Glymphatic Dysfunction Mediates the Influence of White Matter Hyperintensities on Episodic Memory in Cerebral Small Vessel Disease
Abstract
:1. Introduction
2. Methods
2.1. Participants
2.2. MRI Scanning
2.3. MRI Analysis
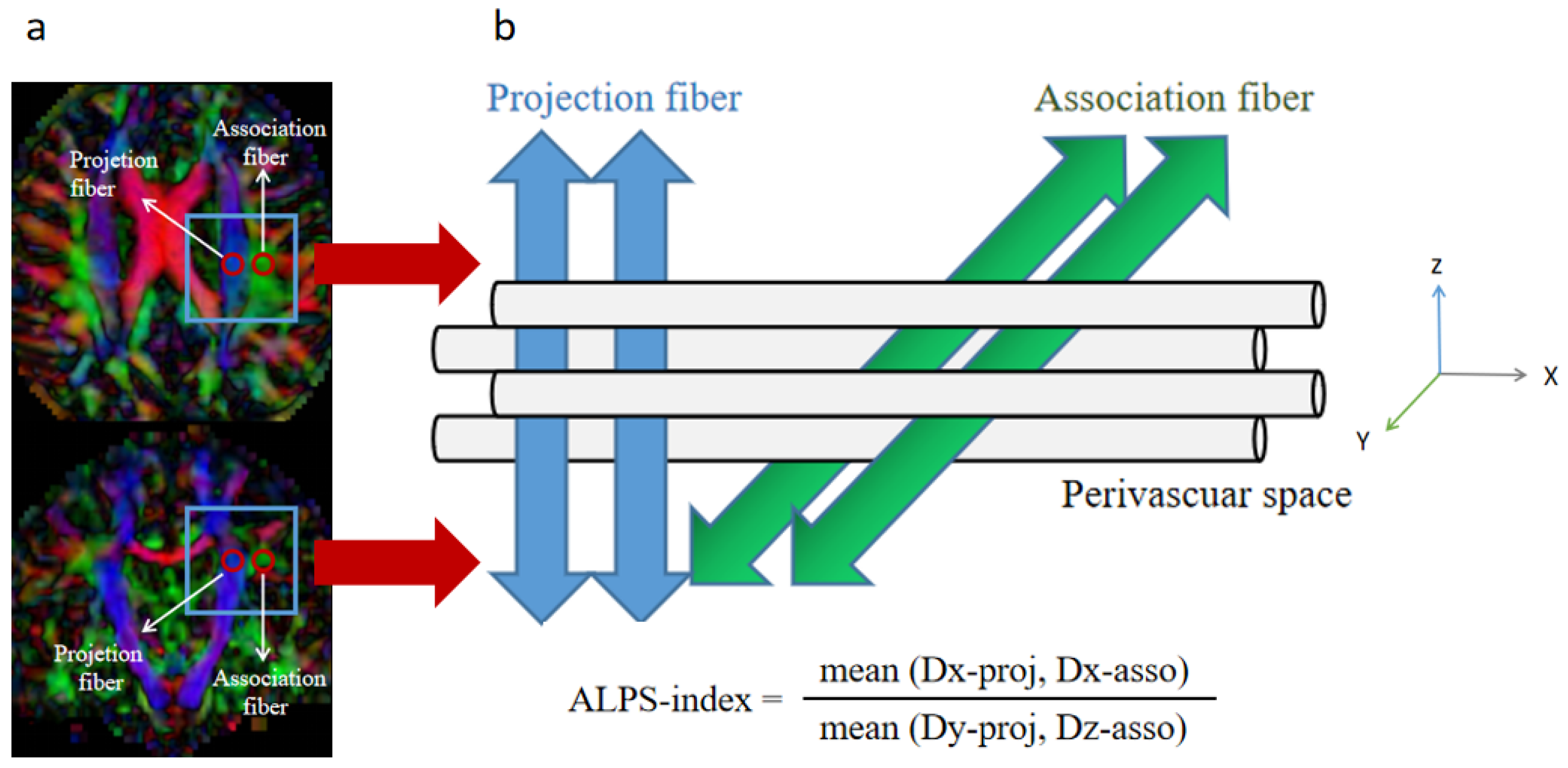
2.3.1. DTI-ALPS Analysis
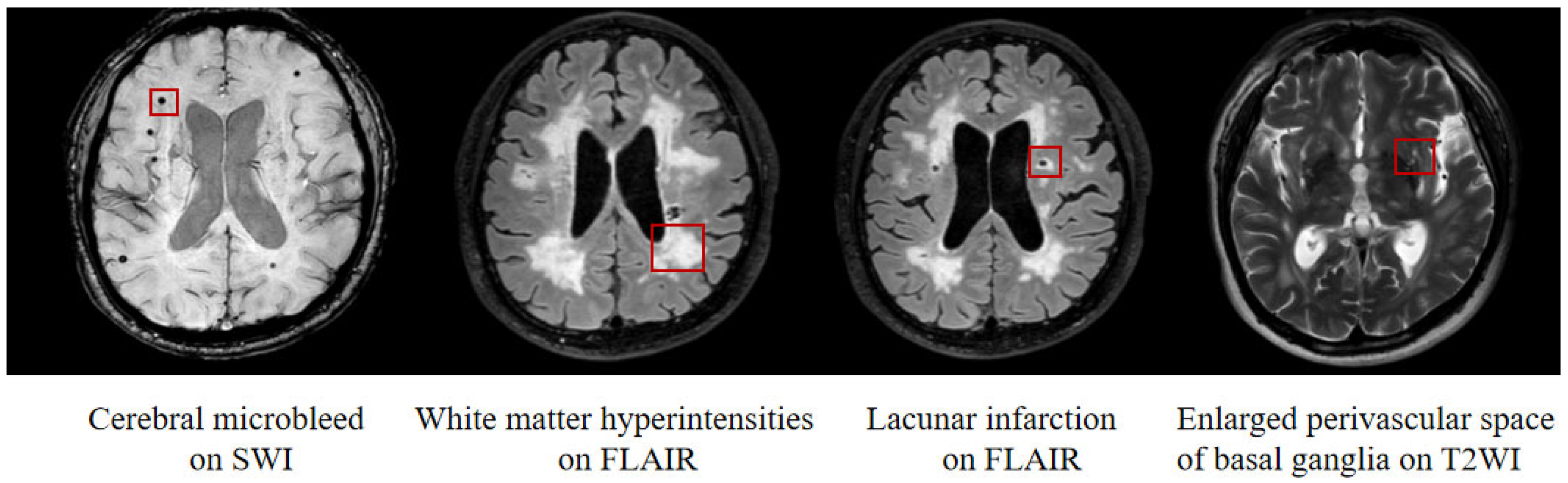
2.3.2. Imaging Biomarkers of CSVD
2.3.3. Neuropsychological Measurements
2.4. Statistical Analysis
3. Results
3.1. Demographics and Cognitive Function
3.2. CSVD Neuroimaging Features
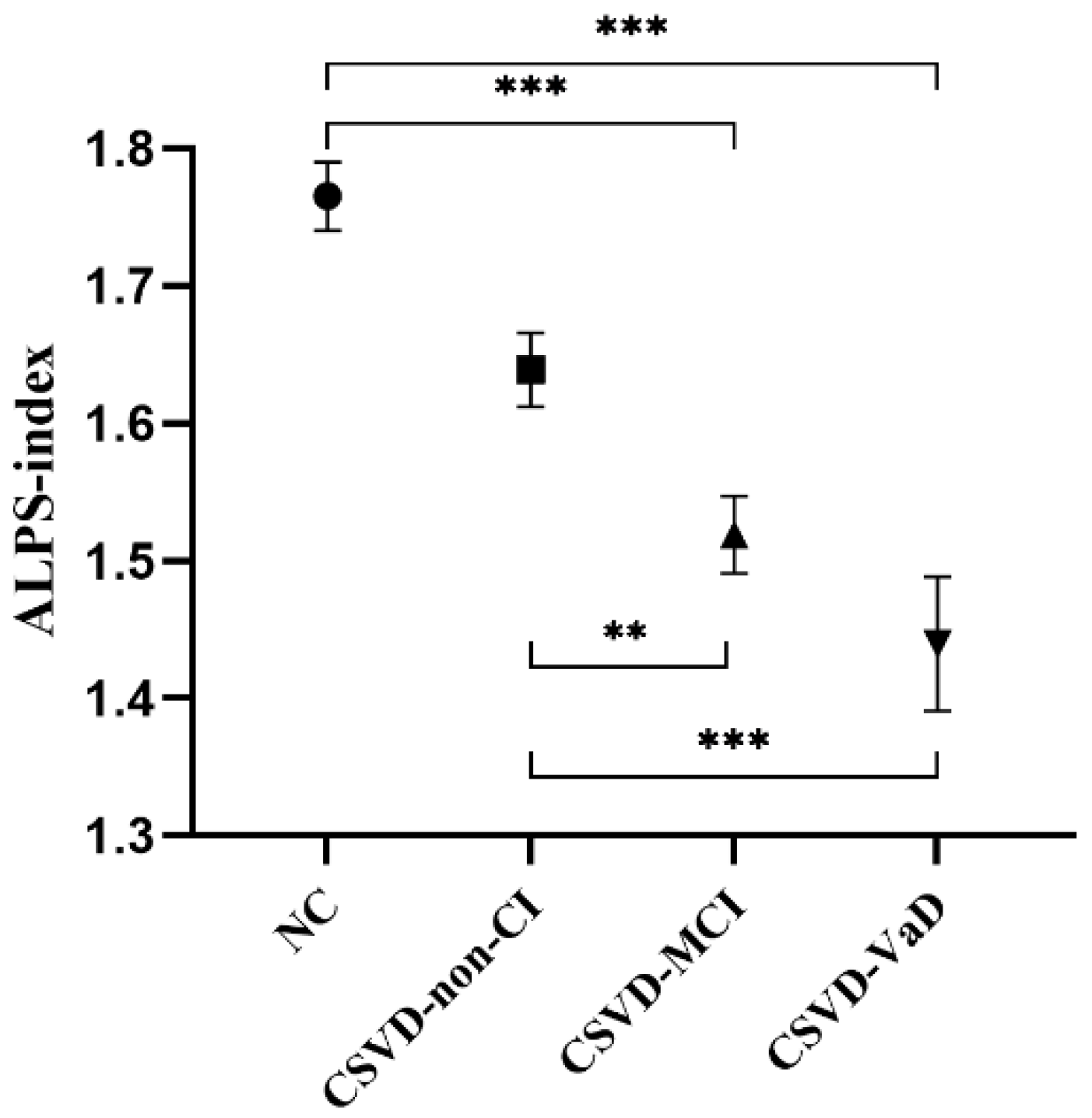
3.3. ALPS-Index
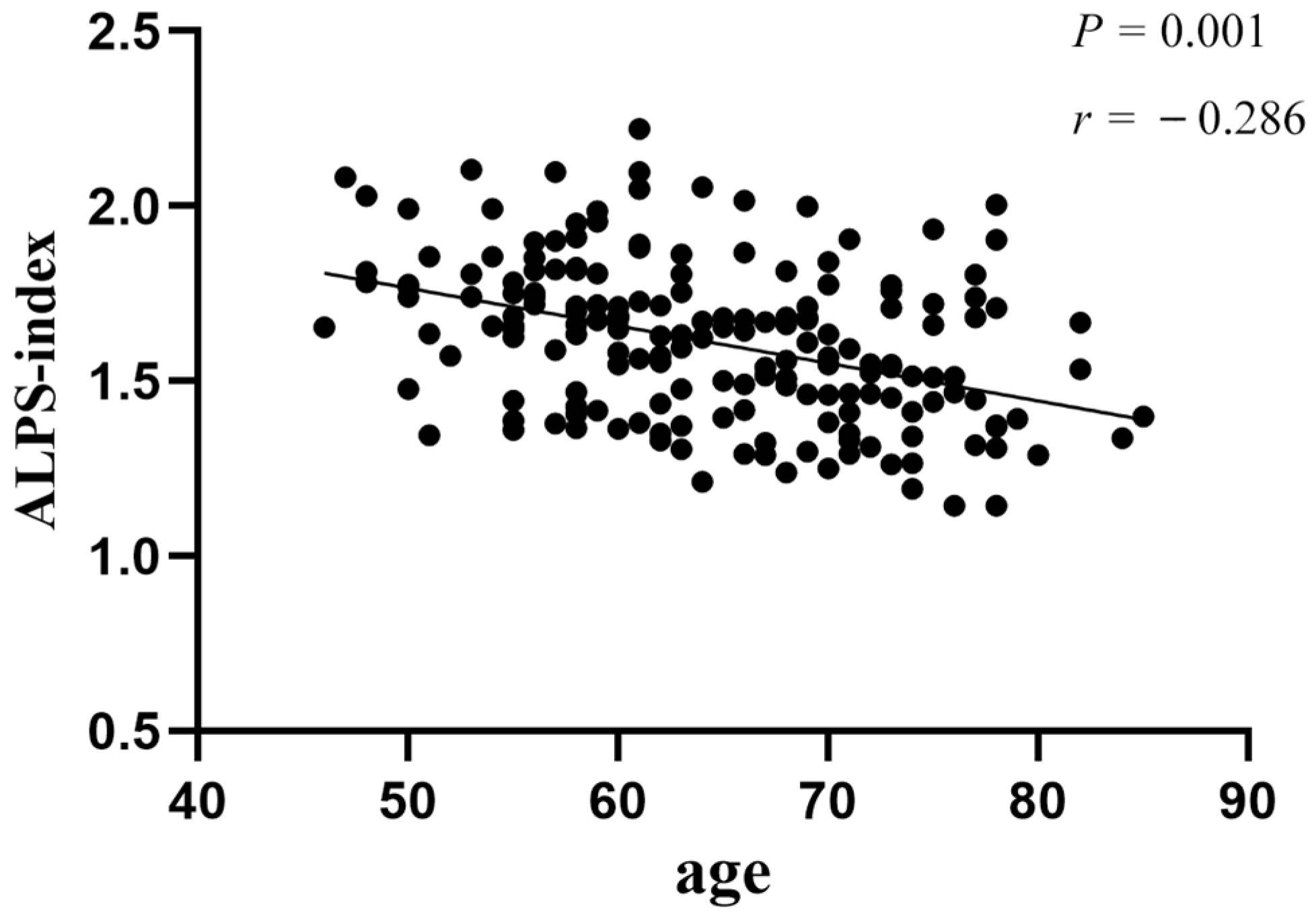
3.4. Correlation Analysis between Demographics and ALPS-Index
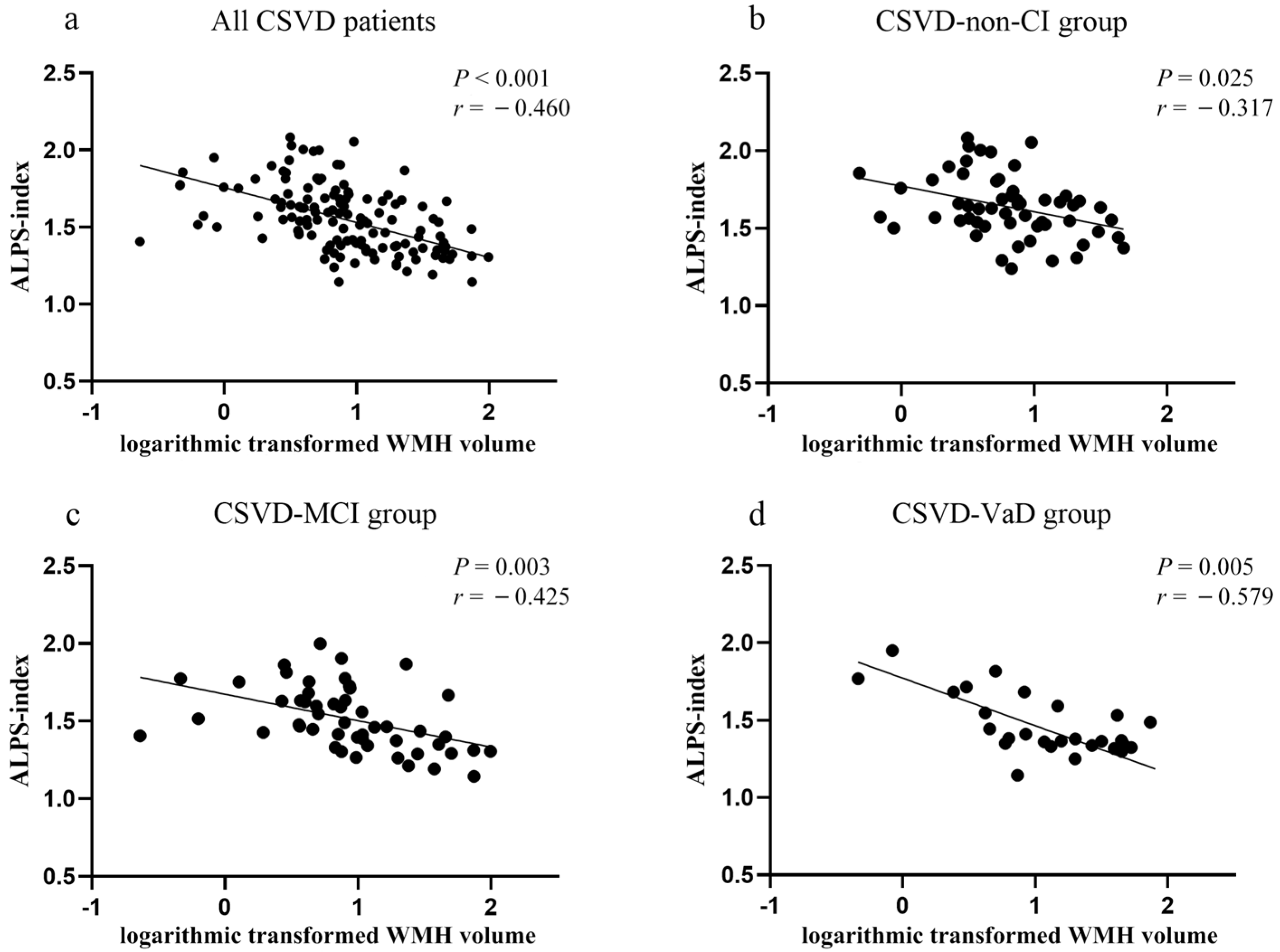
3.5. Correlation Analysis between ALPS-Index and CSVD Neuroimaging Features
3.6. Relationship between ALPS-Index and Cognitive Performance
3.7. Mediation Analysis
4. Discussion
5. Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rudilosso, S.; Rodriguez-Vazquez, A.; Urra, X.; Arboix, A. The Potential Impact of Neuroimaging and Translational Research on the Clinical Management of Lacunar Stroke. Int. J. Mol. Sci. 2022, 23, 1497. [Google Scholar] [CrossRef] [PubMed]
- Wardlaw, J.M.; Smith, C.; Dichgans, M. Small vessel disease: Mechanisms and clinical implications. Lancet Neurol. 2019, 18, 684–696. [Google Scholar] [CrossRef] [PubMed]
- Cannistraro, R.J.; Badi, M.; Eidelman, B.H.; Dickson, D.W.; Middlebrooks, E.H.; Meschia, J.F. CNS small vessel disease A clinical review. Neurology 2019, 92, 1146–1156. [Google Scholar] [CrossRef] [PubMed]
- Kasper, S.; Bancher, C.; Eckert, A.; Forstl, H.; Frolich, L.; Hort, J.; Korczyn, A.D.; Kressig, R.W.; Levin, O.; Palomo, M.S.M. Management of mild cognitive impairment (MCI): The need for national and international guidelines. World J. Biol. Psychiatry 2020, 21, 579–594. [Google Scholar] [CrossRef] [PubMed]
- Iliff, J.J.; Wang, M.H.; Liao, Y.H.; Plogg, B.A.; Peng, W.G.; Gundersen, G.A.; Benveniste, H.; Vates, G.E.; Deane, R.; Goldman, S.A.; et al. A Paravascular Pathway Facilitates CSF Flow Through the Brain Parenchyma and the Clearance of Interstitial Solutes, Including Amyloid beta. Sci. Transl. Med. 2012, 4, 11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Plog, B.A.; Nedergaard, M. The Glymphatic System in Central Nervous System Health and Disease: Past, Present, and Future. In Annual Review of Pathology: Mechanisms of Disease; Abbas, A.K., Aster, J.C., Eds.; Annual Reviews: Palo Alto, CA, USA, 2018; Volume 13, pp. 379–394. [Google Scholar]
- Nedergaard, M.; Goldman, S.A. Glymphatic failure as a final common pathway to dementia. Science 2020, 370, 50. [Google Scholar] [CrossRef]
- Mestre, H.; Kostrikov, S.; Mehta, R.I.; Nedergaard, M. Perivascular spaces, glymphatic dysfunction, and small vessel disease. Clin. Sci. 2017, 131, 2257–2274. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Zhang, R.T.; Ye, Y.Q.; Wang, S.Y.; Jiaerken, Y.; Hong, H.; Li, K.C.; Zeng, Q.Z.; Luo, X.; Xu, X.P.; et al. The Influence of Demographics and Vascular Risk Factors on Glymphatic Function Measured by Diffusion Along Perivascular Space. Front. Aging Neurosci. 2021, 13, 8. [Google Scholar] [CrossRef]
- Mestre, H.; Mori, Y.; Nedergaard, M. The Brain’s Glymphatic System: Current Controversies. Trends Neurosci. 2020, 43, 458–466. [Google Scholar] [CrossRef]
- Iliff, J.J.; Wang, M.H.; Zeppenfeld, D.M.; Venkataraman, A.; Plog, B.A.; Liao, Y.H.; Deane, R.; Nedergaard, M. Cerebral Arterial Pulsation Drives Paravascular CSF-Interstitial Fluid Exchange in the Murine Brain. J. Neurosci. 2013, 33, 18190–18199. [Google Scholar] [CrossRef] [PubMed]
- Ringstad, G.; Vatnehol, S.A.S.; Eide, P.K. Glymphatic MRI in idiopathic normal pressure hydrocephalus. Brain 2017, 140, 2691–2705. [Google Scholar] [CrossRef] [Green Version]
- Taoka, T.; Masutani, Y.; Kawai, H.; Nakane, T.; Matsuoka, K.; Yasuno, F.; Kishimoto, T.; Naganawa, S. Evaluation of glymphatic system activity with the diffusion MR technique: Diffusion tensor image analysis along the perivascular space (DTI-ALPS) in Alzheimer’s disease cases. Jpn. J. Radiol. 2017, 35, 172–178. [Google Scholar] [CrossRef]
- Zhang, W.H.; Zhou, Y.; Wang, J.A.; Gong, X.X.; Chen, Z.C.; Zhang, X.T.; Cai, J.S.; Chen, S.Y.; Fang, L.; Sun, J.Z.; et al. Glymphatic clearance function in patients with cerebral small vessel disease. Neuroimage 2021, 238, 118257. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.X.; Li, S.H.; Li, C.M.; Wang, R.; Chen, M.; Chen, H.B.; Su, W. Diffusion Tensor Imaging Along the Perivascular Space Index in Different Stages of Parkinson’s Disease. Front. Aging Neurosci. 2021, 13, 7. [Google Scholar] [CrossRef] [PubMed]
- Steward, C.E.; Venkatraman, V.K.; Lui, E.; Malpas, C.B.; Ellis, K.A.; Cyarto, E.V.; Vivash, L.; O’Brien, T.J.; Velakoulis, D.; Ames, D.; et al. Assessment of the DTI-ALPS Parameter Along the Perivascular Space in Older Adults at Risk of Dementia. J. Neuroimaging 2021, 31, 569–578. [Google Scholar] [CrossRef] [PubMed]
- Pantoni, L. Cerebral small vessel disease: From pathogenesis and clinical characteristics to therapeutic challenges. Lancet Neurol. 2010, 9, 689–701. [Google Scholar] [CrossRef] [PubMed]
- Katzman, R.; Zhang, M.Y.; Ouang Ya, Q.; Wang, Z.Y.; Liu, W.T.; Yu, E.; Wong, S.C.; Salmon, D.P.; Grant, I. A Chinese version of the Mini-Mental State Examination; impact of illiteracy in a Shanghai dementia survey. J. Clin. Epidemiol. 1988, 41, 971–978. [Google Scholar] [CrossRef]
- Lu, J.; Li, D.; Li, F.; Zhou, A.H.; Wang, F.; Zuo, X.M.; Jia, X.F.; Song, H.Q.; Jia, J.P. Montreal Cognitive Assessment in Detecting Cognitive Impairment in Chinese Elderly Individuals: A Population-Based Study. J. Geriatr. Psychiatry Neurol. 2011, 24, 184–190. [Google Scholar] [CrossRef]
- Gorelick, P.B.; Scuteri, A.; Black, S.E.; DeCarli, C.; Greenberg, S.M.; Iadecola, C.; Launer, L.J.; Laurent, S.; Lopez, O.L.; Nyenhuis, D.; et al. Vascular Contributions to Cognitive Impairment and Dementia A Statement for Healthcare Professionals From the American Heart Association/American Stroke Association. Stroke 2011, 42, 2672–2713. [Google Scholar] [CrossRef]
- Schmidt, P.; Gaser, C.; Arsic, M.; Buck, D.; Forschler, A.; Berthele, A.; Hoshi, M.; Ilg, R.; Schmid, V.J.; Zimmer, C.; et al. An automated tool for detection of FLAIR-hyperintense white-matter lesions in Multiple Sclerosis. Neuroimage 2012, 59, 3774–3783. [Google Scholar] [CrossRef]
- Huang, L.L.; Chen, X.; Sun, W.S.; Chen, H.F.; Ye, Q.; Yang, D.; Li, M.C.; Luo, C.M.; Ma, J.Y.; Shao, P.F.; et al. Early Segmental White Matter Fascicle Microstructural Damage Predicts the Corresponding Cognitive Domain Impairment in Cerebral Small Vessel Disease Patients by Automated Fiber Quantification. Front. Aging Neurosci. 2021, 12, 598242. [Google Scholar] [CrossRef]
- Tang, J.; Zhang, M.; Liu, N.; Xue, Y.; Ren, X.; Huang, Q.; Shi, L.; Fu, J. The Association Between Glymphatic System Dysfunction and Cognitive Impairment in Cerebral Small Vessel Disease. Front. Aging Neurosci. 2022, 14, 916633. [Google Scholar] [CrossRef]
- Xu, J.; Su, Y.; Fu, J.; Wang, X.; Nguchu, B.A.; Qiu, B.; Dong, Q.; Cheng, X. Glymphatic dysfunction correlates with severity of small vessel disease and cognitive impairment in cerebral amyloid angiopathy. Eur. J. Neurol. 2022, 29, 2895–2904. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.L.; Chen, P.C.; Lu, C.H.; Tsai, N.W.; Yu, C.C.; Chou, K.H.; Lai, Y.R.; Taoka, T.; Lin, W.C. Associations among Cognitive Functions, Plasma DNA, and Diffusion Tensor Image along the Perivascular Space (DTI-ALPS) in Patients with Parkinson’s Disease. Oxidative Med. Cell. Longev. 2021, 2021, 10. [Google Scholar] [CrossRef]
- Hamilton, O.K.L.; Backhouse, E.V.; Janssen, E.; Jochems, A.C.C.; Maher, C.; Ritakari, T.E.; Stevenson, A.J.; Xia, L.H.; Deary, I.J.; Wardlaw, J.M. Cognitive impairment in sporadic cerebral small vessel disease: A systematic review and meta-analysis. Alzheimer’s Dement. 2021, 17, 665–685. [Google Scholar] [CrossRef] [PubMed]
- Hu, A.M.; Ma, Y.L.; Li, Y.X.; Han, Z.Z.; Yan, N.; Zhang, Y.M. Association between Changes in White Matter Microstructure and Cognitive Impairment in White Matter Lesions. Brain Sci. 2022, 12, 482. [Google Scholar] [CrossRef] [PubMed]
- Rubinstein, D.Y.; Camarillo-Rodriguez, L.; Serruya, M.D.; Herweg, N.A.; Waldman, Z.J.; Wanda, P.A.; Sharan, A.D.; Weiss, S.A.; Sperling, M.R. Contribution of left supramarginal and angular gyri to episodic memory encoding: An intracranial EEG study. Neuroimage 2021, 225, 117514. [Google Scholar] [CrossRef] [PubMed]
- Orth, M.; Wagnon, C.; Neumann-Dunayevska, E.; Kaller, C.P.; Kloppel, S.; Meier, B.; Henke, K.; Peter, J. The left prefrontal cortex determines relevance at encoding and governs episodic memory formation. Cereb. Cortex, 2022; online ahead of print. [Google Scholar] [CrossRef]
- Siow, T.Y.; Toh, C.H.; Hsu, J.L.; Liu, G.H.; Lee, S.H.; Chen, N.H.; Fu, C.J.; Castillo, M.; Fang, J.T. Association of Sleep, Neuropsychological Performance, and Gray Matter Volume With Glymphatic Function in Community-Dwelling Older Adults. Neurology 2022, 98, E829–E838. [Google Scholar] [CrossRef]
- Benveniste, H.; Nedergaard, M. Cerebral small vessel disease: A glymphopathy? Curr. Opin. Neurobiol. 2022, 72, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Lockhart, S.N.; Schaich, C.L.; Craft, S.; Sachs, B.C.; Rapp, S.R.; Jung, Y.; Whitlow, C.T.; Sai, K.K.S.; Cleveland, M.; Williams, B.J.; et al. Associations among vascular risk factors, neuroimaging biomarkers, and cognition: Preliminary analyses from the Multi-Ethnic Study of Atherosclerosis (MESA). Alzheimer’s Dement. 2022, 18, 551–560. [Google Scholar] [CrossRef]
- Wong, S.M.; Jansen, J.F.A.; Zhang, C.E.; Hoff, E.I.; Staals, J.; van Oostenbrugge, R.J.; Backes, W.H. Blood-brain barrier impairment and hypoperfusion are linked in cerebral small vessel disease. Neurology 2019, 92, E1669–E1677. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.H.; Li, F.F.; Xing, D.X.; Song, H.Y.; Chen, J.S.; Duan, Y.; Yang, B.Q. A Novel Imaging Biomarker for Cerebral Small Vessel Disease Associated with Cognitive Impairment: The Deep-Medullary-Veins Score. Front. Aging Neurosci. 2021, 13, 7. [Google Scholar] [CrossRef]
- Liu, X.L.; Ouyang, F.B.; Hu, L.T.; Sun, P.; Yang, J.; Sun, Y.J.; Liao, M.S.; Lan, L.F.; Pei, Z.; Fan, Y.H. Mesenchymal Stem Cells Improve Cognitive Impairment and Reduce A beta Deposition via Promoting AQP4 Polarity and Relieving Neuroinflammation in Rats with Chronic Hypertension-Induced Cerebral Small-Vessel Disease. Front. Aging Neurosci. 2022, 14, 16. [Google Scholar]
- Jia, Y.L.; Liu, C.L.; Li, H.; Li, X.N.; Wu, J.; Zhao, Y.M.; Xu, M.Y.; Yu, H.T.; Guan, Z.T.; Sun, S.N.; et al. Enlarged Perivascular Space and Its Correlation with Polysomnography Indicators of Obstructive Sleep Apnea. Nat. Sci. Sleep 2021, 13, 863–872. [Google Scholar] [CrossRef]
- Shen, M.; Wei, G.R.; Cheng, M.; Jiang, H. Association between Enlarged Perivascular Spaces and Internal Carotid Artery Stenosis: A Study in Patients Diagnosed by Digital Subtraction Angiography. J. Stroke Cerebrovasc. Dis. 2020, 29, 8. [Google Scholar] [CrossRef] [PubMed]
- Gertje, E.C.; van Westen, D.; Panizo, C.; Mattsson-Carlgren, N.; Hansson, O. Association of Enlarged Perivascular Spaces and Measures of Small Vessel and Alzheimer Disease. Neurology 2021, 96, E193–E202. [Google Scholar] [CrossRef]
- Gyanwali, B.; Vrooman, H.; Venketasubramanian, N.; Wong, T.Y.; Cheng, C.Y.; Chen, C.; Hilal, S. Cerebral Small Vessel Disease and Enlarged Perivascular Spaces-Data from Memory Clinic and Population-Based Settings. Front. Neurol. 2019, 10, 8. [Google Scholar] [CrossRef] [PubMed]
- Mestre, H.; Du, T.; Sweeney, A.M.; Liu, G.J.; Samson, A.J.; Peng, W.G.; Mortensen, K.N.; Staeger, F.F.; Bork, P.A.R.; Bashford, L.; et al. Cerebrospinal fluid influx drives acute ischemic tissue swelling. Science 2020, 367, eaax7171. [Google Scholar] [CrossRef] [PubMed]
- Alber, J.; Alladi, S.; Bae, H.-J.; Barton, D.A.; Beckett, L.A.; Bell, J.M.; Berman, S.E.; Biessels, G.J.; Black, S.E.; Bos, I.; et al. White matter hyperintensities in vascular contributions to cognitive impairment and dementia (VCID): Knowledge gaps and opportunities. Alzheimer’s Dement. 2019, 5, 107–117. [Google Scholar] [CrossRef]
- Clancy, U.; Gilmartin, D.; Jochems, A.C.C.; Knox, L.; Doubal, F.N.; Wardlaw, J.M. Neuropsychiatric symptoms associated with cerebral small vessel disease: A systematic review and meta-analysis. Lancet Psychiatry 2021, 8, 225–236. [Google Scholar] [CrossRef] [PubMed]
- Dao, E.; Tam, R.; Hsiung, G.Y.R.; ten Brinke, L.; Crockett, R.; Barha, C.K.; Yoo, Y.; Al Keridy, W.; Doherty, S.H.; Laule, C.; et al. Exploring the Contribution of Myelin Content in Normal Appearing White Matter to Cognitive Outcomes in Cerebral Small Vessel Disease. J. Alzheimer’s Dis. 2021, 80, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Grau-Olivares, M.; Arboix, A.; Junque, C.; Arenaza-Urquijo, E.M.; Rovira, M.; Bartres-Faz, D. Progressive gray matter atrophy in lacunar patients with vascular mild cognitive impairment. Cerebrovasc. Dis. 2010, 30, 157–166. [Google Scholar] [CrossRef] [PubMed]


| Variables | NC Group (n = 52) | CSVS-Non-CI Group (n = 56) | CSVD-MCI Group (n = 52) | CSVD-VaD Group (n = 29) | F\χ2 | p Value |
|---|---|---|---|---|---|---|
| Gender (F/M) | 25/27 | 28/28 | 26/26 | 15/14 | 0.106 | 0.991 |
| Age (years, mean ± s.d.) | 58.88 ± 6.41 | 66.28 ± 9.25 | 67.48 ± 6.74 | 66.07 ± 8.75 | 12.87 | <0.001 a–c |
| Education (years, mean ± s.d.) | 14.48 ± 3.58 | 12.96 ± 3.46 | 10.19 ± 3.07 | 9.41 ± 3.19 | 21.75 | <0.001 b–e |
| Vascular risk factors | ||||||
| Hypertension, n (%) | 24 (46.2%) | 40 (71.4%) | 39 (75.0%) | 19 (65.5%) | 13.203 | 0.004 a–c |
| Diabetes mellitus, n (%) | 8 (15.4%) | 12 (21.4%) | 16 (31.4%) | 6 (20.7%) | 3.745 | 0.298 |
| Hyperlipidemia, n (%) | 13 (25.0%) | 12 (21.4%) | 8 (15.7%) | 2 (6.9%) | 1.36 | 0.253 |
| Smoking, n (%) | 10 (19.2%) | 10 (17.9%) | 12 (23.5%) | 5 (17.2%) | 0.491 | 0.920 |
| Alcohol drinking, n (%) | 10 (19.2%) | 9 (16.7%) | 11 (21.6%) | 1 (3.4%) | 4.41 | 0.214 |
| Variables | NC Group (n = 52) | CSVS-Non-CI Group (n = 56) | CSVD-MCI Group (n = 52) | CSVD-VaD Group (n = 29) | Statistics | p Value |
|---|---|---|---|---|---|---|
| General cognitive function | ||||||
| MMSE | 29.36 ± 0.77 | 28.84 ± 1.06 | 27.48 ± 1.39 | 20.24 ± 2.73 | 277.736 | <0.001 b–f |
| MoCA | 27.40 ± 1.30 | 26.87 ± 1.41 | 21.07 ± 2.60 | 14.52 ± 3.42 | 289.684 | <0.001 b–f |
| Cognitive domains | ||||||
| episodic memory | 0.34 ± 0.09 | 0.44 ± 0.08 | −0.23 ± 0.08 | −1.03 ± 0.11 | 38.164 | <0.001 b–f |
| Z-linguistic function | 0.53 ± 0.10 | 0.30 ± 0.10 | −0.31 ± 0.10 | −1.21 ± 0.15 | 30.603 | <0.001 b–f |
| Z-processing speed | 0.40 ± 0.10 | 0.32 ± 0.09 | −0.38 ± 0.09 | −0.88 ± 0.14 | 24.675 | <0.001 b–f |
| Z-executive function | 0.17 ± 0.11 | 0.27 ± 0.09 | −0.26 ± 0.10 | −0.53 ± 0.15 | 8.724 | <0.05 b–e |
| Z-visuospatial | 0.30 ± 0.10 | 0.34 ± 0.09 | −0.06 ± 0.10 | −1.54 ± 0.15 | 37.914 | <0.01 c–f |
| Variables | NC Group (n = 52) | CSVS-Non-CI Group (n = 56) | CSVD-MCI Group (n = 52) | CSVD-VaD Group (n = 29) | Statistics | p Value |
|---|---|---|---|---|---|---|
| ALPS-index (mean ± s. d.) | 1.76 ± 0.18 | 1.64 ± 0.20 | 1.52 ± 0.20 | 1.44 ± 0.26 | 13.706 | <0.001 b–e |
| neuroimaging features | ||||||
| WMH volume (mL, median (interquartile range)) | 0.93 (0.87) | 6.65 (8.84) | 8.00 (15.69) | 13.14 (34.93) | 95.429 | <0.001 a–c,e |
| CMB numbers median (interquartile range) | 0 | 0 (2.00) | 0 (5.00) | 2.00 (28.00) | 56.604 | <0.001 a–c,e,f |
| LI numbers median (interquartile range) | 0 | 0 (1.00) | 1.00 (2.00) | 1.00 (2.00) | 54.170 | <0.05 a–e |
| EPVS-bg, n (%) | 0 (0%) | 19 (33.9%) | 19 (36.5%) | 11 (37.9%) | 25.293 | <0.001 a–c |
| global gray matter volume (mL, mean ± s.d.) | 547.52 ± 46.58 | 546.67 ± 50.30 | 539.32 ± 51.20 | 543.96 ± 57.33 | 1.103 | 0.349 |
| global white matter volume (mL, mean ± s.d.) | 463.36 ± 60.50 | 466.46 ± 57.20 | 448.13 ± 52.42 | 449.37 ± 69.49 | 0.514 | 0.673 |
| cerebrospinal fluid volume (mL, mean ± s.d.) | 309.21 ± 37.08 | 337.38 ± 42.64 | 334.80 ± 56.48 | 341.77 ± 60.29 | 5.246 | 0.001 c |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ke, Z.; Mo, Y.; Li, J.; Yang, D.; Huang, L.; Yang, Z.; Qin, R.; Mao, C.; Lv, W.; Huang, Y.; et al. Glymphatic Dysfunction Mediates the Influence of White Matter Hyperintensities on Episodic Memory in Cerebral Small Vessel Disease. Brain Sci. 2022, 12, 1611. https://doi.org/10.3390/brainsci12121611
Ke Z, Mo Y, Li J, Yang D, Huang L, Yang Z, Qin R, Mao C, Lv W, Huang Y, et al. Glymphatic Dysfunction Mediates the Influence of White Matter Hyperintensities on Episodic Memory in Cerebral Small Vessel Disease. Brain Sciences. 2022; 12(12):1611. https://doi.org/10.3390/brainsci12121611
Chicago/Turabian StyleKe, Zhihong, Yuting Mo, Jiangnan Li, Dan Yang, Lili Huang, Zhiyuan Yang, Ruomeng Qin, Chenglu Mao, Weiping Lv, Yanan Huang, and et al. 2022. "Glymphatic Dysfunction Mediates the Influence of White Matter Hyperintensities on Episodic Memory in Cerebral Small Vessel Disease" Brain Sciences 12, no. 12: 1611. https://doi.org/10.3390/brainsci12121611







