Aberrant Amplitude of Low-Frequency Fluctuation and Degree Centrality within the Default Mode Network in Patients with Vascular Mild Cognitive Impairment
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Image Data Acquisition
2.3. MRI Data Processing
2.4. Statistical Analysis
3. Results
3.1. Demographic and Neuropsychological Tests Results
3.2. VBM Results
3.3. ALFF Results
3.4. DC Results
3.5. Linear Regression Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Geriatric Neurology Group; Chinese Society of Geriatrics; Clinical Practice Guideline for Cognitive Impairment of Cerebral Small Vessel Disease Writing Group. Clinical practice guideline for cognitive impairment of cerebral small vessel disease of China (2019). Chin. J. Geriatr. 2019, 38, 345–354. [Google Scholar] [CrossRef]
- La, C.; Nair, V.A.; Mossahebi, P.; Young, B.M.; Chacon, M.; Jensen, M.; Birn, R.M.; Meyerand, M.E.; Prabhakaran, V. Implication of the slow-5 oscillations in the disruption of the default-mode network in healthy aging and stroke. Brain Connect. 2016, 6, 482–495. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zang, Y.F.; He, Y.; Zhu, C.Z.; Cao, Q.J.; Sui, M.Q.; Liang, M.; Tian, L.X.; Jiang, T.Z.; Wang, Y.F. Altered baseline brain activity in children with ADHD revealed by resting-state functional MRI. Brain Dev. 2007, 29, 83–91. [Google Scholar] [CrossRef]
- Yi, L.; Wang, J.; Jia, L.; Zhao, Z.; Lu, J.; Li, K.; Jia, J.; He, Y.; Jiang, C.; Han, Y. Structural and functional changes in subcortical vascular mild cognitive impairment: A combined voxel-based morphometry and resting-state fMRI study. PLoS ONE 2012, 7, e44758. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, Y.; Shi, Y.; Zhang, J.; Kong, D.; Guo, L.; Bo, G.; Feng, Y. Neurologic factors in patients with vascular mild cognitive impairment based on fMRI. World Neurosurg. 2021, 149, 461–469. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Chen, H.; Liang, H.; Wang, W.; Liang, Y.; Liang, Y.; Zhang, Y. Low-frequency fluctuations amplitude signals exhibit abnormalities of intrinsic brain activities and reflect cognitive impairment in leukoaraiosis patients. Med. Sci. Monit. 2019, 25, 5219–5228. [Google Scholar] [CrossRef]
- Ter Telgte, A.; van Leijsen, E.M.C.; Wiegertjes, K.; Klijn, C.J.M.; Tuladhar, A.M.; de Leeuw, F.E. Cerebral small vessel disease: From a focal to a global perspective. Nat. Rev. Neurol. 2018, 14, 387–398. [Google Scholar] [CrossRef]
- Grau-Olivares, M.; Arboix, A.; Junque, C.; Arenaza-Urquijo, E.M.; Rovira, M.; Bartres-Faz, D. Progressive gray matter atrophy in lacunar patients with vascular mild cognitive impairment. Cerebrovasc. Dis. 2010, 30, 157–166. [Google Scholar] [CrossRef]
- Li, M.; Meng, Y.; Wang, M.; Yang, S.; Wu, H.; Zhao, B.; Wang, G. Cerebral gray matter volume reduction in subcortical vascular mild cognitive impairment patients and subcortical vascular dementia patients, and its relation with cognitive deficits. Brain Behav. 2017, 7, e00745. [Google Scholar] [CrossRef]
- Lei, Y.; Su, J.; Guo, Q.; Yang, H.; Gu, Y.; Mao, Y. Regional gray matter atrophy in vascular mild cognitive impairment. J. Stroke Cerebrovasc. Dis. 2016, 25, 95–101. [Google Scholar] [CrossRef]
- Liu, C.; Li, C.; Yin, X.; Yang, J.; Zhou, D.; Gui, L.; Wang, J. Abnormal intrinsic brain activity patterns in patients with subcortical ischemic vascular dementia. PLoS ONE 2014, 9, e87880. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.L.; Fan, F.M.; Lu, J.; Li, H.J.; Jia, L.F.; Han, Y.; Li, K.C. Changes of gray matter volume and amplitude of low-frequency oscillations in amnestic MCI: An integrative multi-modal MRI study. Acta Radiol. 2015, 56, 614–621. [Google Scholar] [CrossRef] [PubMed]
- Lyu, H.; Wang, J.; Xu, J.; Zheng, H.; Yang, X.; Lin, S.; Chen, J.; Zhou, L.; Hu, Y.; Guo, Z. Structural and functional disruptions in subcortical vascular mild cognitive impairment with and without depressive symptoms. Front. Aging Neurosci. 2019, 11, 241. [Google Scholar] [CrossRef] [PubMed]
- Zuo, X.N.; Ehmke, R.; Mennes, M.; Imperati, D.; Castellanos, F.X.; Sporns, O.; Milham, M.P. Network centrality in the human functional connectome. Cereb. Cortex 2012, 22, 1862–1875. [Google Scholar] [CrossRef] [PubMed]
- Sachdev, P.; Kalaria, R.; O’Brien, J.; Skoog, I.; Alladi, S.; Black, S.E.; Blacker, D.; Blazer, D.; Chen, C.; Chui, H.; et al. Diagnostic criteria for vascular cognitive disorders: A VASCOG statement. Alzheimer Dis. Assoc. Disord. 2014, 28, 206. [Google Scholar] [CrossRef] [Green Version]
- Yan, C.G.; Wang, X.D.; Zuo, X.N.; Zang, Y.F. DPABI: Data processing & analysis for (resting-state) brain imaging. Neuroinformatics 2016, 14, 339–351. [Google Scholar] [CrossRef]
- Yeo, B.T.; Krienen, F.M.; Sepulcre, J.; Sabuncu, M.R.; Lashkari, D.; Hollinshead, M.; Roffman, J.L.; Smoller, J.W.; Zollei, L.; Polimeni, J.R.; et al. The organization of the human cerebral cortex estimated by intrinsic functional connectivity. J. Neurophysiol. 2011, 106, 1125–1165. [Google Scholar] [CrossRef]
- Yin, Y.; He, X.; Xu, M.; Hou, Z.; Song, X.; Sui, Y.; Liu, Z.; Jiang, W.; Yue, Y.; Zhang, Y.; et al. Structural and functional connectivity of default mode network underlying the cognitive impairment in late-onset depression. Sci. Rep. 2016, 6, 37617. [Google Scholar] [CrossRef] [Green Version]
- Greicius, M.D.; Krasnow, B.; Reiss, A.L.; Menon, V. Functional connectivity in the resting brain: A network analysis of the default mode hypothesis. Proc. Natl. Acad. Sci. USA 2003, 100, 253–258. [Google Scholar] [CrossRef] [Green Version]
- Cavanna, A.E.; Trimble, M.R. The precuneus: A review of its functional anatomy and behavioural correlates. Brain 2006, 129, 564–583. [Google Scholar] [CrossRef] [Green Version]
- Seghier, M.L. The angular gyrus: Multiple functions and multiple subdivisions. Neuroscientist 2013, 19, 43–61. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Talati, A.; Hirsch, J. Functional specialization within the medial frontal gyrus for perceptual go/no-go decisions based on “what,” “when,” and “where” related information: An fMRI study. J. Cogn. Neurosci. 2005, 17, 981–993. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Yang, J.; Yin, X.; Liu, C.; Zhang, L.; Zhang, X.; Gui, L.; Wang, J. Abnormal intrinsic brain activity patterns in leukoaraiosis with and without cognitive impairment. Behav. Brain Res. 2015, 292, 409–413. [Google Scholar] [CrossRef] [PubMed]
- Ni, L.; Liu, R.; Yin, Z.; Zhao, H.; Nedelska, Z.; Hort, J.; Zhou, F.; Wu, W.; Zhang, X.; Li, M.; et al. Aberrant spontaneous brain activity in patients with mild cognitive impairment and concomitant lacunar infarction: A resting-state functional MRI study. J. Alzheimers Dis. 2016, 50, 1243–1254. [Google Scholar] [CrossRef] [PubMed]
- Zuo, M.; Xu, Y.; Zhang, X.; Li, M.; Jia, X.; Niu, J.; Li, D.; Han, Y.; Yang, Y. Aberrant brain regional homogeneity and functional connectivity of entorhinal cortex in vascular mild cognitive impairment: A resting-state functional MRI study. Front. Neurol. 2018, 9, 1177. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Teper, E.; O’Brien, J.T. Vascular factors and depression. Int. J. Geriatr. Psychiatry 2008, 23, 993–1000. [Google Scholar] [CrossRef]
- Steffens, D.C.; Otey, E.; Alexopoulos, G.S.; Butters, M.A.; Cuthbert, B.; Ganguli, M.; Geda, Y.E.; Hendrie, H.C.; Krishnan, R.R.; Kumar, A.; et al. Perspectives on depression, mild cognitive impairment, and cognitive decline. Arch. Gen. Psychiatry 2006, 63, 130–138. [Google Scholar] [CrossRef]
- Burrage, E.; Marshall, K.L.; Santanam, N.; Chantler, P.D. Cerebrovascular dysfunction with stress and depression. Brain Circ. 2018, 4, 43–53. [Google Scholar] [CrossRef]
- Xie, X.; Shi, Y.; Zhang, J. Structural network connectivity impairment and depressive symptoms in cerebral small vessel disease. J. Affect. Disord. 2017, 220, 8–14. [Google Scholar] [CrossRef]
- Duering, M.; Righart, R.; Csanadi, E.; Jouvent, E.; Herve, D.; Chabriat, H.; Dichgans, M. Incident subcortical infarcts inducefocal thinning in connected cortical regions. Neurology 2012, 79, 2025–2028. [Google Scholar] [CrossRef]
- Carmichael, O.; Schwarz, C.; Drucker, D.; Fletcher, E.; Harvey, D.; Beckett, L.; Jack, C.R., Jr.; Weiner, M.; DeCarli, C.; Alzheimer’s Disease Neuroimaging Initiative. Longitudinal changes in white matter disease and cognition in the first year of the Alzheimer disease neuroimaging initiative. Arch. Neurol. 2010, 67, 1370–1378. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wen, Z.; Zhou, F.Q.; Huang, X.; Dan, H.D.; Xie, B.J.; Shen, Y. Altered functional connectivity of primary visual cortex in late blindness. Neuropsychiatr. Dis. Treat. 2018, 14, 3317–3327. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, Y.H.; Young, I.M.; Conner, A.K.; Glenn, C.A.; Chakraborty, A.R.; Nix, C.E.; Bai, M.Y.; Dhanaraj, V.; Fonseka, R.D.; Hormovas, J.; et al. Anatomy and White Matter Connections of the Inferior Temporal Gyrus. World Neurosurg. 2020, 143, e656–e666. [Google Scholar] [CrossRef]
- Borkowski, K.; Klodowski, K.; Figiel, H.; Krzyzak, A.T. A theoretical validation of the B-matrix spatial distribution approach to diffusion tensor imaging. Magn. Reson. Imaging 2017, 36, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Borkowski, K.; Krzyzak, A.T. The generalized Stejskal-Tanner equation for non-uniform magnetic field gradients. J. Magn. Reson. 2018, 296, 23–28. [Google Scholar] [CrossRef]
- Borkowski, K.; Krzyzak, A.T. Assessment of the systematic errors caused by diffusion gradient inhomogeneity in DTI-computer simulations. NMR Biomed. 2019, 32, e4130. [Google Scholar] [CrossRef] [PubMed]


| HC (N = 31) | VaMCI (N = 31) | p-Value | |
|---|---|---|---|
| Age (years) | 59.35 (8.15) | 62.87 (7.07) | 0.075 |
| Gender (male/female) | 14/17 | 20/11 | 0.446 |
| Education level (years) | 11(9, 12) | 9 (8, 11) | 0.017 |
| MoCA score | 28 (26, 30) | 23(20, 24) | <0.001 |
| HAMD score | 0 (0, 1) | 3 (0, 8) | 0.006 |
| HAMA score | 0 (0, 3) | 3 (0, 6) | 0.009 |
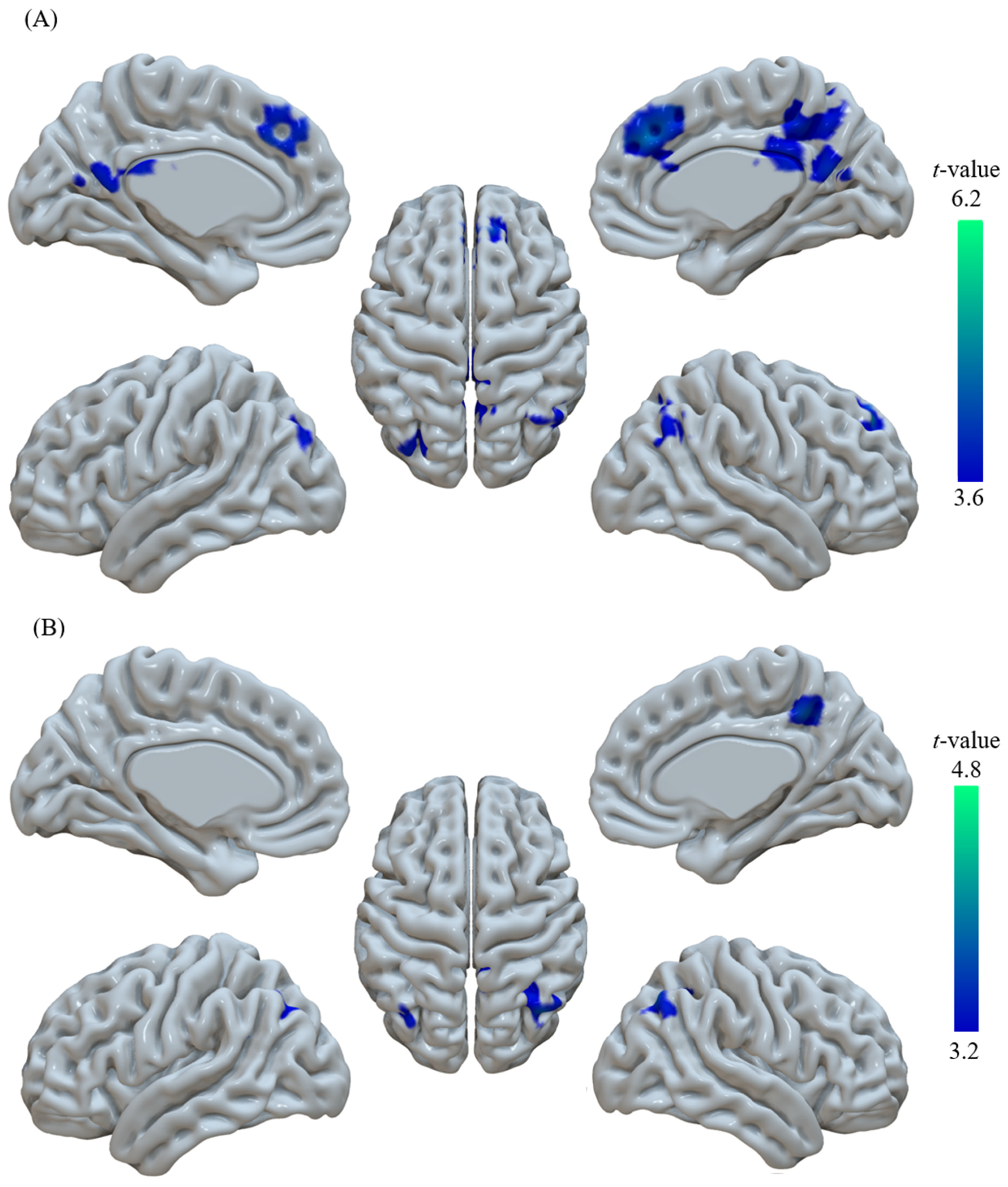
| Region | Cluster Size | MNI Coordinate | t-Value |
|---|---|---|---|
| (voxel) | (x, y, z) | ||
| R.PCu | 258 | (15, −45, 45) | 5.05 |
| R.AG | 125 | (54, −63, 45) | 4.91 |
| R.medFG | 58 | (6, 39, 36) | 6.08 |
| L.PCu | 44 | (−33, −81, 39) | 4.35 |
| Region | Cluster Size | MNI Coordinate | t-Value |
|---|---|---|---|
| (voxel) | (x, y, z) | ||
| L.AG | 258 | (−36, −60, 33) | 4.15 |
| R.PCu | 224 | (6, −45, 42) | 4.24 |
| R.AG | 220 | (42, −72, 48) | 4.69 |
| Unstandardized Coefficient | Standardized Coefficient | t | p | |||
|---|---|---|---|---|---|---|
| B | SE(B) | β | ||||
| MoCA | Constant | 23.528 | 0.488 | 48.193 | <0.001 *** | |
| Age | −0.160 | −1.103 | 0.280 | |||
| Gender | 0.006 | 0.042 | 0.967 | |||
| Edu. level | 0.043 | 0.292 | 0.773 | |||
| GMV | 0.092 | 0.627 | 0.536 | |||
| ALFF | 2.122 | 0.489 | 0.627 | 4.338 | <0.001 *** | |
| DC | −0.077 | −0.476 | 0.638 | |||
| Registered R square: 0.373; ANOVA: <0.001 | ||||||
| HAMA | Constant | 1.727 | 0.836 | 2.065 | 0.048 * | |
| Age | 0.224 | 1.249 | 0.222 | |||
| Gender | 2.773 | 1.041 | 0.443 | 2.663 | 0.013 * | |
| Edu. level | 0.065 | 0.368 | 0.716 | |||
| GMV | −0.323 | −2.043 | 0.051 | |||
| ALFF | 0.050 | 0.292 | 0.773 | |||
| DC | −0.237 | −1.349 | 0.188 | |||
| Registered R square: 0.169; ANOVA: 0.013 | ||||||
| HAMD | Constant | 0.220 | 1.104 | 0.199 | 0.844 | |
| Age | 0.083 | 0.517 | 0.610 | |||
| Gender | 2.963 | 1.251 | 0.322 | 2.368 | 0.025 * | |
| Edu. level | −0.084 | −0.589 | 0.561 | |||
| GMV | −3.426 | 0.735 | −0.633 | −4.662 | <0.001 *** | |
| ALFF | −0.086 | −0.615 | 0.544 | |||
| DC | −0.114 | −0.775 | 0.445 | |||
| Registered R square: 0.448; ANOVA: <0.001 | ||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, H.; Jia, X.; Li, Y.; Jia, X.; Yang, Q. Aberrant Amplitude of Low-Frequency Fluctuation and Degree Centrality within the Default Mode Network in Patients with Vascular Mild Cognitive Impairment. Brain Sci. 2021, 11, 1534. https://doi.org/10.3390/brainsci11111534
Li H, Jia X, Li Y, Jia X, Yang Q. Aberrant Amplitude of Low-Frequency Fluctuation and Degree Centrality within the Default Mode Network in Patients with Vascular Mild Cognitive Impairment. Brain Sciences. 2021; 11(11):1534. https://doi.org/10.3390/brainsci11111534
Chicago/Turabian StyleLi, Haoyuan, Xiuqin Jia, Yingying Li, Xuejia Jia, and Qi Yang. 2021. "Aberrant Amplitude of Low-Frequency Fluctuation and Degree Centrality within the Default Mode Network in Patients with Vascular Mild Cognitive Impairment" Brain Sciences 11, no. 11: 1534. https://doi.org/10.3390/brainsci11111534





