The Neuroprotective Effects of Cannabis-Derived Phytocannabinoids and Resveratrol in Parkinson’s Disease: A Systematic Literature Review of Pre-Clinical Studies
Abstract
:1. Introduction
1.1. Neurotoxin and Genetic Models of PD
1.2. Cannabis-Derived Cannabinoids and Resveratrol as Neuroprotective Agents
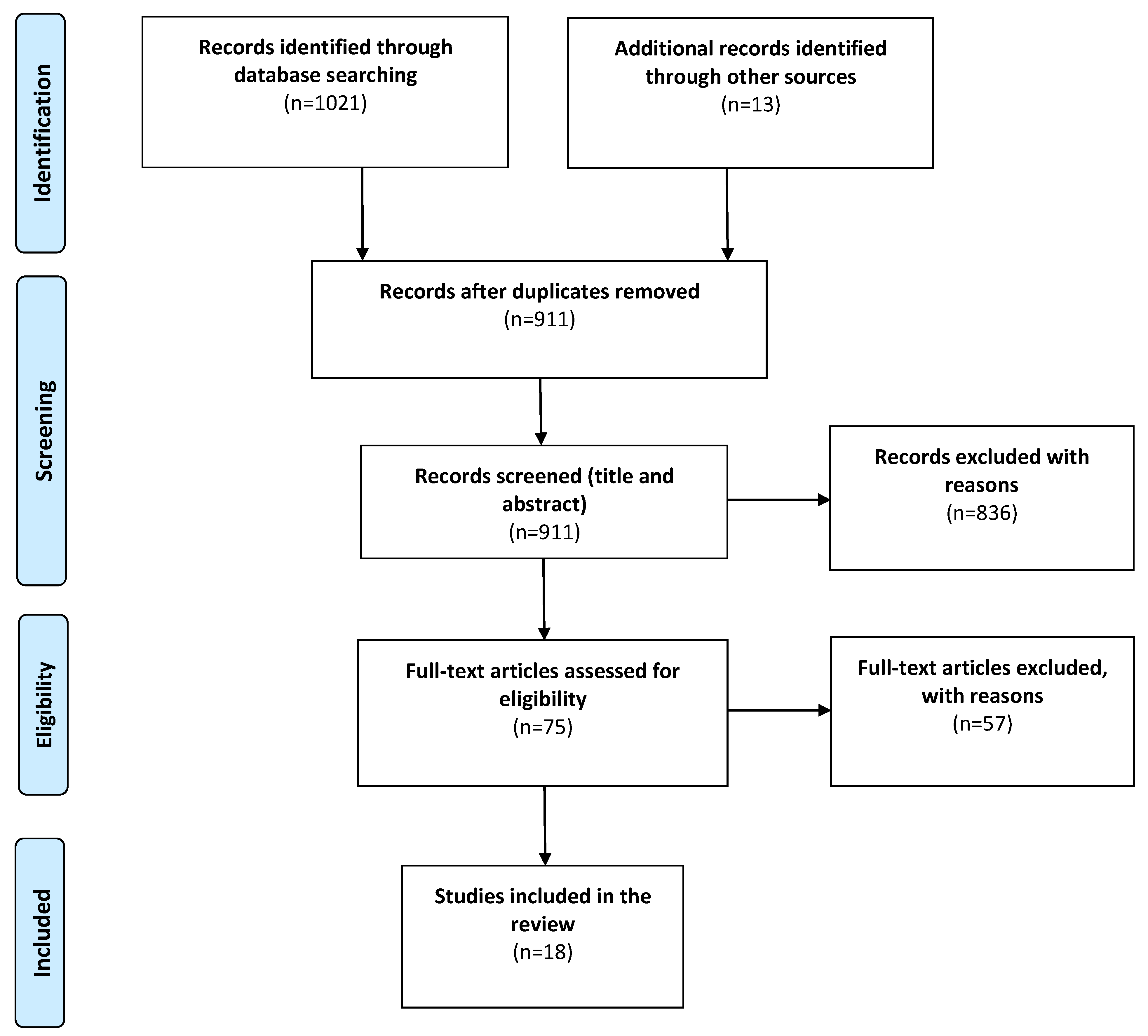
2. Materials and Methods
2.1. Search Strategy
2.2. Eligibility Criteria
2.3. Data Acquisition and Analysis
3. Results
3.1. Study Characteristics
3.2. Dosing
3.3. Behavioral Outcomes
3.3.1. Open Field Testing and Movement
3.3.2. Rotarod and Grasp Strength
3.3.3. Pole and Beam Test
3.3.4. Gait Assessment and Stepping Test
3.3.5. Catalepsy
3.3.6. Apomorphine-Induced Circling Behavior
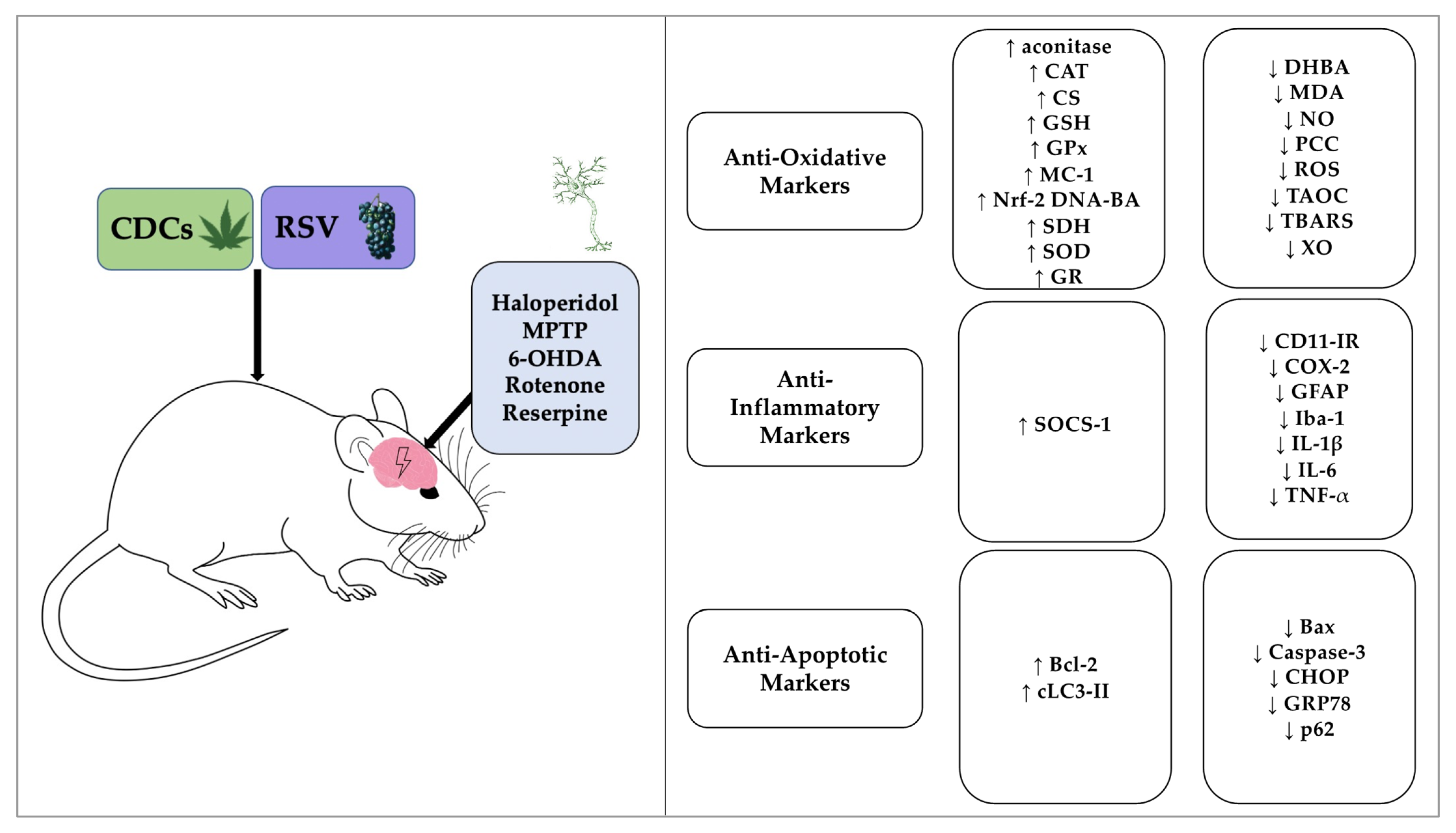
3.4. Biochemical and Immunohistochemical Outcomes
3.5. The Effectiveness of CDCs or RSV to Combat Oxidative Stress
3.6. The Effectiveness of CDCs or RSV to Combat Neuroinflammation
3.7. The Anti-Apoptotic Effects of CDCs or RSV
4. Discussion
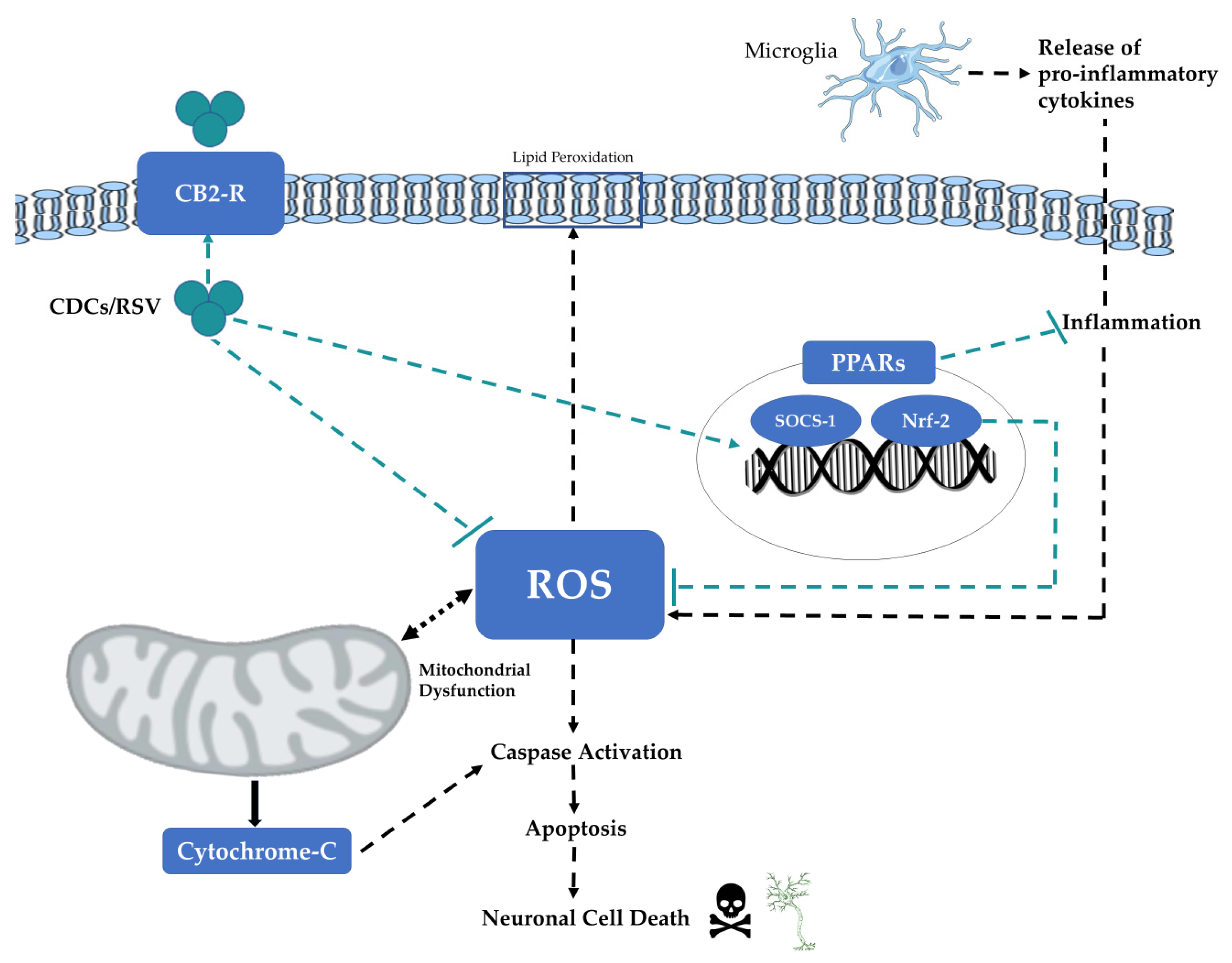
4.1. Neuroprotective Effects and Mechanisms
4.1.1. Behavioral Improvements Indicative of Neuroprotection
4.1.2. Anti-Oxidative Effects of Neuroprotective Agents
4.1.3. Anti-Inflammatory Effects of Neuroprotective Agents
4.1.4. Anti-Apoptotic Effects of RSV
4.2. Pro-Dopaminergic Properties of CDC’s May Involve Cannabinoid Receptors
4.3. Clinical Trials Using Cannabinoids and RSV
Bioavailability of CDCs and RSV
4.4. Study Limitations
5. Conclusions and Future Perspectives
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Xia, R.; Mao, Z.H. Progression of motor symptoms in Parkinson’s disease. Neurosci. Bull. 2012, 28, 39–48. [Google Scholar] [CrossRef] [PubMed]
- McFarland, N.R.; Hess, C.W. Recognizing Atypical Parkinsonisms: “Red Flags” and Therapeutic Approaches. Semin. Neurol. 2017, 37, 215–227. [Google Scholar] [CrossRef] [Green Version]
- Pringsheim, T.; Jette, N.; Frolkis, A.; Steeves, T.D. The prevalence of Parkinson’s disease: A systematic review and meta-analysis. Mov. Disord. 2014, 29, 1583–1590. [Google Scholar] [CrossRef]
- Dorsey, E.R.; Sherer, T.; Okun, M.S.; Bloem, B.R. The Emerging Evidence of the Parkinson Pandemic. J. Parkinsons Dis. 2018, 8, S3–S8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rocca, W.A. The burden of Parkinson’s disease: A worldwide perspective. Lancet Neurol. 2018, 17, 928–929. [Google Scholar] [CrossRef] [Green Version]
- Kouli, A.; Torsney, K.M.; Kuan, W.L. Parkinson’s Disease: Etiology, Neuropathology, and Pathogenesis. In Parkinson’s Disease: Pathogenesis and Clinical Aspects; Stoker, T.B., Greenland, J.C., Eds.; Codon Publications: Brisbane, Australia, 2018. [Google Scholar]
- Bloem, B.R.; Okun, M.S.; Klein, C. Parkinson’s disease. Lancet 2021, 397, 2284–2303. [Google Scholar] [CrossRef]
- Beyer, K.; Domingo-Sàbat, M.; Ariza, A. Molecular pathology of Lewy body diseases. Int. J. Mol. Sci. 2009, 10, 724–745. [Google Scholar] [CrossRef]
- Emamzadeh, F.N. Alpha-synuclein structure, functions, and interactions. J. Res. Med. Sci. 2016, 21, 29. [Google Scholar] [CrossRef]
- Hindle, J.V. Ageing, neurodegeneration and Parkinson’s disease. Age Ageing 2010, 39, 156–161. [Google Scholar] [CrossRef] [Green Version]
- Lill, C.M.; Klein, C. Epidemiology and causes of Parkinson’s disease. Nervenarzt 2017, 88, 345–355. [Google Scholar] [CrossRef]
- Johnson, C.C.; Gorell, J.M.; Rybicki, B.A.; Sanders, K.; Peterson, E.L. Adult nutrient intake as a risk factor for Parkinson’s disease. Int. J. Epidemiol. 1999, 28, 1102–1109. [Google Scholar] [CrossRef] [Green Version]
- Klein, C.; Westenberger, A. Genetics of Parkinson’s disease. Cold Spring Harb. Perspect. Med. 2012, 2, a008888. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alster, P.; Madetko, N.; Koziorowski, D.; Friedman, A. Microglial Activation and Inflammation as a Factor in the Pathogenesis of Progressive Supranuclear Palsy (PSP). Front. Neurosci. 2020, 14, 893. [Google Scholar] [CrossRef] [PubMed]
- Koller, W.C.; Rueda, M.G. Mechanism of action of dopaminergic agents in Parkinson’s disease. Neurology 1998, 50, S11–S14. [Google Scholar] [CrossRef] [PubMed]
- Tambasco, N.; Romoli, M.; Calabresi, P. Levodopa in Parkinson’s Disease: Current Status and Future Developments. Curr. Neuropharmacol. 2018, 16, 1239–1252. [Google Scholar] [CrossRef]
- Jankovic, J. Current approaches to the treatment of Parkinson’s disease. Neuropsychiatr. Dis. Treat. 2008, 4, 743–757. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Riederer, P.; Laux, G. MAO-inhibitors in Parkinson’s Disease. Exp. Neurobiol. 2011, 20, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Alborghetti, M.; Nicoletti, F. Different Generations of Type-B Monoamine Oxidase Inhibitors in Parkinson’s Disease: From Bench to Bedside. Curr. Neuropharmacol. 2019, 17, 861–873. [Google Scholar] [CrossRef]
- Zeng, X.-S.; Geng, W.-S.; Jia, J.-J. Neurotoxin-Induced Animal Models of Parkinson Disease: Pathogenic Mechanism and Assessment. ASN Neuro 2018, 10, 1–15. [Google Scholar] [CrossRef]
- El-Gamal, M.; Salama, M.; Collins-Praino, L.E.; Baetu, I.; Fathalla, A.M.; Soliman, A.M.; Mohamed, W.; Moustafa, A.A. Neurotoxin-Induced Rodent Models of Parkinson’s Disease: Benefits and Drawbacks. Neurotox. Res. 2021, 39, 897–923. [Google Scholar] [CrossRef]
- Jagmag, S.A.; Tripathi, N.; Shukla, S.D.; Maiti, S.; Khurana, S. Evaluation of Models of Parkinson’s Disease. Front. Neurosci. 2016, 9, 503. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Freitas, C.M.; Busanello, A.; Schaffer, L.F.; Peroza, L.R.; Krum, B.N.; Leal, C.Q.; Ceretta, A.P.C.; Da Rocha, J.B.T.; Fachinetto, R. Behavioral and neurochemical effects induced by reserpine in mice. Psychopharmacology 2015, 233, 457–467. [Google Scholar] [CrossRef]
- Dawson, T.M.; Ko, H.S.; Dawson, V. Genetic Animal Models of Parkinson’s Disease. Neuron 2010, 66, 646–661. [Google Scholar] [CrossRef] [Green Version]
- Aryal, S.; Skinner, T.; Bridges, B.; Weber, J.T. The Pathology of Parkinson’s Disease and Potential Benefit of Dietary Polyphenols. Molecules 2020, 25, 4382. [Google Scholar] [CrossRef]
- Nair, M.P.; Figueroa, G.; Casteleiro, G.; Muñoz, K.; Agudelo, M. Alcohol Versus Cannabinoids: A Review of Their Opposite Neuro-Immunomodulatory Effects and Future Therapeutic Potentials. J. Alcohol. Drug Depend. 2015, 3, 184. [Google Scholar] [CrossRef]
- Caruana, M.; Cauchi, R.; Vassallo, N. Putative Role of Red Wine Polyphenols against Brain Pathology in Alzheimer’s and Parkinson’s Disease. Front. Nutr. 2016, 3, 31. [Google Scholar] [CrossRef] [Green Version]
- Kujawska, M.; Jodynis-Liebert, J. Polyphenols in Parkinson’s Disease: A Systematic Review of in Vivo Studies. Nutrients 2018, 10, 642. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mouhamed, Y.; Vishnyakov, A.; Qorri, B.; Sambi, M.; Frank, S.S.; Nowierski, C.; Lamba, A.; Bhatti, U.; Szewczuk, M.R. Therapeutic potential of medicinal marijuana: An educational primer for health care professionals. Drug Healthc. Patient Saf. 2018, 10, 45–66. [Google Scholar] [CrossRef] [Green Version]
- Pertwee, R.G. The diverse CB1 and CB2 receptor pharmacology of three plant cannabinoids: Delta9-tetrahydrocannabinol, cannabidiol and delta9-tetrahydrocannabivarin. Br. J. Pharmacol. 2008, 153, 199–215. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gertsch, J.; Leonti, M.; Raduner, S.; Racz, I.; Chen, J.Z.; Xie, X.Q.; Altmann, K.H.; Karsak, M.; Zimmer, A. Beta-caryophyllene is a dietary cannabinoid. Proc. Natl. Acad. Sci. USA 2008, 105, 9099–9104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Romano, B.; Pagano, E.; Orlando, P.; Capasso, R.; Cascio, M.G.; Pertwee, R.; Di Marzo, V.; Izzo, A.A.; Borrelli, F. Pure Δ9 -tetrahydrocannabivarin and a Cannabis sativa extract with high content in Δ9 -tetrahydrocannabivarin inhibit nitrite production in murine peritoneal macrophages. Pharmacol. Res. 2016, 113, 199–208. [Google Scholar] [CrossRef]
- Yang, M.; Lv, Y.; Tian, X.; Lou, J.; An, R.; Zhang, Q.; Li, M.; Xu, L.; Dong, Z. Neuroprotective Effect of β-Caryophyllene on Cerebral Ischemia-Reperfusion Injury via Regulation of Necroptotic Neuronal Death and Inflammation: In Vivo and in Vitro. Front. Neurosci. 2017, 11, 583. [Google Scholar] [CrossRef]
- O’Sullivan, S.E. An update on PPAR activation by cannabinoids. Br. J. Pharmacol. 2016, 173, 1899–1910. [Google Scholar] [CrossRef] [Green Version]
- Ramírez-Garza, S.L.; Laveriano-Santos, E.P.; Marhuenda-Muñoz, M.; Storniolo, C.E.; Tresserra-Rimbau, A.; Vallverdú-Queralt, A.; Lamuela-Raventós, R.M. Health Effects of Resveratrol: Results from Human Intervention Trials. Nutrients 2018, 10, 1892. [Google Scholar] [CrossRef] [Green Version]
- Salehi, B.; Mishra, A.P.; Nigam, M.; Sener, B.; Kilic, M.; Sharifi-Rad, M.; Fokou, P.V.T.; Martins, N.; Sharifi-Rad, J. Resveratrol: A Double-Edged Sword in Health Benefits. Biomedicines 2018, 6, 91. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, H.; Li, C.; Kwok, S.T.; Zhang, Q.W.; Chan, S.W. A Review of the Pharmacological Effects of the Dried Root of Polygonum cuspidatum (Hu Zhang) and Its Constituents. Evid. Based Complement. Altern. Med. 2013, 2013, 208349. [Google Scholar] [CrossRef] [Green Version]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. J. Clin. Epidemiol. 2009, 62, 1006–1012. [Google Scholar] [CrossRef] [PubMed]
- Hooijmans, C.R.; Rovers, M.M.; de Vries, R.B.; Leenaars, M.; Ritskes-Hoitinga, M.; Langendam, M.W. SYRCLE’S risk of bias tool for animal studies. BMC Med. Res. Methodol. 2014, 14, 43. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lastres-Becker, I.; Molina-Holgado, F.; Ramos, J.A.; Mechoulam, R.; Fernández-Ruiz, J. Cannabinoids provide neuroprotection against 6-hydroxydopamine toxicity in vivo and in vitro: Relevance to Parkinson’s disease. Neurobiol. Dis. 2005, 19, 96–107. [Google Scholar] [CrossRef] [PubMed]
- García, C.; Palomo-Garo, C.; García-Arencibia, M.; Ramos, J.; Pertwee, R.; Fernández-Ruiz, J. Symptom-relieving and neuroprotective effects of the phytocannabinoid Δ9-THCV in animal models of Parkinson’s disease. Br. J. Pharmacol. 2011, 163, 1495–1506. [Google Scholar] [CrossRef] [Green Version]
- Jin, F.; Wu, Q.; Lu, Y.-F.; Gong, Q.-H.; Shi, J.-S. Neuroprotective effect of resveratrol on 6-OHDA-induced Parkinson’s disease in rats. Eur. J. Pharmacol. 2008, 600, 78–82. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.; Ahmad, A.; Ishrat, T.; Hoda, N.; Khuwaja, G.; Raza, S.S.; Khan, A.; Javed, H.; Vaibhav, K.; Islam, F. Resveratrol attenuates 6-hydroxydopamine-induced oxidative damage and dopamine depletion in rat model of Parkinson’s disease. Brain Res. 2010, 1328, 139–151. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Xu, H.; Fu, Q.; Ma, R.; Xiang, J. Protective effect of resveratrol derived from Polygonum cuspidatum and its liposomal form on nigral cells in Parkinsonian rats. J. Neurol. Sci. 2011, 304, 29–34. [Google Scholar] [CrossRef]
- Huang, N.; Zhang, Y.; Chen, M.; Jin, H.; Nie, J.; Luo, Y.; Zhou, S.; Shi, J.; Jin, F. Resveratrol delays 6-hydroxydopamine-induced apoptosis by activating the PI3K/Akt signaling pathway. Exp. Gerontol. 2019, 124, 110653. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Salam, O.M.; El-Shamarka, M.E.S.; Salem, N.A.; El-Din, M.; Gaafar, A. Effects of Cannabis sativa extract on haloperidol-induced catalepsy and oxidative stress in the mice. EXCLI J. 2012, 11, 45–58. [Google Scholar]
- Ojha, S.; Javed, H.; Azimullah, S.; Haque, M.E. β-Caryophyllene, a phytocannabinoid attenuates oxidative stress, neuroinflammation, glial activation, and salvages dopaminergic neurons in a rat model of Parkinson disease. Mol. Cell. Biochem. 2016, 418, 59–70. [Google Scholar] [CrossRef]
- Gaballah, H.H.; Zakaria, S.S.; Elbatsh, M.M.; Tahoon, N.M. Modulatory effects of resveratrol on endoplasmic reticulum stress-associated apoptosis and oxido-inflammatory markers in a rat model of rotenone-induced Parkinson’s disease. Chem. Interact. 2016, 251, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Peres, F.F.; Levin, R.; Suiama, M.A.; Diana, M.C.; Gouvêa, D.A.; de Almeida, V.; Santos, C.M.; Lungato, L.; Zuardi, A.W.; Hallak, J.E.C.; et al. Cannabidiol Prevents Motor and Cognitive Impairments Induced by Reserpine in Rats. Front. Pharmacol. 2016, 7, 343. [Google Scholar] [CrossRef] [Green Version]
- Palle, S.; Neerati, P. Improved neuroprotective effect of resveratrol nanoparticles as evinced by abrogation of rotenone-induced behavioral deficits and oxidative and mitochondrial dysfunctions in rat model of Parkinson’s disease. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2018, 391, 445–453. [Google Scholar] [CrossRef]
- Zhang, L.F.; Yu, X.L.; Ji, M.; Liu, S.Y.; Wu, X.L.; Wang, Y.J.; Liu, R.T. Resveratrol alleviates motor and cognitive deficits and neuropathology in the A53T α-synuclein mouse model of Parkinson’s disease. Food Funct. 2018, 9, 6414–6426. [Google Scholar] [CrossRef]
- Guo, Y.J.; Dong, S.Y.; Cui, X.X.; Feng, Y.; Liu, T.; Yin, M.; Kuo, S.H.; Tan, E.K.; Zhao, W.J.; Wu, Y.C. Resveratrol alleviates MPTP-induced motor impairments and pathological changes by autophagic degradation of α-synuclein via SIRT1-deacetylated LC3. Mol. Nutr. Food Res. 2016, 60, 2161–2175. [Google Scholar] [CrossRef]
- Lofrumento, N.E.; Nicolardi, G.; Cianciulli, A.; De Nuccio, F.; La Pesa, V.; Carofiglio, V.; Dragone, T.; Calvello, R.; Panaro, M.A. Neuroprotective effects of resveratrol in an MPTP mouse model of Parkinson’s-like disease: Possible role of SOCS-1 in reducing pro-inflammatory responses. Innate Immun. 2013, 20, 249–260. [Google Scholar] [CrossRef] [PubMed]
- Anandhan, A.; Tamilselvam, K.; Vijayraja, D.; Ashokkumar, N.; Rajasankar, S.; Manivasagam, T. Resveratrol attenuates oxidative stress and improves behaviour in 1 -methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) challenged mice. Ann. Neurosci. 2010, 17, 113–119. [Google Scholar] [CrossRef] [Green Version]
- Xia, D.; Sui, R.; Zhang, Z. Administration of resveratrol improved Parkinson’s disease-like phenotype by suppressing apoptosis of neurons via modulating the MALAT1/miR-129/SNCA signaling pathway. J. Cell. Biochem. 2019, 120, 4942–4951. [Google Scholar] [CrossRef]
- Lu, K.T.; Ko, M.C.; Chen, B.Y.; Huang, J.C.; Hsieh, C.W.; Lee, M.C.; Chiou, R.Y.; Wung, B.S.; Peng, C.H.; Yang, Y.L. Neuroprotective effects of resveratrol on MPTP-induced neuron loss mediated by free radical scavenging. J. Agric. Food Chem. 2008, 56, 6910–6913. [Google Scholar] [CrossRef]
- Viveros-Paredes, J.M.; González-Castañeda, R.E.; Gertsch, J.; Chaparro-Huerta, V.; López-Roa, R.I.; Vázquez-Valls, E.; Beas-Zarate, C.; Camins-Espuny, A.; Flores-Soto, M.E. Neuroprotective Effects of β-Caryophyllene against Dopaminergic Neuron Injury in a Murine Model of Parkinson’s Disease Induced by MPTP. Pharmaceuticals 2017, 10, 60. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Amende, I.; Kale, A.; McCue, S.; Glazier, S.; Morgan, J.P.; Hampton, T.G. Gait dynamics in mouse models of Parkinson’s disease and Huntington’s disease. J. Neuroeng. Rehabil. 2005, 2, 20. [Google Scholar] [CrossRef] [Green Version]
- Ungerstedt, U.; Arbuthnott, G.W. Quantitative recording of rotational behavior in rats after 6-hydroxy-dopamine lesions of the nigrostriatal dopamine system. Brain Res. 1970, 24, 485–493. [Google Scholar] [CrossRef]
- Su, C.F.; Jiang, L.; Zhang, X.W.; Iyaswamy, A.; Li, M. Resveratrol in Rodent Models of Parkinson’s Disease: A Systematic Review of Experimental Studies. Front. Pharmacol. 2021, 12, 644219. [Google Scholar] [CrossRef] [PubMed]
- Mallet, N.; Delgado, L.; Chazalon, M.; Miguelez, C.; Baufreton, J. Cellular and Synaptic Dysfunctions in Parkinson’s Disease: Stepping out of the Striatum. Cells 2019, 8, 1005. [Google Scholar] [CrossRef] [Green Version]
- Rubin, J.E.; McIntyre, C.C.; Turner, R.; Wichmann, T. Basal ganglia activity patterns in parkinsonism and computational modeling of their downstream effects. Eur. J. Neurosci. 2012, 36, 2213–2228. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.H.; Cha, M.; Lee, B.H. Neuroprotective Effect of Antioxidants in the Brain. Int. J. Mol. Sci. 2020, 21, 7152. [Google Scholar] [CrossRef] [PubMed]
- Smeyne, M.; Smeyne, R.J. Glutathione metabolism and Parkinson’s disease. Free Radic. Biol. Med. 2013, 62, 13–25. [Google Scholar] [CrossRef] [Green Version]
- Gelders, G.; Baekelandt, V.; Van Der Perren, A. Linking Neuroinflammation and Neurodegeneration in Parkinson’s Disease. J. Immunol. Res. 2018, 2018, 4784268. [Google Scholar] [CrossRef] [Green Version]
- Licastro, F.; Porcellini, E. Activation of Endogenous Retrovirus, Brain Infections and Environmental Insults in Neurodegeneration and Alzheimer’s Disease. Int. J. Mol. Sci. 2021, 22, 7263. [Google Scholar] [CrossRef]
- Rigano, D.; Sirignano, C.; Taglialatela-Scafati, O. The potential of natural products for targeting PPAR α. Acta Pharm. Sin. B 2017, 7, 427–438. [Google Scholar] [CrossRef]
- Hussain, T.; Tan, B.; Yin, Y.; Blachier, F.; Tossou, M.C.B.; Rahu, N. Oxidative Stress and Inflammation: What Polyphenols Can Do for Us? Oxid. Med. Cell. Longev. 2016, 2016, 7432797. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Sá Coutinho, D.; Pacheco, M.T.; Frozza, R.L.; Bernardi, A. Anti-Inflammatory Effects of Resveratrol: Mechanistic Insights. Int. J. Mol. Sci. 2018, 19, 1812. [Google Scholar] [CrossRef] [Green Version]
- Vanhaesebroeck, B.; Alessi, D.R. The PI3K-PDK1 connection: More than just a road to PKB. Biochem. J. 2000, 346, 561–576. [Google Scholar] [CrossRef]
- Malagelada, C.; Jin, Z.H.; Greene, L.A. RTP801 is induced in Parkinson’s disease and mediates neuron death by inhibiting Akt phosphorylation/activation. J. Neurosci. 2008, 28, 14363–14371. [Google Scholar] [CrossRef] [Green Version]
- Wu, Y.; Li, X.; Zhu, J.X.; Xie, W.; Le, W.; Fan, Z.; Jankovic, J.; Pan, T. Resveratrol-activated AMPK/SIRT1/autophagy in cellular models of Parkinson’s disease. Neurosignals 2011, 19, 163–174. [Google Scholar] [CrossRef]
- Lee, I.H. Mechanisms and disease implications of sirtuin-mediated autophagic regulation. Exp. Mol. Med. 2019, 51, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Costa, L.; Amaral, C.; Teixeira, N.; Correia-Da-Silva, G.; Fonseca, B.M. Cannabinoid-induced autophagy: Protective or death role? Prostaglandins Other Lipid Mediat. 2016, 122, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Colla, E. Linking the Endoplasmic Reticulum to Parkinson’s Disease and Alpha-Synucleinopathy. Front. Neurosci. 2019, 13, 560. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hoozemans, J.J.; van Haastert, E.S.; Nijholt, D.A.; Rozemuller, A.J.; Scheper, W. Activation of the unfolded protein response is an early event in Alzheimer’s and Parkinson’s disease. Neurodegener. Dis. 2012, 10, 212–215. [Google Scholar] [CrossRef]
- Gorbatyuk, M.S.; Shabashvili, A.; Chen, W.; Meyers, C.; Sullivan, L.F.; Salganik, M.; Lin, J.H.; Lewin, A.S.; Muzyczka, N.; Gorbatyuk, O.S. Glucose regulated protein 78 diminishes α-synuclein neurotoxicity in a rat model of Parkinson disease. Mol. Ther. 2012, 20, 1327–1337. [Google Scholar] [CrossRef]
- Lu, H.C.; Mackie, K. An Introduction to the Endogenous Cannabinoid System. Biol. Psychiatry 2016, 79, 516–525. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Buhmann, C.; Mainka, T.; Ebersbach, G.; Gandor, F. Evidence for the use of cannabinoids in Parkinson’s disease. J. Neural Transm. 2019, 126, 913–924. [Google Scholar] [CrossRef] [PubMed]
- Turcotte, C.; Blanchet, M.R.; Laviolette, M.; Flamand, N. The CB2 receptor and its role as a regulator of inflammation. Cell. Mol. Life Sci. 2016, 73, 4449–4470. [Google Scholar] [CrossRef] [Green Version]
- Silvestro, S.; Schepici, G.; Bramanti, P.; Mazzon, E. Molecular Targets of Cannabidiol in Experimental Models of Neurological Disease. Molecules 2020, 25, 5186. [Google Scholar] [CrossRef]
- Leehey, M.A.; Liu, Y.; Hart, F.; Epstein, C.; Cook, M.; Sillau, S.; Klawitter, J.; Newman, H.; Sempio, C.; Forman, L.; et al. Safety and Tolerability of Cannabidiol in Parkinson Disease: An Open Label, Dose-Escalation Study. Cannabis Cannabinoid Res. 2020, 5, 326–336. [Google Scholar] [CrossRef] [PubMed]
- Chagas, M.H.; Zuardi, A.W.; Tumas, V.; Pena-Pereira, M.A.; Sobreira, E.T.; Bergamaschi, M.M.; dos Santos, A.C.; Teixeira, A.L.; Hallak, J.E.; Crippa, J.A. Effects of cannabidiol in the treatment of patients with Parkinson’s disease: An exploratory double-blind trial. J. Psychopharmacol. 2014, 28, 1088–1098. [Google Scholar] [CrossRef]
- Gambini, J.; Inglés, M.; Olaso, G.; Lopez-Grueso, R.; Bonet-Costa, V.; Gimeno-Mallench, L.; Mas-Bargues, C.; Abdelaziz, K.M.; Gomez-Cabrera, M.C.; Vina, J.; et al. Properties of Resveratrol: In Vitro and in Vivo Studies about Metabolism, Bioavailability, and Biological Effects in Animal Models and Humans. Oxid. Med. Cell. Longev. 2015, 2015, 837042. [Google Scholar] [CrossRef] [Green Version]
- Chimento, A.; De Amicis, F.; Sirianni, R.; Sinicropi, M.S.; Puoci, F.; Casaburi, I.; Saturnino, C.; Pezzi, V. Progress to Improve Oral Bioavailability and Beneficial Effects of Resveratrol. Int. J. Mol. Sci. 2019, 20, 1381. [Google Scholar] [CrossRef] [Green Version]
- Walle, T. Bioavailability of resveratrol. Ann. N. Y. Acad. Sci. 2011, 1215, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Almeida, L.; Vaz-da-Silva, M.; Falcão, A.; Soares, E.; Costa, R.; Loureiro, A.I.; Fernandes-Lopes, C.; Rocha, J.F.; Nunes, T.; Wright, L.; et al. Pharmacokinetic and safety profile of trans-resveratrol in a rising multiple-dose study in healthy volunteers. Mol. Nutr. Food Res. 2009, 53 (Suppl. 1), S7–S15. [Google Scholar] [CrossRef]
- Boocock, D.J.; Faust, G.E.; Patel, K.R.; Schinas, A.M.; Brown, V.A.; Ducharme, M.P.; Booth, T.D.; Crowell, J.A.; Perloff, M.; Gescher, A.J.; et al. Phase I dose escalation pharmacokinetic study in healthy volunteers of resveratrol, a potential cancer chemopreventive agent. Cancer Epidemiol. Biomark. Prev. 2007, 16, 1246–1252. [Google Scholar] [CrossRef] [Green Version]
- Millar, S.A.; Stone, N.L.; Yates, A.S.; O’Sullivan, S.E. A Systematic Review on the Pharmacokinetics of Cannabidiol in Humans. Front. Pharmacol. 2018, 9, 1365. [Google Scholar] [CrossRef] [PubMed]
- Ohlsson, A.; Lindgren, J.E.; Andersson, S.; Agurell, S.; Gillespie, H.; Hollister, L.E. Single-dose kinetics of deuterium-labelled cannabidiol in man after smoking and intravenous administration. Biomed. Environ. Mass Spectrom. 1986, 13, 77–83. [Google Scholar] [CrossRef]
- Perucca, E.; Bialer, M. Critical Aspects Affecting Cannabidiol Oral Bioavailability and Metabolic Elimination, and Related Clinical Implications. CNS Drugs 2020, 34, 795–800. [Google Scholar] [CrossRef]
- Ohlsson, A.; Lindgren, J.E.; Wahlen, A.; Agurell, S.; Hollister, L.E.; Gillespie, H.K. Single dose kinetics of deuterium labelled delta-1-tetrahydrocannabinol in heavy and light cannabis users. Biomed. Environ. Mass Spectrom. 1982, 9, 6–10. [Google Scholar] [CrossRef] [PubMed]
- Karschner, E.L.; Darwin, W.D.; Goodwin, R.S.; Wright, S.; Huestis, M.A. Plasma Cannabinoid Pharmacokinetics following Controlled Oral Δ9-Tetrahydrocannabinol and Oromucosal Cannabis Extract Administration. Clin. Chem. 2011, 57, 66–75. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Collet, J.-P.; Shapiro, S.; Ware, M.A. Adverse effects of medical cannabinoids: A systematic review. Can. Med. Assoc. J. 2008, 178, 1669–1678. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| trans-Resveratrol (RSV) |  |
| Cannabidiol (CBD) | 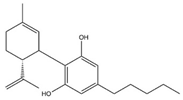 |
| ∆9-Tetrahydrocannabinol (THC) | 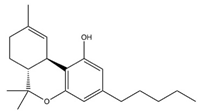 |
| Tetrahydrocannabivarin (THCV) | 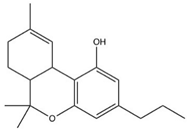 |
| β-caryophyllene (BCP) | 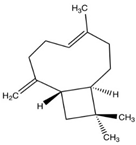 |
| Author and Year | Species and Strain, Study Population | Neurotoxin Model, Dose, Route of Administration, Length of Dosing | Main Intervention Groups: Compound, Dose, Frequency | Outcomes in Main Interventional Groups |
|---|---|---|---|---|
| Ojha et al. (2016) [47] | Wistar rats. Four Groups (n = 8) | Rotenone induced (2.5 mg/kg bw per day, i.p.); 28 days | BCP (50 mg/kg bw per day dissolved in olive oil i.p.); 28 days | ↓, DAN loss ↓, OS markers ↓, Inflammatory markers |
| Viveros-Paredes et al. (2017) [57] | C57BL/6J mice. Six groups (n = 6) | MPTP induced (30 mg/kg bw per day, i.p.); 5 days | BCP (10 mg/kg bw per day i.p.); 5 days. BCP (10 mg/kg bw per day o.g.); 5 days | ↓, pole test time (s), ↑, stride length in gait test, ↓, time in beam test (s) ↑, TH–positive neurones ↓, inflammatory markers |
| Peres et al. (2016) [49] | Wistar rats. Four Groups (n = 10) | Reserpine induced (1 mg/kg bw per day, s.c.); 2 days | CBD (0.5 mg/kg and 5 mg/kg bw per day dissolved in saline and 1% Tween-80, i.p.); 3 days | ↓, catalepsy |
| Lastres-Becker et al. (2005) [40] | SD rats. Four groups (n = 7+) | 6-OHDA induced (8 μg m.f.b.i.); 14 days | CBD (3 mg/kg bw per day dissolved Tween-80:saline (1:16) i.p.); 14 days THC (3 mg/kg bw per day, dissolved in Tween-80:saline (1:16) i.p.); 14 days. | ↑, T H mRNA |
| Abdel-Salam et al. (2012) [46] | SA mice. Seven groups (n = 6) | Haloperidol induced (1 mg/kg bw per day, i.p.); 18 days | Cannabis extract THC (10 mg/kg and 20 mg/kg bw per day, dissolved in 96% ethanol s.c.); 18 days | ↓, catalepsy ↓, OS markers |
| Garcia et al. (2011) [41] | SD rats, CB2 −/− mice, wild-type littermates. Twelve groups (n = 5–6) | 6-OHDA induced rats (200 μg per day, i.c.v.); 14 days. LPS induced mice (5 μg, i.c.v.); 14 days | THCV (2 mg/kg bw dissolved in Tween-80:saline (1:16) i.p., single dose 14 days post-lesion.) THCV (2 mg/kg bw per day dissolved in Tween-80:saline (1:16) i.p.); 14 days | ↑, activity in CAA test ↓, DAN loss, ↑, TH–positive neurons |
| Zhang et al. (2018) [51] | A53T α-synuclein mice, wild-type littermates. Five groups (n = 8) | A53T α-synuclein mouse model. | RSV (10 mg/kg and 50 mg/kg bw per day, o.g.; 35 days) | ↓, activity in open field test, ↑, cognitive performance, ↓, hindlimb clasping, ↓, time pole test (s) ↓, α-synuclein ↓, OS markers ↓, inflammatory markers |
| Lu et al. (2008) [56] | Balb/C mice. Four groups (n = 15) | MPTP induced (30 mg/kg bw per day, i.p.); 7 days | RSV (20 mg/kg bw per day, dissolved in 20% ethanol i.v.); 7 days | ↑, retention time on rotarod, ↓, grasp strength ↓, OS markers |
| Anandhan et al. (2010) [54] | Albino C57BL/6 mice. Four groups (n = 12) | MPTP induced (30 mg/kg bw per day i.p.); 4 days | RSV (50 mg/kg bw per day, p.o.); 7 days | ↑, activity in open field test, ↑, retention time on the rotarod, ↓, time in beam test ↑, DA ↓, OS markers |
| Lofrumento et al. (2014) [53] | C57BL/6N mice. Four groups (n = 6) | MPTP induced (20 mg/kg bw, 4 doses, i.p.); 8 h | RSV (50 mg/kg bw per day, o.g.); 21 days | ↓, apomorphine-induced circling behavior ↓, DAN loss ↓, inflammatory markers |
| Guo et al. (2016) [52] | C57BL/6 mice. Four groups (n = 12) | MPTP induced (30 mg/kg bw per day, i.p.); 5 days | RSV (100 mg/kg bw per day i.g.; 33 days) | ↑, activity in open field test, ↑, catalepsy, ↑, rotarod performance ↑, TH–positive neurons ↓, pro-apoptotic markers |
| Xia et al. (2019) [55] | Mice. Three groups (n = 8) | MPTP induced (20 mg/kg bw, 4 doses, i.p.); 8 h | RSV (50 mg/kg bw per day, i.g.); 21 days | ↑, TH–positive neurons, ↓, α-synuclein |
| Jin et al. (2008) [42] | SD rats. Six groups (n = 10) | 6-OHDA induced. (10 μg s.i.) | RSV (10, 20 and 40 mg/kg in distilled water bw per day, o.g.); 70 days | ↓, apomorphine-induced circling behavior ↓, DAN injury ↓, inflammatory markers |
| Khan et al. (2010) [43] | Wistar rats. Four groups (n = 8) | 6-OHDA induced (10 μg s.i.) | RSV (20 mg/kg bw per day, dissolved in 20% ethanol, i.p.); 15 days | ↓, apomorphine-induced circling behavior, ↑, performance in stepping test, ↑, retention time on the rotarod ↑, DA ↑, DOPAC ↓, OS markers |
| Wang et al. (2011) [44] | Wistar rats. Five groups (n = 10) | 6-OHDA induced (15 μg i.c.v.) | RSV (1 mL, g.g.); 14 days RSV liposome (20 mg/kg bw per day, i.g.); 14 days | ↓, apomorphine-induced circling behavior ↑, TH-positive neurons ↓, OS markers ↓, pro-apoptotic markers |
| Huang et al. (2019) [45] | SD rats. Five groups (n = 10) | 6-OHDA induced (8 μg i.p.) | RSV (15 mg/kg and 30 mg/kg bw per day, p.o.); 36 days | ↑, activity in open field test, ↑, catalepsy, ↑, rotarod performance ↑, TH–positive neurons ↓, pro-apoptotic markers |
| Gaballah et al. (2016) [48] | Wistar Albino rats. Four groups (n = 10–15) | Rotenone induced (1.5 mg/kg bw, 11 doses s.c.); 21 days | RSV (20 mg/kg bw per day, o.g.); 21 days | ↓, catalepsy, ↑, retention time on rotarod ↑, DA ↓, OS markers ↓, inflammatory markers ↓, pro-apoptotic markers |
| Palle and Neerati (2018) [50] | Wistar Albino rats. Four groups (n = 12) | Rotenone induced (2 mg/kg bw per day s.c.); 35 days | RSV (40 mg/kg bw per day, p.o.) RSV nanoparticles (40 mg/kg bw per day, p.o.); 35 days | ↑, rearing count ↑, rotarod performance ↓, OS markers |
| Author and Year | Intervention | Behavioral Outcomes in PD Model Group (vs. Controls) | Level of Significance | Behavioral Changes in Main Intervention Group (vs. PD Model Group) | Level of Significance |
|---|---|---|---|---|---|
| Viveros-Paredes et al. (2017) [57] | BCP | ↑, pole test time (s) ↓, stride length in gait test ↑, time in beam test (s) | (p < 0.001) (p < 0.01) (p < 0.01) | ↓, pole test time (s) ↑, stride length in gait test ↓, time in beam test (s) | (p < 0.01) (p < 0.05) (p < 0.01) |
| Peres et al. (2016) [49] | CBD | ↓, activity in open field test ↑, catalepsy ↑, vacuous chewing | (p < 0.05) | ↑, memory deficit but not locomotor activity in open field test ↓, catalepsy ↓, vacuous chewing | (p < 0.05) |
| Abdel-Salam et al. (2012) [46] | THC | ↑, catalepsy | (p < 0.05) | ↓, catalepsy | (p < 0.05) |
| Garcia et al. (2011) [41] | THCV | ↓, activity in CAA test | (p < 0.05) | ↑, activity in CAA test | (p < 0.05) |
| Jin et al. (2008) [42] | RSV | ↑, apomorphine-induced circling behavior | - | ↓, apomorphine-induced circling behavior | (p < 0.01) |
| Lu et al. (2008) [56] | RSV | ↓, retention time on rotarod ↑, grasp strength | (p < 0.05) | ↑, retention time on rotarod ↓, grasp strength | (p < 0.05) |
| Anandhan et al. (2010) [54] | RSV | ↓, activity in open field test ↑, time in beam test ↓, retention time on rotarod | (p < 0.05) | ↑, activity in open field test ↓, time in beam test ↑, retention time on the rotarod | (p < 0.05) |
| Khan et al. (2010) [43] | RSV | ↑, apomorphine-induced circling behavior ↓, retention time on rotarod ↓, performance in stepping test | (p < 0.01) (p < 0.001) (p < 0.01) | ↓, apomorphine-induced circling behavior ↑, retention time on the rotarod ↑, performance in stepping test | (p < 0.05) (p < 0.05) (p < 0.05) |
| Wang et al. (2011) [44] | RSV | ↑, apomorphine-induced circling behavior | (p < 0.01) | ↓, apomorphine-induced circling behavior | (p < 0.01) |
| Gaballah et al. (2016) [48] | RSV | ↑, catalepsy ↓, retention time on rotarod | (p < 0.05) | ↓, catalepsy ↑, retention time on rotarod | (p < 0.05) |
| Guo et al. (2016) [52] | RSV | ↓, activity in open field test ↓, stride length in gait test ↑, time pole test (s) | (p < 0.001) (p < 0.001) (p < 0.01) | ↑, activity in open field test ↑, stride length in gait test ↓, time pole test (s) | (p < 0.001) (p < 0.001) (p < 0.01) |
| Palle and Neerati (2018) [50] | RSV | ↓, rearing count ↓, retention time on rotarod | (p < 0.05) | ↑, rearing count ↑, retention time on rotarod | (p < 0.05) |
| Zhang et al. (2018) [51] | RSV | ↑, activity in open field test ↑, time pole test (s) ↑, hindlimb clasping ↓, cognitive performance | (p < 0.05) (p < 0.001) (p < 0.001) (p < 0.05) | ↓, activity in open field test ↓, time pole test (s) ↓, hindlimb clasping ↑, cognitive performance | (p < 0.05) (p < 0.01) (p < 0.05) (p < 0.05) |
| Huang et al. (2019) [45] | RSV | ↓, retention time on rotarod ↓, activity in open field test ↑, catalepsy | (p < 0.05) (p < 0.01) (p < 0.05) | ↑, rotarod performance ↑, activity in open field test ↓, catalepsy | (p < 0.05) (p < 0.01) (p < 0.05) |
| Author and Year | Intervention | Changes in Dopamine and α-Synuclein in PD Model Groups (vs. Control) | Level of Significance | Changes in Dopaminergic System and α-Synuclein in Main Interventional Groups (vs. PD Model) | Level of Significance |
|---|---|---|---|---|---|
| Ojha et al. (2016) [47] | BCP | ↑, DAN loss | (p < 0.05) | ↓, DAN loss | (p < 0.05) |
| Viveros-Paredes et al. (2017) [57] | BCP | ↓, TH–positive neurons | (p < 0.001) | ↑, TH–positive neurons | (p < 0.05) |
| Lastres-Becker et al. (2005) [40] | THC or CBD | ↓, TH activity ↓, TH mRNA | (p < 0.05) (p < 0.01) | ↑, TH activity ↑, TH mRNA | (p < 0.05) (p < 0.05) |
| Garcia et al. (2011) [41] | THCV | ↑, DAN loss ↓, TH–positive neurons | (p < 0.05) (p < 0.005) | ↓, DAN loss ↑, TH–positive neurons | (p > 0.05) (p < 0.05) |
| Jin et al. (2008) [42] | RSV | ↑, DAN injury | - | ↓, DAN injury | - |
| Anandhan et al. (2010) [54] | RSV | ↓, DA ↓, DOPAC ↓, HVA | (p < 0.05) | ↑, DA ↑, DOPAC ↑, HVA | (p < 0.05) |
| Wang et al. (2011) [44] | RSV | ↓, TH-positive cells | (p < 0.05) | ↑, TH-positive cells | (p < 0.01) |
| Lofrumento et al. (2014) [53] | RSV | ↓, TH immunoreactivity | (p < 0.01) | ↑, TH immunoreactivity | (p < 0.01) |
| Guo et al. (2016) [52] | RSV | ↓, TH–positive neurons | (p < 0.001) | ↑, TH–positive neurons | (p < 0.01) |
| Gaballah et al. (2016) [48] | RSV | ↓, DA | (p < 0.05) | ↑, DA | (p < 0.05) |
| Zhang et al. (2018) [51] | RSV | ↑, α-synuclein ↑, A-11 positive oligomers ↑, W20 positive oligomers ↓, TH–positive neurons | (p < 0.001) (p < 0.001) (p < 0.001) (p < 0.01) | ↓, α-synuclein ↓, A-11 positive oligomers ↓, W20 positive oligomers ↑, TH–positive neurons | (p < 0.001) (p < 0.05) (p < 0.05) - |
| Huang et al. (2019) [45] | RSV | ↓, TH–positive neurons | (p < 0.05) | ↑, TH–positive neurons | (p < 0.05) |
| Xia et al. (2019) [55] | RSV | ↓, TH-positive neurons ↑, α-synuclein | (p < 0.05) | ↑, TH–positive neurons ↓, α-synuclein | (p < 0.05) |
| Author and Year | Intervention | Changes in Oxidative Stress in PD Model Group (vs. Control) | Level of Significance | Changes in Oxidative Stress Markers in Main Intervention Group (vs. PD Model Group) | Level of Significance |
|---|---|---|---|---|---|
| Ojha et al. (2016) [47] | BCP | ↓, GSH ↑, MDA ↓, SOD and CAT | (p < 0.01) | ↑, GSH ↓, MDA ↑, SOD and CAT | (p < 0.05) |
| Abdel-Salam et al. (2012) [46] | THC | ↑, MDA ↑, NO ↓, GSH | (p < 0.05) | ↓, MDA ↓, NO ↑, GSH | (p < 0.05) |
| Lu et al. (2008) [56] | RSV | ↑, DHBA | (p < 0.05) | ↓, DHBA | (p < 0.05) |
| Anandhan et al. (2010) [54] | RSV | ↓, GSH ↑, SOD and CAT ↑, TBARS ↓, GPx | (p < 0.05) | ↑, GSH ↓, SOD and CAT ↓, TBARS ↑, GPx | (p < 0.05) |
| Khan et al. (2010) [43] | RSV | ↑, TBARS ↑, PCC ↓, GSH ↓, GPx ↓, GR ↓, SOD ↓, CAT | (p < 0.001) (p < 0.01) (p < 0.01) (p < 0.01) (p < 0.05) (p < 0.05) (p < 0.01) | ↓, TBARS ↓, PCC ↑, GSH ↑, GPx ↑, GR ↑, SOD ↑, CAT | (p < 0.01) (p < 0.05) (p < 0.05) (p < 0.05) (p < 0.05) (p < 0.01) (p < 0.001) |
| Wang et al. (2011) [44] | RSV | ↓, TAOC ↑, ROS | (p < 0.01) | ↑, TAOC ↓, ROS | (p < 0.01) |
| Gaballah et al. (2016) [48] | RSV | ↑, XO ↑, PCC ↓, GPx ↑, Nrf-2 DNA-binding activity | (p < 0.05) | ↓, XO ↓, PCC ↑, GPx ↑, Nrf-2 DNA-binding activity | (p < 0.0001) |
| Zhang et al. (2018) [51] | RSV | ↑, ROS ↑, MDA ↓, SOD and CAT | (p < 0.01) | ↓, ROS ↓, MDA ↑, SOD and CAT | (p < 0.01) |
| Palle and Neerati (2018) [50] | RSV | ↓, SDH ↓, CS ↓, aconitase ↓, MC-I activity ↓, GSH ↓, CAT ↑, MDA | (p < 0.05) | ↑, SDH ↑, CS ↑, aconitase ↑, MC-I activity ↑, GSH ↑, CAT ↓, MDA | (p < 0.05) |
| Author and Year | Intervention | Inflammatory Changes in PD Model Group (vs. Control) | Level of Significance | Inflammatory Changes in Main Intervention Group (vs. PD Model Group) | Level of Significance |
|---|---|---|---|---|---|
| Ojha et al. (2016) [47] | BCP | ↑, GFAP ↑, Iba-1 ↑, IL-1β ↑, IL-6 ↑, TNF-α | (p < 0.05) | ↓, GFAP ↓, Iba-1 ↓, IL-1β ↓, IL-6 ↓, TNF-α | (p < 0.05) |
| Viveros-Paredes et al. (2017) [57] | BCP | ↑, GFAP-IR cells ↑, Iba-1-IR cells ↑, IL-1β ↑, IL-6 ↑, TNF-α | (p < 0.01) (p < 0.05) (p < 0.01) (p < 0.05) (p < 0.05) | ↓, GFAP-IR cells ↓, IBA-1-IR cells ↓, IL-1β ↓, IL-6 | (p < 0.01) (p < 0.01) (p < 0.01) (p < 0.05) |
| Garcia et al. (2011) [41] | THCV | ↑, microglial activation | (p < 0.005) | ↓, microglial activation | (p < 0.05) |
| Jin et al. (2008) [42] | RSV | ↑, COX-2 ↑, TNF-α mRNA | (p < 0.01) | ↓, COX-2 ↓, TNF-α mRNA | (p < 0.01) |
| Lofrumento et al. (2014) [53] | RSV | ↑, GFAP mRNA expression ↑, CD11-immunoreactivity ↑, IL-1β mRNA ↑, TNF-α mRNA ↑, IL-6 mRNA ↑, IL-1β R1 ↑, TNF-α R1 ↑, IL-6Rα ↓, SOCS-1 | (p < 0.01) (p < 0.01) (p < 0.01) (p < 0.01) (p < 0.05) (p < 0.05) (p < 0.05) (p < 0.01) (p < 0.01) | ↓, GFAP mRNA expression ↓, CD11-immunoreactivity ↓, IL-1β mRNA ↓, TNF-α mRNA ↓, IL-6 mRNA ↓, IL-1β RI ↓, TNF-α RI ↓, IL-6Rα ↑, SOCS-1 | (p < 0.01) (p < 0.05) (p < 0.01) (p < 0.05) (p < 0.01) (p < 0.05) (p < 0.05) (p < 0.05) (p < 0.01) |
| Gaballah et al. (2016) [48] | RSV | ↑, striatal IL-1β levels | (p < 0.05) | ↓, striatal IL-1β levels | (p < 0.05) |
| Zhang et al. (2018) [51] | RSV | ↑, GFAP ↑, Iba-1 ↑, IL-1β ↑, IL-6 ↑, TNF-α | (p < 0.001) (p < 0.001) (p < 0.01) (p < 0.001) (p < 0.05) | ↓, GFAP ↓, Iba-1 ↓, IL-1β ↓, IL-6 ↓, TNF-α | (p < 0.05) (p < 0.01) (p < 0.05) (p < 0.05) (p < 0.01) |
| Author and Year | Intervention | Apoptotic Changes in PD Model Group (vs. Control) | Level of Significance | Apoptotic Changes in Main Intervention Group (vs. PD Model Group) | Level of Significance |
|---|---|---|---|---|---|
| Wang et al. (2011) [44] | RSV | ↓, apoptotic nigral cells | (p < 0.01) | ↓, apoptotic nigral cells | (p < 0.01) |
| Gaballah et al. (2016) [48] | RSV | ↑, CHOP and GRP78 ↑, striatal caspase-3 activity | (p < 0.05) | ↓, CHOP and GRP78 ↓, striatal caspase-3 activity | (p < 0.05) |
| Guo et al. (2016) [52] | RSV | ↑, cleaved caspase 3 ↓, deacetylated LC3-II ↑, p62 | (p < 0.001) (p < 0.001) (p < 0.01) | ↓, cleaved caspase 3 ↑, deacetylated LC3-II ↓, p62 | (p < 0.01) (p < 0.001) (p < 0.001) |
| Huang et al. (2019) [45] | RSV | ↑, Bax ↑, activated caspase 3 ↓, Bcl-2 ↓, Pro-caspase-3 expression | (p < 0.01) (p < 0.01) (p < 0.05) (p < 0.01) | ↓, Bax ↓, activated caspase 3 ↑, Bcl-2 ↑, Pro-caspase-3 expression | (p < 0.01) (p < 0.01) (p < 0.05) (p < 0.01) |
| Author & Year | Study Type | Population | Intervention | Study Outcome |
|---|---|---|---|---|
| Leehey et al. (2020) [82] NCT02818777 | Safety and tolerability of CBD in PD, open label dose-escalation study | 13 participants, mean age 68 (SD = 6) with PD | CBD: (Epidiolex; 100 mg/mL); 5 to 20–25 mg/kg/day; 10–15 days | Improved MDS-UPDRS scores. Improved sleep and emotional dyscontrol scores. Mild adverse effects including diarrhea, somnolence, fatigue, weight gain, dizziness, abdominal pain, weight loss, headache, nausea, anorexia, increased appetite. Increase liver enzyme profile, cholestatic in nature. |
| Chagas et al. (2014) [83] NCT unavailable | CBD as a treatment for patients with PD, exploratory double-blind trial | 119 individuals; PD | CBD; 75 mg/day or CBD 300 mg/day; 36 days | No significant changes in UPDRS scores, plasma BDNF levels or H1-MRS measures. Improved PDQ scores and overall emotional well-being. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Prakash, S.; Carter, W.G. The Neuroprotective Effects of Cannabis-Derived Phytocannabinoids and Resveratrol in Parkinson’s Disease: A Systematic Literature Review of Pre-Clinical Studies. Brain Sci. 2021, 11, 1573. https://doi.org/10.3390/brainsci11121573
Prakash S, Carter WG. The Neuroprotective Effects of Cannabis-Derived Phytocannabinoids and Resveratrol in Parkinson’s Disease: A Systematic Literature Review of Pre-Clinical Studies. Brain Sciences. 2021; 11(12):1573. https://doi.org/10.3390/brainsci11121573
Chicago/Turabian StylePrakash, Samay, and Wayne G. Carter. 2021. "The Neuroprotective Effects of Cannabis-Derived Phytocannabinoids and Resveratrol in Parkinson’s Disease: A Systematic Literature Review of Pre-Clinical Studies" Brain Sciences 11, no. 12: 1573. https://doi.org/10.3390/brainsci11121573
APA StylePrakash, S., & Carter, W. G. (2021). The Neuroprotective Effects of Cannabis-Derived Phytocannabinoids and Resveratrol in Parkinson’s Disease: A Systematic Literature Review of Pre-Clinical Studies. Brain Sciences, 11(12), 1573. https://doi.org/10.3390/brainsci11121573







