Motor Chunking in Internally Guided Sequencing
Abstract
1. Introduction
2. Methods
2.1. Participants
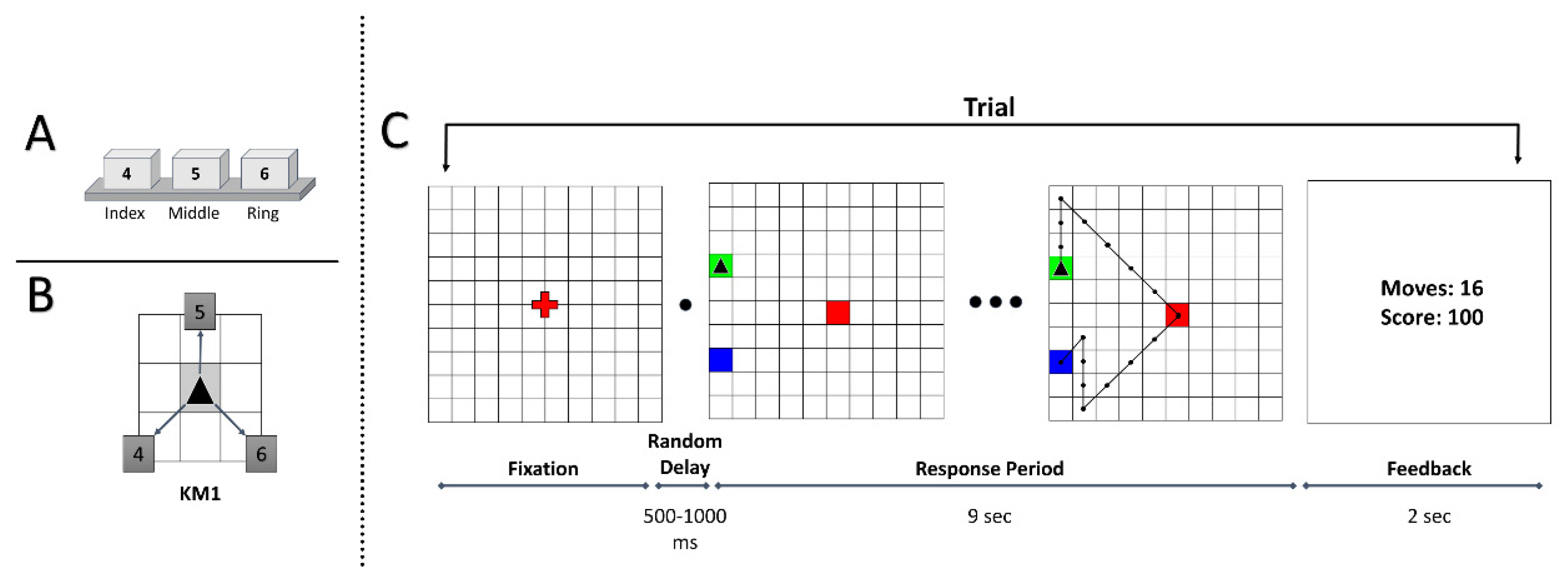
2.2. Apparatus
2.3. Task Paradigm
2.4. Behavioral Measures
3. Results
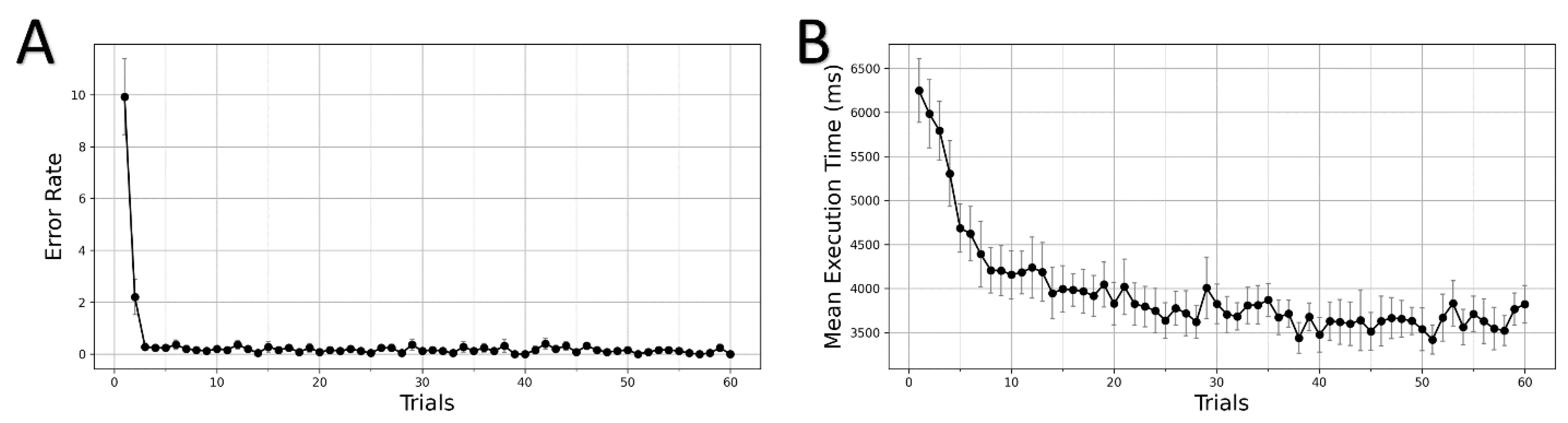
3.1. Learning in GST
3.2. Motor Chunking in GST
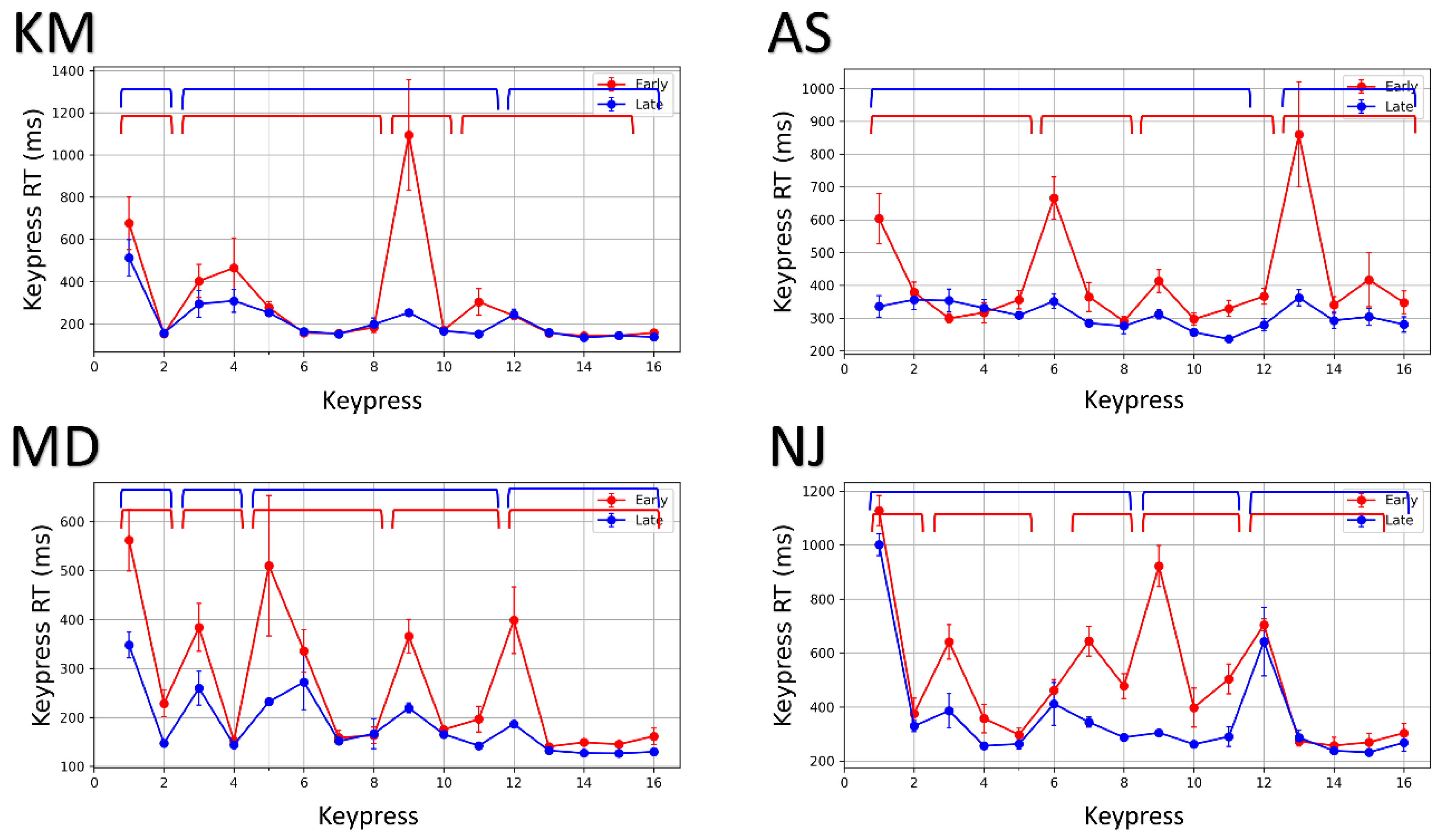
3.2.1. Identifying Chunk Patterns from Keypress RTs
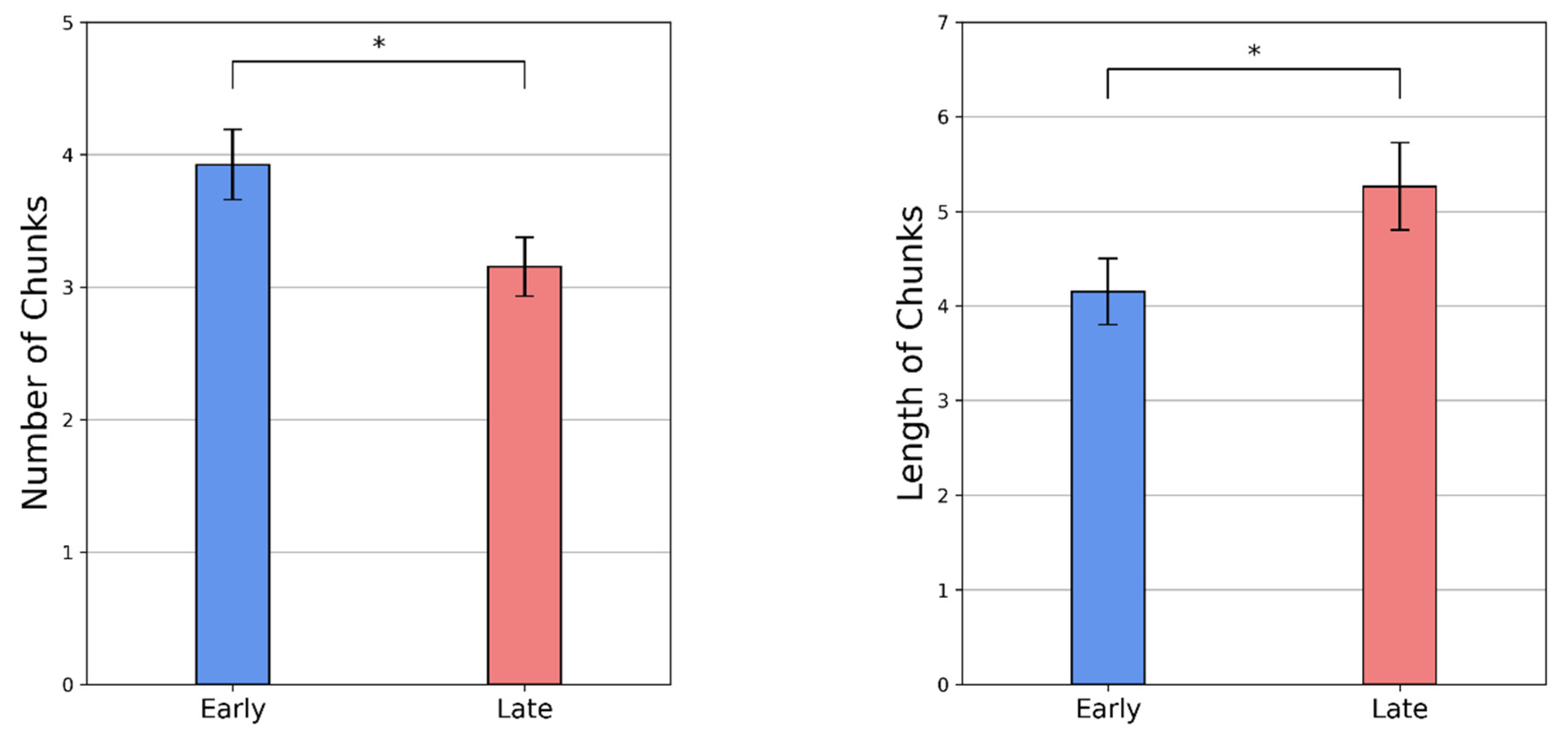
3.2.2. Re-Organization of Action Sequences with Practice
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Adolph, K.E.; Joh, A.S.; Franchak, J.M.; Ishak, S.; Gill, S.V. Flexibility in the development of action. In Oxford Handbook of Human Action; Social cognition and social neuroscience; Morsella, E., Bargh, J.A., Gollwitzer, P.M., Eds.; Oxford University Press: Oxford, UK, 2009; pp. 399–426. [Google Scholar]
- Ben-Sasson, A.; Gill, S.V. Motor and language abilities from early to late toddlerhood: Using formalized assessments to capture continuity and discontinuity in development. Res. Dev. Disabil. 2014, 35, 1425–1432. [Google Scholar] [CrossRef]
- Barnhoorn, J.S.; Van Asseldonk, E.H.F.; Verwey, W.B. Differences in chunking behavior between young and older adults diminish with extended practice. Psychol. Res. 2019, 83, 275–285. [Google Scholar] [CrossRef] [PubMed]
- Sommerville, J.A.; Decety, J. Weaving the fabric of social interaction: Articulating developmental psychology and cognitive neuroscience in the domain of motor cognition. Psychon. Bull. Rev. 2006, 13, 179–200. [Google Scholar] [CrossRef]
- Gill, S.V.; Yang, Z.; Hung, Y.C. Effects of singular and dual task constraints on motor skill variability in childhood. Gait Posture 2017, 53, 121–126. [Google Scholar] [CrossRef][Green Version]
- Krakauer, J.W.; Hadjiosif, A.M.; Xu, J.; Wong, A.L.; Haith, A.M. Motor learning. Compr. Physiol. 2019, 9, 613–663. [Google Scholar] [CrossRef] [PubMed]
- Wolpert, D.M.; Diedrichsen, J.; Flanagan, J.R. Principles of sensorimotor learning. Nat. Rev. Neurosci. 2011, 12, 739–751. [Google Scholar] [CrossRef] [PubMed]
- Willingham, D.B. A Neuropsychological Theory of Motor Skill Learning. Psychol. Rev. 1998, 105, 558–584. [Google Scholar] [CrossRef]
- Newell, K.M. Motor skill acquisition. Annu. Rev. Psychol. 1991, 42, 213–237. [Google Scholar] [CrossRef] [PubMed]
- Clegg, B.A.; DiGirolamo, G.J.; Keele, S.W. Sequence learning. Trends Cogn. Sci. 1998, 2, 275–281. [Google Scholar] [CrossRef]
- Haibach, P.; Reid, G.; Douglas, C. Motor Learning and Development, 2nd ed.; Human Kinetics: Champaign, IL, USA, 2017; ISBN 9781492536598. [Google Scholar]
- Schmidt, R.A.; Lee, T.D.; Winstein, C.J.; Wulf, G.; Zelaznik, H.N. Motor Control and Learning: A Behavioural Emphasis, 6th ed.; Human Kinetics: Champaign, IL, USA, 2018; ISBN 1450412297. [Google Scholar]
- Shadmehr, R.; Mussa-Ivaldi, F.A. Adaptive representation of dynamics during learning of a motor task. J. Neurosci. 1994, 14, 3208–3224. [Google Scholar] [CrossRef]
- Izawa, J.; Rane, T.; Donchin, O.; Shadmehr, R. Motor adaptation as a process of reoptimization. J. Neurosci. 2008, 28, 2883–2891. [Google Scholar] [CrossRef] [PubMed]
- Sakai, K.; Kitaguchi, K.; Hikosaka, O. Chunking during human visuomotor sequence learning. Exp. Brain Res. 2003, 152, 229–242. [Google Scholar] [CrossRef]
- Verwey, W.B.; Eikelboom, T. Evidence for lasting sequence segmentation in the discrete sequence-production task. J. Mot. Behav. 2003, 35, 171–181. [Google Scholar] [CrossRef]
- Bo, J.; Seidler, R.D. Visuospatial working memory capacity predicts the organization of acquired explicit motor sequences. J. Neurophysiol. 2009, 101, 3116–3125. [Google Scholar] [CrossRef]
- Kennerley, S.W.; Sakai, K.; Rushworth, M.F.S. Organization of Action Sequences and the Role of the Pre-SMA. J. Neurophysiol. 2004, 91, 978–993. [Google Scholar] [CrossRef] [PubMed]
- Verwey, W.B.; Abrahamse, E.L.; Jiménez, L. Segmentation of short keying sequences does not spontaneously transfer to other sequences. Hum. Mov. Sci. 2009, 28, 348–361. [Google Scholar] [CrossRef]
- Lashley, K. The problem of serial order in behavior. In Cerebral Mechanisms in Behavior; the Hixon Symposium; Jeffress, L.A., Ed.; Wiley: Hoboken, NJ, USA, 1951; pp. 112–146. [Google Scholar]
- Rosenbaum, D.A.; Kenny, S.B.; Derr, M.A. Hierarchical control of rapid movement sequences. J. Exp. Psychol. Hum. Percept. Perform. 1983, 9, 86–102. [Google Scholar] [CrossRef]
- Zeigler, H.P.; Gallistel, C.R. The Organization of Action: A New Synthesis. Am. J. Psychol. 1981, 94, 190. [Google Scholar] [CrossRef][Green Version]
- Wymbs, N.F.; Bassett, D.S.; Mucha, P.J.; Porter, M.A.; Grafton, S.T. Differential Recruitment of the Sensorimotor Putamen and Frontoparietal Cortex during Motor Chunking in Humans. Neuron 2012, 74, 936–946. [Google Scholar] [CrossRef] [PubMed]
- Ramkumar, P.; Acuna, D.E.; Berniker, M.; Grafton, S.T.; Turner, R.S.; Kording, K.P. Chunking as the result of an efficiency computation trade-off. Nat. Commun. 2016, 7, 12176. [Google Scholar] [CrossRef] [PubMed]
- Logan, G.D. Automatic control: How experts act without thinking. Psychol. Rev. 2018, 125, 453–485. [Google Scholar] [CrossRef]
- Yamaguchi, M.; Logan, G.D. Pushing typists back on the learning curve: Revealing chunking in skilled typewriting. J. Exp. Psychol. Hum. Percept. Perform. 2014, 40, 592–612. [Google Scholar] [CrossRef]
- Thalmann, M.; Souza, A.S.; Oberauer, K. How does chunking help working memory? J. Exp. Psychol. Learn. Mem. Cogn. 2019, 45, 37–55. [Google Scholar] [CrossRef]
- Abrahamse, E.L.; Ruitenberg, M.F.L.; de Kleine, E.; Verwey, W.B. Control of automated behavior: Insights from the discrete sequence production task. Front. Hum. Neurosci. 2013, 7, 82. [Google Scholar] [CrossRef] [PubMed]
- Verwey, W.B.; Dronkert, Y. Practicing a Structured Continuous Key-Pressing Task: Motor Chunking or Rhythm Consolidation? J. Mot. Behav. 1996, 28, 71–79. [Google Scholar] [CrossRef]
- Verwey, W.B. Buffer Loading and Chunking in Sequential Keypressing. J. Exp. Psychol. Hum. Percept. Perform. 1996, 22, 544–562. [Google Scholar] [CrossRef]
- Bapi, R.S.; Doya, K.; Harner, A.M. Evidence for effector independent and dependent representations and their differential time course of acquisition during motor sequence learning. Exp. Brain Res. 2000, 132, 149–162. [Google Scholar] [CrossRef]
- Povel, D.J.; Collard, R. Structural factors in patterned finger tapping. Acta Psychol. 1982, 52, 107–123. [Google Scholar] [CrossRef]
- Restle, F.; Burnside, B.L. Tracking of serial patterns. J. Exp. Psychol. 1972, 95, 299–307. [Google Scholar] [CrossRef]
- Robertson, E.M. The serial reaction time task: Implicit motor skill learning? J. Neurosci. 2007, 27, 10073–10075. [Google Scholar] [CrossRef]
- Curran, T.; Keele, S.W. Attentional and Nonattentional Forms of Sequence Learning. J. Exp. Psychol. Learn. Mem. Cogn. 1993, 19, 189–202. [Google Scholar] [CrossRef]
- Nissen, M.J.; Bullemer, P. Attentional requirements of learning: Evidence from performance measures. Cogn. Psychol. 1987, 19, 1–32. [Google Scholar] [CrossRef]
- Stadler, M.A. Implicit serial learning: Questions inspired by Hebb (1961). Mem. Cognit. 1993, 21, 819–827. [Google Scholar] [CrossRef][Green Version]
- Koch, I.; Hoffmann, J. Patterns, chunks, and hierarchies in serial reaction-time tasks. Psychol. Res. 2000, 63, 22–35. [Google Scholar] [CrossRef] [PubMed]
- Drucker, J.H.; Sathian, K.; Crosson, B.; Krishnamurthy, V.; McGregor, K.M.; Bozzorg, A.; Gopinath, K.; Krishnamurthy, L.C.; Wolf, S.L.; Hart, A.R.; et al. Internally guided lower limb movement recruits compensatory cerebellar activity in people with Parkinson’s disease. Front. Neurol. 2019, 10, 537. [Google Scholar] [CrossRef] [PubMed]
- Jueptner, M.; Weiller, C. A review of differences between basal ganglia and cerebellar control of movements as revealed by functional imaging studies. Brain 1998, 121, 1437–1449. [Google Scholar] [CrossRef]
- Gowen, E.; Miall, R.C. Differentiation between external and internal cuing: An fMRI study comparing tracing with drawing. Neuroimage 2007, 36, 396–410. [Google Scholar] [CrossRef]
- Van Donkelaar, P.; Stein, J.F.; Passingham, R.E.; Miall, R.C. Neuronal activity in the primate motor thalamus during visually triggered and internally generated limb movements. J. Neurophysiol. 1999, 82, 934–945. [Google Scholar] [CrossRef]
- Logan, G.D. Toward an Instance Theory of Automatization. Psychol. Rev. 1988, 95, 492–527. [Google Scholar] [CrossRef]
- Herwig, A.; Waszak, F. Intention and attention in ideomotor learning. Q. J. Exp. Psychol. 2009, 62, 219–227. [Google Scholar] [CrossRef]
- Fermin, A.S.R.; Yoshida, T.; Yoshimoto, J.; Ito, M.; Tanaka, S.C.; Doya, K. Model-based action planning involves cortico-cerebellar and basal ganglia networks. Sci. Rep. 2016, 6, 1–14. [Google Scholar] [CrossRef]
- Fermin, A.; Yoshida, T.; Ito, M.; Yoshimoto, J.; Doya, K. Evidence for model-based action planning in a sequential finger movement task. J. Mot. Behav. 2010, 42, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Abrahamse, E.L.; Jiménez, L.; Verwey, W.B.; Clegg, B.A. Representing serial action and perception. Psychon. Bull. Rev. 2010, 17, 603–623. [Google Scholar] [CrossRef]
- Ziessler, M.; Nattkemper, D. Learning of Event Sequences is Based on Response-Effect Learning: Further Evidence from a Serial Reaction Task. J. Exp. Psychol. Learn. Mem. Cogn. 2001, 27, 595–613. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, J.; Sebald, A.; Stöcker, C. Irrelevant Response Effects Improve Serial Learning in Serial Reaction Time Tasks. J. Exp. Psychol. Learn. Mem. Cogn. 2001, 27, 470–482. [Google Scholar] [CrossRef] [PubMed]
- Hommel, B.; Müsseler, J.; Aschersleben, G.; Prinz, W. The Theory of Event Coding (TEC): A framework for perception and action planning. Behav. Brain Sci. 2001, 24, 849–878. [Google Scholar] [CrossRef] [PubMed]
- Hommel, B. Acquisition and control of voluntary action. Volunt. Action Brains Minds Soc. 2003, 34–48. [Google Scholar]
- Jueptner, M.; Jenkins, I.H.; Brooks, D.J.; Frackowiak, R.S.J.; Passingham, R.E. The sensory guidance of movement: A comparison of the cerebellum and basal ganglia. Exp. Brain Res. 1996, 112, 462–474. [Google Scholar] [CrossRef]
- Bapi, R.S.; Pammi, V.S.C.; Miyapuram, K.P. Ahmed Investigation of sequence processing: A cognitive and computational neuroscience perspective. Curr. Sci. 2005, 89, 1690–1698. [Google Scholar]
- Acuna, D.E.; Wymbs, N.F.; Reynolds, C.A.; Picard, N.; Turner, R.S.; Strick, P.L.; Grafton, S.T.; Kording, K.P. Multifaceted aspects of chunking enable robust algorithms. J. Neurophysiol. 2014, 112, 1849–1856. [Google Scholar] [CrossRef][Green Version]
| SUBJECT | EARLY PHASE | LATE PHASE | ||
|---|---|---|---|---|
| No. of Chunks | Avg. Length of Chunks | No. of Chunks | Avg. Length of Chunks | |
| K.M. | 4 | 3.75 | 3 | 5.33 |
| A.S. | 4 | 4.00 | 2 | 7.50 |
| M.D. | 5 | 3.20 | 4 | 4.00 |
| N.J. | 5 | 2.80 | 3 | 5.33 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bera, K.; Shukla, A.; Bapi, R.S. Motor Chunking in Internally Guided Sequencing. Brain Sci. 2021, 11, 292. https://doi.org/10.3390/brainsci11030292
Bera K, Shukla A, Bapi RS. Motor Chunking in Internally Guided Sequencing. Brain Sciences. 2021; 11(3):292. https://doi.org/10.3390/brainsci11030292
Chicago/Turabian StyleBera, Krishn, Anuj Shukla, and Raju S. Bapi. 2021. "Motor Chunking in Internally Guided Sequencing" Brain Sciences 11, no. 3: 292. https://doi.org/10.3390/brainsci11030292
APA StyleBera, K., Shukla, A., & Bapi, R. S. (2021). Motor Chunking in Internally Guided Sequencing. Brain Sciences, 11(3), 292. https://doi.org/10.3390/brainsci11030292






