Intervention Effect of Non-Invasive Brain Stimulation on Cognitive Functions among People with Traumatic Brain Injury: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
- i.
- Examined the intervention efficacy of NIBS (tDCS and TMS) on cognitive functions (overall and sub-domains including attention, executive function, and memory) in TBI; and
- ii.
- Examined whether potential moderators (e.g., number of session and duration) affected the efficacy of NIBS in treating different cognitive outcomes among people with TBI.
- i.
- Active NIBS would help improve the cognitive functions and sub-domains when compared with sham NIBS among people with TBI, and
- ii.
- The moderators (number of session and duration) would affect the efficacy of NIBS in treating cognitive impairments among people with TBI.
2. Materials and Methods
- i.
- Participant (P)
- ii.
- Intervention (I)
- iii.
- Comparison (C)
- iv.
- Outcomes (O)
2.1. Search Strategy and Study Selection
2.2. Assessment for Quality of Reporting, Methodological Quality and Risk of Bias
2.3. Data Extraction
2.4. Data Analysis
3. Results
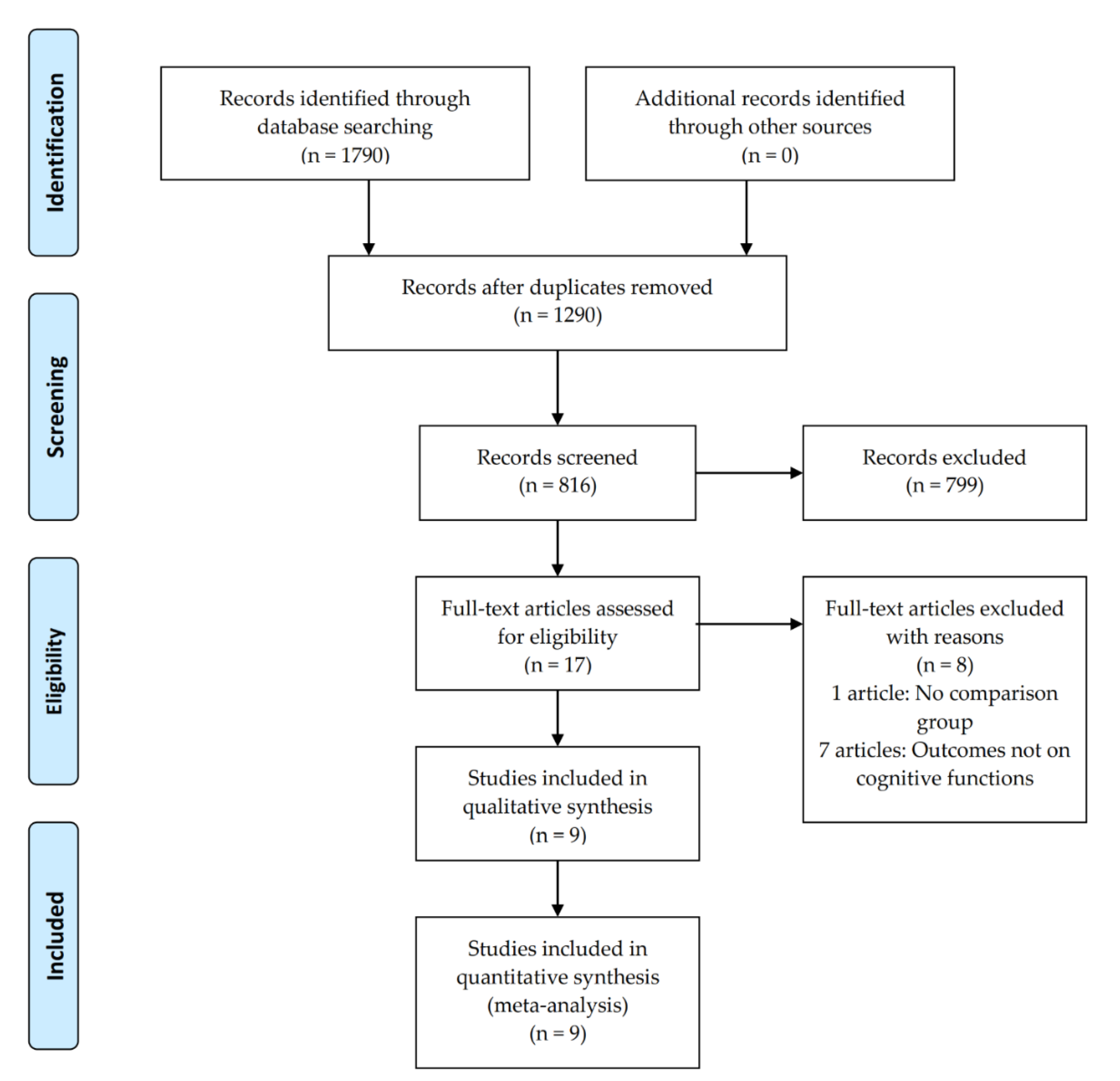
3.1. Study Selection
3.2. Characteristics of Included Studies
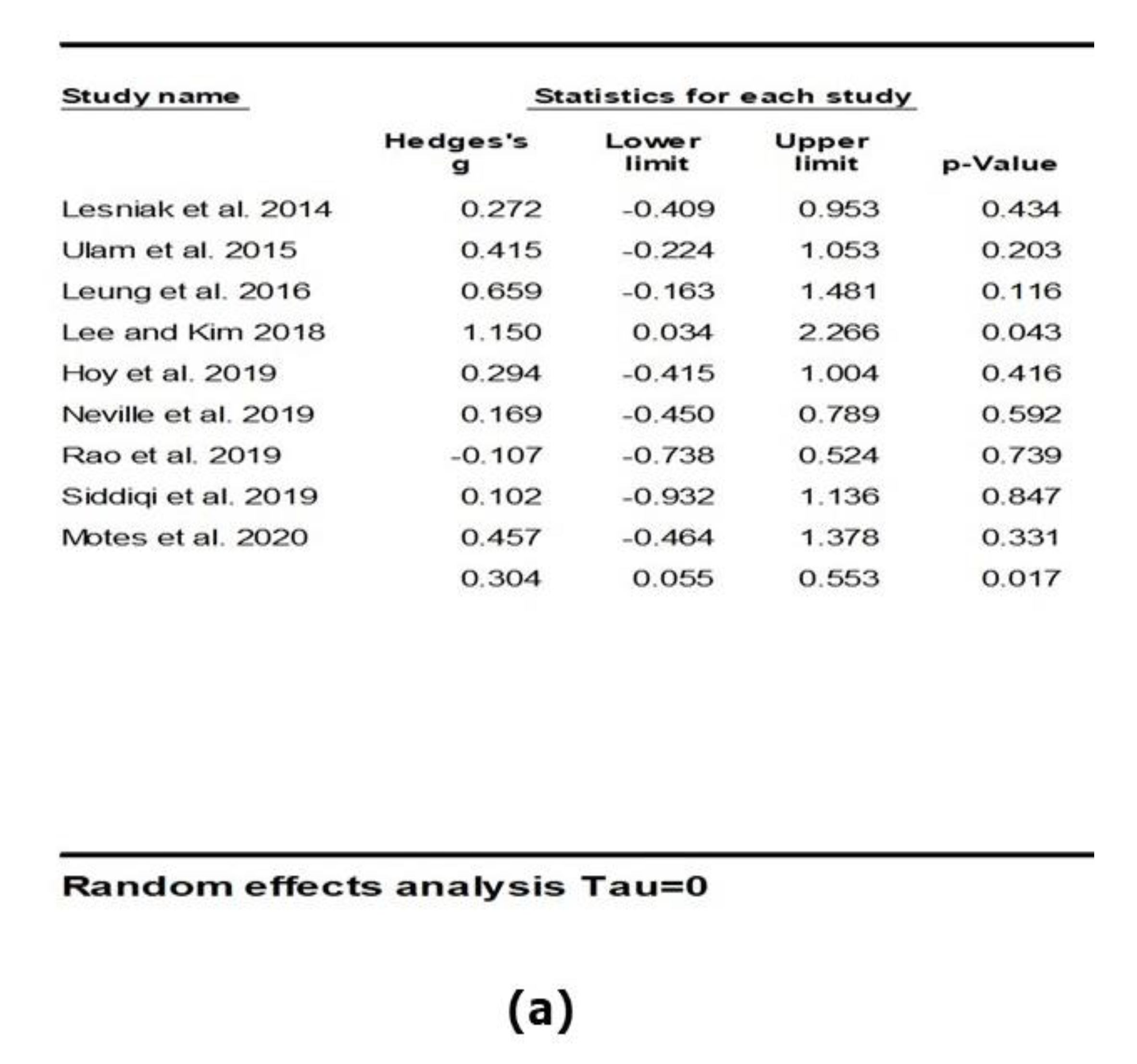
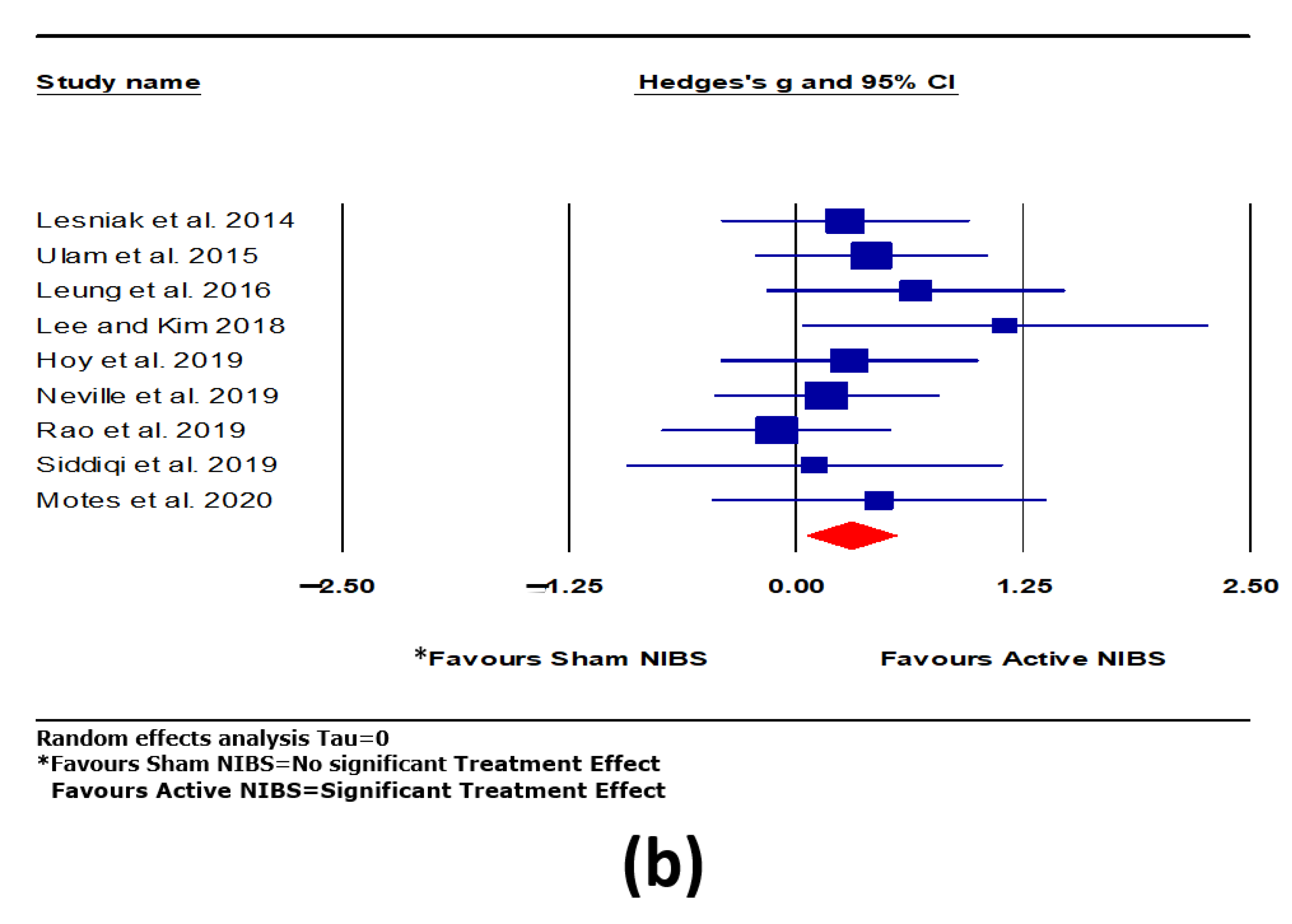
3.3. Intervention Effect on Overall Cognitive Outcomes
3.4. Intervention Effect on Domain-Specific Outcomes
3.4.1. Attention
3.4.2. Memory
3.4.3. Executive Function
3.4.4. Other Cognitive Domains
3.5. Identification and Analysis of Potential Moderators
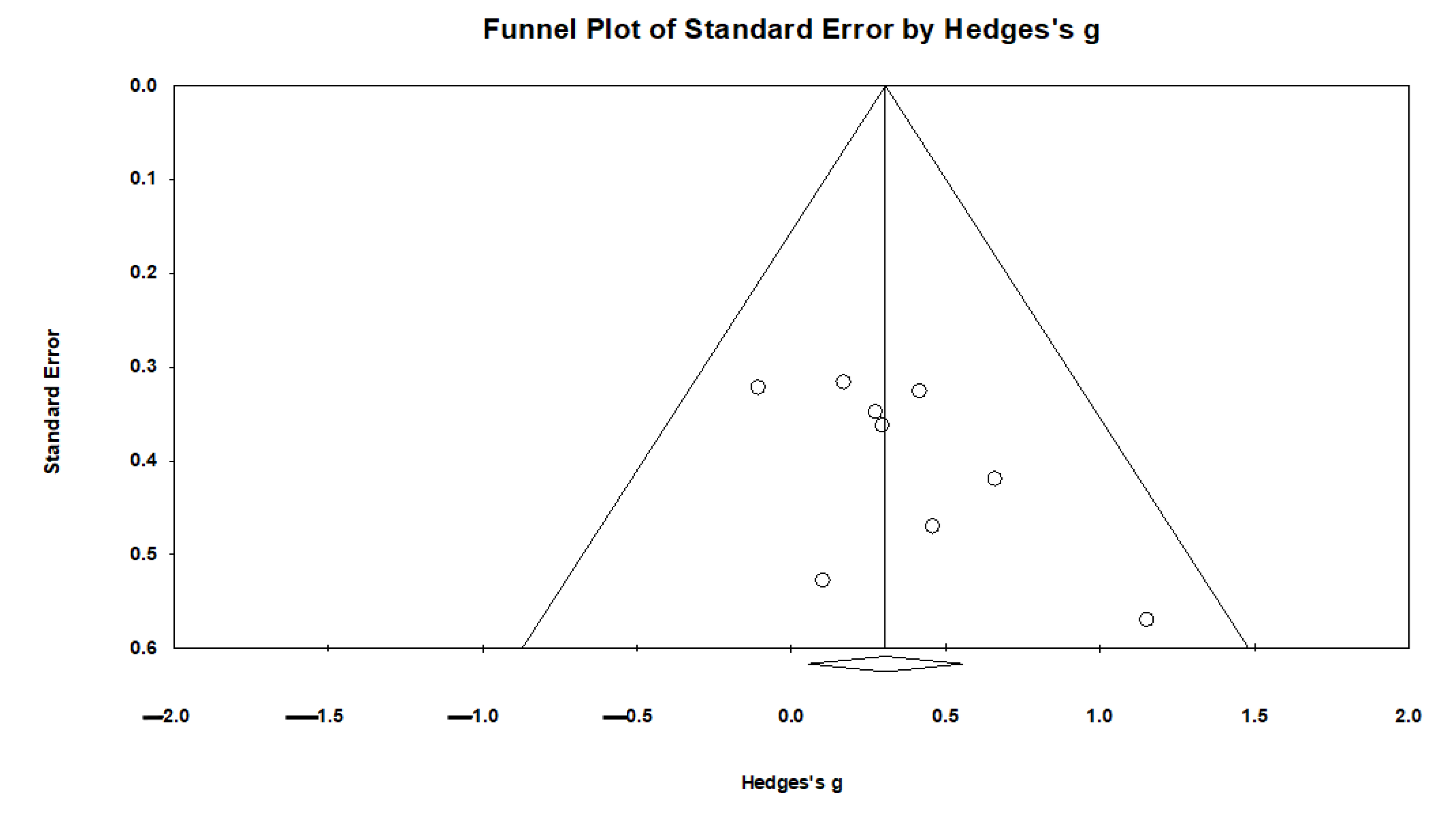
3.6. Sensitivity Analyses
4. Discussion
Limitations and Future Directions
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- McKee, A.C.; Daneshvar, D.H. The neuropathology of traumatic brain injury. In Handbook of Clinical Neurology; Elsevier: Amsterdam, The Netherlands, 2015; Volume 127, pp. 45–66. [Google Scholar]
- Wortzel, H.S.; Arciniegas, D.B. Treatment of post-traumatic cognitive impairments. Curr. Treat. Opt. Neurol. 2012, 14, 493–508. [Google Scholar] [CrossRef]
- Whiteneck, G.G.; Cuthbert, J.P.; Corrigan, J.D.; Bogner, J.A. Risk of Negative Outcomes after Traumatic Brain Injury: A Statewide Population-Based Survey. J. Head Trauma Rehabil. 2016, 31, E43–E54. [Google Scholar] [CrossRef] [PubMed]
- Willemse-van Son, A.H.P.; Ribbers, G.M.; Verhagen, A.P.; Stam, H.J. Prognostic factors of long-term functioning and productivity after traumatic brain injury: A systematic review of prospective cohort studies. Clin. Rehabil. 2007, 21, 1024–1037. [Google Scholar] [CrossRef] [PubMed]
- Santiago, L.A.; Oh, B.C.; Dash, P.K.; Holcomb, J.B.; Wade, C.E. A clinical comparison of penetrating and blunt traumatic brain injuries. Brain Inj. 2012, 26, 107–125. [Google Scholar] [CrossRef]
- Lloyd, J.; Wilson, M.L.; Tenovuo, O.; Saarijärvi, S. Outcomes from mild and moderate traumatic brain injuries among children and adolescents: A systematic review of studies from 2008–2013. Brain Inj. 2015, 29, 539–549. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Surveillance Report of Traumatic Brain Injury-Related Emergency Department Visits, Hospitalizations, and Deaths—United States. 2014. Available online: https://www.cdc.gov/traumaticbraininjury/get_the_facts.html (accessed on 11 July 2020).
- Dewan, M.C.; Rattani, A.; Gupta, S.; Baticulon, R.E.; Hung, Y.-C.; Punchak, M.; Agrawal, A.; Adeleye, A.O.; Shrime, M.G.; Rubiano, A.M.; et al. Estimating the global incidence of traumatic brain injury. J. Neurosurg. 2018, 130, 1080–1097. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Luo, C.; Chen, X.; Tao, L. Assessment of cognitive dysfunction in traumatic brain injury patients: A review. Forensic Sci. Res. 2017, 2, 174–179. [Google Scholar] [CrossRef]
- Dikmen, S.S.; Corrigan, J.D.; Levin, H.S.; Machamer, J.; Stiers, W.; Weisskopf, M.G. Cognitive Outcome Following Traumatic Brain Injury. J. Head Trauma Rehabil. 2009, 24, 430–438. [Google Scholar] [CrossRef] [PubMed]
- de Freitas Cardoso, M.G.; Faleiro, R.M.; de Paula, J.J.; Kummer, A.; Caramelli, P.; Teixeira, A.L.; de Souza, L.C.; Miranda, A.S. Cognitive Impairment Following Acute Mild Traumatic Brain Injury. Front. Neurol. 2019, 10, 198. [Google Scholar] [CrossRef]
- Chen, A.J.W.; D’Esposito, M. Traumatic Brain Injury: From Bench to Bedside to Society. Neuron 2010, 66, 11–14. [Google Scholar] [CrossRef][Green Version]
- Lew, H.L.; Poole, J.H.; Chiang, J.Y.; Lee, E.H.; Warden, D. Event-related potential in facial affect recognition: Potential clinical utility in patients with traumatic brain injury. J. Rehabil. Res. Dev. 2005, 42, 29–34. [Google Scholar] [CrossRef]
- Wintermark, M.; Sanelli, P.C.; Anzai, Y.; Tsiouris, A.J.; Whitlow, C.T.; Druzgal, T.J.; Gean, A.D.; Lui, Y.W.; Norbash, A.M.; Raji, C. Imaging evidence and recommendations for traumatic brain injury: Conventional neuroimaging techniques. J. Am. Coll. Radiol. 2015, 12, e1–e14. [Google Scholar] [CrossRef]
- Anzai, Y.; Minoshima, S. Imaging of traumatic brain injury: Current and future. Imaging Med. 2011, 3, 153–165. [Google Scholar] [CrossRef]
- Mena, J.H.; Sanchez, A.I.; Rubiano, A.M.; Peitzman, A.B.; Sperry, J.L.; Gutierrez, M.I.; Puyana, J.C. Effect of the modified Glasgow Coma Scale score criteria for mild traumatic brain injury on mortality prediction: Comparing classic and modified Glasgow Coma Scale score model scores of 13. J. Trauma 2011, 71, 1185–1193. [Google Scholar] [CrossRef]
- Teasdale, G.; Jennett, B. Assessment of coma and impaired consciousness: A Practical Scale. Lancet 1974, 304, 81–84. [Google Scholar] [CrossRef]
- Paszkiel, S. Characteristics of Question of Blind Source Separation Using Moore-Penrose Pseudoinversion for Reconstruction of EEG Signal. In Automation 2017; Springer: Cham, Switzerland, 2017; pp. 393–400. [Google Scholar]
- Paszkiel, S.; Hunek, W.; Shylenko, A. Project and Simulation of a Portable Device for Measuring Bioelectrical Signals from the Brain for States Consciousness Verification with Visualization on LEDs. In Challenges in Automation, Robotics and Measurement Techniques; Springer: Cham, Switzerland, 2016; pp. 25–35. [Google Scholar]
- Talsky, A.; Pacione, L.R.; Shaw, T.; Wasserman, L.; Lenny, A.; Verma, A.; Hurwitz, G.; Waxman, R.; Morgan, A.; Bhalerao, S. Pharmacological interventions for traumatic brain injury. BC Med. J. 2010, 53, 26–31. [Google Scholar]
- Gultekin, R.; Huang, S.; Clavisi, O.; Pattuwage, L.; König, T.C.; Gruen, R. Pharmacological interventions in traumatic brain injury: Can we rely on systematic reviews for evidence? Injury 2016, 47, 516–524. [Google Scholar] [CrossRef] [PubMed]
- Wiart, L.; Luauté, J.; Stefan, A.; Plantier, D.; Hamonet, J. Non pharmacological treatments for psychological and behavioural disorders following traumatic brain injury (TBI). A systematic literature review and expert opinion leading to recommendations. Ann. Phys. Rehabil. Med. 2016, 59, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Gary, K.W.; Neimeier, J.P.; Ward, J.; Lapane, K.L. Randomized controlled trials in adult traumatic brain injury. Brain Inj. 2012, 26, 1523–1548. [Google Scholar] [CrossRef] [PubMed]
- Dhaliwal, S.K.; Meek, B.P.; Modirrousta, M.M. Non-Invasive Brain Stimulation for the Treatment of Symptoms Following Traumatic Brain Injury. Front. Psychiatry 2015, 6, 119. [Google Scholar] [CrossRef]
- Demirtas-Tatlidede, A.; Vahabzadeh-Hagh, A.M.; Bernabeu, M.; Tormos, J.M.; Pascual-Leone, A. Noninvasive brain stimulation in traumatic brain injury. J. Head Trauma Rehabil. 2012, 27, 274–292. [Google Scholar] [CrossRef]
- Hallett, M. Transcranial Magnetic Stimulation: A Primer. Neuron 2007, 55, 187–199. [Google Scholar] [CrossRef]
- Hara, T.; Shanmugalingam, A.; McIntyre, A.; Burhan, A.M. The Effect of Non-Invasive Brain Stimulation (NIBS) on Attention and Memory Function in Stroke Rehabilitation Patients: A Systematic Review and Meta-Analysis. Diagnostics 2021, 11, 227. [Google Scholar] [CrossRef]
- Hara, T.; Shanmugalingam, A.; McIntyre, A.; Burhan, A.M. The Effect of Non-Invasive Brain Stimulation (NIBS) on Executive Functioning, Attention and Memory in Rehabilitation Patients with Traumatic Brain Injury: A Systematic Review. Diagnostics 2021, 11, 627. [Google Scholar] [CrossRef]
- Fleeman, J.A.; Stavisky, C.; Carson, S.; Dukelow, N.; Maier, S.; Coles, H.; Wager, J.; Rice, J.; Essaff, D.; Scherer, M. Integrating cognitive rehabilitation: A preliminary program description and theoretical review of an interdisciplinary cognitive rehabilitation program. NeuroRehabilitation 2015, 37, 471–486. [Google Scholar] [CrossRef]
- Kim, W.-S.; Lee, K.; Kim, S.; Cho, S.; Paik, N.-J. Transcranial direct current stimulation for the treatment of motor impairment following traumatic brain injury. J. Neuroeng. Rehabil. 2019, 16, 14. [Google Scholar] [CrossRef] [PubMed]
- Clayton, E.; Kinley-Cooper, S.K.; Weber, R.A.; Adkins, D.L. Brain stimulation: Neuromodulation as a potential treatment for motor recovery following traumatic brain injury. Brain Res. 2016, 1640, 130–138. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, B.; Li, N.; Li, Y.; Hou, J.; Duan, G.; Wu, D. Transcranial Direct Current Stimulation Over Prefrontal Areas Improves Psychomotor Inhibition State in Patients with Traumatic Brain Injury: A Pilot Study. Front. Neurosci. 2020, 14, 386. [Google Scholar] [CrossRef]
- Martens, K.M.; Pechacek, K.M.; Modrak, C.G.; Milleson, V.J.; Zhu, B.; Vonder Haar, C. Cathodal Transcranial Direct-Current Stimulation Selectively Decreases Impulsivity after Traumatic Brain Injury in Rats. J. Neurotrauma 2019, 36, 2827–2830. [Google Scholar] [CrossRef] [PubMed]
- Kang, E.-K.; Kim, D.-Y.; Paik, N.-J. Transcranial direct current stimulation of the left prefrontal cortex improves attention in patients with traumatic brain injury: A pilot study. J. Rehabil. Med. 2012, 44, 346–350. [Google Scholar] [CrossRef] [PubMed]
- Yoon, K.J.; Lee, Y.-T.; Chae, S.W.; Park, C.R.; Kim, D.Y. Effects of anodal transcranial direct current stimulation (tDCS) on behavioral and spatial memory during the early stage of traumatic brain injury in the rats. J. Neurol. Sci. 2016, 362, 314–320. [Google Scholar] [CrossRef] [PubMed]
- Zaninotto, A.L.; El-Hagrassy, M.M.; Green, J.R.; Babo, M.; Paglioni, V.M.; Benute, G.G.; Paiva, W.S. Transcranial direct current stimulation (tDCS) effects on traumatic brain injury (TBI) recovery: A systematic review. Dement. Neuropsychol. 2019, 13, 172–179. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Zaninotto, A.L.; Neville, I.S.; Paiva, W.S.; Nunn, D.; Fregni, F. Clinical utility of brain stimulation modalities following traumatic brain injury: Current evidence. Neuropsychiatr. Dis. Treat. 2015, 11, 1573–1586. [Google Scholar] [CrossRef] [PubMed]
- Middleton, A.; Fritz, S.L.; Liuzzo, D.M.; Newman-Norlund, R.; Herter, T.M. Using clinical and robotic assessment tools to examine the feasibility of pairing tDCS with upper extremity physical therapy in patients with stroke and TBI: A consideration-of-concept pilot study. NeuroRehabilitation 2014, 35, 741–754. [Google Scholar] [CrossRef] [PubMed]
- Yoon, K.J.; Lee, Y.-T.; Chung, P.-W.; Lee, Y.K.; Kim, D.Y.; Chun, M.H. Effects of Repetitive Transcranial Magnetic Stimulation on Behavioral Recovery during Early Stage of Traumatic Brain Injury in Rats. J. Korean Med. Sci. 2015, 30, 1496–1502. [Google Scholar] [CrossRef] [PubMed]
- Pachalska, M.; Łukowicz, M.; Kropotov, J.D.; Herman-Sucharska, I.; Talar, J. Evaluation of differentiated neurotherapy programs for a patient after severe TBI and long term coma using event-related potentials. Med. Sci. Monit. 2011, 17, CS120–CS128. [Google Scholar] [CrossRef][Green Version]
- Nardone, R.; Sebastianelli, L.; Versace, V.; Brigo, F.; Golaszewski, S.; Manganotti, P.; Saltuari, L.; Trinka, E. Repetitive transcranial magnetic stimulation in traumatic brain injury: Evidence from animal and human studies. Brain Res. Bull. 2020, 159, 44–52. [Google Scholar] [CrossRef]
- Hoy, K.E.; McQueen, S.; Elliot, D.; Herring, S.E.; Maller, J.J.; Fitzgerald, P.B. A pilot investigation of repetitive transcranial magnetic stimulation for post-traumatic brain injury depression: Safety, tolerability, and efficacy. J. Neurotrauma 2019, 36, 2092–2098. [Google Scholar] [CrossRef]
- Pink, A.E.; Williams, C.; Alderman, N.; Stoffels, M. The use of repetitive transcranial magnetic stimulation (rTMS) following traumatic brain injury (TBI): A scoping review. Neuropsychol. Rehabil. 2019, 1–27. [Google Scholar] [CrossRef]
- Lee, S.A.; Kim, M.-K. Effect of Low Frequency Repetitive Transcranial Magnetic Stimulation on Depression and Cognition of Patients with Traumatic Brain Injury: A Randomized Controlled Trial. Med. Sci. Monit. 2018, 24, 8789–8794. [Google Scholar] [CrossRef]
- Garland, J.S.; Jaskot, E.-M.; Taylor, J.; Buccellato, K.; Atwal, N.; Pasquina, P.F. 135 Evolution of a Study of Bilateral Prefrontal Transcranial Magnetic Stimulation (TMS) to Treat the Symptoms of Mild TBI (mTBI) and PTSD. CNS Spectr. 2020, 25, 285–286. [Google Scholar] [CrossRef][Green Version]
- Iliceto, A.; Seiler, R.L.; Sarkar, K. Repetitive Transcranial Magnetic Stimulation for Treatment of Depression in a Patient with Severe Traumatic Brain Injury. Ochsner. J. 2018, 18, 264–267. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A.; Group, P.-P. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst. Rev. 2015, 4, 1. [Google Scholar] [CrossRef] [PubMed]
- Shamseer, L.; Moher, D.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015: Elaboration and explanation. BMJ Br. Med. J. 2015, 349, g7647. [Google Scholar] [CrossRef] [PubMed]
- Eriksen, M.B.; Frandsen, T.F. The impact of patient, intervention, comparison, outcome (PICO) as a search strategy tool on literature search quality: A systematic review. J. Med. Libr. Assoc. 2018, 106, 420–431. [Google Scholar] [CrossRef] [PubMed]
- Moola, S.; Munn, Z.; Tufanaru, C.; Aromataris, E.; Sears, K.; Sfetcu, R.; Currie, M.; Lisy, K.; Qureshi, R.; Mattis, P.; et al. Chapter 7: Systematic reviews of etiology and risk. In JBI Manual for Evidence Synthesis; The Joanna Briggs Institute: Adelaide, Australia, 2020. [Google Scholar] [CrossRef]
- Tufanaru, C.; Munn, Z.; Aromataris, E.; Campbell, J.; Hopp, L. Chapter 3: Systematic reviews of effectiveness. In Joanna Briggs Institute Reviewer’s Manual; The Joanna Briggs Institute: Adelaide, Australia, 2020. [Google Scholar] [CrossRef]
- McFadden, W.C.; Walsh, H.; Richter, F.; Soudant, C.; Bryce, C.H.; Hof, P.R.; Fowkes, M.; Crary, J.F.; McKenzie, A.T. Perfusion fixation in brain banking: A systematic review. Acta Neuropathol. Commun. 2019, 7, 146. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V.A. Cochrane Handbook for Systematic Reviews of Interventions; John Wiley & Sons: Hoboken, NJ, USA, 2019. [Google Scholar]
- Dasanayaka, N.N.; Sirisena, N.; Samaranayake, N. The effects of meditation on length of telomeres in healthy individuals: A systematic review. Syst. Rev. 2020, 10, 1–6. [Google Scholar] [CrossRef]
- Bown, M.J.; Sutton, A.J. Quality Control in Systematic Reviews and Meta-analyses. Eur. J. Vasc. Endovasc. Surg. 2010, 40, 669–677. [Google Scholar] [CrossRef]
- Siddiqi, S.H.; Trapp, N.T.; Hacker, C.D.; Laumann, T.O.; Kandala, S.; Hong, X.; Trillo, L.; Shahim, P.; Leuthardt, E.C.; Carter, A.R.; et al. Repetitive Transcranial Magnetic Stimulation with Resting-State Network Targeting for Treatment-Resistant Depression in Traumatic Brain Injury: A Randomized, Controlled, Double-Blinded Pilot Study. J. Neurotrauma 2019, 36, 1361–1374. [Google Scholar] [CrossRef]
- Rao, G.; Lopez-Jimenez, F.; Boyd, J.; D’Amico, F.; Durant Nefertiti, H.; Hlatky Mark, A.; Howard, G.; Kirley, K.; Masi, C.; Powell-Wiley Tiffany, M.; et al. Methodological Standards for Meta-Analyses and Qualitative Systematic Reviews of Cardiac Prevention and Treatment Studies: A Scientific Statement From the American Heart Association. Circulation 2017, 136, e172–e194. [Google Scholar] [CrossRef] [PubMed]
- Motes, M.; Spence, J.; Yeatman, K.; Jones, P.; Lutrell, M.; O’Hair, R.; Shakal, S.; De La Rosa, B.; To, W.; Vanneste, S.; et al. High-Definition Transcranial Direct Current Stimulation to Improve Verbal Retrieval Deficits in Chronic Traumatic Brain Injury. J. Neurotrauma 2020, 37, 170–177. [Google Scholar] [CrossRef]
- Hallock, H.; Collins, D.; Lampit, A.; Deol, K.; Fleming, J.; Valenzuela, M. Cognitive Training for Post-Acute Traumatic Brain Injury: A Systematic Review and Meta-Analysis. Front. Hum. Neurosci. 2016, 10, 537. [Google Scholar] [CrossRef]
- Olkin, I.; Gleser, L. Stochastically dependent effect sizes. In The Handbook of Research Synthesis and Meta-Analysis, 2nd ed.; Cooper, H., Hedges, L., Valentine, J., Eds.; Russell Sage Foundation: New York, NY, USA, 2009; pp. 357–376. [Google Scholar]
- Sterne, J.A.C.; Sutton, A.J.; Ioannidis, J.P.A.; Terrin, N.; Jones, D.R.; Lau, J.; Carpenter, J.; Rücker, G.; Harbord, R.M.; Schmid, C.H.; et al. Recommendations for examining and interpreting funnel plot asymmetry in meta-analyses of randomised controlled trials. BMJ 2011, 343, d4002. [Google Scholar] [CrossRef] [PubMed]
- Strauss, E.; Sherman, E.M.; Spreen, O. A Compendium of Neuropsychological Tests: Administration, Norms, and Commentary; Oxford University Press: Oxford, UK, 2006. [Google Scholar]
- Lampit, A.; Hallock, H.; Valenzuela, M. Computerized cognitive training in cognitively healthy older adults: A systematic review and meta-analysis of effect modifiers. PLoS Med. 2014, 11, e1001756. [Google Scholar] [CrossRef] [PubMed]
- Borenstein, M.; Hedges, L.; Higgins, J.; Rothstein, H. Introduction to Meta-Analysis; John Wiley & Sons Ltd.: Chichester, UK, 2009. [Google Scholar]
- Rao, V.; Bechtold, K.; McCann, U.; Roy, D.; Peters, M.; Vaishnavi, S.; Yousem, D.; Mori, S.; Yan, H.; Leoutsakos, J.; et al. Low-Frequency Right Repetitive Transcranial Magnetic Stimulation for the Treatment of Depression After Traumatic Brain Injury: A Randomized Sham-Controlled Pilot Study. J. Neuropsychiatry Clin. Neurosci. 2019, 31, 306–318. [Google Scholar] [CrossRef]
- Ulam, F.; Shelton, C.; Richards, L.; Davis, L.; Hunter, B.; Fregni, F.; Higgins, K. Cumulative effects of transcranial direct current stimulation on EEG oscillations and attention/working memory during subacute neurorehabilitation of traumatic brain injury. Clin. Neurophysiol. 2015, 126, 486–496. [Google Scholar] [CrossRef]
- Leung, A.; Shukla, S.; Fallah, A.; Song, D.; Lin, L.; Golshan, S.; Tsai, A.; Jak, A.; Polston, G.; Lee, R. Repetitive Transcranial Magnetic Stimulation in Managing Mild Traumatic Brain Injury-Related Headaches. Neuromodul. Technol. Neural Interface 2016, 19, 133–141. [Google Scholar] [CrossRef]
- Lesniak, M.; Polanowska, K.; Seniów, J.; Czlonkowska, A. Effects of Repeated Anodal tDCS Coupled with Cognitive Training for Patients With Severe Traumatic Brain Injury: A Pilot Randomized Controlled Trial. J. Head Trauma Rehabil. 2014, 29, E20–E29. [Google Scholar] [CrossRef]
- Neville, I.S.; Zaninotto, A.L.; Hayashi, C.Y.; Rodrigues, P.A.; Galhardoni, R.; Ciampi de Andrade, D.; Brunoni, A.R.; Amorim, R.L.O.; Teixeira, M.J.; Paiva, W.S. Repetitive TMS does not improve cognition in patients with TBI: A randomized double-blind trial. Neurology 2019, 93, e190–e199. [Google Scholar] [CrossRef]
- Li, H.; Li, J.; Li, N.; Li, B.; Wang, P.; Zhou, T. Cognitive intervention for persons with mild cognitive impairment: A meta-analysis. Ageing Res. Rev. 2011, 10, 285–296. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, P.A.; Schwippel, T.; Wolz, I.; Svaldi, J. Meta-analysis of the effects of transcranial direct current stimulation on inhibitory control. Brain Stimul. 2020, 13, 1159–1167. [Google Scholar] [CrossRef] [PubMed]
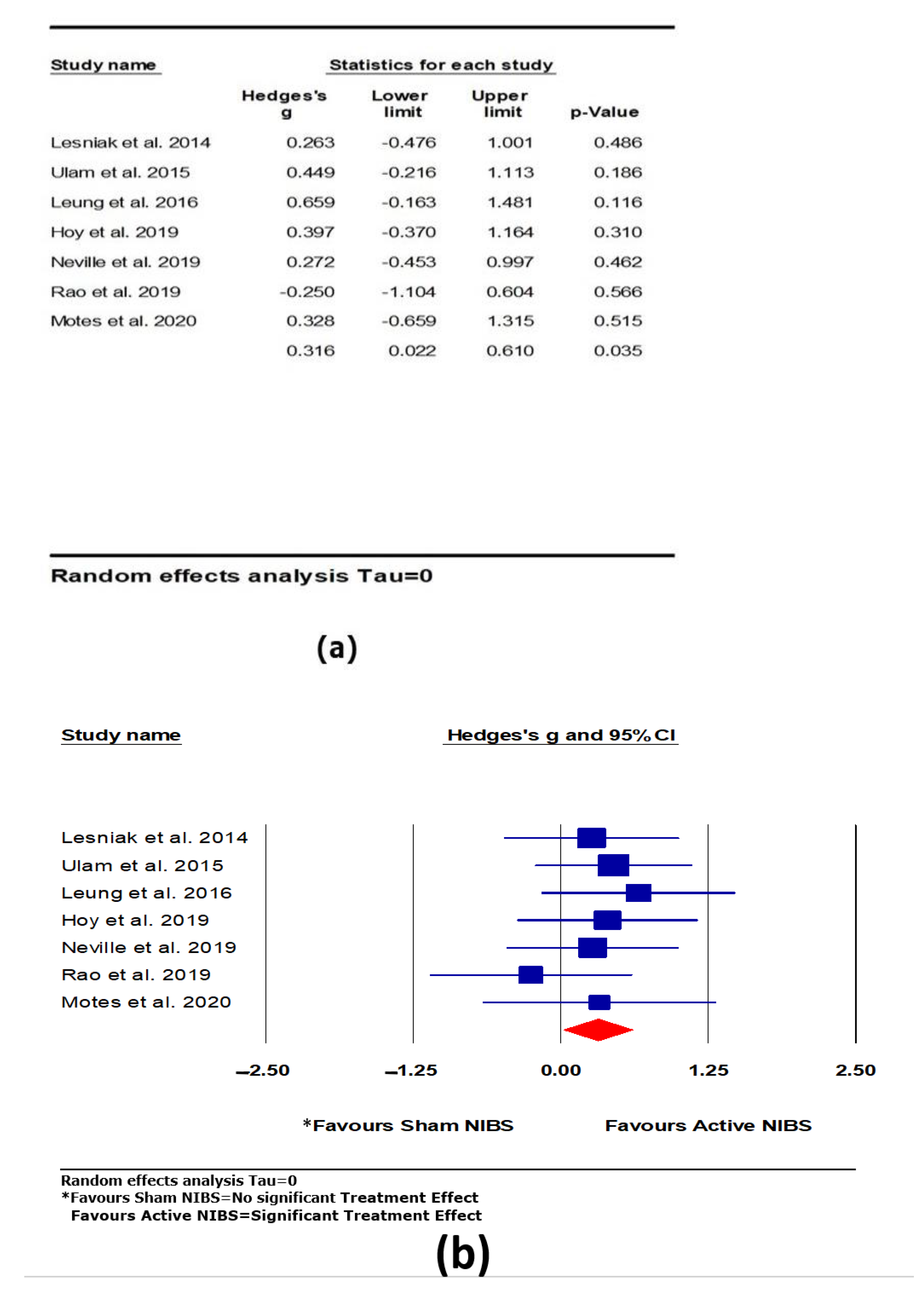
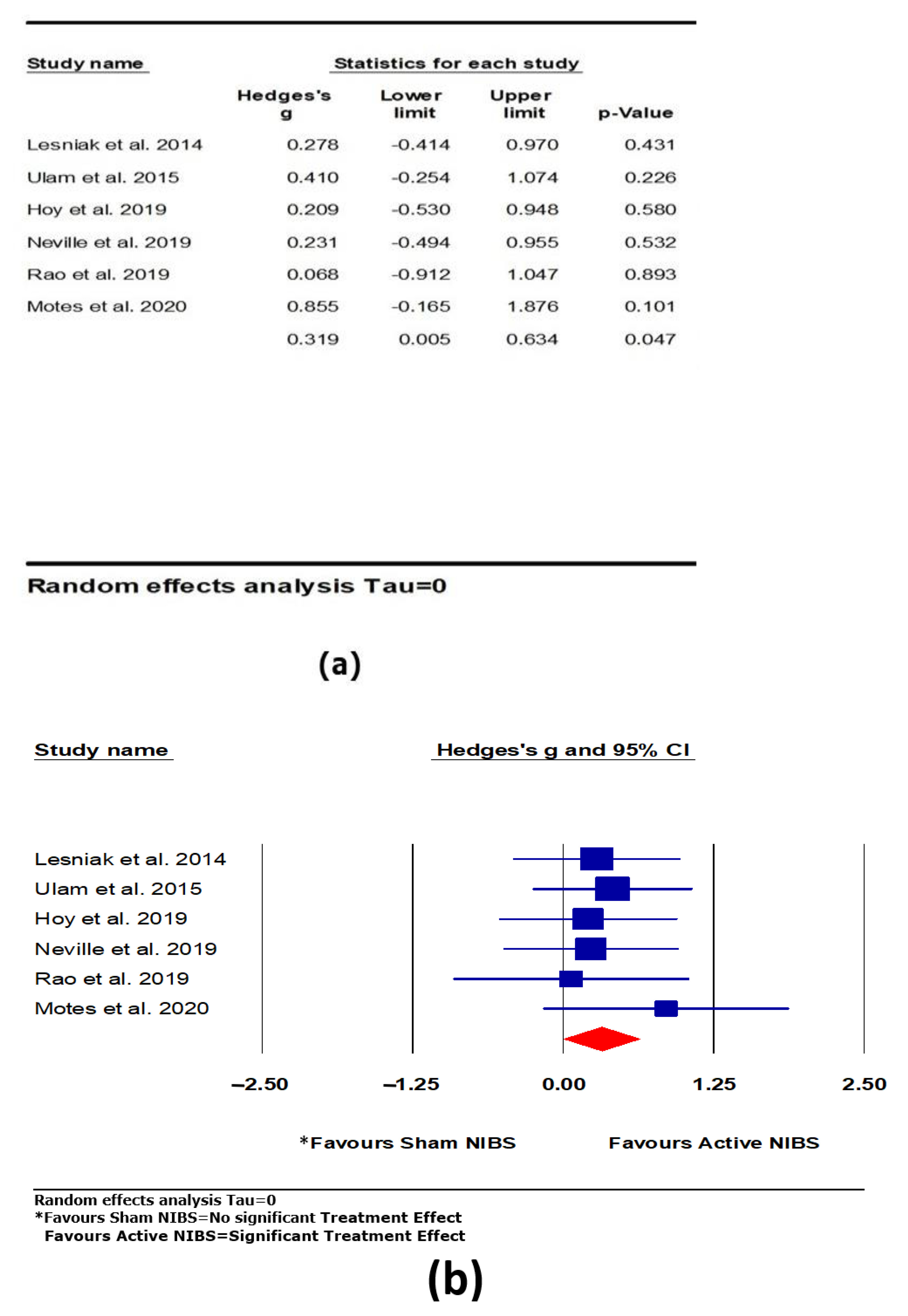
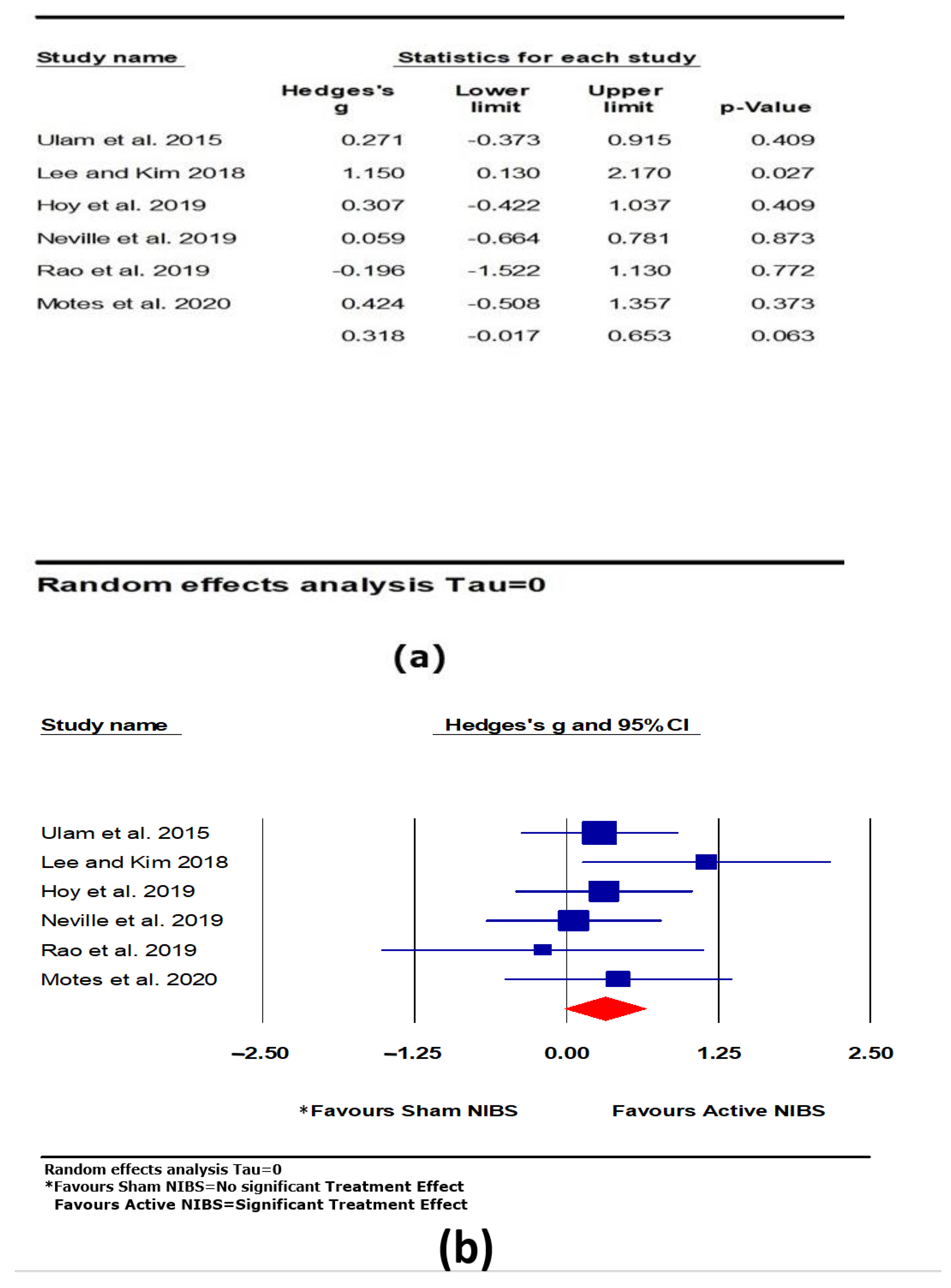
| Author and Year | N (Active, Sham) | Age Active, Age Sham | Study Design | Treatment Type | No of Sessions | Duration (min/day) | Outcomes | Quality Appraisal | Risk of Bias |
|---|---|---|---|---|---|---|---|---|---|
| Hoy et al. (2019) [38] | 21 (11, 10) | 41.27 ± 10.04, 51.80 ± 13.38 | RCT | Bilateral dlPFC rTMS | 20 | ― 20 days | Attention Executive function Memory | High | Low |
| Lee and Kim (2018) [40] | 13 (7, 6) | 42.42 ± 11.32, 41.33 ± 11.02 | RCT | Right dlPFC rTMS | 10 | 30 min/day | Executive function | High | Low |
| Lesniak et al. (2014) [55] | 23 (12, 11) | 28.3 ± 9, 29.3 ± 7.7 | RCT | Left dlPFC Anode tDCS | 15 | 10 min/day | Attention Memory | High | Low |
| Leung et al. (2016) [56] | 24 (12, 12) | 41 ± 14, 41 ± 12 | RCT | LMC rTMS | 3 | ― 3 days a | Attention | High | High |
| Motes et al. (2020) [57] | 15 (9, 6) | 40.9 ± 5.0, 40.8 ± 10.9 | Non-RCT | preSMA/dACC Anode tDCS | 10 | 20 min/day | Attention Executive function Memory Language | High | N/A |
| Neville et al. (2019) [58] | 30 (17, 13) | 32.62 ± 12.81, 29.0 ± 10.35 | RCT | Left dlPFC rTMS | 10 | ― 10 days | Attention Executive function Memory Motor function | High | Low |
| Rao et al. (2019) [59] | 30 (13, 17) | 39.8 ± 14.2, 40.2 ± 14.6 | RCT | LFR dlPFC rTMS | 20 | ― 20 days | Global cognition Attention Executive function Memory | High | Low |
| Siddiqi et al. (2019) [60] | 15 (9, 6) | 43 ± 13, 50 ± 18 | RCT | Bilateral dlPFC rTMS | 20 | ― 20 days b | Fluid cognition Crystallised cognition Overall cognition | High | Low |
| Ulam et al. (2015) [61] | 26 (13, 13) | 31.34 ± 9.8, 35.70 ± 14.7 | RCT | Left dlPFC Anode tDCS | 10 | 20 min/day | Attention Executive function Memory Social perception | High | Low |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahorsu, D.K.; Adjaottor, E.S.; Lam, B.Y.H. Intervention Effect of Non-Invasive Brain Stimulation on Cognitive Functions among People with Traumatic Brain Injury: A Systematic Review and Meta-Analysis. Brain Sci. 2021, 11, 840. https://doi.org/10.3390/brainsci11070840
Ahorsu DK, Adjaottor ES, Lam BYH. Intervention Effect of Non-Invasive Brain Stimulation on Cognitive Functions among People with Traumatic Brain Injury: A Systematic Review and Meta-Analysis. Brain Sciences. 2021; 11(7):840. https://doi.org/10.3390/brainsci11070840
Chicago/Turabian StyleAhorsu, Daniel Kwasi, Emma Sethina Adjaottor, and Bess Yin Hung Lam. 2021. "Intervention Effect of Non-Invasive Brain Stimulation on Cognitive Functions among People with Traumatic Brain Injury: A Systematic Review and Meta-Analysis" Brain Sciences 11, no. 7: 840. https://doi.org/10.3390/brainsci11070840
APA StyleAhorsu, D. K., Adjaottor, E. S., & Lam, B. Y. H. (2021). Intervention Effect of Non-Invasive Brain Stimulation on Cognitive Functions among People with Traumatic Brain Injury: A Systematic Review and Meta-Analysis. Brain Sciences, 11(7), 840. https://doi.org/10.3390/brainsci11070840







