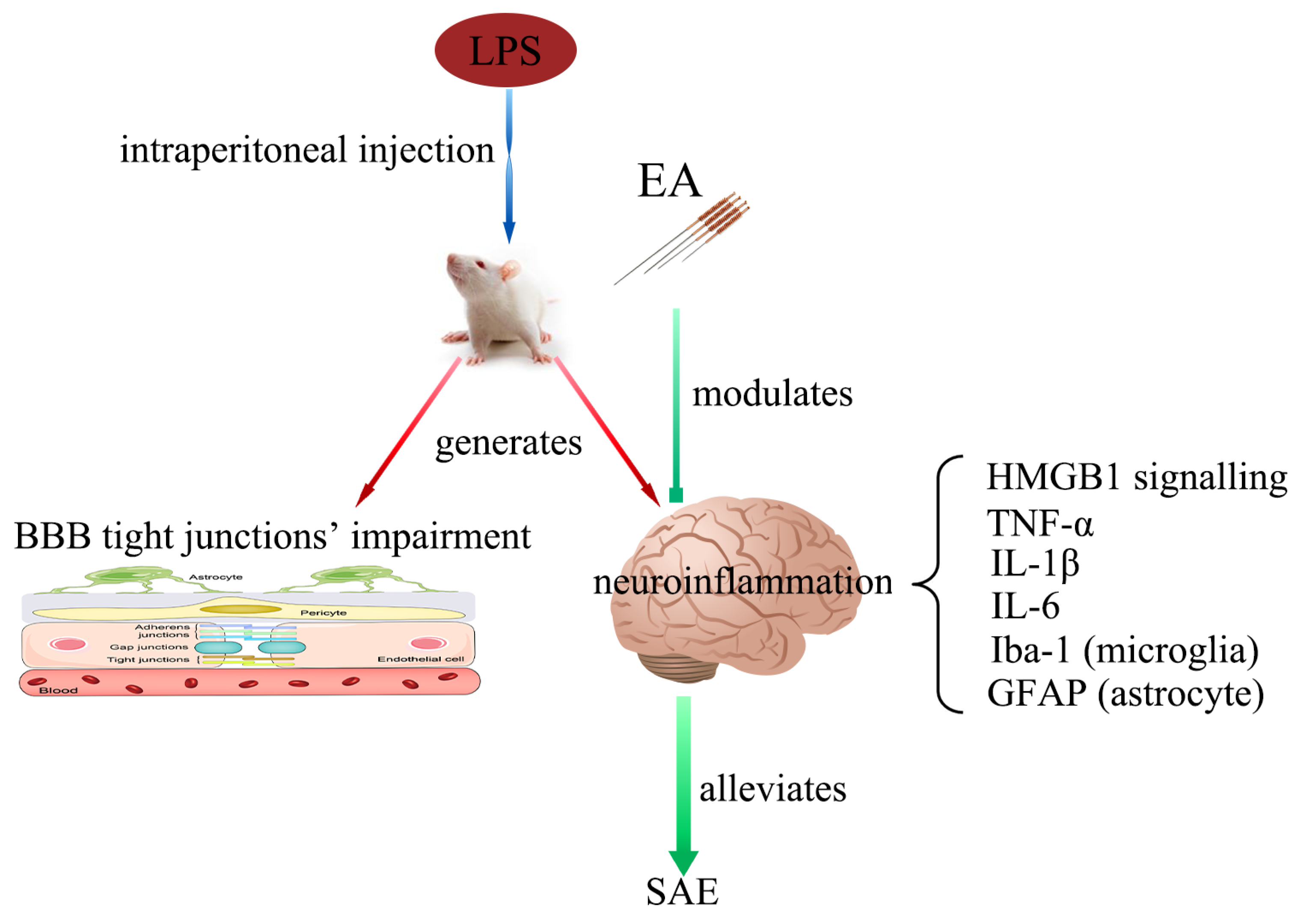
Electroacupuncture Alleviates Neuroinflammation by Inhibiting the HMGB1 Signaling Pathway in Rats with Sepsis-Associated Encephalopathy
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals
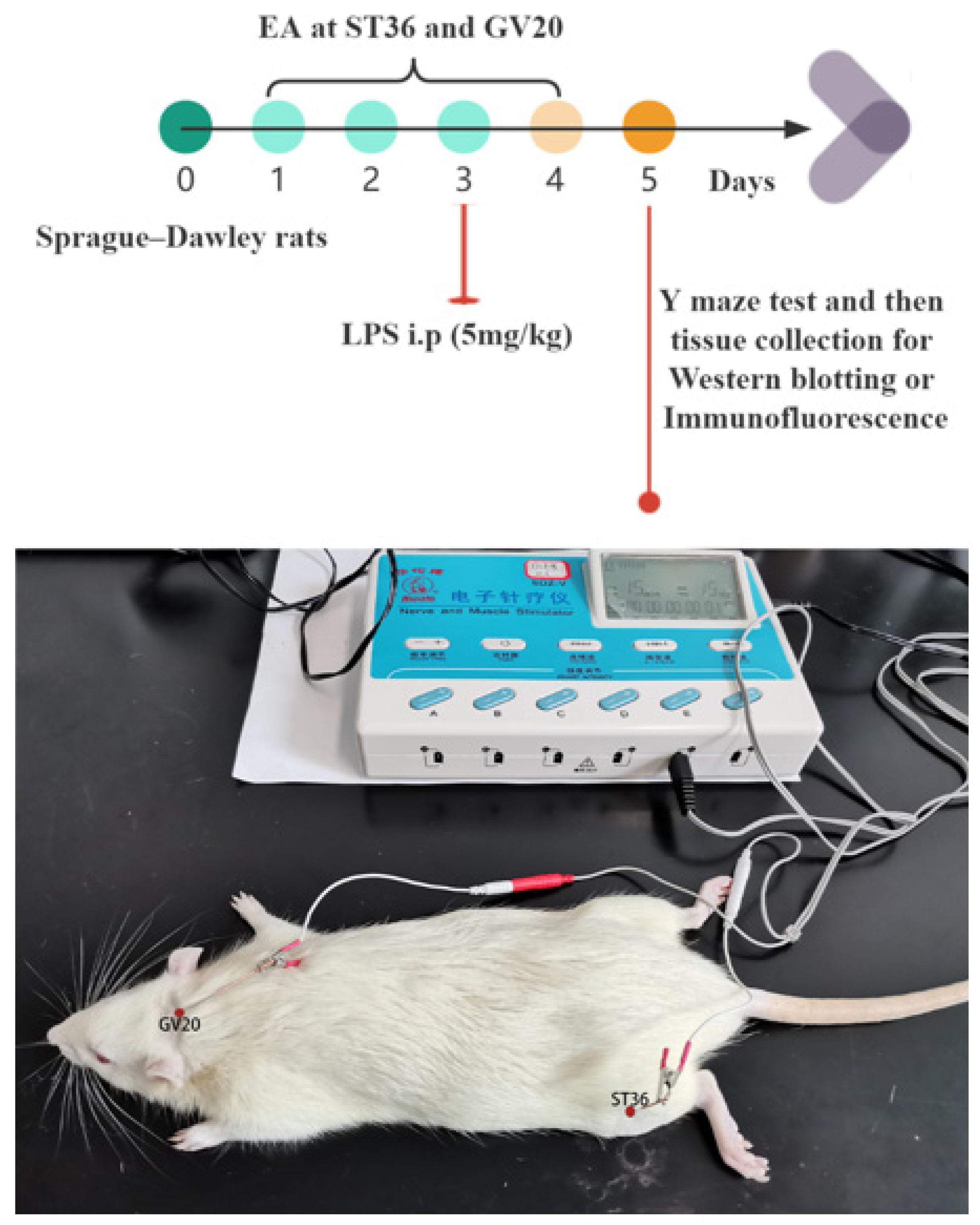
2.2. Electroacupuncture Treatment
2.3. Sepsis-Associated Encephalopathy Model
2.4. Y Maze Test
2.5. Western Blotting
2.6. Immunofluorescence (IF)
2.7. Statistical Analyses
3. Results
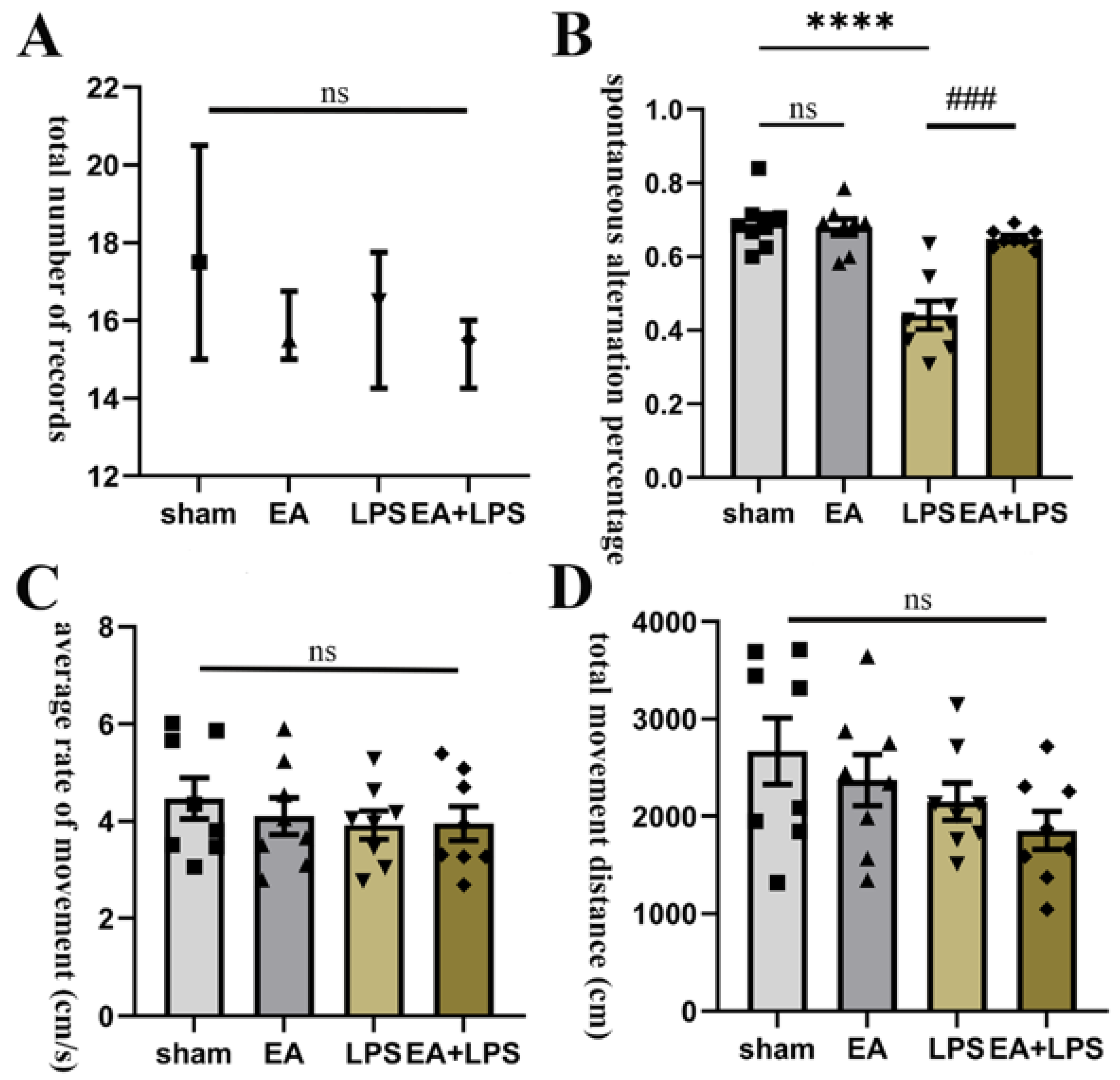
3.1. EA Mitigated Working Memory Impairment Caused by LPS
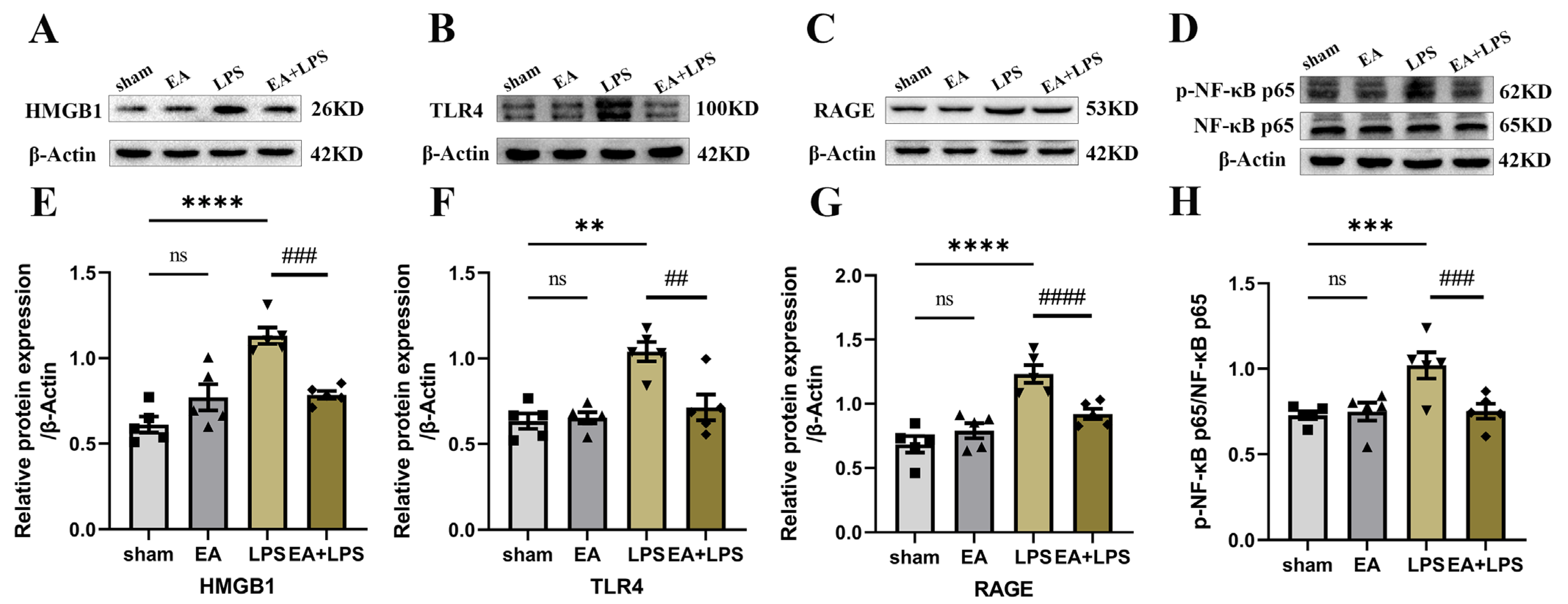
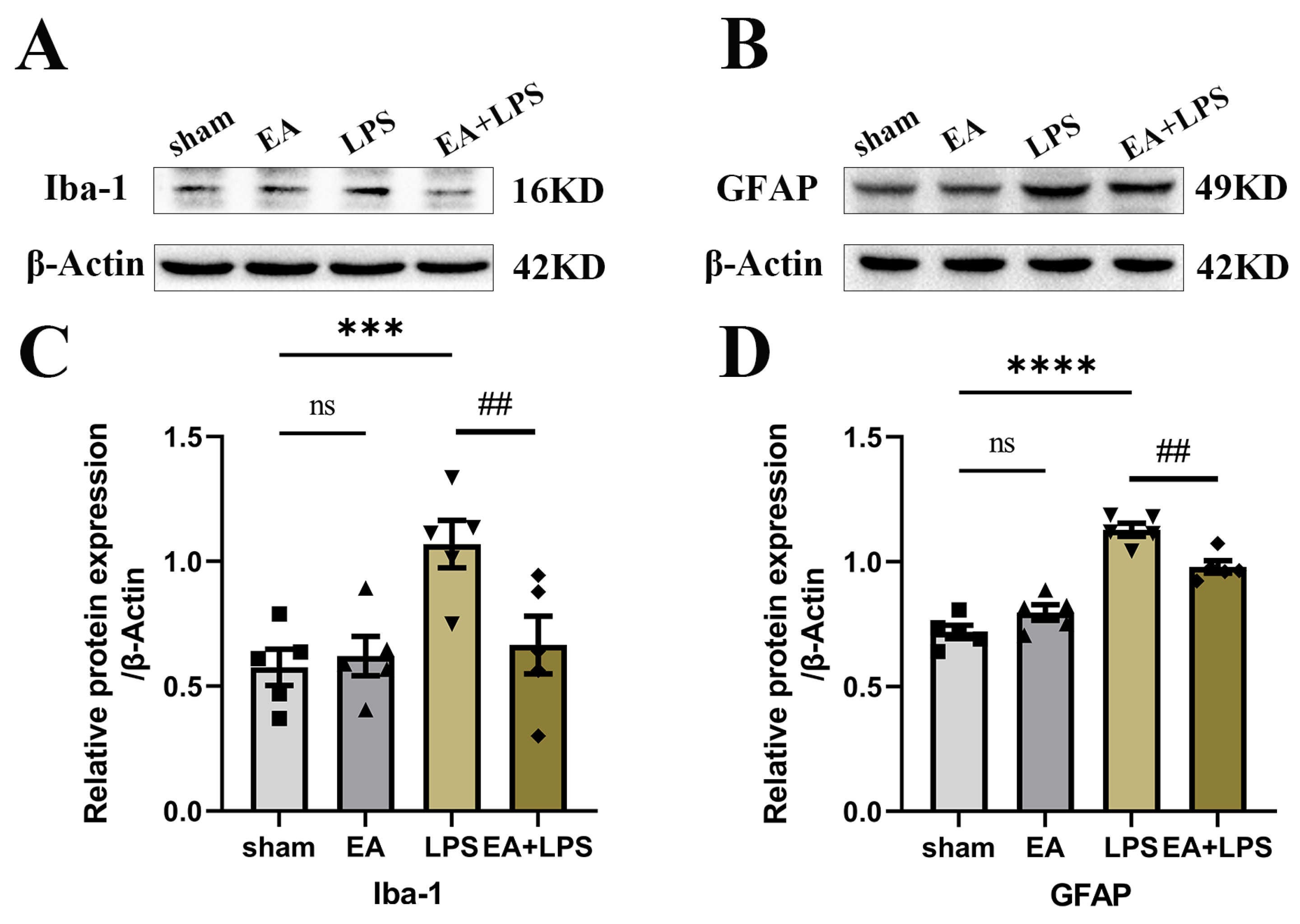
3.2. EA Modulated the HMGB1 Signaling in the Hippocampus
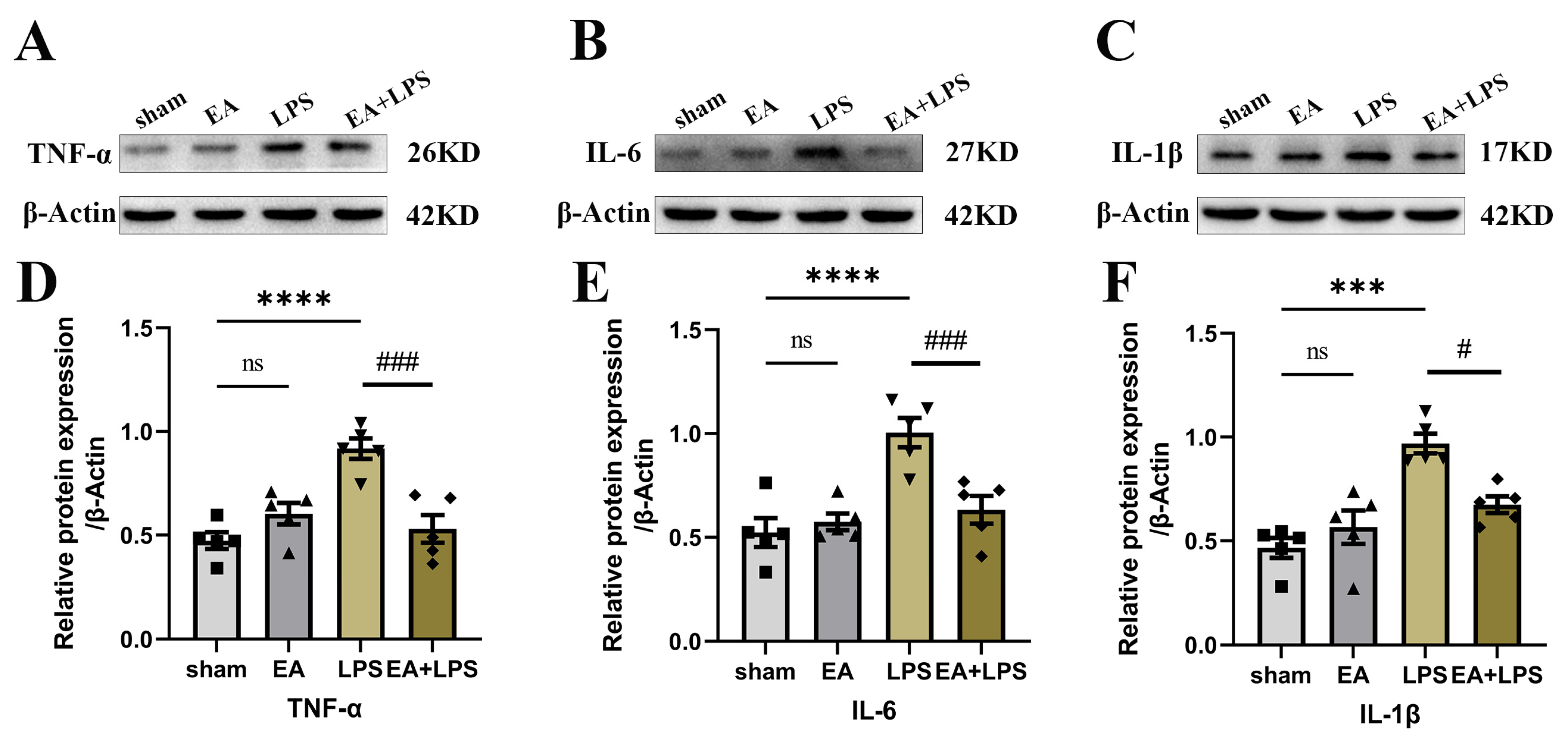
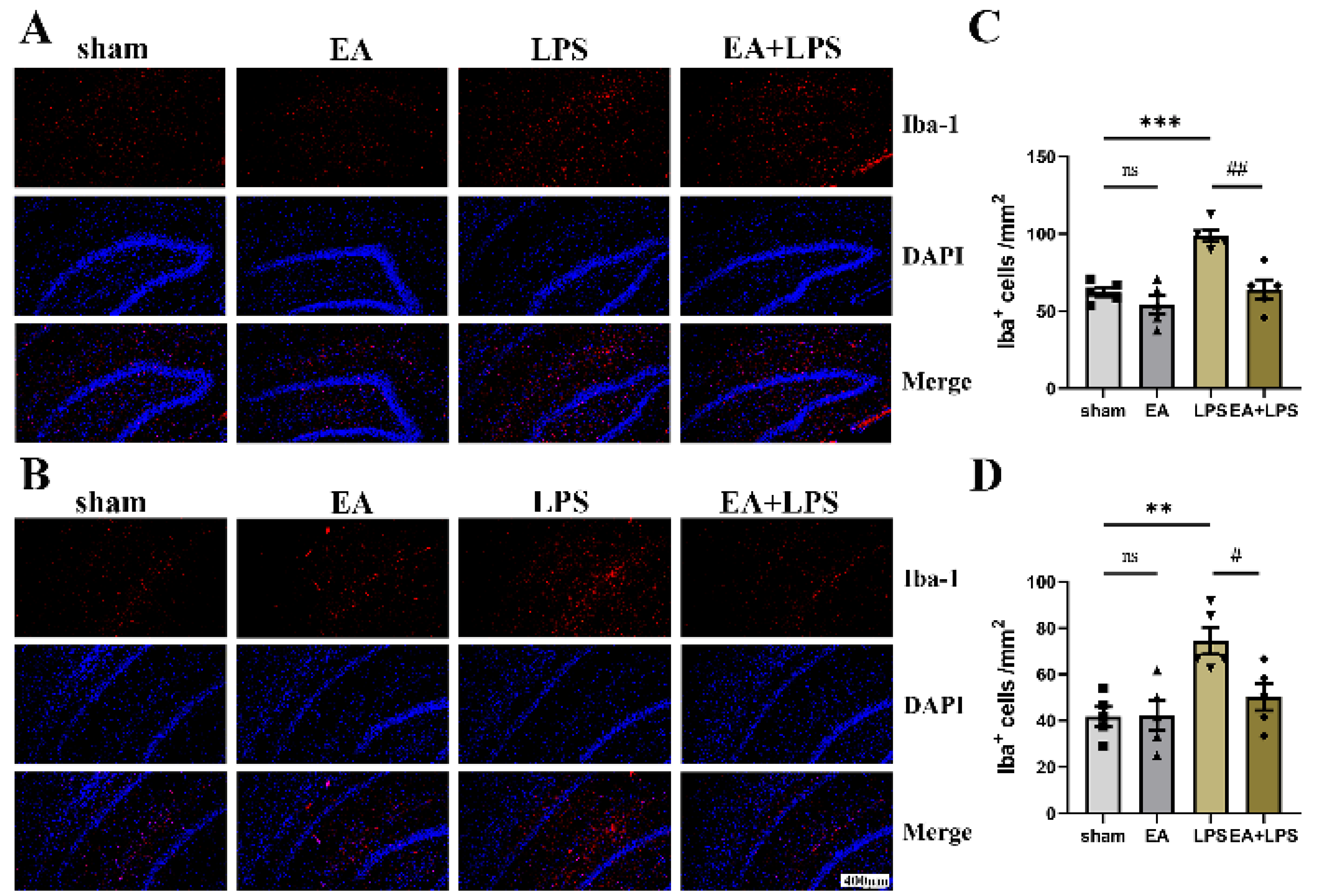
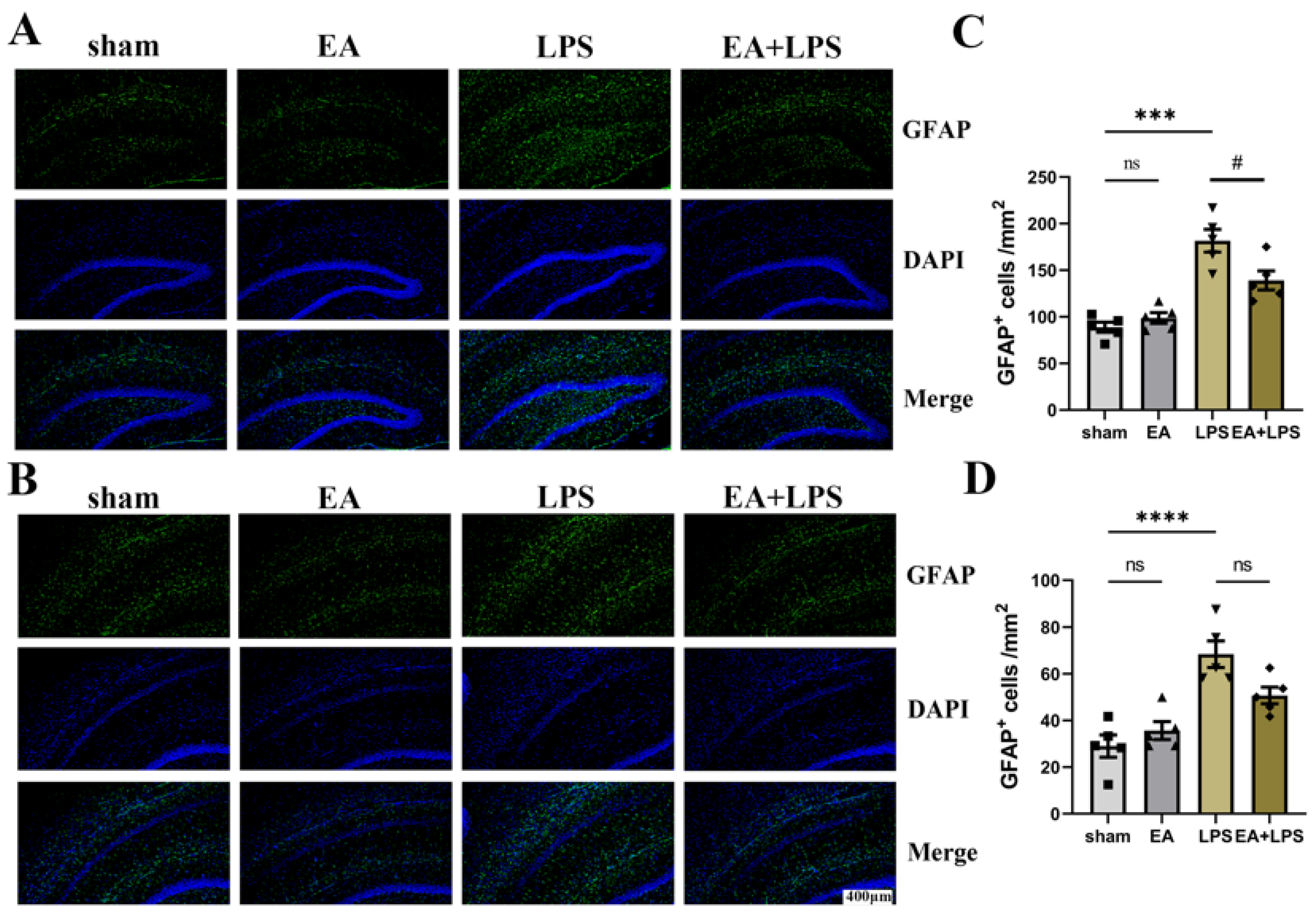
3.3. EA Alleviated Neuroinflammation in the Hippocampus
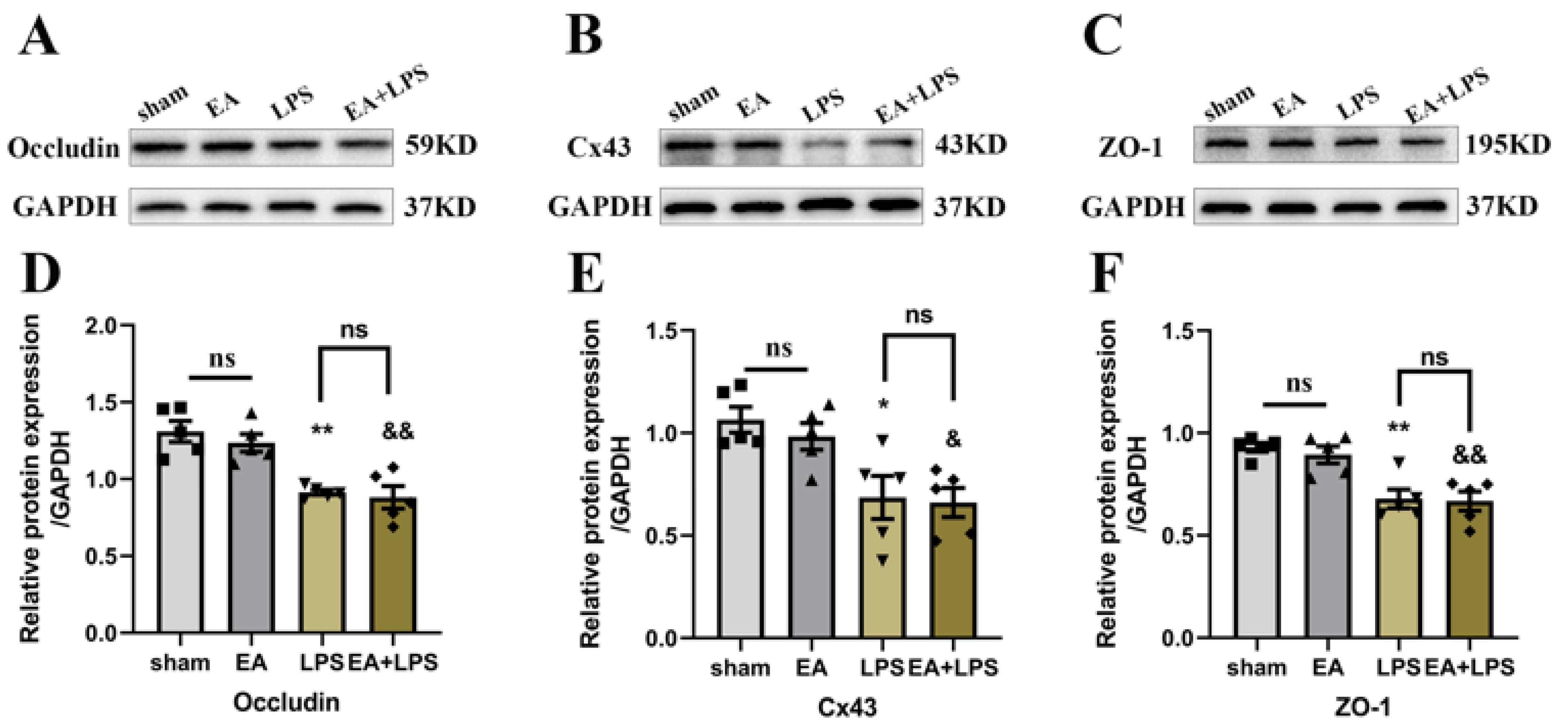
3.4. EA Did Not Affect the Tight Junctions’ Expression of BBB in the Hippocampus
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Singer, M.; Deutschman, C.S.; Seymour, C.W.; Shankar-Hari, M.; Annane, D.; Bauer, M.; Bellomo, R.; Bernard, G.R.; Chiche, J.D.; Coopersmith, C.M.; et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016, 315, 801–810. [Google Scholar] [CrossRef]
- Zhao, L.; An, R.; Yang, Y.; Yang, X.; Liu, H.; Yue, L.; Li, X.; Lin, Y.; Reiter, R.J.; Qu, Y. Melatonin alleviates brain injury in mice subjected to cecal ligation and puncture via attenuating inflammation, apoptosis, and oxidative stress: The role of SIRT1 signaling. J. Pineal Res. 2015, 59, 230–239. [Google Scholar] [CrossRef]
- Tauber, S.C.; Djukic, M.; Gossner, J.; Eiffert, H.; Bruck, W.; Nau, R. Sepsis-associated encephalopathy and septic encephalitis: An update. Expert Rev. Anti-Infect Ther. 2021, 19, 215–231. [Google Scholar] [CrossRef]
- Catarina, A.V.; Branchini, G.; Bettoni, L.; De Oliveira, J.R.; Nunes, F.B. Sepsis-Associated Encephalopathy: From Pathophysiology to Progress in Experimental Studies. Mol. Neurobiol. 2021, 58, 2770–2779. [Google Scholar] [CrossRef]
- Li, Y.; Yin, L.; Fan, Z.; Su, B.; Chen, Y.; Ma, Y.; Zhong, Y.; Hou, W.; Fang, Z.; Zhang, X. Microglia: A Potential Therapeutic Target for Sepsis-Associated Encephalopathy and Sepsis-Associated Chronic Pain. Front. Pharmacol. 2020, 11, 600421. [Google Scholar] [CrossRef]
- DiSabato, D.J.; Quan, N.; Godbout, J.P. Neuroinflammation: The devil is in the details. J. Neurochem. 2016, 139 (Suppl. S2), 136–153. [Google Scholar] [CrossRef] [Green Version]
- Manabe, T.; Heneka, M.T. Cerebral dysfunctions caused by sepsis during ageing. Nat. Rev. Immunol. 2021, 22, 444–458. [Google Scholar] [CrossRef]
- Nishibori, M.; Wang, D.; Ousaka, D.; Wake, H. High Mobility Group Box-1 and Blood-Brain Barrier Disruption. Cells 2020, 9, 2650. [Google Scholar] [CrossRef]
- Sulhan, S.; Lyon, K.A.; Shapiro, L.A.; Huang, J.H. Neuroinflammation and blood-brain barrier disruption following traumatic brain injury: Pathophysiology and potential therapeutic targets. J. Neurosci. Res. 2020, 98, 19–28. [Google Scholar] [CrossRef] [Green Version]
- Hudson, B.I.; Lippman, M.E. Targeting RAGE Signaling in Inflammatory Disease. Annu. Rev. Med. 2018, 69, 349–364. [Google Scholar] [CrossRef]
- Paudel, Y.N.; Shaikh, M.F.; Chakraborti, A.; Kumari, Y.; Aledo-Serrano, A.; Aleksovska, K.; Alvim, M.K.M.; Othman, I. HMGB1: A Common Biomarker and Potential Target for TBI, Neuroinflammation, Epilepsy, and Cognitive Dysfunction. Front. Neurosci. 2018, 12, 628. [Google Scholar] [CrossRef] [Green Version]
- Ni, J.; Zhao, Y.; Su, J.; Liu, Z.; Fang, S.; Li, L.; Deng, J.; Fan, G. Toddalolactone Protects Lipopolysaccharide-Induced Sepsis and Attenuates Lipopolysaccharide-Induced Inflammatory Response by Modulating HMGB1-NF-kappaB Translocation. Front. Pharm. 2020, 11, 109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chavan, S.S.; Huerta, P.T.; Robbiati, S.; Valdes-Ferrer, S.I.; Ochani, M.; Dancho, M.; Frankfurt, M.; Volpe, B.T.; Tracey, K.J.; Diamond, B. HMGB1 mediates cognitive impairment in sepsis survivors. Mol. Med. 2012, 18, 930–937. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.; Yang, J.; Tong, H.; Zhu, C.; Pang, Y. HMGB1 augments cognitive impairment in sepsis-associated encephalopathy by binding to MD-2 and promoting NLRP3-induced neuroinflammation. Psychogeriatrics 2022, 22, 167–179. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Gao, Y.X.; Guo, S.G.; Lu, X.; Yu, S.Y.; Ge, Z.Z.; Zhu, H.D.; Li, Y. Sepsis-Associated Encephalopathy: Insight into Injury and Pathogenesis. CNS Neurol. Disord. Drug Targets 2021, 20, 112–124. [Google Scholar] [CrossRef]
- Acar, H.V. Acupuncture and related techniques during perioperative period: A literature review. Complement. Ther. Med. 2016, 29, 48–55. [Google Scholar] [CrossRef]
- Li, F.; He, T.; Xu, Q.; Lin, L.T.; Li, H.; Liu, Y.; Shi, G.X.; Liu, C.Z. What is the Acupoint? A preliminary review of Acupoints. Pain Med. 2015, 16, 1905–1915. [Google Scholar] [CrossRef] [Green Version]
- Ifrim Chen, F.; Antochi, A.D.; Barbilian, A.G. Acupuncture and the retrospect of its modern research. Rom. J. Morphol. Embryol. 2019, 60, 411–418. [Google Scholar]
- Wang, W.W.; Xie, C.L.; Lu, L.; Zheng, G.Q. A systematic review and meta-analysis of Baihui (GV20)-based scalp acupuncture in experimental ischemic stroke. Sci. Rep. 2014, 4, 3981. [Google Scholar] [CrossRef] [Green Version]
- Wang, M.; Gao, Y.H.; Xu, J.; Chi, Y.; Wei, X.B.; Lewith, G.; Liu, J.P. Zusanli (ST36) acupoint injection for preventing postoperative ileus: A systematic review and meta-analysis of randomized clinical trials. Complement. Ther. Med. 2015, 23, 469–483. [Google Scholar] [CrossRef] [Green Version]
- Lai, F.; Ren, Y.; Lai, C.; Chen, R.; Yin, X.; Tan, C.; Li, J.; Yang, C.; Liang, G.; Li, J.; et al. Acupuncture at Zusanli (ST36) for Experimental Sepsis: A Systematic Review. Evid.-Based Complement. Altern. Med. 2020, 2020, 3620741. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.E.; Kim, S.N. Anti-Inflammatory Effects of Acupuncture at ST36 Point: A Literature Review in Animal Studies. Front. Immunol. 2021, 12, 813748. [Google Scholar] [CrossRef] [PubMed]
- Song, G.; Fiocchi, C.; Achkar, J.P. Acupuncture in Inflammatory Bowel Disease. Inflamm. Bowel Dis. 2019, 25, 1129–1139. [Google Scholar] [CrossRef] [PubMed]
- Xin, Y.Y.; Wang, J.X.; Xu, A.J. Electroacupuncture ameliorates neuroinflammation in animal models. Acupunct. Med. 2022, 40, 474–483. [Google Scholar] [CrossRef] [PubMed]
- Chiu, C.; Legrand, M. Epidemiology of sepsis and septic shock. Curr. Opin. Anaesthesiol. 2021, 34, 71–76. [Google Scholar] [CrossRef]
- Gotts, J.E.; Matthay, M.A. Sepsis: Pathophysiology and clinical management. BMJ 2016, 353, i1585. [Google Scholar] [CrossRef] [Green Version]
- Guidet, B.; Maury, E. Sex and severe sepsis. Crit. Care 2013, 17, 144. [Google Scholar] [CrossRef] [Green Version]
- Zhang, M.Q.; Macala, K.F.; Fox-Robichaud, A.; Mendelson, A.A.; Lalu, M.M.; Sepsis Canada National Preclinical Sepsis Platform. Sex- and Gender-Dependent Differences in Clinical and Preclinical Sepsis. Shock 2021, 56, 178–187. [Google Scholar] [CrossRef]
- Yin, C.S.; Jeong, H.S.; Park, H.J.; Baik, Y.; Yoon, M.H.; Choi, C.B.; Koh, H.G. A proposed transpositional acupoint system in a mouse and rat model. Res. Vet. Sci. 2008, 84, 159–165. [Google Scholar] [CrossRef]
- Xu, H.; Zhang, Y.; Sun, H.; Chen, S.; Wang, F. Effects of acupuncture at GV20 and ST36 on the expression of matrix metalloproteinase 2, aquaporin 4, and aquaporin 9 in rats subjected to cerebral ischemia/reperfusion injury. PLoS ONE 2014, 9, e97488. [Google Scholar] [CrossRef]
- Sha, R.; Zhang, B.; Han, X.; Peng, J.; Zheng, C.; Zhang, F.; Huang, X. Electroacupuncture Alleviates Ischemic Brain Injury by Inhibiting the miR-223/NLRP3 Pathway. Med. Sci. Monit. 2019, 25, 4723–4733. [Google Scholar] [CrossRef] [PubMed]
- Savran, M.; Ozmen, O.; Erzurumlu, Y.; Savas, H.B.; Asci, S.; Kaynak, M. The Impact of Prophylactic Lacosamide on LPS-Induced Neuroinflammation in Aged Rats. Inflammation 2019, 42, 1913–1924. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Pei, L.; Liang, Z. mRNA and Long Non-coding RNA Expression Profiles in Rats Reveal Inflammatory Features in Sepsis-Associated Encephalopathy. Neurochem. Res. 2017, 42, 3199–3219. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi, D.S.; Campos, A.C.P.; Qu, H.; Forrester, S.J.; Pagano, R.L.; Lassegue, B.; Sadikot, R.T.; Griendling, K.K.; Hernandes, M.S. Poldip2 mediates blood-brain barrier disruption in a model of sepsis-associated encephalopathy. J. Neuroinflamm. 2019, 16, 241. [Google Scholar] [CrossRef]
- Nelson, R.J.; Bumgarner, J.R.; Walker, W.H., 2nd; DeVries, A.C. Time-of-day as a critical biological variable. Neurosci. Biobehav. Rev. 2021, 127, 740–746. [Google Scholar] [CrossRef]
- Song, C.; Zhang, Y.; Cheng, L.; Shi, M.; Li, X.; Zhang, L.; Zhao, H. Tea polyphenols ameliorates memory decline in aging model rats by inhibiting brain TLR4/NF-kappaB inflammatory signaling pathway caused by intestinal flora dysbiosis. Exp. Gerontol. 2021, 153, 111476. [Google Scholar] [CrossRef]
- Paxinos, G.; Watson, C. The Rat Brain in Stereotaxic Coordinates, 6th ed.; Academic Press; Elsevier: Amsterdam, The Netherlands; Boston, MA, USA, 2007. [Google Scholar]
- Godinho-Pereira, J.; Garcia, A.R.; Figueira, I.; Malho, R.; Brito, M.A. Behind Brain Metastases Formation: Cellular and Molecular Alterations and Blood-Brain Barrier Disruption. Int. J. Mol. Sci. 2021, 22, 7057. [Google Scholar] [CrossRef]
- Yuan, S.; Liu, K.J.; Qi, Z. Occludin regulation of blood-brain barrier and potential therapeutic target in ischemic stroke. Brain Circ. 2020, 6, 152–162. [Google Scholar] [CrossRef]
- Ekstrom, A.D.; Ranganath, C. Space, time, and episodic memory: The hippocampus is all over the cognitive map. Hippocampus 2018, 28, 680–687. [Google Scholar] [CrossRef]
- Ito, H.; Hosomi, S.; Koyama, Y.; Matsumoto, H.; Imamura, Y.; Ogura, H.; Oda, J. Sepsis-Associated Encephalopathy: A Mini-Review of Inflammation in the Brain and Body. Front. Aging Neurosci. 2022, 14, 912866. [Google Scholar] [CrossRef]
- Orhun, G.; Tuzun, E.; Bilgic, B.; Ergin Ozcan, P.; Sencer, S.; Barburoglu, M.; Esen, F. Brain Volume Changes in Patients with Acute Brain Dysfunction Due to Sepsis. Neurocrit. Care 2020, 32, 459–468. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Zhao, Z.; Lu, L.; Liu, J.; Sun, J.; Wu, X.; Dong, J. Icariin and icaritin ameliorated hippocampus neuroinflammation via inhibiting HMGB1-related pro-inflammatory signals in lipopolysaccharide-induced inflammation model in C57BL/6J mice. Int. Immunopharmacol. 2019, 68, 95–105. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, K.A.; Hansen, M.K.; Rachal Pugh, C.; Deak, M.M.; Biedenkapp, J.C.; Milligan, E.D.; Johnson, J.D.; Wang, H.; Maier, S.F.; Tracey, K.J.; et al. Further characterization of high mobility group box 1 (HMGB1) as a proinflammatory cytokine: Central nervous system effects. Cytokine 2003, 24, 254–265. [Google Scholar] [CrossRef] [PubMed]
- Andersson, U.; Yang, H.; Harris, H. Extracellular HMGB1 as a therapeutic target in inflammatory diseases. Expert Opin. Ther. Targets 2018, 22, 263–277. [Google Scholar] [CrossRef]
- Wang, H.; Bloom, O.; Zhang, M.; Vishnubhakat, J.M.; Ombrellino, M.; Che, J.; Frazier, A.; Yang, H.; Ivanova, S.; Borovikova, L.; et al. HMG-1 as a late mediator of endotoxin lethality in mice. Science 1999, 285, 248–251. [Google Scholar] [CrossRef]
- Yang, H.; Wang, H.; Chavan, S.S.; Andersson, U. High Mobility Group Box Protein 1 (HMGB1): The Prototypical Endogenous Danger Molecule. Mol. Med. 2015, 21 (Suppl. S1), S6–S12. [Google Scholar] [CrossRef]
- Zusso, M.; Lunardi, V.; Franceschini, D.; Pagetta, A.; Lo, R.; Stifani, S.; Frigo, A.C.; Giusti, P.; Moro, S. Ciprofloxacin and levofloxacin attenuate microglia inflammatory response via TLR4/NF-kB pathway. J. Neuroinflamm. 2019, 16, 148. [Google Scholar] [CrossRef] [Green Version]
- Paudel, Y.N.; Angelopoulou, E.; Piperi, C.; Balasubramaniam, V.; Othman, I.; Shaikh, M.F. Enlightening the role of high mobility group box 1 (HMGB1) in inflammation: Updates on receptor signalling. Eur. J. Pharmacol. 2019, 858, 172487. [Google Scholar] [CrossRef]
- Paudel, Y.N.; Angelopoulou, E.; Piperi, C.; Othman, I.; Aamir, K.; Shaikh, M.F. Impact of HMGB1, RAGE, and TLR4 in Alzheimer’s Disease (AD): From Risk Factors to Therapeutic Targeting. Cells 2020, 9, 383. [Google Scholar] [CrossRef] [Green Version]
- Yang, H.; Wang, H.; Andersson, U. Targeting Inflammation Driven by HMGB1. Front. Immunol. 2020, 11, 484. [Google Scholar] [CrossRef] [Green Version]
- Ray, R.; Juranek, J.K.; Rai, V. RAGE axis in neuroinflammation, neurodegeneration and its emerging role in the pathogenesis of amyotrophic lateral sclerosis. Neurosci. Biobehav. Rev. 2016, 62, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.F.; Yao, Y.M.; Sheng, Z.Y. Novel insights for high mobility group box 1 protein-mediated cellular immune response in sepsis: A systemic review. World J. Emerg. Med. 2012, 3, 165–171. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cho, W.; Koo, J.Y.; Park, Y.; Oh, K.; Lee, S.; Song, J.S.; Bae, M.A.; Lim, D.; Lee, D.S.; Park, S.B. Treatment of Sepsis Pathogenesis with High Mobility Group Box Protein 1-Regulating Anti-inflammatory Agents. J. Med. Chem. 2017, 60, 170–179. [Google Scholar] [CrossRef] [PubMed]
- Ren, C.; Tong, Y.L.; Li, J.C.; Dong, N.; Hao, J.W.; Zhang, Q.H.; Yao, Y.M. Early antagonism of cerebral high mobility group box-1 protein is benefit for sepsis induced brain injury. Oncotarget 2017, 8, 92578–92588. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Terrando, N.; Yang, T.; Wang, X.; Fang, J.; Cao, M.; Andersson, U.; Erlandsson, H.H.; Ouyang, W.; Tong, J. Systemic HMGB1 Neutralization Prevents Postoperative Neurocognitive Dysfunction in Aged Rats. Front. Immunol. 2016, 7, 441. [Google Scholar] [CrossRef] [Green Version]
- Yu, M.; Huang, H.; Dong, S.; Sha, H.; Wei, W.; Liu, C. High mobility group box-1 mediates hippocampal inflammation and contributes to cognitive deficits in high-fat high-fructose diet-induced obese rats. Brain Behav. Immun. 2019, 82, 167–177. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Mishra, K.P.; Chanda, S.; Ganju, L.; Singh, S.B. CXCR7: A key neuroprotective molecule against alarmin HMGB1 mediated CNS pathophysiology and subsequent memory impairment. Brain Behav. Immun. 2019, 82, 319–337. [Google Scholar] [CrossRef]
- Li, Y.; Liu, T.; Li, Y.; Han, D.; Hong, J.; Yang, N.; He, J.; Peng, R.; Mi, X.; Kuang, C.; et al. Baicalin Ameliorates Cognitive Impairment and Protects Microglia from LPS-Induced Neuroinflammation via the SIRT1/HMGB1 Pathway. Oxid. Med. Cell Longev. 2020, 2020, 4751349. [Google Scholar] [CrossRef]
- Chen, X.; Wu, S.; Chen, C.; Xie, B.; Fang, Z.; Hu, W.; Chen, J.; Fu, H.; He, H. Omega-3 polyunsaturated fatty acid supplementation attenuates microglial-induced inflammation by inhibiting the HMGB1/TLR4/NF-kappaB pathway following experimental traumatic brain injury. J. Neuroinflamm. 2017, 14, 143. [Google Scholar] [CrossRef]
- Xu, X.; Lu, Y.N.; Cheng, J.H.; Lan, H.W.; Lu, J.M.; Jin, G.N.; Xu, G.H.; Jin, C.H.; Ma, J.; Piao, H.N.; et al. Ginsenoside Rh2 reduces depression in offspring of mice with maternal toxoplasma infection during pregnancy by inhibiting microglial activation via the HMGB1/TLR4/NF-kappaB signaling pathway. J. Ginseng. Res. 2022, 46, 62–70. [Google Scholar] [CrossRef]
- Xu, X.; Piao, H.N.; Aosai, F.; Zeng, X.Y.; Cheng, J.H.; Cui, Y.X.; Li, J.; Ma, J.; Piao, H.R.; Jin, X.; et al. Arctigenin protects against depression by inhibiting microglial activation and neuroinflammation via HMGB1/TLR4/NF-kappaB and TNF-alpha/TNFR1/NF-kappaB pathways. Br. J. Pharmacol. 2020, 177, 5224–5245. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Xu, H.; Cavagnaro, M.J.; Li, X.; Fang, J. Blocking HMGB1/RAGE Signaling by Berberine Alleviates A1 Astrocyte and Attenuates Sepsis-Associated Encephalopathy. Front. Pharmacol. 2021, 12, 760186. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.Y.; Fang, M.; Zhu, G.F.; Zhou, Y.; Zeng, H.K. Role of microglia in the pathogenesis of sepsis-associated encephalopathy. CNS Neurol. Disord. Drug Targets 2013, 12, 720–725. [Google Scholar] [CrossRef] [PubMed]
- Moraes, C.A.; Zaverucha-do-Valle, C.; Fleurance, R.; Sharshar, T.; Bozza, F.A.; d’Avila, J.C. Neuroinflammation in Sepsis: Molecular Pathways of Microglia Activation. Pharmaceuticals 2021, 14, 416. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Lin, F.; Ren, M.; Liu, X.; Xie, W.; Zhang, A.; Qian, M.; Mo, Y.; Wang, J.; Lv, Y. The PICK1/TLR4 complex on microglia is involved in the regulation of LPS-induced sepsis-associated encephalopathy. Int. Immunopharmacol. 2021, 100, 108116. [Google Scholar] [CrossRef]
- Cui, W.; Chen, J.; Yu, F.; Liu, W.; He, M. GYY4137 protected the integrity of the blood-brain barrier via activation of the Nrf2/ARE pathway in mice with sepsis. FASEB J. 2021, 35, e21710. [Google Scholar] [CrossRef]
- Huang, C.T.; Lue, J.H.; Cheng, T.H.; Tsai, Y.J. Glycemic control with insulin attenuates sepsis-associated encephalopathy by inhibiting glial activation via the suppression of the nuclear factor kappa B and mitogen-activated protein kinase signaling pathways in septic rats. Brain Res. 2020, 1738, 146822. [Google Scholar] [CrossRef]
- Zhu, T.; Zhao, Y.; Hu, H.; Zheng, Q.; Luo, X.; Ling, Y.; Ying, Y.; Shen, Z.; Jiang, P.; Shu, Q. TRPM2 channel regulates cytokines production in astrocytes and aggravates brain disorder during lipopolysaccharide-induced endotoxin sepsis. Int. Immunopharmacol. 2019, 75, 105836. [Google Scholar] [CrossRef]
- Hu, J.; Cheng, Y.; Chen, P.; Huang, Z.; Yang, L. Caffeine Citrate Protects Against Sepsis-Associated Encephalopathy and Inhibits the UCP2/NLRP3 Axis in Astrocytes. J. Interferon. Cytokine Res. 2022, 42, 267–278. [Google Scholar] [CrossRef]
- Shulyatnikova, T.; Verkhratsky, A. Astroglia in Sepsis Associated Encephalopathy. Neurochem. Res. 2020, 45, 83–99. [Google Scholar] [CrossRef]
- Stefanini, F.; Kushnir, L.; Jimenez, J.C.; Jennings, J.H.; Woods, N.I.; Stuber, G.D.; Kheirbek, M.A.; Hen, R.; Fusi, S. A Distributed Neural Code in the Dentate Gyrus and in CA1. Neuron 2020, 107, 703–716.e4. [Google Scholar] [CrossRef] [PubMed]
- Duncan, K.; Ketz, N.; Inati, S.J.; Davachi, L. Evidence for area CA1 as a match/mismatch detector: A high-resolution fMRI study of the human hippocampus. Hippocampus 2012, 22, 389–398. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schlichting, M.L.; Zeithamova, D.; Preston, A.R. CA1 subfield contributions to memory integration and inference. Hippocampus 2014, 24, 1248–1260. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chung, H.Y.; Wickel, J.; Brunkhorst, F.M.; Geis, C. Sepsis-Associated Encephalopathy: From Delirium to Dementia? J. Clin. Med. 2020, 9, 703. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuperberg, S.J.; Wadgaonkar, R. Sepsis-Associated Encephalopathy: The Blood-Brain Barrier and the Sphingolipid Rheostat. Front. Immunol. 2017, 8, 597. [Google Scholar] [CrossRef] [Green Version]
- Obermeier, B.; Daneman, R.; Ransohoff, R.M. Development, maintenance and disruption of the blood-brain barrier. Nat. Med. 2013, 19, 1584–1596. [Google Scholar] [CrossRef] [Green Version]
- Ren, C.; Yao, R.Q.; Zhang, H.; Feng, Y.W.; Yao, Y.M. Sepsis-associated encephalopathy: A vicious cycle of immunosuppression. J. Neuroinflamm. 2020, 17, 14. [Google Scholar] [CrossRef]
- Molnar, L.; Fulesdi, B.; Nemeth, N.; Molnar, C. Sepsis-associated encephalopathy: A review of literature. Neurol. India 2018, 66, 352–361. [Google Scholar] [CrossRef] [Green Version]
- Sadraie, S.; Kiasalari, Z.; Razavian, M.; Azimi, S.; Sedighnejad, L.; Afshin-Majd, S.; Baluchnejadmojarad, T.; Roghani, M. Berberine ameliorates lipopolysaccharide-induced learning and memory deficit in the rat: Insights into underlying molecular mechanisms. Metab. Brain Dis. 2019, 34, 245–255. [Google Scholar] [CrossRef] [PubMed]
- Shu, H.; Wang, M.; Song, M.; Sun, Y.; Shen, X.; Zhang, J.; Jin, X. Acute Nicotine Treatment Alleviates LPS-Induced Impairment of Fear Memory Reconsolidation Through AMPK Activation and CRTC1 Upregulation in Hippocampus. Int. J. Neuropsychopharmacol. 2020, 23, 687–699. [Google Scholar] [CrossRef]
- Marefati, N.; Beheshti, F.; Vafaee, F.; Barabadi, M.; Hosseini, M. The Effects of Incensole Acetate on Neuro-inflammation, Brain-Derived Neurotrophic Factor and Memory Impairment Induced by Lipopolysaccharide in Rats. Neurochem. Res. 2021, 46, 2473–2484. [Google Scholar] [CrossRef] [PubMed]
- Gu, M.; Mei, X.L.; Zhao, Y.N. Sepsis and Cerebral Dysfunction: BBB Damage, Neuroinflammation, Oxidative Stress, Apoptosis and Autophagy as Key Mediators and the Potential Therapeutic Approaches. Neurotox. Res. 2021, 39, 489–503. [Google Scholar] [CrossRef] [PubMed]
- Trapero, I.; Cauli, O. Interleukin 6 and cognitive dysfunction. Metab. Brain Dis. 2014, 29, 593–608. [Google Scholar] [CrossRef] [PubMed]
- Pan, W.; Stone, K.P.; Hsuchou, H.; Manda, V.K.; Zhang, Y.; Kastin, A.J. Cytokine signaling modulates blood-brain barrier function. Curr. Pharm. Des. 2011, 17, 3729–3740. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tian, M.; Wang, W.; Wang, K.; Jin, P.; Lenahan, C.; Wang, Y.; Tan, J.; Wen, H.; Deng, S.; Zhao, F.; et al. Dexmedetomidine alleviates cognitive impairment by reducing blood-brain barrier interruption and neuroinflammation via regulating Th1/Th2/Th17 polarization in an experimental sepsis model of mice. Int. Immunopharmacol. 2021, 101, 108332. [Google Scholar] [CrossRef]
- Jiang, Y.; Zhang, K.; Yu, Y.; Wang, Y.; Lian, N.; Xie, K.; Yu, Y. Molecular hydrogen alleviates brain injury and cognitive impairment in a chronic sequelae model of murine polymicrobial sepsis. Exp. Brain Res. 2020, 238, 2897–2908. [Google Scholar] [CrossRef]
- Ismail Hassan, F.; Didari, T.; Baeeri, M.; Gholami, M.; Haghi-Aminjan, H.; Khalid, M.; Navaei-Nigjeh, M.; Rahimifard, M.; Solgi, S.; Abdollahi, M.; et al. Metformin Attenuates Brain Injury by Inhibiting Inflammation and Regulating Tight Junction Proteins in Septic Rats. Cell J. 2020, 22, 29–37. [Google Scholar] [CrossRef]
- Yu, Y.; Feng, J.; Lian, N.; Yang, M.; Xie, K.; Wang, G.; Wang, C.; Yu, Y. Hydrogen gas alleviates blood-brain barrier impairment and cognitive dysfunction of septic mice in an Nrf2-dependent pathway. Int. Immunopharmacol. 2020, 85, 106585. [Google Scholar] [CrossRef]
- Li, H.Q.; Li, Y.; Chen, Z.X.; Zhang, X.G.; Zheng, X.W.; Yang, W.T.; Chen, S.; Zheng, G.Q. Electroacupuncture Exerts Neuroprotection through Caveolin-1 Mediated Molecular Pathway in Intracerebral Hemorrhage of Rats. Neural Plast. 2016, 2016, 7308261. [Google Scholar] [CrossRef] [Green Version]
- Zou, R.; Wu, Z.; Cui, S. Electroacupuncture pretreatment attenuates blood-brain barrier disruption following cerebral ischemia/reperfusion. Mol. Med. Rep. 2015, 12, 2027–2034. [Google Scholar] [CrossRef] [Green Version]
- Chen, Y.; Lei, Y.; Mo, L.Q.; Li, J.; Wang, M.H.; Wei, J.C.; Zhou, J. Electroacupuncture pretreatment with different waveforms prevents brain injury in rats subjected to cecal ligation and puncture via inhibiting microglial activation, and attenuating inflammation, oxidative stress and apoptosis. Brain Res. Bull. 2016, 127, 248–259. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Gong, P.; Zhang, J.; Mao, X.; Zhao, Y.; Wang, H.; Gan, L.; Lin, X. Specific Frequency Electroacupuncture Stimulation Transiently Enhances the Permeability of the Blood-Brain Barrier and Induces Tight Junction Changes. Front. Neurosci. 2020, 14, 582324. [Google Scholar] [CrossRef] [PubMed]
- Pan, W.X.; Fan, A.Y.; Chen, S.; Alemi, S.F. Acupuncture modulates immunity in sepsis: Toward a science-based protocol. Auton. Neurosci. 2021, 232, 102793. [Google Scholar] [CrossRef]
- Li, C.; Yu, T.Y.; Zhang, Y.; Wei, L.P.; Dong, S.A.; Shi, J.; Du, S.H.; Yu, J.B. Electroacupuncture Improves Cognition in Rats With Sepsis-Associated Encephalopathy. J. Surg. Res. 2020, 256, 258–266. [Google Scholar] [CrossRef]
- Meng, J.B.; Jiao, Y.N.; Zhang, G.; Xu, X.J.; Ji, C.L.; Hu, M.H.; Lai, Z.Z.; Zhang, M. Electroacupuncture Improves Intestinal Dysfunction in Septic Patients: A Randomised Controlled Trial. Biomed. Res. Int. 2018, 2018, 8293594. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Han, Y.G.; Qin, X.; Zhang, T.; Lei, M.; Sun, F.Y.; Sun, J.J.; Yuan, W.F. Electroacupuncture prevents cognitive impairment induced by lipopolysaccharide via inhibition of oxidative stress and neuroinflammation. Neurosci. Lett. 2018, 683, 190–195. [Google Scholar] [CrossRef] [PubMed]
- Zhan, J.; Qin, W.; Zhang, Y.; Jiang, J.; Ma, H.; Li, Q.; Luo, Y. Upregulation of neuronal zinc finger protein A20 expression is required for electroacupuncture to attenuate the cerebral inflammatory injury mediated by the nuclear factor-kB signaling pathway in cerebral ischemia/reperfusion rats. J. Neuroinflamm. 2016, 13, 258. [Google Scholar] [CrossRef] [Green Version]
- Jiang, T.; Wu, M.; Zhang, Z.; Yan, C.; Ma, Z.; He, S.; Yuan, W.; Pu, K.; Wang, Q. Electroacupuncture attenuated cerebral ischemic injury and neuroinflammation through alpha7nAChR-mediated inhibition of NLRP3 inflammasome in stroke rats. Mol. Med. 2019, 25, 22. [Google Scholar] [CrossRef] [Green Version]
- Huang, J.; You, X.; Liu, W.; Song, C.; Lin, X.; Zhang, X.; Tao, J.; Chen, L. Electroacupuncture ameliorating post-stroke cognitive impairments via inhibition of peri-infarct astroglial and microglial/macrophage P2 purinoceptors-mediated neuroinflammation and hyperplasia. BMC Complement. Altern. Med. 2017, 17, 480. [Google Scholar] [CrossRef] [Green Version]
- Zhang, K.; Liu, R.; Gao, Y.; Ma, W.; Shen, W. Electroacupuncture Relieves LPS-Induced Depression-Like Behaviour in Rats Through IDO-Mediated Tryptophan-Degrading Pathway. Neuropsychiatr. Dis. Treat. 2020, 16, 2257–2266. [Google Scholar] [CrossRef]
- Liu, P.R.; Zhou, Y.; Zhang, Y.; Diao, S. Electroacupuncture alleviates surgery-induced cognitive dysfunction by increasing alpha7-nAChR expression and inhibiting inflammatory pathway in aged rats. Neurosci. Lett. 2017, 659, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Hou, Z.; Qiu, R.; Wei, Q.; Liu, Y.; Wang, M.; Mei, T.; Zhang, Y.; Song, L.; Shao, X.; Shang, H.; et al. Electroacupuncture Improves Cognitive Function in Senescence-Accelerated P8 (SAMP8) Mice via the NLRP3/Caspase-1 Pathway. Neural Plast. 2020, 2020, 8853720. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xin, Y.; Wang, J.; Chu, T.; Zhou, Y.; Liu, C.; Xu, A. Electroacupuncture Alleviates Neuroinflammation by Inhibiting the HMGB1 Signaling Pathway in Rats with Sepsis-Associated Encephalopathy. Brain Sci. 2022, 12, 1732. https://doi.org/10.3390/brainsci12121732
Xin Y, Wang J, Chu T, Zhou Y, Liu C, Xu A. Electroacupuncture Alleviates Neuroinflammation by Inhibiting the HMGB1 Signaling Pathway in Rats with Sepsis-Associated Encephalopathy. Brain Sciences. 2022; 12(12):1732. https://doi.org/10.3390/brainsci12121732
Chicago/Turabian StyleXin, Yueyang, Jinxu Wang, Tiantian Chu, Yaqun Zhou, Cheng Liu, and Aijun Xu. 2022. "Electroacupuncture Alleviates Neuroinflammation by Inhibiting the HMGB1 Signaling Pathway in Rats with Sepsis-Associated Encephalopathy" Brain Sciences 12, no. 12: 1732. https://doi.org/10.3390/brainsci12121732





