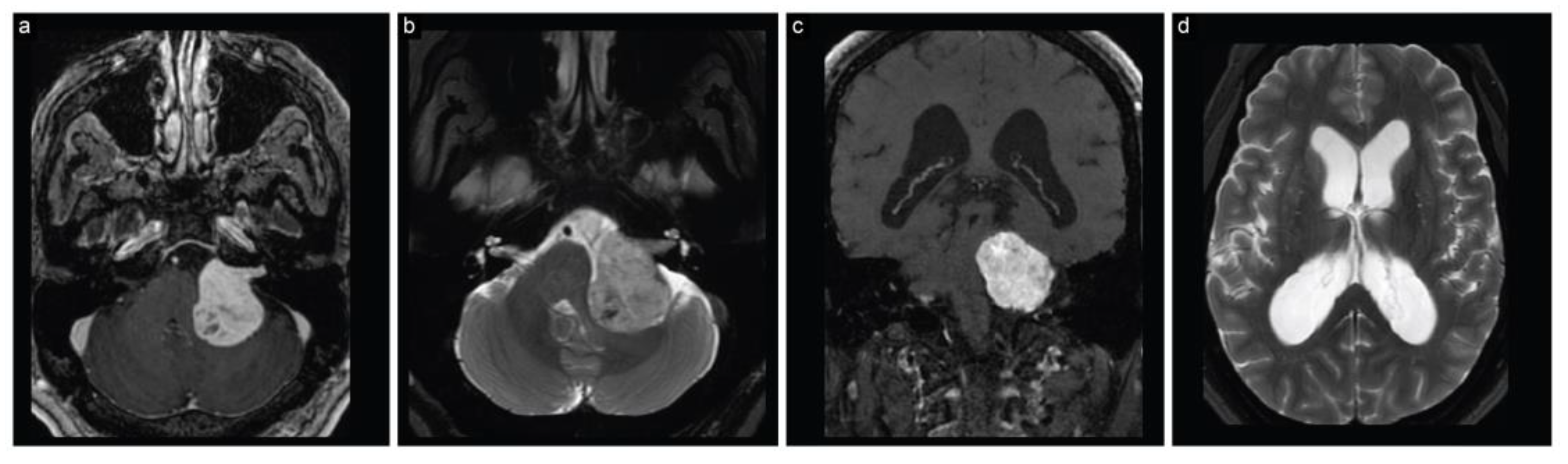
2.1. Case 1
2.2. Case 2
3. Discussion
Indications and Timing Interval for the Staged Surgeries
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Baskaya, M.K.; Pyle, G.M.; Roche, J.P. Vestibular Schwannoma Surgery: A Video Guide; Springer: Berlin/Heidelberg, Germany, 2019. [Google Scholar]
- Chamoun, R.; MacDonald, J.; Shelton, C.; Couldwell, W.T. Surgical approaches for resection of vestibular schwannomas: Translabyrinthine, retrosigmoid, and middle fossa approaches. Neurosurg. Focus 2012, 33, E9. [Google Scholar] [CrossRef] [PubMed]
- Nickele, C.M.; Akture, E.; Gubbels, S.P.; Baskaya, M.K. A stepwise illustration of the translabyrinthine approach to a large cystic vestibular schwannoma. Neurosurg. Focus 2012, 33, 5. [Google Scholar] [CrossRef] [PubMed]
- Ocak, P.E.; Dogan, I.; Ocak, U.; Dinc, C.; Baskaya, M.K. Facial nerve outcome and extent of resection in cystic versus solid vestibular schwannomas in radiosurgery era. Neurosurg. Focus 2018, 44, 8. [Google Scholar] [CrossRef]
- Sayyahmelli, S.; Roche, J.; Baskaya, M.K. Microsurgical Gross Total Resection of a Large Residual/Recurrent Vestibular Schwannoma via Translabyrinthine Approach. J. Neurol. Surg. Part B 2018, 79, S387–S388. [Google Scholar] [CrossRef]
- Marchioni, D.; Carney, M.; Rubini, A.; Nogueira, J.F.; Masotto, B.; Alicandri-Ciufelli, M.; Presutti, L. The Fully Endoscopic Acoustic Neuroma Surgery. Otolaryngol. Clin. N. Am. 2016, 49, 1227–1236. [Google Scholar] [CrossRef]
- Molinari, G.; Calvaruso, F.; Presutti, L.; Marchioni, D.; Alicandri-Ciufelli, M.; Friso, F.; Fernandez, I.J.; Francoli, P.; Di Maro, F. Vestibular schwannoma removal through expanded transcanal transpromontorial approach: A multicentric experience. Eur. Arch. Oto-Rhino-Laryn. 2023, 280, 2165–2172. [Google Scholar] [CrossRef]
- Bi, Y.K.; Ni, Y.J.; Gao, D.D.; Zhu, Q.W.; Zhou, Q.Y.; Tang, J.J.; Liu, J.; Shi, F.; Li, H.C.; Yin, J.; et al. Endoscope-Assisted Retrosigmoid Approach for Vestibular Schwannomas With Intracanalicular Extensions: Facial Nerve Outcomes. Front. Oncol. 2022, 11, 774462. [Google Scholar] [CrossRef]
- Presutti, L.; Magnaguagno, F.; Pavesi, G.; Cunsolo, E.; Pinna, G.; Alicandri-Ciufelli, M.; Marchioni, D.; Prontera, A.; Gioacchini, F.M. Combined endoscopic-microscopic approach for vestibular schwannoma removal: Outcomes in a cohort of 81 patients. Acta Otorhinolaryngol. Ital. 2014, 34, 427–433. [Google Scholar]
- Comey, C.H.; Jannetta, P.J.; Sheptak, P.E.; Jho, H.D.; Burkhart, L.E. Staged Removal of Acoustic Tumors—Techniques and Lessons Learned from a Series of 83 Patients. Neurosurgery 1995, 37, 915–920. [Google Scholar] [CrossRef]
- Kim, E.; Nam, S.-I. Staging in vestibular schwannoma surgery: A modified technique. J. Korean Neurosurg. Soc. 2008, 43, 57–60. [Google Scholar] [CrossRef][Green Version]
- Patni, A.H.; Kartush, J.M. Staged resection of large acoustic neuromas. Otolaryngol. Head Neck Surg. 2005, 132, 11–19. [Google Scholar] [CrossRef]
- Abe, T.; Izumiyama, H.; Imaizumi, Y.; Kobayashi, S.; Shimazu, M.; Sasaki, K.; Matsumoto, K.; Kushima, M. Staged resection of large hypervascular vestibular schwannomas in young adults. Skull Base-Interdiscip. Appr. 2001, 11, 199–206. [Google Scholar] [CrossRef][Green Version]
- Dandy, W.E. An operation for the total removal of cerebellopontine (acoustic) tumors. Surg Gynecol Obs. 1925, 41, 129–148. [Google Scholar]
- Raslan, A.M.; Liu, J.K.; McMenomey, S.O.; Delashaw, J.B., Jr. Staged resection of large vestibular schwannomas. J. Neurosurg. 2012, 116, 1126–1133. [Google Scholar] [CrossRef] [PubMed]
- Sheptak, P.E.; Jannetta, P.J. The two-stage excision of huge acoustic neurinomas. J. Neurosurg. 1979, 51, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Ballance, C.A. Some Points in the Surgery of the Brain and Its Membranes; Macmillan: London, UK, 1907. [Google Scholar]
- Cushing, H. Tumors of the Nervus Acusticus and the Syndrome of the Cerebellopontile Angle; WB Saunders: Philadelphia, PA, USA, 1917. [Google Scholar]
- House, W.F. Surgical exposure of the internal auditory canal and its contents through the middle, cranial fossa. Laryngoscope 1961, 71, 1363–1385. [Google Scholar] [PubMed]
- Kurze, T.; Doyle, J.B. Extradural Intracranial (Middle Fossa) Approach to Internal Auditory Canal. J. Neurosurg. 1962, 19, 1033–1037. [Google Scholar] [CrossRef]
- Rand, R.; Kurze, T. Micro-neurosurgical resection of acoustic tumors by a transmeatal posterior fossa approach. Bull. Los Angeles Neurol. Soc. 1965, 30, 17–20. [Google Scholar]
- Arts, H.A.; Telian, S.A.; El-Kashlan, H.; Thompson, B.G. Hearing preservation and facial nerve outcomes in vestibular schwannoma surgery: Results using the middle cranial fossa approach. Otol. Neurotol. 2006, 27, 234–241. [Google Scholar] [CrossRef]
- Brackmann, D.E.; Cullen, R.D.; Fisher, L.M. Facial nerve function after translabyrinthine vestibular schwannoma surgery. Otolaryngol. Head Neck Surg. 2007, 136, 773–777. [Google Scholar] [CrossRef]
- Irving, R.M.; Jackler, R.K.; Pitts, L.H. Hearing preservation in patients undergoing vestibular schwannoma surgery: Comparison of middle fossa and retrosigmoid approaches. J. Neurosurg. 1998, 88, 840–845. [Google Scholar] [CrossRef]
- Lanman, T.H.; Brackmann, D.E.; Hitselberger, W.E.; Subin, B. Report of 190 consecutive cases of large acoustic tumors (vestibular schwannoma) removed via the translabyrinthine approach. J. Neurosurg. 1999, 90, 617–623. [Google Scholar] [CrossRef]
- Noudel, R.; Gomis, P.; Duntze, J.; Marnet, D.; Bazin, A.; Roche, P.H. Hearing preservation and facial nerve function after microsurgery for intracanalicular vestibular schwannomas: Comparison of middle fossa and restrosigmoid approaches. Acta Neurochir. 2009, 151, 935–945. [Google Scholar] [CrossRef]
- Samii, M.; Gerganov, V.; Samii, A. Improved preservation of hearing and facial nerve function in vestibular schwannoma surgery via the retrosigmoid approach in a series of 200 patients. J. Neurosurg. 2006, 105, 527–535. [Google Scholar] [CrossRef]
- Samii, M.; Gerganov, V.M.; Samii, A. Functional outcome after complete surgical removal of giant vestibular schwannomas Clinical article. J. Neurosurg. 2010, 112, 860–867. [Google Scholar] [CrossRef] [PubMed]
- Yamakami, I.; Uchino, Y.; Kobayashi, E.; Yamaura, A.; Oka, N. Removal of large acoustic neurinomas (vestibular schwannomas) by the retrosigmoid approach with no mortality and minimal morbidity. J. Neurol. Neurosurg. Psychiatry 2004, 75, 453–458. [Google Scholar] [CrossRef] [PubMed]
- Yasargil, M. Microneurosurgery, Volume IVB: Microneurosurgery of CNS Tumors; Georg Thieme Verlag: Stuttgart, Germany, 1996. [Google Scholar]
- Yaşargil, M.; Smith, R.; Gasser, J. Microsurgical Approach to Acoustic Neurinomas. Adv. Technol. Stand. Neurosurg. 1977, 4, 93–129. [Google Scholar]
- Yaşargil, M.G. Microsurgery Applied to Neurosurgery; Georg Thieme: Stuttgart, Germany, 1969. [Google Scholar]
- Kaylie, D.M.; Gilbert, E.; Horgan, M.A.; Delashaw, J.B.; McMenomey, S.O. Acoustic neuroma surgery outcomes. Otol. Neurotol. 2001, 22, 686–689. [Google Scholar] [CrossRef]
- Godefroy, W.P.; van der Mey, A.G.L.; de Bruine, F.T.; Hoekstra, E.R.; Malessy, M.J.A. Surgery for Large Vestibular Schwannoma: Residual Tumor and Outcome. Otol. Neurotol. 2009, 30, 629–634. [Google Scholar] [CrossRef]
- Silva, J.; Cerejo, A.; Duarte, F.; Silveira, F.; Vaz, R. Surgical Removal of Giant Acoustic Neuromas. World Neurosurg. 2012, 77, 731–735. [Google Scholar] [CrossRef]
- Sughrue, M.E.; Yang, I.; Rutkowski, M.J.; Aranda, D.; Parsa, A.T. Preservation of facial nerve function after resection of vestibular schwannoma. Br. J. Neurosurg. 2010, 24, 666–671. [Google Scholar] [CrossRef] [PubMed]
- Gazia, F.; Callejo, A.; Perez-Grau, M.; Lareo, S.; Prades, J.; Roca-Ribas, F.; Amilibia, E. Pre- and intra-operative prognostic factors of facial nerve function in cerebellopontine angle surgery. Eur. Arch. Oto-Rhino-Laryn. 2022, 280, 1055–1062. [Google Scholar] [CrossRef] [PubMed]
- Andersson, G.; Kinnefors, A.; Ekvall, L.; RaskAndersen, H. Tinnitus and translabyrinthine acoustic neuroma surgery. Audiol. Neuro-Otol. 1997, 2, 403–409. [Google Scholar] [CrossRef] [PubMed]
- Fahy, C.; Nikolopoulos, T.P.; O’Donoghue, G.M. Acoustic neuroma surgery and tinnitus. Eur. Arch. Oto-Rhino-Laryn. 2002, 259, 299–301. [Google Scholar] [CrossRef]
- Kojima, T.; Oishi, N.; Nishiyama, T.; Ogawa, K. Severity of Tinnitus Distress Negatively Impacts Quality of Life in Patients With Vestibular Schwannoma and Mimics Primary Tinnitus. Front. Neurol. 2019, 10, 389. [Google Scholar] [CrossRef]
- West, N.; Bunne, M.; Sass, H.; Caye-Thomasen, P. Cochlear Implantation for Patients with a Vestibular Schwannoma: Effect on Tinnitus Handicap. J. Int. Adv. Otol. 2022, 18, 382–387. [Google Scholar] [CrossRef] [PubMed]
- Conway, R.M.; Tu, N.C.; Sioshansi, P.C.; Porps, S.L.; Schutt, C.A.; Hong, R.S.; Jacob, J.T.; Babu, S.C. Early Outcomes of Simultaneous Translabyrinthine Resection and Cochlear Implantation. Laryngoscope 2021, 131, E2312–E2317. [Google Scholar] [CrossRef]
- Salem, N.; Galal, A.; Piras, G.; Sykopetrites, V.; Di Rubbo, V.; Talaat, M.; Sobhy, O.; Sanna, M. Management of Vestibular Schwannoma with Normal Hearing. Audiol. Neuro-Otol. 2023, 28, 12–21. [Google Scholar] [CrossRef]
- Sorrentino, F.; Tealdo, G.; Cazzador, D.; Favaretto, N.; Brotto, D.; Montino, S.; Caserta, E.; Bovo, R.; Denaro, L.; Baro, V.; et al. Cochlear implant in vestibular schwannomas: Long-term outcomes and critical analysis of indications. Eur. Arch. Oto-Rhino-Laryn. 2022, 279, 4709–4718. [Google Scholar] [CrossRef]
- Wick, C.C.; Butler, M.J.; Yeager, L.H.; Kallogjeri, D.; Durakovic, N.; McJunkin, J.L.; Shew, M.A.; Herzog, J.A.; Buchman, C.A. Cochlear Implant Outcomes Following Vestibular Schwannoma Resection: Systematic Review. Otol. Neurotol. 2020, 41, 1190–1197. [Google Scholar] [CrossRef]
- Gonzalez-Lopez, P.; Harput, M.V.; Türe, H.; Atalay, B.; Türe, U. Efficacy of placing a thin layer of gelatin sponge over the subdural space during dural closure in preventing meningo-cerebral adhesion. World Neurosurg. 2015, 83, 93–101. [Google Scholar] [CrossRef] [PubMed]





Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Keles, A.; Ozaydin, B.; Erginoglu, U.; Baskaya, M.K. Two-Stage Surgical Management for Acutely Presented Large Vestibular Schwannomas: Report of Two Cases. Brain Sci. 2023, 13, 1548. https://doi.org/10.3390/brainsci13111548
Keles A, Ozaydin B, Erginoglu U, Baskaya MK. Two-Stage Surgical Management for Acutely Presented Large Vestibular Schwannomas: Report of Two Cases. Brain Sciences. 2023; 13(11):1548. https://doi.org/10.3390/brainsci13111548
Chicago/Turabian StyleKeles, Abdullah, Burak Ozaydin, Ufuk Erginoglu, and Mustafa K. Baskaya. 2023. "Two-Stage Surgical Management for Acutely Presented Large Vestibular Schwannomas: Report of Two Cases" Brain Sciences 13, no. 11: 1548. https://doi.org/10.3390/brainsci13111548
APA StyleKeles, A., Ozaydin, B., Erginoglu, U., & Baskaya, M. K. (2023). Two-Stage Surgical Management for Acutely Presented Large Vestibular Schwannomas: Report of Two Cases. Brain Sciences, 13(11), 1548. https://doi.org/10.3390/brainsci13111548




