Rekindling Action Language: A Neuromodulatory Study on Parkinson’s Disease Patients
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Experimental Task
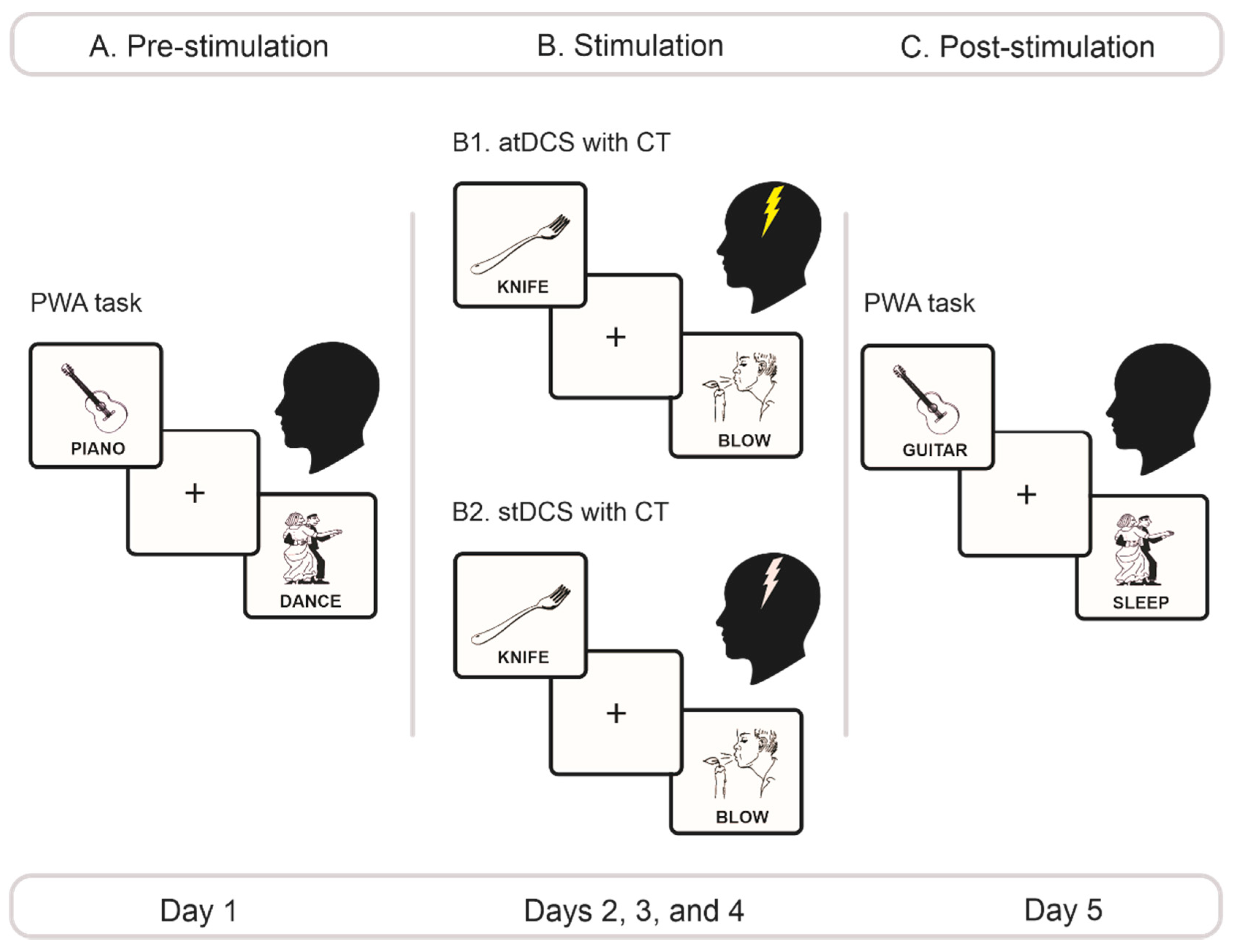
2.3. Experimental Protocol
2.3.1. Pre-Stimulation Phase
2.3.2. Stimulation Phase
2.3.3. Post-Stimulation Phase
2.4. Data Analysis
Behavioral Data Analysis
3. Results
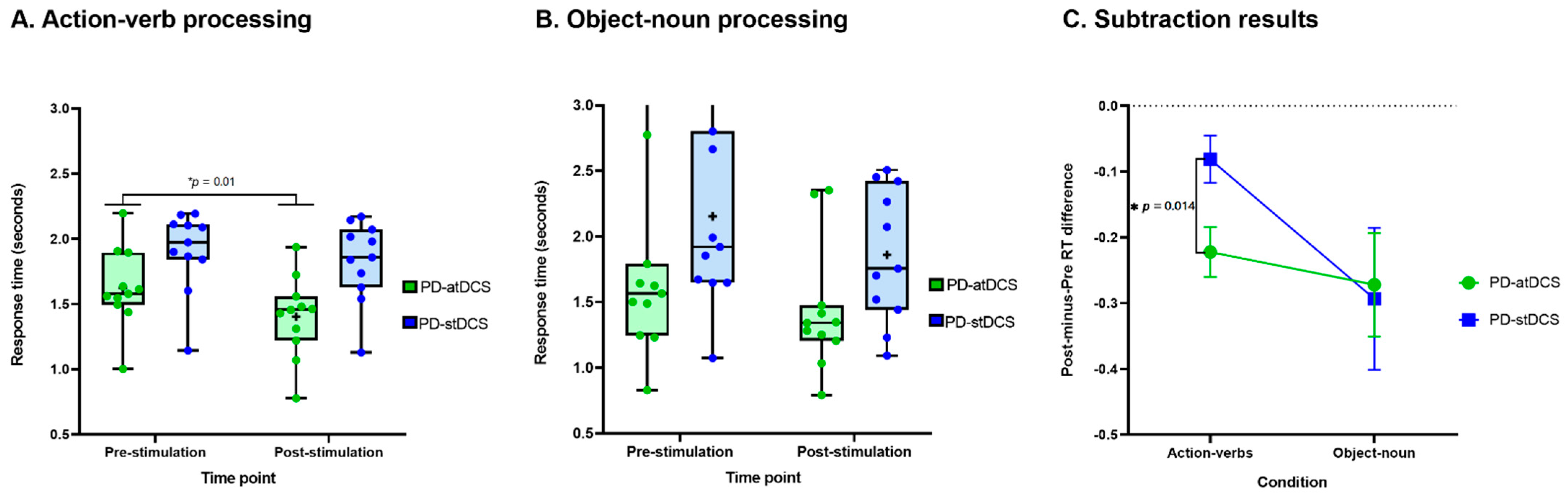
3.1. Behavioral Results
3.1.1. Action-Verb Processing
3.1.2. Object-Noun Processing
4. Discussion
5. Conclusions
6. Limitations and Avenues for Further Research
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ascherio, A.; Schwarzschild, M.A. The epidemiology of Parkinson’s disease: Risk factors and prevention. Lancet Neurol. 2016, 15, 1257–1272. [Google Scholar] [CrossRef]
- Dorsey, E.R.; Elbaz, A.; Nichols, E.; Abd-Allah, F.; Abdelalim, A.; Adsuar, J.C.; Ansha, M.G.; Brayne, C.; Choi, J.-Y.J.; Collado-Mateo, D.; et al. Global, regional, and national burden of Parkinson’s disease, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2018, 17, 939–953. [Google Scholar] [CrossRef] [Green Version]
- Rodriguez-Oroz, M.C.; Jahanshahi, M.; Krack, P.; Litvan, I.; Macias, R.; Bezard, E.; Obeso, J.A. Initial clinical manifestations of Parkinson’s disease: Features and pathophysiological mechanisms. Lancet Neurol. 2009, 8, 1128–1139. [Google Scholar] [CrossRef] [Green Version]
- Gratwicke, J.; Jahanshahi, M.; Foltynie, T. Parkinson’s disease dementia: A neural networks perspective. Brain 2015, 138, 1454–1476. [Google Scholar] [CrossRef]
- Halliday, G.; Leverenz, J.; Schneider, J.; Adler, C. The neurobiological basis of cognitive impairment in Parkinson’s disease. Mov. Disord. 2014, 29, 634–650. [Google Scholar] [CrossRef] [Green Version]
- Birba, A.; García-Cordero, I.; Kozono, G.; Legaz, A.; Ibanez, A.; Sedeño, L.; García, A. Losing ground: Frontostriatal atrophy disrupts language embodiment in Parkinson’s and Huntington’s disease. Neurosci. Biobehav. Rev. 2017, 80, 673–687. [Google Scholar] [CrossRef]
- Gallese, V.; Cuccio, V. The neural exploitation hypothesis and its implications for an embodied approach to language and cognition: Insights from the study of action verbs processing and motor disorders in Parkinson’s disease. Cortex 2018, 100, 215–225. [Google Scholar] [CrossRef]
- García, A.M.; Ibáñez, A. When embodiment breaks down: Language deficits as novel avenues into movement disorders. Cortex 2018, 100, 1–7. [Google Scholar] [CrossRef]
- Smith, K.M.; Caplan, D.N. Communication impairment in Parkinson’s disease: Impact of motor and cognitive symptoms on speech and language. Brain Lang. 2018, 185, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Pulvermüller, F.; Fadiga, L. Active perception: Sensorimotor circuits as a cortical basis for language. Nat. Rev. Neurosci. 2010, 11, 351–360. [Google Scholar] [CrossRef]
- Thompson, E.L.; Bird, G.; Catmur, C. Conceptualizing and testing action understanding. Neurosci. Biobehav. Rev. 2019, 105, 106–114. [Google Scholar] [CrossRef]
- Herrera, E.; Cuetos, F.; Ribacoba, R. Verbal fluency in Parkinson’s disease patients on/off dopamine medication. Neuropsychologia 2012, 50, 3636–3640. [Google Scholar] [CrossRef]
- Norel, R.; Agurto, C.; Heisig, S.; Rice, J.J.; Zhang, H.; Ostrand, R.; Wacnik, P.W.; Ho, B.K.; Ramos, V.L.; Cecchi, G.A. Speech-based characterization of dopamine replacement therapy in people with Parkinson’s disease. Park. Dis. 2020, 6, 1–8. [Google Scholar]
- Cardona, J.F.; Gershanik, O.; Gelormini-Lezama, C.; Houck, A.L.; Cardona, S.; Kargieman, L.; Trujillo, N.; Arevalo, A.; Amoruso, L.; Manes, F.; et al. Action-verb processing in Parkinson’s disease: New pathways for motor–language coupling. Brain Struct. Funct. 2013, 218, 1355–1373. [Google Scholar] [CrossRef]
- Bocanegra, Y.; García, A.M.; Pineda, D.; Buriticá, O.; Villegas, A.; Lopera, F.; Gómez, D.; Gómez-Arias, C.; Cardona, J.F.; Trujillo, N.; et al. Syntax, action verbs, action semantics, and object semantics in Parkinson’s disease: Dissociability, progression, and executive influences. Cortex 2015, 69, 237–254. [Google Scholar] [CrossRef]
- García, A.M.; Carrillo, F.; Orozco-Arroyave, J.R.; Trujillo, N.; Bonilla, J.F.V.; Fittipaldi, S.; Adolfi, F.; Nöth, E.; Sigman, M.; Slezak, D.F.; et al. How language flows when movements don’t: An automated analysis of spontaneous discourse in Parkinson’s disease. Brain Lang. 2016, 162, 19–28. [Google Scholar] [CrossRef] [PubMed]
- García, A.M.; Bocanegra, Y.; Herrera, E.; Moreno, L.; Carmona, J.; Baena, A.; Lopera, F.; Pineda, D.; Melloni, M.; Legaz, A.; et al. Parkinson’s disease compromises the appraisal of action meanings evoked by naturalistic texts. Cortex 2018, 100, 111–126. [Google Scholar] [CrossRef] [PubMed]
- Péran, P.; Cardebat, D.; Cherubini, A.; Piras, F.; Luccichenti, G.; Peppe, A.; Caltagirone, C.; Rascol, O.; Demonet, J.-F.; Sabatini, U. Object naming and action-verb generation in Parkinson’s disease: A fMRI study. Cortex 2009, 45, 960–971. [Google Scholar] [CrossRef] [PubMed]
- Péran, P.; Rascol, O.; Demonet, J.-F.; Celsis, P.; Nespoulous, J.-L.; Dubois, B.; Cardebat, D. Deficit of verb generation in nondemented patients with Parkinson’s disease. Mov. Disord. 2002, 18, 150–156. [Google Scholar] [CrossRef] [PubMed]
- Boulenger, V.; Mechtouff, L.; Thobois, S.; Broussolle, E.; Jeannerod, M.; Nazir, T.A. Word processing in Parkinson’s disease is impaired for action verbs but not for concrete nouns. Neuropsychologia 2008, 46, 743–756. [Google Scholar] [CrossRef] [Green Version]
- Ibáñez, A.; Cardona, J.F.; Dos Santos, Y.V.; Blenkmann, A.; Aravena, P.; Roca, M.; Hurtado, E.; Nerguizian, M.; Amoruso, L.; Gómez-Arévalo, G.; et al. Motor-language coupling: Direct evidence from early Parkinson’s disease and intracranial cortical recordings. Cortex 2013, 49, 968–984. [Google Scholar] [CrossRef]
- Abrevaya, S.; Sedeño, L.; Fitipaldi, S.; Pineda, D.; Lopera, F.; Buritica, O.; Villegas, A.; Bustamante, C.; Gomez, D.; Trujillo, N.; et al. The Road Less Traveled: Alternative Pathways for Action-Verb Processing in Parkinson’s Disease. J. Alzheimers Dis. 2016, 55, 1429–1435. [Google Scholar] [CrossRef]
- García, A.; Moguilner, S.; Torquati, K.; Marco, E.G.; Herrera, E.; Muñoz, E.; Castillo, E.M.; Kleineschay, T.; Sedeño, L.; Ibáñez, A. How meaning unfolds in neural time: Embodied reactivations can precede multimodal semantic effects during language processing. NeuroImage 2019, 197, 439–449. [Google Scholar] [CrossRef]
- Hauk, O.; Johnsrude, I.; Pulvermüller, F. Somatotopic Representation of Action Words in Human Motor and Premotor Cortex. Neuron 2004, 41, 301–307. [Google Scholar] [CrossRef] [Green Version]
- Hauk, O.; Shtyrov, Y.; Pulvermüller, F. The time course of action and action-word comprehension in the human brain as revealed by neurophysiology. J. Physiol. Paris 2008, 102, 50–58. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tomasino, B.; Fink, G.R.; Sparing, R.; Dafotakis, M.; Weiss, P.H. Action verbs and the primary motor cortex: A comparative TMS study of silent reading, frequency judgments, and motor imagery. Neuropsychologia 2008, 46, 1915–1926. [Google Scholar] [CrossRef] [PubMed]
- Birba, A.; Vitale, F.; Padrón, I.; Dottori, M.; de Vega, M.; Zimerman, M.; Sedeño, L.; Ibáñez, A.; García, A.M. Electrifying discourse: Anodal tDCS of the primary motor cortex selectively reduces action appraisal in naturalistic narratives. Cortex 2020, 132, 460–472. Available online: https://linkinghub.elsevier.com/retrieve/pii/S0010945220303038 (accessed on 28 August 2020). [CrossRef] [PubMed]
- Birba, A.; Beltrán, D.; Caro, M.M.; Trevisan, P.; Kogan, B.; Sedeño, L.; Ibáñez, A.; García, A.M. Motor-system dynamics during naturalistic reading of action narratives in first and second language. Neuroimage 2020, 216, 116820. Available online: https://linkinghub.elsevier.com/retrieve/pii/S1053811920303074 (accessed on 2 February 2021). [CrossRef]
- Nitsche, M.A.; Paulus, W. Excitability changes induced in the human motor cortex by weak transcranial direct current stimulation. Exp. Brain Res. 2000, 527, 633–639. [Google Scholar] [CrossRef]
- Lefaucheur, J. A comprehensive database of puublished tDCS clinical trials (2005–2016). Neurophysiol. Clin. 2016, 46, 319–398. [Google Scholar] [CrossRef]
- Stagg, C.; Antal, A.; Nitsche, M. Physiology of Transcranial Direct Current Stimulation. J. ECT 2018, 34, 144–152. [Google Scholar] [CrossRef]
- Lefaucheur, J.-P.; Antal, A.; Ayache, S.S.; Benninger, D.; Brunelin, J.; Cogiamanian, F.; Cotelli, M.; De Ridder, D.; Ferrucci, R.; Langguth, B.; et al. Evidence-based guidelines on the therapeutic use of transcranial direct current stimulation (tDCS). Clin. Neurophysiol. 2017, 128, 56–92. Available online: http://www.ncbi.nlm.nih.gov/pubmed/27866120 (accessed on 2 February 2021). [CrossRef] [PubMed]
- Hummel, F.C.; Celnik, P.; Pascual-Leone, A.; Fregni, F.; Byblow, W.D.; Buetefisch, C.M.; Rothwell, J.; Cohen, L.G.; Gerloff, C. Controversy: Noninvasive and invasive cortical stimulation show efficacy in treating stroke patients. Brain Stimul. 2008, 1, 370–382. [Google Scholar] [CrossRef]
- Tahtis, V.; Kaski, D. Parkinson’s disease treatments: Focus on transcranial direct current stimulation (tDCS). J. Park. Restless Legs Syndr. 2017, 7, 55–70. [Google Scholar] [CrossRef] [Green Version]
- Elsner, B. Cochrane Database of Systematic Reviews Transcranial Direct Current Stimulation (tDCS) for Idiopathic Parkinson’s Disease (Review). 2016. Available online: www.cochranelibrary.com (accessed on 2 February 2021).
- Nitsche, M.A.; Seeber, A.; Frommann, K.; Klein, C.C.; Rochford, C.; Nitsche, M.S.; Fricke, K.; Liebetanz, D.; Lang, N.; Antal, A.; et al. Modulating parameters of excitability during and after transcranial direct current stimulation of the human motor cortex. J. Physiol. 2005, 568, 291–303. [Google Scholar] [CrossRef] [PubMed]
- Andrews, S.C.; Hoy, K.E.; Enticott, P.; Daskalakis, Z.J.; Fitzgerald, P.B. Improving working memory: The effect of combining cognitive activity and anodal transcranial direct current stimulation to the left dorsolateral prefrontal cortex. Brain Stimul. 2011, 4, 84–89. [Google Scholar] [CrossRef]
- Perceval, G.; Martin, A.; Copland, D.A.; Laine, M.; Meinzer, M. High-definition tDCS of the temporo-parietal cortex enhances access to newly learned words. Sci. Rep. 2017, 7, 1–9. [Google Scholar] [CrossRef]
- Niccolai, V.; Klepp, A.; Indefrey, P.; Schnitzler, A.; Biermann-Ruben, K. Semantic discrimination impacts tDCS modulation of verb processing. Sci. Rep. 2017, 7, 17162. [Google Scholar] [CrossRef] [Green Version]
- Liuzzi, G.; Freundlieb, N.; Ridder, V.; Hoppe, J.; Heise, K.-F.; Zimerman, M.; Dobel, C.; Enriquez-Geppert, S.; Gerloff, C.; Zwitserlood, P.; et al. The involvement of the left motor cortex in learning of a novel action word lexicon. Curr. Biol. 2010, 20, 1745–1751. [Google Scholar] [CrossRef] [Green Version]
- Branscheidt, M.; Hoppe, J.; Freundlieb, N.; Zwitserlood, P.; Liuzzi, G. tDCS over the motor cortex shows differential effects on action and object words in associative word learning in healthy aging. Front. Aging Neurosci. 2017, 9, 137. [Google Scholar] [CrossRef]
- Saruco, E.; Di Rienzo, F.; Nunez-Nagy, S.; González, M.A.R.; Jackson, P.; Collet, C.; Saimpont, A.; Guillot, A. Anodal tDCS over the primary motor cortex improves motor imagery benefits on postural control: A pilot study. Sci. Rep. 2017, 7, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saimpont, A.; Mercier, C.; Malouin, F.; Guillot, A.; Collet, C.; Doyon, J.; Jackson, P.L. Anodal transcranial direct current stimulation enhances the effects of motor imagery training in a finger tapping task. Eur. J. Neurosci. 2015, 43, 113–119. [Google Scholar] [CrossRef]
- Foerster, Á.; Rocha, S.; Wiesiolek, C.; Chagas, A.P.; Machado, G.; Silva, E.; Fregni, F.; Monte-Silva, K. Site-specific effects of mental practice combined with transcranial direct current stimulation on motor learning. Eur. J. Neurosci. 2012, 37, 786–794. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lang, N.; Siebner, H.R.; Ward, N.S.; Lee, L.; Nitsche, M.A.; Paulus, W.; Rothwell, J.; Lemon, R.N.; Frackowiak, R. How does transcranial DC stimulation of the primary motor cortex alter regional neuronal activity in the human brain? Eur. J. Neurosci. 2005, 22, 495–504. [Google Scholar] [CrossRef] [PubMed]
- Polanía, R.; Paulus, W.; Nitsche, M. Modulating cortico-striatal and thalamo-cortical functional connectivity with transcranial direct current stimulation. Hum. Brain Mapp. 2012, 33, 2499–2508. [Google Scholar] [CrossRef] [PubMed]
- Moguilner, S.; Birba, A.; Fino, D.; Isoardi, R.; Huetagoyena, C.; Otoya, R.; Tirapu, V.; Cremaschi, F.; Sedeño, L.; Ibáñez, A.; et al. Multimodal neurocognitive markers of frontal lobe epilepsy: Insights from ecological text processing. Neuroimage 2021, 235, 117998. [Google Scholar] [CrossRef]
- Suarez-García, D.M.A.; Grisales-Cárdenas, J.S.; Zimerman, M.; Cardona, J.F. Transcranial Direct Current Stimulation to Enhance Cognitive Impairment in Parkinson’s Disease: A Systematic Review and Meta-Analysis. Front. Neurol. 2020, 11, 1–8. [Google Scholar] [CrossRef]
- Benninger, D.H.; Lomarev, M.; Lopez, G.; Wassermann, E.M.; Li, X.; Considine, E.; Hallett, M. Transcranial direct current stimulation for the treatment of Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 2010, 81, 1105–1111. [Google Scholar] [CrossRef] [Green Version]
- Costa-Ribeiro, A.; Maux, A.; Bosford, T.; Aoki, Y.; Castro, R.; Baltar, A.; Shirahige, L.; Filho, A.M.; Nitsche, M.A.; Silva, K.M. Transcranial direct current stimulation associated with gait training in Parkinson’s disease: A pilot randomized clinical trial. Dev. Neurorehabilit. 2016, 20, 121–128. Available online: http://www.tandfonline.com/doi/full/10.3109/17518423.2015.1131755 (accessed on 2 February 2021). [CrossRef]
- Costa-ribeiro, A.; Maux, A.; Bosford, T.; Tenório, Y.; Marques, D.; Carneiro, M.; Nitsche, M.A.; Filho, A.M.; Monte-Silva, K. Dopamine-independent effects of combining transcranial direct current stimulation with cued gait training on cortical excitability and functional mobility in parkinson’s disease. J. Rehabil. Med. 2016, 48, 819–823. [Google Scholar] [CrossRef] [Green Version]
- Schabrun, S.M.; Lamont, R.M.; Brauer, S.G. Transcranial direct current stimulation to enhance dual-task gait training in Parkinson’s disease: A pilot RCT. PLoS ONE 2016, 11, e0158497. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Broeder, S.; Heremans, E.; Pereira, M.P.; Nackaerts, E.; Meesen, R.; Verheyden, G.; Nieuwboer, A. Does transcranial direct current stimulation during writing alleviate upper limb freezing in people with Parkinson’s disease? A pilot study. Hum. Mov. Sci. 2019, 65, 142–153. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dasilva, A.F.; Mendonca, M.E.; Zaghi, S.; Lopes, M.; Dossantos, M.F.; Spierings, E.L.; Bajwa, Z.; Datta, A.; Bikson, M.; Fregni, F. tDCS-Induced Analgesia and Electrical Fields in Pain-Related Neural Networks in Chronic Migraine. Headache 2012, 52, 1283–1295. [Google Scholar] [CrossRef] [PubMed]
- Lattari, E.; Costa, S.S.; Campos, C.; De Oliveira, A.J.; Machado, S.; Neto, G.A.M. Can transcranial direct current stimulation on the dorsolateral prefrontal cortex improves balance and functional mobility in Parkinson’s disease? Neurosci. Lett. 2017, 636, 165–169. [Google Scholar] [CrossRef] [Green Version]
- Kaski, D.; Dominguez, R.O.; Allum, J.H.; Islam, A.F.; Bronstein, A.M. Combining physical training with transcranial direct current stimulation to improve gait in Parkinson’s disease: A pilot randomized controlled study. Clin. Rehabil. 2014, 28, 1115–1124. [Google Scholar] [CrossRef]
- Valentino, F.; Cosentino, G.; Brighina, F.; Pozzi, N.; Sandrini, G.; Fierro, B.; Savettieri, G.; D’Amelio, M.; Pacchetti, C. Transcranial Direct Current Stimulation for Treatment of Freezing of Gait: A cross-over Study. Mov. Disord. 2014, 29, 1064–1069. [Google Scholar] [CrossRef]
- Fregni, F.; Boggio, P.S.; Santos, M.C.; Lima, M.; Vieira, A.L.; Rigonatti, S.P.; Silva, M.T.A.; Barbosa, E.R.; Nitsche, M.A.; Pascual-Leone, A. Noninvasive Cortical Stimulation with Transcranial Direct Current Stimulation in Parkinson’s Disease. Mov. Disord. 2006, 21, 1693–1702. [Google Scholar] [CrossRef]
- Agarwal, S.; Pawlak, N.; Cucca, A.; Sharma, K.; Dobbs, B.; Shaw, M.; Charvet, L.; Biagioni, M. Remotely-supervised transcranial direct current stimulation paired with cognitive training in Parkinson’s disease: An open-label study. J. Clin. Neurosci. 2018, 57, 51–57. [Google Scholar] [CrossRef]
- Manenti, R.; Brambilla, M.; Benussi, A.; Rosini, S.; Cobelli, C.; Ferrari, C.; Petesi, M.; Orizio, I.; Padovani, A.; Borroni, B.; et al. Mild cognitive impairment in Parkinson’s disease is improved by transcranial direct current stimulation combined with physical therapy. Mov. Disord. 2016, 31, 715–724. [Google Scholar] [CrossRef]
- Manenti, R.; Brambilla, M.; Rosini, S.; Orizio, I.; Ferrari, C.; Borroni, B.; Cotelli, M. Time up and go task performance improves after transcranial direct current stimulation in patient affected by Parkinson’s disease. Neurosci. Lett. 2014, 580, 74–77. [Google Scholar] [CrossRef]
- Salimpour, Y.; Mari, Z.K.; Shadmehr, R. Altering Effort Costs in Parkinson’s Disease with Noninvasive Cortical Stimulation. J. Neurosci. 2015, 35, 12287–12302. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yotnuengnit, P.; Bhidayasiri, R.; Donkhan, R.; Chaluaysrimuang, J.; Piravej, K. Effects of Transcranial Direct Current Stimulation Plus Physical Therapy on Gait in Patients with Parkinson Disease: A Randomized Controlled Trial. Am. J. Phys. Med. Rehabil. 2017, 97, 7–15. [Google Scholar] [CrossRef]
- Schoellmann, A.; Scholten, M.; Wasserka, B.; Govindan, R.B.; Krüger, R.; Gharabaghi, A.; Plewnia, C.; Weiss, D. Anodal tDCS modulates cortical activity and synchronization in Parkinson’s disease depending on motor processing. NeuroImage Clin. 2019, 22, 101689. [Google Scholar] [CrossRef]
- Bueno, M.E.B.; Neto, L.I.D.N.; Terra, M.B.; Barboza, N.M.; Okano, A.H.; Smaili, S.M. Effectiveness of acute transcranial direct current stimulation on non-motor and motor symptoms in Parkinson’s disease. Neurosci. Lett. 2019, 696, 46–51. [Google Scholar] [CrossRef] [PubMed]
- Pereira, J.B.; Junqué, C.; Bartrés-Faz, D.; Martí, M.J.; Sala-Llonch, R.; Compta, Y.; Falcón, C.; Vendrell, P.; Pascual-Leone, Á.; Valls-Solé, J.; et al. Modulation of verbal fluency networks by transcranial direct current stimulation (tDCS) in Parkinson’s disease. Brain Stimul. 2013, 6, 16–24. [Google Scholar] [CrossRef]
- Cotelli, M.; Borroni, B.; Manenti, R.; Zanetti, M.; Arevalo, A.; Cappa, S.F.; Padovani, A. Action and object naming in Parkinson’s disease without dementia. Eur. J. Neurol. 2007, 14, 632–637. [Google Scholar] [CrossRef]
- Bocanegra, Y.; García, A.; Lopera, F.; Pineda, D.; Baena, A.; Ospina, P.; Alzate, D.; Buriticá, O.; Moreno, L.; Ibanez, A.; et al. Unspeakable motion: Selective action-verb impairments in Parkinson’s disease patients without mild cognitive impairment. Brain Lang. 2017, 168, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Oldfield, R.C. The assessment and analysis of handedness: The Edinburgh inventory. Neuropsychologia 1971, 9, 97–113. [Google Scholar] [CrossRef]
- Hughes Daniel, S.; Kilford, L.; Lees, A. Accuracy of clinical diagnosis of idiopathic Parkinson’s disease: A clinico-pathological study of 100 cases. Neurol Neurosurg. Psychiatry 1992, 55, 181–184. [Google Scholar] [CrossRef] [Green Version]
- Goetz, C.G.; Tilley, B.C.; Shaftman, S.R.; Stebbins, G.T.; Fahn, S.; Martinez-Martin, P.; Poewe, W.; Sampaio, C.; Stern, M.B.; Dodel, R.; et al. Movement Disorder Society-sponsored revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS): Scale presentation and clinimetric testing results. Mov. Disord. 2008, 23, 2129–2170. [Google Scholar] [CrossRef]
- Hoehn, M.M.; Yahr, M.D. Parkinsonism: Onset, progression, and mortality. Neurology 1967, 17, 427–442. [Google Scholar] [CrossRef] [Green Version]
- Mahoney, F.I.; Barthel, D.W. Functional Evaluation: The Barthel Index. Md. State Med. J. 1965, 14, 61–65. Available online: http://www.ncbi.nlm.nih.gov/pubmed/14258950 (accessed on 2 February 2021).
- Lawton, M.P.; Brody, E.M. Assessment of older people: Self-Maintaining and Instrumental Activities of Daily Living. Gerontologist 1969, 9, 179–186. Available online: http://gerontologist.oxfordjournals.org/ (accessed on 2 February 2021). [CrossRef] [PubMed]
- Martínez, J.; Onís, M.; Dueñas, R.; Albert, C.; Aguado, C.; Luque, R. The Spanish version of the Yesavage abbreviated questionnaire (GDS) to screen depressive dysfunc-tions in patients older than 65 years. Medifam 2002, 12, 620–630. [Google Scholar]
- Ospina, N. Adaptación y Validación en Colombia del Addenbrooke’s Cognitive Examination-Revisado (ACE-R) en Pacientes con Deterioro Cognoscitivo Leve y Demencia; Universidad Nacional de Colombia: Bogotá, Colombia, 2015. [Google Scholar]
- Reyes, M.; Lloret, S.; Gerscovich, E.; Martin, M.; Leiguarda, R.; Merello, M. Addenbrooke’s Cognitive Examination validation in Parkinson’s disease. Eur. J. Neurol. 2009, 16, 142–147. [Google Scholar] [CrossRef] [PubMed]
- Rittman, T.; Ghosh, B.; McColgan, P.; Breen, D.P.; Evans, J.; Williams-Gray, C.H.; Barker, R.A.; Rowe, J.B. The Addenbrooke’s Cognitive Examination for the differential diagnosis and longitudinal assessment of patients with parkinsonian disorders. J. Neurol. Neurosurg. Psychiatry 2013, 84, 544–551. [Google Scholar] [CrossRef] [Green Version]
- McColgan, P.; Evans, J.R.; Breen, D.P.; Mason, S.L.; Barker, R.A.; Williams-Gray, C.H. Addenbrooke’s Cognitive Examination-Revised for mild cognitive impairment in Parkinson’s disease. Mov. Disord. 2012, 27, 1173–1177. [Google Scholar] [CrossRef]
- Torralva, T.; Roca, M.; Gleichgerrcht, E.; López, P.L.; Manes, F. INECO Frontal Screening (IFS): A brief, sensitive, and specific tool to assess executive functions in dementia. J. Int. Neuropsychol. Soc. 2009, 15, 777–786. [Google Scholar] [CrossRef] [Green Version]
- García, A.; Sedeño, L.; Trujillo, N.; Bocanegra, Y.; Gomez, D.; Pineda, D.; Villegas, A.; Muñoz, E.; Arias, W.; Ibáñez, A. Language Deficits as a Preclinical Window into Parkinson’s Disease: Evidence from Asymptomatic Parkin and Dardarin Mutation Carriers. J. Int. Neuropsychol. Soc. 2017, 23, 150–158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cardona, J.F.; Kargieman, L.; Sinay, V.; Gershanik, O.; Gelormini, C.; Amoruso, L.; Roca, M.; Pineda, D.; Trujillo, N.; Michon, M.; et al. How embodied is action language? Neurological evidence from motor diseases. Cognition 2014, 131, 311–322. [Google Scholar] [CrossRef]
- Druks, J.; Masterson, J. An Object and Action Naming Battery; Psychology Press: Hove, UK, 2000. [Google Scholar]
- Bates, E.; D’Amico, S.; Jacobsen, T.; Székely, A.; Andonova, E.; Devescovi, A.; Herron, D.; Lu, C.C.; Pechmann, T.; Pléh, C.; et al. Timed picture naming in seven languages. Psychon. Bull. Rev. 2003, 10, 344–380. [Google Scholar] [CrossRef] [Green Version]
- García, A.M.; Bocanegra, Y.; Herrera, E.; Pino, M.; Muñoz, E.; Sedeño, L.; Ibáñez, A. Action-semantic and syntactic deficits in subjects at risk for Huntington’s disease. J. Neuropsychol. 2017, 12, 389–408. [Google Scholar] [CrossRef] [PubMed]
- Bak, T.H. The neuroscience of action semantics in neurodegenerative brain diseases. Curr. Opin. Neurol. 2013, 26, 671–677. Available online: http://www.ncbi.nlm.nih.gov/pubmed/24184973 (accessed on 2 February 2021). [CrossRef] [PubMed]
- Da Silva, D.; Lemos, T.; De Sá Ferreira, A.; Horsczaruk, C.; Pedron, C.; De Carvalho Rodrigues, E.; de Oliveira, L.A.S. Effects of Acute Transcranial Direct Current Stimulation on Gait Kinematics of Individuals with Parkinson Disease. Top. Geriatr. Rehabil. 2018, 34, 262–268. [Google Scholar] [CrossRef]
- Adenzato, M.; Manenti, R.; Enrici, I.; Gobbi, E.; Brambilla, M.; Alberici, A.; Cotelli, M.S.; Padovani, A.; Borroni, B. Transcranial direct current stimulation enhances theory of mind in Parkinson’s disease patients with mild cognitive impairment: A randomized, double-blind, sham-controlled study. Transl. Neurodegener. 2019, 8, 1–13. [Google Scholar] [CrossRef]
- Boggio, P.S.; Ferrucci, R.; Rigonatti, S.P.; Covre, P.; Nitsche, M.; Pascual-Leone, A.; Fregni, F. Effects of transcranial direct current stimulation on working memory in patients with Parkinson’s disease. J. Neurol. Sci. 2006, 249, 31–38. [Google Scholar] [CrossRef]
- Kaski, D.; Kaski, D.; Allum, J.; Allum, J.; Bronstein, A.; Bronstein, A.; Dominguez, R.; Dominguez, R.; Kaski, D.; Kaski, D.; et al. Applying anodal tDCS during tango dancing in a patient with Parkinson’s disease. Neurosci. Lett. 2014, 568, 39–43. [Google Scholar] [CrossRef]
- Dubreuil-Vall, L.; Chau, P.; Ruffini, G.; Widge, A.S.; Camprodon, J.A. tDCS to the left DLPFC modulates cognitive and physiological correlates of executive function in a state-dependent manner. Brain Stimul. 2019, 12, 1456–1463. [Google Scholar] [CrossRef]
- Manor, B.; Zhou, J.; Harrison, R.; Lo, O.-Y.; Travison, T.G.; Hausdorff, J.M.; Pascual-Leone, A.; Lipsitz, L. Transcranial Direct Current Stimulation May Improve Cognitive-Motor Function in Functionally Limited Older Adults. Neurorehabilit. Neural Repair. 2018, 32, 788–798. [Google Scholar] [CrossRef]
- Foerster, Á.; Dutta, A.; Kuo, M.; Paulus, W.; Nitsche, M.A. Effects of anodal transcranial direct current stimulation over lower limb primary motor cortex on motor learning in healthy individuals. Eur. J. Neurosci. 2018, 47, 779–789. [Google Scholar] [CrossRef]
- Gandiga, P.C.; Hummel, F.C.; Cohen, L.G. Transcranial DC stimulation (tDCS): A tool for double-blind sham-controlled clinical studies in brain stimulation. Clin. Neurophysiol. 2006, 117, 845–850. [Google Scholar] [CrossRef]
- García, A.; Abrevaya, S.; Kozono, G.; Cordero, I.G.; Córdoba, M.; Kauffman, M.; Pautassi, R.M.; Muñoz, E.; Sedeño, L.; Ibáñez, A. The cerebellum and embodied semantics: Evidence from a case of genetic ataxia due toSTUB1mutations. J. Med. Genet. 2017, 54, 114–124. [Google Scholar] [CrossRef] [PubMed]
- Cervetto, S.; Abrevaya, S.; Caro, M.M.; Kozono, G.; Muñoz, E.; Ferrari, J.; Sedeño, L.; Ibáñez, A.; García, A.M. Action semantics at the bottom of the brain: Insights from dysplastic cerebellar gangliocytoma. Front. Psychol. 2018, 9, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Trevisan, P.; Sedeño, L.; Birba, A.; Ibáñez, A.; García, A.M. A moving story: Whole-body motor training selectively improves the appraisal of action meanings in naturalistic narratives. Sci. Rep. 2017, 7, 1–10. [Google Scholar] [CrossRef]
- Dottori, M.; Hesse, E.; Santilli, M.; Vilas, M.G.; Caro, M.M.; Fraiman, D.; Sedeño, L.; Ibáñez, A.; García, A. Task-specific signatures in the expert brain: Differential correlates of translation and reading in professional interpreters. Neuroimage 2020, 209, 116519. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Lawrence Erlbaum Associate: New York, NY, USA, 1988. [Google Scholar]
- Rodríguez-Ferreiro, J.; Menéndez, M.; Ribacoba, R.; Cuetos, F. Action naming is impaired in Parkinson disease patients. Neuropsychologia 2009, 47, 3271–3274. [Google Scholar] [CrossRef] [PubMed]
- Fernandino, L.; Conant, L.; Binder, J.; Blindauer, K.; Hiner, B.; Spangler, K.; Desai, R. Where is the action? Action sentence processing in Parkinson’s disease. Neuropsychologia 2013, 51, 1510–1517. [Google Scholar] [CrossRef] [Green Version]
- Fernandino, L.; Conant, L.; Binder, J.; Blindauer, K.; Hiner, B.; Spangler, K.; Desai, R. Parkinson’s disease disrupts both automatic and controlled processing of action verbs. Brain Lang. 2013, 127, 65–74. [Google Scholar] [CrossRef] [Green Version]
- Buccino, G.; Volta, R.D.; Arabia, G.; Morelli, M.; Chiriaco, C.; Lupo, A.; Silipo, F.; Quattrone, A. Processing graspable object images and their nouns is impaired in Parkinson’s disease patients. Cortex 2018, 100, 32–39. [Google Scholar] [CrossRef]
- Silveri, M.C.; Traficante, D.; Monaco, M.R.L.; Iori, L.; Sarchioni, F.; Burani, C. Word selection processing in Parkinson’s disease: When nouns are more difficult than verbs. Cortex 2018, 100, 8–20. [Google Scholar] [CrossRef] [Green Version]
- Signorini, M.; Volpato, C. Action fluency in Parkinson’s disease: A follow-up study. Mov. Disord. 2006, 21, 467–472. [Google Scholar] [CrossRef]
- Herrera, E.; Cuetos, F. Action naming in Parkinson’s disease patients on/off dopamine. Neurosci. Lett. 2012, 513, 219–222. [Google Scholar] [CrossRef]
- Bindman, L.J.; Lippold, O.C.J.; Redfearn, J.W.T. Long-lasting Changes in the Level of the Electrical Activity of the Cerebral Cortex produced by Polarizing Currents. Nature 1962, 196, 584–585. [Google Scholar] [CrossRef]
- Nitsche, M.A.; Paulus, W. Sustained excitability elevations induced by transcranial DC motor cortex stimulation in humans. Neurology 2001, 57, 1899–1901. [Google Scholar] [CrossRef]
- Monti, A.; Ferrucci, R.; Fumagalli, M.; Mameli, F.; Cogiamanian, F.; Ardolino, G.; Priori, A. Transcranial direct current stimulation (tDCS) and language. J. Neurol. Neurosurg. Psychiatry 2012, 84, 832–842. [Google Scholar] [CrossRef] [PubMed]
- Chesters, J.; Möttönen, R.; Watkins, K. Facilitating fluency in adults who stutter. Brain 2018, 141, 944–946. [Google Scholar]
- Biundo, R.; Weis, L.; Fiorenzato, E.; Gentile, G.; Giglio, M.; Schifano, R.; Campo, M.C.; Marcon, V.; Martinez-Martin, P.; Bisiacchi, P.; et al. Double-blind randomized trial of t-DCS versus sham in parkinson patients with mild cognitive impairment receiving cognitive training. Brain Stimul. 2015, 8, 1223–1225. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, B.J.; Gasson, N.; Johnson, A.; Booth, L.; Loftus, A.M. Cognitive Training and Transcranial Direct Current Stimulation for Mild Cognitive Impairment in Parkinson’s Disease: A Randomized Controlled Trial. Park. Dis. 2018, 2018, 1–12. [Google Scholar] [CrossRef]
- Vitale, F.; Padrón, I.; Avenanti, A.; de Vega, M. Enhancing Motor Brain Activity Improves Memory for Action Language: A tDCS Study. Cereb. Cortex 2021, 31, 1569–1581. [Google Scholar] [CrossRef] [PubMed]
- D’Ausilio, A.; Pulvermüller, F.; Salmas, P.; Bufalari, I.; Begliomini, C.; Fadiga, L. The Motor Somatotopy of Speech Perception. Curr. Biol. 2009, 19, 381–385. [Google Scholar] [CrossRef] [PubMed]
- Pulvermüller, F. Semantic embodiment, disembodiment or misembodiment? In search of meaning in modules and neuron circuits. Brain Lang. 2013, 127, 86–103. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pulvermüller, F. Neurobiological Mechanisms for Semantic Feature Extraction and Conceptual Flexibility. Top. Cogn. Sci. 2018, 10, 590–620. [Google Scholar] [CrossRef]
- Vukovic, N.; Feurra, M.; Shpektor, A.; Myachykov, A.; Shtyrov, Y. Primary motor cortex functionally contributes to language comprehension: An online rTMS study. Neuropsychologia 2017, 96, 222–229. [Google Scholar] [CrossRef] [Green Version]
- Lau, C.; Liu, M.; Chang, K.; Chang, A.; Bai, C.; Tseng, C.; Walsh, V.; Wang, H. Effect of single-session transcranial direct current stimulation on cognition in Parkinson’s disease. CNS Neurosci. Ther. 2019, 25, 1237–1243. [Google Scholar] [CrossRef]
- Marangolo, P.; Fiori, V.; Di Paola, M.; Cipollari, S.; Razzano, C.; Oliveri, M.; Caltagirone, C. Differential involvement of the left frontal and temporal regions in verb naming: A tDCS treatment study. Restor. Neurol. Neurosci. 2013, 31, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Cosentino, G.; Valentino, F.; Todisco, M.; Alfonsi, E.; Davì, R.; Savettieri, G.; Fierro, B.; D’Amelio, M.; Brighina, F. Effects of more-affected vs. Less-affected motor cortex tDCS in Parkinson’s disease. Front. Hum. Neurosci. 2017, 11, 309. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fiori, V.; Kunz, L.; Kuhnke, P.; Marangolo, P.; Hartwigsen, G. Transcranial direct current stimulation (tDCS) facilitates verb learning by altering effective connectivity in the healthy brain. Neuroimage 2018, 181, 550–559. [Google Scholar] [CrossRef]
- Suárez-García, D.; Birba, A.; Zimerman, M.; Diazgranados, J.; Lopes, P.; Ibáñez, A.; Grisales, J.S.; Londoño, J.F.C.; García, A. Clinical, Functional and Cognitive Data from Parkinson’s Disease Patients in an Action-Verb Processing tDCS Study. 2021. Available online: https://figshare.com/articles/dataset/Clinical_functional_and_cognitive_data_from_Parkinson_s_disease_patients_in_an_action-verb_processing_tDCS_study/14424149/1 (accessed on 15 April 2021).
| PD-atDCS (n = 11) | PD-stDCS (n = 11) | p-Value | Effect Size | |
|---|---|---|---|---|
| Age | 62.82 (7.49) | 66.45 (5.69) | 0.21 a | 0.53 c |
| Years of education | 11.45 (5.39) | 8.09 (5.24) | 0.15 a | 0.63 c |
| Sex (F:M) | 3:8 | 2:9 | 0.25 b | --- |
| Years since diagnosis | 4.45 (0.30) | 4.18 (3.84) | 0.85 a | 0.09 c |
| Hoehn and Yahr | 1.09 (0.30) | 1.09 (0.30) | 1.00 a | 0.00 c |
| MDS-UPDRS-III | 18.64 (7.35) | 19.45 (9.47) | 0.82 a | 0.95 c |
| GDS | 0.45 (0.82) | 0.91 (1.30) | 0.33 a | 0.42 c |
| Lawton and Brody | 8.00 (0.00) | 7.91 (0.30) | 0.32 a | 0.59 c |
| Barthel Index | 100 (0.00) | 98.64 (3.23) | 0.17 a | 0.59 c |
| ACE-R | 92.00 (6.08) | 87.27 (6.18) | 0.08 a | 0.77 c |
| IFS battery | 19.64 (4.69) | 19.09 (6.42) | 0.82 a | 0.09 c |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Suárez-García, D.M.A.; Birba, A.; Zimerman, M.; Diazgranados, J.A.; Lopes da Cunha, P.; Ibáñez, A.; Grisales-Cárdenas, J.S.; Cardona, J.F.; García, A.M. Rekindling Action Language: A Neuromodulatory Study on Parkinson’s Disease Patients. Brain Sci. 2021, 11, 887. https://doi.org/10.3390/brainsci11070887
Suárez-García DMA, Birba A, Zimerman M, Diazgranados JA, Lopes da Cunha P, Ibáñez A, Grisales-Cárdenas JS, Cardona JF, García AM. Rekindling Action Language: A Neuromodulatory Study on Parkinson’s Disease Patients. Brain Sciences. 2021; 11(7):887. https://doi.org/10.3390/brainsci11070887
Chicago/Turabian StyleSuárez-García, Diana M. A., Agustina Birba, Máximo Zimerman, Jesús A. Diazgranados, Pamela Lopes da Cunha, Agustín Ibáñez, Johan S. Grisales-Cárdenas, Juan Felipe Cardona, and Adolfo M. García. 2021. "Rekindling Action Language: A Neuromodulatory Study on Parkinson’s Disease Patients" Brain Sciences 11, no. 7: 887. https://doi.org/10.3390/brainsci11070887
APA StyleSuárez-García, D. M. A., Birba, A., Zimerman, M., Diazgranados, J. A., Lopes da Cunha, P., Ibáñez, A., Grisales-Cárdenas, J. S., Cardona, J. F., & García, A. M. (2021). Rekindling Action Language: A Neuromodulatory Study on Parkinson’s Disease Patients. Brain Sciences, 11(7), 887. https://doi.org/10.3390/brainsci11070887






