Neural Activity Associated with Symptoms Change in Depressed Adolescents following Self-Processing Neurofeedback
Abstract
:1. Introduction
1.1. Self-Referential Processing and Depression
1.2. Emotional Regulation and Depression
1.3. Neurofeedback in Adult and Adolescent Populations
2. Materials and Methods
2.1. Online Analyses
2.2. Offline Analyses
3. Results
3.1. Brain Activity Associated with Symptom Change during Feedback vs. Counting Backwards
3.2. Brain Activity Associated with Symptom during Self-Processing
4. Discussion
4.1. Symptoms Change and Neurofeedback
4.2. Post- vs. Pre- Neurofeedback Self Processing and Symptoms Improvement
4.3. Limitations and Future Directions
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Twenge, J.M.; Cooper, A.B.; Joiner, T.E.; Duffy, M.E.; Binau, S.G. Age, period, and cohort trends in mood disorder indicators and suicide-related outcomes in a nationally representative dataset, 2005–2017. J. Abnorm. Psychol. 2019, 128, 185–199. [Google Scholar] [CrossRef] [PubMed]
- The Substance Abuse and Mental Health Services Administration. 2020 National Survey of Drug Use and Health (NSDUH) Releases; The Substance Abuse and Mental Health Services Administration: Rockville, MD, USA, 2020. [Google Scholar]
- Hankin, B.L.; Abramson, L.Y.; Moffitt, T.E.; Silva, P.A.; Mcgee, R.; Angell, K.E. Development of depression from preadolescence to young adulthood: Emerging gender differences in a 10-year longitudinal study. J. Abnorm. Psychol. 1998, 107, 128–140. [Google Scholar] [CrossRef] [PubMed]
- Andersen, S.L.; Teicher, M.H. Stress, sensitive periods and maturational events in adolescent depression. Trends Neurosci. 2008, 31, 183–191. [Google Scholar] [CrossRef]
- Nolen-Hoeksema, S. The role of rumination in depressive disorders and mixed anxiety/depressive symptoms. J. Abnorm. Psychol. 2000, 109, 504–511. [Google Scholar] [CrossRef]
- Zisook, S.; Rush, A.J.; Lesser, I.; Wisniewski, S.R.; Trivedi, M.; Husain, M.M.; Balasubramani, G.K.; Alpert, J.E.; Fava, M. Preadult onset vs. adult onset of major depressive disorder: A replication study. Acta Psychiatr. Scand. 2007, 115, 196–205. [Google Scholar] [CrossRef] [PubMed]
- Watkins, E.R. Constructive and unconstructive repetitive thought. Psychol. Bull. 2008, 134, 163–206. [Google Scholar] [CrossRef] [PubMed]
- Lyubomirsky, S.; Layous, K.; Chancellor, J.; Nelson, S.K. Thinking About Rumination: The Scholarly Contributions and Intellectual Legacy of Susan Nolen-Hoeksema. Annu. Rev. Clin. Psychol. 2015, 11, 1–22. [Google Scholar] [CrossRef]
- Gruber, J.; Harvey, A.G.; Johnson, S.L. Reflective and ruminative processing of positive emotional memories in bipolar disorder and healthy controls. Behav. Res. Ther. 2009, 47, 697–704. [Google Scholar] [CrossRef]
- Nejad, A.B.; Fossati, P.; Lemogne, C. Self-Referential Processing, Rumination, and Cortical Midline Structures in Major Depression. Front. Hum. Neurosci. 2013, 7, 666. [Google Scholar] [CrossRef]
- Molnar-Szakacs, I.; Uddin, L. Self-Processing and the Default Mode Network: Interactions with the Mirror Neuron System. Front. Hum. Neurosci. 2013, 7, 571. [Google Scholar] [CrossRef] [Green Version]
- Schwartz, J.; Ordaz, S.J.; Ho, T.C.; Gotlib, I.H. Longitudinal decreases in suicidal ideation are associated with increases in salience network coherence in depressed adolescents. J. Affect. Disord. 2019, 245, 545–552. [Google Scholar] [CrossRef] [PubMed]
- Young, C.; Raz, G.; Everaerd, D.; Beckmann, C.F.; Tendolkar, I.; Hendler, T.; Fernandez, G.; Hermans, E.J. Dynamic Shifts in Large-Scale Brain Network Balance as a Function of Arousal. J. Neurosci. 2016, 37, 281–290. [Google Scholar] [CrossRef] [PubMed]
- Geng, H.; Li, X.; Chen, J.; Li, X.; Gu, R. Decreased Intra- and Inter-Salience Network Functional Connectivity is Related to Trait Anxiety in Adolescents. Front. Behav. Neurosci. 2016, 9, 350. [Google Scholar] [CrossRef]
- Demertzi, A.; Whitfield-Gabrieli, S. Intrinsic Brain Activity and Consciousness. In The Neurology of Consciousness: Cognitive Neuroscience and Neuropathology; Academic Press: Cambridge, MA, USA, 2016; pp. 95–105. [Google Scholar]
- Chen, T.; Cai, W.; Ryali, S.; Supekar, K.; Menon, V. Distinct Global Brain Dynamics and Spatiotemporal Organization of the Salience Network. PLoS Biol. 2016, 14, e1002469. [Google Scholar] [CrossRef] [PubMed]
- Diamond, A. Executive functions. Annu. Rev. Psychol. 2013, 64, 135–168. [Google Scholar] [CrossRef]
- Manning, K.J.; Steffens, D.C. Chapter 11—Systems Neuroscience in Late-Life Depression; Frodl, T.B.T.S.N.i.D., Ed.; Academic Press: San Diego, CA, USA, 2016; pp. 325–340. [Google Scholar]
- Hamilton, J.P.; Farmer, M.; Fogelman, P.; Gotlib, I.H. Depressive Rumination, the Default-Mode Network, and the Dark Matter of Clinical Neuroscience. Biol. Psychiatry 2015, 78, 224–230. [Google Scholar] [CrossRef]
- Chen, H.; Mechanic, D.; Hansell, S. A Longitudinal Study of Self-Awareness and Depressed Mood in Adolescence. J. Youth Adolesc. 1998, 27, 719–734. [Google Scholar] [CrossRef]
- Northoff, G.; Bermpohl, F. Cortical midline structures and the self. Trends Cogn. Sci. 2004, 8, 102–107. [Google Scholar] [CrossRef]
- Raichle, M.E.; MacLeod, A.M.; Snyder, A.Z.; Powers, W.J.; Gusnard, D.A.; Shulman, G.L. A default mode of brain function. Proc. Natl. Acad. Sci. USA 2001, 98, 676–682. [Google Scholar] [CrossRef]
- Uddin, L.Q. Salience processing and insular cortical function and dysfunction. Nat. Rev. Neurosci. 2015, 16, 55–61. [Google Scholar] [CrossRef]
- Stuhrmann, A.; Dohm, K.; Kugel, H.; Zwanzger, P.; Redlich, R.; Grotegerd, D.; Rauch, A.; Arolt, V.; Heindel, W.; Suslow, T.; et al. Mood-congruent amygdala responses to subliminally presented facial expressions in major depression: Associations with anhedonia. J. Psychiatry Neurosci. 2013, 38, 249–258. [Google Scholar] [CrossRef]
- Hu, B.; Rao, J.; Li, X.; Cao, T.; Li, J.; Majoe, D.; Gutknecht, J. Emotion Regulating Attentional Control Abnormalities in Major Depressive Disorder: An Event-Related Potential Study. Sci. Rep. 2017, 7, 13530. [Google Scholar] [CrossRef] [PubMed]
- Quevedo, K.; Harms, M.; Sauder, M.; Scott, H.; Mohamed, S.; Thomas, K.M.; Schallmo, M.-P.; Smyda, G. The neurobiology of self face recognition among depressed adolescents. J. Affect. Disord. 2017, 229, 22–31. [Google Scholar] [CrossRef]
- Zhu, Y.; Gao, H.; Tong, L.; Li, Z.; Wang, L.; Zhang, C.; Yang, Q.; Yan, B. Emotion Regulation of Hippocampus Using Real-Time fMRI Neurofeedback in Healthy Human. Front. Hum. Neurosci. 2019, 13, 242. [Google Scholar] [CrossRef]
- Young, K.D.; Zotev, V.; Phillips, R.; Misaki, M.; Yuan, H.; Drevets, W.C.; Bodurka, J. Real-Time fMRI Neurofeedback Training of Amygdala Activity in Patients with Major Depressive Disorder. PLoS ONE 2014, 9, e88785. [Google Scholar] [CrossRef]
- Rance, M.; Walsh, C.; Sukhodolsky, D.G.; Pittman, B.; Qiu, M.; Kichuk, S.A.; Wasylink, S.; Koller, W.N.; Bloch, M.; Gruner, P.; et al. Time course of clinical change following neurofeedback. NeuroImage 2018, 181, 807–813. [Google Scholar] [CrossRef]
- Yuan, H.; Young, K.D.; Phillips, R.; Zotev, V.; Misaki, M.; Bodurka, J. Resting-state functional connectivity modulation and sustained changes after real-time functional magnetic resonance imaging neurofeedback training in depression. Brain Connect. 2014, 4, 690–701. [Google Scholar] [CrossRef] [PubMed]
- Alegria, A.A.; Wulff, M.; Brinson, H.; Barker, G.J.; Norman, L.J.; Brandeis, D.; Stahl, D.; David, A.S.; Taylor, E.; Giampietro, V.; et al. Real-time fMRI neurofeedback in adolescents with attention deficit hyperactivity disorder. Hum. Brain Mapp. 2017, 38, 3190–3209. [Google Scholar] [CrossRef]
- Rubia, K.; Criaud, M.; Wulff, M.; Alegria, A.; Brinson, H.; Barker, G.; Stahl, D.; Giampietro, V. Functional connectivity changes associated with fMRI neurofeedback of right inferior frontal cortex in adolescents with ADHD. NeuroImage 2019, 188, 43–58. [Google Scholar] [CrossRef] [PubMed]
- Hashemian, P.; Sadjadi, S.A. Evaluation of neurofeedback therapy in adolescents with major depressive disorder who take fluoxetine. Afr. J. Psychiatry 2015, 18, 2. [Google Scholar]
- de Charms, R.C.; Christoff, K.; Glover, G.H.; Pauly, J.M.; Whitfield, S.; Gabrieli, J.D. Learned regulation of spatially localized brain activation using real-time fMRI. NeuroImage 2004, 21, 436–443. [Google Scholar] [CrossRef] [PubMed]
- Kaufman, J.; Birmaher, B.; Brent, D.; Rao, U.M.A.; Flynn, C.; Moreci, P.; Williamson, D.; Ryan, N. Schedule for Affective Disorders and Schizophrenia for School-Age Children-Present and Lifetime Version (K-SADS-PL): Initial Reliability and Validity Data. J. Am. Acad. Child Adolesc. Psychiatry 1997, 36, 980–988. [Google Scholar] [CrossRef] [PubMed]
- Poznanski, E.O.; Cook, S.C.; Carroll, B.J. Children’s Depression Rating Scale. Pediatrics 1979, 64, 442–450. [Google Scholar] [CrossRef] [PubMed]
- Weschler, D. Weschler Abbreviated Scale of Intelligence (WASI); Harcourt Inc.: San Diego, CA, USA, 1999. [Google Scholar]
- Messer, S.C.; Angold, A.; Costello, E.J.; Loeber, R.; Van Kammen, W.; Stouthamer-Loeber, M. Development of a short questionnaire for use in epidemiological studies of depression in children and adolescents: Factor composition and structure across development. Int. J. Methods Psychiatr. Res. 1995, 5, 251–262. [Google Scholar]
- Treynor, W.; Gonzalez, R.; Nolen-Hoeksema, S. Rumination Reconsidered: A Psychometric Analysis. Cogn. Ther. Res. 2003, 27, 247–259. [Google Scholar] [CrossRef]
- Hinds, O.; Ghosh, S.; Thompson, T.W.; Yoo, J.J.; Whitfield-Gabrieli, S.; Triantafyllou, C.; Gabrieli, J.D. Computing moment-to-moment BOLD activation for real-time neurofeedback. NeuroImage 2011, 54, 361–368. [Google Scholar] [CrossRef]
- Zawadzki, M.J. Rumination is independently associated with poor psychological health: Comparing emotion regulation strategies. Psychol. Health 2015, 30, 1146–1163. [Google Scholar] [CrossRef]
- Timm, C.; Ubl, B.; Zamoscik, V.; Ebner-Priemer, U.; Reinhard, I.; Huffziger, S.; Kirsch, P.; Kuehner, C. Cognitive and affective trait and state factors influencing the long-term symptom course in remitted depressed patients. PLoS ONE 2017, 12, e0178759. [Google Scholar]
- Visted, E.; Vøllestad, J.; Nielsen, M.B.; Schanche, E. Emotion Regulation in Current and Remitted Depression: A Systematic Review and Meta-Analysis. Front. Psychol. 2018, 9, 756. [Google Scholar] [CrossRef]
- Peirce, J.; Gray, J.R.; Simpson, S.; MacAskill, M.; Höchenberger, R.; Sogo, H.; Kastman, E.; Lindeløv, J.K. PsychoPy2: Experiments in behavior made easy. Behav. Res. Methods 2019, 51, 195–203. [Google Scholar] [CrossRef]
- Thesen, S.; Heid, O.; Mueller, E.; Schad, L.R. Prospective acquisition correction for head motion with image-based tracking for real-time fMRI. Magn. Reson. Med. 2000, 44, 457–465. [Google Scholar] [CrossRef]
- Adamaszek, M.; D’Agata, F.; Ferrucci, R.; Habas, C.; Keulen, S.; Kirkby, K.C.; Leggio, M.; Mariën, P.; Molinari, M.; Moulton, E.; et al. Consensus Paper: Cerebellum and Emotion. Cerebellum 2017, 16, 552–576. [Google Scholar] [CrossRef]
- Makino, Y.; Yokosawa, K.; Takeda, Y.; Kumada, T. Visual search and memory search engage extensive overlapping cerebral cortices: An fMRI study. NeuroImage 2004, 23, 525–533. [Google Scholar] [CrossRef]
- Cavanna, A.E.; Trimble, M.R. The Precuneus: A Review of Its Functional Anatomy and Behavioural Correlates; Oxford University Press: Oxford, UK, 2006; pp. 564–583. [Google Scholar]
- Andrea, E.C. The Precuneus and Consciousness. CNS Spectr. 2007, 12, 545–552. [Google Scholar]
- Kircher, T.T.; Brammer, M.; Bullmore, E.; Simmons, A.; Bartels, M.; David, A.S. The neural correlates of intentional and incidental self processing. Neuropsychologia 2002, 40, 683–692. [Google Scholar] [CrossRef]
- Kjaer, T.W.; Nowak, M.; Lou, H.C. Reflective Self-Awareness and Conscious States: PET Evidence for a Common Midline Parietofrontal Core. NeuroImage 2002, 17, 1080–1086. [Google Scholar] [CrossRef]
- Sandman, C.F.; Young, K.S.; Burklund, L.J.; Saxbe, D.E.; Lieberman, M.D.; Craske, M.G. Changes in functional connectivity with cognitive behavioral therapy for social anxiety disorder predict outcomes at follow-up. Behav. Res. Ther. 2020, 129, 103612. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Chiang-Shan, R.L. Functional connectivity mapping of the human precuneus by resting state fMRI. NeuroImage 2012, 59, 3548–3562. [Google Scholar] [CrossRef]
- D’Argembeau, A.; Collette, F.; Van der Linden, M.; Laureys, S.; Del Fiore, G.; Degueldre, C.; Luxen, A.; Salmon, E. Self-referential reflective activity and its relationship with rest: A PET study. NeuroImage 2005, 25, 616–624. [Google Scholar] [CrossRef]
- Seghier, M.L. The Angular Gyrus:Multiple Functions and Multiple Subdivisions. Neuroscientist 2013, 19, 43–61. [Google Scholar] [CrossRef]
- Ma, C.; Ding, J.; Li, J.; Guo, W.; Long, Z.; Liu, F.; Gao, Q.; Zeng, L.; Zhao, J.; Chen, H. Resting-State Functional Connectivity Bias of Middle Temporal Gyrus and Caudate with Altered Gray Matter Volume in Major Depression. PLoS ONE 2012, 7, e45263. [Google Scholar] [CrossRef] [PubMed]
- Quevedo, K.; Ng, R.; Scott, H.; Martin, J.; Smyda, G.; Keener, M.; Oppenheimer, C.W. The neurobiology of self-face recognition in depressed adolescents with low or high suicidality. J. Abnorm. Psychol. 2016, 125, 1185–1200. [Google Scholar] [CrossRef] [PubMed]
- Auerbach, R.P.; Millner, A.J.; Stewart, J.G.; Esposito, E.C. Identifying differences between depressed adolescent suicide ideators and attempters. J. Affect. Disord. 2015, 186, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Sergent, J.; Ohta, S.; Macdonald, B. Functional neuroanatomy of face and object processing: A positron emission tomography study. Brain 1992, 115, 15–36. [Google Scholar] [CrossRef] [PubMed]
- Kitada, R.; Johnsrude, I.S.; Kochiyama, T.; Lederman, S.J. Brain networks involved in haptic and visual identification of facial expressions of emotion: An fMRI study. NeuroImage 2010, 49, 1677–1689. [Google Scholar] [CrossRef]
- Rossion, B.; Schiltz, C.; Crommelinck, M. The functionally defined right occipital and fusiform “face areas” discriminate novel from visually familiar faces. NeuroImage 2003, 19, 877–883. [Google Scholar] [CrossRef]
- Sugiura, M.; Shah, N.J.; Zilles, K.; Fink, G.R. Cortical Representations of Personally Familiar Objects and Places: Functional Organization of the Human Posterior Cingulate Cortex. J. Cogn. Neurosci. 2005, 17, 183–198. [Google Scholar] [CrossRef]
- Lau, E.F.; Phillips, C.; Poeppel, D. A cortical network for semantics: (de)constructing the N400. Nature reviews. Neuroscience 2008, 9, 920–933. [Google Scholar]
- Davey, J.; Thompson, H.E.; Hallam, G.; Karapanagiotidis, T.; Murphy, C.; De Caso, I.; Krieger-Redwood, K.; Bernhardt, B.C.; Smallwood, J.; Jefferies, E. Exploring the role of the posterior middle temporal gyrus in semantic cognition: Integration of anterior temporal lobe with executive processes. NeuroImage 2016, 137, 165–177. [Google Scholar] [CrossRef]
- Igelström, K.M.; Graziano, M.S.A. The inferior parietal lobule and temporoparietal junction: A network perspective. Neuropsychologia 2017, 105, 70–83. [Google Scholar] [CrossRef]
- Van Overwalle, F. Social cognition and the brain: A meta-analysis. Hum. Brain Mapp. 2009, 30, 829–858. [Google Scholar] [CrossRef] [PubMed]
- Blackwood, N.J.; Bentall, R.P.; Simmons, A.; Murray, R.M.; Howard, R.J. Self-responsibility and the self-serving bias: An fMRI investigation of causal attributions. NeuroImage 2003, 20, 1076–1085. [Google Scholar] [CrossRef]
- Schmahmann, J.D. The cerebellum and cognition. Neurosci. Lett. 2019, 688, 62–75. [Google Scholar]
- Van Overwalle, F.; Baetens, K.; Mariën, P.; Vandekerckhove, M. Social cognition and the cerebellum: A meta-analysis of over 350 fMRI studies. NeuroImage 2014, 86, 554–572. [Google Scholar] [CrossRef] [PubMed]
- Ge, R.; Downar, J.; Blumberger, D.M.; Daskalakis, Z.J.; Vila-Rodriguez, F. Functional connectivity of the anterior cingulate cortex predicts treatment outcome for rTMS in treatment-resistant depression at 3-month follow-up. Brain Stimul. 2020, 13, 206–214. [Google Scholar] [CrossRef] [PubMed]
- MacDuffie, K.E.; MacInnes, J.; Dickerson, K.C.; Eddington, K.M.; Strauman, T.J.; Adcock, R.A. Single session real-time fMRI neurofeedback has a lasting impact on cognitive behavioral therapy strategies. NeuroImage Clin. 2018, 19, 868–875. [Google Scholar] [CrossRef]


| Healthy Controls | Depressed | |
|---|---|---|
| n = 19 | n = 34 | |
| Suicide attempters | n = 0 a | n = 15 b |
| Age at Intake: M (SD) | 16.26 (1.19) | 16.08 (1.27) |
| Age at Scanning: M (SD) | 16.35 (1.23) | 16.11 (1.25) |
| IQ:M (SD) | 115.32 (9.12) a | 108.35 (10.84) b |
| Sex | ||
| Male | 7 (36.84%) | 10 (29.41%) |
| Female | 12 (63.16%) | 24 (70.59%) |
| Puberty: M (SD) | 4.53 (0.65) | 4.53 (0.68) |
| Ethnicity White | 14 (73.68%) | 27 (79.41%) |
| African American/Black | 0 | 2 (5.88%) |
| Native American | 0 | 2 (5.88%) |
| Asian | 3 (15.79%) | 0 |
| Other Ethnicity | 2 (10.53%) | 3 (8.82%) |
| Family Structure Married | 15 (78.95%) | 22 (64.71%) |
| Living with partner | 1 (5.26%) | 3 (8.82%) |
| Separated-Divorced | 3 (15.79%) | 5 (14.71%) |
| Single | 0 | 4 (11.76%) |
| Income | ||
| ≥ 35K | 0 | 6 (17.65%) |
| 35–75 K | 7 (36.84%) | 9 (26.47%) |
| + > 75 K | 12 (63.16%) | 19 (55.88%) |
| Depression before neurofeedback: M (SD) | 3.76 (3.95) a | 29.73 (13.79) b |
| Depression after neurofeedback: M (SD) | 2.26 (2.46) a | 20.35 (16.62) b * |
| Rumination before neurofeedback: M (SD) | 29.31 (6.97) a | 50.71 (11.68) b |
| Rumination after neurofeedback: M (SD) | 27.89 (7.25) a | 44.06 (14.76) b * |
| Depression Severity (CDRS): M (SD) | 19.21 (3.56) a | 49.85 (16.14) b |
| Depression Diagnosis (K-SADS-PL) | ||
| Major Depressive Disorder (MDD) | 0 | 14 |
| MDD with Psychotic Features | 0 | 1 |
| Dysthymia | 0 | 4 |
| Melancholic Depression | 0 | 1 |
| Depressive Disorder-NOS | 0 | 15 |
| Eating Disorders (K-SADS-PL) | 0 | 2 |
| Anxiety Disorders (K-SADS-PL) | 0 | 22 |
| PTSD (K-SADS-PL) | 0 | 6 |
| Disruptive Behavior Disorders (K-SADS-PL) | 0 | 6 |
| Substance Use Presence (K-SADS-PL) | 0 | 2 |
| Medication | ||
| Antidepressants | 0 | 26 |
| Antipsychotics | 0 | 2 |
| Mood stabilizers | 0 | 0 |
| Anxiolytics | 0 | 10 |
| Direction of Prediction | Voxels | Hemisphere | MNI Coordinates | T | |||
|---|---|---|---|---|---|---|---|
| X | Y | Z | |||||
| Areas linked to depression change during neurofeedback vs. counting-backwards (ESOM-NF). | |||||||
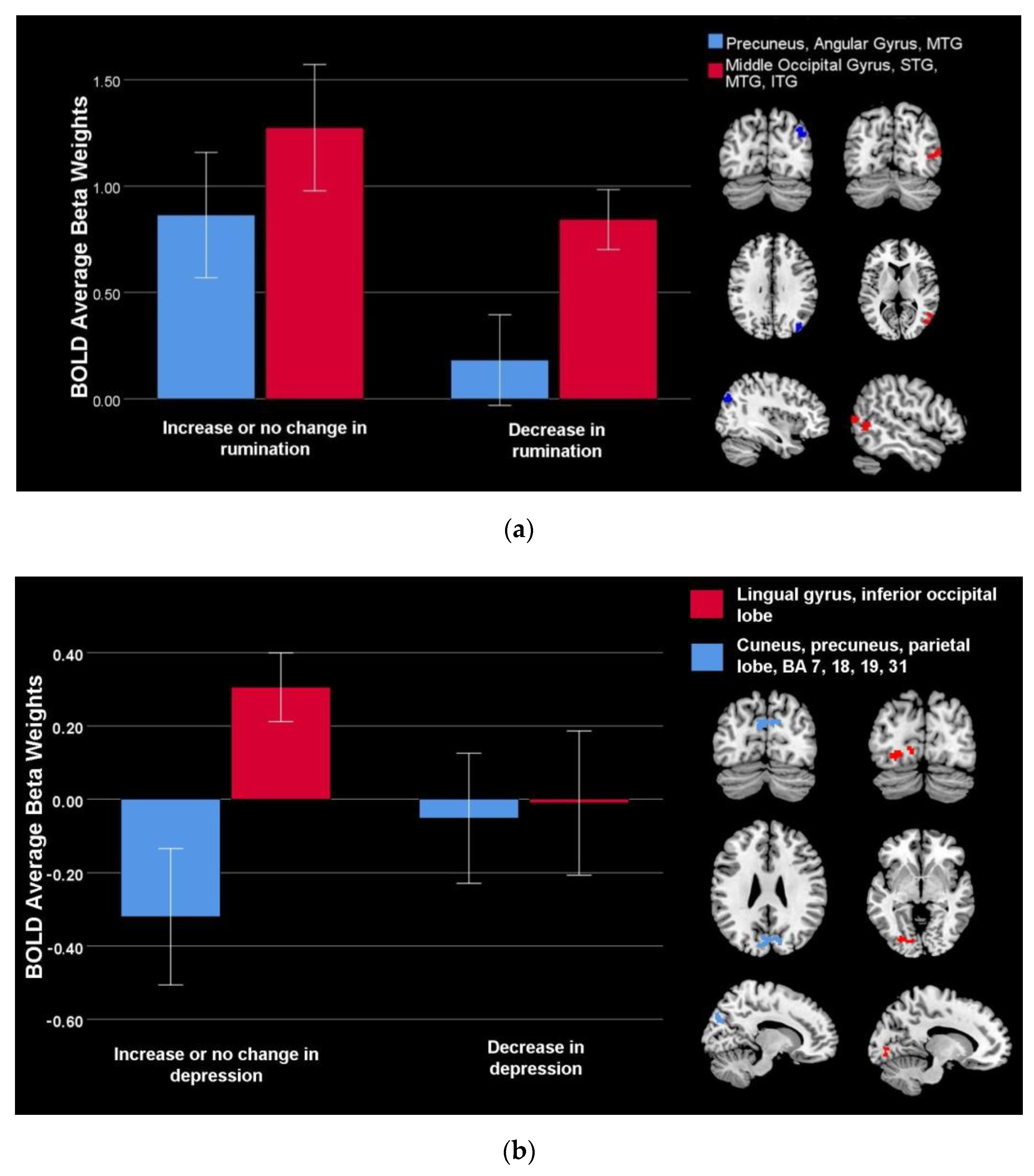
| Lingual gyrus, inferior occipital lobe | Positive | 112 | Left | −28 | −80 | −6 | 3.58 |
| Cuneus, precuneus, parietal lobe, BA 7, 18, 19, 31 | Negative | 276 | Bilateral | −4 | −82 | 26 | 4.06 |
| Areas linked to rumination change during neurofeedback vs. counting backwards (ESOM-NF). | |||||||
| Middle occipital gyrus, superior temporal gyrus, inferior temporal gyrus, middle temporal gyrus, BA 19, 37, 39 | Positive | 152 | Right | 52 | −58 | 6 | 4.40 |
| Precuneus, angular gyrus, middle temporal gyrus, BA 19 and 39 | Positive | 98 | Right | 38 | −82 | 34 | 4.81 |
| Areas linked to depression change during self-recognition post- vs. pre- neurofeedback (ESOM-Post minus ESOM-Pre). | |||||||
| Cerebellum | Positive | 429 | Right | 24 | −80 | −26 | 5.07 |
| Middle temporal gyrus (BA 21), superior temporal gyrus (BA 22) | Positive | 547 | Left | −64 | −26 | −2 | 4.92 |
| Frontal lobe, middle frontal gyrus | Positive | 121 | Left | −18 | −10 | 42 | 4.50 |
| Cerebellum | Positive | 283 | Left | −18 | −78 | −42 | 4.50 |
| Supramarginal gyrus, middle temporal gyrus, superior temporal gyrus | Positive | 127 | Left | −38 | −54 | 32 | 3.94 |
| Middle temporal gyrus (BA 21), superior temporal gyrus (BA 22) | Positive | 111 | Right | 68 | −40 | 0 | 3.79 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahrweiler, N.; Santana-Gonzalez, C.; Zhang, N.; Quandt, G.; Ashtiani, N.; Liu, G.; Engstrom, M.; Schultz, E.; Liengswangwong, R.; Teoh, J.Y.; et al. Neural Activity Associated with Symptoms Change in Depressed Adolescents following Self-Processing Neurofeedback. Brain Sci. 2022, 12, 1128. https://doi.org/10.3390/brainsci12091128
Ahrweiler N, Santana-Gonzalez C, Zhang N, Quandt G, Ashtiani N, Liu G, Engstrom M, Schultz E, Liengswangwong R, Teoh JY, et al. Neural Activity Associated with Symptoms Change in Depressed Adolescents following Self-Processing Neurofeedback. Brain Sciences. 2022; 12(9):1128. https://doi.org/10.3390/brainsci12091128
Chicago/Turabian StyleAhrweiler, Natasha, Carmen Santana-Gonzalez, Na Zhang, Grace Quandt, Nikki Ashtiani, Guanmin Liu, Maggie Engstrom, Erika Schultz, Ryan Liengswangwong, Jia Yuan Teoh, and et al. 2022. "Neural Activity Associated with Symptoms Change in Depressed Adolescents following Self-Processing Neurofeedback" Brain Sciences 12, no. 9: 1128. https://doi.org/10.3390/brainsci12091128
APA StyleAhrweiler, N., Santana-Gonzalez, C., Zhang, N., Quandt, G., Ashtiani, N., Liu, G., Engstrom, M., Schultz, E., Liengswangwong, R., Teoh, J. Y., Kozachok, K., & Quevedo, K. (2022). Neural Activity Associated with Symptoms Change in Depressed Adolescents following Self-Processing Neurofeedback. Brain Sciences, 12(9), 1128. https://doi.org/10.3390/brainsci12091128






