Deficits in Early Sensory and Cognitive Processing Are Related to Phase and Nonphase EEG Activity in Multiple Sclerosis Patients
Abstract
1. Introduction
2. Methods
2.1. Participants
2.2. Cognitive Task
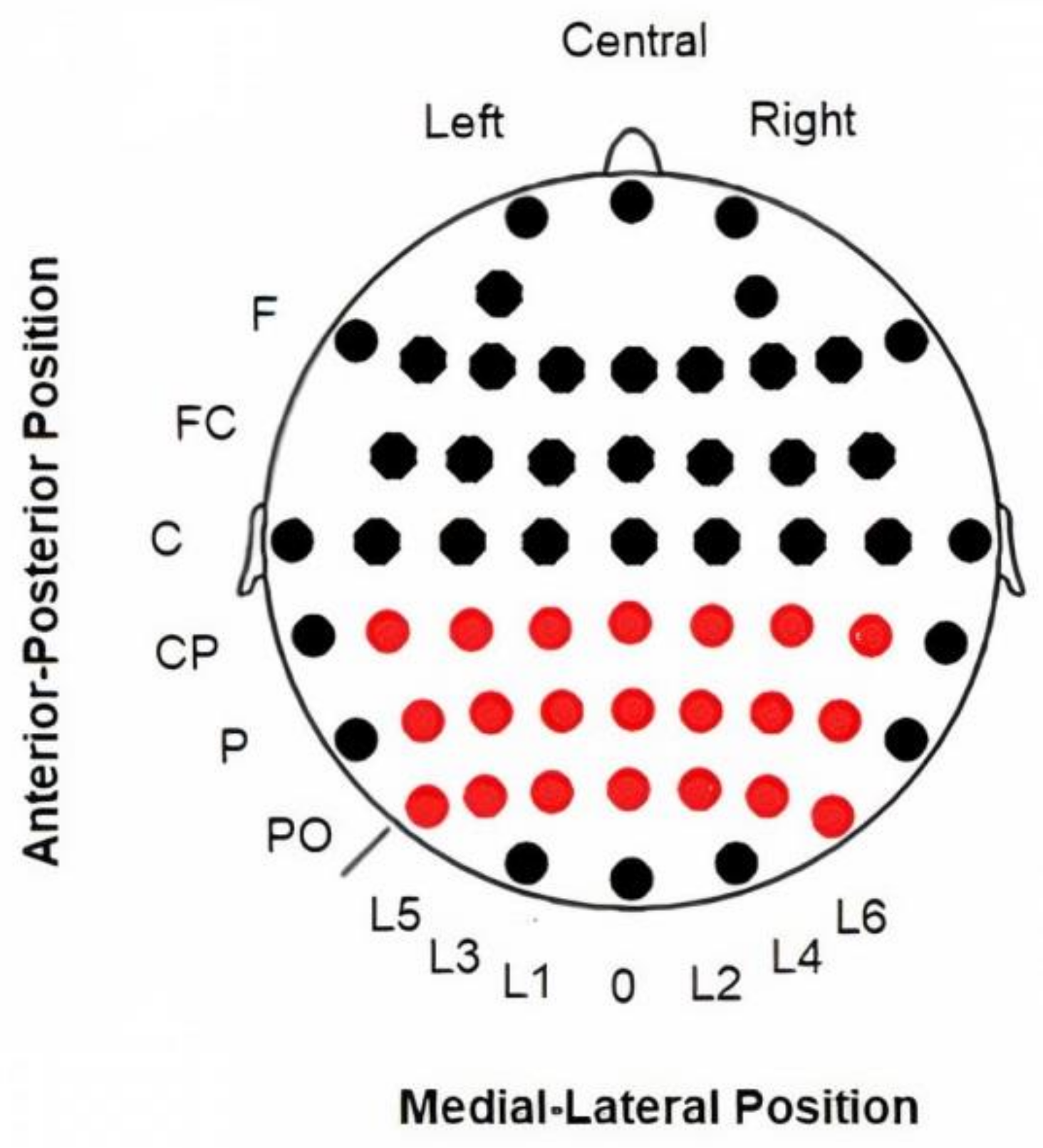
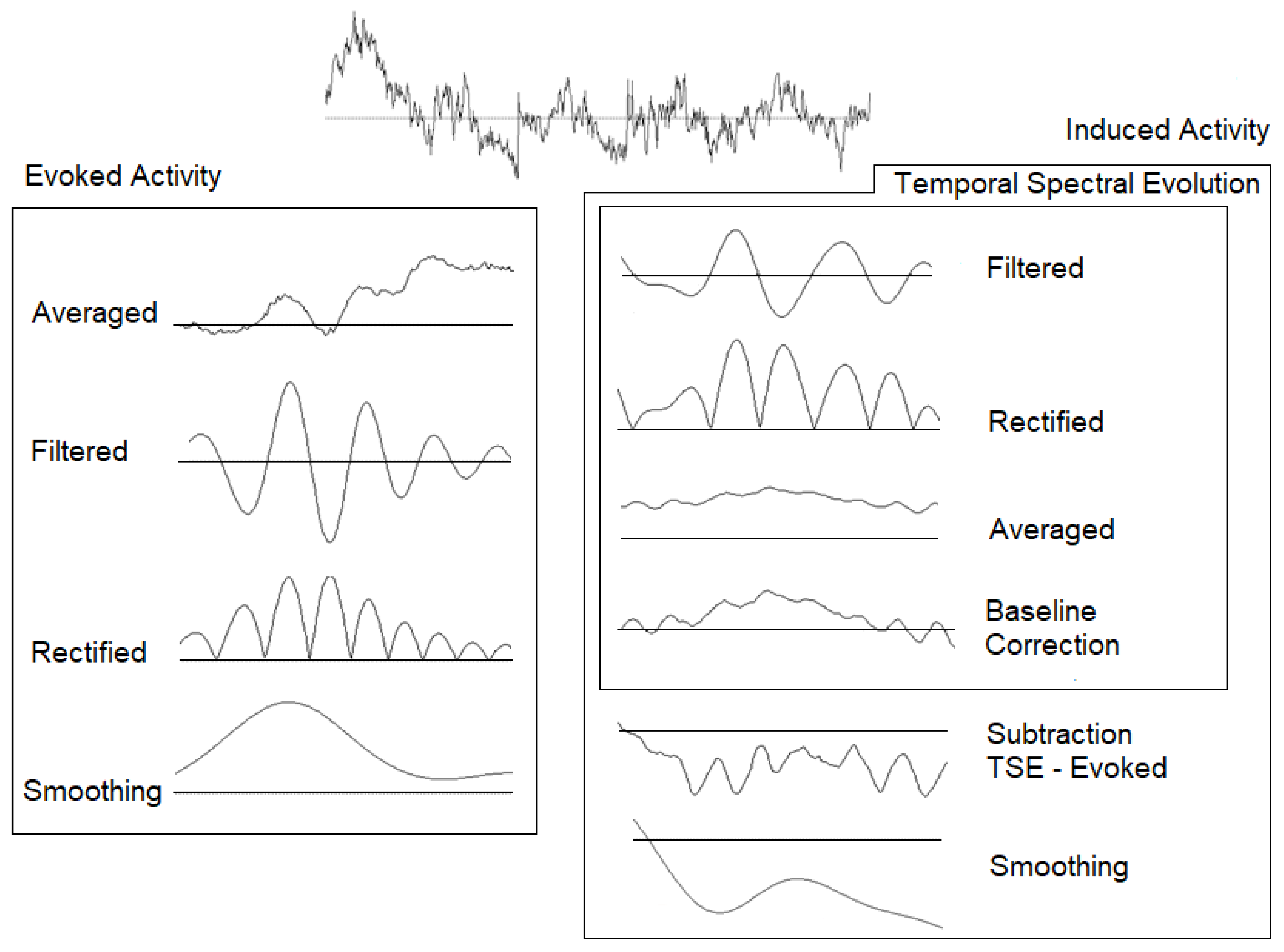
2.3. Electroencephalography (EEG) Recording and Analyses
2.4. Statistical Analyses
2.4.1. Behavioural Responses
2.4.2. Alpha Band
2.4.3. Gamma Band
3. Result
3.1. Behavioural Data
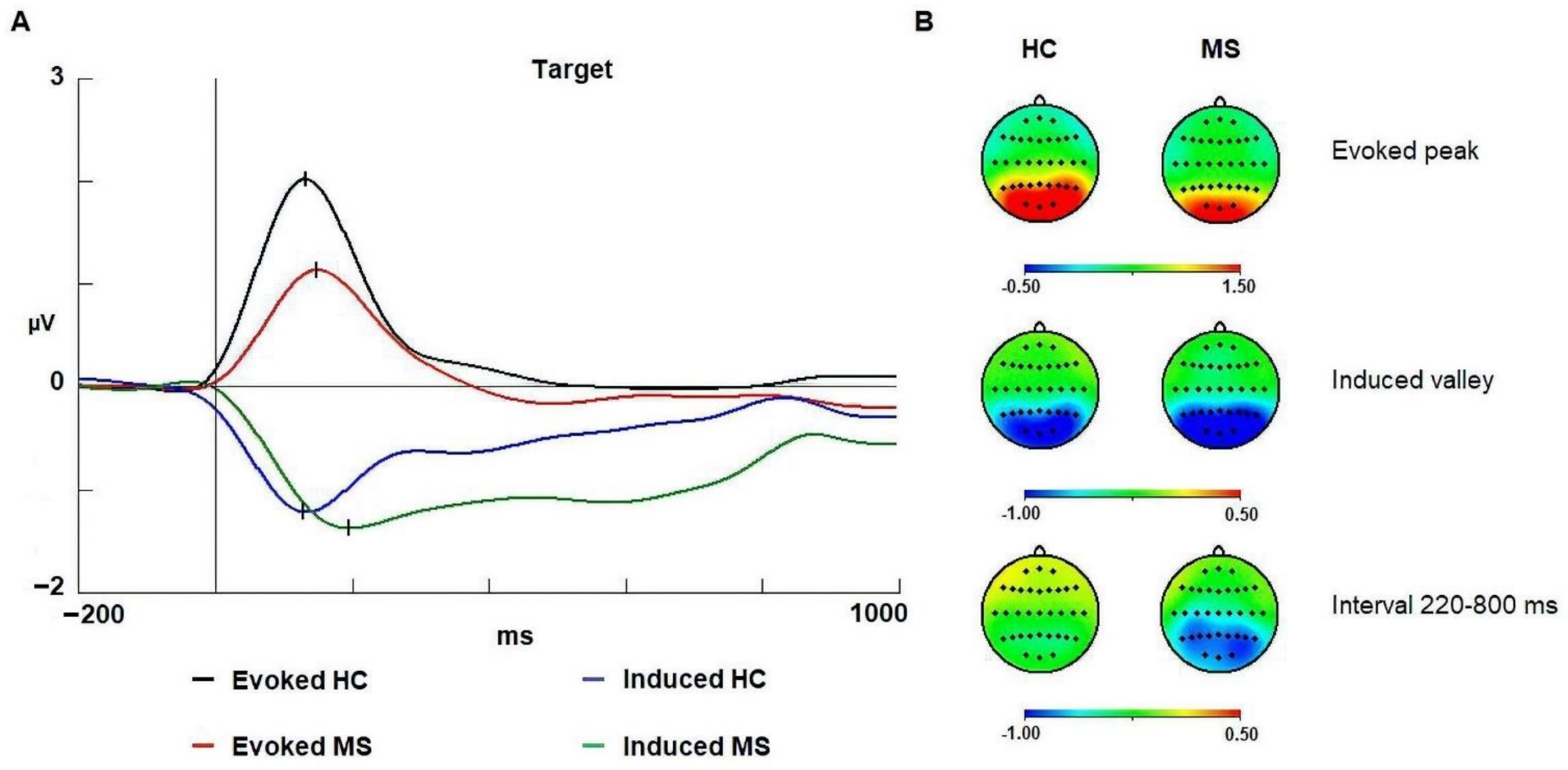
3.2. Alpha Band
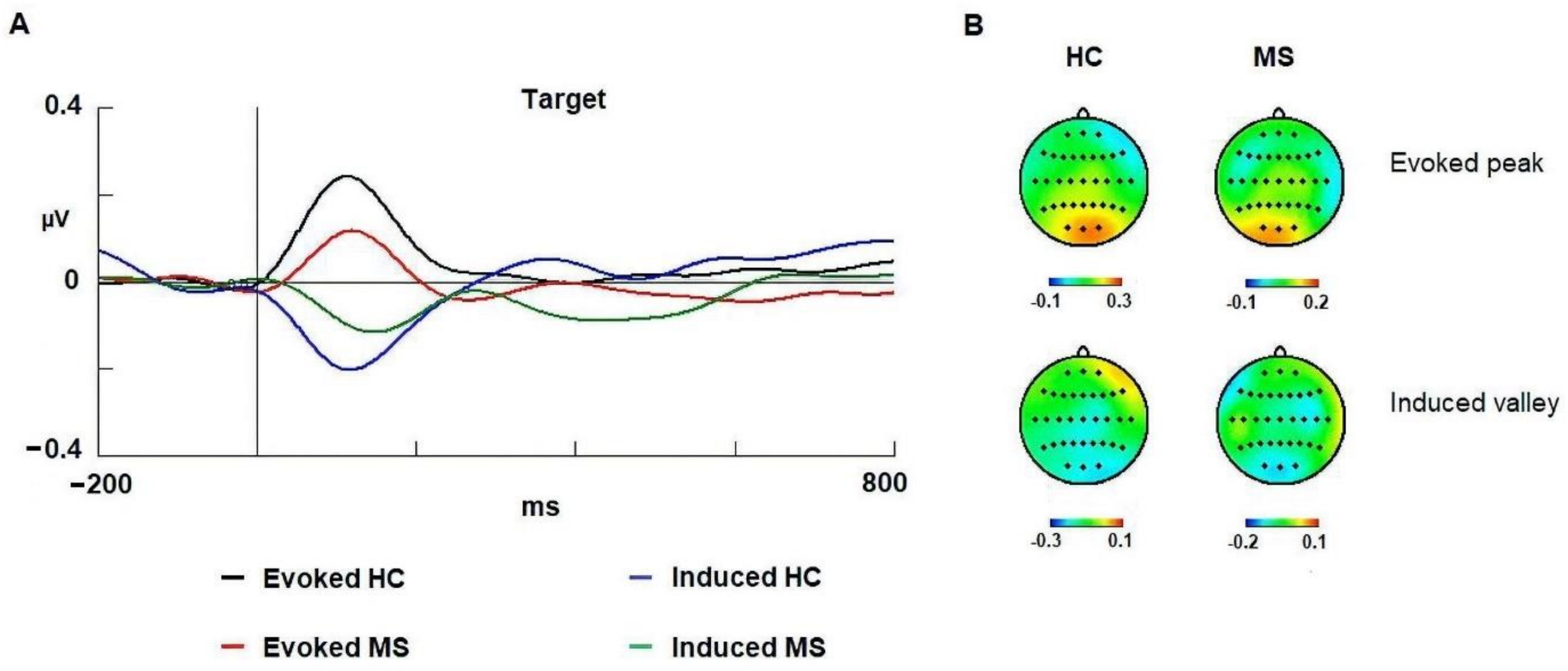
3.3. Gamma Band
4. Discussion
4.1. Evoked Alpha
4.2. Induced Alpha
4.3. Evoked Gamma
4.4. Induced Gamma
4.5. Evoked and Induced Activity
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Calabresi, P.A. Multiple sclerosis and demyelinating conditions of the central nervous system. In Goldman’s Cecil Medicine, 24th ed.; Goldman, L., Schafer, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2016; Volume 2, pp. 2347–2355. [Google Scholar]
- Tarlinton, R.E.; Khaibullin, T.; Granatov, E.; Martynova, E.; Rizvanov, A.; Khaiboullina, S. The Interaction between Viral and Environmental Risk Factors in the Pathogenesis of Multiple Sclerosis. Int. J. Mol. Sci. 2019, 20, 303. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Rosa, J.J.; Vazquez-Marrufo, M.; Vaquero, E.; Duque, P.; Borges, M.; Gomez-Gonzalez, C.M.; Izquierdo, G. Cluster analysis of behavioural and event-related potentials during a contingent negative variation paradigm in remitting-relapsing and benign forms of multiple sclerosis. BMC Neurol. 2011, 11, 64. [Google Scholar] [CrossRef] [PubMed]
- Moral-Torres, E.; Fernández-Fernández, O.; Carrascal-Rueda, P.; Ruíz-Beato, E.; Estella-Pérez, E.; Manzanares-Estrada, R.; Gómez-García, T.; Jiménez, M.; Hidalgo-Vega, A.; Merino, M. Social value of a set of proposals for the ideal approach of multiple sclerosis within the Spanish National Health System: A social return on investment study. BMC Health Serv. Res. 2020, 20, 84. [Google Scholar] [CrossRef]
- Ellger, T.; Bethke, F.; Frese, A.; Luettmann, R.J.; Buchheister, A.; Ringelstein, E.B.; Evers, S. Event-related potentials in different subtypes of multiple sclerosis: A cross-sectional study. J. Neurol. Sci. 2002, 205, 35–40. [Google Scholar] [CrossRef]
- Piras, M.R.; Magnano, I.; Canu, E.D.G.; Paulus, K.S.; Satta, W.M.; Soddu, A.; Conti, M.; Achene, A.; Solinas, G.; Aiello, I. Longitudinal study of cognitive dysfunction in multiple sclerosis: Neuropsychological, neuroradiological, and neurophysiological findings. J. Neurol. Neurosurg. Psychiatry 2003, 74, 878–885. [Google Scholar] [CrossRef]
- Vázquez-Marrufo, M.; González-Rosa, J.; Vaquero-Casares, E.; Duque, P.; Borges, M.; Izquierdo, G. Cognitive evoked potentials in remitting-relapsing and benign forms of multiple sclerosis. Rev. Neurol. 2009, 48, 453–458. [Google Scholar]
- López-Góngora, M.; Escartín, A.; Martínez-Horta, S.; Fernández-Bobadilla, R.; Querol, L.; Romero, S.; Mañanas, M.A.; Riba, J. Neurophysiological evidence of compensatory brain mechanisms in early stage multiple sclerosis. PLoS ONE 2015, 10, e0136786. [Google Scholar]
- Whelan, R.; Lonergan, R.; Kiiski, H.; Nolan, H.; Kinsella, K.; Bramham, J.; O´Brien, M.; Reilly, R.B.; Hutchinson, M.; Tubridy, N. A high-density ERP study reveals latency, amplitude, and topographical differences in multiple sclerosis patients versus controls. Clin. Neurophysiol. 2010, 121, 1420–1426. [Google Scholar] [CrossRef]
- Kiiski, H.; Reilly, R.B.; Lonergan, R.; Kelly, S.; O´Brien, M.; Kinsella, K.; Bramham, J.; Burke, T.; O´Donnchadha, S.; Nolan, H.; et al. Change in PASAT performance correlates with change in P3 erp amplitude over a 12-month period in multiple sclerosis patients. J. Neurol. Sci. 2011, 305, 45–52. [Google Scholar] [CrossRef]
- Kimiskidis, V.K.; Papaliagkas, V.; Sotirakoglou, K.; Kouvatsou, Z.K.; Kapina, V.K.; Papadaki, E.; Tsimourtou, V.; Masoura, E.; Kazis, D.A.; Papayiannopoulos, S.; et al. Cognitive event-related potentials in multiple sclerosis: Correlation with MRI and neuropsychological findings. Mult. Scler. Relat. Disord. 2016, 10, 192–197. [Google Scholar] [CrossRef]
- Kiiski, H.; Jollans, L.; Donnchadha, S.Ó.; Nolan, H.; Lonergan, R.; Kelly, S.; O’Brien, M.C.; Kinsella, K.; Bramham, J.; Burke, T.; et al. Machine learning EEG to predict cognitive functioning and processing speed over a 2-year period in multiple sclerosis patients and controls. Brain Topogr. 2018, 31, 346–363. [Google Scholar] [CrossRef]
- Vázquez-Marrufo, M.; González-Rosa, J.J.; Vaquero, E.; Duque, P.; Escera, C.; Borges, M.; Izquierdo, G.; Gómez, C.M. Abnormal ERPs and high frequency bands power in multiplesclerosis. Int. J. Neurosci. 2008, 118, 27–38. [Google Scholar] [CrossRef]
- Sailer, M.; Heinze, H.J.; Tendolkar, I.; Decker, U.; Kreye, O.; Rolbicki, U.V.; Münte, T.F. Influence of cerebral lesion volume and lesion distribution on event-related brain potentials in multiple sclerosis. J. Neurol. 2001, 248, 1049–1055. [Google Scholar] [CrossRef]
- Pfurtscheller, G.; Maresh, H.; Schuy, S. Inter- and intrahemispheric differences in the peak frequency of rhythmic within the alpha band. Electroencephalogr. Clin. Neurophysiol. 1977, 42, 77–83. [Google Scholar] [CrossRef]
- Klimesch, W.; Sauseng, P.; Hanslmayr, S. EEG alpha oscillations: The inhibition-timing hypothesis. Brain Res. Rev. 2007, 53, 63–88. [Google Scholar] [CrossRef]
- Romei, V.; Gross, J.; Thut, G. Sounds reset rhythms of visual cortex and corresponding human visual perception. Curr. Biol. 2012, 22, 807–813. [Google Scholar] [CrossRef]
- Gruber, T.; Klimesch, W.; Sauseng, P.; Doppelmayr, M. Alpha phase synchronization predicts P1 and N1 latency and amplitude size. Cereb. Cortex 2005, 15, 371–377. [Google Scholar] [CrossRef]
- Hanslmayr, S.; Klimesch, W.; Sauseng, P.; Gruber, W.; Doppelmayr, M.; Freunberger, R.; Pecherstorfer, T.; Birbaumer, N. Alpha phase reset contributes to the generation of ERPs. Cereb. Cortex 2007, 17, 1–8. [Google Scholar] [CrossRef]
- Vázquez-Marrufo, M.; Sarrias-Arrabal, E.; Martín-Clemente, R.; Galvao-Carmona, A.; Navarro, G.; Izquierdo, G. Altered phase and nonphase EEG activity expose impaired maintenance of a spatial-object attentional focus in multiple sclerosis patients. Sci. Rep. 2020, 10, 20721. [Google Scholar] [CrossRef]
- Klimesch, W. EEG alpha and theta oscillations reflect cognitive and memory performance: A review and analysis. Brain Res. Rev. 1999, 29, 169–195. [Google Scholar] [CrossRef]
- Pfurtscheller, G.; Neuper, C.; Mohl, W. Event-related desynchronization (ERD) during visual processing. Int. J. Psychophysiol. 1994, 16, 147–153. [Google Scholar] [CrossRef]
- Klimesch, W.; Doppelmayr, M.; Pachinger, T.; Russegger, H. Event-Related desynchronization in the alpha band and the processing of semantic information. Cogn. Brain Res. 1997, 6, 83–94. [Google Scholar] [CrossRef]
- Klimesch, W.; Russegger, H.; Doppelmayr, M.; Pachinger, T. A method for the calculation of induced band power: Implications for the significance of brain oscillations. Electroencephalogr. Clin. Neurophysiol. 1998, 108, 123–130. [Google Scholar] [CrossRef]
- Vázquez-Marrufo, M.; Galvao-Carmona, A.; Benítez-Lugo, M.L.; Ruíz-Peña, J.L.; Borges, M.; Izquierdo, G. Retest reliability of individual alpha ERD topography assessed by human electroencephalography. PLoS ONE 2017, 12, e0187244. [Google Scholar] [CrossRef]
- Fries, P. Rhythms for cognition: Communication through coherence. Neuron 2015, 88, 220–235. [Google Scholar] [CrossRef]
- Mably, A.J.; Colgin, L.L. Gamma oscillations in cognitive disorders. Curr. Opin. Neurobiol. 2018, 52, 182–187. [Google Scholar] [CrossRef]
- Tallon-Baudry, C.; Bertrand, O.; Delpuech, C.; Pernier, J. Stimulus specificity of phase-locked and non-phase-locked 40 Hz visual responses in human. J. Neurosci. 1996, 16, 4240–4249. [Google Scholar] [CrossRef]
- Gruber, T.; Trujillo-Barreto, N.J.; Giabbiconi, C.M.; Valdes-Sosa, P.A.; Muller, M.M. Brain electrical tomography (BET) analysis of induced gamma band responses during a simple object recognition task. Neuroimage 2006, 29, 888–900. [Google Scholar] [CrossRef]
- Tallon-Baudry, C. The roles of gamma-band oscillatory synchrony in human visual cognition. Front. Biosci. 2009, 14, 321–332. [Google Scholar] [CrossRef]
- Schadow, J.; Lenz, D.; Thaerig, S.; Busch, N.A.; Fründ, I.; Rieger, J.W.; Herrmann, C.S. Stimulus intensity affects early sensory processing: Visual contrast modulates evoked gamma-band activity in human EEG. Int. J. Psychophysiol 2007, 66, 28–36. [Google Scholar] [CrossRef]
- Müsch, K.; Hamamé, C.M.; Perrone-Bertolotti, M.; Minotti, L.; Kahane, P.; Engel, A.K.; Lachaux, J.P.; Schneider, T.R. Selective attention modulates high-frequency in the face processing network. Cortex 2014, 60, 34–51. [Google Scholar] [CrossRef]
- Fan, J.; Byrne, J.; Worden, M.S.; Guise, K.G.; McCandliss, B.D.; Fossella, J.; Posner, M.I. The relation of brain oscillations to attentional networks. J. Neurosci. 2007, 27, 6197–6206. [Google Scholar]
- Gruber, T.; Müller, M.M.; Keil, A.; Elbert, T. Selective visual-spatial attention alters induced gamma band responses in the human EEG. Clin. Neurophysiol. 1999, 110, 2074–2085. [Google Scholar] [CrossRef]
- Kiiski, H.; Reilly, R.B.; Lonergan, R.; Kelly, S.; O’Brien, M.; Kinsella, K.; Bramham, J.; Burke, T.; O’Donnchadha, S.; Nolan, H.; et al. Only low frequency event-related EEG activity is compromised in Multiple Sclerosis: Insights from an independent component clustering analysis. PLoS ONE 2012, 7, e45536. [Google Scholar] [CrossRef]
- Keune, P.; Hansen, S.; Weber, E.; Zapf, F.; Habich, J.; Muenssinger, J.; Wolf, S.; Schönenberg, M.; Oschmann, P. Exploring resting-state EEG brain oscillatory activity in relation to cognitive functioning in multiple sclerosis. Clin. Neuropsysiol. 2017, 128, 1746–1754. [Google Scholar] [CrossRef]
- Stickland, R.; Allen, M.; Magazzini, L.; Singh, K.D.; Wise, R.G.; Tomassini, V. Neurovascular coupling during visual stimulation in multiple sclerosis: A MEG-fMRI study. Neuroscience 2019, 403, 54–69. [Google Scholar] [CrossRef]
- Vázquez-Marrufo, M.; González-Rosa, J.J.; Vaquero, E.; Duque, P.; Borges, M.; Gómez, C.; Izquierdo, G. Quantitative electroencephalography reveals different physiological profiles between benign and remitting-relapsing multiple sclerosis patients. BMC Neurol. 2008, 8, 44. [Google Scholar] [CrossRef]
- Salmelin, R.; Hari, R. Spatiotemporal characteristics of sensoriomotor neuromagnetic rhythms related to thumb movement. Neuroscience 1994, 60, 537–550. [Google Scholar] [CrossRef]
- Lehtela, L.; Salmelin, R.; Hari, R. Evidence for reactive magnetic 10-Hz rhythm in the human auditory cortex. Neurosci. Lett. 1997, 222, 111–114. [Google Scholar] [CrossRef]
- Vázquez-Marrufo, M.; Vaquero, E.; Cardoso, M.J.; Gómez, C.M. Temporal spectral evolution of electroencephalogram. Methodology and applications. Rev. Neurol. 2001, 33, 525–530. [Google Scholar]
- Vázquez-Marrufo, M.; Vaquero, E.; Cardoso, M.J.; Gómez, C.M. Temporal evolution of α and β bands during visual spatial attention. Cogn. Brain Res. 2001, 12, 315–320. [Google Scholar] [CrossRef]
- Polman, C.H.; Reingold, S.C.; Banwell, B.; Clanet, M.; Cohen, J.A.; Filippi, M.; Fujihara, K.; Havrdova, E.; Hutchinson, M.; Kappos, L.; et al. Diagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteria. Ann. Neurol. 2011, 69, 292–302. [Google Scholar] [CrossRef] [PubMed]
- American Electroencephalographic Society. Guideline thirteen: Guidelines for standard electrode position nomenclature. J. Clin. Neurophysiol. 1994, 11, 111–113. [Google Scholar] [CrossRef]
- Gratton, G.; Coles, M.G.; Donchin, E. A new method for off-line removal of ocular artifact. Electroencephalogr. Clin. Neurophysiol. 1983, 55, 468–484. [Google Scholar] [CrossRef]
- Cohen, M. Analyzing Neural Time Series Data. Theory and Practice, 1st ed.; MIT Press: Cambridge, MA, USA, 2014. [Google Scholar]
- Vázquez-Marrufo, M.; García-Valdecasas, M.; Caballero-Díaz, R.; Martín-Clemente, R.; Galvao-Carmona, A. Multiple evoked and induced alpha modulations in a visual attention task: Latency, amplitude and topographical profiles. PLoS ONE 2019, 14, e0223055. [Google Scholar] [CrossRef]
- Keil, A.; Müller, M.M. Feature selection in the human brain: Electrophysiological correlates of sensory enhancement and feature integration. Brain Res. 2010, 1313, 172–184. [Google Scholar] [CrossRef]
- Vázquez-Marrufo, M.; Galvao-Carmona, A.; Caballero-Díaz, R.; Borges, M.; Paramo, M.D.; Benítez-Lugo, M.L.; Ruíz-Peña, J.L.; Izquierdo, G. Altered individual behavioral and EEG parameters are related to the EDSS score in relapsing-remitting multiple sclerosis patients. PLoS ONE 2019, 14, e0219594. [Google Scholar] [CrossRef]
- Klaver, R.; De Vries, H.E.; Schenk, G.J.; Geurts, J.J.G. Grey matter damage in multiple sclerosis: A pathology perspective. Prion 2013, 7, 66–75. [Google Scholar] [CrossRef]
- Barry, R.J.; Fogarty, J.S.; De Blasio, F.M.; Karamacoska, D. EEG phase states at stimulus onset in a variable-ISI Go/NoGo task: Effects on ERP components. Biol. Psychol. 2018, 134, 89–102. [Google Scholar] [CrossRef]
- Di Russo, F.; Berchicci, M.; Bianco, V.; Mussini, E.; Perri, R.; Pitzalis, S.; Quinzi, F.; Tranquilli, S.; Spinelli, D. Sustained visuospatial attention enhances lateralized anticipatory ERP activity in sensory areas. Brain Struct. Funct. 2021, 226, 457–470. [Google Scholar] [CrossRef]
- Galvao-Carmona, A.; González-Rosa, J.J.; Hidalgo-Muñoz, A.R.; Páramo, D.; Benítez, M.L.; Izquierdo, G.; Vázquez-Marrufo, M. Disentangling the Attention network test: Behavioral, event related potentials and neural source analyses. Front. Hum. Neurosci. 2014, 8, 813. [Google Scholar] [CrossRef]
- Liu, P.; Yang, W.; Yuan, X.; Bi, C.; Chen, A.; Huang, X. Individual alerting efficiency modulates time perception. Front. Psychol. 2015, 6, 386. [Google Scholar] [CrossRef]
- Urbanek, C.; Weinges-Evers, N.; Bellmann-Strobl, J.; Bock, M.; Dörr, J.; Hahn, E.; Neuhaus, A.H.; Opgen-Rhein, C.; Tam, T.M.T.; Herges, K.; et al. Attention Network Test reveals alerting network dysfunction in multiple sclerosis. Mult. Scler. 2010, 16, 93–99. [Google Scholar] [CrossRef]
- Crivelli, L.; Farez, M.F.; González, C.D.; Fiol, M.; Amengual, A.; Leiguarda, R.; Correale, J. Alerting network dysfunction in early Multiple Sclerosis. J. Int. Neuropsychol. Soc. 2012, 18, 757–763. [Google Scholar] [CrossRef]
- Vázquez-Marrufo, M.; Galvao-Carmona, A.; González-Rosa, J.J.; Hidalgo-Muñoz, A.R.; Borges, M.; Ruiz-Peña, J.L.; Izquierdo, G. Neural correlates of alerting and orienting impairment in multiple sclerosis patients. PLoS ONE 2014, 9, e97226. [Google Scholar] [CrossRef]
- Sarrias-Arrabal, E.; Izquierdo-Ayuso, G.; Vázquez-Marrufo, M. Attentional networks in neurodegenerative diseases: Anatomical and functional evidence from the Attention Network Test. Neurología 2020. [Google Scholar] [CrossRef]
- Mishra, J.; Martínez, A.; Schroeder, C.E.; Hillyard, S.A. Spatial attention boosts short-latency neural responses in human visual cortex. Neuroimage 2012, 59, 1968–1978. [Google Scholar] [CrossRef]
- Tsagkas, C.; Mallar-Chakravarty, M.; Gaetano, L.; Naegelin, Y.; Amann, M.; Parmar, K.; Papadopoulou, A.; Wuerfel, J.; Kappos, L.; Sprenger, T.; et al. Longitudinal patterns of cortical thinning in multiple sclerosis. Hum. Brain Mapp. 2020, 41, 2198–2215. [Google Scholar] [CrossRef]
- Vázquez-Marrufo, M.; Caballero-Díaz, R.; Martín-Clemente, R.; Galvao-Carmona, A.; González-Rosa, J.J. Individual test-retest reliability of evoked and induced alpha activity in human EEG data. PLoS ONE 2020, 15, e0239612. [Google Scholar] [CrossRef]
- Tewarie, P.; Steenwijk, M.D.; Tijms, B.M.; Daams, M.; Balk, L.J.; Stam, C.J.; Uitdehaag, B.M.J.; Polman, C.H.; Geurts, J.J.G.; Barkhof, F.; et al. Disruption of structural and functional networks in long-standing Multiple Sclerosis. Hum. Brain Mapp. 2014, 35, 5946–5961. [Google Scholar] [CrossRef]
- Stefanics, G.; Jakab, A.; Bernáth, L.; Kellényi, L.; Hernándi, I. EEG early evoked gamma-band synchronization reflects objects recognition in visual oddball tasks. Brain Topogr. 2004, 16, 261–264. [Google Scholar] [CrossRef] [PubMed]
- Tallon-Baudry, C.; Bertrand, O.; Delpuech, C.; Pernier, J. Oscillatory gamma band (30 ± 70 Hz) activity induced by a visual search task in human. J. Neurosci. 1997, 17, 722–734. [Google Scholar] [CrossRef] [PubMed]
| MS | HC | |
|---|---|---|
| Sex (m/f) | 8/22 | 11/19 |
| Age (years, mean ± SD) | 41.5 ± 9.04 | 37.43 ± 10.68 |
| Handedless (left/right) | 3/27 | 3/27 |
| Duration of disease (years, mean ± SD) | 8.46 ± 4.3 | - |
| EDSS (mean, range) | 2.95 ± 1.6 | - |
| Reaction Time | Accuracy | ||||
|---|---|---|---|---|---|
| Group | Healthy Controls | Multiple Sclerosis | Subject | Healthy Controls | Multiple Sclerosis |
| Mean | 338 | 394 | Mean | 96 | 78 |
| SD | 40.6 | 38.2 | SD | 5.3 | 26.4 |
| p-value | <0.001 | p-value | <0.001 | ||
| Alpha Band | |||||||
| Latency (mean ± SD) | Amplitude (mean ± SD) | ||||||
| HC | MS | p-value | HC | MS | p-value | ||
| Evoked | 132 ± 32 | 167 ± 54 | 0.034 | Evoked | 2.15 ± 0.83 | 1.30 ± 1.05 | 0.003 |
| Induced | 136 ± 37 | 189 ± 60 | <0.001 | Induced | –1.33 ± 0.60 | –1.57 ± 1.22 | 0.292 |
| Gamma Band | |||||||
| Latency (mean ± SD) | Amplitude (mean ± SD) | ||||||
| HC | MS | p-value | HC | MS | p-value | ||
| Evoked | 117 ± 37 | 126 ± 36 | 1.000 | Evoked | 0.275 ± 0.2 | 0.142 ± 0.16 | 0.058 |
| Induced | 126 ± 41 | 152 ± 46 | 0.098 | Induced | –0.246 ± 0.14 | –0.17 ± 0.15 | 0.067 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sarrias-Arrabal, E.; Eichau, S.; Galvao-Carmona, A.; Domínguez, E.; Izquierdo, G.; Vázquez-Marrufo, M. Deficits in Early Sensory and Cognitive Processing Are Related to Phase and Nonphase EEG Activity in Multiple Sclerosis Patients. Brain Sci. 2021, 11, 629. https://doi.org/10.3390/brainsci11050629
Sarrias-Arrabal E, Eichau S, Galvao-Carmona A, Domínguez E, Izquierdo G, Vázquez-Marrufo M. Deficits in Early Sensory and Cognitive Processing Are Related to Phase and Nonphase EEG Activity in Multiple Sclerosis Patients. Brain Sciences. 2021; 11(5):629. https://doi.org/10.3390/brainsci11050629
Chicago/Turabian StyleSarrias-Arrabal, Esteban, Sara Eichau, Alejandro Galvao-Carmona, Elvira Domínguez, Guillermo Izquierdo, and Manuel Vázquez-Marrufo. 2021. "Deficits in Early Sensory and Cognitive Processing Are Related to Phase and Nonphase EEG Activity in Multiple Sclerosis Patients" Brain Sciences 11, no. 5: 629. https://doi.org/10.3390/brainsci11050629
APA StyleSarrias-Arrabal, E., Eichau, S., Galvao-Carmona, A., Domínguez, E., Izquierdo, G., & Vázquez-Marrufo, M. (2021). Deficits in Early Sensory and Cognitive Processing Are Related to Phase and Nonphase EEG Activity in Multiple Sclerosis Patients. Brain Sciences, 11(5), 629. https://doi.org/10.3390/brainsci11050629







